Islands of Tuberculosis Elimination: An Evaluation of Community-Based Active Case Finding in North Sumatra, Indonesia
Abstract
1. Introduction
2. Materials and Methods
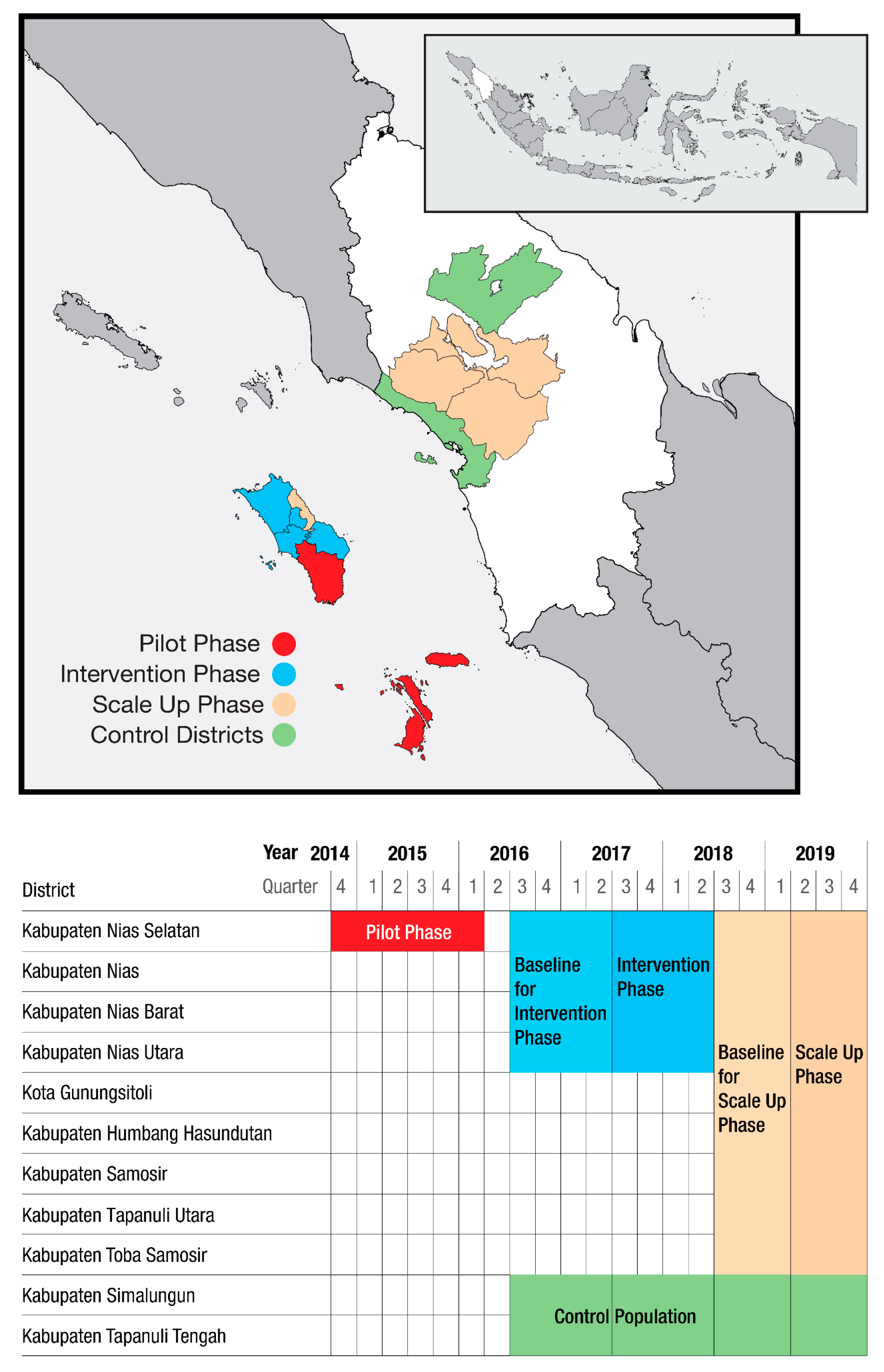
2.1. Setting
2.2. Timeline and Coverage
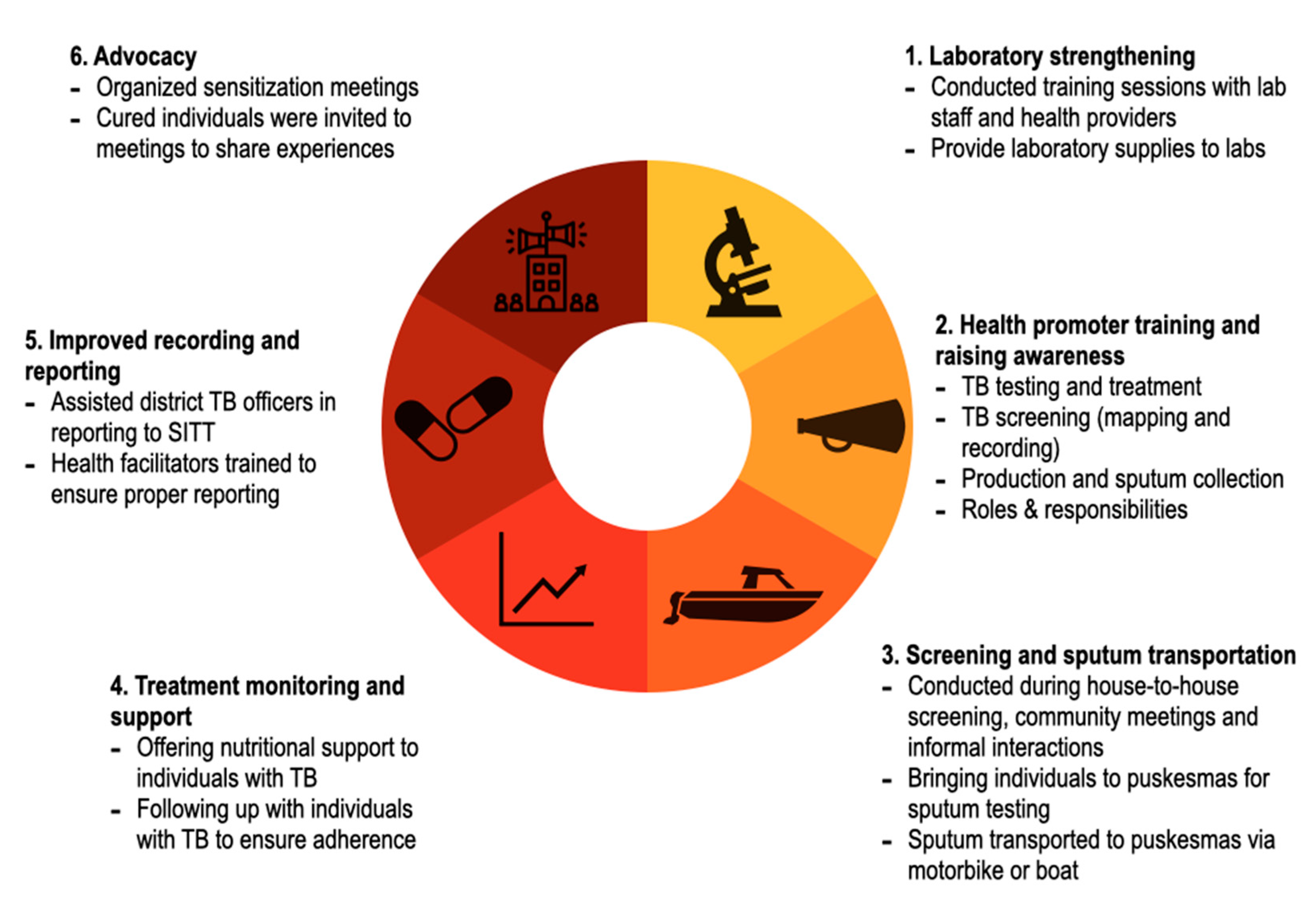
2.3. Community-Based Outreach Intervention
2.4. Data Collection
2.5. Data Analysis
2.6. Ethics Statement
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Global Tuberculosis Report 2019. Available online: https://www.who.int/teams/global-tuberculosis-programme/global-report-2019 (accessed on 18 September 2020).
- Stop TB Partnership. UN High Level Meeting on TB: Key Targets & Commitments for 2022. 2018. Available online: http://stoptb.org/assets/documents/global/advocacy/unhlm/UNHLM_Targets&Commitments.pdf (accessed on 18 September 2020).
- Creswell, J.; Sahu, S.; Blok, L.; Bakker, M.I.; Stevens, R.; Ditiu, L. A multi-site evaluation of innovative approaches to increase tuberculosis case notification: Summary results. PLoS ONE 2014, 9, e94465. [Google Scholar] [CrossRef]
- Stop TB Partnership. Partnership: The Paradigm Shift. 2016–2020. Global Plan to End TB; United Nations Office for Project Services: Geneva, Switzerland, 2015. [Google Scholar]
- De Vries, S.G.; Cremers, A.L.; Heuvelings, C.C.; Greve, P.F.; Visser, B.J.; Bélard, S.; Janssen, S.; Spijker, R.; Shaw, B.; Hill, R.A.; et al. Barriers and facilitators to the uptake of tuberculosis diagnostic and treatment services by hard-to-reach populations in countries of low and medium tuberculosis incidence: A systematic review of qualitative literature. Lancet Infect. Dis. 2017, 17, e128–e143. [Google Scholar] [CrossRef]
- World Health Organization. Joint Initiative “FIND. TREAT. All. #ENDTB”. 2018. Available online: https://www.who.int/tb/joint-initiative/en/ (accessed on 18 September 2020).
- Stop TB Partnership. The Strategic Initiative to Find the Missing People with TB. Available online: https://stoptb-strategicinitiative.org/ (accessed on 20 August 2020).
- Dowdy, D.W.; Basu, S.; Andrews, J.R. Is passive diagnosis enough? The impact of subclinical disease on diagnostic strategies for tuberculosis. Am. J. Respir. Crit. Care Med. 2013, 187, 543–551. [Google Scholar] [CrossRef]
- Ho, J.; Fox, G.J.; Marais, B.J. Passive case finding for tuberculosis is not enough. Int. J. Mycobacteriol. 2016, 5, 374–378. [Google Scholar] [CrossRef]
- Yuen, C.M.; Amanullah, F.; Dharmadhikari, A.; Nardell, E.A.; Seddon, J.A.; Vasilyeva, I.; Zhao, Y.; Keshavjee, S.; Becerra, M.C. Turning off the tap: Stopping tuberculosis transmission through active case-finding and prompt effective treatment. Lancet 2015, 386, 2334–2343. [Google Scholar] [CrossRef]
- Datiko, D.G.; Yassin, M.A.; Theobald, S.J.; Blok, L.; Suvanand, S.; Creswell, J.; Cuevas, L.E. Health extension workers improve tuberculosis case finding and treatment outcome in Ethiopia: A large-scale implementation study. BMJ Glob. Health 2017, 2, e000390. [Google Scholar] [CrossRef]
- Yassin, M.A.; Datiko, D.G.; Tulloch, O.; Markos, P.; Aschalew, M.; Shargie, E.B.; Dangisso, M.H.; Komatsu, R.; Sahu, S.; Blok, L.; et al. Innovative community-based approaches doubled tuberculosis case notification and improve treatment outcome in Southern Ethiopia. PLoS ONE 2013, 8, e63174. [Google Scholar] [CrossRef]
- Codlin, A.J.; Monyrath, C.; Ky, M.; Gerstel, L.; Creswell, J.; Eang, M.T. Results from a roving, active case finding initiative to improve tuberculosis detection among older people in rural Cambodia using the Xpert MTB/RIF assay and chest X-ray. J. Clin. Tuberc. Other Mycobact. Dis. 2018, 13, 22–27. [Google Scholar] [CrossRef]
- Eang, M.T.; Satha, P.; Yadav, R.P.; Morishita, F.; Nishikiori, N.; van-Maaren, P.; Lambregts-van Weezenbeek, C. Early detection of tuberculosis through community-based active case finding in Cambodia. BMC Public Health 2012, 12, 469. [Google Scholar] [CrossRef]
- Morishita, F.; Eang, M.T.; Nishikiori, N.; Yadav, R.P. Increased case notification through active case finding of tuberculosis among household and neighbourhood contacts in Cambodia. PLoS ONE 2016, 11, e0150405. [Google Scholar] [CrossRef]
- Myint, O.; Saw, S.; Isaakidis, P.; Khogali, M.; Reid, A.; Hoa, N.B.; Kyaw, T.T.; Zaw, K.K.; Khaing, T.M.; Aung, S.T. Active case-finding for tuberculosis by mobile teams in Myanmar: Yield and treatment outcomes. Infect. Dis. Poverty 2017, 6, 77. [Google Scholar] [CrossRef]
- Prasad, B.; Satyanarayana, S.; Chadha, S.S.; Das, A.; Thapa, B.; Mohanty, S.; Pandurangan, S.; Babu, E.R.; Tonsing, J.; Sachdeva, K.S. Experience of active tuberculosis case finding in nearly 5 million households in India. Public Health Action 2016, 6, 15–18. [Google Scholar] [CrossRef]
- Shewade, H.D.; Gupta, V.; Satyanarayana, S.; Kharate, A.; Sahai, K.N.; Murali, L.; Kamble, S.; Deshpande, M.; Kumar, N.; Kumar, S.; et al. Active case finding among marginalised and vulnerable populations reduces catastrophic costs due to tuberculosis diagnosis. Glob. Health Action 2018, 11, 1494897. [Google Scholar] [CrossRef]
- Mitchell, E.M.; Golub, J.; Portocarrero, A. Acceptability of TB Screening among at-Risk and Vulnerable Groups; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- World Health Organization. Toolkit to Develop a National Strategic Plan for TB Prevention, Care and Control. 2015. Available online: https://apps.who.int/iris/bitstream/handle/10665/153811/9789241507974_eng.pdf (accessed on 4 August 2020).
- Surya, A.; Setyaningsih, B.; Suryani Nasution, H.; Gita Parwati, C.; Yuzwar, Y.E.; Osberg, M.; Hanson, C.L.; Hymoff, A.; Mingkwan, P.; Makayova, J.; et al. Quality tuberculosis Care in Indonesia: Using patient pathway analysis to optimize public–private collaboration. J. Infect. Dis. 2017, 216 (Suppl. 7), S724–S732. [Google Scholar] [CrossRef]
- Indonesia National TB Program. Current Status of Integrated Community Based TB Service Delivery and the Global Fund Work Plan to Find Missing TB Cases. 2018. Available online: https://www.who.int/tb/features_archive/indonesia_11apr18.pdf?ua=1 (accessed on 25 September 2020).
- McAllister, S.; Wiem Lestari, B.; Sujatmiko, B.; Siregar, A.; Sihaloho, E.D.; Fathania, D.; Dewi, N.F.; Koesoemadinata, R.C.; Hill, P.C.; Alisjahbana, B. Feasibility of two active case finding approaches for detection of tuberculosis in Bandung City, Indonesia. Public Health Action 2017, 7, 206–211. [Google Scholar] [CrossRef][Green Version]
- Ahmad, R.A.; Mahendradhata, Y.; Cunningham, J.; Utarini, A.; de Vlas, S.J. How to optimize tuberculosis case finding: Explorations for Indonesia with a health system model. BMC Infect. Dis. 2009, 9, 87. [Google Scholar] [CrossRef]
- Stop TB Partnership. The TB REACH Initiative. Available online: http://www.stoptb.org/global/awards/tbreach/ (accessed on 24 September 2020).
- Kementerian Kesehatan Republik Indonesia. Estimasi Penduduk Menurut Umur Tunggal Dal Jenis Kelamin Menurut Kabupaten/Kota Tahun 2014. Available online: https://web.archive.org/web/20140208021950/http:/depkes.go.id/downloads/Penduduk%20Kab%20Kota%20Umur%20Tunggal%202014.pdf (accessed on 25 September 2020).
- Wikipedia. North Sumatra. 2020. Available online: https://en.wikipedia.org/wiki/North_Sumatra (accessed on 25 September 2020).
- Blok, L.; Creswell, J.; Stevens, R.; Brouwer, M.; Ramis, O.; Weil, O.; Klatser, P.; Sahu, S.; Bakker, M.I. A pragmatic approach to measuring, monitoring and evaluating interventions for improved tuberculosis case detection. Int. Health 2014, 6, 181–188. [Google Scholar] [CrossRef]
- Corbett, E.L.; Bandason, T.; Duong, T.; Dauya, E.; Makamure, B.; Churchyard, G.J.; Williams, B.G.; Munyati, S.S.; Butterworth, A.E.; Mason, P.R.; et al. Comparison of two active case-finding strategies for community-based diagnosis of symptomatic smear-positive tuberculosis and control of infectious tuberculosis in Harare, Zimbabwe (DETECTB): A cluster-randomised trial. Lancet 2010, 376, 1244–1253. [Google Scholar] [CrossRef]
- Miller, A.C.; Golub, J.E.; Cavalcante, S.C.; Durovni, B.; Moulton, L.H.; Fonseca, Z.; Arduini, D.; Chaisson, R.E.; Soares, E.C. Controlled trial of active tuberculosis case finding in a Brazilian favela. Int. J. Tuberc. Lung Dis. 2010, 14, 720–726. [Google Scholar]
- Marks, G.B.; Nguyen, N.V.; Nguyen, P.T.; Nguyen, T.A.; Nguyen, H.B.; Tran, K.H.; Nguyen, S.V.; Luu, K.B.; Tran, D.T.; Vo, Q.T.; et al. Community-wide screening for tuberculosis in a high-prevalence setting. N. Engl. J. Med. 2019, 381, 1347–1357. [Google Scholar] [CrossRef]
- Ayles, H.; Muyoyeta, M.; Du Toit, E.; Schaap, A.; Floyd, S.; Simwinga, M.; Shanaube, K.; Chishinga, N.; Bond, V.; Dunbar, R.; et al. Effect of household and community interventions on the burden of tuberculosis in southern Africa: The ZAMSTAR community-randomised trial. Lancet 2013, 382, 1183–1194. [Google Scholar] [CrossRef]
- Gurung, S.C.; Dixit, K.; Rai, B.; Caws, M.; Paudel, P.R.; Dhital, R.; Acharya, S.; Budhathoki, G.; Malla, D.; Levy, J.W.; et al. The role of active case finding in reducing patient incurred catastrophic costs for tuberculosis in Nepal. Infect. Dis. Poverty 2019, 8, 99. [Google Scholar] [CrossRef]
- Kranzer, K.; Afnan-Holmes, H.; Tomlin, K.; Golub, J.E.; Shapiro, A.E.; Schaap, A.; Corbett, E.L.; Lönnroth, K.; Glynn, J.R. The benefits to communities and individuals of screening for active tuberculosis disease: A systematic review [State of the art series. Case finding/screening. Number 2 in the series]. Int. J. Tuberc. Lung Dis. 2013, 17, 432–446. [Google Scholar] [CrossRef]
- Albert, H.; Purcell, R.; Wang, Y.Y.; Kao, K.; Mareka, M.; Katz, Z.; Maama, B.L. Mots’ oane T Designing an optimized diagnostic network to improve access to TB diagnosis and treatment in Lesotho. PLoS ONE 2020, 15, e0233620. [Google Scholar] [CrossRef]
- John, S.; Gidado, M.; Dahiru, T.; Fanning, A.; Codlin, A.J.; Creswell, J. Tuberculosis among nomads in Adamawa, Nigeria: Outcomes from two years of active case finding. Int. J. Tuberc. Lung Dis. 2015, 19, 463–468. [Google Scholar] [CrossRef]
- Vyas, A.; Creswell, J.; Codlin, A.J.; Stevens, R.; Rao, V.G.; Kumar, B.; Khaparde, S.; Sahu, S. Community-based active case-finding to reach the most vulnerable: Tuberculosis in tribal areas of India. Int. J. Tuberc. Lung Dis. 2019, 23, 750–755. [Google Scholar] [CrossRef]
- Abdullahi, S.A.; Smelyanskaya, M.; John, S.; Adamu, H.I.; Ubochioma, E.; Kennedy, I.; Abubakar, F.A.; Ago, H.A.; Stevens, R.; Creswell, J. Providing TB and HIV outreach services to internally displaced populations in Northeast Nigeria: Results of a controlled intervention study. PLoS Med. 2020, 17, e1003218. [Google Scholar] [CrossRef]
- Onozaki, I.; Law, I.; Sismanidis, C.; Zignol, M.; Glaziou, P.; Floyd, K. National tuberculosis prevalence surveys in Asia, 1990–2012: An overview of results and lessons learned. Trop. Med. Int. Health 2015, 20, 1128–1145. [Google Scholar] [CrossRef]
- World Health Organization. Approaches to Improve Sputum Smear Microscopy for Tuberculosis Diagnosis: Expert Group Meeting Report. 2009. Available online: https://www.who.int/tb/laboratory/egmreport_microscopymethods_nov09.pdf (accessed on 4 July 2020).
- World Health Organization. Systematic Screening for Active Tuberculosis: Principles and Recommendations; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- rGLC Country Support Mission Report. 2017. Available online: https://www.who.int/docs/default-source/searo/tuberculosis/pmdt-indonesia-2017.pdf?sfvrsn=b07db6e9_2 (accessed on 20 August 2020).
- World Health Organization. Molecular Assays Intended as Initial Tests for the Diagnosis of Pulmonary and Extrapulmonary TB and Rifampicin Resistance in Adults and Children: Rapid Communication; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Creswell, J.; Rai, B.; Wali, R.; Sudrungrot, S.; Adhikari, L.M.; Pant, R.; Pyakurel, S.; Uranw, D.; Codlin, A.J. Introducing new tuberculosis diagnostics: The impact of Xpert® MTB/RIF testing on case notifications in Nepal. Int. J. Tuberc. Lung Dis. 2015, 19, 545–551. [Google Scholar] [CrossRef]
- Theron, G.; Zijenah, L.; Chanda, D.; Clowes, P.; Rachow, A.; Lesosky, M.; Bara, W.; Mungofa, S.; Pai, M.; Hoelscher, M.; et al. Feasibility, accuracy, and clinical effect of point-of-care Xpert MTB/RIF testing for tuberculosis in primary-care settings in Africa: A multicentre, randomised, controlled trial. Lancet 2014, 383, 424–435. [Google Scholar] [CrossRef]
- TB CARE I Core Project: Intensified Implementation of GeneXpert MTB/RIF in 3 Countries. KNCV Tuberculosis Foundation to Eliminate TB 2013. Available online: https://www.kncvtbc.org/uploaded/2015/09/TB_CARE_I_GeneXpert_Core_Project_Final_Report.pdf (accessed on 20 August 2020).
- Van’t Hoog, A.H.; Onozaki, I.; Lonnroth, K. Choosing algorithms for TB screening: A modelling study to compare yield, predictive value and diagnostic burden. BMC Infect. Dis. 2014, 14, 532. [Google Scholar] [CrossRef] [PubMed]
- Suárez, P.G.; Watt, C.J.; Alarcón, E.; Portocarrero, J.; Zavala, D.; Canales, R.; Luelmo, F.; Espinal, M.A.; Dye, C. The dynamics of tuberculosis in response to 10 years of intensive control effort in Peru. J. Infect. Dis. 2001, 184, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Creswell, J.; Codlin, A.J.; Oliva Flores, S.; Samayoa, M.; Ramis, O.; Guardado, M.E. Will more sensitive diagnostics identify tuberculosis missed by clinicians? Evaluating Xpert MTB/RIF testing in Guatemala. Gac. Sanit. 2020, 34, 127–132. [Google Scholar] [CrossRef]
- Mhimbira, F.A.; Cuevas, L.E.; Dacombe, R.; Mkopi, A.; Sinclair, D. Interventions to increase tuberculosis case detection at primary healthcare or community-level services. Cochrane Database Syst. Rev. 2017. [Google Scholar] [CrossRef]
| Pilot Phase # | Intervention Phase + | Scale-up Phase & | Overall Total | |||
|---|---|---|---|---|---|---|
| Nias | Mainland | Total | ||||
| People Screened | N/A | 124,430 | 126,384 | 126,390 | 252,774 | 377,204 |
| People Referred for Testing | 3261 | 6084 | 5464 | 4280 | 9744 | 19,089 |
| People Tested | 2983 | 5807 | 5287 | 3675 | 8962 | 17,752 |
| People with B+ TB | 215 | 509 | 429 | 394 | 823 | 1547 |
| People Initiated on Treatment | 215 | 492 | 388 | 370 | 758 | 1465 |
| Presumptive Rate (Presumptive/Screened) | 4.9% | 4.3% | 3.4% | 3.9% | 4.2% ** | |
| % of presumptive people tested | 91.5% | 95.4% | 96.8% | 85.9% | 92.0% | 93.0% |
| % yield of TB testing | 7.2% | 8.8% | 8.1% | 13.1% | 9.2% | 8.7% |
| % B+ linked to treatment | 100.0% | 96.7% | 90.4% | 91.7% | 92.1% | 94.7% |
| B+ TB Notifications Intervention Districts | B+ TB Notifications Control Districts | |
|---|---|---|
| Pilot phase Nias * | ||
| Baseline ^ | 444 | NA |
| Intervention | 509 | NA |
| ACF direct yield | 215 | NA |
| % notifications identified by ACF | 42% | NA |
| Number and (%) change from baseline | 65 (+15%) | NA |
| Intervention phase Nias + | ||
| Baseline | 495 | 1424 |
| Intervention | 916 | 1653 |
| ACF direct yield | 492 | NA |
| % notifications identified by ACF | 54% | NA |
| Number and (%) change from baseline | 421 (+85%) | 229 (+16%) |
| Scale-up phase Nias & | ||
| Baseline | 551 | 1176 |
| Intervention | 673 | 1184 |
| ACF direct yield | 388 | NA |
| % notifications identified by ACF | 58% | NA |
| Number and (%) change from baseline | 122 (+22%) | 8 (+1%) |
| Scale-up phase Mainland North Sumatra & | ||
| Baseline | 686 | 1176 |
| Intervention | 672 | 1184 |
| ACF direct yield | 370 | NA |
| % notifications identified by ACF | 55% | NA |
| Number and (%) change from baseline | −14 (−2%) | 8 (+1%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siahaan, E.S.; Bakker, M.I.; Pasaribu, R.; Khan, A.; Pande, T.; Hasibuan, A.M.; Creswell, J. Islands of Tuberculosis Elimination: An Evaluation of Community-Based Active Case Finding in North Sumatra, Indonesia. Trop. Med. Infect. Dis. 2020, 5, 163. https://doi.org/10.3390/tropicalmed5040163
Siahaan ES, Bakker MI, Pasaribu R, Khan A, Pande T, Hasibuan AM, Creswell J. Islands of Tuberculosis Elimination: An Evaluation of Community-Based Active Case Finding in North Sumatra, Indonesia. Tropical Medicine and Infectious Disease. 2020; 5(4):163. https://doi.org/10.3390/tropicalmed5040163
Chicago/Turabian StyleSiahaan, Elvi S., Mirjam I. Bakker, Ratna Pasaribu, Amera Khan, Tripti Pande, Alwi Mujahit Hasibuan, and Jacob Creswell. 2020. "Islands of Tuberculosis Elimination: An Evaluation of Community-Based Active Case Finding in North Sumatra, Indonesia" Tropical Medicine and Infectious Disease 5, no. 4: 163. https://doi.org/10.3390/tropicalmed5040163
APA StyleSiahaan, E. S., Bakker, M. I., Pasaribu, R., Khan, A., Pande, T., Hasibuan, A. M., & Creswell, J. (2020). Islands of Tuberculosis Elimination: An Evaluation of Community-Based Active Case Finding in North Sumatra, Indonesia. Tropical Medicine and Infectious Disease, 5(4), 163. https://doi.org/10.3390/tropicalmed5040163








