Anticorrosion Properties of Epoxy Composite Coating Reinforced by Molybdate-Intercalated Functionalized Layered Double Hydroxide
Abstract
1. Introduction
2. Experimental
2.1. Materials
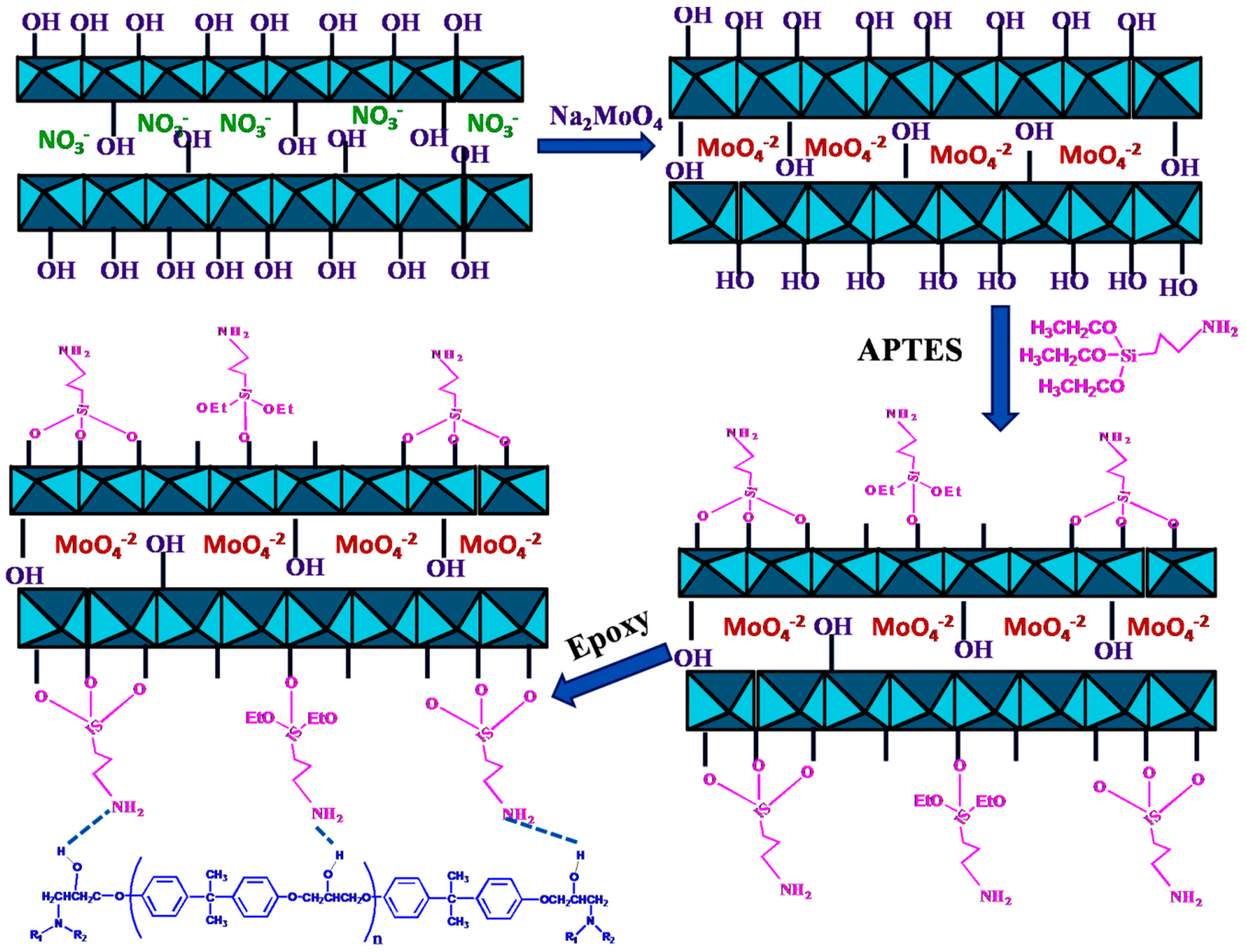
2.2. Preparation of MgAl–NO3−–LDH and MoO42−-Intercalated MgAl–NO3−–LDH
2.3. Preparation of APTES-Surface-Modified Mo–LDH
2.4. Preparation of APTES–Mo–LDH/Epoxy Coatings
2.5. Characterization
3. Results and Discussion
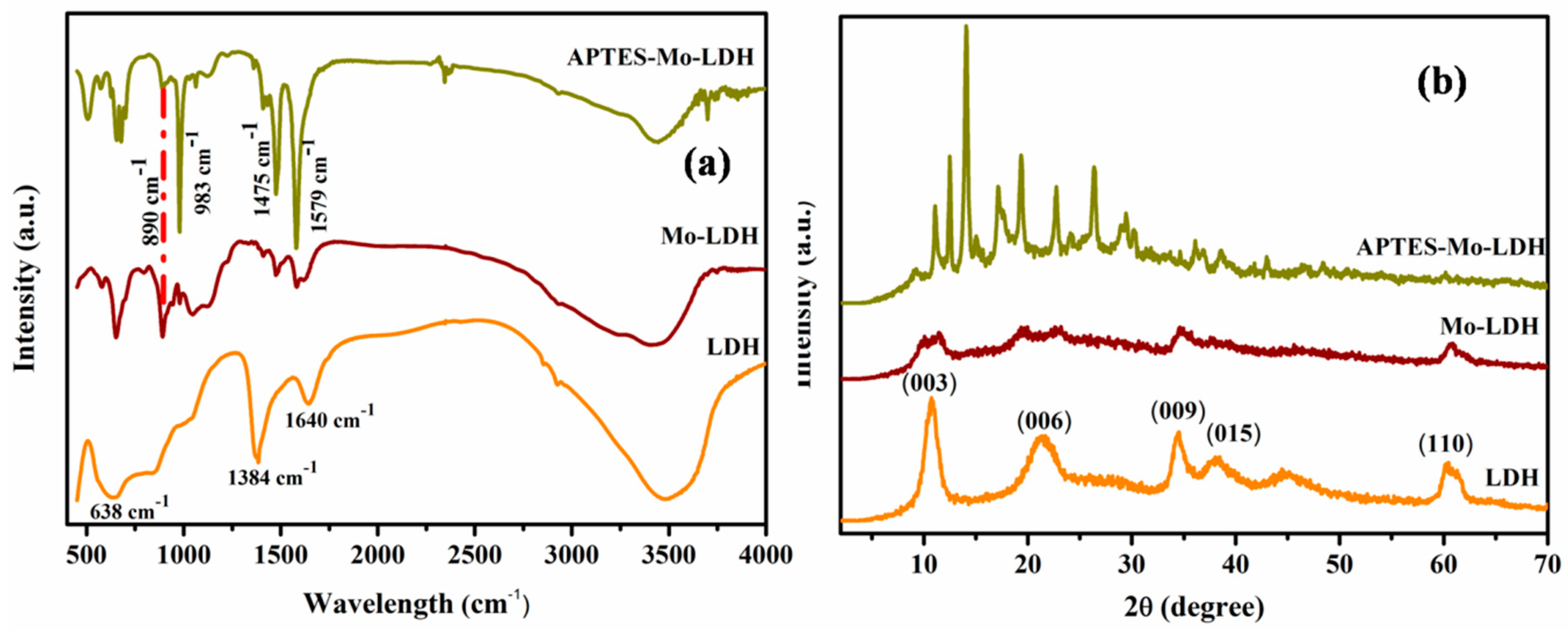
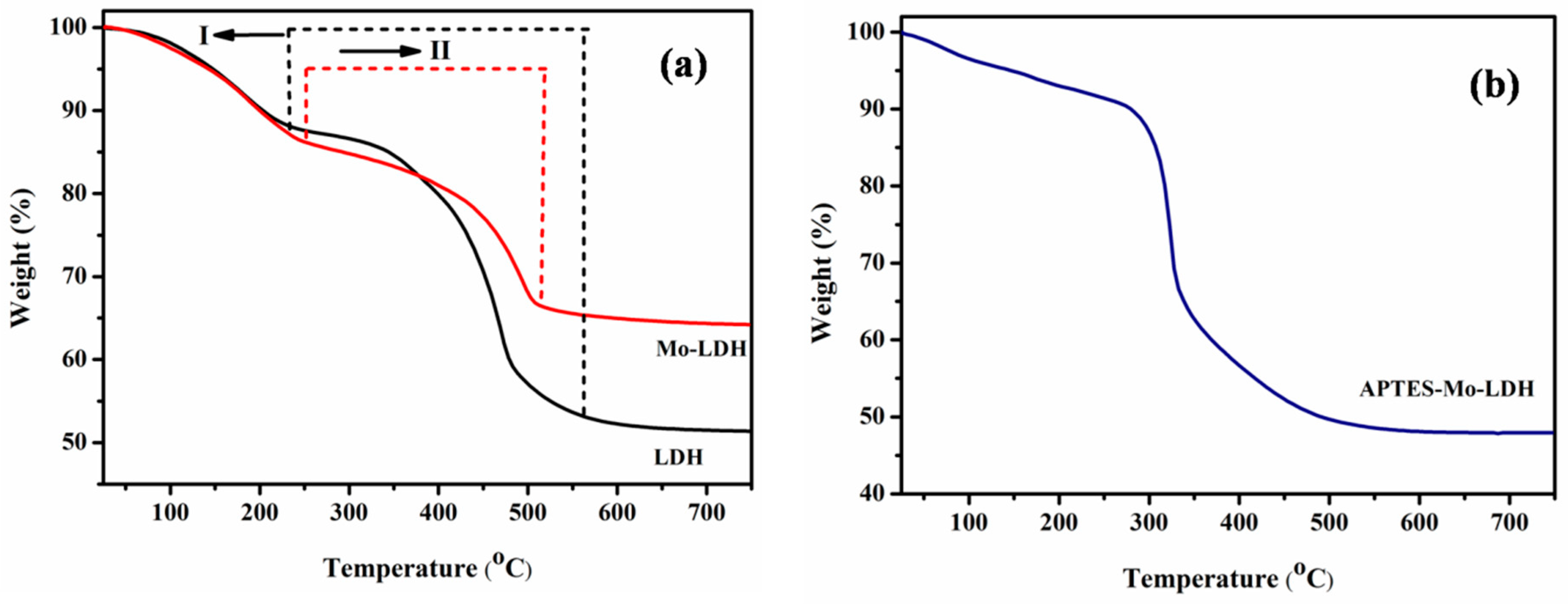
3.1. Characterization of LDH, Mo–LDH, and APTES–Mo–LDH
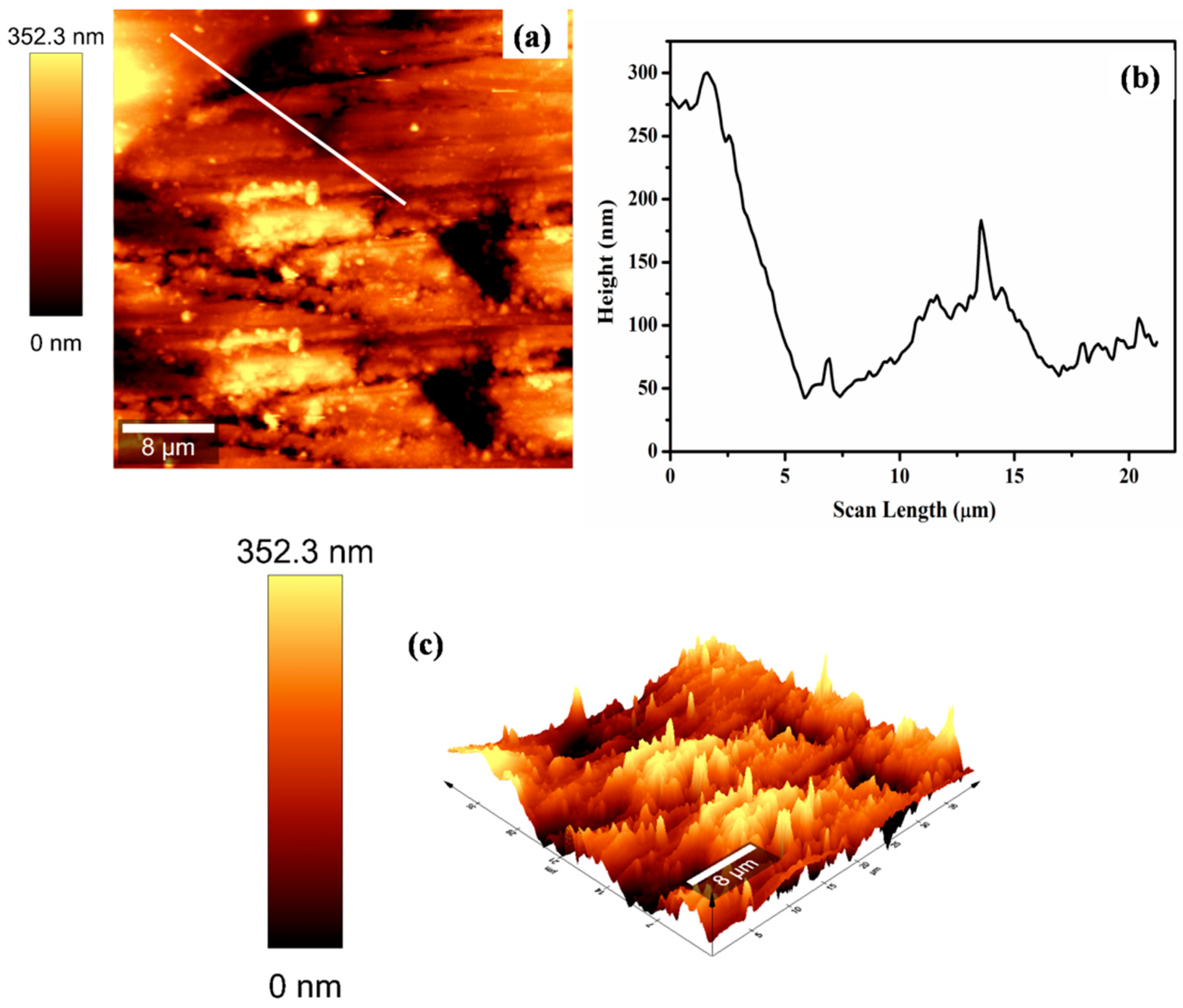
3.2. FE-SEM Analysis of Coatings
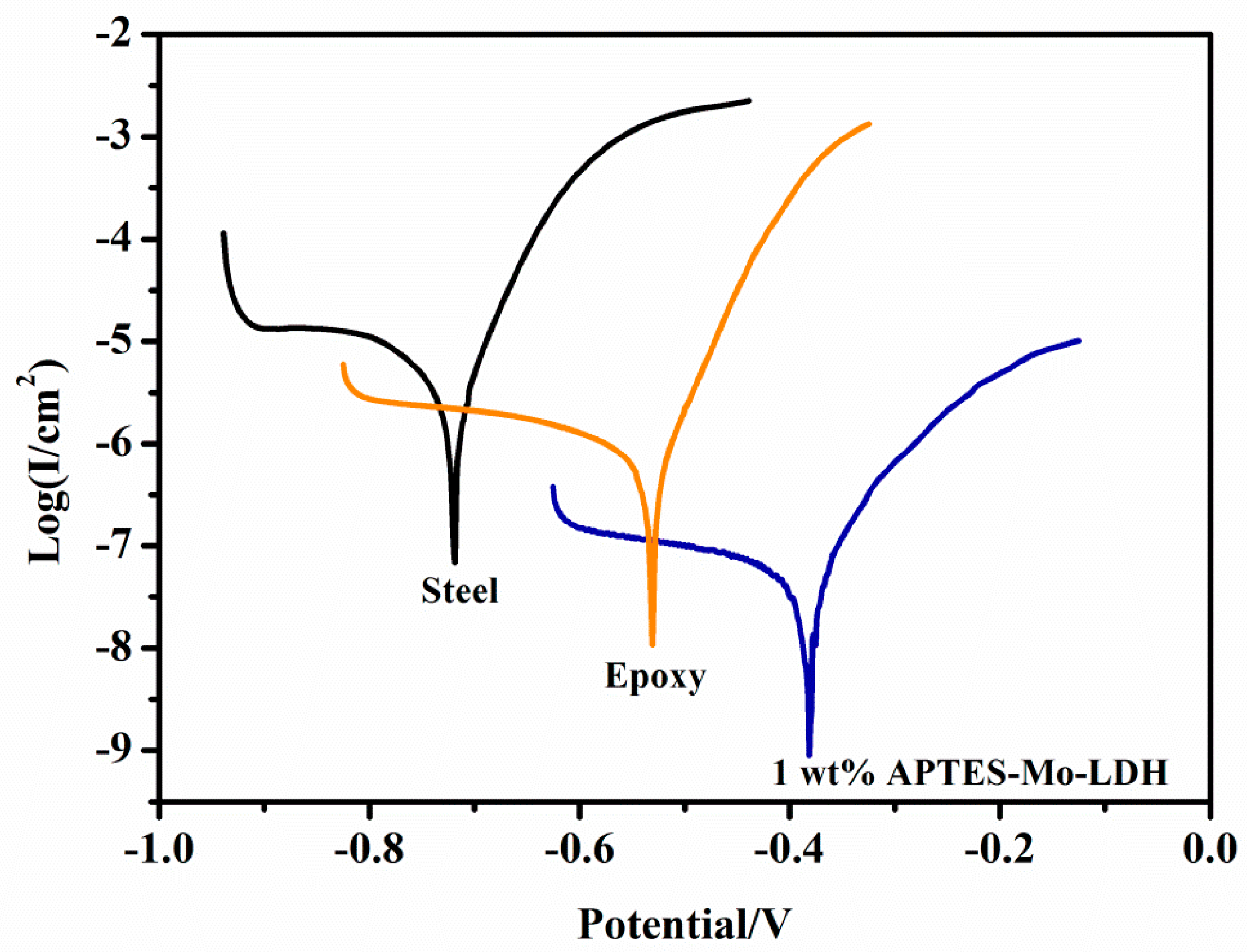
3.3. Impedance Measurements and Potentiodynamic Polarization Analysis
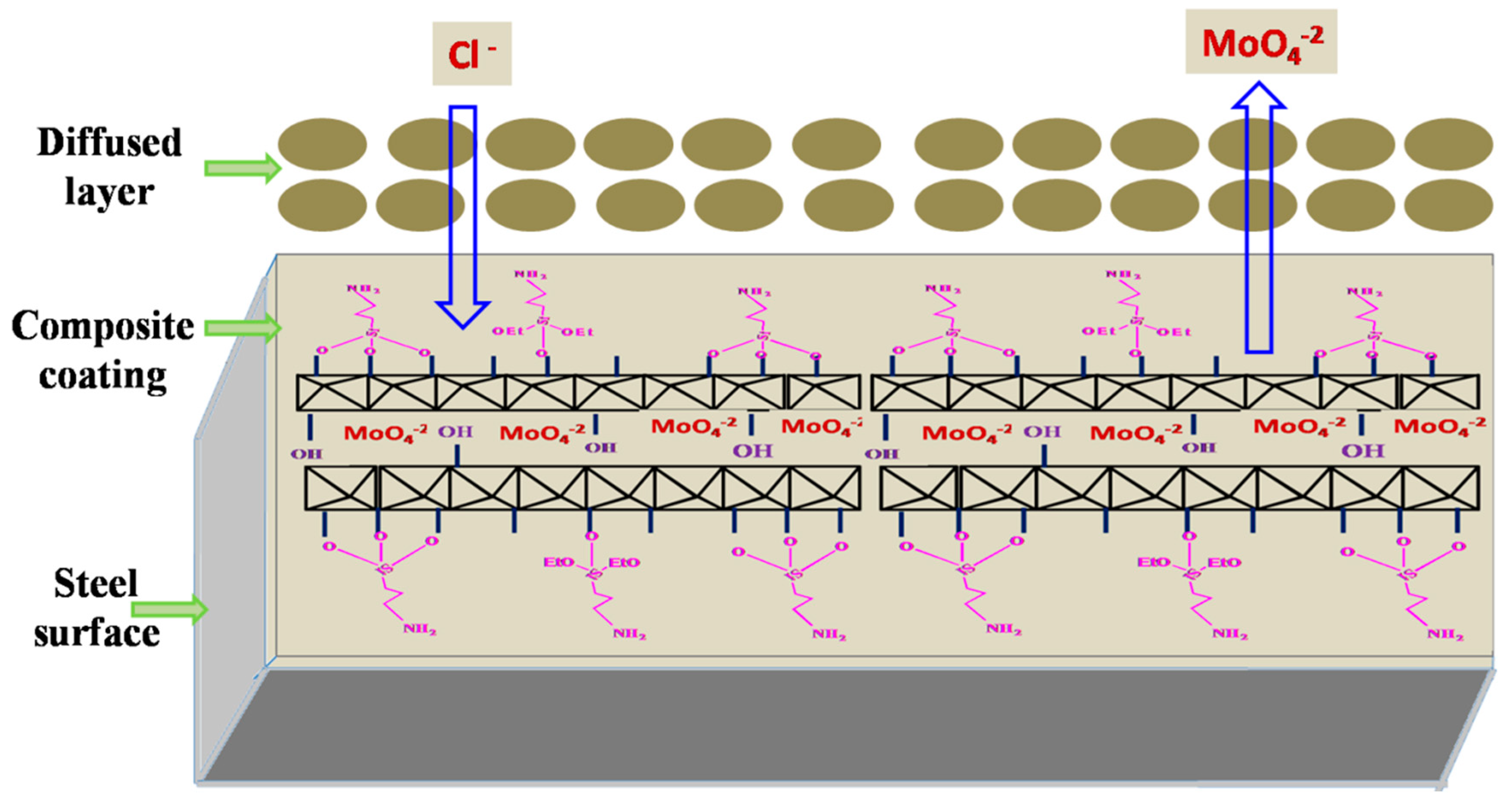
3.4. Inhibition Mechanism of the Composite Coating
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Xu, B.; Gong, W.; Zhang, K.; Yang, W.; Liu, Y.; Yin, X. Theoretical prediction and experimental study of 1-Butyl-2-(4-methylphenyl)benzimidazole as a novel corrosion inhibitor for mild steel in hydrochloric acid. J. Taiwan. Inst. Chem. Eng. 2015, 51, 193–200. [Google Scholar] [CrossRef]
- Rahman, O.; Ahmad, S. Physico-mechanical and electrochemical corrosion behavior of soy alkyd/Fe3O4 nanocomposite coatings. RSC Adv. 2014, 4, 14936–14947. [Google Scholar] [CrossRef]
- Abdollahi, H.; Ershad-Langroudi, A.; Salimi, A.; Rahimi, A. Anticorrosive Coatings prepared using Epoxy-Silica Hybrid Nanocomposite Materials. Ind. Eng. Chem. Res. 2014, 53, 10858–10869. [Google Scholar] [CrossRef]
- Ji, W.G.; Hu, J.M.; Zhang, J.Q.; Cao, C.N. Reducing the water absorption in epoxy coatings by silane monomer incorporation. Corros. Sci. 2006, 48, 3731–3739. [Google Scholar] [CrossRef]
- Ramezanzadeh, B.; Attar, M.M. Studying the corrosion resistance and hydrolytic degradation of an epoxy coating containing ZnO nanoparticles. Mater. Chem. Phys. 2011, 130, 1208–1219. [Google Scholar] [CrossRef]
- Raps, D.; Hack, T.; Wehr, J.; Zheludkevich, M.L.; Bastos, A.C.; Ferreira, M.G.S.; Nuyken, O. Electrochemical study of inhibitor-containing organic–inorganic hybrid coatings on AA2024. Corros. Sci. 2009, 51, 1012–1021. [Google Scholar] [CrossRef]
- Blustein, G.; Di Sarli, A.R.; Jaén, J.A.; Romagnoli, R.; Del Amo, B. Study of iron benzoate as a novel steel corrosion inhibitor pigment for protective paint films. Corros. Sci. 2007, 49, 4202–4231. [Google Scholar] [CrossRef]
- Williams, G.; McMurray, H.N. Anion-Exchange Inhibition of Filiform Corrosion on Organic Coated AA2024-T3 Aluminum Alloy by Hydrotalcite-Like Pigments. Electrochem. Solid-State Lett. 2003, 6, B9–B11. [Google Scholar] [CrossRef]
- Buchheit, R.G.; Guan, H.; Mahajanam, S.; Wong, F. Active Corrosion Protection and Corrosion Sensing in Chromate-Free Organic Coatings. Prog. Org. Coat. 2003, 47, 174–182. [Google Scholar] [CrossRef]
- Stimpfling, T.; Leroux, F.; Hintze-Bruening, H. Organo-Modified Layered Double Hydroxide in Coating Formulation to Protect AA2024 from Corrosion. Colloids Surf. A 2014, 458, 147–154. [Google Scholar] [CrossRef]
- Poznyak, S.K.; Tedim, J.; Rodrigues, L.M.; Salak, A.N.; Zheludkevich, M.L.; Dick, L.F.P.; Ferreira, M.G.S. Novel Inorganic Host Layered Double Hydroxides Intercalated with Guest Organic Inhibitors for Anticorrosion Applications. ACS Appl. Mater. Interfaces 2009, 1, 2353–2362. [Google Scholar] [CrossRef] [PubMed]
- Zheludkevich, M.L.; Poznyak, S.K.; Rodrigues, L.M.; Raps, D.; Hack, T.; Dick, L.F.; Nunes, T.; Ferreira, M.G.S. Active protection coatings with layered double hydroxide nanocontainers of corrosion inhibitor. Corros. Sci. 2010, 52, 602–611. [Google Scholar] [CrossRef]
- Alibakhshi, E.; Ghasemi, E.; Mahdavian, M.; Ramezanzadeh, B.; Farashi, S. Fabrication and Characterization of PO43− Intercalated Zn-Al-Layered Double Hydroxide Nanocontainer. J. Electrochem. Soc. 2016, 163, C495–C505. [Google Scholar] [CrossRef]
- Alibakhshi, E.; Ghasemi, E.; Mahdavian, M.; Ramezanzadeh, B. Fabrication and characterization of layered double hydroxide/silane nanocomposite coatings for protection of mild steel. J. Taiwan Inst. Chem. Eng. 2017, 80, 924–934. [Google Scholar] [CrossRef]
- Chhetri, S.; Adak, N.C.; Samanta, P.; Murmu, N.C.; Kuila, T. Rheological, Mechanical, and Thermal Properties of Silane Grafted Layered Double Hydroxide/Epoxy Composites. Ind. Eng. Chem. Res. 2018, 57, 8729–8739. [Google Scholar] [CrossRef]
- Wessling, B. Passivation of metals by coating with polyaniline: Corrosion potential shift and morphological changes. Adv. Mater. 1994, 6, 226–228. [Google Scholar] [CrossRef]
- Guo, X.X.; Xu, S.; Zhao, L.; Lu, W.; Zhang, F.; Evans, D.G.; Duan, X. One-step hydrothermal crystallization of a layered double hydroxide/alumina bilayer film on aluminum and its corrosion resistance properties. Langmuir 2009, 25, 9894–9897. [Google Scholar] [CrossRef]
- Ding, P.; Qu, B. Synthesis and characterization of exfoliated polystyrene/ZnAl layered double hydroxide nanocomposite via emulsion polymerization. J. Colloid Interface Sci. 2005, 291, 13–18. [Google Scholar] [CrossRef]
- Phuruangrat, A.; Thongtem, T.; Thongtem, S. Preparation, characterization and photoluminescence of nanocrystalline calcium molybdate. J. Alloys Compd. 2009, 481, 568–572. [Google Scholar] [CrossRef]
- Tao, Q.; He, H.; Frost, R.L.; Yuan, P.; Zhu, J. Nanomaterials based upon silylated layered double hydroxides. Appl. Clay Sci. 2009, 255, 4334–4340. [Google Scholar] [CrossRef]
- Kang, D.; Yu, X.; Tong, S.; Ge, M.; Zuo, J.; Cao, C.; Song, W. Performance and mechanism of Mg/Fe layered double hydroxides for fluoride and arsenate removal from aqueous solution. Chem. Eng. J. 2013, 228, 731–740. [Google Scholar] [CrossRef]
- Tao, Q.; He, H.; Li, T.; Frost, R.L.; Zhang, D.; He, Z. Tailoring surface properties and structure of layered double hydroxides using silanes with different number of functional groups. J. Solid State Chem. 2014, 213, 176–181. [Google Scholar] [CrossRef]
- Sun, W.; Wang, L.; Wu, T.; Pan, Y.; Liu, G. Synthesis of low-electrical-conductivity graphene/pernigraniline composites and their application in corrosion protection. Carbon 2014, 79, 605–614. [Google Scholar] [CrossRef]
- Qian, M.; Soutar, A.M.; Tan, X.H.; Zeng, X.T.; Wijesinghe, S.L. Two-part epoxysiloxane hybrid corrosion protection coatings for carbon steel. Thin Solid Films 2009, 517, 5237–5242. [Google Scholar] [CrossRef]
- Dai, N.; Zhang, L.C.; Zhang, J.; Chen, Q.; Wu, M. Corrosion behavior of selective laser melted Ti-6Al-4 V alloy in NaCl solution. Corros. Sci. 2016, 102, 484–489. [Google Scholar] [CrossRef]
- Qin, P.; Liu, Y.; Sercombe, T.B.; Li, Y.; Zhang, C.; Cao, C.; Sun, H.; Zhang, L.C. Improved Corrosion Resistance on Selective Laser Melting Produced Ti-5Cu Alloy after Heat Treatment. ACS Biomater. Sci. Eng. 2018, 4, 2633–2642. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, J.; Dai, N.; Qin, P.; Attar, H.; Zhang, L.C. Corrosion Behaviour of Selective Laser Melted Ti-TiBBiocomposite in Simulated Body Fluid. Electrochem. Acta. 2017, 232, 89–97. [Google Scholar] [CrossRef]
- Bai, Y.; Gai, X.; Li, S.; Zhang, L.C.; Liu, Y.; Hao, Y.; Zhang, X.; Yang, R.; Gao, Y. Improved corrosion behaviour of electron beam melted Ti-6Al–4V alloy in phosphate buffered saline. Corros. Sci. 2017, 123, 289–296. [Google Scholar] [CrossRef]
- Farr, J.P.G.; Seeney, A.M.; Bentley, A.J.; Earwaker, L.G. A technique for the in situ elemental analysis of electrode surfaces. Surf. Technol. 1984, 23, 99. [Google Scholar]
- Farr, J.P.G.; Saremi, M.; Seeney, A.M.; Bentley, A.J.; Earwaker, L.G. Molybdates in aqueous corrosion inhibition—III. Effects of molybdate in the anodic filming of steel. Polyhedron 1986, 5, 547. [Google Scholar]
- Mu, G.; Li, X.; Qu, Q.; Zhou, J. Molybdate and tungstate as corrosion inhibitors for cold rolling steel in hydrochloric acid solution. Corros. Sci. 2006, 48, 445–459. [Google Scholar] [CrossRef]

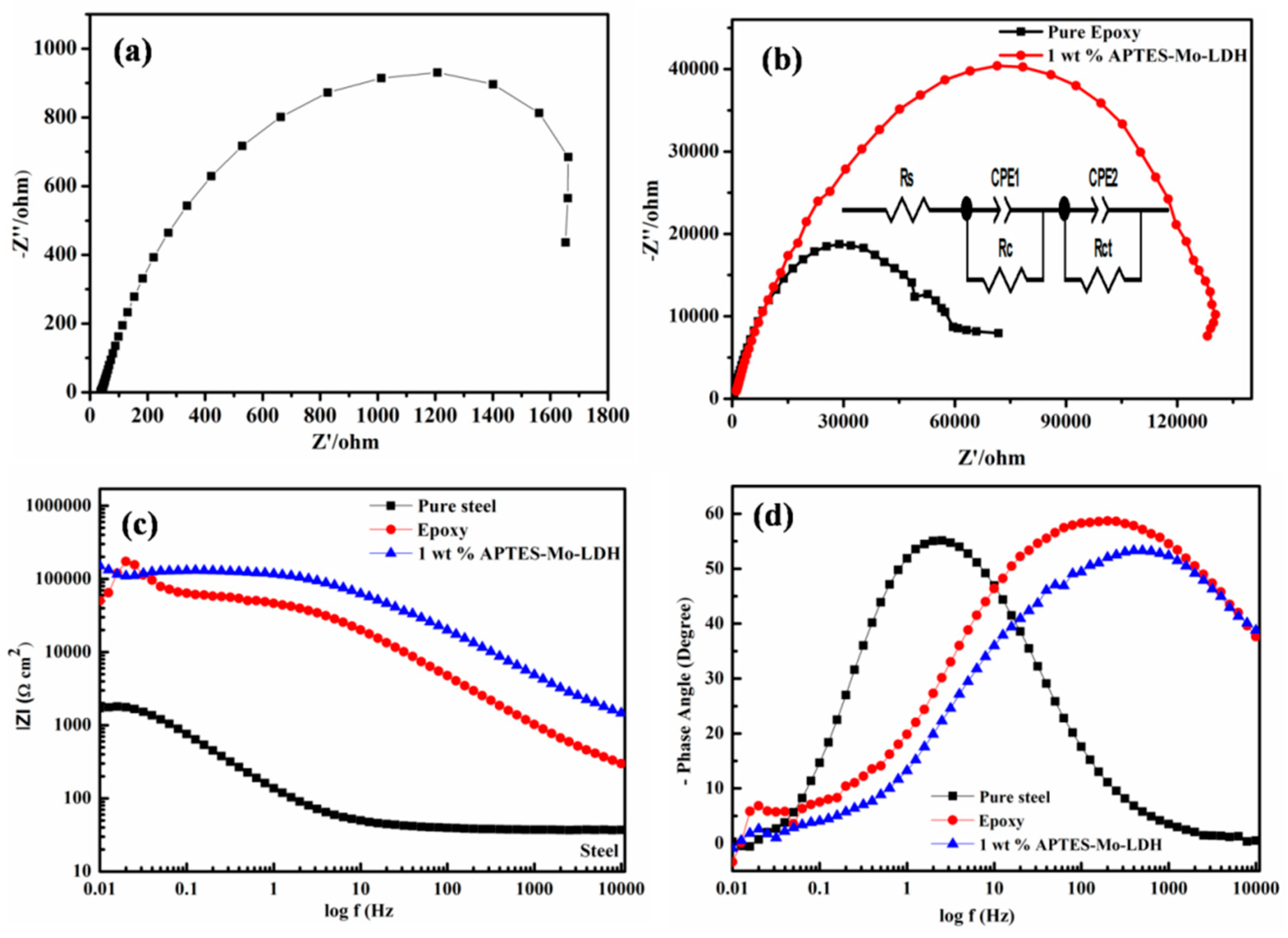
| Sample Type | Rs (Ω·cm2) | CPE1 (mF·cm−2) | Rc (Ω·cm2) | CPE2 (mF·cm−2) | Rct (Ω·cm2) | η % |
|---|---|---|---|---|---|---|
| Steel | 37.25 | 0.000496 | 900 | 0.000411 | 290 | - |
| Epoxy coating | 162.5 | 9.39 × 10−7 | 54,000 | 0.032 | 32,000 | - |
| Composite coating | 1000 | 1.9 × 10−7 | 62,000 | 7.19 × 10−7 | 72,000 | 99 |
| Sample Type | ECorr versus Ag/AgCl (mV) | ICorr (µa/m2) | βa (mV·dec−1) | βc (mV·dec−1) | PE (%) | RCorr (MPY) |
|---|---|---|---|---|---|---|
| Steel | −718.689 | 0.002 | 42.1 | 85.0 | - | 611.55 |
| Epoxy coating | −531.443 | 0.000 396 | 43.0 | 102.0 | 80.2 | 121.00 |
| Composite coating | −381.843 | 0.000 029 | 53.1 | 139.1 | 98.5 | 8.86 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chhetri, S.; Samanta, P.; Murmu, N.C.; Kuila, T. Anticorrosion Properties of Epoxy Composite Coating Reinforced by Molybdate-Intercalated Functionalized Layered Double Hydroxide. J. Compos. Sci. 2019, 3, 11. https://doi.org/10.3390/jcs3010011
Chhetri S, Samanta P, Murmu NC, Kuila T. Anticorrosion Properties of Epoxy Composite Coating Reinforced by Molybdate-Intercalated Functionalized Layered Double Hydroxide. Journal of Composites Science. 2019; 3(1):11. https://doi.org/10.3390/jcs3010011
Chicago/Turabian StyleChhetri, Suman, Pranab Samanta, Naresh Chandra Murmu, and Tapas Kuila. 2019. "Anticorrosion Properties of Epoxy Composite Coating Reinforced by Molybdate-Intercalated Functionalized Layered Double Hydroxide" Journal of Composites Science 3, no. 1: 11. https://doi.org/10.3390/jcs3010011
APA StyleChhetri, S., Samanta, P., Murmu, N. C., & Kuila, T. (2019). Anticorrosion Properties of Epoxy Composite Coating Reinforced by Molybdate-Intercalated Functionalized Layered Double Hydroxide. Journal of Composites Science, 3(1), 11. https://doi.org/10.3390/jcs3010011





