Wetting Properties of Polyetheretherketone Plasma Activated and Biocoated Surfaces
Abstract
1. Introduction
2. Materials and Methods
2.1. Surface Roughness Characterization
2.2. Surface Free Energy (SFE) Determination
3. Results and Discussion
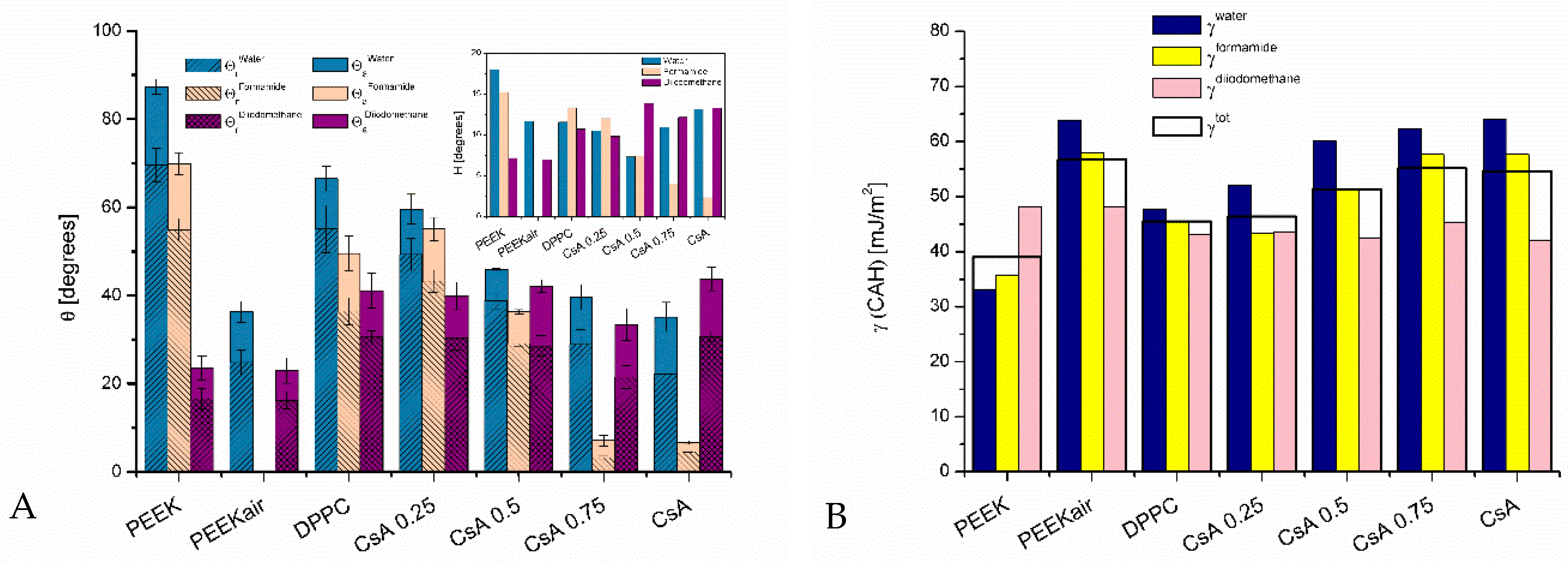
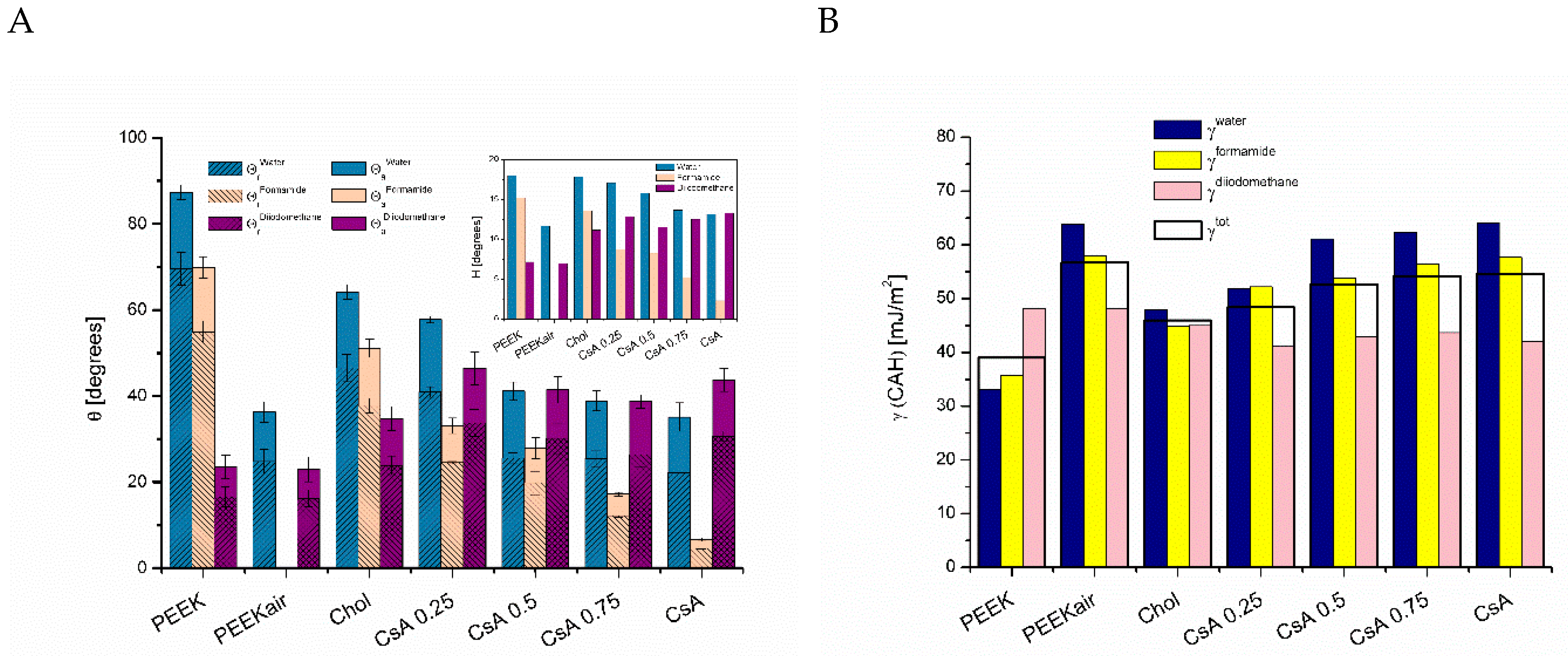
3.1. Contact Angles
3.1.1. Plasma-Activated and Untreated PEEK Surfaces
3.1.2. One-Component Monolayers
3.1.3. Binary Monolayers
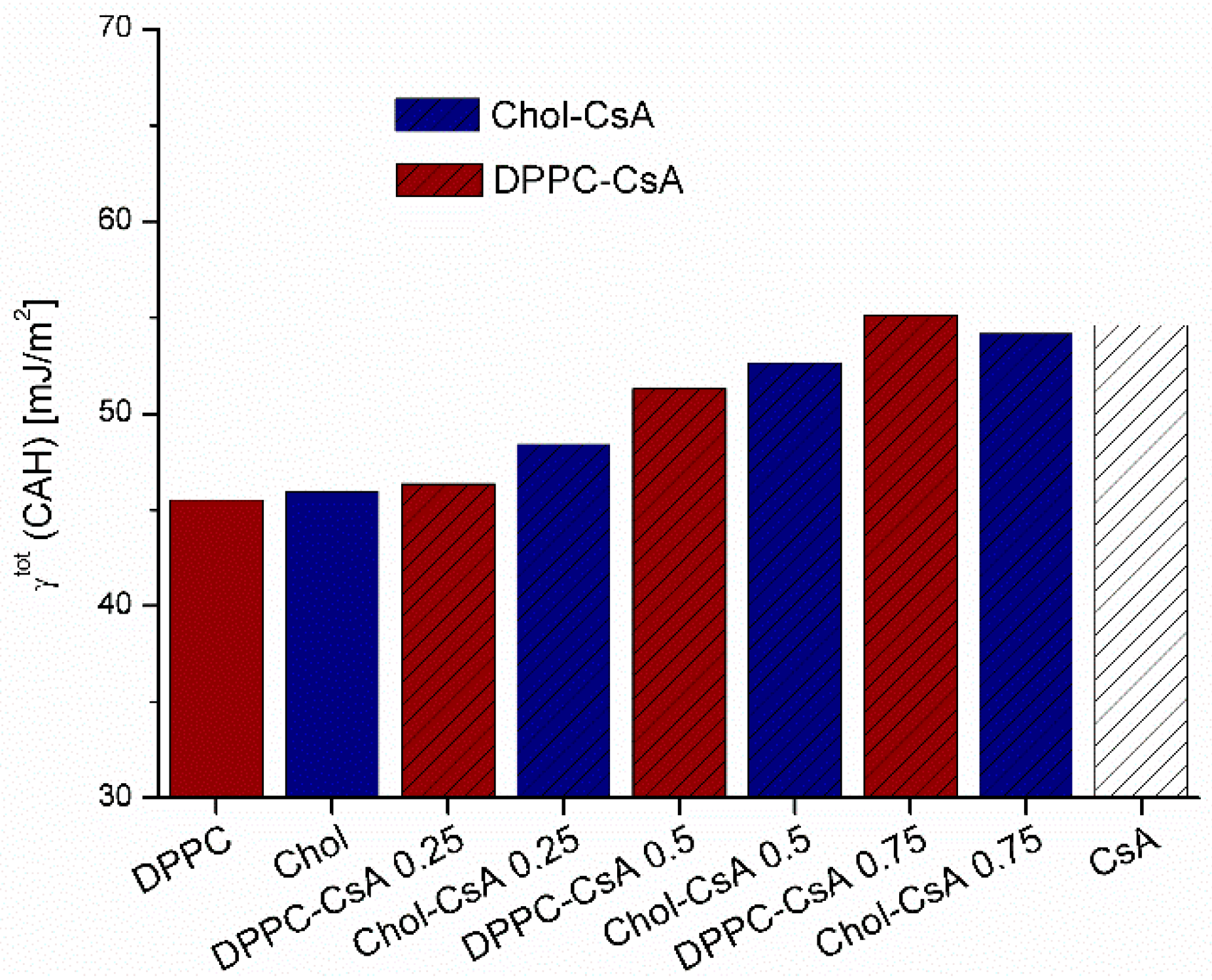
3.2. Contact Angle Hysteresis
3.3. Surface Free Energy
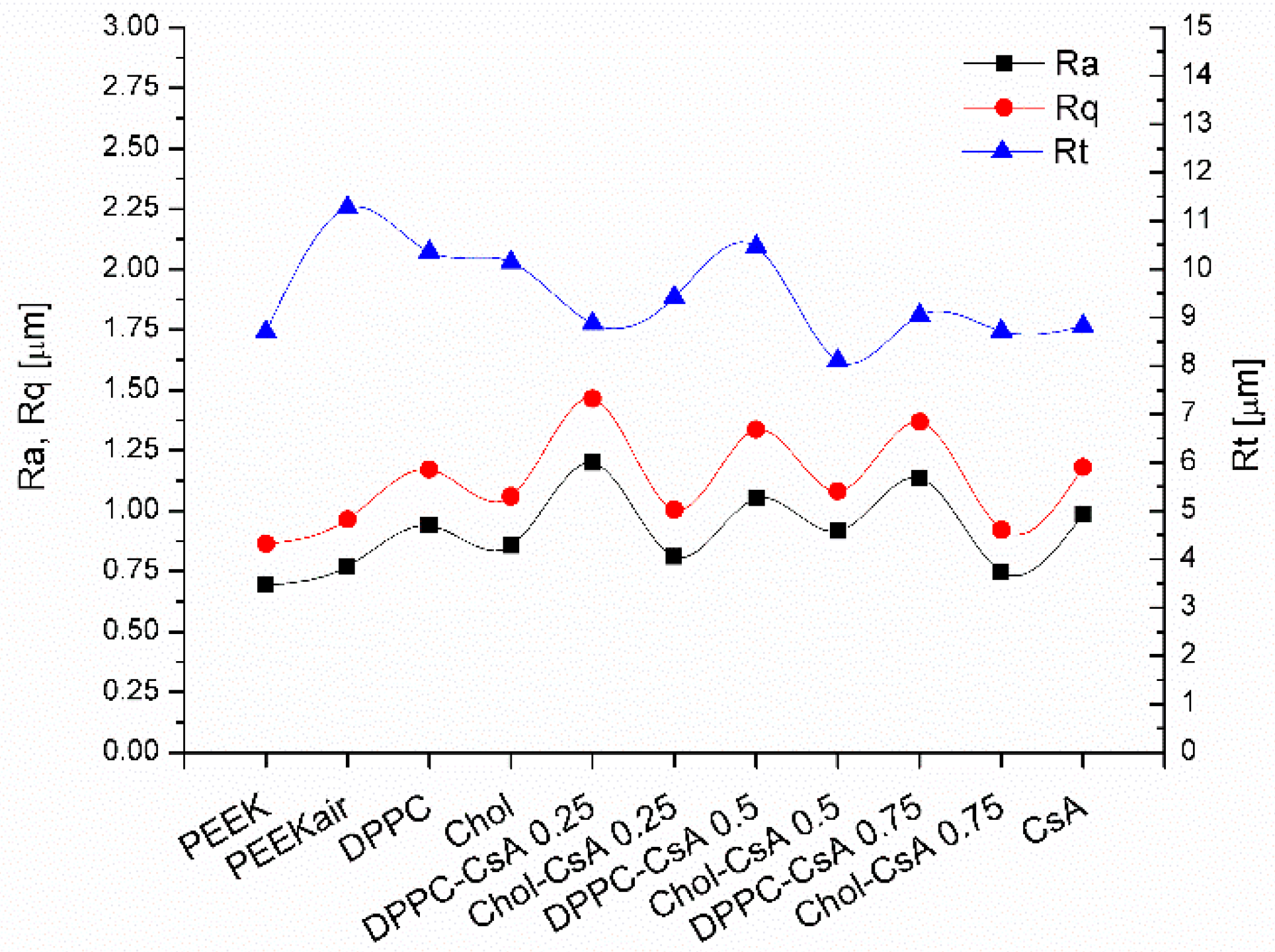
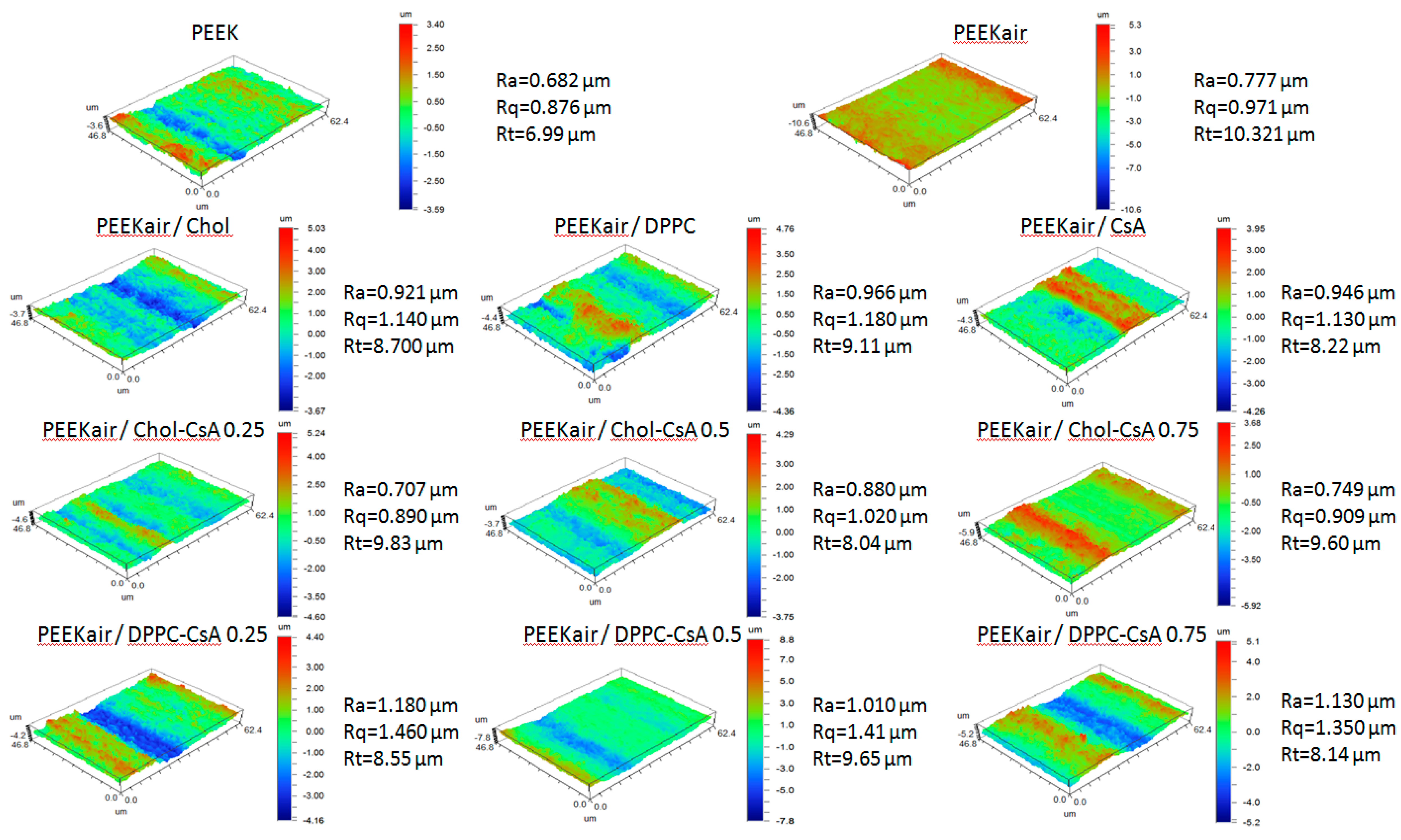
3.4. Surface Topography
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kurtz, S.M. An overview of PEEK biomaterials. In PEEK Biomaterials Handbook, 1st ed.; William Andrew: Kindlington, UK, 2011; pp. 1–8. [Google Scholar]
- Ma, R.; Tang, T. Current strategies to improve the bioactivity of PEEK. Int. J. Mol. Sci. 2014, 15, 5426–5445. [Google Scholar] [CrossRef] [PubMed]
- Wiącek, A.E.; Terpiłowski, K.; Jurak, M.; Worzakowska, M. Low-temperature air plasma modification of chitosan-coated PEEK. Polym. Test. 2016, 50, 325–334. [Google Scholar] [CrossRef]
- Wiącek, A.E.; Terpiłowski, K.; Jurak, M.; Worzakowska, M. Effect of low-temperature plasma on chitosan-coated PEEK polymer characteristics. Eur. Polym. J. 2016, 78, 1–13. [Google Scholar] [CrossRef]
- Evans, N.T.; Torstrick, F.B.; Lee, C.S.D.; Dupont, K.M.; Safranski, D.L.; Chang, W.A.; Macedo, A.E.; Lin, A.; Boothby, J.M.; Whittingslow, D.C.; et al. High strenght, surface porous poly-ether-ether-ketone orthopaedic implants. Acta Biomater. 2015, 13, 159–167. [Google Scholar] [CrossRef]
- Walsh, W.R.; Bertollo, N.; Christou, C.; Schaffiner, D.; Mobbs, R.J. Plasma-sprayed titanium coating to polyetheretherketone improves the bone-implant interface. Spine J. 2015, 15, 1041–1049. [Google Scholar] [CrossRef] [PubMed]
- Lvhua, L.; Yanyan, Z.; Xiong, Z. Bioactive polyetheretherketone implant composites for hard tissue. Prog. Chem. 2017, 29, 450–458. [Google Scholar] [CrossRef]
- Gan, K.; Liu, H.; Jiang, L.; Liu, X.; Song, X.; Niu, D.; Chen, T.; Liu, C. Bioactivity and antibacterial effect of nitrogen plasma immersion ion implantation on polyetheretherketone. Dent. Mater. 2016, 32, 10263–10274. [Google Scholar] [CrossRef]
- Civantos, A.; Martinez-Campos, E.; Ramos, V.; Carlos, E.; Gallardo, A.; Abarrategi, A. Titanium coatings and surface modifications: Toward clinically useful bioactive implants. ASC Biomater. Sci. Eng. 2017, 3, 1245–1261. [Google Scholar] [CrossRef]
- Willumeit, R.; Feyersbend, F. Phospholipids as implant coatings. J. Mater. Sci. Mater. Med. 2007, 18, 367–380. [Google Scholar] [CrossRef]
- Tateishi, T.; Kyomoto, M.; Kakinoki, S.; Yamaoka, T.; Ishihara, K. Reduced platelets and bacteria adhesion on poly(ether etherketone) by photoinduced and self-initiated graft polymerization of 2-methacryloyloxyethyl phosphorylcholine. J. Biomed. Mater. Res. A 2014, 102, 1342–1349. [Google Scholar] [CrossRef]
- Iwasaki, Y.; Ishihara, K. Cell membrane-inspired phospholipid polymers for developing medical devices with excellent biointerfaces. Sci. Technol. Adv. Mater. 2012, 13, 064101. [Google Scholar] [CrossRef] [PubMed]
- Jurak, M.; Wiącek, A.E.; Terpiłowski, K. Properties of PEEK-supported films of biological substances prepared by the Langmuir-Blodgett technique. Colloids Surf. A 2016, 510, 263–274. [Google Scholar] [CrossRef]
- Jurak, M.; Wiącek, A.E.; Mroczka, R.; Łopucki, R. Chitosan/phospholipid coated polyethyleneterephthalate (PET) polimer surfaces activated by air plasma. Colloids Surf. A 2017, 532, 155–164. [Google Scholar] [CrossRef]
- Liefeith, K.; Frant, M. Archaeal tetraether lipid coatings—A strategy for the development of membrane analog spacer systems for the site-specific functionalization of medical surfaces. Biointerphases 2018, 13, 011004. [Google Scholar] [CrossRef]
- De Leo, V.; Mattioli-Belmonte, M.; Cimmarusti, M.T.; Panniello, A.; Dicarlo, M.; Milano, F.; Agostiano, A.; De Giglio, E.; Catucci, L. Liposome—Modified titanium surface: A strategy to locally deliver bioactive molecules. Colloids Surf. B 2017, 158, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Willumeit, R.; Feyerabend, F.; Kamusewitz, H.; Schossig, M. Biological multi-layer systems as implant Surface modification. Materialwissenschaft und Werkstofftechnik 2003, 34, 1084–1093. [Google Scholar] [CrossRef]
- Rochford, E.T.; Poulsson, A.H.; Salavarrieta, V.J.; Lezuo, P.; Richards, R.G.; Moriarty, T.F. Bacterial adhesion to orthopaedic implant materials and a novel oxygen plasma modified PEEK surface. Colloids Surf. B 2014, 113, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Gil’man, A.B. Low-temperature plasma treatment as an effective method for surface modification of polymeric materials. High Energy Chem. 2003, 37, 17–23. [Google Scholar] [CrossRef]
- Chibowski, E. Surface free energy of a solid from contact angle hysteresis. Adv. Colloid Interface Sci. 2003, 103, 149–172. [Google Scholar] [CrossRef]
- Chibowski, E. Some problems of characterization of solid surface via the surface free energy changes. Adsorpt. Sci. Technol. 2017, 35, 647–659. [Google Scholar] [CrossRef]
- Jurak, M. Changes in stability of the DPPC monolayer during its contact with the liquid phase. Chem. Phys. Lipids 2012, 165, 302–310. [Google Scholar] [CrossRef] [PubMed]
- Belman, N.; Jn, K.; Golan, Y.; Israelachvili, J.N.; Pesika, N.S. Origin of the contact angle hysteresis of water on chemisorbed and physisorbed self-assembled monolayers. Langmuir 2012, 28, 14609–14617. [Google Scholar] [CrossRef]
- Jurak, M. Contact angle hysteresis and phase separation in dry phospholipid films with cholesterol deposited on mica surface. Appl. Surf. Sci. 2015, 328, 596–605. [Google Scholar] [CrossRef]
- Wnętrzak, A.; Makyła-Juzak, K.; Chachaj-Brekiesz, A.; Lipiec, E.; Romeu, N.V.; Dynarowicz-Łątka, P. Cyclosporin A distribution in cholesterol-sphingomyelin artificial membranes modeled as Langmuir monolayers. Colloids Surf. B Biointerfaces 2018, 166, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Ha, S.W.; Hauert, R.; Ernst, K.H.; Winter, E.; Wintermantel, E. Surface analysis of chemically-etched and plasma-treated polyetheretherketone (PEEK) for biomedical applications. Surf. Coat. Technol. 1997, 96, 293–299. [Google Scholar] [CrossRef]
- De Bartolo, L.; Gugluizza, A.; Morelli, S.; Cirillo, B.; Gordano, A.; Drioli, E. Novel PEEK-WC membranes with lows plasma protein affinity related to surface free energy parameters. J. Mater. Sci. Mater. Med. 2004, 15, 877–883. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Awaja, F.; James, N.; McKenzie, D.R.; Ruys, A.J. A comparison of the strength of autohesion of plasma treated amorphous and semi-crystalline PEEK films. Polym. Adv. Technol. 2011, 22, 2496–2502. [Google Scholar] [CrossRef]
- Terpiłowski, K.; Jurak, M.; Wiącek, A.E. Influence of nitrogen plasma treatment on the wettability of polyetheretherketone and deposited chitosan layers. Adv. Polym. Technol. 2018, 37, 1557–1569. [Google Scholar] [CrossRef]
- El Taya, N.; Mark, A.E.; Vallat, P.; Brunne, R.M.; Testa, B.; van Gunsteren, W.F. Solvent-dependent conformation and hydrogen-bonding capacity of cyclosporin A: Evidence from partition coefficients and molecular dynamics simulations. J. Med. Chem. 1993, 36, 3757–3764. [Google Scholar] [CrossRef]
- Przykaza, K.; Woźniak, K.; Jurak, M.; Wiącek, A.E. Properties of Langmuir and Langmuir-Blodgett monolayers of cholesterol-cyclosporine A on water and polymer support. Adsorption 2019. submitted. [Google Scholar]
- Przykaza, K.; Woźniak, K.; Jurak, M.; Wiącek, A.E. Characteristics of polypeptide/phospholipid monolayers on water and plasma activated PEEK support. J. Surfactants Deterg. 2019. submitted. [Google Scholar]
- Dynarowicz, P.; Wnętrzak, P.; Makyła-Juzak, K. Cyclosporin A in membrane lipid environment: Implications for antimalarian activity of the drug—The Langmuir monolayer studies. J. Membr. Biol. 2015, 248, 1021–1032. [Google Scholar] [CrossRef] [PubMed]
- Soderlund, T.; Lehtonen, J.Y.A.; Kinnunen, P.K.J. Interactions of cyclosporin A with phospholipid membranes: Effect of cholesterol. Mol. Pharmacol. 1999, 55, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Gaines, G.L. Insoluble Monolayers at Liquid-Gas Interface; Wiley—Interscience: New York, NY, USA, 1966. [Google Scholar]
- Rymuszka, D.; Terpiłowski, K.; Borowski, P.; Hołysz, L. Time-dependent changes of surface properties of polyether ether ketone caused by air plasma treatment. Polym. Int. 2016, 65, 827–834. [Google Scholar] [CrossRef]
- O’Learyl, T.; Ross, P.; Lieber, M.; Levin, I. Effects of cyclosporine A on biomembranes: Vibrational spectroscopic, calorimetric and hemolysis studies. Biophys. J. 1986, 49, 795–801. [Google Scholar] [CrossRef]
- Sedev, R.V.; Petrov, J.G.; Neumann, J.G. Effect of swelling of a polymer surface on advancing and receding contact angles. J. Colloid Interfacial Sci. 1996, 180, 36–42. [Google Scholar] [CrossRef]
- Scomparin, C.; Lecuyer, S.; Ferreira, M.; Charitat, T.; Tinland, B. Diffusion in supported lipid bilayers: Influence of substrate and preparation technique on the internal dynamics. Eur. Phys. J. E 2009, 28, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Richter, R.P.; Maury, N.; Brisson, A.R. On the effect of the solid support on the interleafed distribution of lipids in supported lipid bilayers. Langmuir 2005, 21, 299–304. [Google Scholar] [CrossRef] [PubMed]
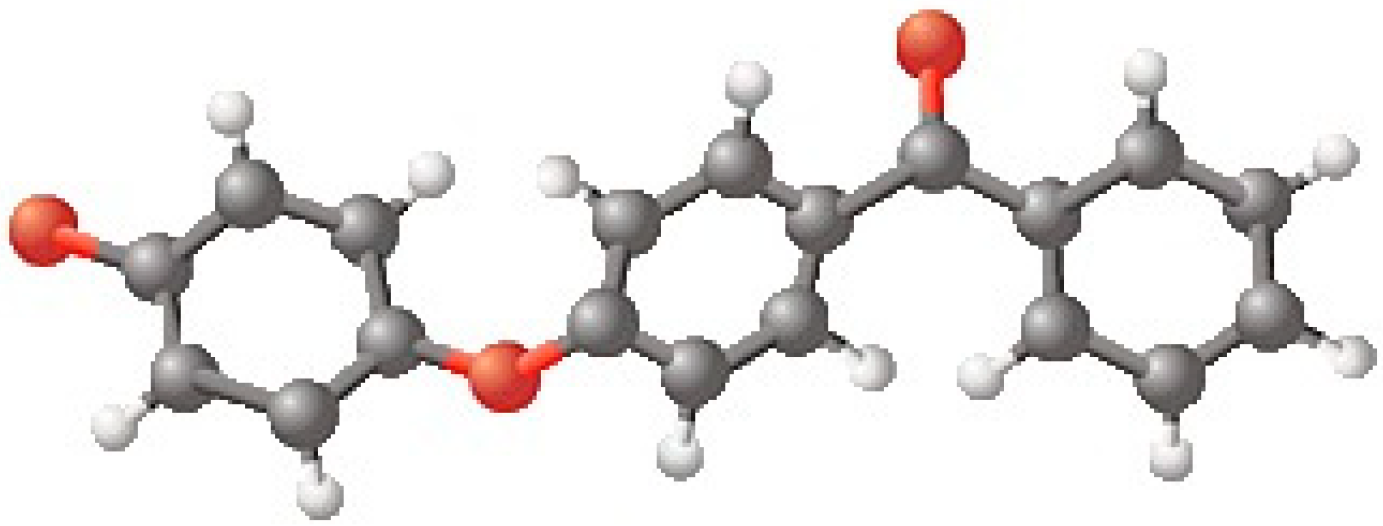
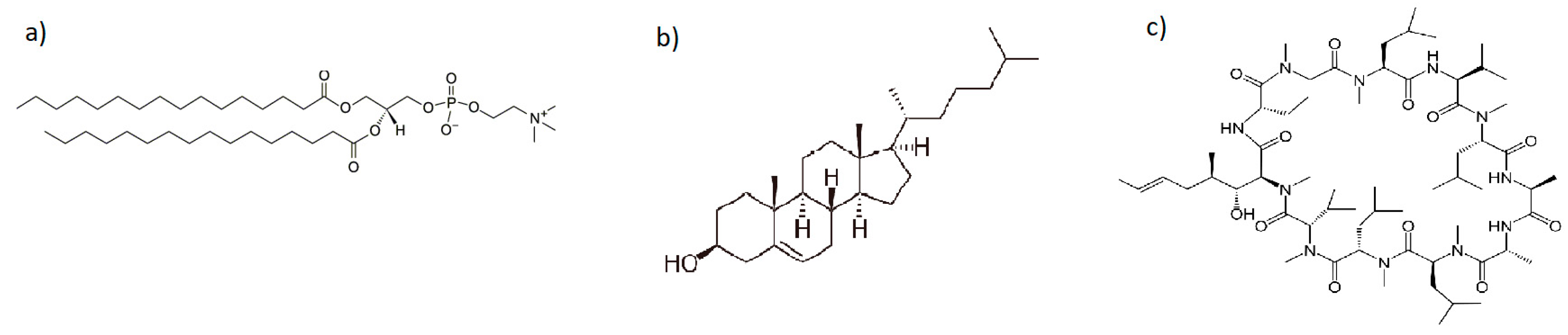
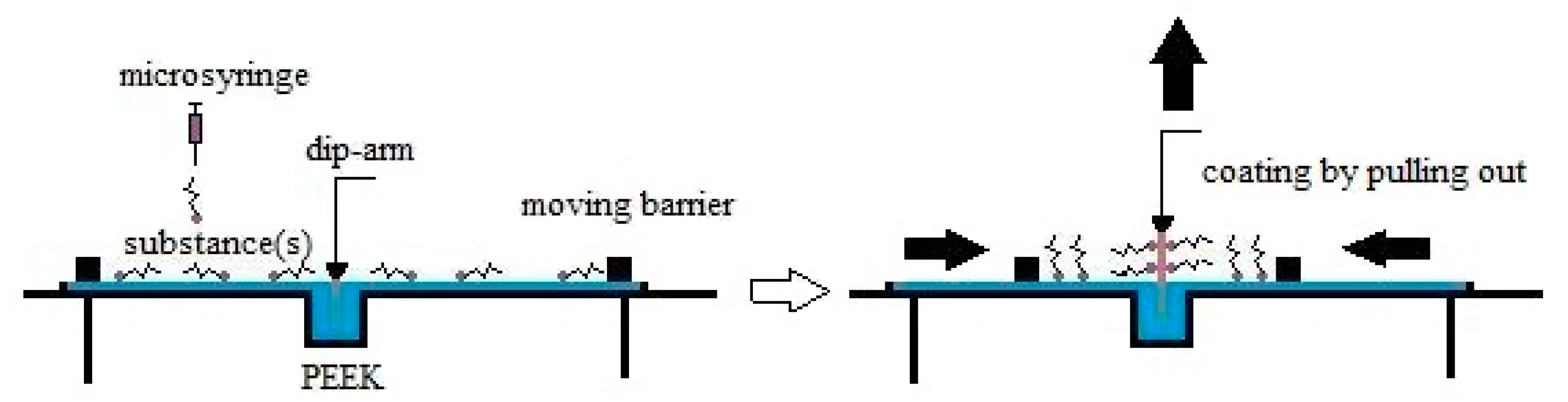
| Substance(s) | Compression Speed [mm/min] | Surface Pressure during Transfer [mN/m] | Transfer Speed [mm/min] |
|---|---|---|---|
| DPPC | 20 | 30 | 5 |
| Cholesterol | 20 | 30 | 5 |
| CsA | 20 | 15 | 5 |
| DPPC-CsA | 20 | 15 | 5 |
| Cholesterol-CsA | 20 | 15 | 5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Przykaza, K.; Woźniak, K.; Jurak, M.; Wiącek, A.E. Wetting Properties of Polyetheretherketone Plasma Activated and Biocoated Surfaces. Colloids Interfaces 2019, 3, 40. https://doi.org/10.3390/colloids3010040
Przykaza K, Woźniak K, Jurak M, Wiącek AE. Wetting Properties of Polyetheretherketone Plasma Activated and Biocoated Surfaces. Colloids and Interfaces. 2019; 3(1):40. https://doi.org/10.3390/colloids3010040
Chicago/Turabian StylePrzykaza, Kacper, Klaudia Woźniak, Małgorzata Jurak, and Agnieszka Ewa Wiącek. 2019. "Wetting Properties of Polyetheretherketone Plasma Activated and Biocoated Surfaces" Colloids and Interfaces 3, no. 1: 40. https://doi.org/10.3390/colloids3010040
APA StylePrzykaza, K., Woźniak, K., Jurak, M., & Wiącek, A. E. (2019). Wetting Properties of Polyetheretherketone Plasma Activated and Biocoated Surfaces. Colloids and Interfaces, 3(1), 40. https://doi.org/10.3390/colloids3010040






