Pulmonary Veno-Occlusive Disease: A Comprehensive Review of Diagnostic Challenges, Therapeutic Limitations, and Evolving Management
Abstract
1. Introduction
2. Epidemiology and Clinical Importance
3. Pathophysiology
4. Etiology
4.1. Genetic Predisposition
4.2. Toxic and Environmental Exposures
4.3. Infectious Agents
4.4. Systemic and Autoimmune Diseases
5. Clinical Presentation
6. Diagnostic Approach
6.1. Echocardiography
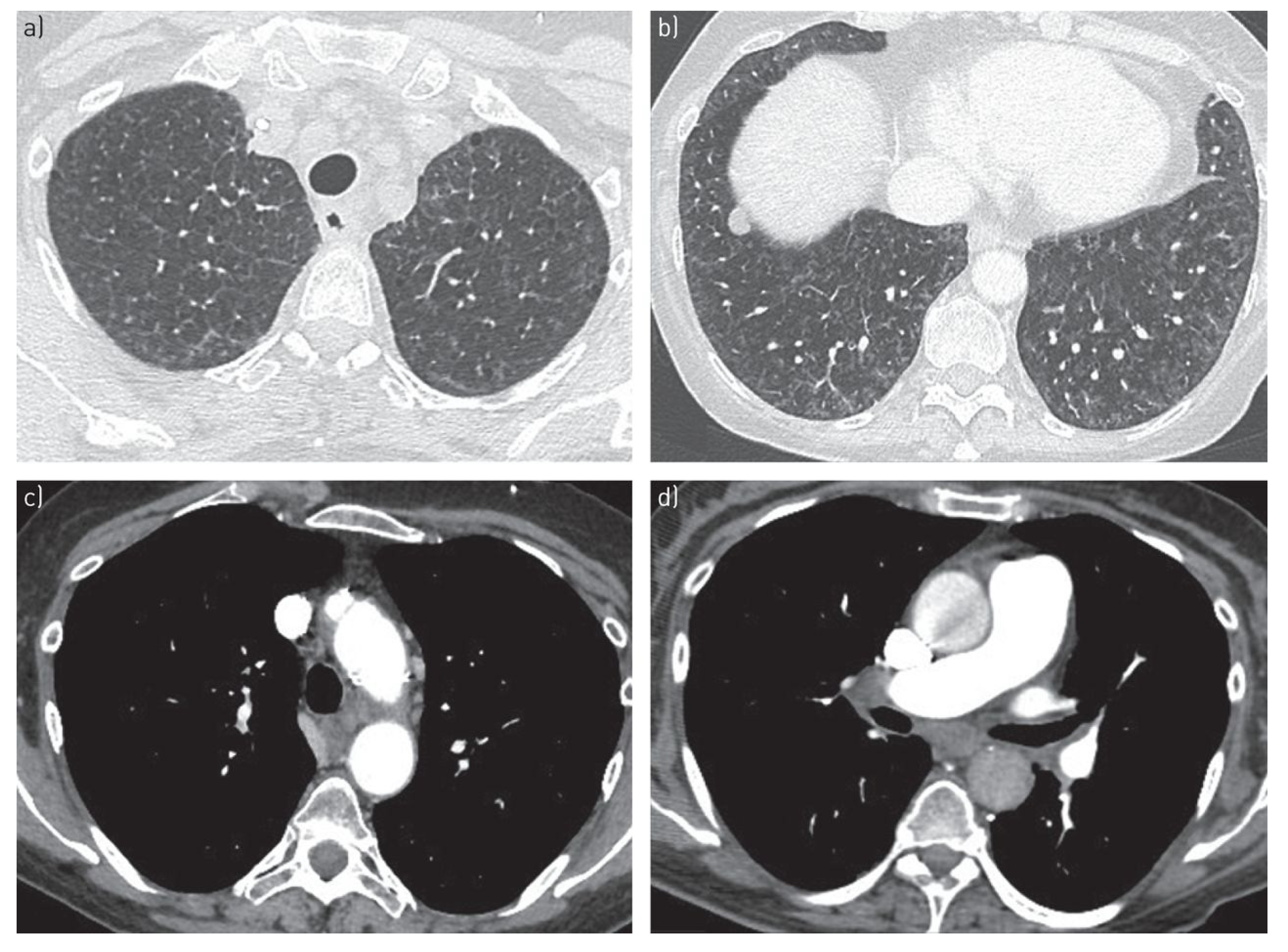
6.2. High-Resolution Computed Tomography (HRCT)
6.3. Ventilation/Perfusion (V/Q) Scan
6.4. Pulmonary Function Testing
6.5. Hemodynamic Profile
6.6. Genetic Testing
6.7. Role of Bronchoscopy and BAL
6.8. Surgical Lung Biopsy
7. Differential Diagnosis
7.1. Idiopathic Pulmonary Arterial Hypertension (IPAH)
7.2. Pulmonary Capillary Hemangiomatosis (PCH)
7.3. Interstitial Lung Disease (ILD)
7.4. Chronic Thromboembolic Pulmonary Hypertension (CTEPH)
8. Management Strategies
8.1. Supportive Measures
8.2. PAH-Targeted Therapy-Use with Extreme Caution
8.3. Lung Transplantation
8.4. Investigational and Emerging Therapies
9. Prognosis
10. Future Directions
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Brown, C.H.; Harrison, C.V. Pulmonary veno-occlusive disease. Lancet 1966, 2, 61–65. [Google Scholar] [CrossRef]
- Heath, D.; Segel, N.; Bishop, J. Pulmonary veno-occlusive disease. Circulation 1966, 34, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Boucly, A.; Girerd, B.; Bourlier, D.; Nemlaghi, S.; Caliez, J.; Savale, L.; Jaïs, X.; Dorfmüller, P.; Simonneau, G.; Sitbon, O.; et al. La maladie veino-occlusive pulmonaire [Pulmonary veno-occlusive disease]. Rev. Mal. Respir. 2018, 35, 160–170. [Google Scholar] [CrossRef]
- Wagenvoort, C.A.; Wagenvoort, N. Primary pulmonary hypertension: A pathologic study of the lung vessels in 156 clinically diagnosed cases. Circulation 1970, 42, 1163–1184. [Google Scholar] [CrossRef]
- Montani, D.; Girerd, B.; Jaïs, X.; Levy, M.; Amar, D.; Savale, L.; Dorfmüller, P.; Seferian, A.; Lau, E.M.; Eyries, M.; et al. Clinical phenotypes and outcomes of heritable and sporadic pulmonary veno-occlusive disease: A population-based study. Lancet Respir. Med. 2017, 5, 125–134. [Google Scholar] [CrossRef]
- Mandel, J.; Mark, E.J.; Hales, C.A. Pulmonary veno-occlusive disease. Am. J. Respir. Crit. Care Med. 2000, 162, 1964–1973. [Google Scholar] [CrossRef] [PubMed]
- Lechartier, B.; Boucly, A.; Solinas, S.; Gopalan, D.; Dorfmüller, P.; Radonic, T.; Sitbon, O.; Montani, D. Pulmonary veno-occlusive disease: Illustrative cases and literature review. Eur. Respir. Rev. 2024, 33, 230156. [Google Scholar] [CrossRef] [PubMed]
- Montani, D.; Price, L.C.; Dorfmuller, P.; Achouh, L.; Jaïs, X.; Yaïci, A.; Sitbon, O.; Musset, D.; Simonneau, G.; Humbert, M. Pulmonary veno-occlusive disease. Eur. Respir. J. 2009, 33, 189–200. [Google Scholar] [CrossRef]
- Wagenvoort, C.A.; Wagenvoort, N.; Takahashi, T. Pulmonary veno-occlusive disease: Involvement of pulmonary arteries and review of the literature. Hum. Pathol. 1985, 16, 1033–1041. [Google Scholar] [CrossRef]
- Eyries, M.; Montani, D.; Girerd, B.; Perret, C.; Leroy, A.; Lonjou, C.; Chelghoum, N.; Coulet, F.; Bonnet, D.; Dorfmüller, P.; et al. EIF2AK4 mutations cause pulmonary veno-occlusive disease, a recessive form of pulmonary hypertension. Nat. Genet. 2014, 46, 65–69. [Google Scholar] [CrossRef]
- Manaud, G.; Nossent, E.J.; Lambert, M.; Ghigna, M.R.; Boët, A.; Vinhas, M.C.; Ranchoux, B.; Dumas, S.J.; Courboulin, A.; Girerd, B.; et al. Comparison of Human and Experimental Pulmonary Veno-Occlusive Disease. Am. J. Respir. Cell Mol. Biol. 2020, 63, 118–131. [Google Scholar] [CrossRef]
- Nossent, E.J.; Antigny, F.; Montani, D.; Bogaard, H.J.; Ghigna, M.R.; Lambert, M.; de Montpréville, V.T.; Girerd, B.; Jaïs, X.; Savale, L.; et al. Pulmonary vascular remodeling patterns and expression of general control nonderepressible 2 (GCN2) in pulmonary veno-occlusive disease. J. Heart Lung Transpl. 2018, 37, 647–655. [Google Scholar] [CrossRef]
- Runo, J.R.; Vnencak-Jones, C.L.; Prince, M.; Loyd, J.E.; Wheeler, L.; Robbins, I.M.; Lane, K.B.; Newman, J.H.; Johnson, J.; Nichols, W.C.; et al. Pulmonary veno-occlusive disease caused by an inherited mutation in bone morphogenetic protein receptor II. Am. J. Respir. Crit. Care Med. 2003, 167, 889–894. [Google Scholar] [CrossRef]
- Takemori, W.; Yamamura, K.; Tomita, Y.; Egami, N.; Eguchi, K.; Nagata, H.; Shirouzu, H.; Ishikawa, Y.; Nakajima, D.; Yoshizawa, A.; et al. Pediatric pulmonary veno-occlusive disease associated with a novel BMPR2 variant. Pediatr. Pulmonol. 2022, 57, 1366–1369. [Google Scholar] [CrossRef]
- Girerd, B.; Montani, D.; Jaïs, X.; Eyries, M.; Yaici, A.; Sztrymf, B.; Savale, L.; Parent, F.; Coulet, F.; Godinas, L.; et al. Genetic counselling in a national referral centre for pulmonary hypertension. Eur. Respir. J. 2016, 47, 541–552. [Google Scholar] [CrossRef]
- van den Heuvel, L.M.; Jansen, S.M.A.; Alsters, S.I.M.; Post, M.C.; van der Smagt, J.J.; Handoko-De Man, F.S.; van Tintelen, J.P.; Gille, H.; Christiaans, I.; Vonk Noordegraaf, A.; et al. Genetic Evaluation in a Cohort of 126 Dutch Pulmonary Arterial Hypertension Patients. Genes 2020, 11, 1191. [Google Scholar] [CrossRef]
- Eyries, M.; Montani, D.; Nadaud, S.; Girerd, B.; Levy, M.; Bourdin, A.; Trésorier, R.; Chaouat, A.; Cottin, V.; Sanfiorenzo, C.; et al. Widening the landscape of heritable pulmonary hypertension mutations in paediatric and adult cases. Eur. Respir. J. 2019, 53, 1801371. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Gonzalez, I.; Tenorio, J.; Palomino-Doza, J.; Martinez Meñaca, A.; Morales Ruiz, R.; Lago-Docampo, M.; Valverde Gomez, M.; Gomez Roman, J.; Enguita Valls, A.B.; Perez-Olivares, C.; et al. Clinical heterogeneity of Pulmonary Arterial Hypertension associated with variants in TBX4. PLoS ONE 2020, 15, e0232216. [Google Scholar] [CrossRef] [PubMed]
- Montani, D.; Lau, E.M.; Descatha, A.; Jaïs, X.; Savale, L.; Andujar, P.; Bensefa-Colas, L.; Girerd, B.; Zendah, I.; Le Pavec, J.; et al. Occupational exposure to organic solvents: A risk factor for pulmonary veno-occlusive disease. Eur. Respir. J. 2015, 46, 1721–1731. [Google Scholar] [CrossRef] [PubMed]
- Perros, F.; Günther, S.; Ranchoux, B.; Godinas, L.; Antigny, F.; Chaumais, M.C.; Dorfmüller, P.; Hautefort, A.; Raymond, N.; Savale, L.; et al. Mitomycin-Induced Pulmonary Veno-Occlusive Disease: Evidence from Human Disease and Animal Models. Circulation 2015, 132, 834–847. [Google Scholar] [CrossRef]
- Ranchoux, B.; Günther, S.; Quarck, R.; Chaumais, M.C.; Dorfmüller, P.; Antigny, F.; Dumas, S.J.; Raymond, N.; Lau, E.; Savale, L.; et al. Chemotherapy-induced pulmonary hypertension: Role of alkylating agents. Am. J. Pathol. 2015, 185, 356–371. [Google Scholar] [CrossRef]
- Escamilla, R.; Hermant, C.; Berjaud, J.; Mazerolles, C.; Daussy, X. Pulmonary veno-occlusive disease in a HIV-infected intravenous drug abuser. Eur. Respir. J. 1995, 8, 1982–1984. [Google Scholar] [CrossRef] [PubMed]
- Mesa, R.A.; Edell, E.S.; Dunn, W.F.; Edwards, W.D. Human immunodeficiency virus infection and pulmonary hypertension: Two new cases and a review of 86 reported cases. Mayo Clin. Proc. 1998, 73, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Launay, D.; Sobanski, V.; Hachulla, E.; Humbert, M. Pulmonary hypertension in systemic sclerosis: Different phenotypes. Eur. Respir. Rev. 2017, 26, 170056. [Google Scholar] [CrossRef]
- George, M.P.; Novelli, E.M.; Shigemura, N.; Simon, M.A.; Feingold, B.; Krishnamurti, L.; Morrell, M.R.; Gries, C.G.; Haider, S.; Johnson, B.A.; et al. First successful lung transplantation for sickle cell disease with severe pulmonary arterial hypertension and pulmonary veno-occlusive disease. Pulm. Circ. 2013, 3, 952–958. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.M.; Dawson, A.; Jenkins, G.H.; Nicholson, A.G.; Hansell, D.M.; Harrison, N.K. Sarcoidosis-related pulmonary veno-occlusive disease presenting with recurrent haemoptysis. Eur. Respir. J. 2009, 34, 517–520. [Google Scholar] [CrossRef]
- Swift, G.L.; Gibbs, A.; Campbell, I.A.; Wagenvoort, C.A.; Tuthill, D. Pulmonary veno-occlusive disease and Hodgkin’s lymphoma. Eur. Respir. J. 1993, 6, 596–598. [Google Scholar] [CrossRef]
- Sahay, S. Evaluation and classification of pulmonary arterial hypertension. J. Thorac. Dis. 2019, 11 (Suppl. S14), S1789–S1799. [Google Scholar] [CrossRef]
- Resten, A.; Maitre, S.; Humbert, M.; Rabiller, A.; Sitbon, O.; Capron, F.; Simonneau, G.; Musset, D. Pulmonary hypertension: CT of the chest in pulmonary venoocclusive disease. Am. J. Roentgenol. 2004, 183, 65–70. [Google Scholar] [CrossRef]
- Montani, D.; Achouh, L.; Dorfmüller, P.; Le Pavec, J.; Sztrymf, B.; Tchérakian, C.; Rabiller, A.; Haque, R.; Sitbon, O.; Jaïs, X.; et al. Pulmonary veno-occlusive disease: Clinical, functional, radiologic, and hemodynamic characteristics and outcome of 24 cases confirmed by histology. Medicine 2008, 87, 220–233. [Google Scholar] [CrossRef]
- Montani, D.; Lau, E.M.; Dorfmüller, P.; Girerd, B.; Jaïs, X.; Savale, L.; Perros, F.; Nossent, E.; Garcia, G.; Parent, F.; et al. Pulmonary veno-occlusive disease. Eur. Respir. J. 2016, 47, 1518–1534. [Google Scholar] [CrossRef]
- Seferian, A.; Helal, B.; Jaïs, X.; Girerd, B.; Price, L.C.; Günther, S.; Savale, L.; Dorfmüller, P.; Parent, F.; Sitbon, O.; et al. Ventilation/perfusion lung scan in pulmonary veno-occlusive disease. Eur. Respir. J. 2012, 40, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Ruopp, N.F.; Cockrill, B.A. Diagnosis and Treatment of Pulmonary Arterial Hypertension: A Review. Jama 2022, 327, 1379–1391. [Google Scholar] [CrossRef] [PubMed]
- Montani, D.; Kemp, K.; Dorfmuller, P.; Sitbon, O.; Simonneau, G.; Humbert, M. Idiopathic pulmonary arterial hypertension and pulmonary veno-occlusive disease: Similarities and differences. Semin. Respir. Crit. Care Med. 2009, 30, 411–420. [Google Scholar] [CrossRef]
- Godinas, L.; Amar, D.; Montani, D.; Lau, E.M.; Jaïs, X.; Savale, L.; Jevnikar, M.; Sitbon, O.; Simonneau, G.; Humbert, M.; et al. Lung capillary blood volume and membrane diffusion in precapillary pulmonary hypertension. J. Heart Lung Transpl. 2016, 35, 647–656. [Google Scholar] [CrossRef]
- Lewis, R.A.; Thompson, A.A.R.; Billings, C.G.; Charalampopoulos, A.; Elliot, C.A.; Hamilton, N.; Hill, C.; Hurdman, J.; Rajaram, S.; Sabroe, I.; et al. Mild parenchymal lung disease and/or low diffusion capacity impacts survival and treatment response in patients diagnosed with idiopathic pulmonary arterial hypertension. Eur. Respir. J. 2020, 55, 2000041. [Google Scholar] [CrossRef]
- Humbert, M.; Kovacs, G.; Hoeper, M.M.; Badagliacca, R.; Berger, R.M.F.; Brida, M.; Carlsen, J.; Coats, A.J.S.; Escribano-Subias, P.; Ferrari, P.; et al. ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur. Heart J. 2022, 43, 3618–3731. [Google Scholar] [CrossRef]
- Hadinnapola, C.; Bleda, M.; Haimel, M.; Screaton, N.; Swift, A.; Dorfmüller, P.; Preston, S.D.; Southwood, M.; Hernandez-Sanchez, J.; Martin, J.; et al. Phenotypic Characterization of EIF2AK4 Mutation Carriers in a Large Cohort of Patients Diagnosed Clinically with Pulmonary Arterial Hypertension. Circulation 2017, 136, 2022–2033. [Google Scholar] [CrossRef]
- Liang, L.; Ma, G.; Chen, K.; Liu, Y.; Wu, X.; Ying, K.; Zhang, R. EIF2AK4 mutation in pulmonary veno-occlusive disease: A case report and review of the literature. Medicine 2016, 95, e5030. [Google Scholar] [CrossRef]
- Montani, D.; O’Callaghan, D.S.; Savale, L.; Jaïs, X.; Yaïci, A.; Maitre, S.; Dorfmuller, P.; Sitbon, O.; Simonneau, G.; Humbert, M. Pulmonary veno-occlusive disease: Recent progress and current challenges. Respir. Med. 2010, 104 (Suppl. S1), S23–S32. [Google Scholar] [CrossRef] [PubMed]
- Ali, N.; Loughborough, W.W.; Rodrigues, J.C.L.; Suntharalingam, J.; Hudson, B.; Hall, T.; Augustine, D.; Mackenzie, R.; Robinson, G. Computed tomographic and clinical features of pulmonary veno-occlusive disease: Raising the radiologist’s awareness. Clin. Radiol. 2019, 74, 655–662. [Google Scholar] [CrossRef]
- Minomo, S.; Kitaichi, M.; Arai, T.; Matsui, H.; Akira, M.; Inoue, Y. Pulmonary Veno-occlusive Disease: A Surgical Lung Biopsy-proven and Autopsied Case Radiologically Mimicking Hypersensitivity Pneumonitis at the Time of a Transbronchial Lung Biopsy. Intern. Med. 2019, 58, 955–964. [Google Scholar] [CrossRef]
- Weatherald, J.; Dorfmüller, P.; Perros, F.; Ghigna, M.R.; Girerd, B.; Humbert, M.; Montani, D. Pulmonary capillary haemangiomatosis: A distinct entity? Eur. Respir. Rev. 2020, 29, 190168. [Google Scholar] [CrossRef]
- Chaisson, N.F.; Dodson, M.W.; Elliott, C.G. Pulmonary Capillary Hemangiomatosis and Pulmonary Veno-occlusive Disease. Clin. Chest Med. 2016, 37, 523–534. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.H.; Delcroix, M.; Jais, X.; Madani, M.M.; Matsubara, H.; Mayer, E.; Ogo, T.; Tapson, V.F.; Ghofrani, H.A.; Jenkins, D.P. Chronic thromboembolic pulmonary hypertension. Eur. Respir. J. 2019, 53, 1801915. [Google Scholar] [CrossRef] [PubMed]
- Aguado, B.; Grynblat, J.; Budhram, B.; Ghigna, M.R.; Boucly, A.; Antigny, F.; Jaïs, X.; Sitbon, O.; Savale, L.; Humbert, M.; et al. Pulmonary veno-occlusive disease: A paradigm of diagnosis and therapeutic challenges in pulmonary hypertension. Curr. Opin. Pulm. Med. 2025, 31, 443–455. [Google Scholar] [CrossRef]
- Doyle, J.; Johnson, R.F. Dyspnea to Diagnosis: A Pulmonary Veno-Occlusive Disease Case. Chest 2024, 166 (Suppl. S4), A6071–A6072. [Google Scholar] [CrossRef]
- Yuan, X.; Hou, X.; Guo, W.; Jiang, H.; Zheng, J.; Paudyal, S.; Lyu, Y. A case report of PVOD patient combined with pulmonary embolism: Anticoagulation or not? Medicine 2017, 96, e6507. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Bian, T.; Yu, Z.; Wei, Y.; Xu, J.; Zhu, J.; Zhang, W. Bilateral Lung Transplantation Provides Better Long-term Survival and Pulmonary Function Than Single Lung Transplantation: A Systematic Review and Meta-analysis. Transplantation 2019, 103, 2634–2644. [Google Scholar] [CrossRef]
- Subramanian, M.; Meyers, B.F. Lung Transplant Procedure of Choice: Bilateral Transplantation Versus Single Transplantation Complications, Quality of Life, and Survival. Clin. Chest Med. 2023, 44, 47–57. [Google Scholar] [CrossRef]
- Quezada-Loaiza, C.A.; de Pablo Gafas, A.; Pérez, V.; Alonso, R.; Juarros, L.; Real, M.I.; López, E.; Cortes, M.; Meneses, J.C.; González, I.H.; et al. Lung Transplantation in Pulmonary Hypertension: A Multidisciplinary Unit’s Management Experience. Transpl. Proc. 2018, 50, 1496–1503. [Google Scholar] [CrossRef]
- Wille, K.M.; Sharma, N.S.; Kulkarni, T.; Lammi, M.R.; Barney, J.B.; Bellot, S.C.; Cantor, R.S.; Naftel, D.C.; Diaz-Guzman, E.; McGiffin, D.C. Characteristics of patients with pulmonary venoocclusive disease awaiting transplantation. Ann. Am. Thorac. Soc. 2014, 11, 1411–1418. [Google Scholar] [CrossRef]
- Charoenpong, P. Lung Transplant Outcomes in Patients with Pulmonary Veno-Occlusive Disease. J. Heart Lung Transpl. 2022, 41 (Suppl. S4), S535–S536. [Google Scholar] [CrossRef]
- Le Pavec, J.; Hascoët, S.; Fadel, E. Heart-lung transplantation: Current indications, prognosis and specific considerations. J. Thorac. Dis. 2018, 10, 5946–5952. [Google Scholar] [CrossRef] [PubMed]
- Mendeloff, E.N.; Meyers, B.F.; Sundt, T.M.; Guthrie, T.J.; Sweet, S.C.; de la Morena, M.; Shapiro, S.; Balzer, D.T.; Trulock, E.P.; Lynch, J.P.; et al. Lung transplantation for pulmonary vascular disease. Ann. Thorac. Surg. 2002, 73, 209–217. [Google Scholar] [CrossRef]
- Yan, H.J.; Zheng, X.Y.; Huang, H.; Xu, L.; Tang, H.T.; Wang, J.J.; Li, C.H.; Zhang, S.X.; Fu, S.Y.; Wen, H.Y.; et al. Double-lung versus heart-lung transplantation for end-stage cardiopulmonary disease: A systematic review and meta-analysis. Surg. Today 2023, 53, 1001–1012. [Google Scholar] [CrossRef] [PubMed]
- Kitai, T.; Okamoto, T.; Miyakoshi, C.; Niikawa, H.; Alvarez, P.A.; Krittanawong, C.; Xanthopoulos, A.; McCurry, K.R. Impact of combined heart and lung transplantation on bronchiolitis obliterans syndrome, cardiac allograft vasculopathy, and long-term survival. J. Heart Lung Transpl. 2019, 38, 1170–1177. [Google Scholar] [CrossRef]
- Sato, H.; Sugimura, K.; Miura, M.; Konno, R.; Kozu, K.; Yaoita, N.; Shimizu, T.; Yamamoto, S.; Aoki, T.; Tatebe, S.; et al. Beneficial Effects of Imatinib in a Patient with Suspected Pulmonary Veno-Occlusive Disease. Tohoku J. Exp. Med. 2019, 247, 69–73. [Google Scholar] [CrossRef]
- Ogawa, A.; Miyaji, K.; Matsubara, H. Efficacy and safety of long-term imatinib therapy for patients with pulmonary veno-occlusive disease and pulmonary capillary hemangiomatosis. Respir. Med. 2017, 131, 215–219. [Google Scholar] [CrossRef]
- Overbeek, M.J.; van Nieuw Amerongen, G.P.; Boonstra, A.; Smit, E.F.; Vonk-Noordegraaf, A. Possible role of imatinib in clinical pulmonary veno-occlusive disease. Eur. Respir. J. 2008, 32, 232–235. [Google Scholar] [CrossRef] [PubMed]
- Gomez Rojas, O.R.; Mitchell, S.T.; Kalra, S.S.; Bryant, A.J.; Ataya, A. Sotatercept Improves Hypoxia and Clinical Status in Patients with Pulmonary Venous Occlusive Disease. Am. J. Respir. Cirt. Care Med. 2025, 211, A7095. [Google Scholar] [CrossRef]
- Humbert, M.; Sitbon, O.; Chaouat, A.; Bertocchi, M.; Habib, G.; Gressin, V.; Yaici, A.; Weitzenblum, E.; Cordier, J.F.; Chabot, F.; et al. Pulmonary arterial hypertension in France: Results from a national registry. Am. J. Respir. Crit. Care Med. 2006, 173, 1023–1030. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.Y.; Duval, S.; Badesch, D.B.; Bull, T.M.; Chakinala, M.M.; De Marco, T.; Frantz, R.P.; Hemnes, A.; Mathai, S.C.; Rosenzweig, E.B.; et al. Mortality in Pulmonary Arterial Hypertension in the Modern Era: Early Insights from the Pulmonary Hypertension Association Registry. J. Am. Heart Assoc. 2022, 11, e024969. [Google Scholar] [CrossRef] [PubMed]
- Hoeper, M.M.; Pausch, C.; Grünig, E.; Staehler, G.; Huscher, D.; Pittrow, D.; Olsson, K.M.; Vizza, C.D.; Gall, H.; Distler, O.; et al. Temporal trends in pulmonary arterial hypertension: Results from the COMPERA registry. Eur. Respir. J. 2022, 59, 2102024. [Google Scholar] [CrossRef]
- Farber, H.W.; Miller, D.P.; Poms, A.D.; Badesch, D.B.; Frost, A.E.; Muros-Le Rouzic, E.; Romero, A.J.; Benton, W.W.; Elliott, C.G.; McGoon, M.D.; et al. Five-Year outcomes of patients enrolled in the REVEAL Registry. Chest 2015, 148, 1043–1054. [Google Scholar] [CrossRef]
- Gall, H.; Felix, J.F.; Schneck, F.K.; Milger, K.; Sommer, N.; Voswinckel, R.; Franco, O.H.; Hofman, A.; Schermuly, R.T.; Weissmann, N.; et al. The Giessen Pulmonary Hypertension Registry: Survival in pulmonary hypertension subgroups. J. Heart Lung Transpl. 2017, 36, 957–967. [Google Scholar] [CrossRef]
- Escribano-Subias, P.; Blanco, I.; López-Meseguer, M.; Lopez-Guarch, C.J.; Roman, A.; Morales, P.; Castillo-Palma, M.J.; Segovia, J.; Gómez-Sanchez, M.A.; Barberà, J.A. Survival in pulmonary hypertension in Spain: Insights from the Spanish registry. Eur. Respir. J. 2012, 40, 596–603. [Google Scholar] [CrossRef]
- Balko, R.; Edriss, H.; Nugent, K.; Test, V. Pulmonary veno-occlusive disease: An important consideration in patients with pulmonary hypertension. Respir. Med. 2017, 132, 203–209. [Google Scholar] [CrossRef]

| Feature | Pulmonary Veno-Occlusive Disease (PVOD) | Idiopathic Pulmonary Arterial Hypertension (IPAH) |
|---|---|---|
| Primary site of pathology | Post-capillary venules and small pulmonary veins; intimal fibrosis and occlusion | Pre-capillary arterioles; medial hypertrophy and plexiform lesions |
| Genetic associations | Biallelic EIF2AK4 mutations; rare overlap with BMPR2, TBX4, SMAD9 variants | BMPR2, TBX4, ACVRL1, SMAD9, CAV1, ENG, others |
| Histopathology | Venular fibrosis and occlusion; capillary congestion; absence of plexiform lesions | Arteriolar remodeling with plexiform lesions and medial hypertrophy |
| Hemodynamics (RHC) | Pre-capillary PH profile (mPAP ≥ 20 mmHg, PCWP ≤ 15 mmHg, PVR > 2 WU), indistinguishable from IPAH | Pre-capillary PH profile (mPAP ≥ 20 mmHg, PCWP ≤ 15 mmHg, PVR > 2 WU) |
| HRCT findings | Classic triad: centrilobular ground-glass opacities, smooth interlobular septal thickening, mediastinal lymphadenopathy | Enlarged pulmonary arteries; otherwise often normal parenchyma |
| Response to PAH therapies | Poor; vasodilators may precipitate pulmonary edema | Often improves with PAH-targeted therapy; vasoreactivity testing may identify responders |
| Bronchoalveolar lavage | Hemosiderin-laden macrophages (evidence of occult hemorrhage reflecting capillary leak and venous involvement) | Typically absent |
| V/Q scan | Usually normal | Usually normal |
| Prognosis | Rapidly progressive; median survival < 2 years without transplant | Variable; median survival 5–7 years with therapy |
| Definitive therapy | Lung transplantation (curative) | Mainstay is medical therapy; transplant reserved for refractory cases |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Published by MDPI on behalf of the Polish Respiratory Society. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Foster, B.; Khan, S.; Suarez Gonzalez, A.; Gillenwater, S. Pulmonary Veno-Occlusive Disease: A Comprehensive Review of Diagnostic Challenges, Therapeutic Limitations, and Evolving Management. Adv. Respir. Med. 2025, 93, 48. https://doi.org/10.3390/arm93060048
Foster B, Khan S, Suarez Gonzalez A, Gillenwater S. Pulmonary Veno-Occlusive Disease: A Comprehensive Review of Diagnostic Challenges, Therapeutic Limitations, and Evolving Management. Advances in Respiratory Medicine. 2025; 93(6):48. https://doi.org/10.3390/arm93060048
Chicago/Turabian StyleFoster, Brian, Sikandar Khan, Ana Suarez Gonzalez, and Samantha Gillenwater. 2025. "Pulmonary Veno-Occlusive Disease: A Comprehensive Review of Diagnostic Challenges, Therapeutic Limitations, and Evolving Management" Advances in Respiratory Medicine 93, no. 6: 48. https://doi.org/10.3390/arm93060048
APA StyleFoster, B., Khan, S., Suarez Gonzalez, A., & Gillenwater, S. (2025). Pulmonary Veno-Occlusive Disease: A Comprehensive Review of Diagnostic Challenges, Therapeutic Limitations, and Evolving Management. Advances in Respiratory Medicine, 93(6), 48. https://doi.org/10.3390/arm93060048






