A Review on Synthesis Methods of Phyllosilicate- and Graphene-Filled Composite Hydrogels
Abstract
:1. Introduction
2. Fabrication of Composite Hydrogels
3. Crosslinking Approach
4. Blending Approach
5. Clay-Based Hydrogels
6. Graphene Oxide- and Graphene-Based Hydrogels
7. Radical Polymerization Method
8. Synthesis Method by High-Energy Irradiations
9. Enzyme-Driven Synthesis Method
10. Functional Group-Induced Gelation
| Hydrogel Matrix | Fillers | Loading (wt%) | Mechanical Strength | Ref. |
|---|---|---|---|---|
| PAM | GO | 0.03 | 65.5 kPa | [118] |
| PAA | GO | 0.5 | 777.3 kPa | [119] |
| poly(AMPS-co-AM) | RGO | 1.0 | ~98 kPa | [120] |
| PAA | Graphene | 3.0 | 19.09 MPa | [121] |
| PAM | Graphene | 3.0 | 1.31 MPa | [122] |
| PAA | Graphene | 0.5 | 0.006 MPa | [123] |
| PAA | GO | 0.073 | 25 kPa | [124] |
| PAA-g-amylose | GO | 3.0 | 42.47 MPa | [125] |
| PAA | Graphene | 2.0 | 411.9 kPa | [126] |
| PAM/Fe3O4 | RGO | 0.15 | 0.945 MPa | [127] |
| PVA/PEG | GO | 1.5 | 4.2 MPa | [128] |
| poly(AA-co-AMPS)/carrageenan | RGO | 2.5 | 488.5 kPa | [108] |
| PAA/gelatin | GO | 0.3 | 0.25 MPa | [129] |
| PAA | GO | 0.1 | 4 MPa | [130] |
11. Characterization Techniques
12. Nuclear Magnetic Resonance (NMR) Spectroscopy
13. Diffusion Ordered NMR Spectroscopy (DOSY)
14. Water Uptake
15. Small-Angle X-ray Scattering (SAXS)
16. Small-Angle Neutron Scattering (SANS)
17. Spectroscopy
18. Summary and Outlook
Author Contributions
Funding
Conflicts of Interest
References
- Sharma, S.; Tiwari, S. A review on biomacromolecular hydrogel classification and its applications. Int. J. Biol. Macromol. 2020, 162, 737–747. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Liu, Y.; Qin, D.; Sun, M.; Wang, T.; Chen, X. Research status of self-healing hydrogel for wound management: A review. Int. J. Biol. Macromol. 2020, 164, 2108–2123. [Google Scholar] [CrossRef] [PubMed]
- Ziv-Polat, O.; Skaat, H.; Shahar, A.; Margel, S. Novel magnetic fibrin hydrogel scaffolds containing thrombin and growth factors conjugated iron oxide nanoparticles for tissue engineering. Int. J. Nanomed. 2012, 7, 1259. [Google Scholar] [CrossRef] [Green Version]
- Cascone, S.; Lamberti, G. Hydrogel-based commercial products for biomedical applications: A review. Int. J. Pharm. 2020, 573, 118803. [Google Scholar] [CrossRef] [PubMed]
- Bai, H.; Li, C.; Wang, X.; Shi, G. A pH-sensitive graphene oxide composite hydrogel. Chem. Commun. 2010, 46, 2376–2378. [Google Scholar] [CrossRef]
- Shah, R.; Gashi, B.; Hoque, S.; Marian, M.; Rosenkranz, A. Enhancing Mechanical and Biomedical Properties of Protheses-Surface and Material Design. Surf. Interfaces 2021, 27, 101498. [Google Scholar] [CrossRef]
- Ganguly, S.; Das, P.; Itzhaki, E.; Hadad, E.; Gedanken, A.; Margel, S. Microwave-synthesized polysaccharide-derived carbon dots as therapeutic cargoes and toughening agents for elastomeric gels. ACS Appl. Mater. Interfaces 2020, 12, 51940–51951. [Google Scholar] [CrossRef]
- Ganguly, S.; Das, P.; Das, N.C. Characterization tools and techniques of hydrogels. In Hydrogels Based on Natural Polymers; Elsevier: Amsterdam, The Netherlands, 2020; pp. 481–517. [Google Scholar]
- Ganguly, S.; Maity, T.; Mondal, S.; Das, P.; Das, N.C. Starch functionalized biodegradable semi-IPN as a pH-tunable controlled release platform for memantine. Int. J. Biol. Macromol. 2017, 95, 185–198. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Kim, H.-W. Emerging properties of hydrogels in tissue engineering. J. Tissue Eng. 2018, 9, 2041731418768285. [Google Scholar] [CrossRef] [Green Version]
- Nicodemus, G.D.; Bryant, S.J. Cell encapsulation in biodegradable hydrogels for tissue engineering applications. Tissue Eng. Part B Rev. 2008, 14, 149–165. [Google Scholar] [CrossRef]
- Hunt, J.A.; Chen, R.; van Veen, T.; Bryan, N. Hydrogels for tissue engineering and regenerative medicine. J. Mater. Chem. B 2014, 2, 5319–5338. [Google Scholar] [CrossRef]
- Zhu, J. Bioactive modification of poly (ethylene glycol) hydrogels for tissue engineering. Biomaterials 2010, 31, 4639–4656. [Google Scholar] [CrossRef] [Green Version]
- Skaat, H.; Ziv-Polat, O.; Shahar, A.; Last, D.; Mardor, Y.; Margel, S. Magnetic scaffolds enriched with bioactive nanoparticles for tissue engineering. Adv. Healthc. Mater. 2012, 1, 168–171. [Google Scholar] [CrossRef] [PubMed]
- Ganguly, S.; Mondal, S.; Das, P.; Bhawal, P.; Maity, P.P.; Ghosh, S.; Dhara, S.; Das, N.C. Design of psyllium-g-poly (acrylic acid-co-sodium acrylate)/cloisite 10A semi-IPN nanocomposite hydrogel and its mechanical, rheological and controlled drug release behaviour. Int. J. Biol. Macromol. 2018, 111, 983–998. [Google Scholar] [CrossRef] [PubMed]
- Das, P.; Ganguly, S.; Margel, S.; Gedanken, A. Tailor made magnetic nanolights: Fabrication to cancer theranostics applications. Nanoscale Adv. 2021, 3, 6762–6796. [Google Scholar] [CrossRef]
- Manandhar, P.; Calvert, P.D.; Buck, J.R. Elastomeric ionic hydrogel sensor for large strains. IEEE Sens. J. 2012, 12, 2052–2061. [Google Scholar] [CrossRef]
- Bhat, A.; Amanor-Boadu, J.M.; Guiseppi-Elie, A. Toward impedimetric measurement of acidosis with a pH-responsive hydrogel sensor. ACS Sens. 2020, 5, 500–509. [Google Scholar] [CrossRef]
- Richter, A.; Paschew, G.; Klatt, S.; Lienig, J.; Arndt, K.-F.; Adler, H.-J.P. Review on hydrogel-based pH sensors and microsensors. Sensors 2008, 8, 561–581. [Google Scholar] [CrossRef] [Green Version]
- Herber, S.; Olthuis, W.; Bergveld, P. A swelling hydrogel-based PCO2 sensor. Sens. Actuators B Chem. 2003, 91, 378–382. [Google Scholar] [CrossRef]
- Salehi, A.A.; Ghannadi-Maragheh, M.; Torab-Mostaedi, M.; Torkaman, R.; Asadollahzadeh, M. Hydrogel materials as an emerging platform for desalination and the production of purified water. Sep. Purif. Rev. 2021, 50, 380–399. [Google Scholar] [CrossRef]
- Amiri, S.; Asghari, A.; Vatanpour, V.; Rajabi, M. Fabrication and characterization of a novel polyvinyl alcohol-graphene oxide-sodium alginate nanocomposite hydrogel blended PES nanofiltration membrane for improved water purification. Sep. Purif. Technol. 2020, 250, 117216. [Google Scholar] [CrossRef]
- Thakur, S.; Sharma, B.; Verma, A.; Chaudhary, J.; Tamulevicius, S.; Thakur, V.K. Recent approaches in guar gum hydrogel synthesis for water purification. Int. J. Polym. Anal. Charact. 2018, 23, 621–632. [Google Scholar] [CrossRef] [Green Version]
- Yazdi, M.K.; Vatanpour, V.; Taghizadeh, A.; Taghizadeh, M.; Ganjali, M.R.; Munir, M.T.; Habibzadeh, S.; Saeb, M.R.; Ghaedi, M. Hydrogel membranes: A review. Mater. Sci. Eng. C 2020, 114, 111023. [Google Scholar] [CrossRef] [PubMed]
- Oveissi, F.; Fletcher, D.F.; Dehghani, F.; Naficy, S. Tough hydrogels for soft artificial muscles. Mater. Des. 2021, 203, 109609. [Google Scholar] [CrossRef]
- Browe, D.P.; Wood, C.; Sze, M.T.; White, K.A.; Scott, T.; Olabisi, R.M.; Freeman, J.W. Characterization and optimization of actuating poly (ethylene glycol) diacrylate/acrylic acid hydrogels as artificial muscles. Polymer 2017, 117, 331–341. [Google Scholar] [CrossRef] [Green Version]
- Sano, K.; Ishida, Y.; Aida, T. Synthesis of anisotropic hydrogels and their applications. Angew. Chem. Int. Ed. 2018, 57, 2532–2543. [Google Scholar] [CrossRef]
- Bassil, M.; Ibrahim, M.; El Tahchi, M. Artificial muscular microfibers: Hydrogel with high speed tunable electroactivity. Soft Matter 2011, 7, 4833–4838. [Google Scholar] [CrossRef]
- Appiah, C.; Arndt, C.; Siemsen, K.; Heitmann, A.; Staubitz, A.; Selhuber-Unkel, C. Living materials herald a new era in soft robotics. Adv. Mater. 2019, 31, 1807747. [Google Scholar] [CrossRef] [PubMed]
- Vallittu, P.K. High-aspect ratio fillers: Fiber-reinforced composites and their anisotropic properties. Dent. Mater. 2015, 31, 1–7. [Google Scholar] [CrossRef]
- Ganguly, S. Preparation/processing of polymer-graphene composites by different techniques. In Polymer Nanocomposites Containing Graphene; Elsevier: Amsterdam, The Netherlands, 2022; pp. 45–74. [Google Scholar] [CrossRef]
- Pacelli, S.; Rampetsreiter, K.; Modaresi, S.; Subham, S.; Chakravarti, A.R.; Lohfeld, S.; Detamore, M.S.; Paul, A. Fabrication of a double-cross-linked interpenetrating polymeric network (IPN) hydrogel surface modified with polydopamine to modulate the osteogenic differentiation of adipose-derived stem cells. ACS Appl. Mater. Interfaces 2018, 10, 24955–24962. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suzuki, K.; Saito, Y.; Tokuoka, Y.; Abe, M.; Sato, T. Poly (ethylene oxide)/poly (propylene oxide)/poly (ethylene oxide) triblock copolymer as a sustained-release carrier for perfume compounds. J. Am. Oil Chem. Soc. 1997, 74, 55–59. [Google Scholar] [CrossRef]
- Vadnere, M.; Amidon, G.; Lindenbaum, S.; Haslam, J.L. Thermodynamic studies on the gel-sol transition of some pluronic polyols. Int. J. Pharm. 1984, 22, 207–218. [Google Scholar] [CrossRef]
- Jeong, B.; Kibbey, M.R.; Birnbaum, J.C.; Won, Y.-Y.; Gutowska, A. Thermogelling biodegradable polymers with hydrophilic backbones: PEG-g-PLGA. Macromolecules 2000, 33, 8317–8322. [Google Scholar] [CrossRef]
- Vernon, B.; Gutowska, A.; Wan Kim, S.; Han Bae, Y. Thermally reversible polymer gels for biohybrid artificial pancreas. In Proceedings of the Macromolecular Symposia; 1996; pp. 155–167. [Google Scholar]
- Gutowska, A.; Jeong, B.; Jasionowski, M. Injectable gels for tissue engineering. Anat. Rec. Off. Publ. Am. Assoc. Anat. 2001, 263, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Sivashanmugam, A.; Kumar, R.A.; Priya, M.V.; Nair, S.V.; Jayakumar, R. An overview of injectable polymeric hydrogels for tissue engineering. Eur. Polym. J. 2015, 72, 543–565. [Google Scholar] [CrossRef]
- Galperin, A.; Long, T.J.; Ratner, B.D. Degradable, thermo-sensitive poly (N-isopropyl acrylamide)-based scaffolds with controlled porosity for tissue engineering applications. Biomacromolecules 2010, 11, 2583–2592. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qureshi, D.; Nayak, S.K.; Maji, S.; Anis, A.; Kim, D.; Pal, K. Environment sensitive hydrogels for drug delivery applications. Eur. Polym. J. 2019, 120, 109220. [Google Scholar] [CrossRef]
- Bai, X.; Lü, S.; Cao, Z.; Gao, C.; Duan, H.; Xu, X.; Sun, L.; Gao, N.; Feng, C.; Liu, M. Self-reinforcing injectable hydrogel with both high water content and mechanical strength for bone repair. Chem. Eng. J. 2016, 288, 546–556. [Google Scholar] [CrossRef]
- Ribeiro, C.A.; Martins, M.V.S.; Bressiani, A.H.; Bressiani, J.C.; Leyva, M.E.; de Queiroz, A.A.A. Electrochemical preparation and characterization of PNIPAM-HAp scaffolds for bone tissue engineering. Mater. Sci. Eng. C 2017, 81, 156–166. [Google Scholar] [CrossRef]
- Liu, J.; Jiang, L.; He, S.; Zhang, J.; Shao, W. Recent progress in PNIPAM-based multi-responsive actuators: A mini-review. Chem. Eng. J. 2021, 133496. [Google Scholar] [CrossRef]
- Ayar, Z.; Shafieian, M.; Mahmoodi, N.; Sabzevari, O.; Hassannejad, Z. A rechargeable drug delivery system based on pNIPAM hydrogel for the local release of curcumin. J. Appl. Polym. Sci. 2021, 138, 51167. [Google Scholar] [CrossRef]
- Zheng, H.; Zhang, S.; Yang, C.; Yin, H.; Liu, W.; Lu, K. Simultaneous removal of Ni (II) and Cr (VI) from aqueous solution by froth flotation using PNIPAM-CS intelligent nano-hydrogels as collector. J. Mol. Liq. 2021, 342, 117551. [Google Scholar] [CrossRef]
- Saikia, A.; Aggarwal, S.; Mandal, U. Preparation and controlled drug release characteristics of thermoresponsive PEG/poly (NIPAM-co-AMPS) hydrogels. Int. J. Polym. Mater. 2013, 62, 39–44. [Google Scholar] [CrossRef]
- Ganguly, S.; Das, P.; Das, T.K.; Ghosh, S.; Das, S.; Bose, M.; Mondal, M.; Das, A.K.; Das, N.C. Acoustic cavitation assisted destratified clay tactoid reinforced in situ elastomer-mimetic semi-IPN hydrogel for catalytic and bactericidal application. Ultrason. Sonochem. 2020, 60, 104797. [Google Scholar] [CrossRef]
- Takeno, H.; Aoki, Y.; Kimura, K. Effects of Silica and Clay Nanoparticles on the Mechanical Properties of Poly (vinyl alcohol) Nanocomposite Hydrogels. Colloids Surf. A Physicochem. Eng. Asp. 2021, 630, 127592. [Google Scholar] [CrossRef]
- Wang, Y.-T.; Zhao, H.-B.; Degracia, K.; Han, L.-X.; Sun, H.; Sun, M.; Wang, Y.-Z.; Schiraldi, D.A. Green approach to improving the strength and flame retardancy of poly (vinyl alcohol)/clay aerogels: Incorporating biobased gelatin. ACS Appl. Mater. Interfaces 2017, 9, 42258–42265. [Google Scholar] [CrossRef]
- Ganguly, S.; Margel, S. Remotely controlled magneto-regulation of therapeutics from magnetoelastic gel matrices. Biotechnol. Adv. 2020, 107611. [Google Scholar] [CrossRef]
- Yang, W.; Qi, G.; Kenny, J.M.; Puglia, D.; Ma, P. Effect of cellulose nanocrystals and lignin nanoparticles on mechanical, antioxidant and water vapour barrier properties of glutaraldehyde crosslinked PVA films. Polymers 2020, 12, 1364. [Google Scholar] [CrossRef]
- Clark, A.H.; Ross-Murphy, S.B. The concentration dependence of biopolymer gel modulus. Br. Polym. J. 1985, 17, 164–168. [Google Scholar] [CrossRef]
- Ganguly, S.; Grinberg, I.; Margel, S. Layer by layer controlled synthesis at room temperature of tri-modal (MRI, fluorescence and CT) core/shell superparamagnetic IO/human serum albumin nanoparticles for diagnostic applications. Polym. Adv. Technol. 2021, 32, 3909–3921. [Google Scholar] [CrossRef]
- Ganguly, S.; Das, N.C. Water uptake kinetics and control release of agrochemical fertilizers from nanoclay-assisted semi-interpenetrating sodium acrylate-based hydrogel. Polym. Plast. Technol. Eng. 2017, 56, 744–761. [Google Scholar] [CrossRef]
- Saha, K.; Dutta, K.; Basu, A.; Adhikari, A.; Chattopadhyay, D.; Sarkar, P. Controlled delivery of tetracycline hydrochloride intercalated into smectite clay using polyurethane nanofibrous membrane for wound healing application. Nano-Struct. Nano-Objects 2020, 21, 100418. [Google Scholar] [CrossRef]
- Haraguchi, K.; Farnworth, R.; Ohbayashi, A.; Takehisa, T. Compositional effects on mechanical properties of nanocomposite hydrogels composed of poly (N, N-dimethylacrylamide) and clay. Macromolecules 2003, 36, 5732–5741. [Google Scholar] [CrossRef]
- Okay, O.; Oppermann, W. Polyacrylamide−clay nanocomposite hydrogels: Rheological and light scattering characterization. Macromolecules 2007, 40, 3378–3387. [Google Scholar] [CrossRef] [Green Version]
- Kamoun, E.A.; Menzel, H. HES-HEMA nanocomposite polymer hydrogels: Swelling behavior and characterization. J. Polym. Res. 2012, 19, 1–14. [Google Scholar] [CrossRef]
- Kaşgöz, H.; Durmus, A. Dye removal by a novel hydrogel-clay nanocomposite with enhanced swelling properties. Polym. Adv. Technol. 2008, 19, 838–845. [Google Scholar] [CrossRef]
- Zhang, J.; Yuan, K.; Wang, Y.-P.; Gu, S.-J.; Zhang, S.-T. Preparation and properties of polyacrylate/bentonite superabsorbent hybrid via intercalated polymerization. Mater. Lett. 2007, 61, 316–320. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, J.; Wang, A. Removal of methylene blue from aqueous solution using chitosan-g-poly (acrylic acid)/montmorillonite superadsorbent nanocomposite. Colloids Surf. A Physicochem. Eng. Asp. 2008, 322, 47–53. [Google Scholar] [CrossRef]
- Kaşgöz, H.; Durmuş, A.; Kaşgöz, A. Enhanced swelling and adsorption properties of AAm-AMPSNa/clay hydrogel nanocomposites for heavy metal ion removal. Polym. Adv. Technol. 2008, 19, 213–220. [Google Scholar] [CrossRef]
- Liu, P.S.; Li, L.; Zhou, N.L.; Zhang, J.; Wei, S.H.; Shen, J. Waste polystyrene foam-graft-acrylic acid/montmorillonite superabsorbent nanocomposite. J. Appl. Polym. Sci. 2007, 104, 2341–2349. [Google Scholar] [CrossRef]
- Liu, P.S.; Li, L.; Zhou, N.L.; Zhang, J.; Wei, S.H.; Shen, J. Synthesis and properties of a poly (acrylic acid)/montmorillonite superabsorbent nanocomposite. J. Appl. Polym. Sci. 2006, 102, 5725–5730. [Google Scholar] [CrossRef]
- Ganguly, S.; Das, N.C. Synthesis of a novel pH responsive phyllosilicate loaded polymeric hydrogel based on poly (acrylic acid-co-N-vinylpyrrolidone) and polyethylene glycol for drug delivery: Modelling and kinetics study for the sustained release of an antibiotic drug. RSC Adv. 2015, 5, 18312–18327. [Google Scholar] [CrossRef]
- Wang, J.; Wang, W.; Wang, A. Synthesis, characterization and swelling behaviors of hydroxyethyl cellulose-g-poly (acrylic acid)/attapulgite superabsorbent composite. Polym. Eng. Sci. 2010, 50, 1019–1027. [Google Scholar] [CrossRef]
- Al, E.; Güçlü, G.; İyim, T.B.; Emik, S.; Özgümüş, S. Synthesis and properties of starch-graft-acrylic acid/Na-montmorillonite superabsorbent nanocomposite hydrogels. J. Appl. Polym. Sci. 2008, 109, 16–22. [Google Scholar] [CrossRef]
- Xu, K.; Wang, J.; Xiang, S.; Chen, Q.; Yue, Y.; Su, X.; Song, C.; Wang, P. Polyampholytes superabsorbent nanocomposites with excellent gel strength. Compos. Sci. Technol. 2007, 67, 3480–3486. [Google Scholar] [CrossRef]
- Qiu, H.; Yu, J. Polyacrylate/(carboxymethylcellulose modified montmorillonite) superabsorbent nanocomposite: Preparation and water absorbency. J. Appl. Polym. Sci. 2008, 107, 118–123. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, L.; Wang, A. Preparation and properties of chitosan-g-poly (acrylic acid)/montmorillonite superabsorbent nanocomposite via in situ intercalative polymerization. Ind. Eng. Chem. Res. 2007, 46, 2497. [Google Scholar] [CrossRef]
- Santiago, F.; Mucientes, A.E.; Osorio, M.; Poblete, F.J. Synthesis and swelling behaviour of poly (sodium acrylate)/sepiolite superabsorbent composites and nanocomposites. Polym. Int. 2006, 55, 843–848. [Google Scholar] [CrossRef]
- Lee, W.F.; Yang, L.G. Superabsorbent polymeric materials. XII. Effect of montmorillonite on water absorbency for poly (sodium acrylate) and montmorillonite nanocomposite superabsorbents. J. Appl. Polym. Sci. 2004, 92, 3422–3429. [Google Scholar] [CrossRef]
- Zhang, H.; Yu, J.; Wang, H.; Xue, L. Investigation of microstructures and ultraviolet aging properties of organo-montmorillonite/SBS modified bitumen. Mater. Chem. Phys. 2011, 129, 769–776. [Google Scholar] [CrossRef]
- Zheng, Y.; Gao, T.; Wang, A. Preparation, swelling, and slow-release characteristics of superabsorbent composite containing sodium humate. Ind. Eng. Chem. Res. 2008, 47, 1766–1773. [Google Scholar] [CrossRef]
- Zhang, J.; Li, A.; Wang, A. Synthesis and characterization of multifunctional poly (acrylic acid-co-acrylamide)/sodium humate superabsorbent composite. React. Funct. Polym. 2006, 66, 747–756. [Google Scholar] [CrossRef]
- Xie, Y.; Wu, J.; Lin, J.; Wei, Y.; Zhong, J. A conductive hydrogel by poly (sodium acrylate)/montmorillonite superabsorbent composite. Polym. Polym. Compos. 2007, 15, 29–33. [Google Scholar] [CrossRef]
- Zhang, Y.; Fan, L.; Zhao, P.; Zhang, L.; Chen, H. Preparation of nanocomposite superabsorbents based on hydrotalcite and poly (acrylic-co-acrylamide) by inverse suspension polymerization. Compos. Interfaces 2008, 15, 747–757. [Google Scholar] [CrossRef]
- Zheng, Y.; Wang, A. Study on superabsorbent composites. XVIII. Preparation, characterization, and property evaluation of poly (acrylic acid-co-acrylamide)/organomontmorillonite/sodium humate superabsorbent composites. J. Appl. Polym. Sci. 2008, 108, 211–219. [Google Scholar] [CrossRef]
- Lee, W.F.; Chen, Y.C. Preparation of reactive mineral powders used for poly (sodium acrylate) composite superabsorbents. J. Appl. Polym. Sci. 2005, 97, 855–861. [Google Scholar] [CrossRef]
- Zhang, Y.; Fan, L.; Cheng, L.; Zhang, L.; Chen, H. Preparation and morphology of high-performance exfoliated poly (sodium acrylate)/hydrotalcite nanocomposite superabsorbents. Polym. Eng. Sci. 2009, 49, 264–271. [Google Scholar] [CrossRef]
- Wang, W.; Wang, A. Preparation, characterization and properties of superabsorbent nanocomposites based on natural guar gum and modified rectorite. Carbohydr. Polym. 2009, 77, 891–897. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, J.; Wang, A. Preparation and swelling properties of superabsorbent nanocomposites based on natural guar gum and organo-vermiculite. Appl. Clay Sci. 2009, 46, 21–26. [Google Scholar] [CrossRef]
- Wang, W.; Wang, A. Preparation, swelling, and stimuli-responsive characteristics of superabsorbent nanocomposites based on carboxymethyl cellulose and rectorite. Polym. Adv. Technol. 2011, 22, 1602–1611. [Google Scholar] [CrossRef]
- Huang, X.; Xu, S.; Zhong, M.; Wang, J.; Feng, S.; Shi, R. Modification of Na-bentonite by polycations for fabrication of amphoteric semi-IPN nanocomposite hydrogels. Appl. Clay Sci. 2009, 42, 455–459. [Google Scholar] [CrossRef]
- Li, P.; Kim, N.H.; Lee, J.H. Swelling behavior of polyacrylamide/laponite clay nanocomposite hydrogels: pH-sensitive property. Compos. Part B Eng. 2009, 40, 275–283. [Google Scholar] [CrossRef]
- Qi, X.; Liu, M.; Chen, Z.; Liang, R. Preparation and properties of diatomite composite superabsorbent. Polym. Adv. Technol. 2007, 18, 184–193. [Google Scholar] [CrossRef]
- Luo, W.; Zhang, W.A.; Chen, P.; Fang, Y.e. Synthesis and properties of starch grafted poly [acrylamide-co-(acrylic acid)]/montmorillonite nanosuperabsorbent via γ-ray irradiation technique. J. Appl. Polym. Sci. 2005, 96, 1341–1346. [Google Scholar] [CrossRef]
- Seki, Y.; Torgürsül, A.; Yurdakoc, K. Preparation and characterization of poly (acrylic acid)-iron rich smectite superabsorbent composites. Polym. Adv. Technol. 2007, 18, 477–482. [Google Scholar] [CrossRef]
- Liu, J.; Wang, Q.; Wang, A. Synthesis and characterization of chitosan-g-poly (acrylic acid)/sodium humate superabsorbent. Carbohydr. Polym. 2007, 70, 166–173. [Google Scholar] [CrossRef]
- Tao, W.; Xiaoqing, W.; Yi, Y.; Wenqiong, H. Preparation of bentonite–poly [(acrylic acid)-acrylamide] water superabsorbent by photopolymerization. Polym. Int. 2006, 55, 1413–1419. [Google Scholar] [CrossRef]
- Pourjavadi, A.; Ayyari, M.; Amini-Fazl, M. Taguchi optimized synthesis of collagen-g-poly (acrylic acid)/kaolin composite superabsorbent hydrogel. Eur. Polym. J. 2008, 44, 1209–1216. [Google Scholar] [CrossRef]
- Hua, S.; Wang, A. Synthesis, characterization and swelling behaviors of sodium alginate-g-poly (acrylic acid)/sodium humate superabsorbent. Carbohydr. Polym. 2009, 75, 79–84. [Google Scholar] [CrossRef]
- Ganguly, S.; Kanovsky, N.; Das, P.; Gedanken, A.; Margel, S. Photopolymerized Thin Coating of Polypyrrole/Graphene Nanofiber/Iron Oxide onto Nonpolar Plastic for Flexible Electromagnetic Radiation Shielding, Strain Sensing, and Non-Contact Heating Applications. Adv. Mater. Interfaces 2021, 8, 2101255. [Google Scholar] [CrossRef]
- Das, P.; Ganguly, S.; Saha, A.; Noked, M.; Margel, S.; Gedanken, A. Carbon-dots-initiated photopolymerization: An in situ synthetic approach for MXene/poly (norepinephrine)/copper hybrid and its application for mitigating water pollution. ACS Appl. Mater. Interfaces 2021, 13, 31038–31050. [Google Scholar] [CrossRef]
- Ganguly, S.; Margel, S. Design of Magnetic Hydrogels for Hyperthermia and Drug Delivery. Polymers 2021, 13, 4259. [Google Scholar] [CrossRef]
- Adhikari, B.; Biswas, A.; Banerjee, A. Graphene oxide-based hydrogels to make metal nanoparticle-containing reduced graphene oxide-based functional hybrid hydrogels. ACS Appl. Mater. Interfaces 2012, 4, 5472–5482. [Google Scholar] [CrossRef]
- Zhang, X.; Sui, Z.; Xu, B.; Yue, S.; Luo, Y.; Zhan, W.; Liu, B. Mechanically strong and highly conductive graphene aerogel and its use as electrodes for electrochemical power sources. J. Mater. Chem. 2011, 21, 6494–6497. [Google Scholar] [CrossRef]
- Huang, H.; Lü, S.; Zhang, X.; Shao, Z. Glucono-δ-lactone controlled assembly of graphene oxide hydrogels with selectively reversible gel–sol transition. Soft Matter 2012, 8, 4609–4615. [Google Scholar] [CrossRef]
- Zu, S.-Z.; Han, B.-H. Aqueous dispersion of graphene sheets stabilized by pluronic copolymers: Formation of supramolecular hydrogel. J. Phys. Chem. C 2009, 113, 13651–13657. [Google Scholar] [CrossRef]
- Sahu, A.; Choi, W.I.; Tae, G. A stimuli-sensitive injectable graphene oxide composite hydrogel. Chem. Commun. 2012, 48, 5820–5822. [Google Scholar] [CrossRef] [PubMed]
- Bardajee, G.R.; Hooshyar, Z. Drug release study by a novel thermo sensitive nanogel based on salep modified graphene oxide. J. Polym. Res. 2017, 24, 1–8. [Google Scholar] [CrossRef]
- Zhang, N.; Li, R.; Zhang, L.; Chen, H.; Wang, W.; Liu, Y.; Wu, T.; Wang, X.; Wang, W.; Li, Y. Actuator materials based on graphene oxide/polyacrylamide composite hydrogels prepared by in situ polymerization. Soft Matter 2011, 7, 7231–7239. [Google Scholar] [CrossRef]
- Si, H.; Luo, H.; Xiong, G.; Yang, Z.; Raman, S.R.; Guo, R.; Wan, Y. One-step in situ biosynthesis of graphene oxide–bacterial cellulose nanocomposite hydrogels. Macromol. Rapid Commun. 2014, 35, 1706–1711. [Google Scholar] [CrossRef]
- Yu, Y.; Shu, Y.; Ye, L. In situ crosslinking of poly (vinyl alcohol)/graphene oxide-glutamic acid nano-composite hydrogel as microbial carrier: Intercalation structure and its wastewater treatment performance. Chem. Eng. J. 2018, 336, 306–314. [Google Scholar] [CrossRef]
- Chen, P.; Wang, X.; Wang, G.; Duo, Y.; Zhang, X.; Hu, X.; Zhang, X. Double network self-healing graphene hydrogel by two step method for anticancer drug delivery. Mater. Technol. 2014, 29, 210–213. [Google Scholar] [CrossRef]
- Zhang, H.; Zhai, D.; He, Y. Graphene oxide/polyacrylamide/carboxymethyl cellulose sodium nanocomposite hydrogel with enhanced mechanical strength: Preparation, characterization and the swelling behavior. RSC Adv. 2014, 4, 44600–44609. [Google Scholar] [CrossRef]
- Ganguly, S.; Mondal, S.; Das, P.; Bhawal, P.; kanti Das, T.; Bose, M.; Choudhary, S.; Gangopadhyay, S.; Das, A.K.; Das, N.C. Natural saponin stabilized nano-catalyst as efficient dye-degradation catalyst. Nano-Struct. Nano-Objects 2018, 16, 86–95. [Google Scholar] [CrossRef]
- Ganguly, S.; Das, P.; Maity, P.P.; Mondal, S.; Ghosh, S.; Dhara, S.; Das, N.C. Green reduced graphene oxide toughened semi-IPN monolith hydrogel as dual responsive drug release system: Rheological, physicomechanical, and electrical evaluations. J. Phys. Chem. B 2018, 122, 7201–7218. [Google Scholar] [CrossRef]
- Das, P.; Ganguly, S.; Margel, S.; Gedanken, A. Immobilization of Heteroatom-Doped Carbon Dots onto Nonpolar Plastics for Antifogging, Antioxidant, and Food Monitoring Applications. Langmuir 2021, 37, 3508–3520. [Google Scholar] [CrossRef]
- Tang, Z.; Gao, L.; Wu, Y.; Su, T.; Wu, Q.; Liu, X.; Li, W.; Wang, Q. BSA–rGO nanocomposite hydrogel formed by UV polymerization and in situ reduction applied as biosensor electrode. J. Mater. Chem. B 2013, 1, 5393–5397. [Google Scholar] [CrossRef]
- Jiao, T.; Guo, H.; Zhang, Q.; Peng, Q.; Tang, Y.; Yan, X.; Li, B. Reduced graphene oxide-based silver nanoparticle-containing composite hydrogel as highly efficient dye catalysts for wastewater treatment. Sci. Rep. 2015, 5, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raafat, A.I.; Ali, A.E.-H. pH-controlled drug release of radiation synthesized graphene oxide/(acrylic acid-co-sodium alginate) interpenetrating network. Polym. Bull. 2017, 74, 2045–2062. [Google Scholar] [CrossRef]
- Dai, Z.; Lu, Q.; Quan, Q.; Mo, R.; Zhou, C.; Hong, P.; Li, C. Novel low temperature (<37 °C) chitosan hydrogel fabrication under the synergistic effect of graphene oxide. New J. Chem. 2017, 41, 671–676. [Google Scholar]
- Lee, Y.; Bae, J.W.; Thi, T.T.H.; Park, K.M.; Park, K.D. Injectable and mechanically robust 4-arm PPO–PEO/graphene oxide composite hydrogels for biomedical applications. Chem. Commun. 2015, 51, 8876–8879. [Google Scholar] [CrossRef]
- Yang, H.; Bremner, D.H.; Tao, L.; Li, H.; Hu, J.; Zhu, L. Carboxymethyl chitosan-mediated synthesis of hyaluronic acid-targeted graphene oxide for cancer drug delivery. Carbohydr. Polym. 2016, 135, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Wang, S.; Li, Y.; Wang, M.; Shi, P.; Huang, X. Covalent functionalization of graphene oxide with biocompatible poly (ethylene glycol) for delivery of paclitaxel. ACS Appl. Mater. Interfaces 2014, 6, 17268–17276. [Google Scholar] [CrossRef]
- Wu, T.; Zhang, B.; Liang, Y.; Liu, T.; Bu, J.; Lin, L.; Wu, Z.; Liu, H.; Wen, S.; Tan, S. Heparin-modified graphene oxide loading anti-cancer drug and growth factor with heat stability, long-term release property and lower cytotoxicity. RSC Adv. 2015, 5, 84334–84342. [Google Scholar] [CrossRef]
- Liu, R.; Liang, S.; Tang, X.-Z.; Yan, D.; Li, X.; Yu, Z.-Z. Tough and highly stretchable graphene oxide/polyacrylamide nanocomposite hydrogels. J. Mater. Chem. 2012, 22, 14160–14167. [Google Scholar] [CrossRef]
- Zhong, M.; Liu, Y.-T.; Xie, X.-M. Self-healable, super tough graphene oxide–poly (acrylic acid) nanocomposite hydrogels facilitated by dual cross-linking effects through dynamic ionic interactions. J. Mater. Chem. B 2015, 3, 4001–4008. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Liu, Z.; Chen, C.; Shi, K.; Zhang, L.; Ju, X.-J.; Wang, W.; Xie, R.; Chu, L.-Y. Reduced graphene oxide-containing smart hydrogels with excellent electro-response and mechanical properties for soft actuators. ACS Appl. Mater. Interfaces 2017, 9, 15758–15767. [Google Scholar] [CrossRef]
- Alam, A.; Meng, Q.; Shi, G.; Arabi, S.; Ma, J.; Zhao, N.; Kuan, H.-C. Electrically conductive, mechanically robust, pH-sensitive graphene/polymer composite hydrogels. Compos. Sci. Technol. 2016, 127, 119–126. [Google Scholar] [CrossRef]
- Alam, A.; Kuan, H.-C.; Zhao, Z.; Xu, J.; Ma, J. Novel polyacrylamide hydrogels by highly conductive, water-processable graphene. Compos. Part A Appl. Sci. Manuf. 2017, 93, 1–9. [Google Scholar] [CrossRef]
- Das, S.; Irin, F.; Ma, L.; Bhattacharia, S.K.; Hedden, R.C.; Green, M.J. Rheology and morphology of pristine graphene/polyacrylamide gels. ACS Appl. Mater. Interfaces 2013, 5, 8633–8640. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Yan, B.; Li, T.; Long, Y.; Li, N.; Ye, M. Mechanical, thermal and swelling properties of poly (acrylic acid)–graphene oxide composite hydrogels. Soft Matter 2012, 8, 1831–1836. [Google Scholar] [CrossRef]
- Abdollahi, R.; Taghizadeh, M.T.; Savani, S. Thermal and mechanical properties of graphene oxide nanocomposite hydrogel based on poly (acrylic acid) grafted onto amylose. Polym. Degrad. Stab. 2018, 147, 151–158. [Google Scholar] [CrossRef]
- Ganguly, S.; Ray, D.; Das, P.; Maity, P.P.; Mondal, S.; Aswal, V.; Dhara, S.; Das, N.C. Mechanically robust dual responsive water dispersible-graphene based conductive elastomeric hydrogel for tunable pulsatile drug release. Ultrason. Sonochem. 2018, 42, 212–227. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.; Lu, J.; Qiu, B.; Shen, B.; Xing, M.; Zhang, J. Developing stretchable and graphene-oxide-based hydrogel for the removal of organic pollutants and metal ions. Appl. Catal. B Environ. 2018, 222, 146–156. [Google Scholar] [CrossRef]
- Meng, Y.; Ye, L.; Coates, P.; Twigg, P. In situ cross-linking of poly (vinyl alcohol)/graphene oxide–polyethylene glycol nanocomposite hydrogels as artificial cartilage replacement: Intercalation structure, unconfined compressive behavior, and biotribological behaviors. J. Phys. Chem. C 2018, 122, 3157–3167. [Google Scholar] [CrossRef] [Green Version]
- Faghihi, S.; Karimi, A.; Jamadi, M.; Imani, R.; Salarian, R. Graphene oxide/poly (acrylic acid)/gelatin nanocomposite hydrogel: Experimental and numerical validation of hyperelastic model. Mater. Sci. Eng. C 2014, 38, 299–305. [Google Scholar] [CrossRef]
- Lee, S.; Lee, H.; Sim, J.H.; Sohn, D. Graphene oxide/poly (acrylic acid) hydrogel by γ-ray pre-irradiation on graphene oxide surface. Macromol. Res. 2014, 22, 165–172. [Google Scholar] [CrossRef]
- Maeda, H. Anion-responsive supramolecular gels. Chem. A Eur. J. 2008, 14, 11274–11282. [Google Scholar] [CrossRef]
- Lloyd, G.O.; Steed, J.W. Anion-tuning of supramolecular gel properties. Nat. Chem. 2009, 1, 437–442. [Google Scholar] [CrossRef] [Green Version]
- Banerjee, S.; Das, R.K.; Maitra, U. Supramolecular gels ‘in action’. J. Mater. Chem. 2009, 19, 6649–6687. [Google Scholar] [CrossRef]
- Steed, J.W. Anion-tuned supramolecular gels: A natural evolution from urea supramolecular chemistry. Chem. Soc. Rev. 2010, 39, 3686–3699. [Google Scholar] [CrossRef]
- Noro, A.; Hayashi, M.; Matsushita, Y. Design and properties of supramolecular polymer gels. Soft Matter 2012, 8, 6416–6429. [Google Scholar] [CrossRef]
- Seiffert, S.; Sprakel, J. Physical chemistry of supramolecular polymer networks. Chem. Soc. Rev. 2012, 41, 909–930. [Google Scholar] [CrossRef] [PubMed]
- Foster, J.A.; Steed, J.W. Exploiting cavities in supramolecular gels. Angew. Chem. Int. Ed. 2010, 49, 6718–6724. [Google Scholar] [CrossRef]
- Steed, J.W. Supramolecular gel chemistry: Developments over the last decade. Chem. Commun. 2011, 47, 1379–1383. [Google Scholar] [CrossRef]
- Appel, E.A.; del Barrio, J.; Loh, X.J.; Scherman, O.A. Supramolecular polymeric hydrogels. Chem. Soc. Rev. 2012, 41, 6195–6214. [Google Scholar] [CrossRef] [PubMed]
- Buerkle, L.E.; Rowan, S.J. Supramolecular gels formed from multi-component low molecular weight species. Chem. Soc. Rev. 2012, 41, 6089–6102. [Google Scholar] [CrossRef] [PubMed]
- Samanta, S.K.; Pal, A.; Bhattacharya, S.; Rao, C. Carbon nanotube reinforced supramolecular gels with electrically conducting, viscoelastic and near-infrared sensitive properties. J. Mater. Chem. 2010, 20, 6881–6890. [Google Scholar] [CrossRef]
- Abdallah, D.J.; Sirchio, S.A.; Weiss, R.G. Hexatriacontane organogels. The first determination of the conformation and molecular packing of a low-molecular-mass organogelator in its gelled state. Langmuir 2000, 16, 7558–7561. [Google Scholar] [CrossRef]
- Echegoyen, L.E.; Portugal, L.; Miller, S.R.; Hernandez, J.C.; Echegoyen, L.; Gokel, G.W. The first evidence for aggregation behavior in a lipophilic [2.2. 2]-cryptand and in 18-membered ring steroidal lariat ethers. Tetrahedron Lett. 1988, 29, 4065–4068. [Google Scholar] [CrossRef]
- Hirst, A.R.; Smith, D.K.; Harrington, J.P. Unique Nanoscale Morphologies Underpinning Organic Gel-Phase Materials. Chem. A Eur. J. 2005, 11, 6552–6559. [Google Scholar] [CrossRef] [PubMed]
- ávan Esch, J.H. Dynamic covalent assembly of stimuli responsive vesicle gels. Chem. Commun. 2012, 48, 9837–9839. [Google Scholar]
- Harada, A.; Kobayashi, R.; Takashima, Y.; Hashidzume, A.; Yamaguchi, H. Macroscopic self-assembly through molecular recognition. Nat. Chem. 2011, 3, 34–37. [Google Scholar] [CrossRef] [PubMed]
- Park, T.; Zimmerman, S.C. Formation of a miscible supramolecular polymer blend through self-assembly mediated by a quadruply hydrogen-bonded heterocomplex. J. Am. Chem. Soc. 2006, 128, 11582–11590. [Google Scholar] [CrossRef]
- Shirakawa, M.; Fujita, N.; Shinkai, S. [60] Fullerene-motivated organogel formation in a porphyrin derivative bearing programmed hydrogen-bonding sites. J. Am. Chem. Soc. 2003, 125, 9902–9903. [Google Scholar] [CrossRef] [PubMed]
- Moffat, J.R.; Seeley, G.J.; Carter, J.T.; Burgess, A.; Smith, D.K. Nanostructured polymers with embedded self-assembled reactive gel networks. Chem. Commun. 2008, 4601–4603. [Google Scholar] [CrossRef] [PubMed]
- Ge, Z.; Hu, J.; Huang, F.; Liu, S. Responsive supramolecular gels constructed by crown ether based molecular recognition. Angew. Chem. 2009, 121, 1830–1834. [Google Scholar] [CrossRef]
- Deng, C.; Fang, R.; Guan, Y.; Jiang, J.; Lin, C.; Wang, L. Sonication-induced self-assembly of flexible tris (ureidobenzyl) amine: From dimeric aggregates to supramolecular gels. Chem. Commun. 2012, 48, 7973–7975. [Google Scholar] [CrossRef]
- Liu, Q.; Wang, Y.; Li, W.; Wu, L. Structural characterization and chemical response of a Ag-coordinated supramolecular gel. Langmuir 2007, 23, 8217–8223. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.-W.; Yang, Y.; Chen, C.-F.; Ma, J.-T. Novel anion-tuning supramolecular gels with dual-channel response: Reversible sol− gel transition and color changes. Langmuir 2010, 26, 9040–9044. [Google Scholar] [CrossRef]
- Das, R.N.; Kumar, Y.P.; Pagoti, S.; Patil, A.J.; Dash, J. Diffusion and birefringence of bioactive dyes in a supramolecular guanosine hydrogel. Chem. A Eur. J. 2012, 18, 6008–6014. [Google Scholar] [CrossRef]
- Huang, F.; Switek, K.A.; Zakharov, L.N.; Fronczek, F.R.; Slebodnick, C.; Lam, M.; Golen, J.A.; Bryant, W.S.; Mason, P.E.; Rheingold, A.L. Bis (m-phenylene)-32-crown-10-based cryptands, powerful hosts for paraquat derivatives. J. Org. Chem. 2005, 70, 3231–3241. [Google Scholar] [CrossRef]
- Yan, X.; Xu, D.; Chi, X.; Chen, J.; Dong, S.; Ding, X.; Yu, Y.; Huang, F. A multiresponsive, shape-persistent, and elastic supramolecular polymer network gel constructed by orthogonal self-assembly. Adv. Mater. 2012, 24, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Xiao, T.; Feng, X.; Ye, S.; Guan, Y.; Li, S.-L.; Wang, Q.; Ji, Y.; Zhu, D.; Hu, X.; Lin, C. Highly controllable ring–chain equilibrium in quadruply hydrogen bonded supramolecular polymers. Macromolecules 2012, 45, 9585–9594. [Google Scholar] [CrossRef]
- Zhang, Z.; Luo, Y.; Chen, J.; Dong, S.; Yu, Y.; Ma, Z.; Huang, F. Formation of Linear Supramolecular Polymers That Is Driven by C—H⋅⋅⋅ π Interactions in Solution and in the Solid State. Angew. Chem. Int. Ed. 2011, 50, 1397–1401. [Google Scholar] [CrossRef]
- Xiao, T.; Li, S.-L.; Zhang, Y.; Lin, C.; Hu, B.; Guan, X.; Yu, Y.; Jiang, J.; Wang, L. Novel self-assembled dynamic [2] catenanes interlocked by the quadruple hydrogen bonding ureidopyrimidinone motif. Chem. Sci. 2012, 3, 1417–1421. [Google Scholar] [CrossRef]
- Rocchigiani, L.; Bellachioma, G.; Ciancaleoni, G.; Crocchianti, S.; Laganà, A.; Zuccaccia, C.; Zuccaccia, D.; Macchioni, A. Anion-Dependent Tendency of Di-Long-Chain Quaternary Ammonium Salts to Form Ion Quadruples and Higher Aggregates in Benzene. ChemPhysChem 2010, 11, 3243–3254. [Google Scholar] [CrossRef]
- Zohourian, M.M.; Kabiri, K. Superabsorbent polymer materials: A review. Iran. Polym. J. 2008, 17, 447–451. [Google Scholar]
- Kabiri, K.; Omidian, H.; Hashemi, S.; Zohuriaan-Mehr, M. Synthesis of fast-swelling superabsorbent hydrogels: Effect of crosslinker type and concentration on porosity and absorption rate. Eur. Polym. J. 2003, 39, 1341–1348. [Google Scholar] [CrossRef]
- Nie, J.; Du, B.; Oppermann, W. Swelling, elasticity, and spatial inhomogeneity of poly (N-isopropylacrylamide)/clay nanocomposite hydrogels. Macromolecules 2005, 38, 5729–5736. [Google Scholar] [CrossRef]
- Ganguly, S.; Das, N.C. Synthesis of Mussel Inspired Polydopamine Coated Halloysite Nanotubes Based Semi-IPN: An Approach to Fine Tuning in Drug Release and Mechanical Toughening. In Proceedings of the Macromolecular Symposia, December 2018; p. 1800076. [Google Scholar]
- Haraguchi, K.; Takehisa, T. Nanocomposite hydrogels: A unique organic–inorganic network structure with extraordinary mechanical, optical, and swelling/de-swelling properties. Adv. Mater. 2002, 14, 1120–1124. [Google Scholar] [CrossRef]
- Lee, W.F.; Chen, Y.C. Effect of bentonite on the physical properties and drug-release behavior of poly (AA-co-PEGMEA)/bentonite nanocomposite hydrogels for mucoadhesive. J. Appl. Polym. Sci. 2004, 91, 2934–2941. [Google Scholar] [CrossRef]
- Lee, W.F.; Chen, Y.C. Effect of hydrotalcite on the physical properties and drug-release behavior of nanocomposite hydrogels based on poly [acrylic acid-co-poly (ethylene glycol) methyl ether acrylate] gels. J. Appl. Polym. Sci. 2004, 94, 692–699. [Google Scholar] [CrossRef]
- Lee, W.F.; Fu, Y.T. Effect of montmorillonite on the swelling behavior and drug-release behavior of nanocomposite hydrogels. J. Appl. Polym. Sci. 2003, 89, 3652–3660. [Google Scholar] [CrossRef]
- Molu, Z.B.; Seki, Y.; Yurdakoc, K. Preparation and characterization of poly (acrylic acid)/pillared clay superabsorbent composite. Polym. Bull. 2010, 64, 171–183. [Google Scholar] [CrossRef]
- Li, A.; Wang, A. Synthesis and properties of clay-based superabsorbent composite. Eur. Polym. J. 2005, 41, 1630–1637. [Google Scholar] [CrossRef]
- Harini, M.; Deshpande, A.P. Rheology of poly (sodium acrylate) hydrogels during cross-linking with and without cellulose microfibrils. J. Rheol. 2009, 53, 31–47. [Google Scholar] [CrossRef]
- Ye, H.; Zhao, J.Q.; Zhang, Y.H. Novel degradable superabsorbent materials of silicate/acrylic-based polymer hybrids. J. Appl. Polym. Sci. 2004, 91, 936–940. [Google Scholar] [CrossRef]
- Doniach, S. Changes in biomolecular conformation seen by small angle X-ray scattering. Chem. Rev. 2001, 101, 1763–1778. [Google Scholar] [CrossRef]
- Das, N.C.; Sokol, P.E. Hybrid photovoltaic devices from regioregular polythiophene and ZnO nanoparticles composites. Renew. Energy 2010, 35, 2683–2688. [Google Scholar] [CrossRef]
- Huang, B.; Hirst, A.R.; Smith, D.K.; Castelletto, V.; Hamley, I.W. A direct comparison of one-and two-component dendritic self-assembled materials: Elucidating molecular recognition pathways. J. Am. Chem. Soc. 2005, 127, 7130–7139. [Google Scholar] [CrossRef] [PubMed]
- Ganguly, S.; Bhawal, P.; Choudhury, A.; Mondal, S.; Das, P.; Das, N.C. Preparation and properties of halloysite nanotubes/poly (ethylene methyl acrylate)-based nanocomposites by variation of mixing methods. Polym. Plast. Technol. Eng. 2018, 57, 997–1014. [Google Scholar] [CrossRef]
- Bhawal, P.; Ganguly, S.; Das, T.K.; Mondal, S.; Das, N. Mechanically robust conductive carbon clusters confined ethylene methyl acrylate–based flexible composites for superior shielding effectiveness. Polym. Adv. Technol. 2018, 29, 95–110. [Google Scholar] [CrossRef]
- Janicki, J. SAXS and WAXD real time studies on nanostructure of selected polymer materials. J. Alloy. Compd. 2004, 382, 61–67. [Google Scholar] [CrossRef]
- Ganguly, S.; Mondal, S.; Das, P.; Bhawal, P.; Das, T.K.; Ghosh, S.; Remanan, S.; Das, N.C. An insight into the physico-mechanical signatures of silylated graphene oxide in poly (ethylene methyl acrylate) copolymeric thermoplastic matrix. Macromol. Res. 2019, 27, 268–281. [Google Scholar] [CrossRef]
- Takeno, H.; Kimura, Y.; Nakamura, W. Mechanical, swelling, and structural properties of mechanically tough clay-sodium polyacrylate blend hydrogels. Gels 2017, 3, 10. [Google Scholar] [CrossRef]
- Ganguly, S.; Maity, P.P.; Mondal, S.; Das, P.; Bhawal, P.; Dhara, S.; Das, N.C. Polysaccharide and poly (methacrylic acid) based biodegradable elastomeric biocompatible semi-IPN hydrogel for controlled drug delivery. Mater. Sci. Eng. C 2018, 92, 34–51. [Google Scholar] [CrossRef]
- Ganguly, S.; Das, N.C. A comparison on self-seeding and isothermal crystallization of polyethylene in solution using small angle neutron scattering. Polymer 2015, 61, 192–197. [Google Scholar] [CrossRef]
- Das, P.; Ganguly, S.; Bose, M.; Ray, D.; Ghosh, S.; Mondal, S.; Aswal, V.K.; Das, A.K.; Banerjee, S.; Das, N.C. Surface quaternized nanosensor as a one-arrow-two-hawks approach for fluorescence turn “on–off–on” bifunctional sensing and antibacterial activity. New J. Chem. 2019, 43, 6205–6219. [Google Scholar] [CrossRef]
- Millon, L.E.; Nieh, M.-P.; Hutter, J.L.; Wan, W. SANS characterization of an anisotropic poly (vinyl alcohol) hydrogel with vascular applications. Macromolecules 2007, 40, 3655–3662. [Google Scholar] [CrossRef] [Green Version]
- Demirdöven, N.; Cheatum, C.M.; Chung, H.S.; Khalil, M.; Knoester, J.; Tokmakoff, A. Two-dimensional infrared spectroscopy of antiparallel β-sheet secondary structure. J. Am. Chem. Soc. 2004, 126, 7981–7990. [Google Scholar] [CrossRef] [Green Version]
- Vivian, J.T.; Callis, P.R. Mechanisms of tryptophan fluorescence shifts in proteins. Biophys. J. 2001, 80, 2093–2109. [Google Scholar] [CrossRef] [Green Version]
- Das, P.; Ganguly, S.; Agarwal, T.; Maity, P.; Ghosh, S.; Choudhary, S.; Gangopadhyay, S.; Maiti, T.K.; Dhara, S.; Banerjee, S. Heteroatom doped blue luminescent carbon dots as a nano-probe for targeted cell labeling and anticancer drug delivery vehicle. Mater. Chem. Phys. 2019, 237, 121860. [Google Scholar] [CrossRef]
- Kim, H.J.; Lee, J.H.; Lee, M. Stimuli-responsive gels from reversible coordination polymers. Angew. Chem. 2005, 117, 5960–5964. [Google Scholar] [CrossRef]
- Das, P.; Ganguly, S.; Mondal, S.; Ghorai, U.K.; Maity, P.P.; Choudhary, S.; Gangopadhyay, S.; Dhara, S.; Banerjee, S.; Das, N.C. Dual doped biocompatible multicolor luminescent carbon dots for bio labeling, UV-active marker and fluorescent polymer composite. Luminescence 2018, 33, 1136–1145. [Google Scholar] [CrossRef]
- Rao, K.V.; Datta, K.; Eswaramoorthy, M.; George, S.J. Light-Harvesting Hybrid Hydrogels: Energy-Transfer-Induced Amplified Fluorescence in Noncovalently Assembled Chromophore–Organoclay Composites. Angew. Chem. Int. Ed. 2011, 50, 1179–1184. [Google Scholar] [CrossRef] [PubMed]
- De Jong, K.P.; Geus, J.W. Carbon nanofibers: Catalytic synthesis and applications. Catal. Rev. 2000, 42, 481–510. [Google Scholar] [CrossRef]
- Greiner, N.R.; Phillips, D.; Johnson, J.; Volk, F. Diamonds in detonation soot. Nature 1988, 333, 440–442. [Google Scholar] [CrossRef]
- Kroto, H.W.; Heath, J.R.; O’Brien, S.C.; Curl, R.F.; Smalley, R.E. C 60: Buckminsterfullerene. Nature 1985, 318, 162–163. [Google Scholar] [CrossRef]
- Ganguly, S.; Das, P.; Bose, M.; Das, T.K.; Mondal, S.; Das, A.K.; Das, N.C. Sonochemical green reduction to prepare Ag nanoparticles decorated graphene sheets for catalytic performance and antibacterial application. Ultrason. Sonochem. 2017, 39, 577–588. [Google Scholar] [CrossRef]
- Das, T.K.; Bhawal, P.; Ganguly, S.; Mondal, S.; Das, N.C. A facile green synthesis of amino acid boosted Ag decorated reduced graphene oxide nanocomposites and its catalytic activity towards 4-nitrophenol reduction. Surf. Interfaces 2018, 13, 79–91. [Google Scholar] [CrossRef]
- Mora, A.; Verma, P.; Kumar, S. Electrical conductivity of CNT/polymer composites: 3D printing, measurements and modeling. Compos. Part B Eng. 2020, 183, 107600. [Google Scholar] [CrossRef]
- Bondon, N.; Raehm, L.; Charnay, C.; Boukherroub, R.; Durand, J.-O. Nanodiamonds for bioapplications, recent developments. J. Mater. Chem. B 2020, 8, 10878–10896. [Google Scholar] [CrossRef] [PubMed]
- Jariwala, D.H.; Patel, D.; Wairkar, S. Surface functionalization of nanodiamonds for biomedical applications. Mater. Sci. Eng. C 2020, 113, 110996. [Google Scholar] [CrossRef]
- Uthappa, U.; Arvind, O.; Sriram, G.; Losic, D.; Kigga, M.; Kurkuri, M.D. Nanodiamonds and their surface modification strategies for drug delivery applications. J. Drug Deliv. Sci. Technol. 2020, 101993. [Google Scholar] [CrossRef]
- Kim, H.; Lee, S. Characterization of carbon nanofiber (CNF)/polymer composite coated on cotton fabrics prepared with various circuit patterns. Fash. Text. 2018, 5, 1–13. [Google Scholar] [CrossRef]
- Saravanan, A.; Maruthapandi, M.; Das, P.; Ganguly, S.; Margel, S.; Luong, J.H.; Gedanken, A. Applications of N-doped carbon dots as antimicrobial agents, antibiotic carriers, and selective fluorescent probes for nitro explosives. ACS Appl. Bio Mater. 2020, 3, 8023–8031. [Google Scholar] [CrossRef]
- Das, P.; Maruthapandi, M.; Saravanan, A.; Natan, M.; Jacobi, G.; Banin, E.; Gedanken, A. Carbon dots for heavy-metal sensing, pH-sensitive cargo delivery, and antibacterial applications. ACS Appl. Nano Mater. 2020, 3, 11777–11790. [Google Scholar] [CrossRef]
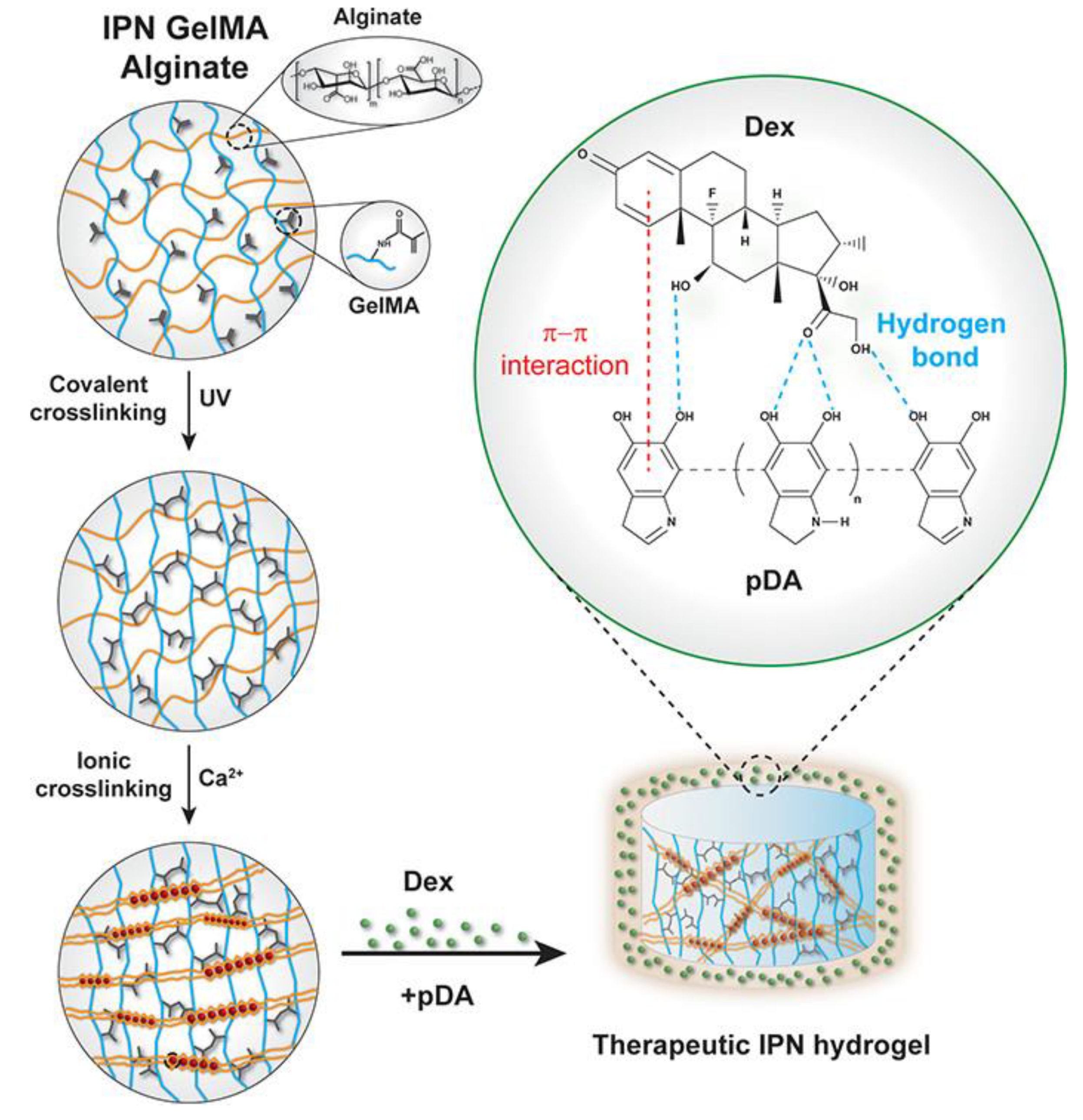

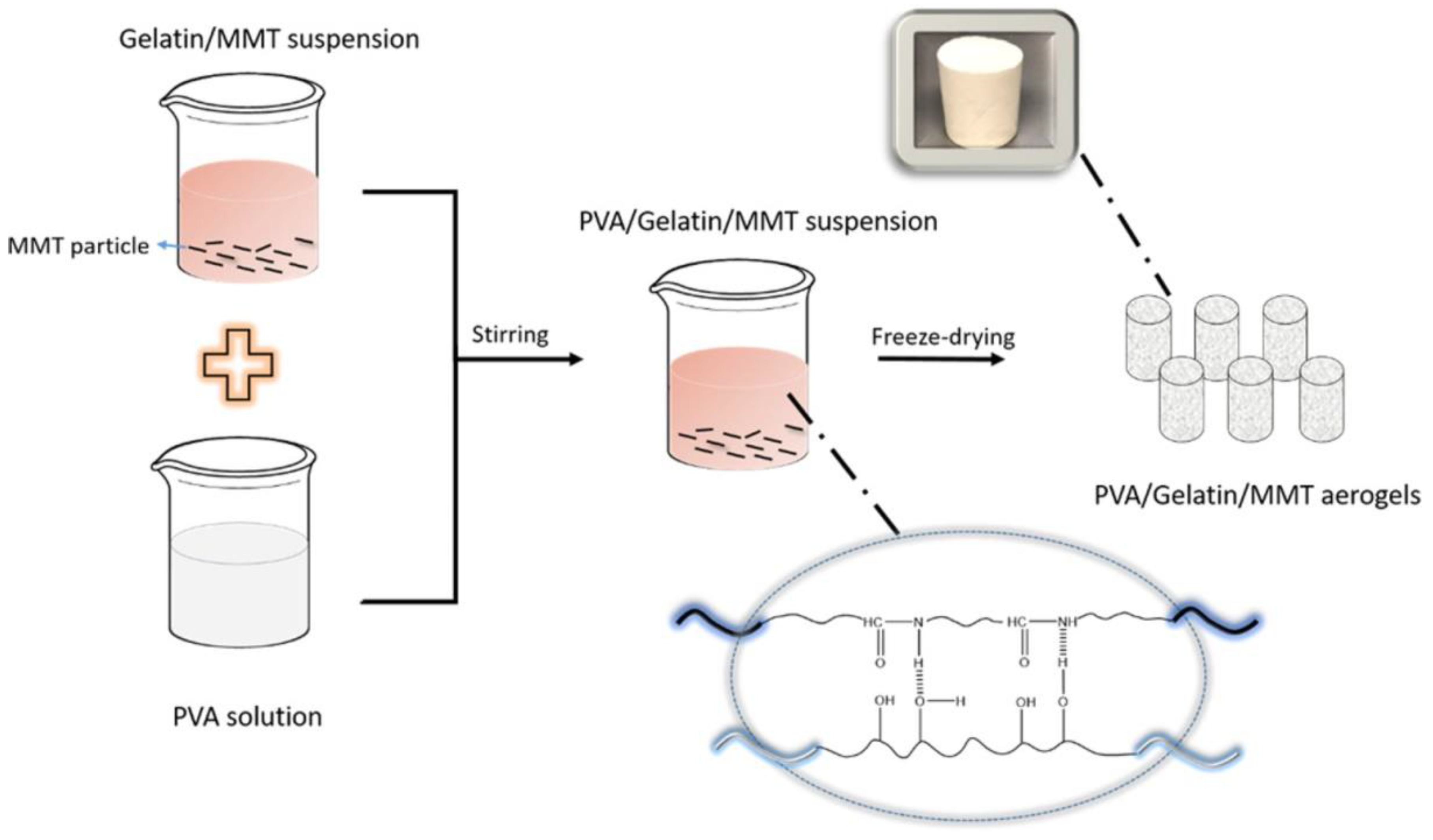

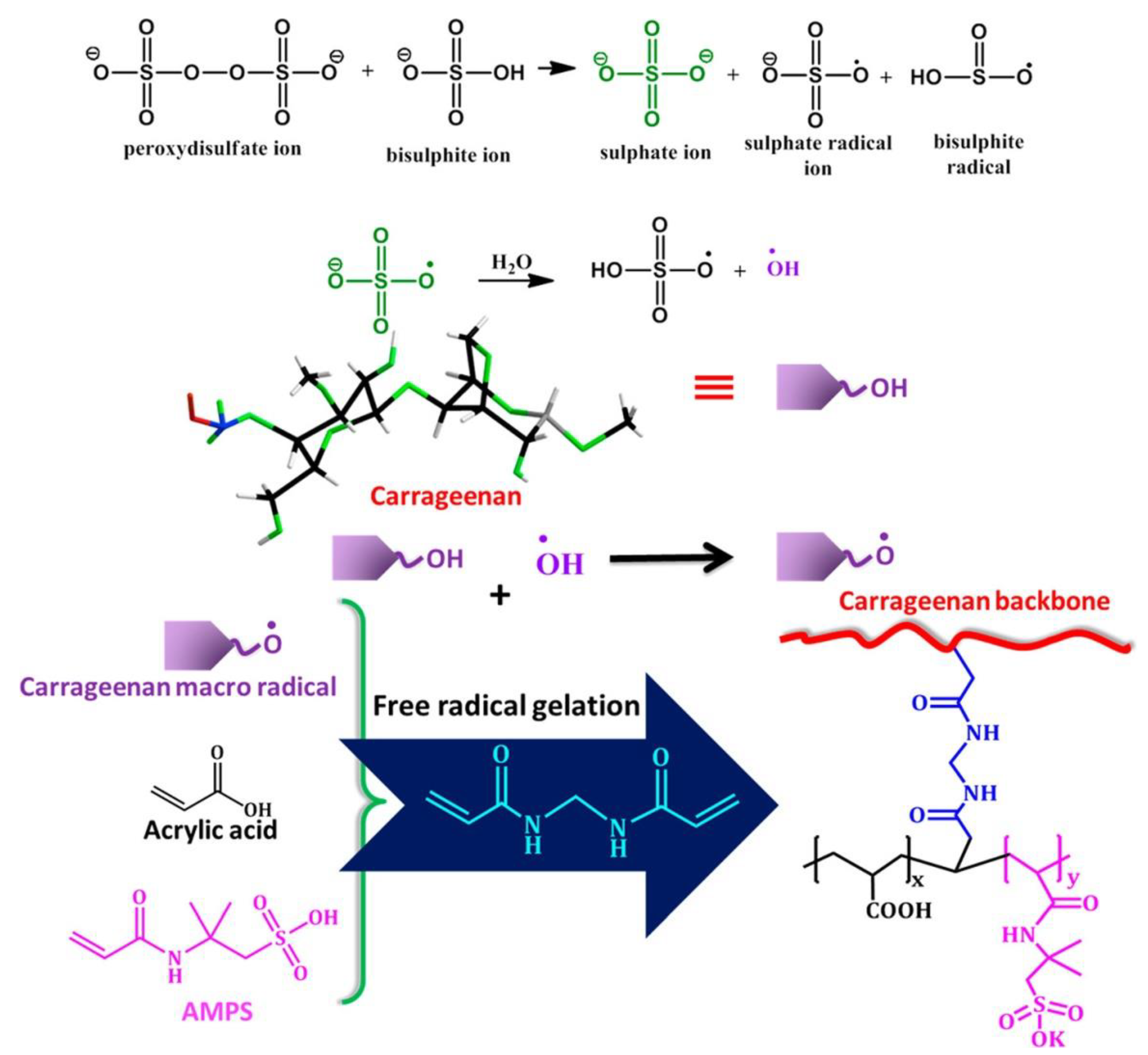
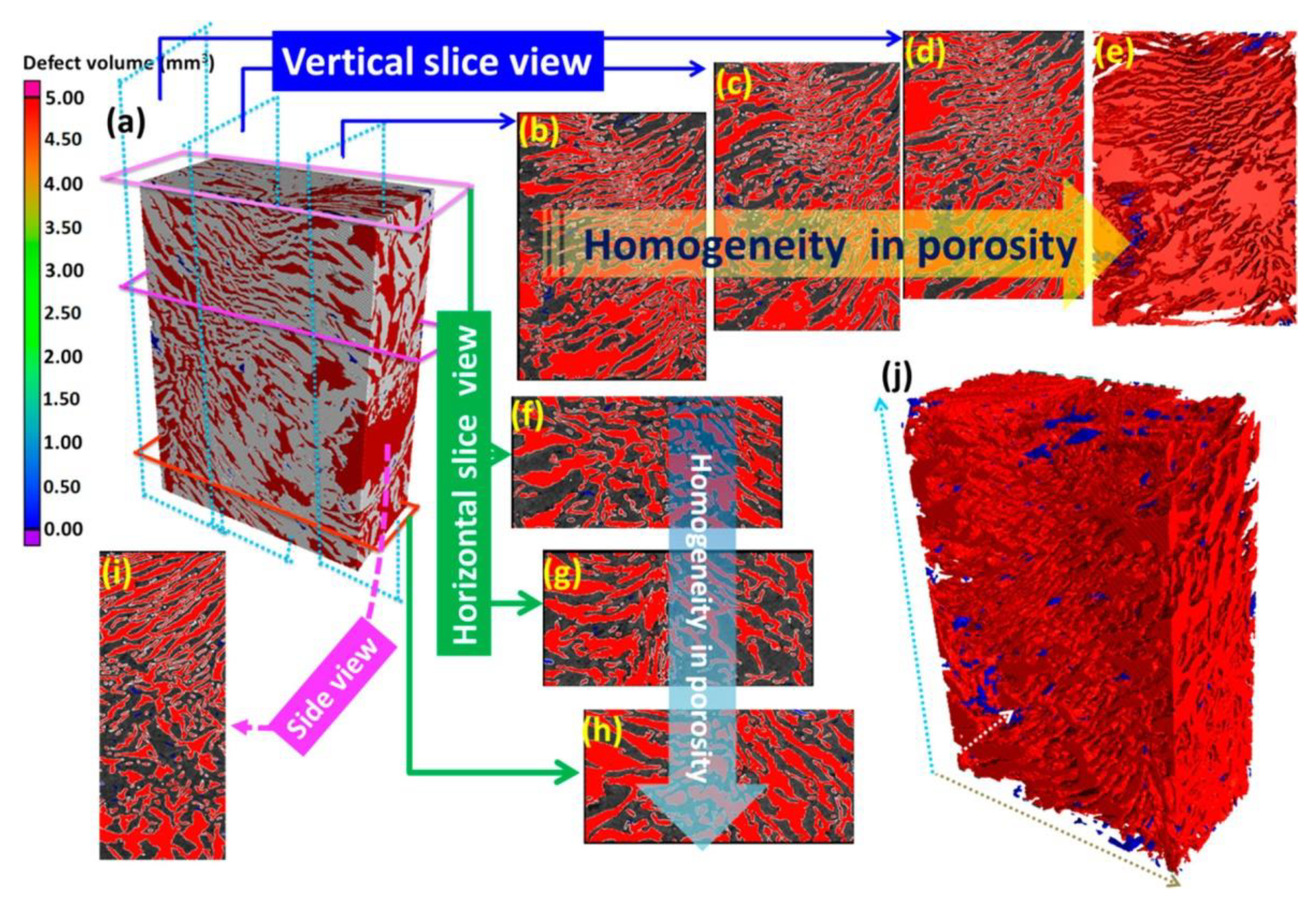
| Monomer | Polymerization Method | Clay Filler | Crosslinker | Ref. |
|---|---|---|---|---|
| AAm/AMPS | Solution | MMT | MBA | [59] |
| AA | Solution | Bentonite | Sugar | [60] |
| Chitosan-g-AA | Solution | MMT | MBA | [61] |
| AAm-AMPS | Solution | MMT | MBA | [62] |
| PS-g-AA | Solution | MMT | MBA | [63] |
| AA | Solution | MMT | MBA | [64] |
| AA-NVP | Solution | MMT | MBA | [65] |
| AA-AAm | Solution | Sepiolite | MBA | [66] |
| Starch-g-AA | Solution | MMT | MBA | [67] |
| AA-DAC | Solution | MMT | MBA | [68] |
| CMC-g-AA | Solution | MMT | MBA | [69] |
| Chitosan-g-AA | Solution | MMT | MBA | [70] |
| AA | Solution | Sepiolite | MBA | [71] |
| AA | Inverse suspension | MMT | MBA | [72] |
| AA | Solution | MMT | MBA | [73] |
| AA | Solution | OMMT | MBA | [74] |
| MAPDMSP | Solution | OMMT | PEGDMA | [75] |
| AA | Solution | O-ATA | MBA | [76] |
| HES-HEMA | Solution | OMMT | Clay | [58] |
| AAm | Solution | ATA | MBA | [77] |
| AA-AAm | Solution | SH-OMMT | MBA | [78] |
| AA | Inverse suspension | OMMT | MBA | [79] |
| AA | Solution | Hydrotalcite | MBA | [80] |
| SA | Solution | AR-O-R | MBA | [81] |
| SA | Solution | O-vermiculite | MBA | [82] |
| SA | solution | Rectorite | MBA | [83] |
| AA-AAm | Inverse suspension | Hydrotalcite | MBA | [77] |
| Starch-g-AA | Solution | Bentonite | MBA | [84] |
| AAm-AMPS | Solution | MMT | Chromium triacetate | [77] |
| AAm | Solution | Laponite | Clay | [85] |
| AA | Solution | Diatomite | MBA | [86] |
| Starch-g-AA-AAm | Solation | MMT | MBA | [87] |
| AA | Solution | Smectite | MBA | [88] |
| NSC-g-AA | Inverse suspension | ATA | MBA | [89] |
| AA-AAm | Solution | Kaolinite | MBA | [90] |
| Collagen-g-AA | Solution | Kaolin | MBA | [91] |
| Sodium alginate-g-AA | Solution | SH | MBA | [92] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ganguly, S.; Margel, S. A Review on Synthesis Methods of Phyllosilicate- and Graphene-Filled Composite Hydrogels. J. Compos. Sci. 2022, 6, 15. https://doi.org/10.3390/jcs6010015
Ganguly S, Margel S. A Review on Synthesis Methods of Phyllosilicate- and Graphene-Filled Composite Hydrogels. Journal of Composites Science. 2022; 6(1):15. https://doi.org/10.3390/jcs6010015
Chicago/Turabian StyleGanguly, Sayan, and Shlomo Margel. 2022. "A Review on Synthesis Methods of Phyllosilicate- and Graphene-Filled Composite Hydrogels" Journal of Composites Science 6, no. 1: 15. https://doi.org/10.3390/jcs6010015
APA StyleGanguly, S., & Margel, S. (2022). A Review on Synthesis Methods of Phyllosilicate- and Graphene-Filled Composite Hydrogels. Journal of Composites Science, 6(1), 15. https://doi.org/10.3390/jcs6010015







