A One Health Approach to Investigating Leptospira Serogroups and Their Spatial Distributions among Humans and Animals in Rio Grande do Sul, Brazil, 2013–2015
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
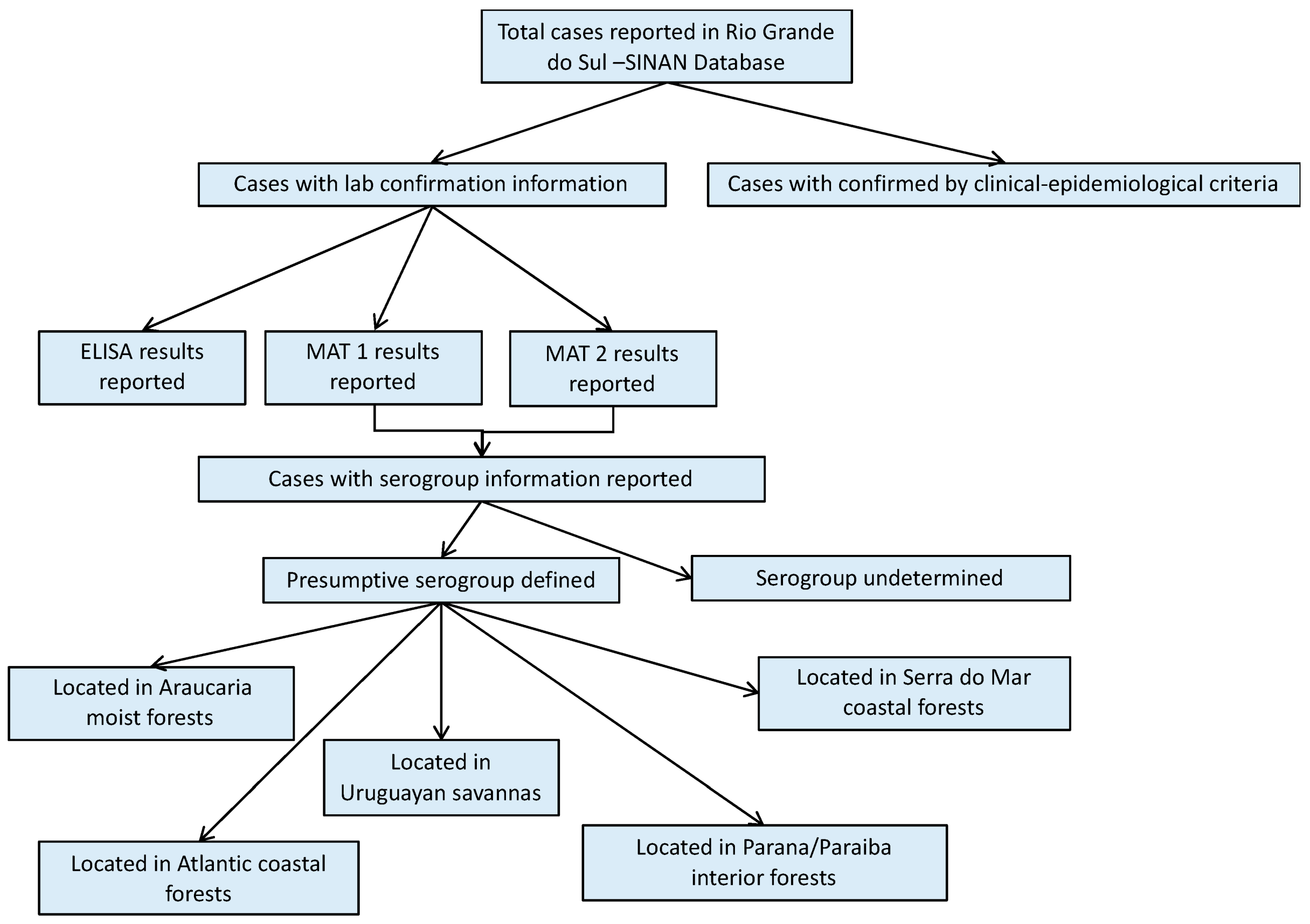
2.2. Study Design and Data Collection
2.3. Definitions
2.3.1. Confirmed Human Leptospirosis Case Definition
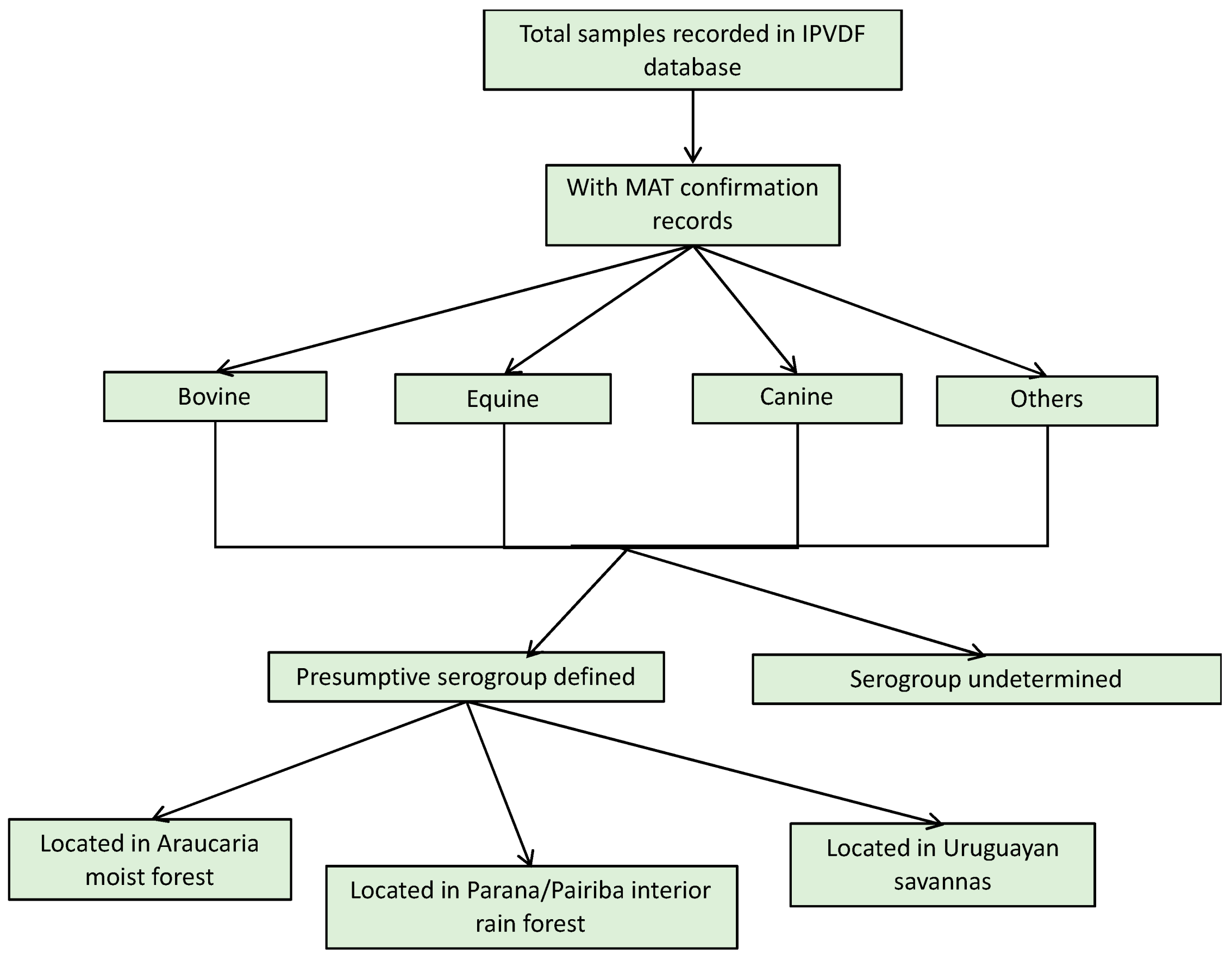
2.3.2. Confirmed Animal Leptospirosis Case Definition
2.3.3. Definitions for Interpreting MAT Results
MAT testing for humans:
- (1)
- Reactive ELISA-IgM sample in an acute sample and seroconversion in MAT from an acute sample to a second sample taken 14–60 days after symptom onset, with a titer greater than or equal to 200;
- (2)
- Increase of four times or more in MAT titers between two samples collected within 14 days after the onset of symptoms (maximum of 60 days) between them;
- (3)
- When two or more samples are not available, a single acute sample greater than or equal to a titer of 800 in MAT.
MAT testing for animals:
Presumptive serogroup MAT definition:
2.3.4. Ecoregion Definition
2.4. Data Analysis and Management
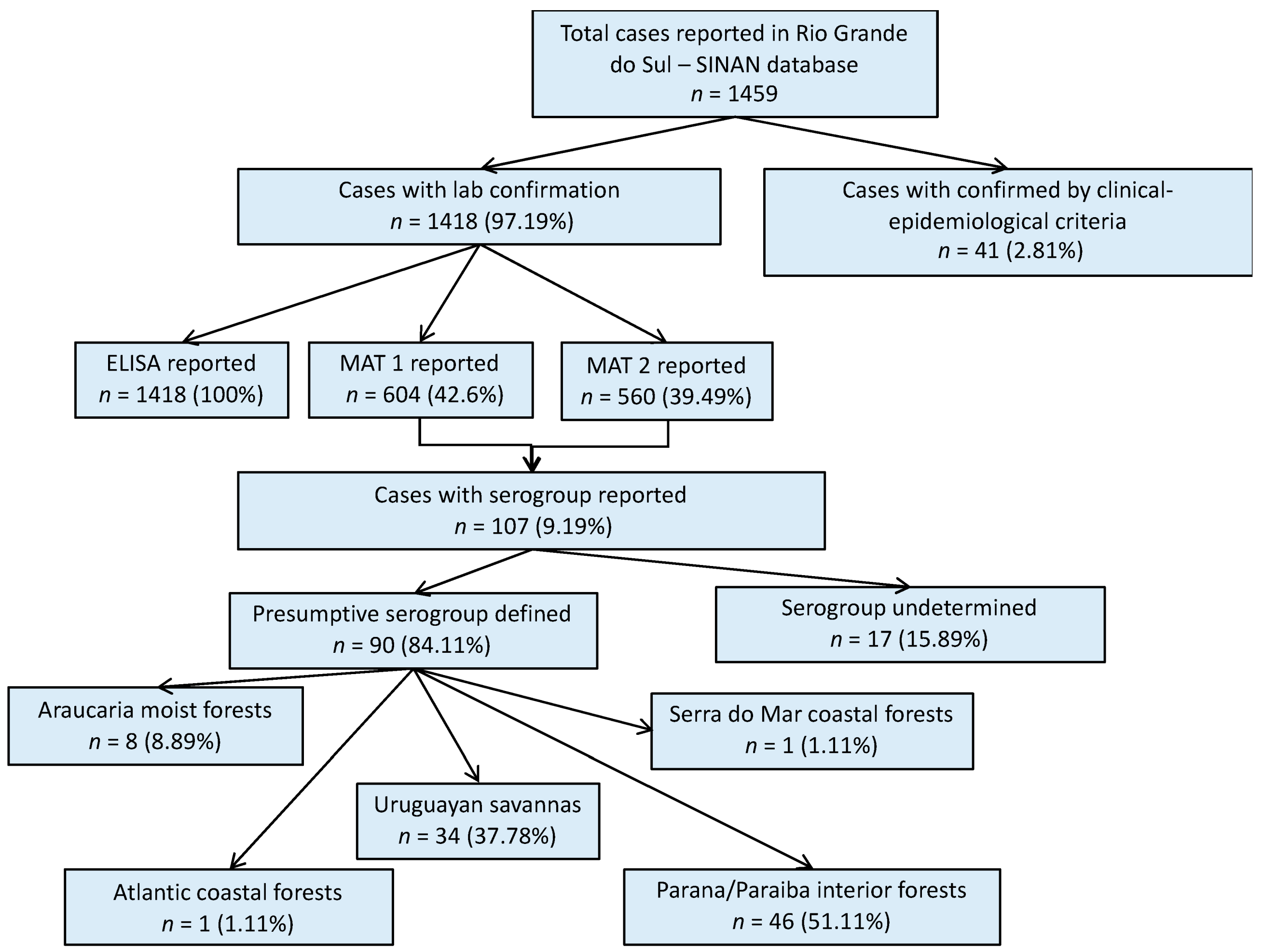
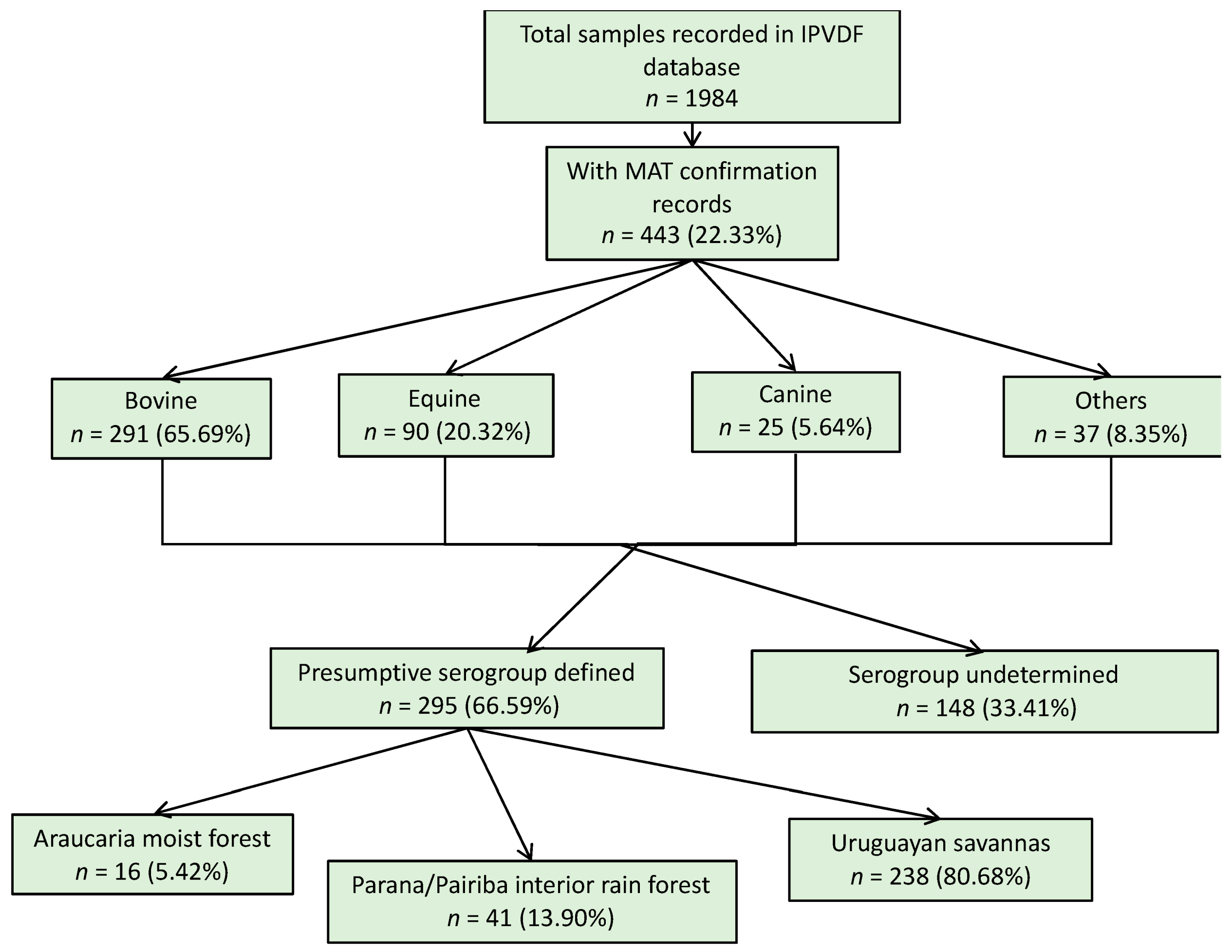
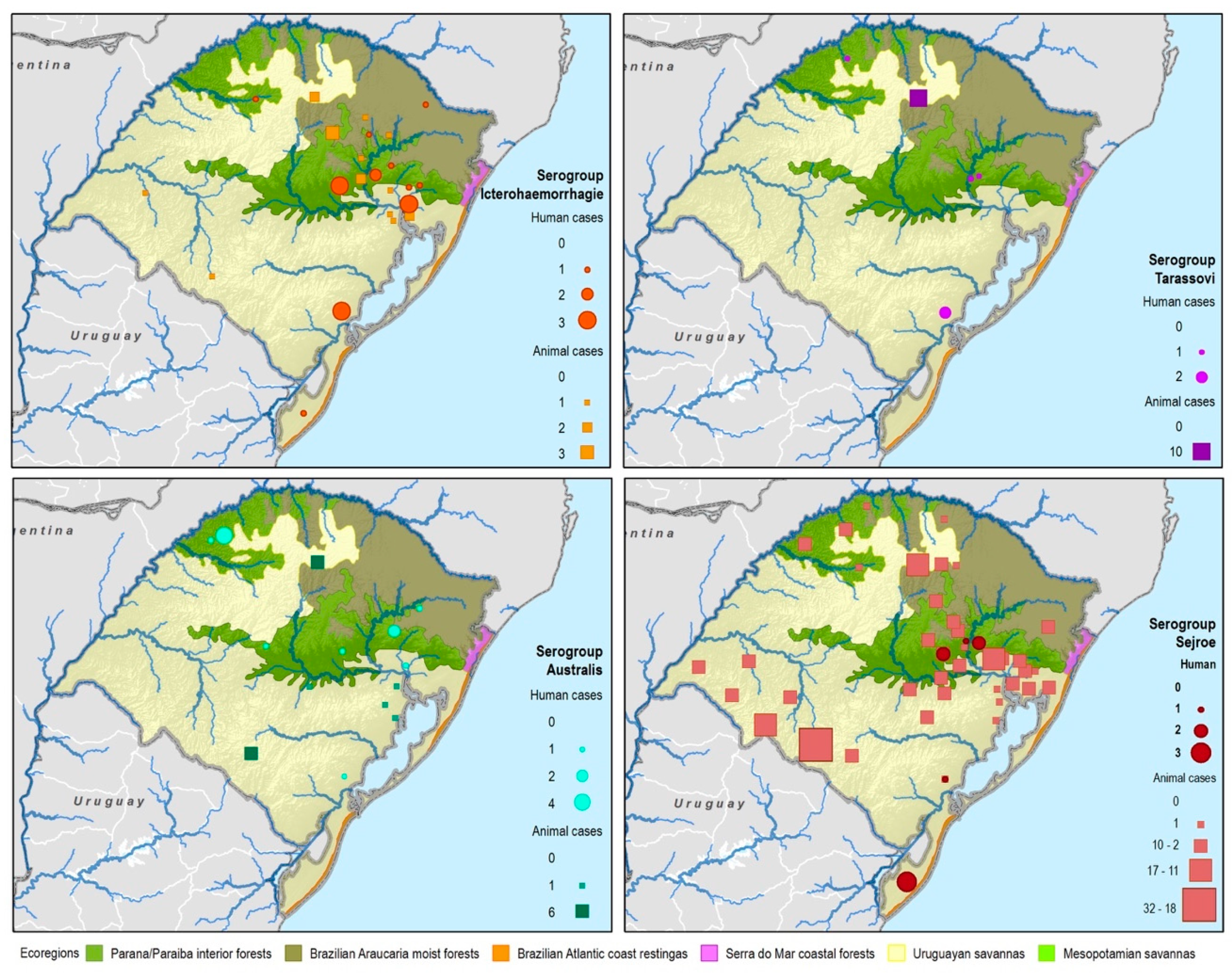
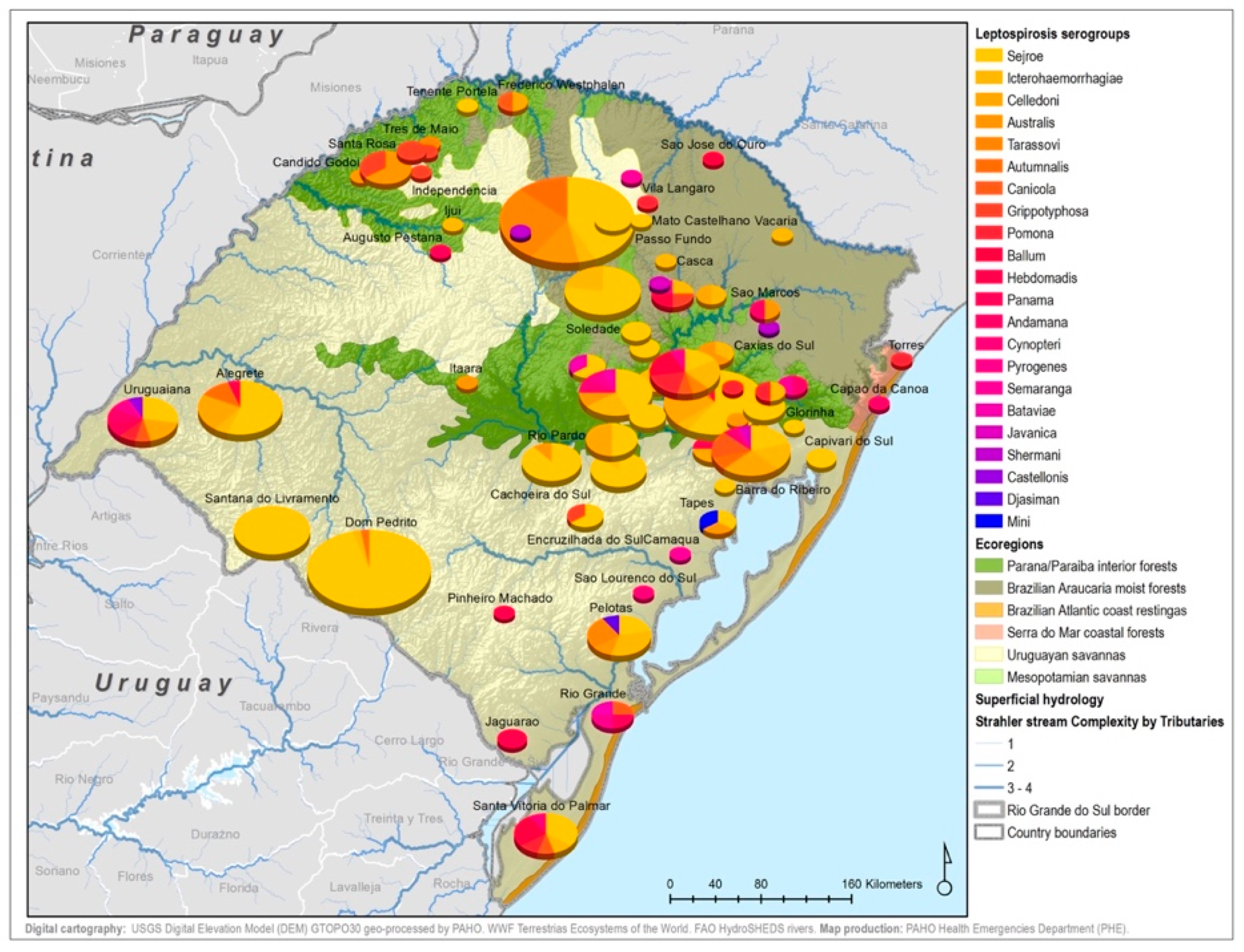
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Variable | 2013 n (% Total) | 2014 n (% Total) | 2015 n (% Total) | TOTAL n (% Total) |
|---|---|---|---|---|
| Sex | ||||
| Female | 2 (2.22) | 4 (4.44) | 5 (5.56) | 11 (12.22) |
| Male | 11 (12.22) | 38 (42.22) | 30 (33.33) | 79 (87.78) |
| Race | ||||
| Black | 2 (2.22) | - | 2 (2.22) | 4 (4.44) |
| Parda | - | 1 (1.11) | - | 1 (1.11) |
| White | 11 (12.22) | 41 (45.56) | 33 (36.67) | 85 (94.44) |
| Human settlement | ||||
| Urban | 10 (11.11) | 25 (27.78) | 23 (25.56) | 58 (64.44) |
| Rural | 3 (3.33) | 16 (17.78) | 12 (13.33) | 31(34.44) |
| Peri-urban | - | 1 (1.11) | - | 1 (1.11) |
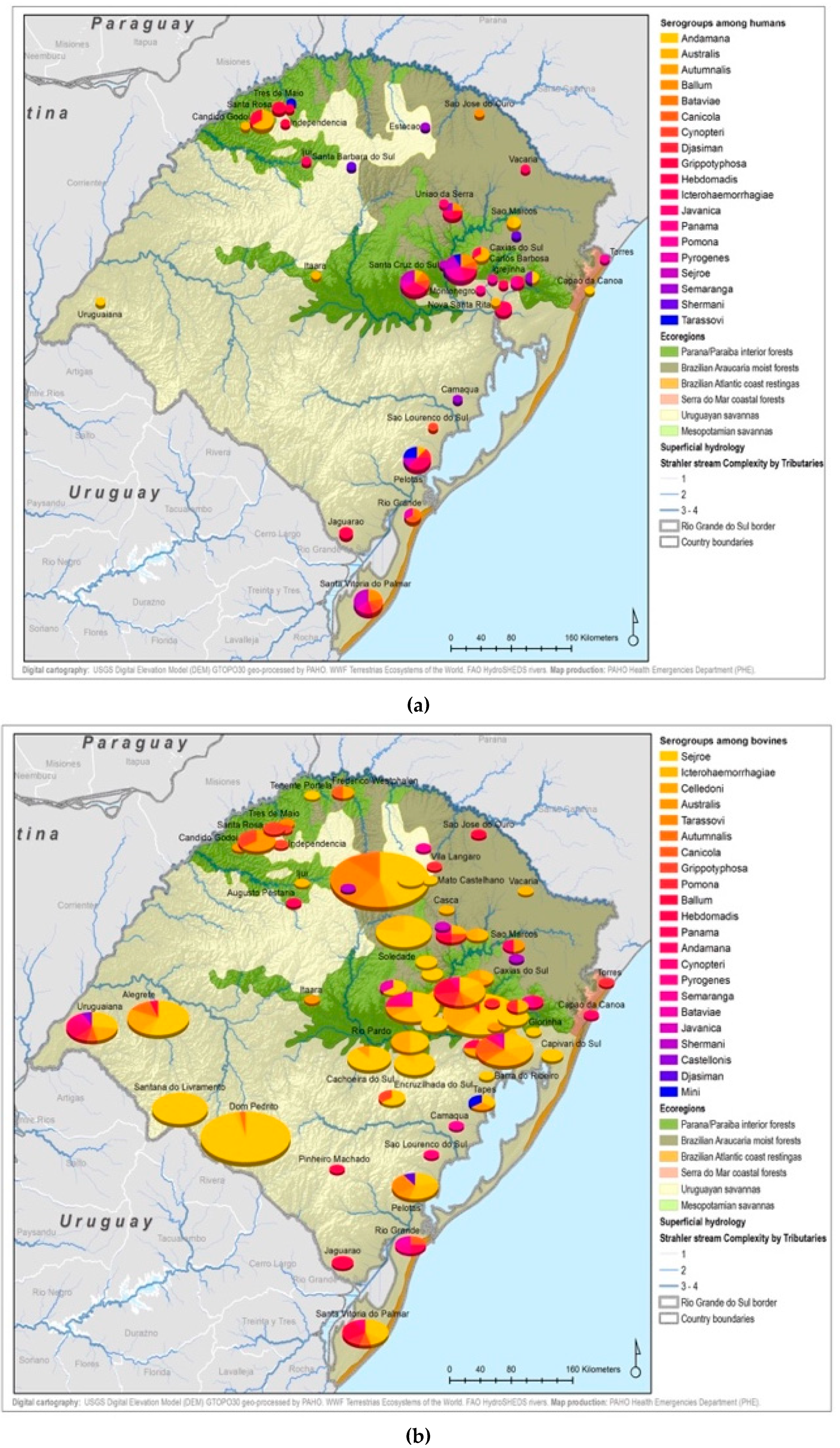
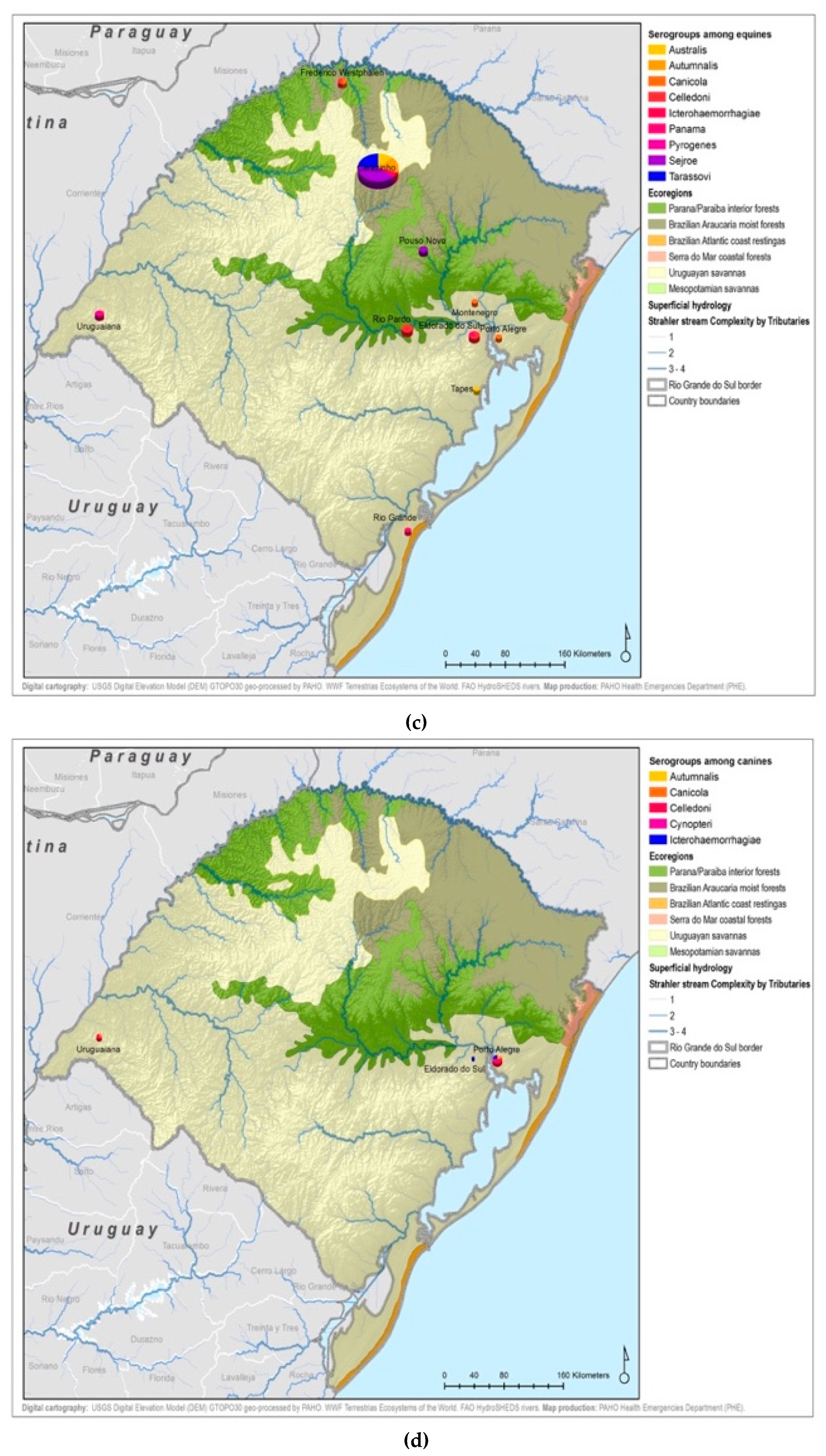

References
- Hotez, P.J.B.M.; Franco-Paredes, C.; Ault, S.K.; Periago, M.R. The neglected tropical diseases of Latin America and the Caribbean: A review of disease burden and distribution and a roadmap for control and elimination. PLoS Negl. Trop. Dis. 2008, 2, e300. [Google Scholar] [CrossRef] [PubMed]
- WHO. Human Leptospirosis: Guidance for Diagnosis, Surveillance and Control; WHO: Washington, DC, USA, 2003; Available online: http://www.who.int/iris/handle/10665/42667 (accessed on 10 February 2018).
- Costa, F.; Hagan, J.E.; Calcagno, J.; Kane, M.; Torgerson, P.; Martinez-Silveira, M.S.; Stein, C.; Abela-Ridder, B.; Ko, A.I. Global Morbidity and Mortality of Leptospirosis: A Systematic Review. PLoS Negl. Trop. Dis. 2015, 9, e0003898. [Google Scholar] [CrossRef] [PubMed]
- Brazilian Ministry of Health. Leptospirosis Diagnosis and Clinical Management. 2014. Portuguese. Available online: http://portalarquivos.saude.gov.br/images/pdf/2014/dezembro/02/Miolo-manual-Leptospirose-17-9-2014.pdf (accessed on 10 February 2018).
- Pereira, M.M.; Schneider, M.C.; Munoz-Zanzi, C.; Costa, F.; Benschop, J.; Hartskeerl, R.; Martinez, J.; Jancloes, M.; Bertherat, E. A road map for leptospirosis research and health policies based on country needs in Latin America. Rev. Panam. Salud Publica 2017, 41, e131. [Google Scholar]
- Schneider, M.C.; Najera, P.; Pereira, M.M.; Machado, G.; dos Anjos, C.B.; Rodrigues, R.O.; Cavagni, G.M.; Muñoz-Zanzi, C.; Corbellini, L.G.; Leone, M.; et al. Leptospirosis in Rio Grande do Sul, Brazil: An Ecosystem Approach in the Animal-Human Interface. PLoS Negl. Trop. Dis. 2015, 9, e0004095. [Google Scholar] [CrossRef] [PubMed]
- Caldas, E. Experience in Leptospirosis in the Republic of Brazil. In Presentation from the International Meeting of Leptospirosis of Nicaragua; PAHO/WHO: Washington, DC, USA, 2012. [Google Scholar]
- Levett, P.N. Leptospirosis. Clin. Microbiol. Rev. 2001, 14, 296–326. [Google Scholar] [CrossRef] [PubMed]
- Chadsuthi, S.; Bicout, D.J.; Wiratsudakul, A.; Suwancharoen, D.; Petkanchanapong, W.; Modchang, C.; Triampo, W.; Ratanakorn, P.; Chalvet-Monfray, K. Investigation on predominant Leptospira serovars and its distribution in humans and livestock in Thailand, 2010–2015. PLoS Negl. Trop. Dis. 2017, 11, e0005228. [Google Scholar] [CrossRef] [PubMed]
- Martins, G.; Lilenbaum, W. The panorama of animal leptospirosis in Rio de Janeiro, Brazil, regarding the seroepidemiology of the infection in tropical regions. BMC Vet. Res. 2013, 9, 237. [Google Scholar] [CrossRef] [PubMed]
- Flores, B.J.; Pérez-Sánchez, T.; Fuertes, H.; Sheleby-Elías, J.; Múzquiz, J.L.; Jirón, W.; Duttmann, C.; Halaihel, N. A cross-sectional epidemiological study of domestic animals related to human leptospirosis cases in Nicaragua. Acta Trop. 2017, 170, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Sathiyamoorthy, A.; Selvaraju, G.; Palanivel, K.M.; Srinivasan, P. Development of indirect enzyme-linked immunosorbent assay for diagnosis of canine leptospirosis. Vet. World 2017, 10, 530–535. [Google Scholar] [CrossRef] [PubMed]
- OIE. Manual of Diagnostic Tests and Vaccines for Terrestrial Animals. 2017. Available online: http://www.oie.int/en/international-standard-setting/terrestrial-manual/access-online/ (accessed on 15 February 2018).
- Desvars, A.; Naze, F.; Vourc’h, G.; Cardinale, E.; Picardeau, M.; Michault, A.; Bourhy, P. Similarities in Leptospira serogroup and species distribution in animals and humans in the Indian ocean island of Mayotte. Am. J. Trop. Med. Hyg. 2012, 87, 134–140. [Google Scholar] [CrossRef] [PubMed]
- IBGE. 2010. Available online: https://censo2010.ibge.gov.br/. Portuguese (accessed on 4 October 2018).
- Adler, B.; de la Pena Moctezuma, A. Leptospira and leptospirosis. Vet. Microbiol. 2010, 140, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Cole, J.R.; Sulzer, C.R.; Pursell, A.R. Improved microtechnique for the leptospiral microscopic agglutination test. Appl. Microbiol. 1973, 25, 976–980. [Google Scholar] [PubMed]
- Lelu, M.; Munñoz-Zanzi, C.; Higgins, B.; Galloway, R. Seroepidemiology of leptospirosis in dogs from rural and slum communities of Los Rios Region, Chile. BMC Vet. Res. 2015, 11, 31. [Google Scholar] [CrossRef] [PubMed]
- FAO. Terrestrial Ecoregions of the World. Available online: http://www.fao.org/land-water/land/land-governance/land-resources-planning-toolbox/category/details/en/c/1036295/ (accessed on 4 October 2018).
- King, L.J.; Anderson, L.R.; Blackmore, C.G.; Blackwell, M.J.; Lautner, E.A.; Marcus, L.C.; Meyer, T.E.; Monath, T.P.; Nave, J.E.; Ohle, J.; et al. One Health Initiative Task Force Report. J. Am. Vet. Med. Assoc. 2008, 233, 259–261. [Google Scholar] [CrossRef] [PubMed]
- Silva, F.J.; Santos, C.E.P.; Glaucenyra, C.P.S.; Santos, R.F.; Curci, V.C.M.; Mathias, L.A. The importance of Leptospira interrogans serovars Icterohaemorrhagiae and Canicola in coastal zone and in southern fields of Rio Grande do Sul, Brazil. Pesq. Vet. Bras. 2014, 34, 34–38. [Google Scholar] [CrossRef]
- Mesquita, M.; Pellegrini, D.C.P.; Simoes Pires Neto, J.A.; Reis, G.R.; Medeiros, C.; Corbellini, L.G. Analise de serie temporal para avaliação do perfil sorológico da leptospirose bovina no estado do Rio Grande do Sul de 1996 a 2006. Arq. Inst. Biol. 2010, 77, 381–387. [Google Scholar]
- Herrmann, G.P.; Lage, A.P.; Moreira, E.C.; Haddad, J.P.A.; Renato de Resende, J.; Rodrigues, R.O.; Leite, R.C. Seroprevalence of agglutinins anti-Leptospira spp. in sheep from the Southeast and Southwest Mesoregions of the State of Rio Grande do Sul, Brazil. Cienc. Rural 2004, 34, 443–448. [Google Scholar] [CrossRef]
- Khairani-Bejo, S.; Bahaman, A.R.; Zamri-Saad, M.; Mutalib, A.R. The Survival of Leptospirosis interrogans Serovar Hardjo in the Malaysian Environment. J. Am. Vet. Adv. 2004, 3, 123–129. [Google Scholar]
- Durski, K.N.; Jancloes, M.; Chowdhary, T.; Bertherat, E. A Global, Multi-Disciplinary, Multi-Sectorial Initiative to Combat Leptospirosis: Global Leptospirosis Environmental Action Network (GLEAN). Int. J. Environ. Res. Public Health 2014, 11, 6000–6008. [Google Scholar] [CrossRef] [PubMed]
- Martins, G.; Hamond, C.; Leite, R.C.-K.; Silva, A.; Ferreira, A.; Brandaão, F.; Oliveira, F.; Lilenbaum, W. Leptospirosis as the most frequent infectious disease impairing productivity in small ruminants in Rio de Janeiro, Brazil. Trop. Anim. Health Prod. 2012, 44, 773–777. [Google Scholar] [CrossRef] [PubMed]
- MAPA Ministeério da Agricultura, Pecuaária e Abastecimento Brasil. Relacaão de Produtos de Uso Veterinaário Licenciados; 2010. Available online: http://www.agricultura.gov.br/pls/portal/docs/PAGE/MAPA/SERVICOS/CPVNOVO/PRODUTOSUSOVET/RELA%C7%C3O%20DE%20PRODUTOS%20COM%20REGISTRO (accessed on 2 September 2010).
- Srikram, A.; Zhang, K.; Bartpho, T.; Lo, M.; Hoke, D.E.; Sermswan, R.W.; Adler, B.; Murray, G.L. Cross-protective immunity against leptospirosis elicited by a live, attenuated lipopolysaccharide mutant. J. Infect. Dis. 2011, 203, 870–879. [Google Scholar] [CrossRef] [PubMed]
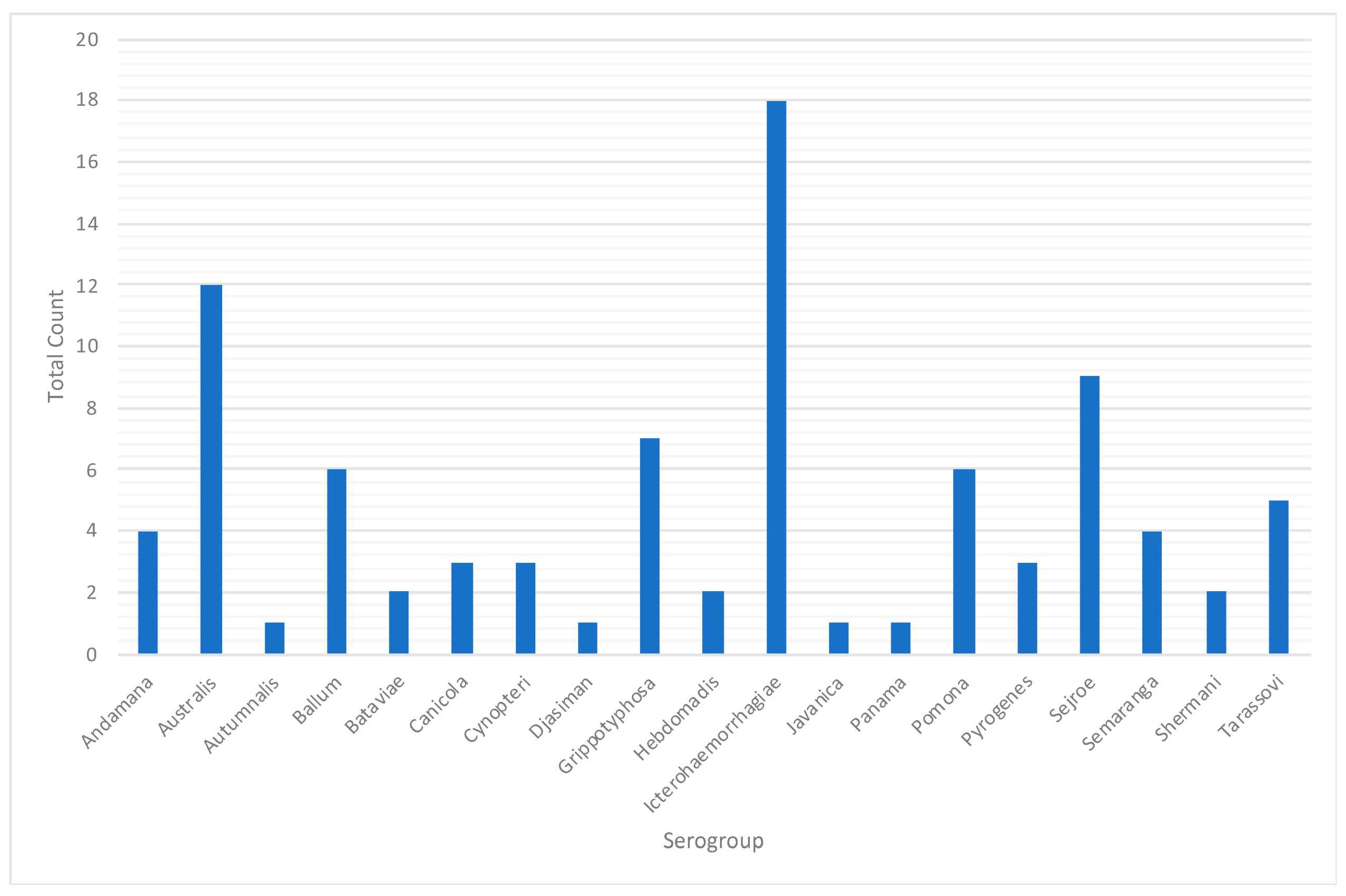
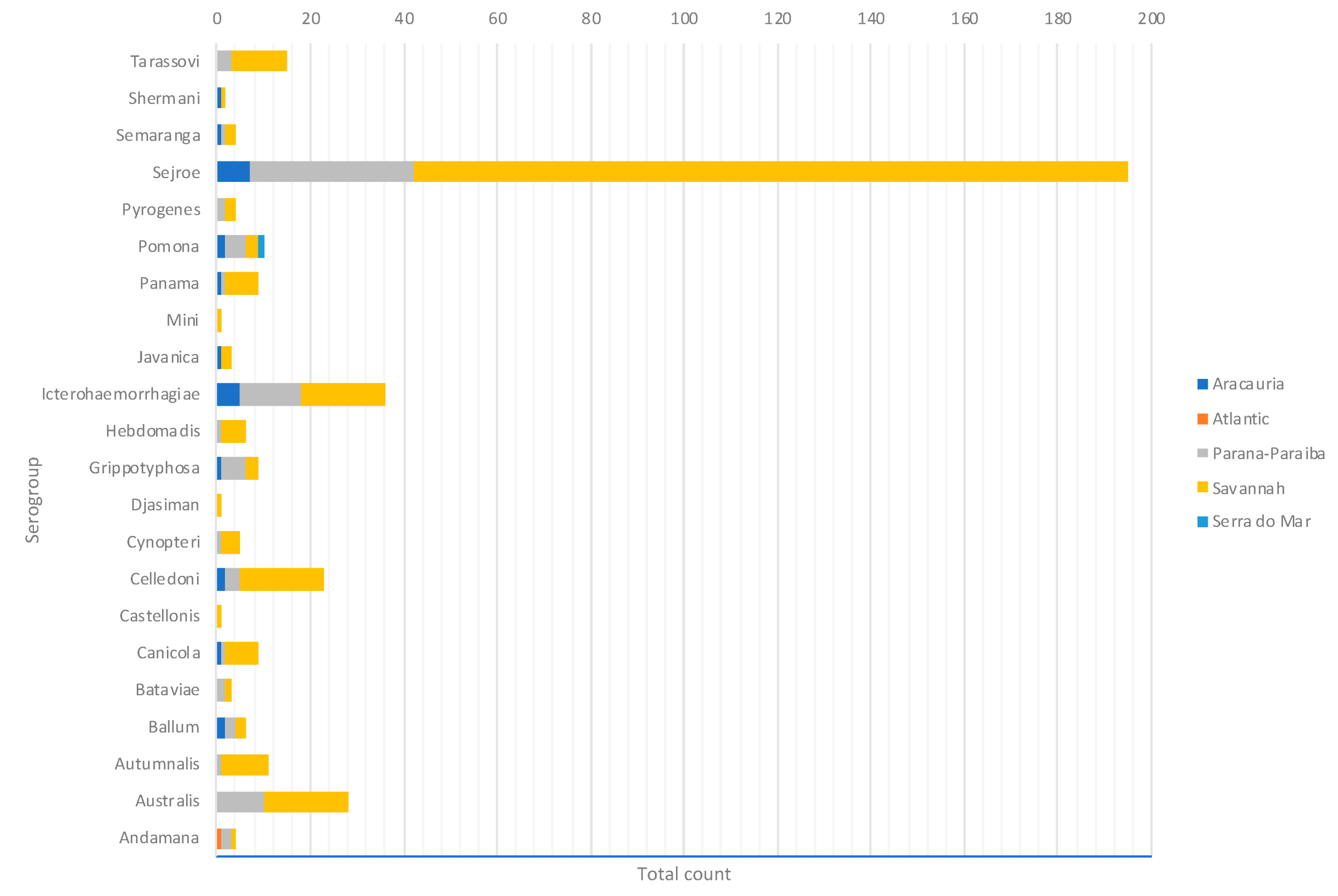
| Serogroup | Species | |||||
|---|---|---|---|---|---|---|
| n (% Serogroup Total) | Total n (% Total) | |||||
| Human | Bovine | Canine | Equine | Others | ||
| Andamana | 4 (100) | - | - | - | - | 4 (1.04) |
| Australis | 12 (42.86) | 7 (25.00) | 1 (3.57) | 8 (28.57) | - | 28 (7.27) |
| Autumnalis | 1 (9.09) | 3 (27.27) | 1 (9.09) | 6 (54.55) | - | 11 (2.86) |
| Ballum | 6 (100) | - | - | - | - | 6 (1.56) |
| Bataviae | 2 (66.67) | 1 (33.33) | - | - | - | 3 (0.78) |
| Canicola | 3 (33.33) | - | 3 (33.33) | 3 (33.33) | - | 9 (2.34) |
| Castellonis | - | 1 (100) | - | - | - | 1 (0.26) |
| Celledoni | - | 9 (39.13) | 6 (26.09) | 7 (30.43) | 1 (4.35) | 23 (5.97) |
| Cynopteri | 3 (60.00) | - | 2 (40.00) | - | - | 5 (1.30) |
| Djasiman | 1 (100) | - | - | - | - | 1 (0.26) |
| Grippotyphosa | 7 (77.78) | 2 (22.22) | - | - | - | 9 (2.34) |
| Hebdomadis | 2 (33.33) | 4 (66.67) | - | - | - | 6 (1.56) |
| Icterohaemorrhagiae | 18 (50.00) | 10 (27.78) | 4 (11.11) | 2 (5.56) | 2 (5.56) | 36 (9.35) |
| Javanica | 1 (33.33) | 1 (33.33) | - | 1 (33.33) | - | 3 (0.78) |
| Mini | - | 1(100) | - | - | - | 1 (0.26) |
| Panama | 1 (11.11) | 2 (22.22) | 2 (22.22) | 4 (44.44) | - | 9 (2.34) |
| Pomona | 6 (60.00) | 4 (40.00) | - | - | - | 10 (2.60) |
| Pyrogenes | 3 (75.00) | - | - | 1 (25.00) | - | 4 (1.04) |
| Sejroe | 9 (4.62) | 142 (72.82) | - | 19 (9.74) | 25 (12.82) | 195 (50.65) |
| Semaranga | 4 (100) | - | - | - | - | 4 (1.04) |
| Shermani | 2 (100) | - | - | - | - | 2 (0.52) |
| Tarassovi | 5 (33.33) | - | - | 10 (66.66) | - | 15 (3.90) |
| TOTAL | 90 | 187 | 19 | 61 | 28 | 385 |
| Variable | N (Total %) |
|---|---|
| Occupation | n = occupation |
| Livestock farmer | 2 (2.22) |
| Mason | 3 (3.33) |
| Agriculture producer | 3 (3.33) |
| Rice farmer | 4 (4.44) |
| Part-time agricultural hire | 5 (5.56) |
| Truck driver | 6 (6.67) |
| Agricultural worker | 12 (13.33) |
| Not mentioned | 55 (61.11) |
| Reported exposure risk factors | n = risk Factors |
| Water tank | 7 (7.78) |
| Sewage | 12 (13.33) |
| Direct contact rodents | 27 (30.00) |
| Water, mud, flooding | 31 (34.44) |
| Rubbish/rubble | 32 (35.56) |
| Proximity to river/stream/dam | 36 (40.00) |
| Grain/food storage | 37 (41.11) |
| Wasteland | 41 (45.56) |
| Planting/harvesting | 49 (54.44) |
| Animal breeding | 65 (72.22) |
| Local rodents | 86 (95.56) |
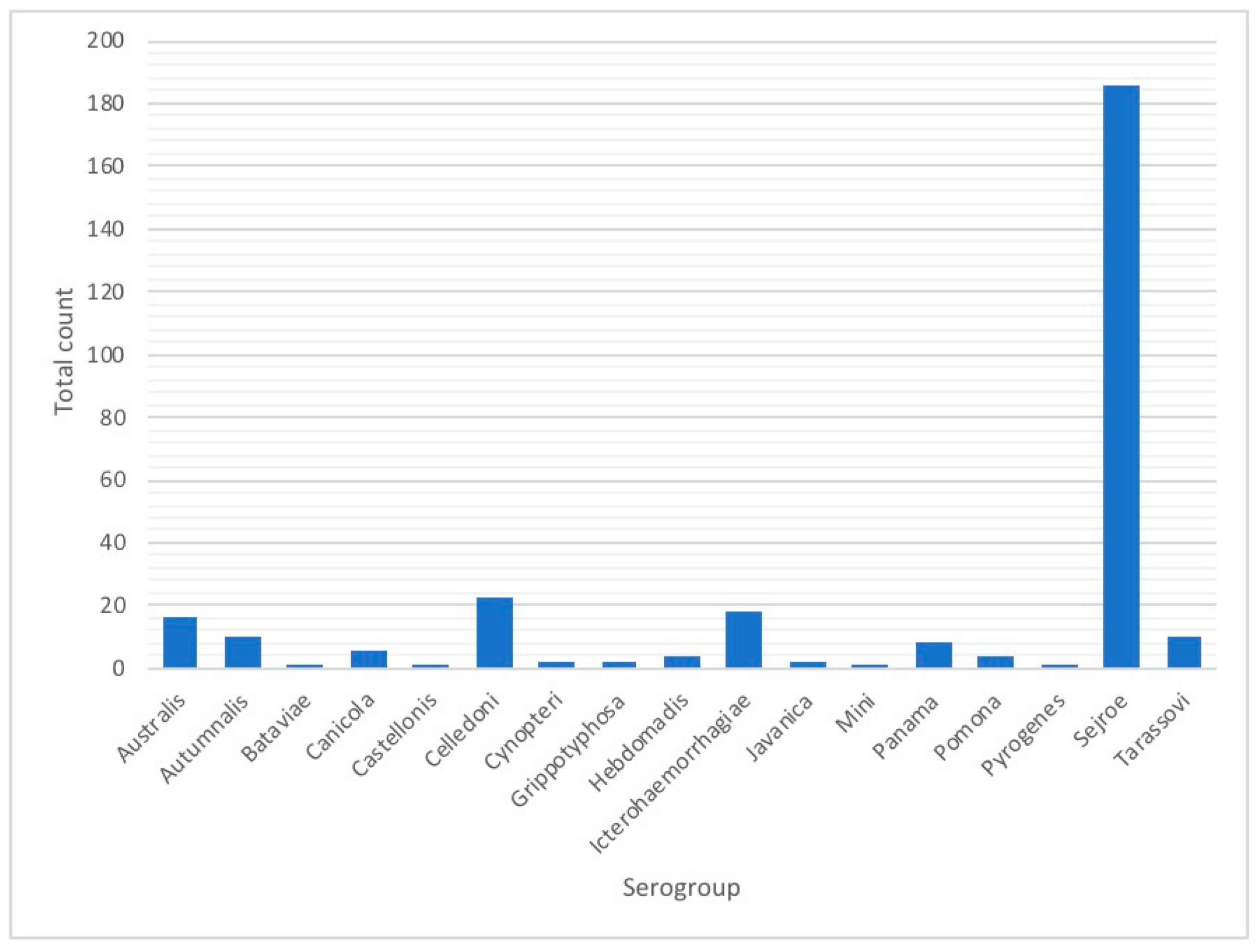
| Variable | Ecoregions | |||||
|---|---|---|---|---|---|---|
| n (% Species by Ecoregion) | ||||||
| Araucaria | Atlantic | Parana-Paraiba | Savannah | Serra do Mar | Total | |
| Species | ||||||
| Human | 8 (33.33) | 1 (100) | 46 (52.87) | 34 (12.5) | 1 (100) | 90 (23.38) |
| Bovine | 11 (45.83) | - | 33 (37.93) | 143 (52.57) | - | 187 (48.57) |
| Equine | 2 (8.33) | - | 5 (5.75) | 54 (19.85) | - | 61 (15.84) |
| Canine | - | - | - | 19 (6.99) | - | 19 (4.94) |
| Others | 3 (12.50) | - | 3 (3.45) | 22 (8.09) | - | 28 (7.27) |
| Serogroups | ||||||
| Andamana | - | 1 (100) | 2 (2.30) | 1 (0.37) | - | 4 (1.04) |
| Australis | - | - | 10 (11.49) | 18 (6.12) | - | 28 (7.27) |
| Autumnalis | - | - | 1 (1.15) | 10 (3.68) | - | 11 (2.86) |
| Ballum | 2 (8.33) | - | 2 (2.30) | 2 (0.74) | - | 6 (1.56) |
| Bataviae | - | - | 2 (2.30) | 1 (0.37) | - | 3 (0.78) |
| Canicola | 1 (4.17) | - | 1 (1.15) | 7 (2.57) | - | 9 (2.34) |
| Castellonis | - | - | - | 1 (0.37) | - | 1 (0.26) |
| Celledoni | 2 (8.33) | - | 3 (3.45) | 18 (6.62) | - | 23 (5.97) |
| Cynopteri | - | - | 1 (1.15) | 4 (1.47) | - | 5 (1.30) |
| Djasiman | - | - | - | 1 (0.37) | - | 1 (0.26) |
| Grippotyphosa | 1 (4.17) | - | 5 (5.75) | 3 (1.10) | - | 9 (2.34) |
| Hebdomadis | - | - | 1 (1.15) | 5 (1.84) | - | 6 (1.56) |
| Icterohaemorrhagiae | 5 (20.83) | - | 13 (14.94) | 18 (6.62) | - | 36 (9.35) |
| Javanica | 1 (4.17) | - | - | 2 (0.74) | - | 3 (0.78) |
| Mini | - | - | - | 1 (0.37) | - | 1 (0.26) |
| Panama | 1 (4.17) | - | 1 (1.15) | 7 (2.57) | - | 9 (2.34) |
| Pomona | 2 (8.33) | - | 4 (4.60) | 3 (1.10) | 1 (100) | 10 (2.60) |
| Pyrogenes | - | - | 2 (2.30) | 2 (0.74) | - | 4 (1.04) |
| Sejroe | 7 (29.17) | - | 35 (40.23) | 153 (56.25) | - | 195 (50.65) |
| Semaranga | 1 (4.17) | - | 1 (1.15) | 2 (0.74) | - | 4 (1.04) |
| Shermani | 1 (4.17) | - | - | 1 (0.37) | - | 2 (0.52) |
| Tarassovi | - | - | 3 (3.45) | 12 (4.41) | - | 15 (3.90) |
| TOTAL | 24 | 1 | 87 | 272 | 1 | 385 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polo, N.; Machado, G.; Rodrigues, R.; Nájera Hamrick, P.; Munoz-Zanzi, C.; Pereira, M.M.; Bercini, M.; Timm, L.N.; Schneider, M.C. A One Health Approach to Investigating Leptospira Serogroups and Their Spatial Distributions among Humans and Animals in Rio Grande do Sul, Brazil, 2013–2015. Trop. Med. Infect. Dis. 2019, 4, 42. https://doi.org/10.3390/tropicalmed4010042
Polo N, Machado G, Rodrigues R, Nájera Hamrick P, Munoz-Zanzi C, Pereira MM, Bercini M, Timm LN, Schneider MC. A One Health Approach to Investigating Leptospira Serogroups and Their Spatial Distributions among Humans and Animals in Rio Grande do Sul, Brazil, 2013–2015. Tropical Medicine and Infectious Disease. 2019; 4(1):42. https://doi.org/10.3390/tropicalmed4010042
Chicago/Turabian StylePolo, Noemi, Gustavo Machado, Rogerio Rodrigues, Patricia Nájera Hamrick, Claudia Munoz-Zanzi, Martha Maria Pereira, Marilina Bercini, Loeci Natalina Timm, and Maria Cristina Schneider. 2019. "A One Health Approach to Investigating Leptospira Serogroups and Their Spatial Distributions among Humans and Animals in Rio Grande do Sul, Brazil, 2013–2015" Tropical Medicine and Infectious Disease 4, no. 1: 42. https://doi.org/10.3390/tropicalmed4010042
APA StylePolo, N., Machado, G., Rodrigues, R., Nájera Hamrick, P., Munoz-Zanzi, C., Pereira, M. M., Bercini, M., Timm, L. N., & Schneider, M. C. (2019). A One Health Approach to Investigating Leptospira Serogroups and Their Spatial Distributions among Humans and Animals in Rio Grande do Sul, Brazil, 2013–2015. Tropical Medicine and Infectious Disease, 4(1), 42. https://doi.org/10.3390/tropicalmed4010042







