Experimental Investigation of Water Based Colloidal Gas Aphron Fluid Stability
Abstract
:1. Introduction
2. Methodology
2.1. Materials
2.2. Tauchig Approach for Design of Experiment
2.3. Preparation of Fluids
2.4. Stability of CGAs
2.5. Visualization of CGAs
2.6. Rheological Characterization and Filtration Reduction Evaluation of the Fluids
3. Results and Discussion
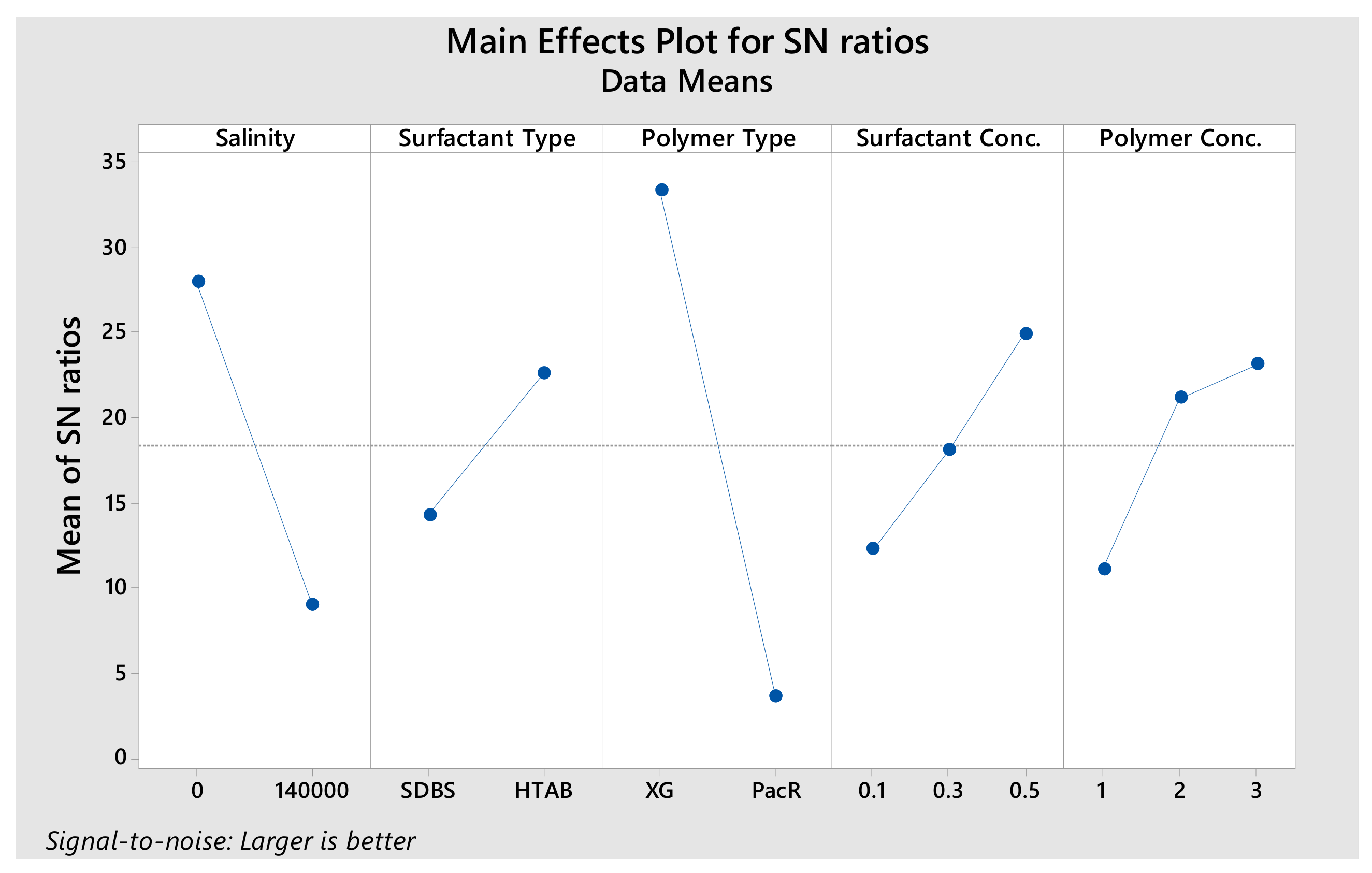
3.1. Design of Experiment Analyzes
3.2. Stability of CGAs
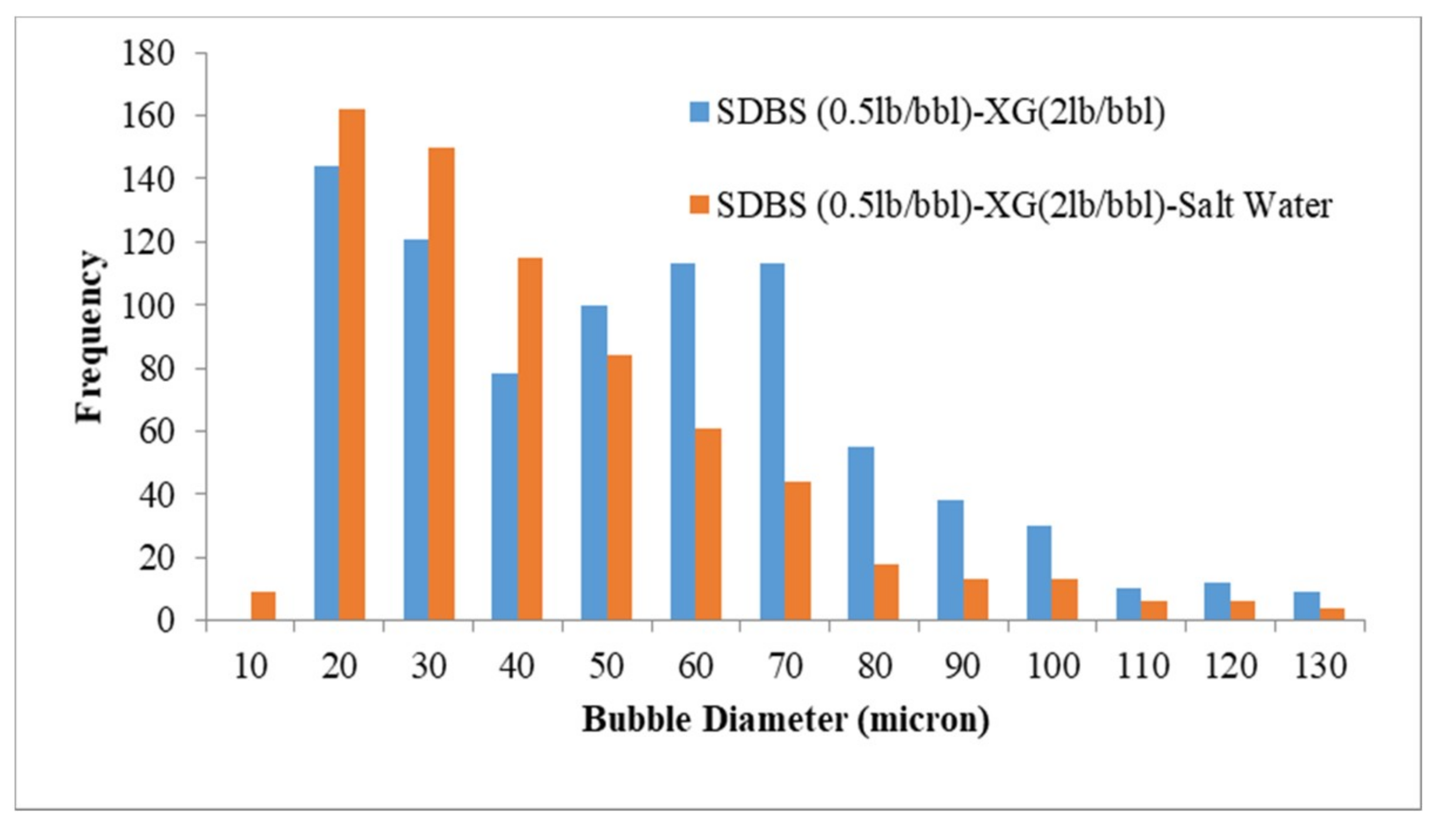
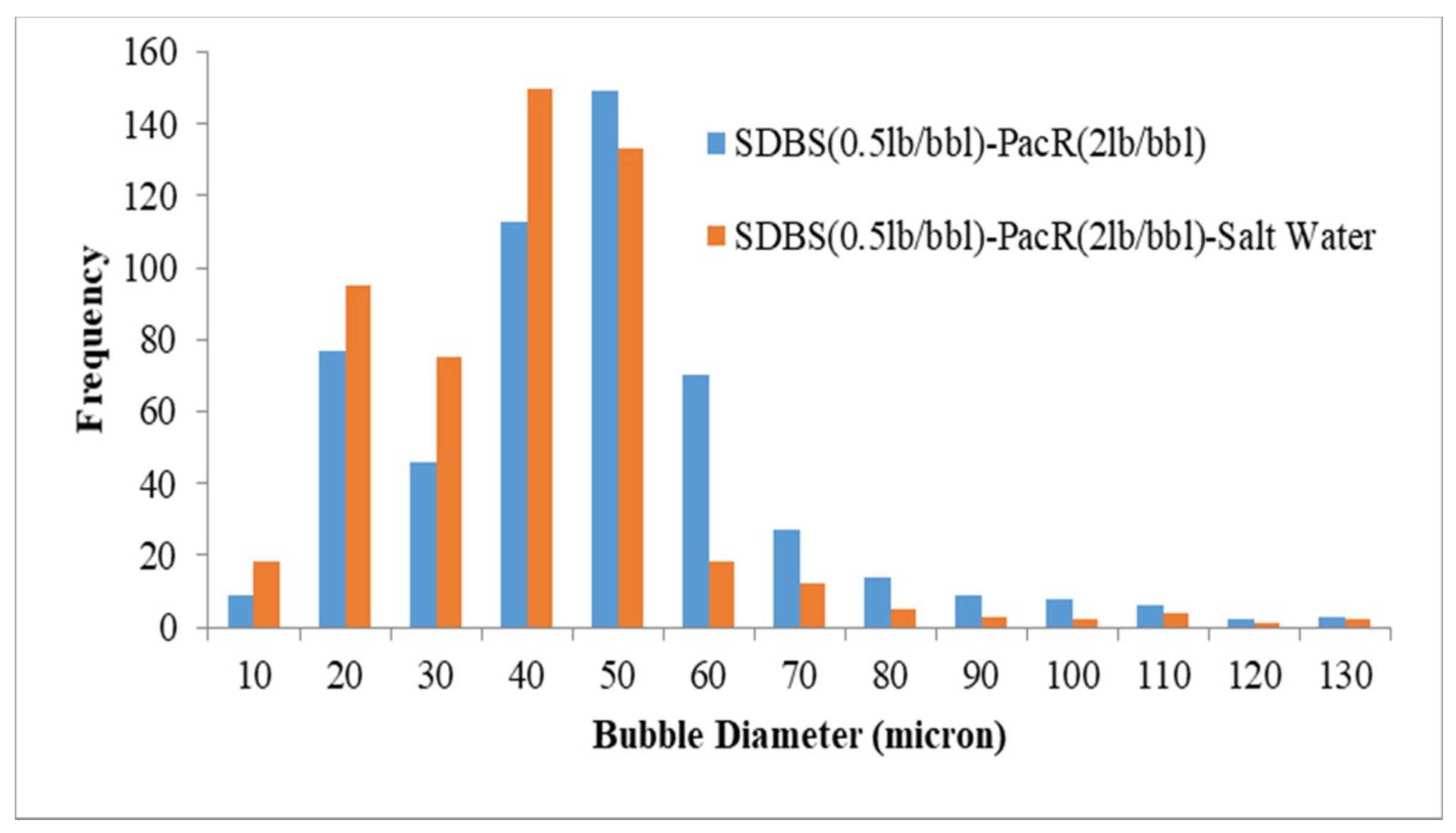
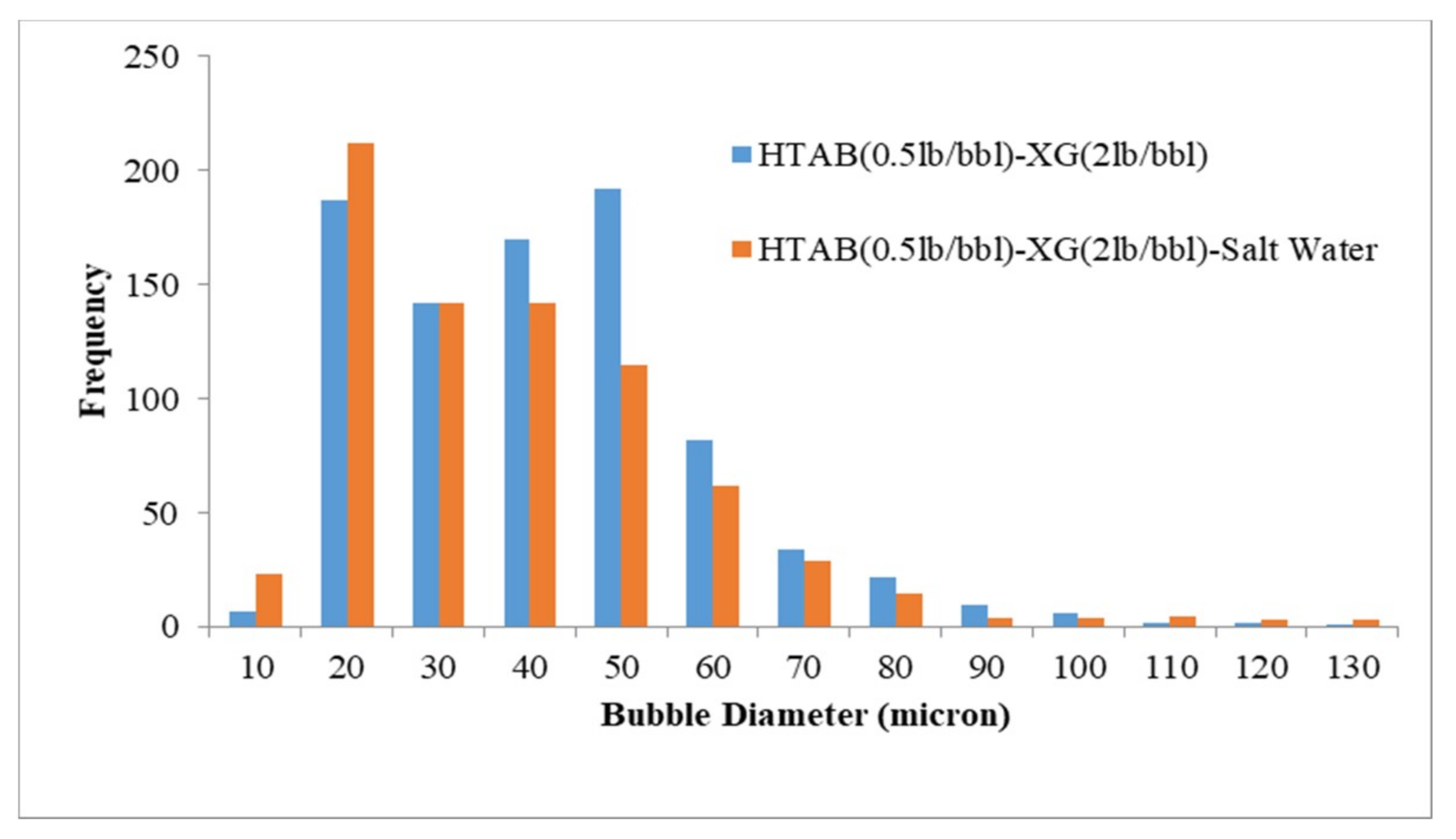
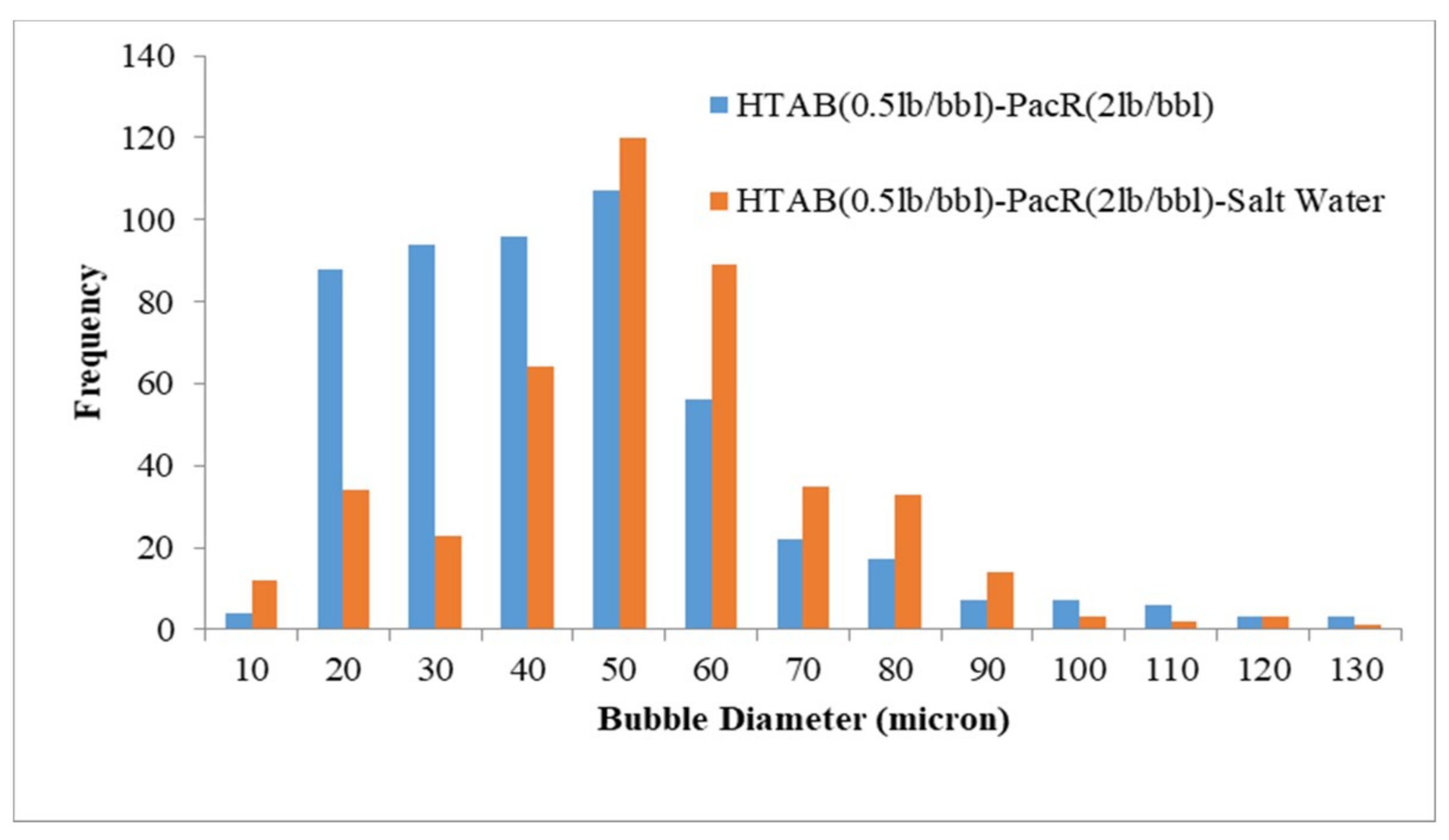
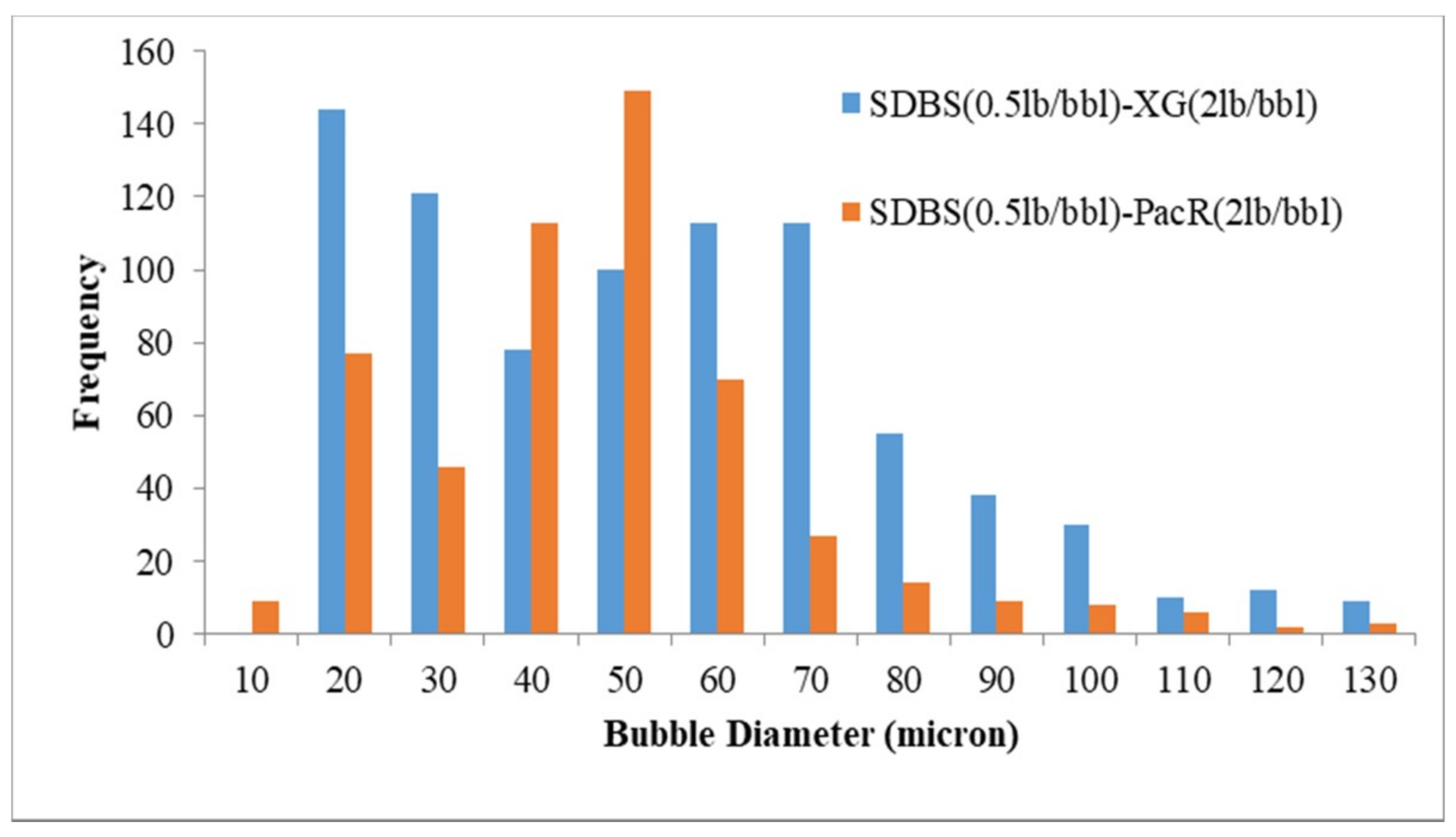
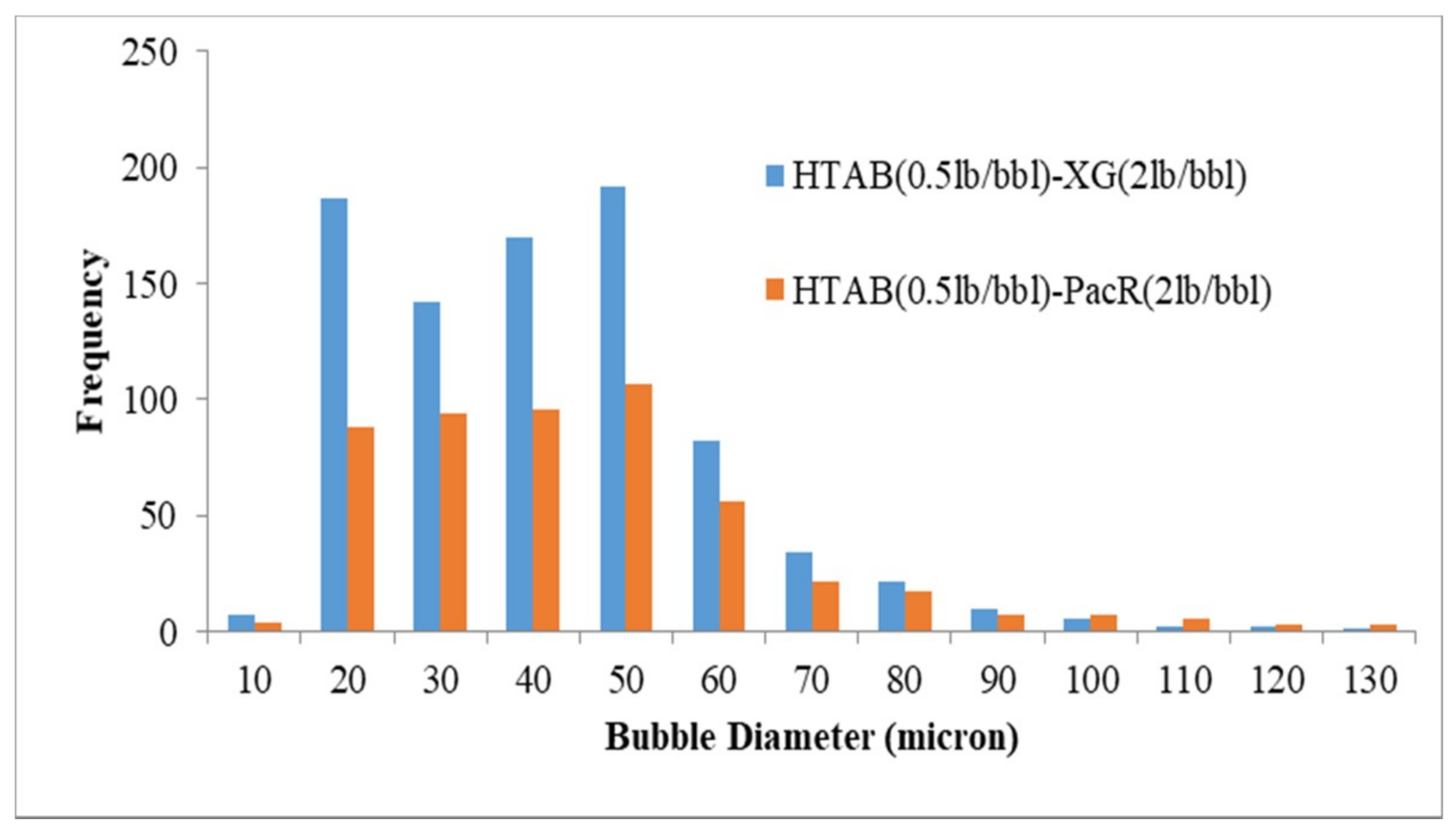
3.3. Visualization and size Distribution of the CGAs
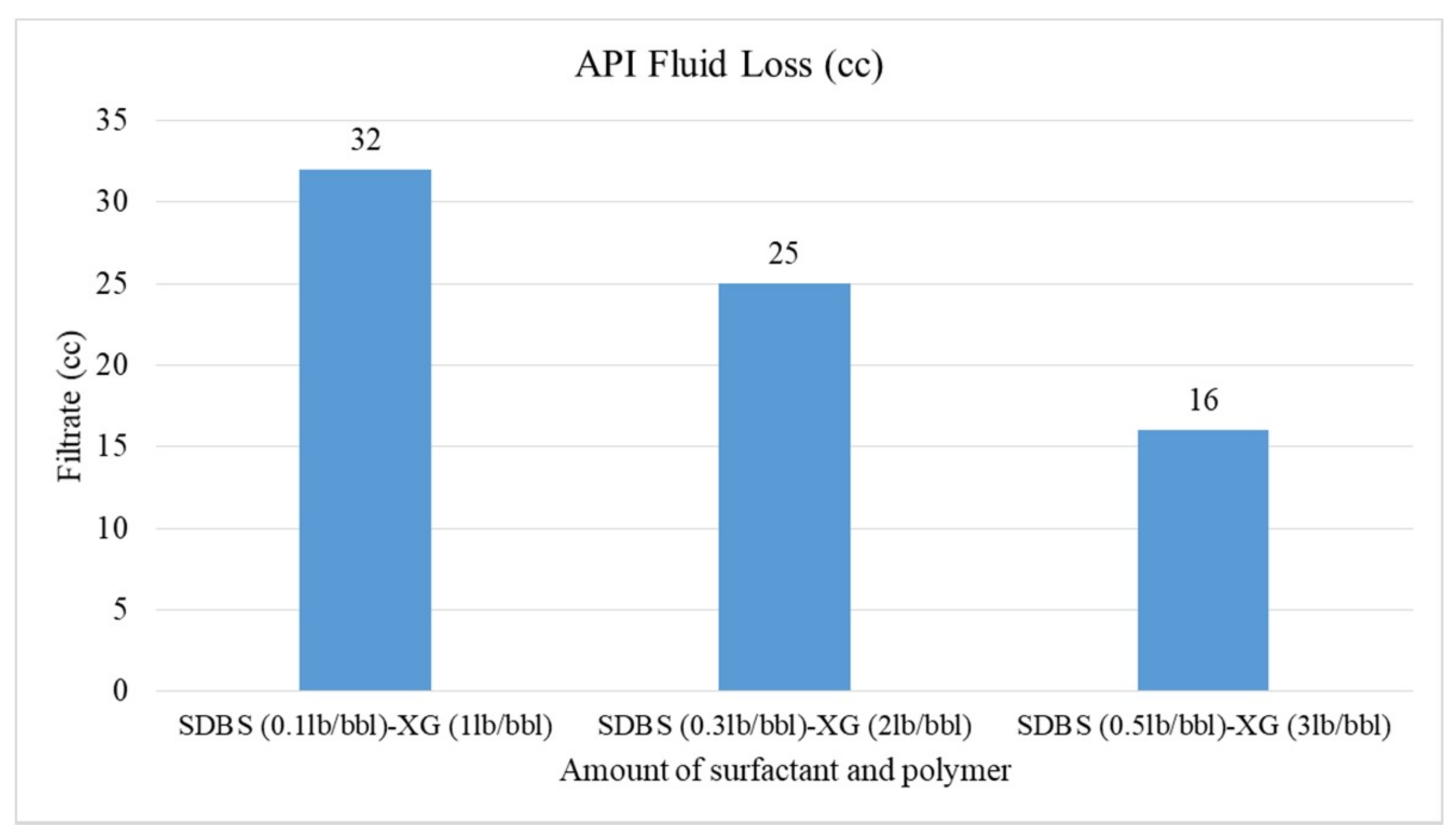
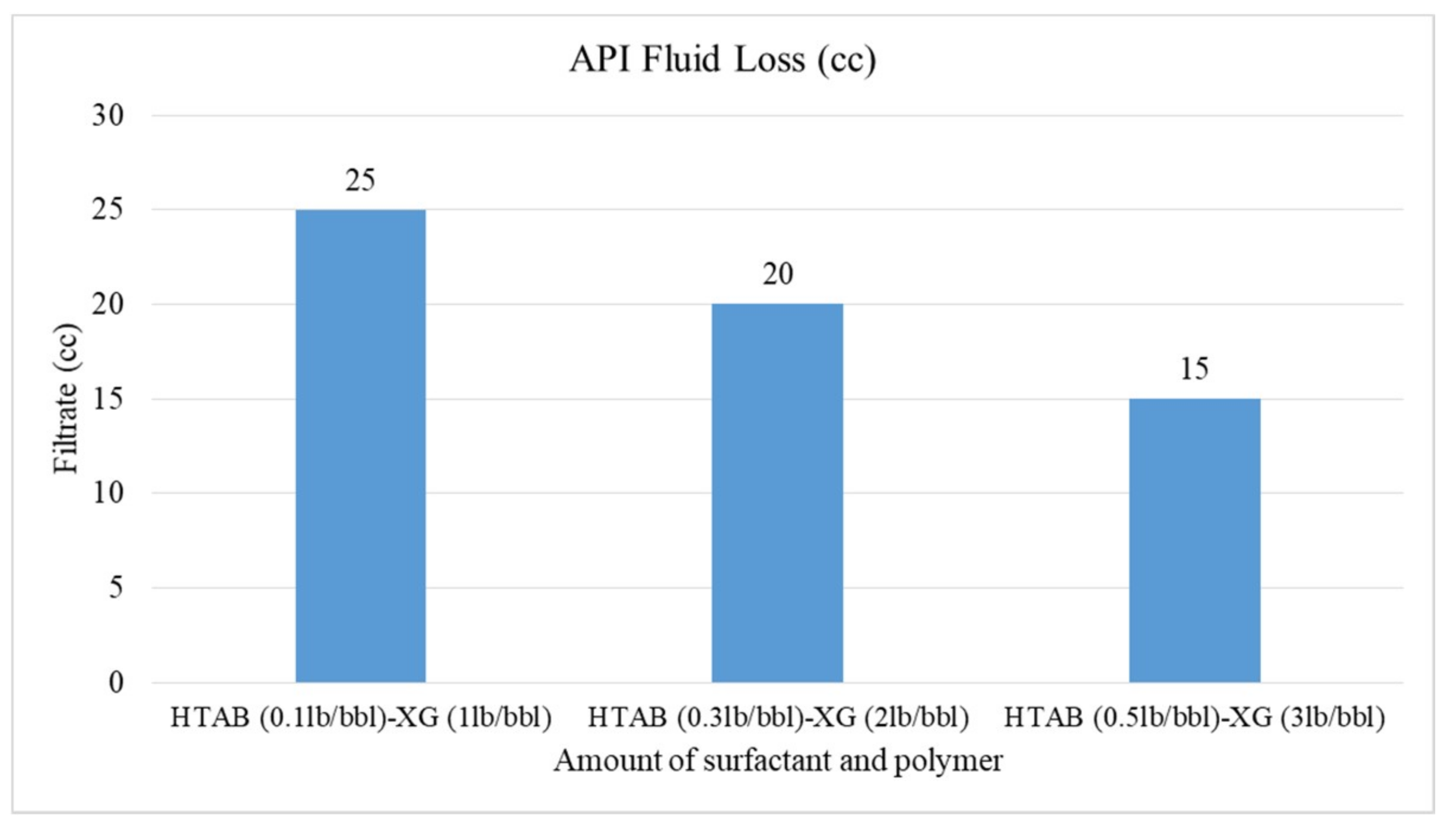
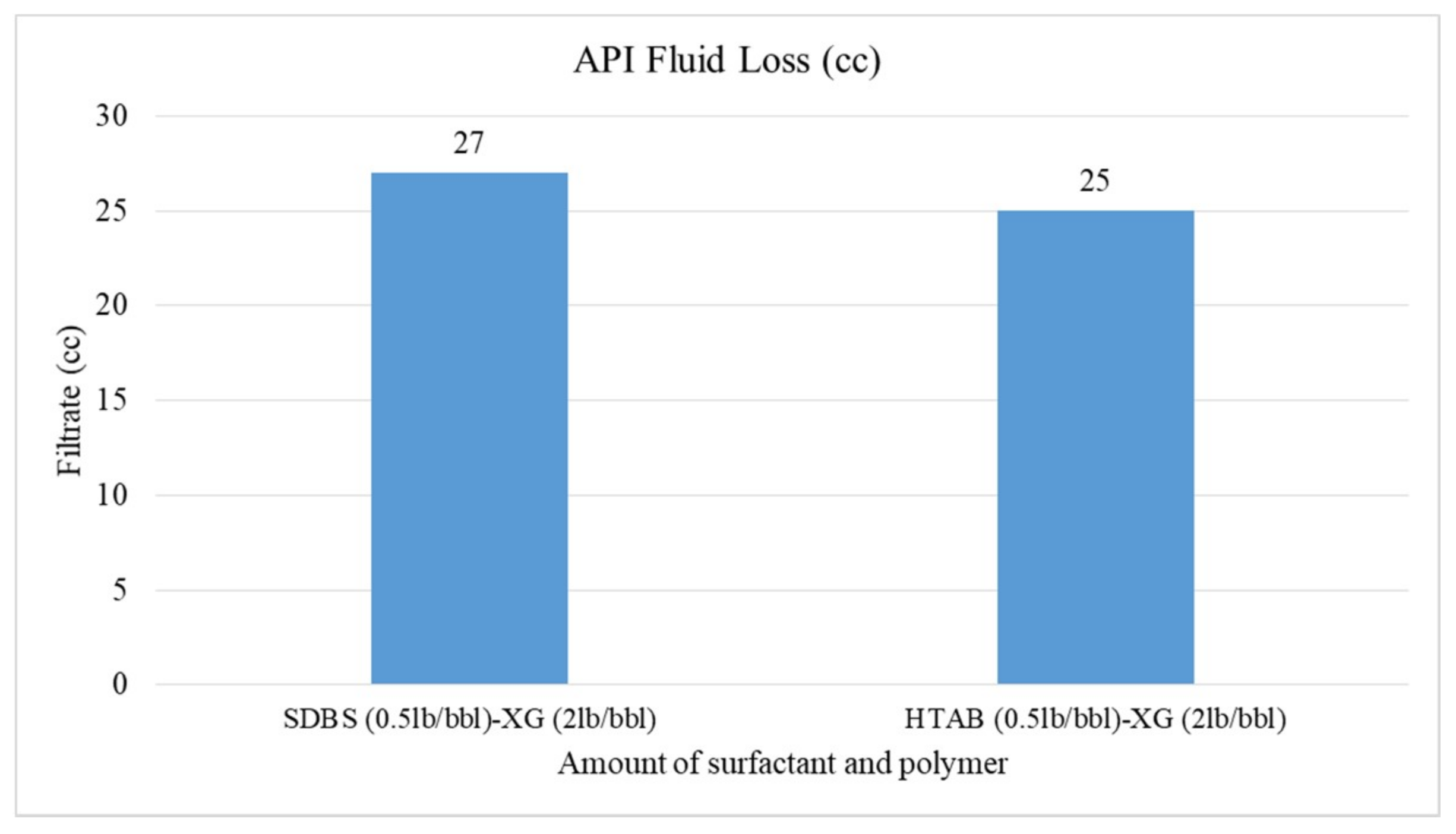
3.4. Rheological Properties and Filtration of CGAs
4. Conclusions
- According to DOE results, polymer type is the most effective and surfactant type is the least effective parameter on stability.
- By using XG polymer, the stability increases and micro-bubbles are more stable and disperse in the fluid than PacR polymer, which in the presence of PacR the micro-bubbles of CGA are very unstable and the liquid phase of the fluid drain rapidly just after aphron generation.
- Both SDBS and HTAB surfactants have a relatively close effect on micro-bubble stability of CGAs fluid.
- Increasing salinity in CGA fluid reduces the stability and the micro-bubbles generated with HTAB surfactant in an electrolyte system are more stable than SDBS surfactant.
- The apparent and plastic viscosities, yield point and gel strength (initial and 10 min) of CGA drilling fluids increase with the increase of the concentration of surfactant and polymer.
- The HTAB surfactant reduces filtration loss of CGA drilling fluid more than SDBS surfactant in the presence of XG polymer
- In an electrolyte system, the filtration loss is more compared to a non-electrolyte system, this is because of reduction in the number of bubbles and less stability of micro-bubbles in presence of salt in the fluid.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sebba, F. Foams and Biliquid Foams, Aphrons; John Wiley & Sons Ltd: Chichester, UK, 1987. [Google Scholar]
- Save, S.V.; Pangarkar, V.G. Characterisation of colloidal gas aphrons. Chem. Eng. Commun. 1994, 127, 35–54. [Google Scholar] [CrossRef]
- Jauregi, P.; Gilmour, S.; Varley, J. Characterisation of colloidal gas aphrons for subsequent use for protein recovery. Chem. Eng. J. 1997, 65, 1–11. [Google Scholar]
- Brookey, T. Micro-Bubbles: New Aphron Drill.-In Fluid Technique Reduces Formation Damage in Horizontal Wells. In Proceedings of the SPE Formation Damage Control Conference, Lafayette, LA, USA, 8–19 February 1998. [Google Scholar]
- Ramirez, F.; Greaves, R.; Montilva, J. Experience using microbubbles-aphron drilling fluid in mature reservoirs of Lake Maracaibo. In Proceedings of the International Symposium and Exhibition on Formation Damage Control, Lafayette, LA, USA, 20–21 February 2002. [Google Scholar]
- Growcock, F.B. Enhanced wellbore stabilization and reservoir productivity with aphron drilling fluid technology; Department of Energy: Houston, TX, USA, 2005. [Google Scholar]
- Bjorndalen, N.; Kuru, E. Physico-Chemical Characterizationof Aphron Based Drilling Fluids. In Proceedings of the Canadian International Petroleum Conference, Calgary, AB, Canada, 7–9 June 2005. [Google Scholar]
- Keshavarzi, B.; Javadi, A.; Bahramian, A.; Miller, R. Formation and stability of colloidal gas aphron based drilling fluid considering dynamic surface properties. J. Petrol. Sci. Eng. 2019, 174, 468–475. [Google Scholar] [CrossRef]
- Huang, T.; Crews, J.B.; Agrawal, G. Nanoparticle pseudocrosslinked micellar fluids: Optimal solution for fluid-loss control with internal breaking. In Proceedings of the SPE International Symposium and Exhibiton on Formation Damage Control, Lafayette, LA, USA, 10–12 February 2010. [Google Scholar]
- Arabloo, M.; Shahri, M.P.; Zamani, M. Characterization of colloidal gas aphron-fluids produced from a new plant-based surfactant. J. Disper. Sci. Technol. 2013, 34, 669–678. [Google Scholar] [CrossRef]
- Tabzar, A.; Arabloo, M.; Ghazanfari, M.H. Rheology, stability and filtration characteristics of Colloidal Gas. Aphron fluids: Role of surfactant and polymer type. J. Nat. Gas Sci. Eng. 2015, 26, 895–906. [Google Scholar]
- Growcock, F.B.; Khan, A.M.; Simon, G.A. Application of water-based and oil-based aphrons in drilling fluids. In Proceedings of the International Symposium on Oilfield Chemistry, Houston, TX, USA, 5–7 February 2003. [Google Scholar]
- Alizadeh, A.; Khamehchi, E. Experimental investigation of the oil based Aphron drilling fluid for determining the most stable fluid formulation. J. Petrol. Sci. Eng. 2019, 174, 525–532. [Google Scholar] [CrossRef]
- Parthasarathy, R.; Jameson, G.; Ahmed, N. Bubble breakup in stirred vessels-predicting the Sauter mean diameter. Chem. Eng. Res. Des. 1991, 69, 295–301. [Google Scholar]
- Roy, D.; Valsaraj, K.; Kottai, S. Separation of organic dyes from wastewater by using colloidal gas aphrons. Sep. Sci. Technol. 1992, 27, 573–588. [Google Scholar] [CrossRef]
- Dai, Y.; Deng, T. Stabilization and characterization of colloidal gas aphron dispersions. J. Colloid Int. Sci. 2003, 261, 360–365. [Google Scholar] [CrossRef]
- Alizadeh, A.; Khamehchi, E. Modeling of micro-bubble surfactant multi-layer drilling fluid stability based on single bubble behavior under pressure and temperature in a deviated gas well. J. Nat. Gas Sci. Eng. 2015, 26, 42–50. [Google Scholar] [CrossRef]
- Alizadeh, A.; Khamehchi, E. A model for predicting size distribution and liquid drainage from micro-bubble surfactant multi-layer fluids using population balance. Colloid Polym. Sci. 2015, 293, 3419–3427. [Google Scholar] [CrossRef]
- Alizadeh, A.; Khamehchi, E. Stability modeling of water-based surfactant covered micro-bubble fluids. J. Surf. Deterg. 2016, 19, 165–171. [Google Scholar] [CrossRef]
- Alizadeh, A.; Khamehchi, E. Mathematical modeling of the colloidal gas aphron motion through porous medium, including colloidal bubble generation and destruction. Colloid Polym. Sci. 2016, 294, 1075–1085. [Google Scholar] [CrossRef]
- Alizadeh, A.; Khamehchi, E. Modeling of Micro-Bubble Drilling Fluid Stability: Micro-Bubble Surfactant Multi-Layer, Drilling Fluid Stability Based on Single Bubble Behavior; LAP Lambert Academic Publishing: Saarbrücken, Germany, 2016. [Google Scholar]
- Alizadeh, A.; Khamehchi, E. Numerical and experimental investigation of micro-bubble fluid infiltration in porous media. Colloid Polym. Sci. 2017, 295, 529–541. [Google Scholar] [CrossRef]
- Alizadeh, A.; Khamehchi, E. Mathematical modeling of the Colloidal Gas. Aphron transport through porous medium using the filtration theory. J. Nat. Gas Sci. Eng. 2017, 44, 37–53. [Google Scholar] [CrossRef]
- Khamehchi, E.; Tabibzadeh, S.; Alizadeh, A. Rheological properties of Aphron based drilling fluids. Petrol. Explor. Dev. 2016, 43, 1076–1081. [Google Scholar] [CrossRef]
- Sadeghialiabadi, H.; Amiri, M. A new stability index for characterizing the colloidal gas aphrons dispersion. Colloids Surf. A Physicochem. Eng. Asp. 2015, 471, 170–177. [Google Scholar] [CrossRef]
- Kuru, E.; Bjorndalen, N.; Jossy, E.; Alvarez, J.M. Reducing formation damage with microbubble-based drilling fluid: Understanding the blocking ability. J. Can. Petrol. Technol. 2008, 47, 11. [Google Scholar] [CrossRef]
- Nareh’ei, M.A.; Shahri, M.P.; Zamani, M. Preparation and characterization of colloidal gas aphron based drilling fluids using a plant-based surfactant. In Proceedings of the SPE Saudi Arabia Section Technical Symposium and Exhibition, Al-Khobar, Saudi Arabia, 8–11 April 2012. [Google Scholar]
- Arabloo, M.; Shahri, M.P. Experimental studies on stability and viscoplastic modeling of colloidal gas aphron (CGA) based drilling fluids. J. Petrol. Sci. Eng. 2014, 113, 8–22. [Google Scholar] [CrossRef]
- Arabloo Nareh’ei, M.; Pordel Shahri, M.; Zamani, M. Rheological and filtration loss characteristics of colloidal gas aphron based drilling fluids. J. Jap. Petrol. Ins. 2012, 55, 182–190. [Google Scholar] [CrossRef]
- Bjorndalen, N.; Kuru, E. Stability of microbubble-based drilling fluids under downhole conditions. J. Canadian Petrol. Tech. 2008, 47, 6. [Google Scholar] [CrossRef]


| Sl. No | Parameters | Level 1 | Level 2 | Level 3 |
|---|---|---|---|---|
| 1 | Salinity, ppm | 0 | 140000 | - |
| 2 | Surfactant Type | SDBS | HTAB | - |
| 3 | Polymer Type | XG | PacR. | - |
| 4 | Surfactant Conc., lb/bbl | 0.1 | 0.3 | 0.5 |
| 5 | Polymer Conc., lb/bbl | 1 | 2 | 3 |
| Test No | Salinity (ppm) | Surfactant Type | Polymer Type | Surfactant Conc. (lb/bbl) | Polymer Conc. (lb/bbl) |
|---|---|---|---|---|---|
| 1 | 0 | SDBS | XG | 0.1 | 1 |
| 2 | 0 | SDBS | XG | 0.3 | 2 |
| 3 | 0 | SDBS | XG | 0.5 | 3 |
| 4 | 0 | SDBS | XG | 0.1 | 1 |
| 5 | 0 | SDBS | XG | 0.3 | 2 |
| 6 | 0 | SDBS | XG | 0.5 | 3 |
| 7 | 0 | SDBS | PacR | 0.1 | 1 |
| 8 | 0 | SDBS | PacR | 0.3 | 2 |
| 9 | 0 | SDBS | PacR | 0.5 | 3 |
| 10 | 0 | HTAB | XG | 0.1 | 1 |
| 11 | 0 | HTAB | XG | 0.3 | 2 |
| 12 | 0 | HTAB | XG | 0.5 | 3 |
| 13 | 0 | HTAB | PacR | 0.1 | 2 |
| 14 | 0 | HTAB | PacR | 0.3 | 3 |
| 15 | 0 | HTAB | PacR | 0.5 | 1 |
| 16 | 0 | HTAB | PacR | 0.1 | 2 |
| 17 | 0 | HTAB | PacR | 0.3 | 3 |
| 18 | 0 | HTAB | PacR | 0.5 | 1 |
| 19 | 140000 | SDBS | PacR | 0.1 | 2 |
| 20 | 140000 | SDBS | PacR | 0.3 | 3 |
| 21 | 140000 | SDBS | PacR | 0.5 | 1 |
| 22 | 140000 | SDBS | PacR | 0.1 | 2 |
| 23 | 140000 | SDBS | PacR | 0.3 | 3 |
| 24 | 140000 | SDBS | PacR | 0.5 | 1 |
| 25 | 140000 | SDBS | XG | 0.1 | 3 |
| 26 | 140000 | SDBS | XG | 0.3 | 1 |
| 27 | 140000 | SDBS | XG | 0.5 | 2 |
| 28 | 140000 | HTAB | PacR | 0.1 | 3 |
| 29 | 140000 | HTAB | PacR | 0.3 | 1 |
| 30 | 140000 | HTAB | PacR | 0.5 | 2 |
| 31 | 140000 | HTAB | XG | 0.1 | 3 |
| 32 | 140000 | HTAB | XG | 0.3 | 1 |
| 33 | 140000 | HTAB | XG | 0.5 | 2 |
| 34 | 140000 | HTAB | XG | 0.1 | 3 |
| 35 | 140000 | HTAB | XG | 0.3 | 1 |
| 36 | 140000 | HTAB | XG | 0.5 | 2 |
| Test No | Stability (min) | Density (gr/cc) | Vol. Fraction (%) |
|---|---|---|---|
| 1 | 48 | 0.44 | 46.97 |
| 2 | 273 | 0.40 | 53.95 |
| 3 | 843 | 0.49 | 44.44 |
| 4 | 50 | 0.42 | 50.00 |
| 5 | 264 | 0.42 | 52.70 |
| 6 | 820 | 0.43 | 48.53 |
| 7 | 5 | 0.39 | 45.31 |
| 8 | 10 | 0.30 | 60.23 |
| 9 | 28 | 0.30 | 68.75 |
| 10 | 16 | 0.55 | 31.37 |
| 11 | 150 | 0.56 | 35.19 |
| 12 | 360 | 0.57 | 32.69 |
| 13 | 1 | 0.60 | 20.45 |
| 14 | 7 | 0.51 | 36.36 |
| 15 | 1 | 0.63 | 25.53 |
| 16 | 0.5 | 0.59 | 22.22 |
| 17 | 6 | 0.52 | 35.19 |
| 18 | 1 | 0.64 | 23.91 |
| 19 | 0.1 | 1.11 | 0.00 |
| 20 | 0.1 | 1.10 | 0.00 |
| 21 | 0.1 | 1.06 | 0.00 |
| 22 | 0.1 | 1.10 | 0.00 |
| 23 | 0.1 | 1.10 | 0.00 |
| 24 | 0.1 | 1.10 | 0.00 |
| 25 | 0.5 | 0.84 | 7.89 |
| 26 | 1 | 0.82 | 7.89 |
| 27 | 45 | 0.70 | 23.91 |
| 28 | 2 | 0.66 | 27.08 |
| 29 | 2 | 0.49 | 43.00 |
| 30 | 6 | 0.46 | 50.00 |
| 31 | 312 | 0.65 | 31.37 |
| 32 | 36 | 0.47 | 49.28 |
| 33 | 42 | 0.43 | 53.95 |
| 34 | 320 | 0.64 | 32.69 |
| 35 | 32 | 0.46 | 50.00 |
| 36 | 38 | 0.43 | 52.70 |
| Level | Salinity | Surfactant Type | Polymer Type | Surfactant Conc. | Polymer Conc. |
|---|---|---|---|---|---|
| 1 | 27.89 | 14.23 | 33.26 | 12.23 | 16.37 |
| 2 | 8.94 | 22.59 | 3.56 | 18.11 | 11.06 |
| 3 | 24.89 | 23.08 | |||
| Delta | 18.95 | 8.37 | 29.70 | 12.65 | 12.02 |
| Rank | 2 | 5 | 1 | 3 | 4 |
| Composition | Average Bubble Size (Micron) |
|---|---|
| SDBS-XG | 55.6 |
| SDBS-PacR | 43.7 |
| HTAB-XG | 37.4 |
| HTAB-PacR | 46.7 |
| SDBS-XG-Salt Water | 41.5 |
| SDBS-PacR-Salt Water | 36.0 |
| HTAB-XG-Salt Water | 35.6 |
| HTAB-PacR-Salt Water | 48.7 |
| Composition | Salinity (ppm) | Surfactant Conc. (lb/bbl) | Polymer Conc. (lb/bbl) | PV (cP) | YP (lbf/100 ft2) | App. Vis. (cP) | Gel 10 sec (lbf/100 ft2) | Gel 10 min (lbf/100 ft2) |
|---|---|---|---|---|---|---|---|---|
| SDBS-XG | 0 | 0.1 | 1 | 8 | 22 | 19 | 8 | 11 |
| SDBS-XG | 0 | 0.3 | 2 | 11 | 31 | 26.5 | 13 | 17 |
| SDBS-XG | 0 | 0.5 | 3 | 18 | 45 | 40.5 | 25 | 29 |
| HTAB-XG | 0 | 0.1 | 1 | 11 | 20 | 21 | 7 | 9 |
| HTAB -XG | 0 | 0.3 | 2 | 15 | 30 | 30 | 13 | 16 |
| HTAB -XG | 0 | 0.5 | 3 | 21 | 39 | 40.5 | 20 | 24 |
| SDBS -XG | 140000 | 0.5 | 2 | 15 | 35 | 32.5 | 17 | 20 |
| HTAB-XG | 140000 | 0.1 | 3 | 13 | 37 | 31.5 | 18 | 22 |
| HTAB-XG | 140000 | 0.3 | 1 | 10 | 23 | 21.5 | 8 | 12 |
| HTAB -XG | 140000 | 0.5 | 2 | 15 | 27 | 28.5 | 12 | 15 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hosseini-Kaldozakh, S.A.; Khamehchi, E.; Dabir, B.; Alizadeh, A.; Mansoori, Z. Experimental Investigation of Water Based Colloidal Gas Aphron Fluid Stability. Colloids Interfaces 2019, 3, 31. https://doi.org/10.3390/colloids3010031
Hosseini-Kaldozakh SA, Khamehchi E, Dabir B, Alizadeh A, Mansoori Z. Experimental Investigation of Water Based Colloidal Gas Aphron Fluid Stability. Colloids and Interfaces. 2019; 3(1):31. https://doi.org/10.3390/colloids3010031
Chicago/Turabian StyleHosseini-Kaldozakh, Seyed Arman, Ehsan Khamehchi, Bahram Dabir, Ali Alizadeh, and Zohreh Mansoori. 2019. "Experimental Investigation of Water Based Colloidal Gas Aphron Fluid Stability" Colloids and Interfaces 3, no. 1: 31. https://doi.org/10.3390/colloids3010031
APA StyleHosseini-Kaldozakh, S. A., Khamehchi, E., Dabir, B., Alizadeh, A., & Mansoori, Z. (2019). Experimental Investigation of Water Based Colloidal Gas Aphron Fluid Stability. Colloids and Interfaces, 3(1), 31. https://doi.org/10.3390/colloids3010031





