Colloidal Stability of Positively Charged Dispersions of Styrene and Acrylic Copolymers in the Presence of TiO2 and CaCO3
Abstract
1. Introduction
2. Materials and Methods
2.1. Size and ζ-Potential Measurements
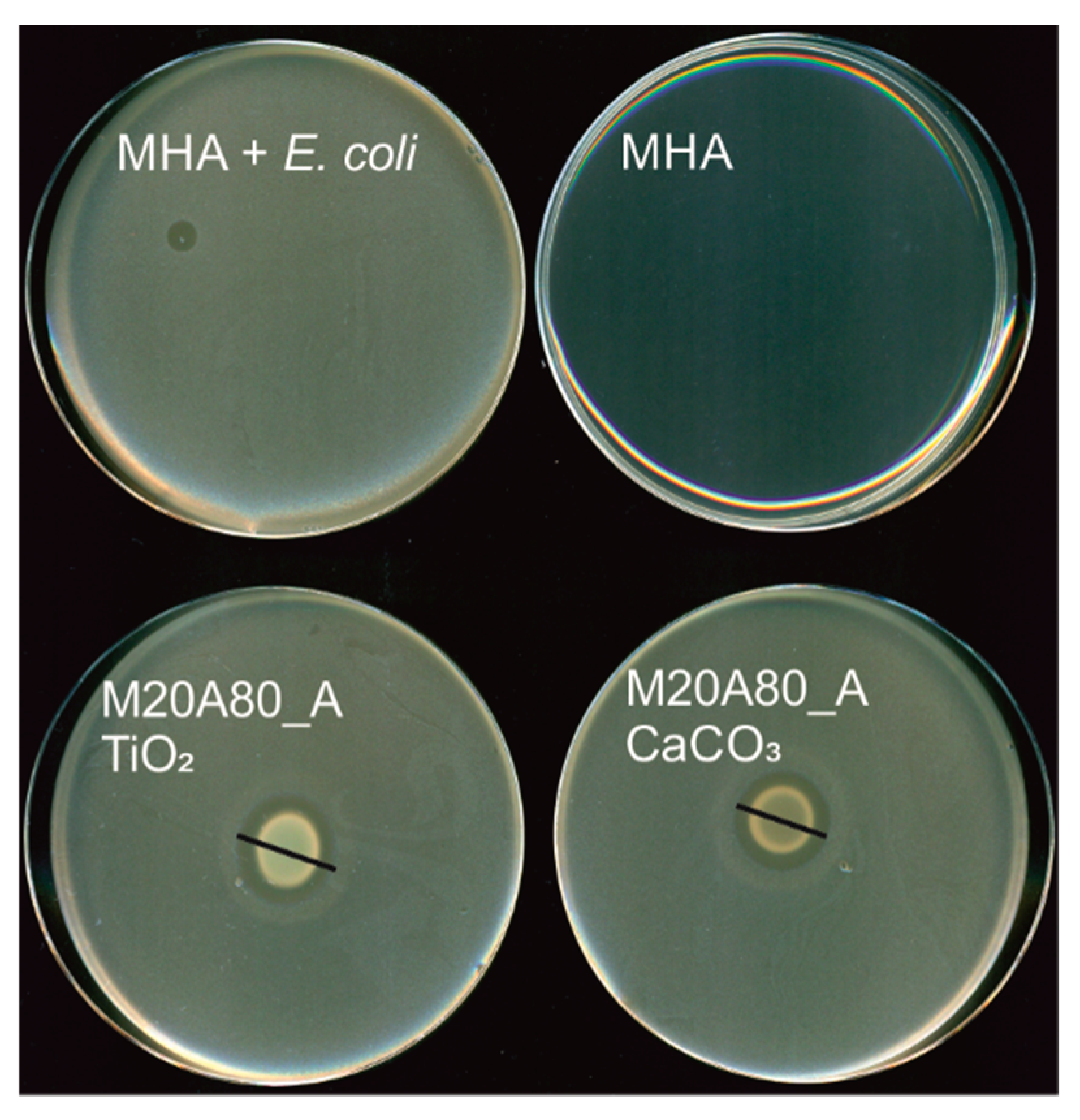
2.2. Antimicrobial Testing
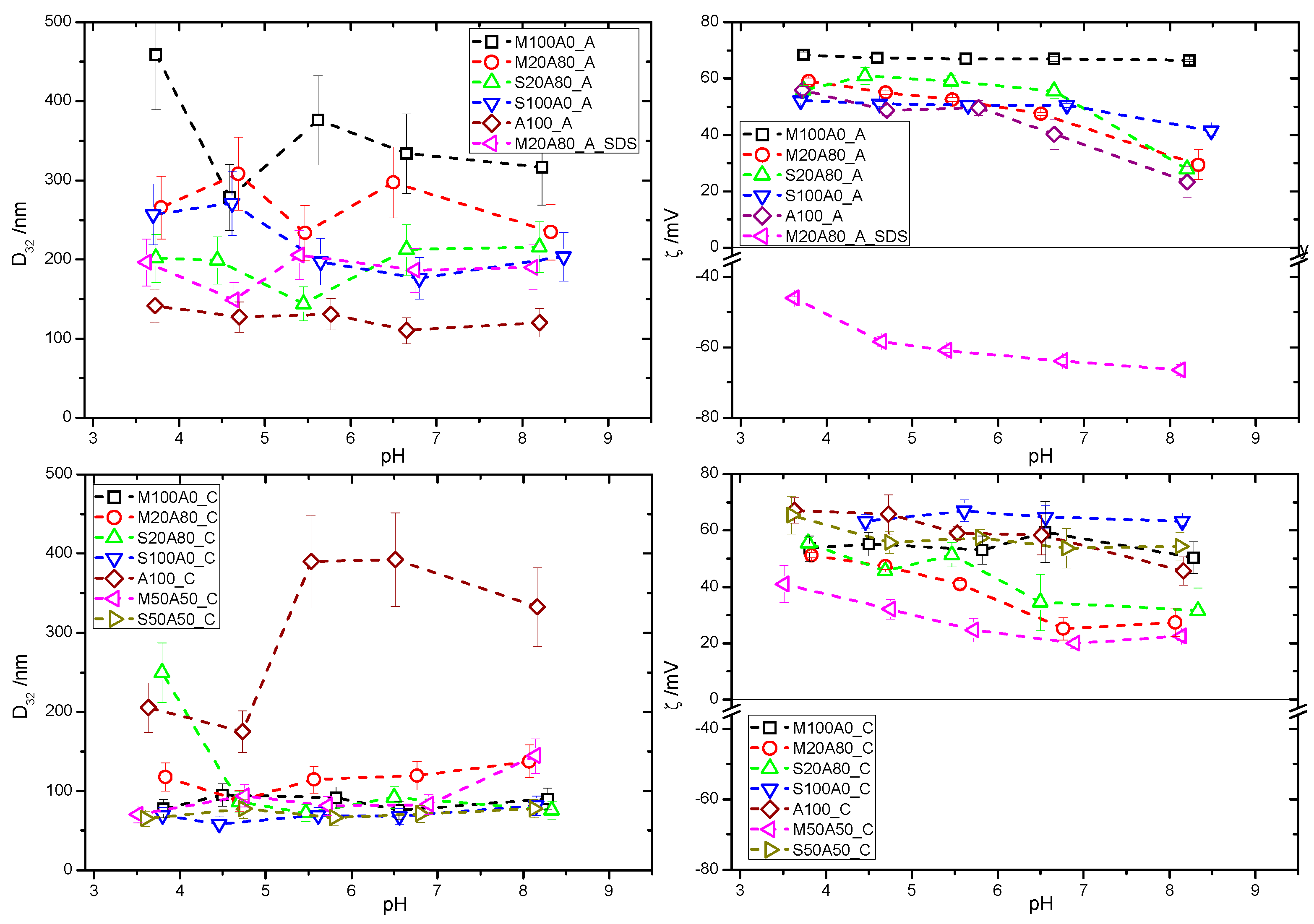
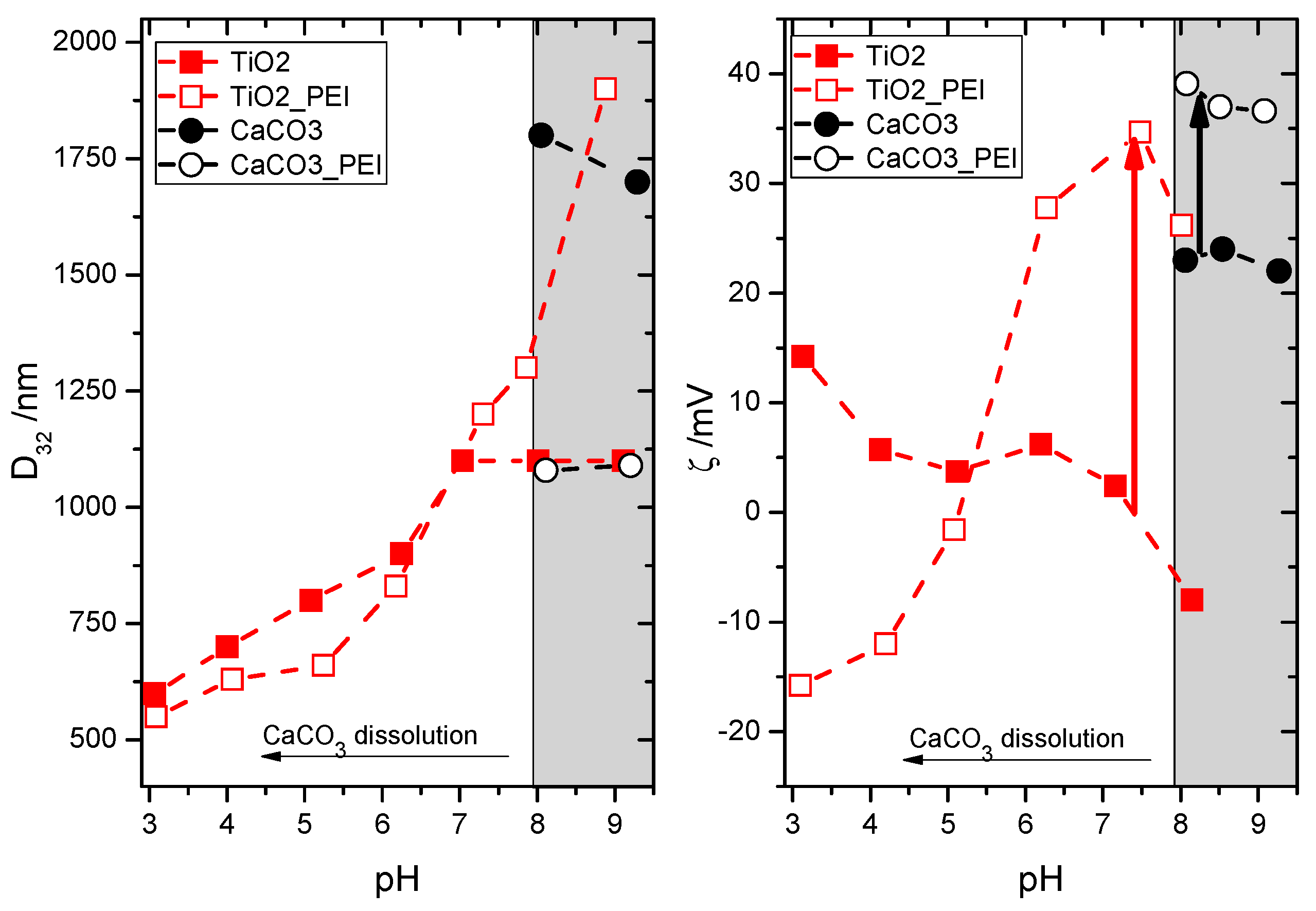
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cloutier, M.; Mantovani, D.; Rosei, F. Antibacterial Coatings: Challenges, Perspectives, and Opportunities. Trends Biotechnol. 2015, 33, 637–652. [Google Scholar] [CrossRef] [PubMed]
- Shaik, S. Is my chemical universe localized or delocalized? is there a future for chemical concepts? New J. Chem. 2007, 31, 2015. [Google Scholar] [CrossRef]
- Findlay, B.; Zhanel, G.G.; Schweizer, F. Cationic amphiphiles, a new generation of antimicrobials inspired by the natural antimicrobial peptide scaffold. Antimicrob. Agents Chemother. 2010, 54, 4049–4058. [Google Scholar] [CrossRef] [PubMed]
- Koprivnjak, T.; Peschel, A. Bacterial resistance mechanisms against host defense peptides. Cell. Mol. Life Sci. 2011, 68, 2243–2254. [Google Scholar] [CrossRef]
- Colomer, A.; Pinazo, A.; García, M.T.; Mitjans, M.; Vinardell, M.P.; Infante, M.R.; Martínez, V.; Pérez, L. PH-sensitive surfactants from lysine: Assessment of their cytotoxicity and environmental behavior. Langmuir 2012, 28, 5900–5912. [Google Scholar] [CrossRef] [PubMed]
- Salay, L.C.; Ferreira, M.; Oliveira, O.N.; Nakaie, C.R.; Schreier, S. Headgroup specificity for the interaction of the antimicrobial peptide tritrpticin with phospholipid Langmuir monolayers. Colloids Surf. B Biointerfaces 2012, 100, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chi, E.Y.; Schanze, K.S.; Whitten, D.G. Membrane activity of antimicrobial phenylene ethynylene based polymers and oligomers. Soft Matter 2012, 8, 8547. [Google Scholar] [CrossRef]
- Yang, L.; Gordon, V.D.; Mishra, A.; Som, A.; Purdy, K.R.; Davis, M.A.; Tew, G.N.; Wong, G.C.L. Synthetic antimicrobial oligomers induce a composition-dependent topological transition in membranes. J. Am. Chem. Soc. 2007, 129, 12141–12147. [Google Scholar] [CrossRef]
- Mensa, B.; Kim, Y.H.; Choi, S.; Scott, R.; Caputo, G.A.; DeGrado, W.F. Antibacterial mechanism of action of arylamide foldamers. Antimicrob. Agents Chemother. 2011, 55, 5043–5053. [Google Scholar] [CrossRef]
- Castillo, J.; Pinazo, A.; Carilla, J.; Infante, M.R.; Alsina, M.A.; Haro, I.; Clapés, P. Interaction of antimicrobial arginine-based cationic surfactants with liposomes and lipid monolayers. Langmuir 2004, 20, 3379–3387. [Google Scholar] [CrossRef]
- Thaker, H.D.; Som, A.; Ayaz, F.; Lui, D.; Pan, W.; Scott, R.W.; Anguita, J.; Tew, G.N. Synthetic mimics of antimicrobial peptides with immunomodulatory responses. J. Am. Chem. Soc. 2012, 134, 11088–11091. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; Zhang, B.; Ma, F.; Jia, C.; Xiao, C.; Zhang, B.; Xing, L.; Li, M. Novel mechanisms of surfactants against Candida albicans growth and morphogenesis. Chem. Biol. Interact. 2015, 227, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Vieira, D.B.; Carmona-Ribeiro, A.M. Cationic lipids and surfactants as antifungal agents: Mode of action. J. Antimicrob. Chemother. 2006, 58, 760–767. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, S.; Matsumura, Y.; Katoh-Kubo, K.; Tsuchido, T. Antibacterial activity of surfactants against Escherichia coli cells is influenced by carbon source and anaerobiosis. J. Appl. Microbiol. 2002, 93, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Montazer, M.; Rangchi, F.; Siavoshi, F. Preparation of Protective Disposable Hygiene Fabrics for Medical Applications. In Medical and Healthcare Textiles; Elsevier: New York, NY, USA, 2010; pp. 164–170. ISBN 9781845692247. [Google Scholar]
- Venkata Nancharaiah, Y.; Reddy, G.K.K.; Lalithamanasa, P.; Venugopalan, V.P. The ionic liquid 1-alkyl-3-methylimidazolium demonstrates comparable antimicrobial and antibiofilm behavior to a cationic surfactant. Biofouling 2012, 28, 1141–1149. [Google Scholar] [CrossRef] [PubMed]
- Fulmer, P.A.; Wynne, J.H. Development of broad-spectrum antimicrobial latex paint surfaces employing active amphiphilic compounds. ACS Appl. Mater. Interfaces 2011, 3, 2878–2884. [Google Scholar] [CrossRef]
- Makarewicz, E.N.; Shiichuk, A.V.; Syrotyns’ka, I.D. Quaternary ammonium salts as antimicrobial additives to water-dispersible paints. Russ. J. Appl. Chem. 2011, 84, 888–891. [Google Scholar] [CrossRef]
- Goodwin, J.W.; Ottewill, R.H.; Pelton, R. Studies on the preparation and characterization of monodisperse polystyrene latices V.: The preparation of cationic latices. Colloid Polym. Sci. 1979, 257, 61–69. [Google Scholar] [CrossRef]
- Antonietti, M.; Lohmann, S.; Van Niel, C. Polymerization in microemulsion. 2. Surface control and functionalization of microparticles. Macromolecules 1992, 25, 1139–1143. [Google Scholar] [CrossRef]
- Kong, X.Z.; Zhu, X.; Jiang, X.; Li, X. Preparation and full characterization of cationic latex of styrene–butyl acrylate. Polymer 2009, 50, 4220–4227. [Google Scholar] [CrossRef]
- Liu, Z.; Xiao, H.; Wiseman, N. Emulsifier-free emulsion copolymerization of styrene with quaternary ammonium cationic monomers. J. Appl. Polym. Sci. 2000, 76, 1129–1140. [Google Scholar] [CrossRef]
- Naves, A.F.; Palombo, R.R.; Carrasco, L.D.M.; Carmona-Ribeiro, A.M. Antimicrobial Particles from Emulsion Polymerization of Methyl Methacrylate in the Presence of Quaternary Ammonium Surfactants. Langmuir 2013, 29, 9677–9684. [Google Scholar] [CrossRef] [PubMed]
- Sanches, L.M.; Petri, D.F.S.; de Melo Carrasco, L.D.; Carmona-Ribeiro, A.M. The antimicrobial activity of free and immobilized poly (diallyldimethylammonium) chloride in nanoparticles of poly (methylmethacrylate). J. Nanobiotechnol. 2015, 13, 58. [Google Scholar] [CrossRef] [PubMed]
- Melo, L.D.; Palombo, R.R.; Petri, D.F.S.; Bruns, M.; Pereira, E.M.A.; Carmona-Ribeiro, A.M. Structure–Activity Relationship for Quaternary Ammonium Compounds Hybridized with Poly(methyl methacrylate). ACS Appl. Mater. Interfaces 2011, 3, 1933–1939. [Google Scholar] [CrossRef] [PubMed]
- Pereira, E.M.A.; Kosaka, P.M.; Rosa, H.; Vieira, D.B.; Kawano, Y.; Petri, D.F.S.; Carmona-Ribeiro, A.M. Hybrid Materials from Intermolecular Associations between Cationic Lipid and Polymers. J. Phys. Chem. B 2008, 112, 9301–9310. [Google Scholar] [CrossRef]
- Ramos, J.; Forcada, J.; Hidalgo-Alvarez, R. Cationic Polymer Nanoparticles and Nanogels: From Synthesis to Biotechnological Applications. Chem. Rev. 2014, 114, 367–428. [Google Scholar] [CrossRef]
- Wojciechowski, K.; Kaczorowski, M.; Mierzejewska, J.; Parzuchowski, P. Antimicrobial dispersions and films from positively charged styrene and acrylic copolymers. Colloids Surf. B Biointerfaces 2018, 172, 532–540. [Google Scholar] [CrossRef]
- Van der Kooij, H.M.; Sprakel, J. Watching paint dry; more exciting than it seems. Soft Matter 2015, 11, 6353–6359. [Google Scholar] [CrossRef]
- Steward, P.A.; Hearn, J.; Wilkinson, M.C. An overview of polymer latex film formation and properties. Adv. Colloid Interface Sci. 2000, 195–267. [Google Scholar] [CrossRef]
- Kiil, S. Drying of latex films and coatings: Reconsidering the fundamental mechanisms. Prog. Org. Coat. 2006, 57, 236–250. [Google Scholar] [CrossRef]
- Keddie, J.L. Film formation of latex. Mater. Sci. Eng. R Rep. 1997, 21, 101–170. [Google Scholar] [CrossRef]
- Lyklema, J. Surface charges and electrokinetic charges: Distinctions and juxtapositionings. Colloids Surf. A Physicochem. Eng. Asp. 2011, 376, 2–8. [Google Scholar] [CrossRef]
- Bergström, L. Hamaker constants of inorganic materials. Adv. Colloid Interface Sci. 1997, 70, 125–169. [Google Scholar] [CrossRef]
- Farrokhpay, S. A review of polymeric dispersant stabilisation of titania pigment. Adv. Colloid Interface Sci. 2009, 151, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Elzbieciak-Wodka, M.; Popescu, M.N.; Ruiz-Cabello, F.J.M.; Trefalt, G.; Maroni, P.; Borkovec, M. Measurements of dispersion forces between colloidal latex particles with the atomic force microscope and comparison with Lifshitz theory. J. Chem. Phys. 2014, 140, 104906. [Google Scholar] [CrossRef] [PubMed]
| Sample | Monomer Content (mol%) | Surfactant | Initiator | Yield (%) | ||
|---|---|---|---|---|---|---|
| ST | MMA | EA | ||||
| M100A0_A | 0.0 | 100.0 | 0.0 | CTAB | Na2S2O8 | 84.6 |
| S100A0_A | 100.0 | 0.0 | 0.0 | CTAB | Na2S2O8 | 75.4 |
| M20A80_A | 0 | 21.5 | 78.5 | CTAB | Na2S2O8 | 60.1 |
| S20A80_A | 24.5 | 0.0 | 75.5 | CTAB | Na2S2O8 | 96.7 |
| A100_A | 0 | 0.0 | 100.0 | CTAB | Na2S2O8 | 75.7 |
| M20A80_A_SDS | 0 | 23.1 | 76.9 | SDS | Na2S2O8 | 87.6 |
| M100A0_C | 0 | 100.0 | 0.0 | CTAB | AAPH | 96.8 |
| S100A0_C | 100.0 | 0.0 | 0.0 | CTAB | AAPH | 96.8 |
| M20A80_C | 0 | 19.6 | 80.4 | CTAB | AAPH | 97.1 |
| S20A80_C | 20.9 | 0.0 | 79.1 | CTAB | AAPH | 99.9 |
| M50A50_C | 0 | 44.8 | 55.2 | CTAB | AAPH | 93.0 |
| S50A50_C | 54.5 | 0.0 | 45.5 | CTAB | AAPH | 92.4 |
| A100_C | 0 | 0.0 | 100.0 | CTAB | AAPH | 94.0 |
| Mineral Particles | CaCO3 + 1% PEI pH 8.2 | TiO2 + 1% PEI pH 7.5 | ||
|---|---|---|---|---|
| Polymer | Size (nm) | ζ (Mv) | Size (nm) | ζ (Mv) |
| M20A80_A | 190 ± 28; 708 ± 106 | +35 ± 4 | 503 ± 45 | +9 ± 1 |
| S20A80_A | 126 ± 19; 495 ± 74 | +37 ± 4 | 92 ± 15; 792 ± 114 | +16 ± 2 |
| M20A80_C | 108 ± 16; 754 ± 113 | +39 ± 4 | 767 ± 113 | +12 ± 2 |
| S20A80_C | 100 ± 15; 667 ± 100 | +39 ± 4 | 84 ± 12; 463 ± 70; 917 ± 140 | +19 ± 2 |
| A100_A | 146 ± 22; 1130 ± 170 | +36 ± 4 | 460 ± 43 | +31 ± 4 |
| A100_C | 75 ± 11; 170 ± 26 | +39 ± 4 | 370 ± 48; 1280 ± 185 | +42 ± 4 |
| Polymer | Additive | P. aeruginosa | E. coli | S. aureus | C. albicans | ||||
|---|---|---|---|---|---|---|---|---|---|
| Not Rinsed | Rinsed with Milli-Q | Not Rinsed | Rinsed with Milli-Q | Not Rinsed | Rinsed with Milli-Q | Not Rinsed | Rinsed with Milli-Q | ||
| M20A80_A | - | 0 | nt | 18 ± 6 | 0 | 82 ± 4 | 30 ± 5 | 14 ± 1 | 0 |
| CaCO3 | 0 | 0 | 16 ± 1 | 0 | 21 ± 3 | 0 | 16 ± 1 | 14 ± 1 | |
| TiO2 | 11 ± 1 | 0 | 18 ± 1 | 0 | 22 ± 4 | 10 ± 2 | 16 ± 1 | 13 ± 1 | |
| S20A80_A | - | 0 | nt | 0 | nt | 30 ± 4 | nt | 13 ± 2 | nt |
| CaCO3 | 0 | 0 | 0 | 0 | 15 ± 1 | 0 | 0 | 0 | |
| TiO2 | 0 | 0 | 0 | 0 | 14 ± 2 | 0 | 0 | 0 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wojciechowski, K.; Gutarowicz, M.; Janke, K.; Jurek, I.; Kaczorowski, M.; Mierzejewska, J.; Parzuchowski, P. Colloidal Stability of Positively Charged Dispersions of Styrene and Acrylic Copolymers in the Presence of TiO2 and CaCO3. Colloids Interfaces 2019, 3, 20. https://doi.org/10.3390/colloids3010020
Wojciechowski K, Gutarowicz M, Janke K, Jurek I, Kaczorowski M, Mierzejewska J, Parzuchowski P. Colloidal Stability of Positively Charged Dispersions of Styrene and Acrylic Copolymers in the Presence of TiO2 and CaCO3. Colloids and Interfaces. 2019; 3(1):20. https://doi.org/10.3390/colloids3010020
Chicago/Turabian StyleWojciechowski, Kamil, Małgorzata Gutarowicz, Kamil Janke, Ilona Jurek, Marcin Kaczorowski, Jolanta Mierzejewska, and Paweł Parzuchowski. 2019. "Colloidal Stability of Positively Charged Dispersions of Styrene and Acrylic Copolymers in the Presence of TiO2 and CaCO3" Colloids and Interfaces 3, no. 1: 20. https://doi.org/10.3390/colloids3010020
APA StyleWojciechowski, K., Gutarowicz, M., Janke, K., Jurek, I., Kaczorowski, M., Mierzejewska, J., & Parzuchowski, P. (2019). Colloidal Stability of Positively Charged Dispersions of Styrene and Acrylic Copolymers in the Presence of TiO2 and CaCO3. Colloids and Interfaces, 3(1), 20. https://doi.org/10.3390/colloids3010020







