Chemical Stability of Tricalcium Phosphate–Iron Composite during Spark Plasma Sintering
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Tricalcium Phosphate-Iron Composite
2.2. Sintering Conditions
2.3. Chemical and Microstructural Characterization
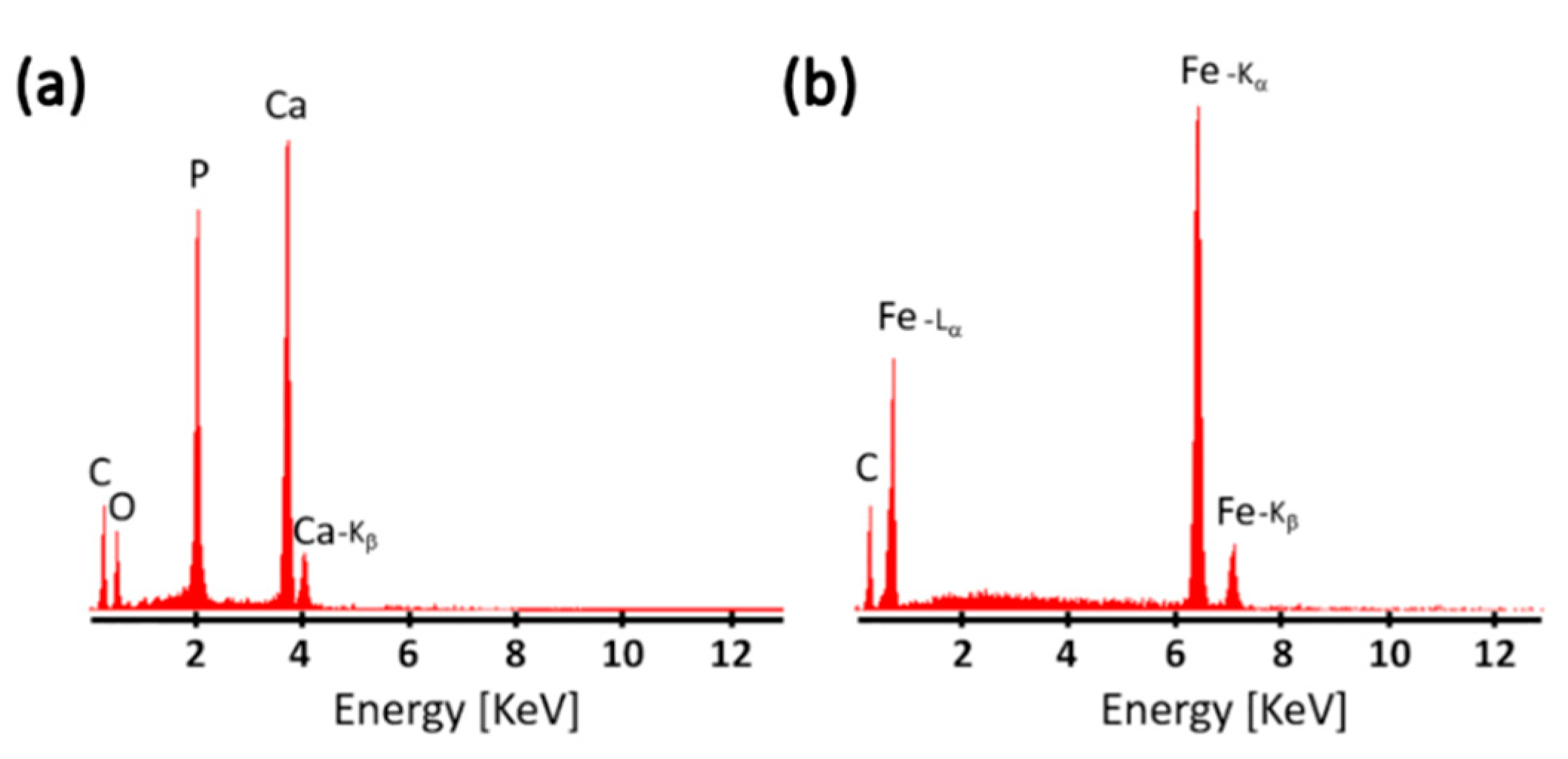
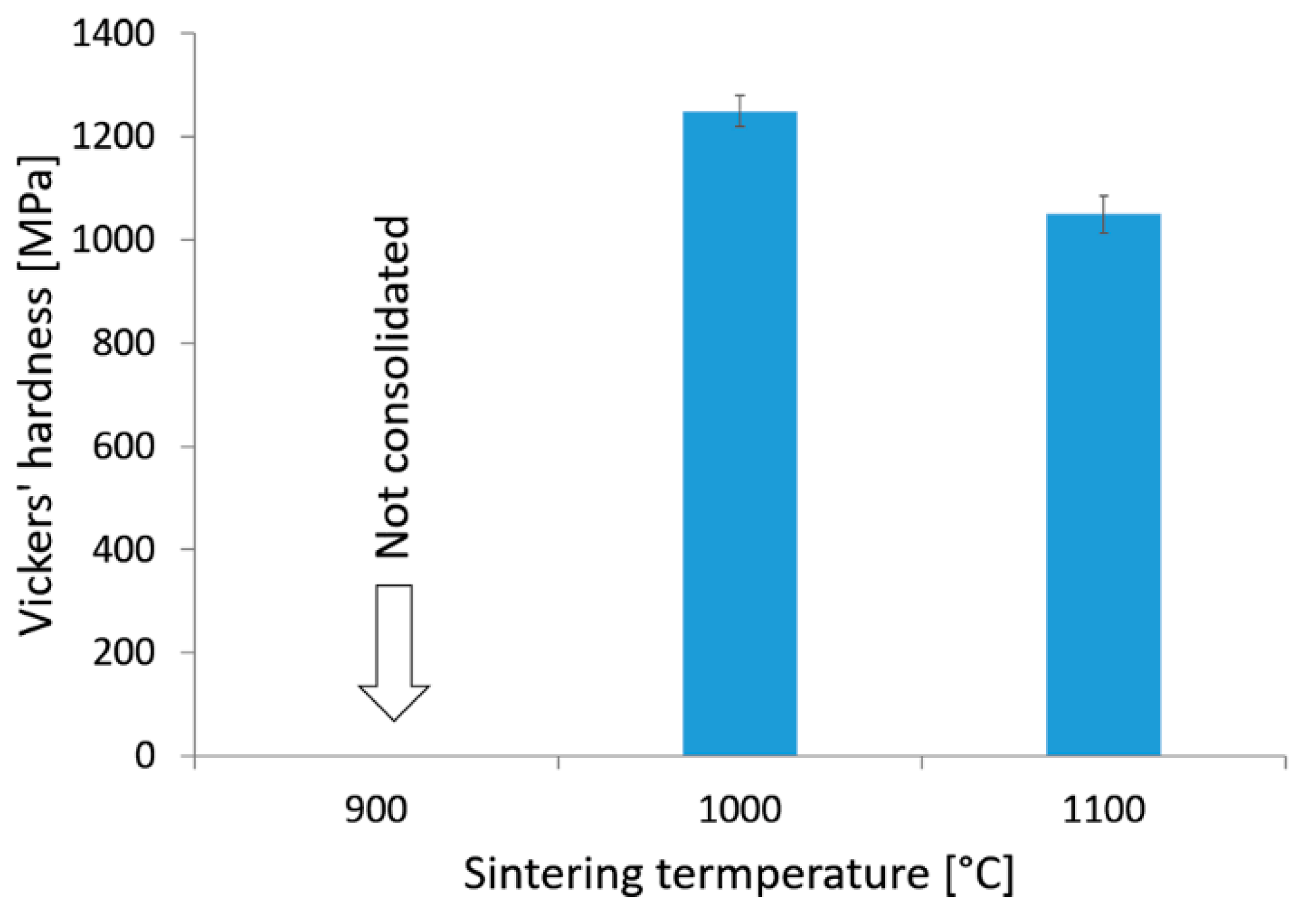
3. Results
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Al-Mayouf, A.M.; Al-Swayih, A.A.; Al-Mobarak, N.A.; Al-Jabab, A.S. Corrosion behavior of a new titanium alloy for dental implant applications in fluoride media. Mater. Chem. Phys. 2004, 86, 320–329. [Google Scholar] [CrossRef]
- LeGeros, R.Z.; LeGeros, J.P. Calcium phosphate bioceramics: Past, present, future. Key Eng. Mater. 2003, 240, 3–10. [Google Scholar] [CrossRef]
- Chu, C.; Xue, X.; Zhu, J.; Yin, Z. Fabrication and characterization of titanium-matrix composite with 20 vol% hydroxyapatite for use as heavy load-bearing hard tissue replacement. J. Mater. Sci. Mater. Med. 2006, 17, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Ye, H.; Liu, X.Y.; Hong, H. Characterization of sintered titanium/hydroxyapatite biocomposite using FTIR spectroscopy. J. Mater. Sci. Mater. Med. 2009, 20, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Chaki, T.K. Sintering behaviour and mechanical properties of hydroxyapatite and dicalcium phosphate. J. Mater. Sci. Mater. Med. 1993, 4, 150–158. [Google Scholar] [CrossRef]
- Elliott, J.C. Structure and Chemistry of the Apatites and Other Calcium Orthophosphates; Elsevier Science: Amsterdam, The Netherlands, 1994; Chapter 3. [Google Scholar]
- Carrodeguas, R.G.; De Aza, S. α-Tricalcium phosphate: Synthesis, properties and biomedical applications. Acta Biomater. 2011, 7, 3536–3546. [Google Scholar] [CrossRef] [PubMed]
- Wulfsberg, G. Oxidation-Reduction Chemistry of the Elements, in: Principles of Descriptive Inorganic Chemistry; University Science Books: Sausalito, CA, USA, 1991; pp. 137–179. [Google Scholar]
- Upadhyaya, G.S. Some issues in sintering science and technology. Mater. Chem. Phys. 2001, 67, 1–5. [Google Scholar] [CrossRef]
- Francis, A.; Yang, Y.; Virtanen, S.; Boccaccini, A.R. Iron and iron-based alloys for temporary cardiovascular applications. J. Mater. Sci. Mater. Med. 2015, 26, 138–153. [Google Scholar] [CrossRef] [PubMed]
- Montufar, E.B.; Maazouz, Y.; Ginebra, M.P. Relevance of the setting reaction to the injectability of tricalcium phosphate pastes. Acta Biomater. 2013, 9, 6188–6198. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Zheng, Y.F. In vitro study on newly designed biodegradable Fe-X composites (X = W, CNT) prepared by spark plasma sintering. J. Biomed. Mater. Res. Part B 2013, 101, 485–497. [Google Scholar] [CrossRef] [PubMed]
- White, A.A.; Best, S.M.; Kionloch, I.A. Hydroxyapatite–carbon nanotube composites for biomedical applications: A review. Int. J. Appl. Ceram. Technol. 2007, 4, 1–13. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, Y.; Sun, B.; Chen, C. Understanding the toxicity of carbon nanotubes. Acc. Chem. Res. 2013, 46, 702–713. [Google Scholar] [CrossRef] [PubMed]
- Sumitomo, N.; Noritake, K.; Hattori, T.; Morikawa, K.; Niwa, S.; Sato, K.; Niinomi, M. Experiment study on fracture fixation with low rigidity titanium alloy. J. Mater. Sci. Mater. Med. 2008, 19, 1581–1586. [Google Scholar] [CrossRef] [PubMed]
- Mondal, D.; Nguyen, L.; Oh, I.H.; Lee, B.T. Microstructure and biocompatibility of composite biomaterials fabricated from titanium and tricalcium phosphate by spark plasma sintering. J. Biomed. Mater. Res. A 2013, 101, 1489–1501. [Google Scholar] [CrossRef] [PubMed]
- Voisin, T.; Durand, L.; Karnatak, N.; Le Gallet, S.; Thomas, M.; Le Berre, Y.; Castagné, J.F.; Couret, A. Temperature control during spark plasma sintering and application to up-scaling and complex shaping. J. Mater. Proc. Tech. 2013, 213, 269–278. [Google Scholar] [CrossRef]
- Himesh, A.; Samar, J.; Kalita, J. Influence of oxide-based sintering additives on densification and mechanical behavior of tricalcium phosphate (TCP). J. Mater. Sci. Mater. Med. 2007, 18, 883–893. [Google Scholar]




© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casas-Luna, M.; Horynová, M.; Tkachenko, S.; Klakurková, L.; Celko, L.; Diaz-de-la-Torre, S.; Montufar, E.B. Chemical Stability of Tricalcium Phosphate–Iron Composite during Spark Plasma Sintering. J. Compos. Sci. 2018, 2, 51. https://doi.org/10.3390/jcs2030051
Casas-Luna M, Horynová M, Tkachenko S, Klakurková L, Celko L, Diaz-de-la-Torre S, Montufar EB. Chemical Stability of Tricalcium Phosphate–Iron Composite during Spark Plasma Sintering. Journal of Composites Science. 2018; 2(3):51. https://doi.org/10.3390/jcs2030051
Chicago/Turabian StyleCasas-Luna, Mariano, Miroslava Horynová, Serhii Tkachenko, Lenka Klakurková, Ladislav Celko, Sebastián Diaz-de-la-Torre, and Edgar B. Montufar. 2018. "Chemical Stability of Tricalcium Phosphate–Iron Composite during Spark Plasma Sintering" Journal of Composites Science 2, no. 3: 51. https://doi.org/10.3390/jcs2030051
APA StyleCasas-Luna, M., Horynová, M., Tkachenko, S., Klakurková, L., Celko, L., Diaz-de-la-Torre, S., & Montufar, E. B. (2018). Chemical Stability of Tricalcium Phosphate–Iron Composite during Spark Plasma Sintering. Journal of Composites Science, 2(3), 51. https://doi.org/10.3390/jcs2030051





