Sorption Membranes and Filter for Water Purification and Disinfection in Outdoor Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Adsorption Experiments
2.3. Analytic Methods
2.4. Preparation of Sorption Membranes
3. Results and Discussion
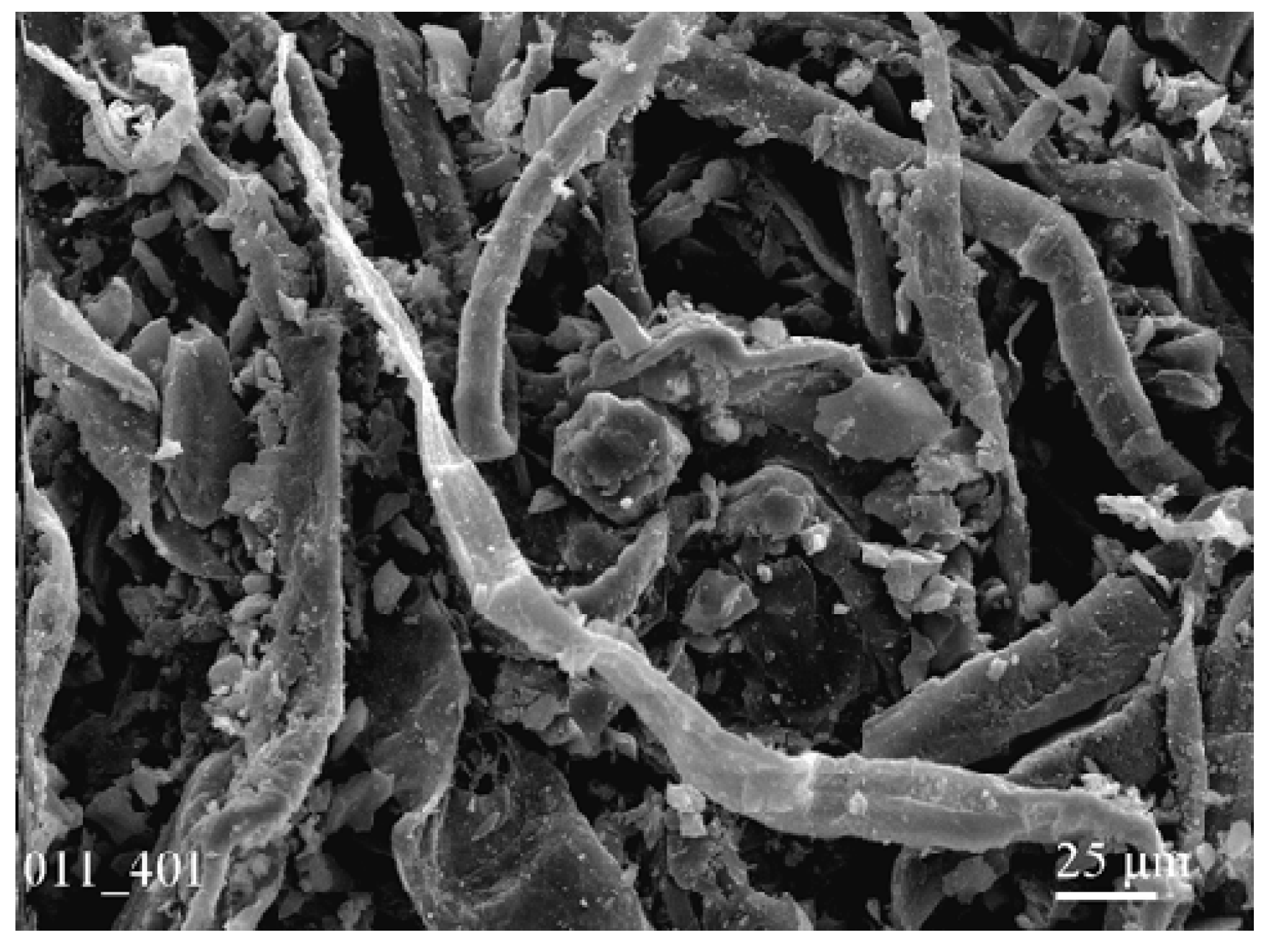
3.1. Rough Mechanical Filtration Membrane
3.2. Bactericidal Membranes
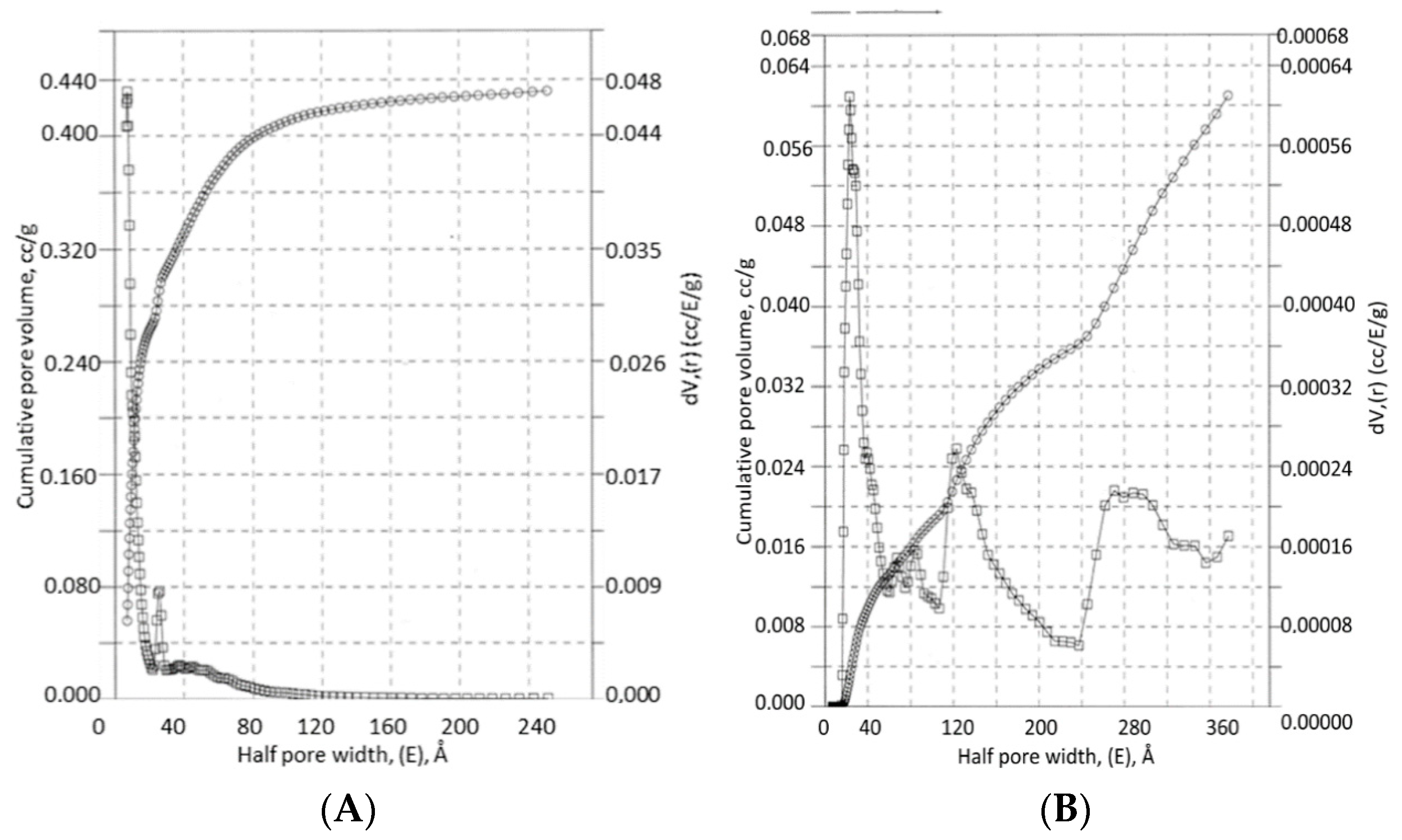
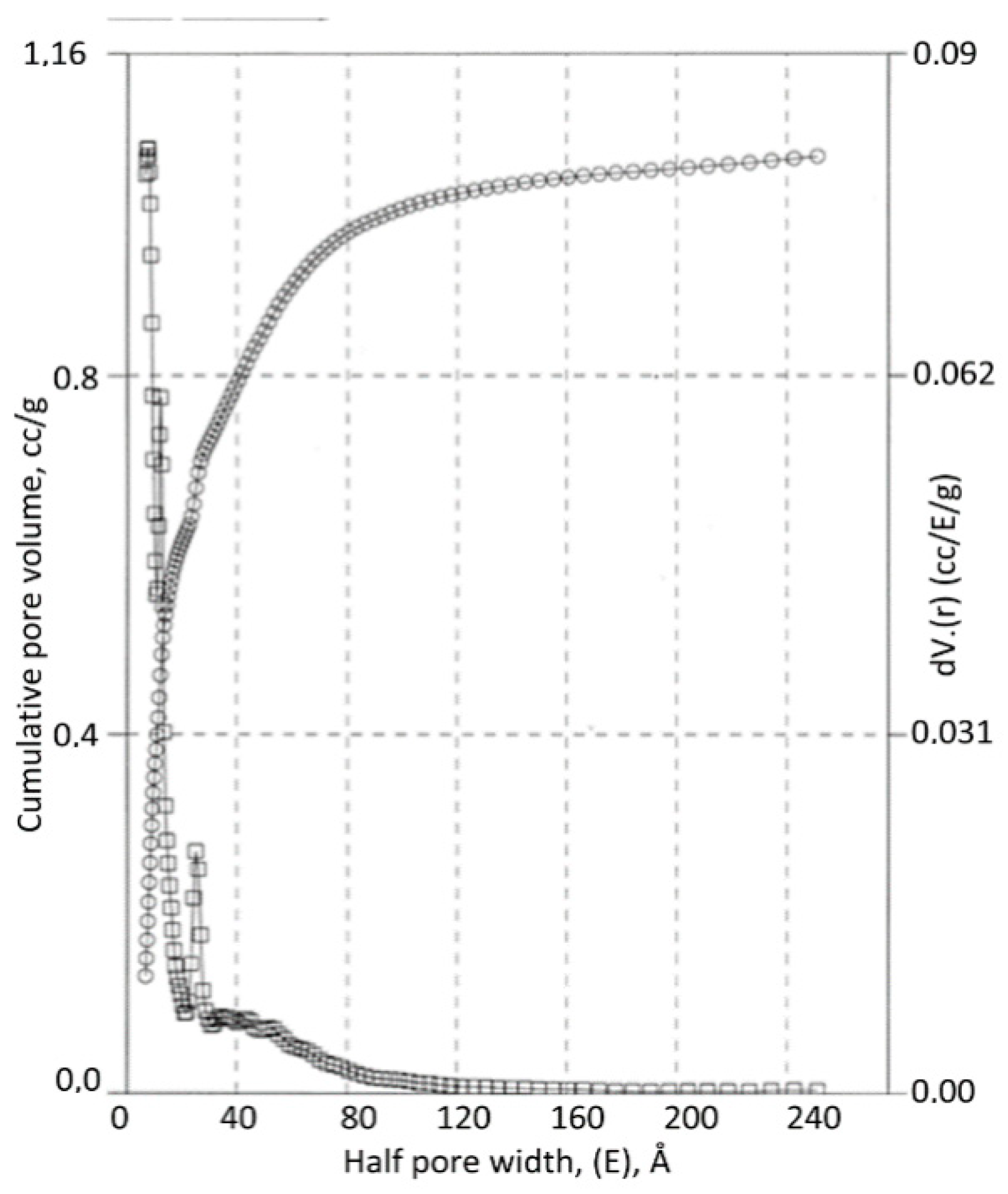
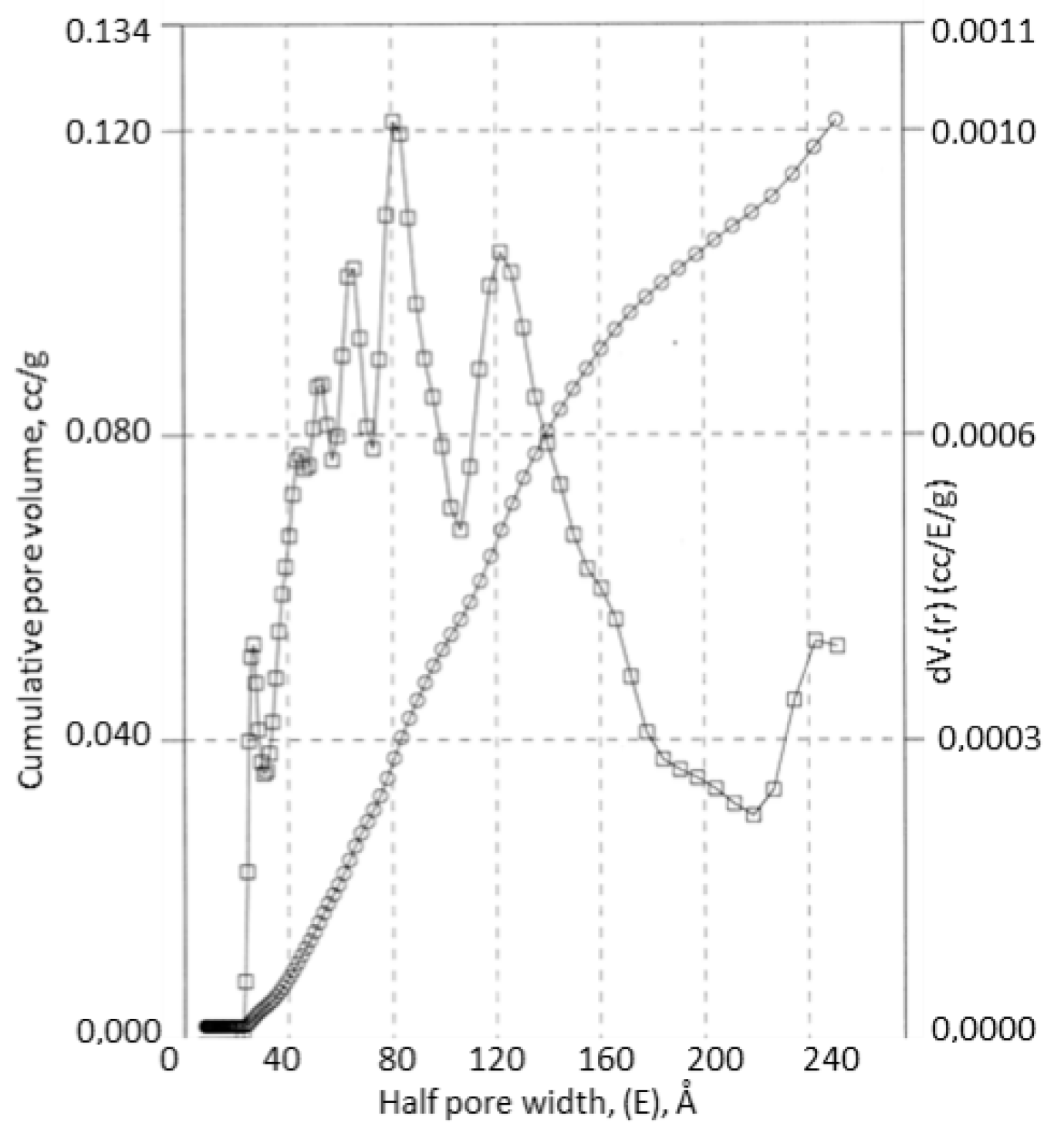
3.3. Carbon-Cellulose Membranes
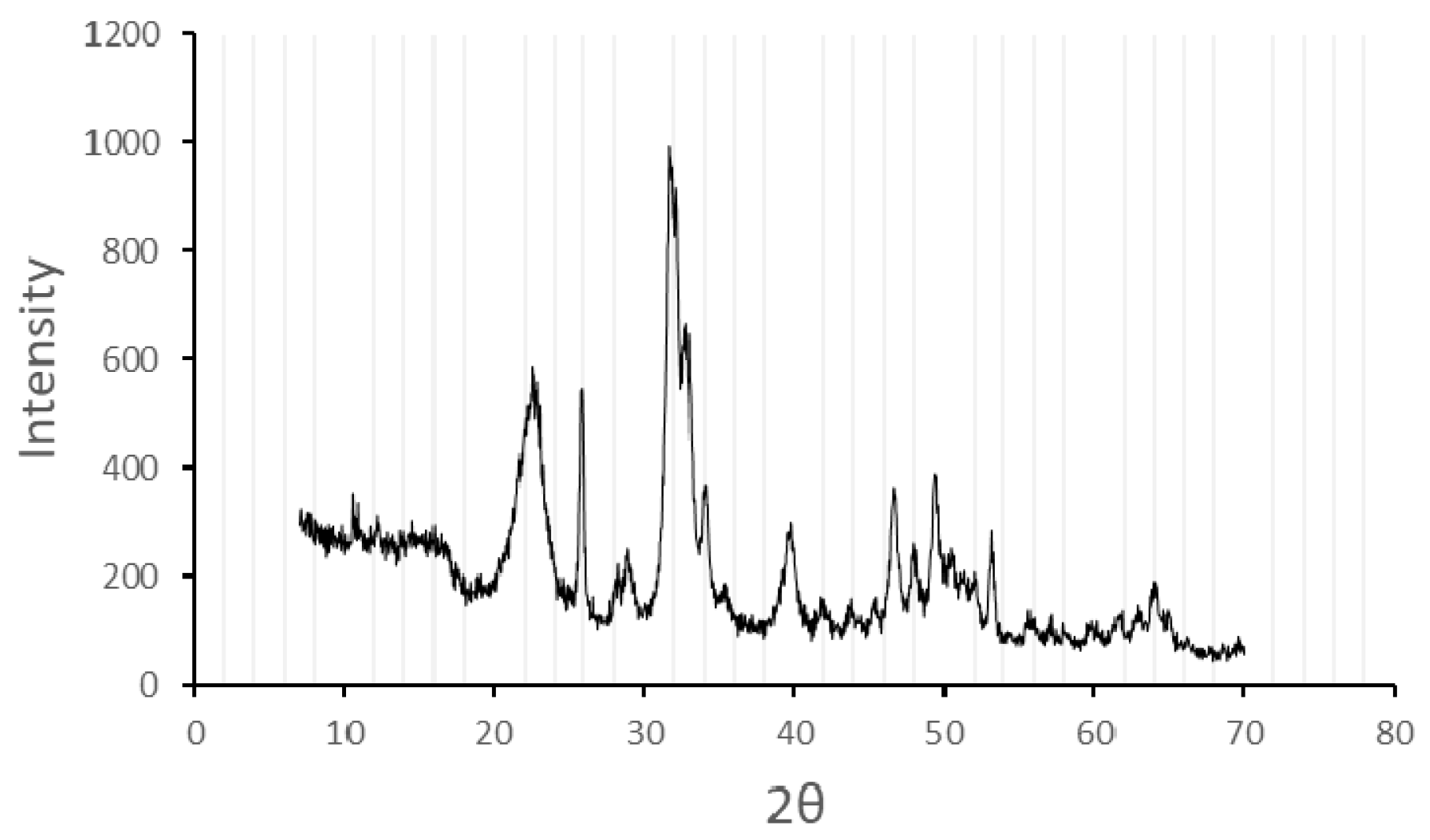
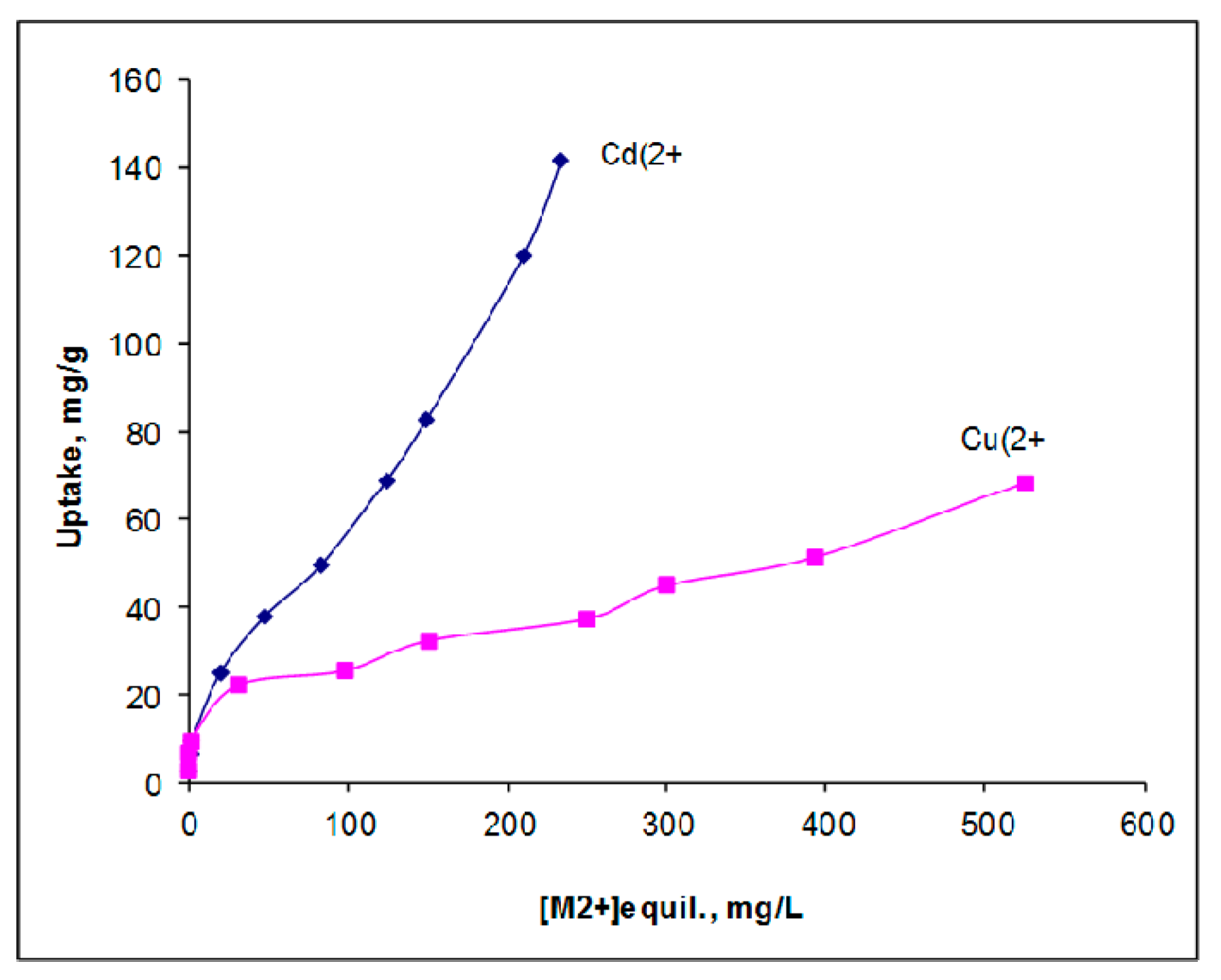
3.4. Apatite-Cellulose Membranes
3.5. Membrane Filter and Contaminated Water Purification
4. Conclusions
Funding
Conflicts of Interest
References
- Figoli, A.; Criscuoli, A. (Eds.) Sustainable Membrane Technology for Water and Wastewater Treatment; Springer Nature, Singapore Pte Ltd.: Singapore, 2017; p. 387. ISBN 978-981-10-5623-9. (eBook). [Google Scholar]
- Basile, A.; Cassano, A.; Rastogi, N. (Eds.) Advances in Membrane Technologies for Water Treatment; Elsevier: Amsterdam, The Netherlands, 2015; p. 667. ISBN 978-1-78242-126-9. (online). [Google Scholar]
- Tansel, B. New Technologies for Water and Wastewater Treatment: A Survey of Recent Patents. Recent Pat. Chem. Eng. 2008, 1, 17–26. [Google Scholar] [CrossRef]
- How to Choose a Water Filter or Purifier. Available online: https://www.rei.com/learn/expert-advice/water-treatment-backcountry.html (accessed on 5 March 2019).
- Monaghan, S.; Inspector (Project Manager), EPA. Water Treatment Manual: Disinfection; Environmental Protection Agency an Ghníomhaireacht um Chaomhnú Comhshaoil: Wexford, Ireland, 2011; p. 200. ISBN 978-184095-421-0. [Google Scholar]
- Rice, E.; Baird, R.; Eaton, A. (Eds.) Standard Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association: Washington, DC, USA, 2017; p. 277. ISBN 9780875532875. [Google Scholar]
- Glymph, T. Wastewater Microbiology: A Handbook for Operators; American Water Work Association: Washington, DC, USA, 2005; p. 182. ISBN 9781583213438. [Google Scholar]
- Gregg, S.J.; Sing, K.S.W. Adsorption, Surface Area and Porosity, 2nd ed.; Academic Press Inc.: San Diego, CA, USA, 1982; p. 313. ISBN 0-12-300956-1. [Google Scholar]
- Conner, W.C.; Cevallos-Candau, J.F.; Weist, E.L. Characterization of Pore Structure: Porosimetry and Sorption. Langmuir 1986, 2, 151–154. [Google Scholar] [CrossRef]
- Kellner, R.; Mermet, J.M.; Otto, M.; Widmer, H.M. (Eds.) Analytical Chemistry; Wiley-VCH: Weinheim, Germany, 1998; p. 942. ISBN 3-527-2881-3. [Google Scholar]
- Monica, E. (Ed.) Pulp and Paper Chemistry and Technology; Walter de Gruyter GmbH & Co. KG.: Berlin, Germany, 2009; Volume 1, p. 420. ISBN 978-3-11-021339-3. [Google Scholar]
- Latha, A.; Arivukarasi, M.; Keerthana, C.; Subashri, R.; Vishnu Priya, V. Paper and Pulp Industry Manufacturing and Treatment Processes—A Review. Int. J. Eng. Res. Technol. 2018, 6. [Google Scholar] [CrossRef]
- Zerin, I.; Datta, E. A Review Article on Applications of Filter Cloth. Int. J. Cloth. Sci. 2018, 5, 1–6. [Google Scholar] [CrossRef]
- Aaron, A.O.; Batchelor, N.W.; Garnier, G. Multi-Layer Filters: Adsorption and Filtration Mechanisms for Improved Separation. Front. Chem. 2018, 6, 417. [Google Scholar]
- Rohrbach, K.; Li, Y.; Zhu, H.; Liu, Z.J.D.; Andreasen, J.; Hu, L. A cellulose based hydrophilic, oleophobic hydrated filter for water/oil separation. R. Soc. Chem. Chem. Commun. 2014, 50, 13296–13299. [Google Scholar] [CrossRef] [PubMed]
- Brock, T.D. Membrane Filtration. A User’s Guide and Reference Manual; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 1983; 381p, ISBN 3-540-12128-5. [Google Scholar]
- Juneja, H.D.; Joshi, M.; Kanfade, J.P. Synthesis and Characterization of Metallic Gel Complexes Derived from Carboxymethyl Cellulose. J. Chem. 2013, 2013, 820328. [Google Scholar] [CrossRef]
- Zhong, T.; Oporto, G.S.; Jaczynsky, J.; Tesfai, A.T.; Armstrong, J. Antimicrobial properties of the hybrid copper nanoparticles-carboxymethyl cellulose. Wood Fiber Sci. 2013, 45, 215–222. [Google Scholar]
- Hou, K.; Gerba, C.P.; Goyal, S.M.; Zerda, K.S. Capture of latex beads, bacteria, endotoxin, and viruses by charge-modified filters. Appl. Environ. Microbiol. 1980, 40, 892–896. [Google Scholar] [PubMed]
- Sobsey, M.D.; Jones, B.L. Concentration of Poliovirus from Tap Water Using Positively Charged Microporous Filters. Appl. Environ. Micribiol. 1979, 588–595. [Google Scholar]
- Chrowdhury, Z.; Summers, S.R.; Westerhoff, G.; Leto, B.J.; Norwack, K.; Corwin, C.; Passantino, L. Activated Carbon. Solution for Improving Water Quality; American Water Work Association: Washington, DC, USA, 2013; p. 353. ISBN 978-1-61300-202-5. (electronic). [Google Scholar]
- Zhuravlev, I.; Zakutevsky, O.; Psareva, T.; Kanibolotsky, V.; Strelko, V.; Taffet, M.; Gallio, G. Uranium sorption on amorphous titanium and zirconium phosphates modified by Al3+ or Fe3+ ions. J. Radioanal. Nucl. Chem. 2002, 254, 85–89. [Google Scholar] [CrossRef]
- Zhuravlev, I.; Kanibolotsky, V.; Strelko, V.; Bortun, A.; Bortun, L.; Khainakov, S.; Clearfield, A. Synthesis and Characterization of the Ion Exchange Properties of Spherically Granulated Sodium Aluminophosphatesilicate. Solvent Extr. Ion Exch. 1999, 17, 635–648. [Google Scholar] [CrossRef]
- Zhuravlev, I.; Kanibolotsky, V.; Strelko, V.; Gallios, G.; Strelko, V., Jr. Novel High Porous Spherically Granulated Ferrophosphatesilicate Gels. Mater. Res. Bull. 2004, 39, 737–744. [Google Scholar] [CrossRef]
- Zhuravlev, I.; Kanibolotsky, V.; Strelko, V.; Gallios, G. Novel Spherically Granulated Inorganic Ion Exchangers Based on Aluminophosphatesilicate and Ferrophosphatesilicate Gels. Sep. Sci. Technol. 2004, 39, 287–300. [Google Scholar] [CrossRef]
- Srinivasan, M.; Ferraris, C.; White, T. Cadmium and lead ion capture with three dimensionally ordered macroporous hydroxyapatite. Environ. Sci. Technol. 2006, 40, 7054–7059. [Google Scholar] [CrossRef] [PubMed]
- Elliott, J.C. Structure and Chemistry of the Apatites and Other Calcium Orthophosphates; Elsevier: Amsterdam, The Netherlands; London, UK; New York, NY, USA; Tokyo, Japan, 1994; p. 401, e-book; ISBN1 9781483290317. Hardcover; ISBN2 9780444815828. [Google Scholar]



| Membrane Sample | Bacteria Content, CFU/mL | |||
|---|---|---|---|---|
| Pseudomonas aeruginosa | Escherichia coli | |||
| Initial | Filtrate | Initial | Filtrate | |
| Cu-CMC-1 | 2300 | 24 | 35 | 13 |
| Cu-CMC-2 | 2300 | 10 | 35 | 0 |
| Cu-CMC-3 | 2300 | 4 | 35 | 0 |
| CCHA-1 | - | - | 35 | 3 |
| CCHA-2 | - | - | 35 | 1 |
| CCHA-3 | - | - | 35 | 0 |
| № | Amount of Membrane | Chromaticity, grad | Oxidizability, mgО2/L |
|---|---|---|---|
| 1 | 1 | 7.6 | 2.7 |
| 2 | 2 | 7.3 | 2.0 |
| 3 | 3 | 6.8 | 1.1 |
| 4 | 4 | 6.4 | 0.8 |
| Water | Physicochemical Properties of Water Quality | Escherichia Coli, Group Microbial Content CFU/L | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| рН | Chromaticity, grad | Turbidity, mg/L | Total Hardness, mgeqv/L | Iron Total, mg/L | Manganese, mg/L | Nitrates, mg/L | Oxidizability, mgO2/L | Dry residue, mg/L | ||
| Requirements for drinking water | 6.5–8.5 | ≤20 | ≤0,58 | ≤7 | ≤0.2 | ≤0.05 | ≤50 | ≤5 | ≤1000 | <3 |
| Initial water | 7.9 | 22.5 | 3.43 | 6.58 | 0.24 | 0.1 | 2.1 | 4.2 | 551 | 1250 |
| 2nd filtered liter | 6.5 | 7.5 | <0.1 | 5.26 | 0.1 | 0.015 | 2.7 | 2.6 | 504 | <3 (norm) |
| 6th filtered liter | 7.68 | 6.8 | <0.1 | 6.3 | 0.01 | 0.031 | 1.9 | 1.0 | 536 | <3 |
| 10th filtered liter | 7.86 | 6.8 | <0.1 | 6.44 | <0.1 | 0.037 | 1.8 | 1.0 | 546 | <3 |
| 14th filtered liter | 7.92 | 6.8 | <0.1 | 6.46 | <0.1 | 0.024 | 2.2 | 0.9 | 547 | <3 |
| 18th filtered liter | 7.96 | 6.8 | <0.1 | 6.58 | <0.1 | 0.018 | 2.1 | 0.9 | 548 | <3 |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhuravlev, I. Sorption Membranes and Filter for Water Purification and Disinfection in Outdoor Conditions. Colloids Interfaces 2019, 3, 35. https://doi.org/10.3390/colloids3010035
Zhuravlev I. Sorption Membranes and Filter for Water Purification and Disinfection in Outdoor Conditions. Colloids and Interfaces. 2019; 3(1):35. https://doi.org/10.3390/colloids3010035
Chicago/Turabian StyleZhuravlev, Igor. 2019. "Sorption Membranes and Filter for Water Purification and Disinfection in Outdoor Conditions" Colloids and Interfaces 3, no. 1: 35. https://doi.org/10.3390/colloids3010035
APA StyleZhuravlev, I. (2019). Sorption Membranes and Filter for Water Purification and Disinfection in Outdoor Conditions. Colloids and Interfaces, 3(1), 35. https://doi.org/10.3390/colloids3010035




