Baseline Mapping of Schistosomiasis and Soil Transmitted Helminthiasis in the Northern and Eastern Health Regions of Gabon, Central Africa: Recommendations for Preventive Chemotherapy
Abstract
1. Introduction
2. Materials and Methods
2.1. Authorization and Ethical Assessment
2.2. Study Area
2.3. Study Type, Period and Population
2.4. Selection and Location of Schools
2.5. Schoolchildren Sampling
2.6. Sample Collection and Parasitological Examination
2.7. Data Analysis
3. Results
3.1. Characteristics of Sampling
3.2. Prevalence
3.2.1. Combined Schistosomiasis and Soil Transmitted Helminthiasis
3.2.2. Combined Schistosomiasis
3.2.3. Schistosomiasis Haematobium
3.2.4. Other Schistosomiasis
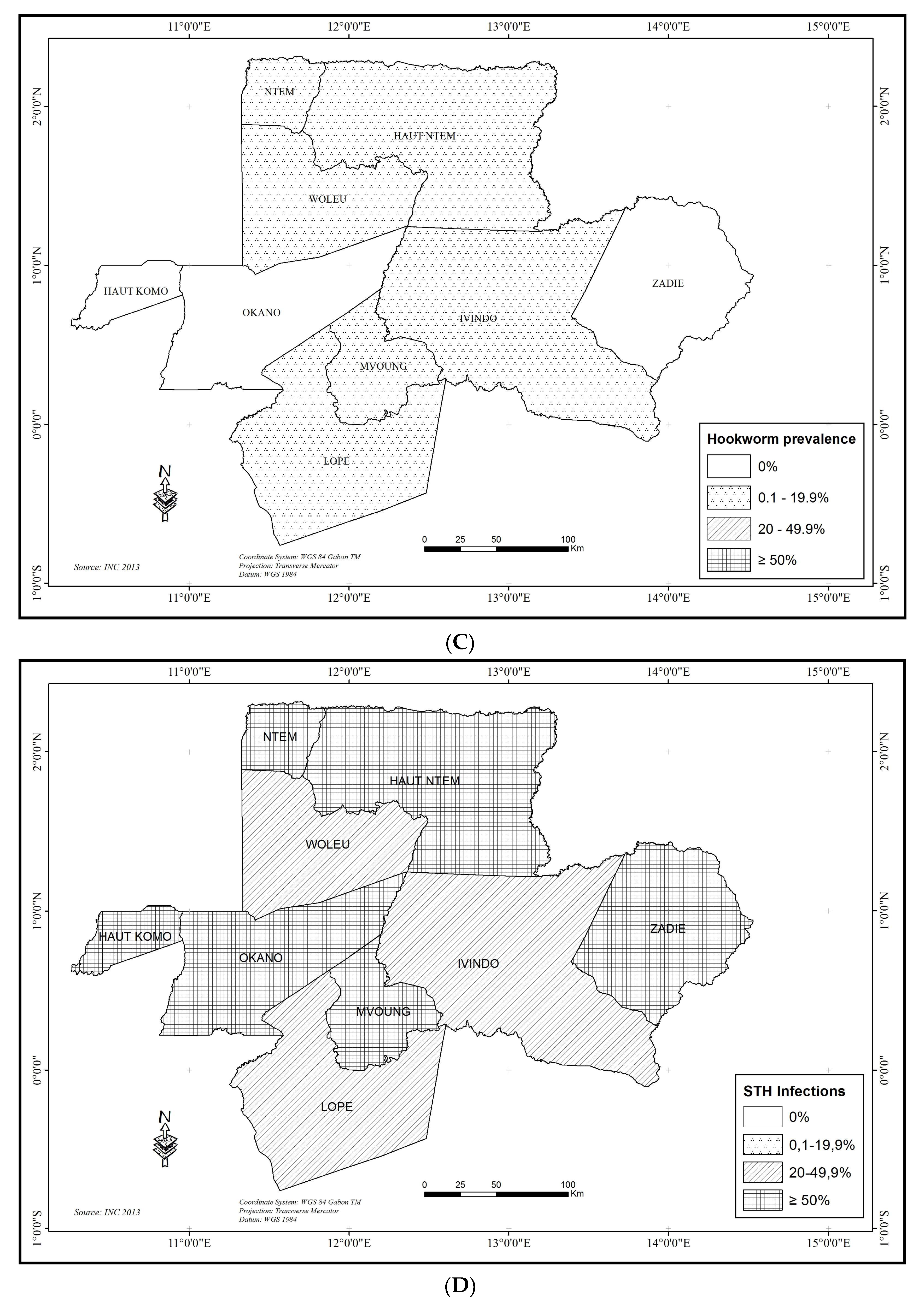
3.2.5. Combined Soil Transmitted Helminthiasis (STH)
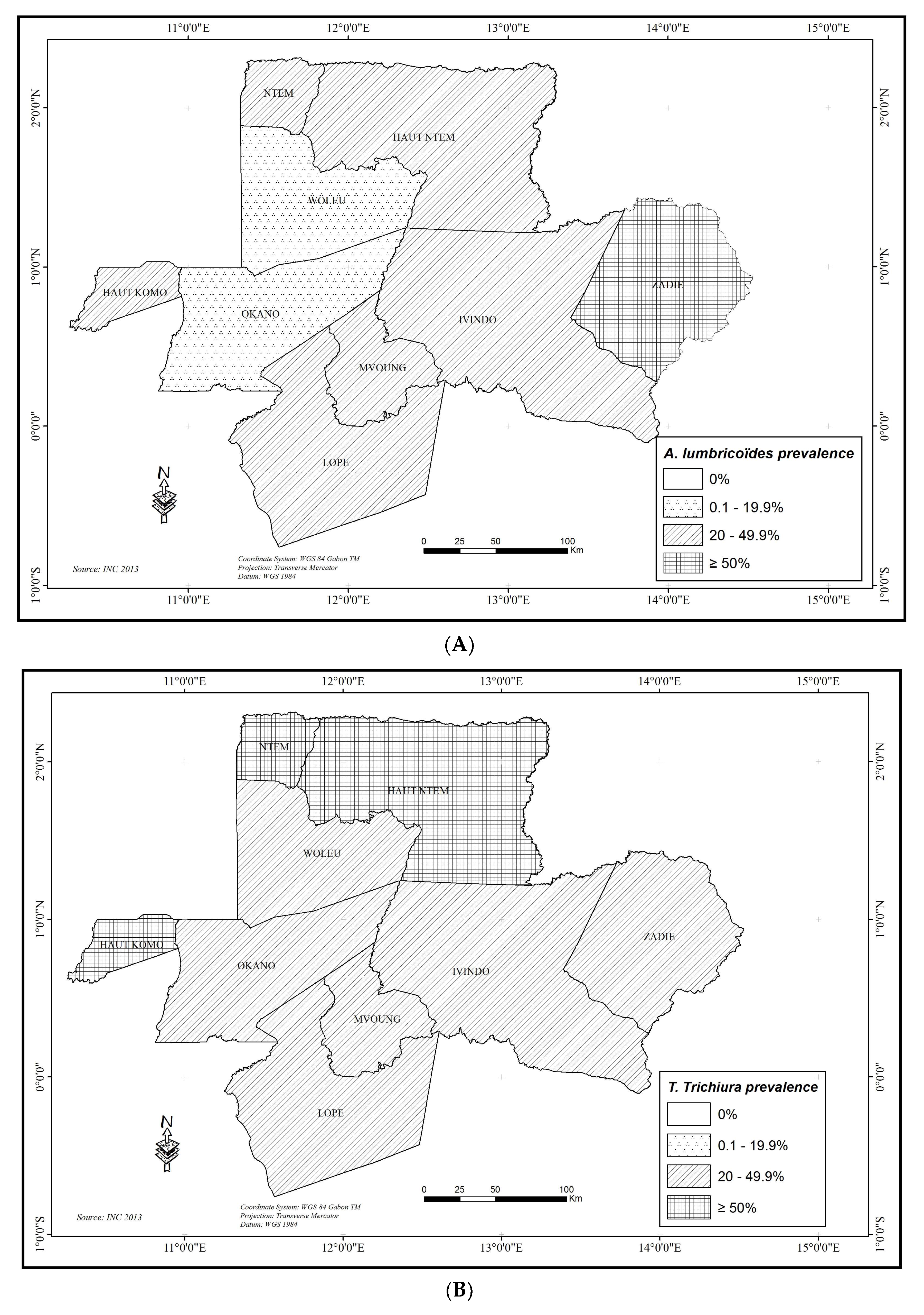
3.2.6. Ascaris lumbricoides
3.2.7. Trichuris trichiura
3.2.8. Hookworms
3.3. Intensity of Infection
3.3.1. Schistosomiasis
3.3.2. Ascaris lumbricoides
3.3.3. Trichuris trichiura
3.3.4. Hookworm
3.4. Community Diagnosis and Recommended Treatment Strategies
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hotez, P.J.; Kamath, A. Neglected tropical diseases in sub-Saharan Africa: Review of their prevalence, distribution, and disease burden. PLoS Negl. Trop. Dis. 2009, 3, e412. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Schistosomiasis and soil-transmitted helminth infections—preliminary estimates of the number of children treated with albendazole or mebendazole. Wkly. Epidemiol. Rec. 2006, 81, 145–163. [Google Scholar]
- Steinmann, P.; Keiser, J.; Bo, R.; Tanner, M.; Utzinger, J. Schistosomiasis and water resources development: Systematic review, meta-analysis, and estimates of people at risk. Lancet Infect. Dis. 2006, 6, 411–425. [Google Scholar] [CrossRef]
- WHO. World Health Organization Schistosomiasis Fact Sheet; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- Adenowoa, A.F.; Oyinloyea, B.E.; Ogunyinkaa, B.I.; Kappo, A.P. Impact of human schistosomiasis in sub-Saharan Africa. Braz. J. Infect. Dis. 2015, 19, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Kane, R.A.; Southgate, V.R.; Rollinson, D.; Littlewood, D.T.J.; Lockyer, A.E.; Pagès, J.R.; Tchuem Tchuente, L.A.; Jourdane, J. A phylogeny based on three mitochondrial genes supports the division of Schistosoma intercalatum into two separate species. Parasitology 2003, 127, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Pagès, J.R.; Jourdane, J.; Southgate, V.R.; Tchuem Tchuente, L.A. Reconnaissance de deux espèces jumelles au sein du taxon Schistosoma intercalatum Fisher, 1934, agent de la schistosomose humaine rectale en Afrique. Description de Schistosoma guineensis n. sp. In Taxonomy, Ecology and Evolution of Metazoan Parasites; tome II; Combes, C., Jourdane, J., Eds.; Presses Universitaire de Perpignan: Perpignan, France, 2003; pp. 139–146. [Google Scholar]
- WHO. Schistosomiasis: Progress Report 2001–2011 and Strategic Plan 2012–2020; WHO/HTM/NTD/PTC; World Health Organization: Geneva, Switzerland, 2013; p. 2. [Google Scholar]
- WHO. Soil-Transmitted Helminthiases: Eliminating Soil-Transmitted Helminthiases as a Public Health Problem in Children: Progress Report 2001–2010 and Strategic Plan 2011–2020; WHO/HTM/NTD/PTC; World Health Organization: Geneva, Switzerland, 2012; p. 4. [Google Scholar]
- WHO. Deworming for Health and Development; Report of the Third Global Meeting of the Partners for Parasite Control; World Health Organization: Geneva, Switzerland, 2005. [Google Scholar]
- Brooker, S.; Clements, A.C.A.; Bundy, D.A.P. Global epidemiology, ecology and control of soil-transmitted helminth infections. Adv. Parasitol. 2006, 62, 221–261. [Google Scholar] [PubMed]
- Mabika-Mamfoumbi, M.; Moussavou-Boussougou, M.N.; Nzenze-Afene, S.; Owono-Medang, M.; Bouyou-Okotet, M.; Kendzo, E.; Kombila, M. Prevalence evaluation of intestinal parasites in rural and sub-urban area in Gabon. Bull. Med. Owendo 2009, 12, 85–88. [Google Scholar]
- Schistosomiasis and Soil-Transmitted Helminth Infections. Fifty-fourth World Health Assembly: Resolution WHA54.192001. 2001. Available online: http://apps.who.int/gb/archive/pdf_fi les/ WHA54/ea54r19.pdf (accessed on 12 April 2018).
- Namwanje, H.; Kabatereine, N.; Olsen, A. A randomised controlled clinical trial on the safety of co-administration of albendazole, ivermectin and praziquantel in infected schoolchildren in Uganda. Trans. R. Soc. Trop. Med. Hyg. 2011, 105, 181–188. [Google Scholar] [CrossRef] [PubMed]
- WHO. Preventive Chemotherapy in Human Helminthiasis: Coordinated Use of Anthelminthic Drugs in Control Interventions; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- Kolaczinski, J.; Kabatereine, N.B.; Onapa, A.; Ndyomugyengi, R.; Kakmebo, A.S.; Brooker, S. Neglected tropical diseases in Uganda: The prospect and challenge of integrated control. Trend Parasitol. 2007, 23, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Hunter, J.M.; Rey, L.; Chu, K.Y.; Adekolu-John, E.O.; Mott, K.E. Parasitic Diseases in Water Resources Development: The Need for Intersectoral Collaboration; World Health Organization: Geneva, Switzerland, 1993. [Google Scholar]
- Brooker, S.; Micheal, E. The potential of geographical information systems and remote sensing in the epidemiology and control of human heliminths infections. Adv. Parasitol. 2000, 47, 246–288. [Google Scholar]
- Direction Générale de la Statistique. Résultats globaux du Recensement Général de la Population et des Logements de 2013 du Gabon (RGPL-2013). Libreville, Publication de la Direction Générale de la Statique (DGS). 2015. Available online: https://www.mays-mouissi.com/wp-content/uploads/2016/07/Recensement-general-de-la-population-et-des-logements-de-2013-RGPL.pdf (accessed on 01 December 2017).
- Peters, P.A.; Mahmoud, A.A.; Warren, K.S.; Ouma, J.H.; Siongok, T.K. Field studies of a rapid, accurate means of quantifying Schistosoma haematobium eggs in urine samples. Bull. World Health Organ. 1976, 54, 159–162. [Google Scholar] [PubMed]
- Coulibaly, J.T.; Knopp, S.; N’Guessan, N.A.; Silue, K.D.; Furst, T.; Lohourignon, L.K.; Brou, J.K.; N’Gbesso, Y.K.; Vounatsou, P.; N’Goran, E.K.; et al. Accuracy of Urine Circulating Cathodic Antigen (CCA) Test for Schistosoma mansoni diagnosis in different settings of Côte d’Ivoire. PLoS Negl. Trop. Dis. 2011, 5, e1384. [Google Scholar] [CrossRef] [PubMed]
- Katz, N.; Chaves, A.; Pellegrino, J. A simple device for quantitative stool thick-smear technique in Schistosomiasis mansoni. Rev. Inst. Med. Trop. Sao Paulo 1972, 14, 397–400. [Google Scholar] [PubMed]
- Nkengazong, L.; Njiokou, F.; Asonganyi, T. Two years impact of single praziquantel treatment on infection of urinary schistosomiasis in the Barombi Kotto focus, Cameroon. Int. J. Biosci. 2013, 3, 98–107. [Google Scholar]
- Mintsa Nguema, R.; Mengue Ngou-Milama, K.; Kombila, M.; Rechard-Lenoble, D.; Tisseyre, P.; Ibikounlé, M.; Moné, H.; Mouahid, G. Morphometric and molecular characterizations of schistosome populations in Estuaire province Gabon. J. Helminthol. 2010, 84, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Tchuem Tchuenté, L.A.; Kamwa Ngassam, R.I.; Sumo, L.; Ngassam, P.; Dongmo Noumedem, C.; Luogbou Nzu, D.D.; Dankoni, E.; Kenfack, C.M.; Gipwe, N.F.; Akame, J.; et al. Mapping of Schistosomiasis and Soil-Transmitted Helminthiasis in the Regions of Centre, East and West Cameroon. PLoS Negl. Trop. Dis. 2012, 6, e1553. [Google Scholar] [CrossRef] [PubMed]
- Tchuem Tchuenté, L.A.; Dongmo Noumedem, C.; Ngassam, P.; Mérimé Kenfack, C.; Feussom Gipwe, N.; Dankoni, E.; Merrime Kenfack, C.; Feussom Gipwe, N.; Akame, J.; Tarini, A.; et al. Mapping of schistosomiasis and soil transmitted helminthiasis in the regions of Littoral, North-West, South and South-West Cameroon and recommendations for treatment. BMC Infect. Dis. 2013, 13, 602. [Google Scholar] [CrossRef] [PubMed]
- Woolhouse, M.E.J.; Chandiwana, S.K. Spatial and temporal heterogeneity in the population dynamics of Bulinus globosus and Biomphalaria pfeifferi and in the epidemiology of their infection with Schistosomes. Parasitology 1989, 98, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Brooker, S. Schistosomes, snails and satellites. Acta Trop. 2002, 82, 207–214. [Google Scholar] [CrossRef]
- Brooker, S.; Hay, S.I.; Issae, W.; Hall, A.; Kihamia, C.M.; Lwambo, N.J.S.; Wint, W.; Rogers, D.J.; Bundy, D.A.P. Predicting the distribution of urinary schistosomiasis in Tanzania using satellite sensor data. Trop. Med. Int. Health 2001, 6, 998–1007. [Google Scholar] [CrossRef] [PubMed]
- Brooker, S.; Hay, S.I.; Tchuem Tchuenté, L.A.; Ratard, R. Modelling human helminth distributions for planning disease control in Cameroon. Photogramm. Eng. Remote Sens. 2002, 68, 175–179. [Google Scholar]
- Brown, S.D. Freshwater Snail of Africa and Their Medical Importance; Taylor and Francis: London, UK, 1994. [Google Scholar]
- Greer, G.J.; Mimpfoundi, R.; Malek, E.A.; Joky, A.; Ngonseu, E.; Ratard, R.C. Human schistosomiasis in Cameroon II. Distribution of the snail hosts. Am. J. Trop. Med. Hyg. 1990, 6, 573–580. [Google Scholar] [CrossRef]
- Kabatereine, N.B.; Brooker, S.; Tukahebwa, E.M.; Kazibwe, F.; Onapa, A. Epidemiology and geography of Schistosoma mansoni in Uganda: Implications for planning control. Trop. Med. Int. Health 2004, 9, 372–380. [Google Scholar] [CrossRef] [PubMed]
- WHO. Report on the WHO Informal Consultation on Schistosomiasis Control; World Health Organization: Geneva, Switzerland, 1999; p. 2. [Google Scholar]
- Burchard, G.D.; Kern, P. Probable hybridization between S. intercalatum and S. haematobium in Western Gabon. Trop. Geogr. Med. 1985, 37, 119–123. [Google Scholar] [PubMed]
- Richard-Lenoble, D.; Kombila, M.; Duong, T.H.; Gendrel, D. Bilharziose à Schistosoma intercalatum, bilharziose récente et oubliée. Rev. Pratc. 1993, 43, 432–439. [Google Scholar]
- Webster, B.L.; Tchuem Tchuenté, L.A.; Jourdane, J.; Southgate, V.R. The interaction of Schistosoma haematobium and S. guineensis in Cameroon. J. Helminthol. 2005, 79, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Mintsa Nguema, R.; Mve Ondo, B.; Mabika Mamfoumbi, M.; Koumba, A.A.; Bouyou Akotet, M.K.; Kombila, M. Recent Examination for Assessing Epidemiological Status of Schistosoma Mansoni in Plaine Orety, Urban Area of Libreville, Gabon, Central Africa. CAJPH 2018, 4, 81–85. [Google Scholar] [CrossRef]
- Toure, S.; Zhang, Y.; Bosque-Oliva, E.; Ky, C.; Ouedraogo, A.; Koukounariet, A.; Gabrielli, A.F.; Sellin, B.; Websterb, J.P.; Fenwickb, A. Two-year impact of single praziquantel treatment on infection in the national control programme on schistosomiasis in Burkina Faso. Bull. World Health Organ. 2008, 86, 780–787. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.H.; Warren, K.S.; Mahmoud, A.A.F. Morbidity in Schistosomiasis Mansoni in relation to intensity of infection: Study of a community in Kisumu, Kenya. Am. J. Trop. Med. Hyg. 1979, 28, 220–229. [Google Scholar] [PubMed]
- Genming, Z.; Brinkmann, U.K.; Qingwu, J.; Shaoji, Z.; Zhide, L.; Hongchang, Y. The relationship between morbidity and intensity of Schistosomiasis japonicum infection of a community in Jiangxi Province, China. Southeast. Asian J. Trop. Med. Public Health 1977, 28, 245–250. [Google Scholar]
- Utzinger, J.; Becker, S.L.; van Lieshout, L.; van Dam, G.J.; Knopp, S. New diagnostic tools in schistosomiasis. Clin. Microbiol. Infect. 2015, 21, 529–542. [Google Scholar] [CrossRef] [PubMed]
- WHO. Basic Laboratory Methods in Medical Parasitology; World Health Organization: Geneva, Switzerland, 1991. [Google Scholar]
- Braun-Munzinger, R.A.; Southgate, B.A. Repeatability and reproducibility of egg counts of Schistosoma haematobium in urine. Trop. Med. Parasitol. 1992, 43, 149–154. [Google Scholar] [PubMed]
- Lamberton, P.H.L.; Kabatereine, N.B.; Oguttu, D.W.; Fenwick, A.; Webster, J.P. Sensitivity and specificity of multiple Kato-Katz thick smears and a Circulating Cathodic Antigen test for Schistosoma mansoni Diagnosis Pre-and Post-repeated-Praziquantel treatment. PLoS Negl. Trop. Dis. 2014, 8, e3139. [Google Scholar] [CrossRef] [PubMed]
- Van Lieshout, L.; Polderman, A.M.; Deelder, A.M. Immunodiagnosis of schistosomiasis by determination of the circulating antigens CAA and CCA, in particular in individuals with recent or light infections. Acta Trop. 2000, 77, 68–80. [Google Scholar] [CrossRef]
- Obeng, B.B.; Aryeetey, Y.A.; de Dood, C.J.; Amoah, A.S.; Larbi, I.A.; Deelder, A.M.; Yazdanbakhsh, M.; Hartgers, F.C.; Boakye, D.A.; Verweij, J.J.; et al. Application of a circulating-cathodic-antigen (CCA) strip test and real-time PCR, in comparison with microscopy, for the detection of Schistosoma haematobium in urine samples from Ghana. Ann. Trop. Med. Parasitol. 2008, 102, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Van Dam, G.J.; Xu, J.; Bergquist, R.; de Dood, C.J.; Utzinger, J.; Qin, Z.Q.; Guanb, W.; Fengb, T.; Yug, X.L.; Zhoug, J.; et al. An ultra-sensitive assay targeting the circulating anodic antigen for the diagnosis of Schistosoma japonicum in a low-endemic area, People’s Republic of China. Acta Trop. 2015, 141, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Knopp, S.; Corstjens, P.L.A.M.; Koukounari, A.; Cercamondi, C.I.; Ame, S.M.; Ali, S.M.; de Dood, C.J.; Mohammed, K.A.; Utzinger, J.; Rollinson, D.; et al. Sensitivity and specificity of a urine circulating anodic antigen test for the diagnosis of Schistosoma haematobium in low endemic settings. PLoS Negl. Trop. Dis. 2015, 9, e0003752. [Google Scholar] [CrossRef] [PubMed]
- Stothard, J.R.; Kabatereine, N.B.; Tukahebwa, E.M.; Kazibwe, F.; Rollinson, D.; Mathieson, W.; Webster, J.P.; Fenwick, A. Use of circulating cathodic antigen (CCA) dipsticks for detection of intestinal and urinary schistosomiasis. Acta Trop. 2006, 97, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Colley, D.G.; Binder, S.; Campbell, C.; King, C.H.; Tchuem Tchuenté, LA.; N’Goran, E.K. A Five-Country Evaluation of a Point-of-Care Circulating Cathodic Antigen Urine Assay for the Prevalence of Schistosoma mansoni. Am. J. Trop. Med. Hyg. 2013, 88, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Tchuem Tchuenté, L.A.; Kueté Fouodo, C.J.; Kamwa Ngassam, R.I.; Sumo, L.; Dongmo Noumedem, C.; Mérimé Kenfack, C.; Gipwe, N.F.; Dankoni Nana, E.; Stothard, J.R.; Rollinson, D. Evaluation of Circulating Cathodic Antigen (CCA) Urine-Tests for Diagnosis of Schistosoma mansoni Infection in Cameroon. PLoS Negl. Trop. Dis. 2012, 6, e1758. [Google Scholar] [CrossRef] [PubMed]
- Ratard, R.C.; Kouemeni, L.E.; Ekani Bessala, M.M.; Ndamkou, K.N.; Greer, G.J.; Spilsbury, J.; Cline, B.L. Human schistosomiasis in Cameroon. I. Distribution of schistosomiasis. Am. J. Trop. Med. Hyg. 1990, 42, 561–572. [Google Scholar] [CrossRef] [PubMed]
- WHO. Prevention and Control of Schistosomiasis and Soil-Transmitted Helminthiasis; World Health Organisation: Geneva, Switzerland, 2002. [Google Scholar]
- Garba, A.; Campagne, G.; Tassie, J.M.; Barkire, A.; Vera, C.; Sellin, B.; Chippaux, J.P. Evaluation à long terme d’un traitement de masse par praziquantel sur la morbidité due à Schistosoma haematobium dans deux villages hyper-endémiques du Nige. Bull. Soc. Pathol. Exot. 2004, 97, 7–11. [Google Scholar] [PubMed]
- Nsowah-Nuamah, N.N.; Aryeetey, M.E.; Jolayemi, E.T.; Yagatsuma, Y.; Mensah, G.; Dontwi, I.K.; Nkrumah, F.K.; Kojima, S. Predicting the timing of second praziquantel treatment and its effect on reduction of egg counts in southern Ghana. Acta Trop. 2004, 90, 263–720. [Google Scholar] [CrossRef] [PubMed]
- Frenzel, K.; Grigull, L.; Odongo-Aginya, E.; Ndugwa, C.M.; Loroni-Lakwo, T.; Schweigmann, U.; Vester, U.; Spannbrucker, N.; Doehring, E. Evidence for a long-term effect of a single dose of praziquantel on Schistosoma mansoni–induced hepatosplenic lesions in Northern Uganda. Am. J. Trop. Med. Hyg. 1999, 60, 927–931. [Google Scholar] [CrossRef] [PubMed]
- Keizer, J.; Utzinger, J. Efficacity of current drugs against Soil Transmitted Helminthiasis infections: Systematic review and meta-analysis. JAMA 2008, 299, 1937–1948. [Google Scholar]
- Albonico, M.; Bickle, Q.; Ramsan, M.; Montresor, A.; Savioli, L.; Taylor, M. Efficacy of mebendazole and levamisole alone or in combination against intestinal nematode infections after repeated targeted mebendazole treatment in Zanzibar. Bull. World Health Organ. 2003, 81, 343–352. [Google Scholar] [PubMed]



| N | Schistosomiasis | Soil Transmittted Helminthiasis | SCH | STH | SCH-STH | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| S. haematobium | S. mansoni | S. guineensis | A. lumbricoides | T. trichiura | Hookworms | |||||
| Overall | 2245 | 1.65 [1.16–2.27] | 0.04 [0.0–0.25] | 0.04 [0.0–0.25] | 35.59 [33.61–37.59] | 43.74 [41.68–45.82] | 1.43 [0.98–2.01] | 1.69 [1.2–2.32] | 56.08 [54.0–58.15] | 56.57 [54.49–58.63] |
| By region | ||||||||||
| Northern | 1236 | 1.46 [0.87–2.29] | 0.08 [0.0–0.45] | 0.08 [0.0–0.45] | 29.13 * [26.60–31.75] | 52.83 * [50.0–55.65] | 1.62 [0.99–2.49] | 1.54 [0.93–2.39] | 58.09 * [55.29–60.86] | 58.5 * [55.69–61.26] |
| Eastern | 1009 | 1.88 [1.14–2.93] | 0.00 [0.0–0.37] | 0.00 [0.0–0.37] | 43.51 [40.42–46.63] | 32.61 [29.72–35.60] | 1.19 [0.62–2.07] | 1.88 [1.14–2.93] | 53.62 [50.48–56.73] | 54.2 [51.08–57.32] |
| By gender | ||||||||||
| Girl | 1116 | 1.25 [0.69–2.1] | 0.00 [0.0–0.33] | 0.09 [0.0–0.5] | 34.14 [31.36–37.01] | 42.47 [39.55–45.43] | 0.81 [0.37–1.53] | 1.34 [0.75–2.2] | 54.84 [51.87–57.79] | 55.11 [52.13–58.05] |
| Boy | 1129 | 2.04 [1.30–3.04] | 0.09 [0.0–0.49] | 0.00 [0.0–0.49] | 37.02 [34.20–39.92] | 45.00 [42.07–47.95] | 2.04 * [1.30–3.04] | 2.04 [1.30–3.04] | 57.31 [54.36–60.21] | 58.02 [55.08–60.91] |
| By school area | ||||||||||
| urban | 1754 | 1.48 [0.97–2.16] | 0.06 [0.0–0.32] | 0.06 [0.0–0.32] | 30.84 [28.69–33.06] * | 40.59 [38.28–42.93] * | 1.37 [0.88–2.03] | 1.54 [1.02–2.23] | 51.60 [49.23–53.96] * | 52.17 [49.80–54.48] * |
| rural | 491 | 2.24 [1.12–3.97] | 0.00 [0.0–0.75] | 0.00 [0.0–0.75] | 52.55 [48.02–57.04] | 54.99 [50.47–59.45] | 1.63 [0.71–3.19] | 2.24 [11.24–3.97] | 72.10 [67.90–76.02] | 72.51 [68.33–76.41] |
| By school category | ||||||||||
| Public | 1420 | 1.97 [1.31–2.84] | 0.07 [0.0–0.39] | 0.07 [0.0–0.39] | 36.76 [34.25–39.33] | 45.35 [43.74–47.98] * | 1.62 [1.03–2.42] | 2.04 [1.37–2.92] | 57.61 [54.99–60.19] | 58.24 [55.62–60.82] * |
| Private | 825 | 1.09 [0.50–2.06] | 0.00 [0.0–0.45] | 0.00 [0.0–0.45] | 33.58 [30.36–36.91] | 40.97 [37.59–44.41] | 1.09 [0.50–2.06] | 1.09 [0.50–2.06] | 53.46 [49.98–56.90] | 53.82 [50.35–57.26] |
| Number of Species | N | Schistosomiasis | STH | Schistosomiasis-STH |
|---|---|---|---|---|
| 1 | 2245 | 37 | 723 | 718 * |
| 2 | 1 | 528 | 537 | |
| 3 | 0 | 8 | 15 | |
| 4 | 0 | 0 | 0 | |
| 5 | 0 | 0 | 0 | |
| 6 | 0 | 0 | 0 | |
| >6 | 0 | 0 | 0 | |
| Negative | 2207 | 986 | 975 |
| Schistosomiasis | Soil Transmittted Helminthiasis | |||||
|---|---|---|---|---|---|---|
| S. haematobium | S. mansoni | S. guineensis | A. lumbricoides | T. trichiura | Hookworms | |
| Overall | 101.9 ± 45.1 (37) | 72 (1) | 240 (1) | 9586.6 ± 618.3 (799) | 1143.2 ± 97.0 (982) | 618.0 ± 499.6 (32) |
| By region | ||||||
| North | 18.3 ± 8.2 (18) | 72 (1) | 240 (1) | 11433.6 ± 1061.7 (360) | 1395.2 ± 126.6 (653) | 130.8 ± 31.1 (20) |
| East | 181.1 ± 85.6 (19) | - | - | 8071.9 ± 707.3 (439) | 642.9 ± 140.2 (329) | 1430.0 ± 1369.1 (12) |
| By gender | ||||||
| Girl | 65.3 ± 32.4 (14) | - | 240 (1) | 10131.3 ± 1053.6 (381) | 1152.6 ± 147.4 (474) | 1861.3 ± 1853.7 (9) |
| Boy | 124.2 ± 70.9 (23) | 72 (1) | - | 9018.1 ± 688.7 (418) | 1130.1 ± 127.7 (508) | 131.5 ± 35.0 (23) |
| By school area | ||||||
| urban | 109.5 ± 61.0 (26) | 72 (1) | 240 (1) | 9144.6 ± 665.8 (541) | 1139.9 ± 113.9 (712) | 791.0 ± 669.2 (24) |
| rural | 174.7 ± 55.3 (11) | - | - | 10513.4 ± 1313.1 (258) | 1151.7 ± 185.9 (270) | 99.0 ± 46.9 (8) |
| By school category | ||||||
| Public | 73.3 ± 26.3 (28) | 72 (1) | 240 (1) | 8874.4 ± 624.4 (522) | 1193.5 ± 113.1 (644) | 97.0 ± 18.9 (23) |
| Private | 504.9 ± 178.5 (9) | - | - | 10928.7 ± 1340.5 (277) | 1047.3 ± 182.1 (338) | 1949.3 ± 1843.5 (9) |
| Department | Category | MDA Interventions in Schools (Enrolled and Non-Enrolled) | Drug |
|---|---|---|---|
| Schistosomiasis infections | |||
| Woleu | Low prevalence | MDA of SAC twice during primary schooling (once on entry, again on leaving) | Praziquantel |
| Ntem | |||
| Haut-Ntem | |||
| Haut-Okano | |||
| Okano | |||
| Ivindo | |||
| Lopé | |||
| Mvoung | |||
| Zadié | Not endemic | No required MDA. Treatment of individual confirmed cases | |
| Soil Transmitted Helminthiasis infections | |||
| Woleu | Moderate prevalence and moderate intensity | Annual MDA | Mebendazole + Levamisole |
| Ntem | High prevalence | Biannual MDA | |
| Haut-Ntem | |||
| Haut-Komo | |||
| Okano | |||
| Ivindo | Moderate prevalence and moderate intensity | Annual MDA | |
| Lopé | |||
| Mvoung | High prevalence | Biannual MDA | |
| Zadié | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mintsa Nguema, R.; Mavoungou, J.F.; Mengue Me Ngou-Milama, K.; Mabicka Mamfoumbi, M.; Koumba, A.A.; Sani Lamine, M.; Diarra, A.; Nkone Asseko, G.; Mourou, J.R.; Bouyou Akotet, M.K.; et al. Baseline Mapping of Schistosomiasis and Soil Transmitted Helminthiasis in the Northern and Eastern Health Regions of Gabon, Central Africa: Recommendations for Preventive Chemotherapy. Trop. Med. Infect. Dis. 2018, 3, 119. https://doi.org/10.3390/tropicalmed3040119
Mintsa Nguema R, Mavoungou JF, Mengue Me Ngou-Milama K, Mabicka Mamfoumbi M, Koumba AA, Sani Lamine M, Diarra A, Nkone Asseko G, Mourou JR, Bouyou Akotet MK, et al. Baseline Mapping of Schistosomiasis and Soil Transmitted Helminthiasis in the Northern and Eastern Health Regions of Gabon, Central Africa: Recommendations for Preventive Chemotherapy. Tropical Medicine and Infectious Disease. 2018; 3(4):119. https://doi.org/10.3390/tropicalmed3040119
Chicago/Turabian StyleMintsa Nguema, Rodrigue, Jacques F. Mavoungou, Krystina Mengue Me Ngou-Milama, Modeste Mabicka Mamfoumbi, Aubin A. Koumba, Mariama Sani Lamine, Abdoulaye Diarra, Ghislaine Nkone Asseko, Jean R. Mourou, Marielle K. Bouyou Akotet, and et al. 2018. "Baseline Mapping of Schistosomiasis and Soil Transmitted Helminthiasis in the Northern and Eastern Health Regions of Gabon, Central Africa: Recommendations for Preventive Chemotherapy" Tropical Medicine and Infectious Disease 3, no. 4: 119. https://doi.org/10.3390/tropicalmed3040119
APA StyleMintsa Nguema, R., Mavoungou, J. F., Mengue Me Ngou-Milama, K., Mabicka Mamfoumbi, M., Koumba, A. A., Sani Lamine, M., Diarra, A., Nkone Asseko, G., Mourou, J. R., Bouyou Akotet, M. K., Moné, H., Mouahid, G., & Atsame, J. (2018). Baseline Mapping of Schistosomiasis and Soil Transmitted Helminthiasis in the Northern and Eastern Health Regions of Gabon, Central Africa: Recommendations for Preventive Chemotherapy. Tropical Medicine and Infectious Disease, 3(4), 119. https://doi.org/10.3390/tropicalmed3040119





