Integrated Management of Skin NTDs—Lessons Learned from Existing Practice and Field Research
Abstract
1. Introduction
2. Active Surveillance
- Selection of the intervention area: try to collect past data and develop mapping methods to identify co-distribution of cases of skin NTDs
- Training of local healthcare workers both on skin NTDs and common skin diseases
- Treatment: develop protocols on how to manage the different diseases anticipated; be prepared to treat or to refer
- Look for opportunities for integration with other community- or school-based activities, e.g., de-worming, vitamin A and micronutrient supplementation, onchocerciasis and/or lymphatic filariasis control, to gain a synergic effect
- Plan for repeat rounds/follow-up activities, decide on the appropriate intervals.
3. Mass Drug Administration and Prophylaxis
- Careful identification of target populations or case definition
- Identifying intervals between rounds and number of rounds; some diseases have long latent periods which may be difficult to assess
- Obtaining strong, coordinated public-private partnerships, including pharmaceutical companies
- Assessment of secondary effects on other diseases (e.g., leprosy vs. tuberculosis), including increasing the risk of drug resistance
- Addressing the issues of stigma and discrimination if implementing for contact cases.
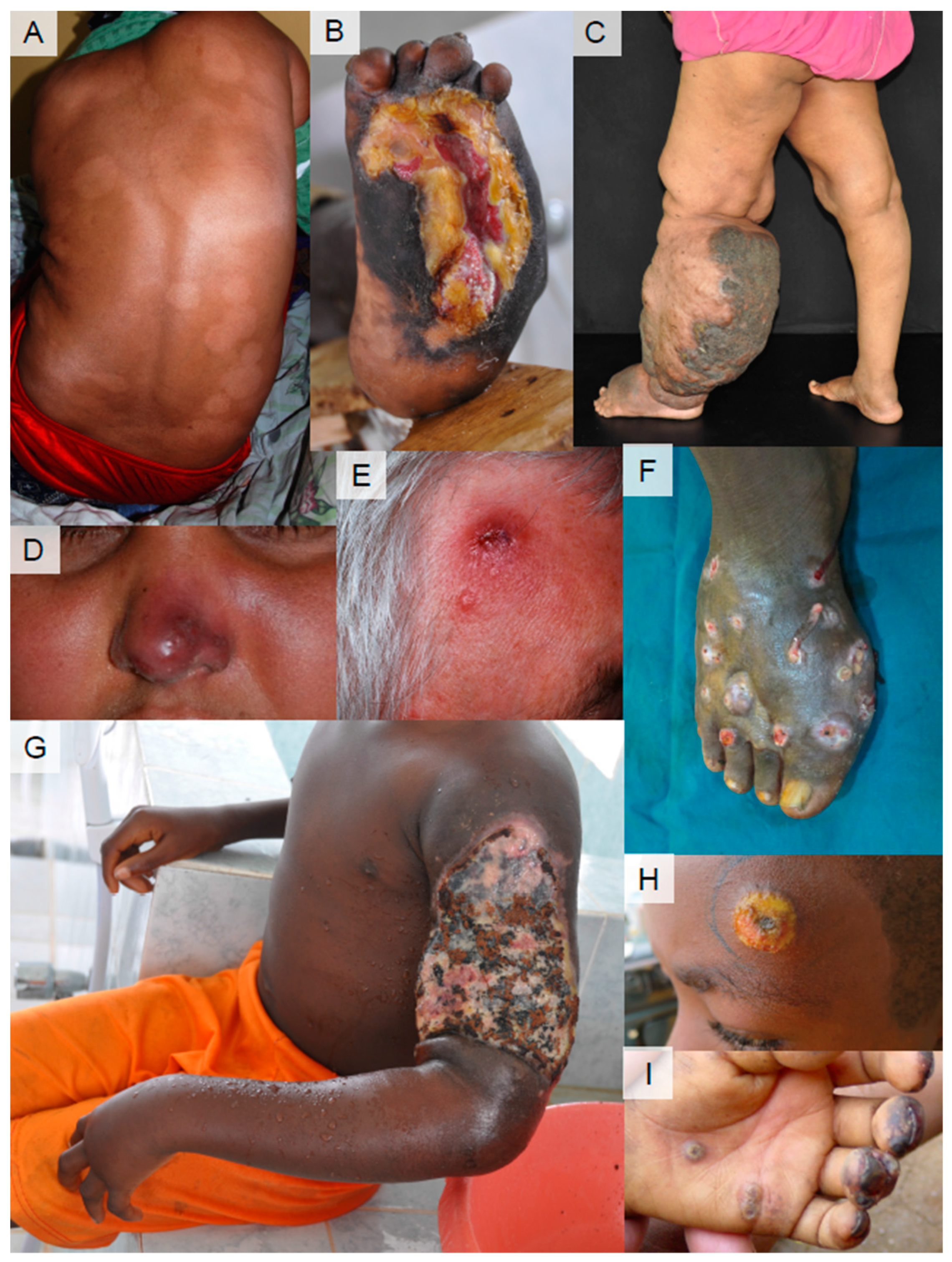
4. Current Status of Diagnosis and Treatment for Skin NTDs
- Training of local healthcare workers on clinical diagnosis
- Training of local healthcare workers on diagnostic tests, including sample taking; make a routine for performing diagnostic tests
- Need for the development of new point-of-care diagnostic tools
- Developments that enhance laboratory confirmation
- Need for further investigation of new drugs and regimens for skin NTDs.
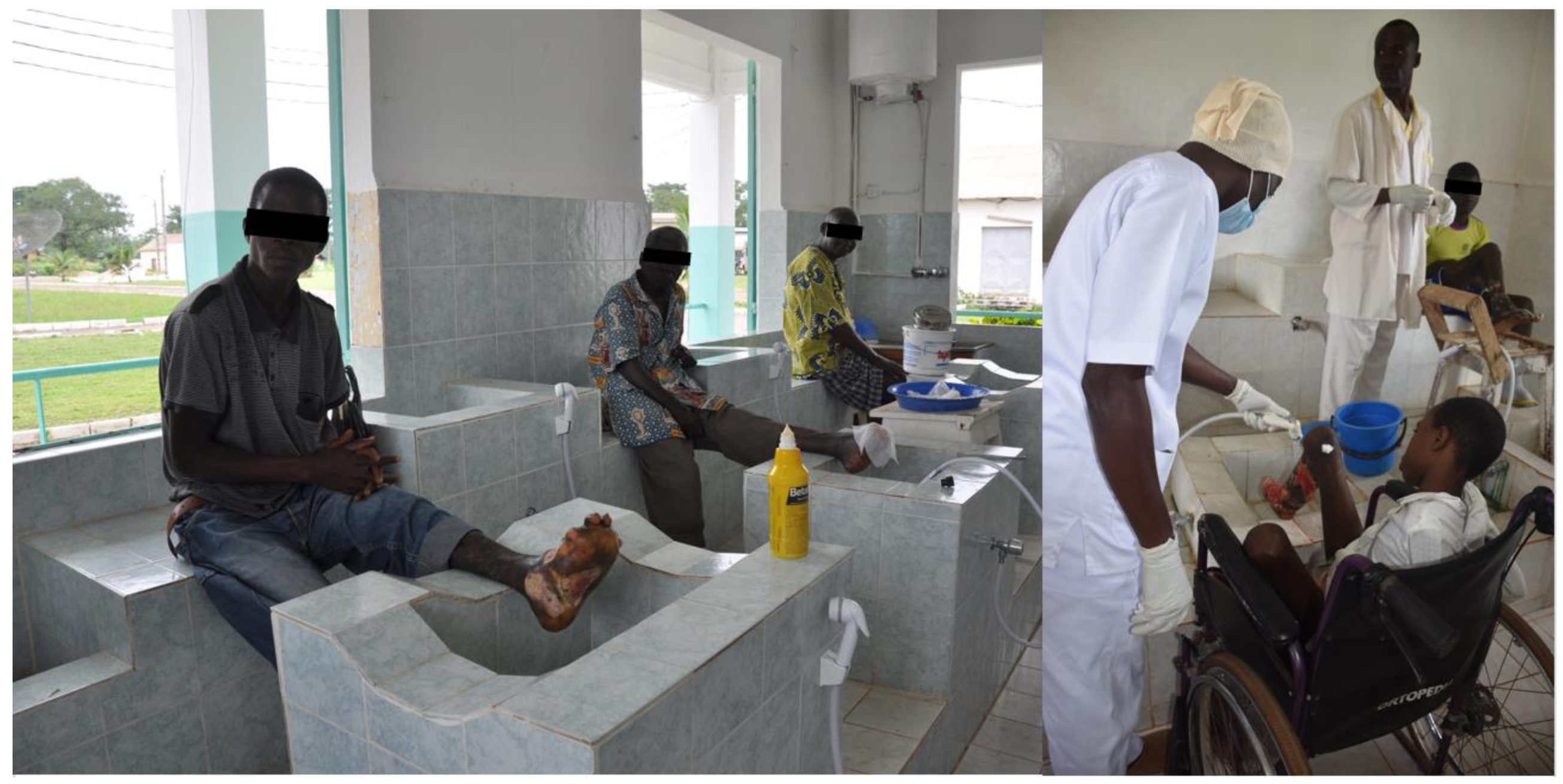
5. Wound and Lymphedema Management–Cross-Cutting Treatment
- Implementation of a simple algorithm utilizing inexpensive and easily obtainable products for wound management/lymphedema management
- Better use of those resources that are available in the local setting
- Cost-analysis
- Training and deployment of helpers including both local health care workers and “the expert patient”.
6. Self-Morbidity Management to Improve Outcomes and Social Inclusion
- Use of locally available methods and materials at low cost
- Patient training for self-care
- Patient empowerment
- Carer training
- Interventions to promote social inclusion.
7. Training and Referrals
8. Next Steps
9. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Hotez, P. Forgotten People, Forgotten Diseases: The Neglected Tropical Diseaes and Their Impact on Global Health and Development, 2nd ed.; ASM Press: Washington, DC, USA, 2013. [Google Scholar]
- Department of Control of Neglected Tropical Diseases, WHO. Neglected Tropical Diseases; WHO: Geneva, Switzerland, 2018; Available online: http://www.who.int/neglected_diseases/diseases/en/ (accessed on 5 August 2018).
- Eberhard, M.L.; Ruiz-Tiben, E.; Hopkins, D.R.; Farrell, C.; Toe, F.; Weiss, A.; Withers, P.C., Jr.; Jenks, M.H.; Thiele, E.A.; Cotton, J.A.; et al. The peculiar epidemiology of dracunculiasis in Chad. Am. J. Trop. Med. Hyg. 2014, 90, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Engelman, D.; Fuller, L.; Solomon, A.; McCarthy, J.; Hay, R.; Lammie, P.; Steer, A. Opportunities for integrated control of neglected tropical diseases that affect the skin. Trends Parasitol. 2016, 32, 843–854. [Google Scholar] [CrossRef] [PubMed]
- Mitja, O.; Marks, M.; Bertran, L.; Kollie, K.; Argaw, D.; Fahal, A.H.; Fitzpatrick, C.; Fuller, L.C.; Garcia Izquierdo, B.; Hay, R.; et al. Integrated control and management of neglected tropical skin diseases. PLoS Negl. Trop. Dis. 2017, 11, e0005136. [Google Scholar] [CrossRef] [PubMed]
- WHO. Eradication of yaws—The Morges strategy. Wkly. Epidemiol. Rec. 2012, 87, 189–194. [Google Scholar]
- Abdulai, A.A.; Agana-Nsiire, P.; Biney, F.; Kwakye-Maclean, C.; Kyei-Faried, S.; Amponsa-Achiano, K.; Simpson, S.V.; Bonsu, G.; Ohene, S.A.; Ampofo, W.K.; et al. Community-based mass treatment with azithromycin for the elimination of yaws in Ghana-Results of a pilot study. PLoS Negl. Trop. Dis. 2018, 12, e0006303. [Google Scholar] [CrossRef] [PubMed]
- Marks, M.; Mitja, O.; Bottomley, C.; Kwakye, C.; Houinei, W.; Bauri, M.; Adwere, P.; Abdulai, A.A.; Dua, F.; Boateng, L.; et al. Comparative efficacy of low-dose versus standard-dose azithromycin for patients with yaws: A randomised non-inferiority trial in Ghana and Papua New Guinea. Lancet Glob. Health 2018, 6, e401–e410. [Google Scholar] [CrossRef]
- Mitja, O.; Godornes, C.; Houinei, W.; Kapa, A.; Paru, R.; Abel, H.; Gonzalez-Beiras, C.; Bieb, S.V.; Wangi, J.; Barry, A.E.; et al. Re-emergence of yaws after single mass azithromycin treatment followed by targeted treatment: A longitudinal study. Lancet 2018, 391, 1599–1607. [Google Scholar] [CrossRef]
- Romani, L.; Whitfeld, M.J.; Koroivueta, J.; Kama, M.; Wand, H.; Tikoduadua, L.; Tuicakau, M.; Koroi, A.; Andrews, R.; Kaldor, J.M.; et al. Mass drug administration for scabies control in a population with endemic disease. N. Engl. J. Med. 2015, 373, 2305–2313. [Google Scholar] [CrossRef] [PubMed]
- Elson, L.; Wright, K.; Swift, J.; Feldmeier, H. Control of tungiasis in absence of a roadmap: Grassroots and global approaches. Trop. Med. Infect. Dis. 2017, 2, 33. [Google Scholar] [CrossRef] [PubMed]
- Yotsu, R.R.; Kouadio, K.; Vagamon, B.; N’Guessan, K.; Akpa, A.J.; Yao, A.; Ake, J.; Abbet Abbet, R.; Tchamba Agbor Agbor, B.; Bedimo, R.; et al. Skin disease prevalence study in schoolchildren in rural Cote d’Ivoire: Implications for integration of neglected skin diseases (skin NTDs). PLoS Negl. Trop. Dis. 2018, 12, e0006489. [Google Scholar] [CrossRef] [PubMed]
- Atre, S.R.; Rangan, S.G.; Shetty, V.P.; Gaikwad, N.; Mistry, N.F. Perceptions, health seeking behaviour and access to diagnosis and treatment initiation among previously undetected leprosy cases in rural Maharashtra, India. Lepr. Rev. 2011, 82, 222–234. [Google Scholar] [PubMed]
- Nicholls, P.G.; Chhina, N.; Bro, A.K.; Barkataki, P.; Kumar, R.; Withington, S.G.; Smith, W.C. Factors contributing to delay in diagnosis and start of treatment of leprosy: Analysis of help-seeking narratives in northern Bangladesh and in West Bengal, India. Lepr. Rev. 2005, 76, 35–47. [Google Scholar] [PubMed]
- Mulder, A.A.; Boerma, R.P.; Barogui, Y.; Zinsou, C.; Johnson, R.C.; Gbovi, J.; van der Werf, T.S.; Stienstra, Y. Healthcare seeking behaviour for Buruli ulcer in Benin: A model to capture therapy choice of patients and healthy community members. Trans. R. Soc. Trop. Med. Hyg. 2008, 102, 912–920. [Google Scholar] [CrossRef] [PubMed]
- Aujoulat, I.; Johnson, C.; Zinsou, C.; Guedenon, A.; Portaels, F. Psychosocial aspects of health seeking behaviours of patients with Buruli ulcer in southern Benin. Trop. Med. Int. Health 2003, 8, 750–759. [Google Scholar] [CrossRef] [PubMed]
- Stienstra, Y.; van der Graaf, W.T.; Asamoa, K.; van der Werf, T.S. Beliefs and attitudes toward Buruli ulcer in Ghana. Am. J. Trop. Med. Hyg. 2002, 67, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Shetty, V.P.; Pandya, S.S.; Arora, S.; Capadia, G.D. Observations from a ‘special selective drive’ conducted under National Leprosy Elimination Programme in Karjat taluka and Gadchiroli district of Maharashtra. Indian J. Lepr. 2009, 81, 189–193. [Google Scholar] [PubMed]
- Daulako, E.C. Population screening and mass chemoprophylaxis in Kiribati. Int. J. Lepr. Mycobact. Dis. 1999, 67, S23–S25. [Google Scholar]
- Diletto, C. Elimination of leprosy in the federated states of micronesia by intensive case finding, treatment with WHO/MDT and administration of chemoprophylaxis. Int. J. Lepr. Mycobact. Dis. 1999, 67, S10–S13. [Google Scholar]
- Tin, K. Population screening and chemoprophylaxis for household contacts of leprosy patients in the Republic of the Marshall Islands. Int. J. Lepr. Mycobact. Dis. 1999, 67, S26–S29. [Google Scholar]
- Msyamboza, K.P.; Mawaya, L.R.; Kubwalo, H.W.; Ng’oma, D.; Liabunya, M.; Manjolo, S.; Msiska, P.P.; Somba, W.W. Burden of leprosy in Malawi: Community camp-based cross-sectional study. BMC Int. Health Hum. Rights 2012, 12, 12. [Google Scholar] [CrossRef] [PubMed]
- Nsagha, D.S.; Bamgboye, E.A.; Yediran, A.B.O.O. Childhood leprosy in Essimbiland of Cameroon: Results of chart review and school survey. Niger. Q. J. Hosp. Med. 2009, 19, 214–219. [Google Scholar] [CrossRef]
- Baretto, J.G.; Guimarães, L.D.S.; Frade, M.A.C.; Rosa, P.S.; Salgado, C.G. High rates of undiagnosed leprosy and subclinical infection amongst school children in the Amazon Region. Mem. Inst. Oswaldo Cruz 2012, 107, 60–67. [Google Scholar] [CrossRef]
- WHO. Brazilian School-Based Deworming and Leprosy Case-Finding Campaign Targets More Than 9 Million Chilren. Available online: http://www.who.int/neglected_diseases/brazil_leprosy_sth_2013/en/ (accessed on 20 August 2018).
- Saikawa, K. Epidemiological implications of school survey in Okinawa (in Japanese). J. Health Welf. Stat. 1978, 25, 6–23. [Google Scholar]
- UmBoock, A.; Awah, P.K.; Mou, F.; Nichter, M. Yaws resurgence in Bankim, Cameroon: The relative effectiveness of different means of detection in rural communities. PLoS Negl. Trop. Dis. 2017, 11, e0005557. [Google Scholar] [CrossRef]
- Ogunbiyi, A.O.; Owoaje, E.; Ndahi, A. Prevalence of skin disorders in school children in Ibadan, Nigeria. Pediatr. Dermatol. 2005, 22, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Hogewoning, A.; Amoah, A.; Bavinck, J.N.; Boakye, D.; Yazdanbakhsh, M.; Adegnika, A.; De Smedt, S.; Fonteyne, Y.; Willemze, R.; Lavrijsen, A. Skin diseases among schoolchildren in Ghana, Gabon, and Rwanda. Int. J. Dermatol. 2013, 52, 589–600. [Google Scholar] [CrossRef] [PubMed]
- Mahe, A.; Prual, A.; Konate, M.; Bobin, P. Skin diseases of children in Mali: A public health problem. Trans. R. Soc. Trop. Med. Hyg. 1995, 89, 467–470. [Google Scholar] [CrossRef]
- Komba, E.V.; Mgonda, Y.M. The spectrum of dermatological disorders among primary school children in Dar es Salaam. BMC Public Health 2010, 10, 765. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, J.I.; Fuller, L.C.; Abraha, A.; Hay, R.J. The prevalence of skin disease among school children in rural Ethiopia—A preliminary assessment of dermatologic needs. Pediatr. Dermatol. 1996, 13, 378–381. [Google Scholar] [CrossRef] [PubMed]
- Murgia, V.; Bilcha, K.D.; Shibeshi, D. Community dermatology in Debre Markos: An attempt to define children’s dermatological needs in a rural area of Ethiopia. Int. J. Dermatol. 2010, 49, 666–671. [Google Scholar] [CrossRef] [PubMed]
- Mitja, O.; Houinei, W.; Moses, P.; Kapa, A.; Paru, R.; Hays, R.; Lukehart, S.; Godornes, C.; Bieb, S.V.; Grice, T.; et al. Mass treatment with single-dose azithromycin for yaws. N. Engl. J. Med. 2015, 372, 703–710. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, G.; Leafasia, J.; Sheridan, J.; Hills, S.; Wate, J.; Wate, C.; Montgomery, J.; Pandeya, N.; Purdie, D. Control of scabies, skin sores and haematuria in children in the Solomon Islands: Another role for ivermectin. Bull. World Health Organ. 2005, 83, 34–42. [Google Scholar]
- Kearns, T.M.; Speare, R.; Cheng, A.C.; McCarthy, J.; Carapetis, J.R.; Holt, D.C.; Currie, B.J.; Page, W.; Shield, J.; Gundjirryirr, R.; et al. Impact of an ivermectin mass drug administration on scabies prevalence in a remote australian aboriginal community. PLoS Negl. Trop. Dis. 2015, 9, e0004151. [Google Scholar] [CrossRef] [PubMed]
- Mason, D.S.; Marks, M.; Sokana, O.; Solomon, A.W.; Mabey, D.C.; Romani, L.; Kaldor, J.; Steer, A.C.; Engelman, D. The prevalence of scabies and impetigo in the Solomon islands: a population-based survey. PLoS Negl. Trop. Dis. 2016, 10, e0004803. [Google Scholar] [CrossRef] [PubMed]
- Heukelbach, J.; Winter, B.; Wilcke, T.; Muehlen, M.; Albrecht, S.; de Oliveira, F.A.; Kerr-Pontes, L.R.; Liesenfeld, O.; Feldmeier, H. Selective mass treatment with ivermectin to control intestinal helminthiases and parasitic skin diseases in a severely affected population. Bull. World Health Organ. 2004, 82, 563–571. [Google Scholar]
- Krotneva, S.P.; Coffeng, L.E.; Noma, M.; Zoure, H.G.; Bakone, L.; Amazigo, U.V.; de Vlas, S.J.; Stolk, W.A. African program for onchocerciasis control 1995–2010: Impact of annual ivermectin mass treatment on off-target infectious diseases. PLoS Negl. Trop. Dis. 2015, 9, e0004051. [Google Scholar] [CrossRef] [PubMed]
- Ottesen, E.A.; Hooper, P.J.; Bradley, M.; Biswas, G. The global programme to eliminate lymphatic filariasis: Health impact after 8 years. PLoS Negl. Trop. Dis. 2008, 2, e317. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, E.K.; Sanuku, N.; Baea, M.; Satofan, S.; Maki, E.; Lombore, B.; Schmidt, M.S.; Siba, P.M.; Weil, G.J.; Kazura, J.W.; et al. Efficacy, safety, and pharmacokinetics of coadministered diethylcarbamazine, albendazole, and ivermectin for treatment of bancroftian filariasis. Clin. Infect. Dis. 2016, 62, 334–341. [Google Scholar] [CrossRef] [PubMed]
- Engelman, D.; Kiang, K.; Chosidow, O.; McCarthy, J.; Fuller, C.; Lammie, P.; Hay, R.; Steer, A.; Members of the International Alliance for the Control of Scabies. Toward the global control of human scabies: Introducing the International Alliance for the Control of Scabies. PLoS Negl. Trop. Dis. 2013, 7, e2167. [Google Scholar] [CrossRef] [PubMed]
- Mounsey, K.E.; Bernigaud, C.; Chosidow, O.; McCarthy, J.S. Prospects for moxidectin as a new oral treatment for human scabies. PLoS Negl. Trop. Dis. 2016, 10, e0004389. [Google Scholar] [CrossRef] [PubMed]
- Mounsey, K.E.; Walton, S.F.; Innes, A.; Cash-Deans, S.; McCarthy, J.S. In vitro efficacy of moxidectin versus Ivermectin against Sarcoptes scabiei. Antimicrob. Agents Chemother. 2017, 61, e00381-17. [Google Scholar] [CrossRef] [PubMed]
- Administration, U.S.F.A.D. Drugs@FDA: FDA Approved Drug Products. 2018. Available online: https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=BasicSearch.process (accessed on 31 August 2018).
- Moet, F.J.; Pahan, D.; Oskam, L.; Richardus, J.H.; Group, C.S. Effectiveness of single dose rifampicin in preventing leprosy in close contacts of patients with newly diagnosed leprosy: Cluster randomised controlled trial. BMJ 2008, 336, 761–764. [Google Scholar] [CrossRef] [PubMed]
- Feenstra, S.G.; Pahan, D.; Moet, F.J.; Oskam, L.; Richardus, J.H. Patient-related factors predicting the effectiveness of rifampicin chemoprophylaxis in contacts: 6 year follow up of the COLEP cohort in Bangladesh. Lepr. Rev. 2012, 83, 292–304. [Google Scholar] [PubMed]
- Bakker, M.I.; Hatta, M.; Kwenang, A.; Van Benthem, B.H.; Van Beers, S.M.; Klatser, P.R.; Oskam, L. Prevention of leprosy using rifampicin as chemoprophylaxis. Am. J. Trop. Med. Hyg. 2005, 72, 443–448. [Google Scholar] [CrossRef] [PubMed]
- WHO. Global leprosy update, 2016: Accelerating reduction of disease burden. Wkly. Epidemiol. Rec. 2017, 35, 501–520. [Google Scholar]
- Tiwari, A.; Dandel, S.; Djupuri, R.; Mieras, L.; Richardus, J.H. Population-wide administration of single dose rifampicin for leprosy prevention in isolated communities: A three year follow-up feasibility study in Indonesia. BMC Infect. Dis. 2018, 18, 324. [Google Scholar] [CrossRef] [PubMed]
- Barth-Jaeggi, T.; Steinmann, P.; Mieras, L.; van Brakel, W.; Richardus, J.H.; Tiwari, A.; Bratschi, M.; Cavaliero, A.; Vander Plaetse, B.; Mirza, F.; et al. Leprosy Post-Exposure Prophylaxis (LPEP) programme: Study protocol for evaluating the feasibility and impact on case detection rates of contact tracing and single dose rifampicin. BMJ Open 2016, 6, e013633. [Google Scholar] [CrossRef] [PubMed]
- Steinmann, P.; Cavaliero, A.; Aerts, A.; Anand, S.; Arif, M.; Ay, S.S.; Aye, T.M.; Barth-Jaeggi, T.; Banstola, N.L.; Bhandari, C.M.; et al. The Leprosy Post-Exposure Prophylaxis (LPEP) programme: Update and interim analysis. Lepr. Rev. 2018, 89, 102–116. [Google Scholar]
- Toutous Trellu, L.; Nkemenang, P.; Comte, E.; Ehounou, G.; Atangana, P.; Mboua, D.J.; Rusch, B.; Njih Tabah, E.; Etard, J.F.; Mueller, Y.K. Differential diagnosis of skin ulcers in a Mycobacterium ulcerans endemic area: Data from a prospective study in cameroon. PLoS Negl. Trop. Dis. 2016, 10, e0004385. [Google Scholar] [CrossRef] [PubMed]
- Nguetta, A.; Coulibaly, N.D.; Kouamé-Elogne, N.C.; Acquah, K.J.R.; Christiane, A.A.; Kouamé, K.; N’Guessan, K.; Aboa, K.; Aubin, Y. Phenotypic and genotypic characterization of mycobacteria isolates from Buruli ulcer suspected patients reveals the involvement of several mycobacteria in chronic skin lesions. Am. J. Microbiol. Res. 2018, 6, 79–87. [Google Scholar] [CrossRef]
- Ablordey, A.; Amissah, D.A.; Aboagye, I.F.; Hatano, B.; Yamazaki, T.; Sata, T.; Ishikawa, K.; Katano, H. Detection of Mycobacterium ulcerans by the loop mediated isothermal amplification method. PLoS Negl. Trop. Dis. 2012, 6, e1590. [Google Scholar] [CrossRef] [PubMed]
- Beissner, M.; Phillips, R.O.; Battke, F.; Bauer, M.; Badziklou, K.; Sarfo, F.S.; Maman, I.; Rhomberg, A.; Piten, E.; Frimpong, M.; et al. Loop-mediated isothermal amplification for laboratory confirmation of buruli ulcer disease-towards a point-of-care test. PLoS Negl. Trop. Dis. 2015, 9, e0004219. [Google Scholar] [CrossRef] [PubMed]
- Wadagni, A.; Frimpong, M.; Phanzu, D.M.; Ablordey, A.; Kacou, E.; Gbedevi, M.; Marion, E.; Xing, Y.; Babu, V.S.; Phillips, R.O.; et al. Simple, rapid Mycobacterium ulcerans disease diagnosis from clinical samples by fluorescence of mycolactone on thin layer chromatography. PLoS Negl. Trop. Dis. 2015, 9, e0004247. [Google Scholar] [CrossRef] [PubMed]
- Dreyer, A.; Roltgen, K.; Dangy, J.P.; Ruf, M.T.; Scherr, N.; Bolz, M.; Tobias, N.J.; Moes, C.; Vettiger, A.; Stinear, T.P.; et al. Identification of the Mycobacterium ulcerans protein MUL_3720 as a promising target for the development of a diagnostic test for Buruli ulcer. PLoS Negl. Trop. Dis. 2015, 9, e0003477. [Google Scholar] [CrossRef] [PubMed]
- WHO. Eradication of Yaws: A Guide for Programme Managers; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- Yotsu, R.; Richardson, M.; Ishii, N. Drugs for Treating Buruli Ulcer; Cochrane Systematic Review: London, UK, 2018. [Google Scholar]
- Ryan, T.J. Public health dermatology: Regeneration and repair of the skin in the developed transitional and developing world. Int. J. Dermatol. 2006, 45, 1233–1237. [Google Scholar] [CrossRef] [PubMed]
- Velding, K.; Klis, S.A.; Abass, K.M.; Tuah, W.; Stienstra, Y.; van der Werf, T. Wound care in Buruli ulcer disease in Ghana and Benin. Am. J. Trop. Med. Hyg. 2014, 91, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Attinger, C.E.; Janis, J.E.; Steinberg, J.; Schwartz, J.; Al-Attar, A.; Couch, K. Clinical approach to wounds: Debridement and wound bed preparation including the use of dressings and wound-healing adjuvants. Plast. Reconstr. Surg. 2006, 117, 72s–109s. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.C.; Kandula, S.; Sherber, N.S. Beyond wet-to-dry: A rational approach to treating chronic wounds. Eplasty 2009, 9, e14. [Google Scholar] [PubMed]
- Fernandez, R.; Griffiths, R. Water for wound cleansing. Cochrane Database Syst. Rev. 2012. [Google Scholar] [CrossRef] [PubMed]
- Cooper, D.D.; Seupaul, R.A. Is water effective for wound cleansing? Ann. Emerg. Med. 2012, 60, 626–627. [Google Scholar] [CrossRef] [PubMed]
- Anandan, V.; Jameela, W.A.; Saraswathy, P.; Sarankumar, S. Platelet rich plasma: Efficacy in treating trophic ulcers in leprosy. J. Clin. Diagn. Res. 2016, 10, WC06–WC09. [Google Scholar] [CrossRef] [PubMed]
- Conde-Montero, E.; Horcajada-Reales, C.; Clavo, P.; Delgado-Sillero, I.; Suarez-Fernandez, R. Neuropathic ulcers in leprosy treated with intralesional platelet-rich plasma. Int. Wound J. 2016, 13, 726–728. [Google Scholar] [CrossRef] [PubMed]
- Murase, C.; Kono, M.; Nakanaga, K.; Ishii, N.; Akiyama, M. Buruli ulcer successfully treated with negative-pressure wound therapy. JAMA Dermatol. 2015, 151, 1137–1139. [Google Scholar] [CrossRef] [PubMed]
- Jebran, A.F.; Schleicher, U.; Steiner, R.; Wentker, P.; Mahfuz, F.; Stahl, H.C.; Amin, F.M.; Bogdan, C.; Stahl, K.W. Rapid healing of cutaneous leishmaniasis by high-frequency electrocauterization and hydrogel wound care with or without DAC N-055: A randomized controlled phase IIa trial in Kabul. PLoS Negl. Trop. Dis. 2014, 8, e2694. [Google Scholar] [CrossRef] [PubMed]
- Jull, A.B.; Walker, N.; Deshpande, S. Honey as a topical treatment for wounds. Cochrane Database Syst. Rev. 2013. [Google Scholar] [CrossRef]
- Stocks, M.E.; Freeman, M.C.; Addiss, D.G. The effect of hygiene-based lymphedema management in lymphatic filariasis-endemic areas: A systematic review and meta-analysis. PLoS Negl. Trop. Dis. 2015, 9, e0004171. [Google Scholar] [CrossRef] [PubMed]
- Dreyer, G.; Medeiros, Z.; Netto, M.J.; Leal, N.C.; de Castro, L.G.; Piessens, W.F. Acute attacks in the extremities of persons living in an area endemic for bancroftian filariasis: Differentiation of two syndromes. Trans. Roy. Soc. Trop. Med. Hyg. 1999, 93, 413–417. [Google Scholar] [CrossRef]
- Brooks, J.; Ersser, S.J.; Cowdell, F.; Gardiner, E.; Mengistu, A.; Matts, P.J. A randomized controlled trial to evaluate the effect of a new skincare regimen on skin barrier function in those with podoconiosis in Ethiopia. Br. J. Dermatol. 2017, 177, 1422–1431. [Google Scholar] [CrossRef] [PubMed]
- Negussie, H.; Molla, M.; Ngari, M.; Berkley, J.A.; Kivaya, E.; Njuguna, P.; Fegan, G.; Tamiru, A.; Kelemework, A.; Lang, T.; et al. Lymphoedema management to prevent acute dermatolymphangioadenitis in podoconiosis in northern Ethiopia (GoLBeT): A pragmatic randomised controlled trial. Lancet Glob. Health 2018, 6, e795–e803. [Google Scholar] [CrossRef]
- Effah, A.; Ersser, S.J.; Hemingway, A. Support needs of people living with Mycobacterium ulcerans (Buruli ulcer) disease in a Ghana rural community: A grounded theory study. Int. J. Dermatol. 2017, 56, 1432–1437. [Google Scholar] [CrossRef] [PubMed]
- Narahari, S.R.; Bose, K.S.; Aggithaya, M.G.; Swamy, G.K.; Ryan, T.J.; Unnikrishnan, B.; Washington, R.G.; Rao, B.P.; Rajagopala, S.; Manjula, K.; et al. Community level morbidity control of lymphoedema using self care and integrative treatment in two lymphatic filariasis endemic districts of South India: A non randomized interventional study. Trans. R. Soc. Trop. Med. Hyg. 2013, 107, 566–577. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, J.; Deribe, K.; Tamiru, A.; Amberbir, T.; Medhin, G.; Malik, M.; Hanlon, C.; Davey, G. Depression and disability in people with podoconiosis: A comparative cross-sectional study in rural Northern Ethiopia. Int. Health 2016, 8, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Bennis, I.; De Brouwere, V.; Belrhiti, Z.; Sahibi, H.; Boelaert, M. Psychosocial burden of localised cutaneous Leishmaniasis: A scoping review. BMC Public Health 2018, 18, 358. [Google Scholar] [CrossRef] [PubMed]
- Bakhiet, S.M.; Fahal, A.H.; Musa, A.M.; Mohamed, E.S.W.; Omer, R.F.; Ahmed, E.S.; El Nour, M.; Mustafa, E.R.M.; Sheikh, A.R.M.E.; Suliman, S.H.; et al. A holistic approach to the mycetoma management. PLoS Negl. Trop. Dis. 2018, 12, e0006391. [Google Scholar] [CrossRef] [PubMed]
- Wiese, S.; Elson, L.; Feldmeier, H. Tungiasis-related life quality impairment in children living in rural Kenya. PLoS Negl. Trop. Dis. 2018, 12, e0005939. [Google Scholar] [CrossRef] [PubMed]
- Walker, S.L.; Lebas, E.; De Sario, V.; Deyasso, Z.; Doni, S.N.; Marks, M.; Roberts, C.H.; Lambert, S.M. The prevalence and association with health-related quality of life of tungiasis and scabies in schoolchildren in southern Ethiopia. PLoS Negl. Trop. Dis. 2017, 11, e0005808. [Google Scholar] [CrossRef] [PubMed]
- Guest, J.F.; Ayoub, N.; McIlwraith, T.; Uchegbu, I.; Gerrish, A.; Weidlich, D.; Vowden, K.; Vowden, P. Health economic burden that different wound types impose on the UK’s National Health Service. Int. Wound J. 2017, 14, 322–330. [Google Scholar] [CrossRef] [PubMed]
- Tchero, H.; Kangambega, P.; Lin, L.; Mukisi-Mukaza, M.; Brunet-Houdard, S.; Briatte, C.; Retali, G.R.; Rusch, E. Cost of diabetic foot in France, Spain, Italy, Germany and United Kingdom: A systematic review. Ann. Endocrinol. 2018, 79, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Stahl, H.C.; Ahmadi, F.; Nahzat, S.M.; Dong, H.J.; Stahl, K.W.; Sauerborn, R. Health economic evaluation of moist wound care in chronic cutaneous leishmaniasis ulcers in Afghanistan. Infect. Dis. Poverty 2018, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Walsh, D.S.; De Jong, B.C.; Meyers, W.M.; Portaels, F. Leprosy and Buruli ulcer: Similarities suggest combining control and prevention of disability strategies in countries endemic for both diseases. Lepr. Rev. 2015, 86, 1–5. [Google Scholar] [PubMed]
- Douglass, J.; Graves, P.; Gordon, S. Self-care for management of secondary lymphedema: A systematic review. PLoS Negl. Trop. Dis. 2016, 10, e0004740. [Google Scholar] [CrossRef] [PubMed]
- Sathiaraj, Y.; Norman, G.; Richard, J. Long term sustainability and efficacy of self-care education on knowledge and practice of wound prevention and management among leprosy patients. Indian J. Lepr. 2010, 82, 79–83. [Google Scholar] [PubMed]
- Ryan, T. Wound healing in the developing world. Dermatol. Clin. 1993, 11, 791–800. [Google Scholar] [CrossRef]
- Deribe, K.; Kebede, B.; Tamiru, M.; Mengistu, B.; Kebede, F.; Martindale, S.; Sime, H.; Mulugeta, A.; Kebede, B.; Sileshi, M.; et al. Integrated morbidity management for lymphatic filariasis and podoconiosis, Ethiopia. Bull. World Health Organ. 2017, 95, 652–656. [Google Scholar] [CrossRef] [PubMed]
- WHO. Recognizing Neglected Tropical Diseases through Changes on the Skin: A Training Guide for Front-Line Health Workers; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- Barbieri, R.R.; Sales, A.M.; Hacker, M.A.; Nery, J.A.; Duppre, N.C.; Machado, A.M.; Moraes, M.O.; Sarno, E.N. Impact of a reference center on leprosy control under a decentralized public health care policy in Brazil. PLoS Negl. Trop. Dis. 2016, 10, e0005059. [Google Scholar] [CrossRef] [PubMed]
- Stanton, M.C.; Mkwanda, S.Z.; Debrah, A.Y.; Batsa, L.; Biritwum, N.K.; Hoerauf, A.; Cliffe, M.; Best, A.; Molineux, A.; Kelly-Hope, L.A. Developing a community-led SMS reporting tool for the rapid assessment of lymphatic filariasis morbidity burden: Case studies from Malawi and Ghana. BMC Infect. Dis. 2015, 15, 214. [Google Scholar] [CrossRef] [PubMed]
- Hay, R.; Estrada, R.; Grossmann, H. Managing skin disease in resource-poor environments—The role of community-oriented training and control programs. Int. J. Dermatol. 2011, 50, 558–563. [Google Scholar] [CrossRef] [PubMed]
- Faye, O.; Bagayoko, C.O.; Dicko, A.; Ciseé, L.; Berthé, S.; Traoré, B.; Fofana, Y.; Niang, M.; Traoré, S.T.; Karabinta, Y.; et al. A teledermatology pilot programme for the management of skin diseases in primary health care centres: Experiences from a Resource-Limited Country (Mali, West Africa). Trop. Med. Infect. Dis. 2018, 3, 88. [Google Scholar] [CrossRef] [PubMed]
- Mieras, L.F.; Taal, A.T.; Post, E.B.; Ndeve, A.G.Z.; van Hees, C.L.M. The development of a mobile application to support peripheral health workers to diagnose and treat people with skin diseases in resource-poor settings. Trop. Med. Infect. Dis. 2018, 3, 102. [Google Scholar] [CrossRef] [PubMed]
- Tomczyk, S.; Deribe, K.; Brooker, S.J.; Clark, H.; Rafique, K.; Knopp, S.; Utzinger, J.; Davey, G. Association between footwear use and neglected tropical diseases: A systematic review and meta-analysis. PLoS Negl. Trop. Dis. 2014, 8, e3285. [Google Scholar] [CrossRef] [PubMed]
- WHO. Guidelines for the Diagnosis, Treatment and Prevention of Leprosy; WHO, Regional Office for South-East Asia: New Delhi, India, 2018. [Google Scholar]
- Engelman, D.; Fuller, L.C.; Steer, A.C.; International Alliance for the Control of Scabies. Consensus criteria for the diagnosis of scabies: A Delphi study of international experts. PLoS Negl. Trop. Dis. 2018, 12, e0006549. [Google Scholar] [CrossRef] [PubMed]

| Pathogen | Rapid Diagnostic Test | PCR | Microscopy | Culture | Serology | Others | |
|---|---|---|---|---|---|---|---|
| Buruli ulcer | Mycobacterium ulcerans | X | O | O | O | X | LAMP test, thin layer chromatography, antigen detection assays under development |
| Cutaneous leishmaniasis (CL)/mucocutaneous leishmaniasis (ML) | Leishmania species | X | O | O Skin smears | O | X | LAMP test, antigen detection assays under development (Montenegro skin test) |
| Lymphatic filariasis (LF) | Microfilaria (Wuchereria bancrofti, Brugia malayi, etc.) | O | O | O Blood smears | X | ∆ Anti-filarial antibodies | Ultrasonography |
| Onchocerciasis | Microfilaria (Onchocerca volvulus) | O | O | O Skin snips | X | ∆ Anti-filarial antibodies | Direct observation of adult worms from nodule(s), slit-lamp eye exam, serological and antigen tests under development |
| Leprosy | Mycobacterium leprae | X | O | O | X | ∆ Anti-PGL-I antibody | Thickened nerves, loss of muscle strength, anesthetic skin lesion |
| Mycetoma | Fungal or bacterial species | X | O | ∆ | O | X | X-rays, CT, ultrasonography, etc. |
| Podoconiosis | Irritant alkalic clay soils | N/A | N/A | N/A | N/A | N/A | Location, history, clinical findings; negative results for LF and other lymphedema-causing diseases; genetic susceptibility |
| Scabies | Sarcoptes scabiei var. hominis | X | ∆ | O | X | X | Dermatoscopy, burrow ink test |
| Tungiasis | Tunga penetrans (sand fleas) | X | ∆ | O | X | X | Direct observation of adult fleas and eggs from skin lesion(s), dermatoscopy |
| Yaws | Treponema pallidum subsp. pertenue | O | O | O | O | PRP, TPHA, FTA-ABS, etc. | Diagnostics for differentiation of Treponema pallidum species under development |
| Medical Treatment | Surgery | Wound or Lymphedema Management | Self-Morbidity Management | Prevention | |
|---|---|---|---|---|---|
| Buruli ulcer | Standard: Oral rifampicin + clarithromycin for 8 weeks Other tested regimens: Oral rifampicin + either 1 or 2 of [ciprofloxacin, ethambutol, mofloxacin, amikacin, etc.] | Yes | Yes | Yes | Limited, route of transmission unknown (Stay away from contaminated water sources) |
| Cutaneous leishmaniasis (CL)/mucocutaneous leishmaniasis (ML) | Individualized treatment depending on species (no standard) Amphotericin B deoxycholate, pentavalent antimonials, fluconazole, ketoconazole, miltefosine, paromomycin ointment, etc. Simple CL lesion(s) with low ML-risk: natural healing may occur Complex CL lesion(s) with high-ML risk, severe lesion(s), immunocompromised persons, etc.: treat all cases | No | Yes | No | Limited (Avoid sand fly bites) |
| Lymphatic filariasis (LF) | Oral albendazole ± [diethylcarbamazine (DEC) or ivermectin] When long-term treatment is possible: Oral DEC (1–12 days) ± doxycycline for 4 to 6 weeks Note: DEC contraindicated in onchocerciasis endemic sites | Yes | Yes | Yes | Avoid mosquito bites, MDAs, vector control, etc. |
| Onchocerciasis | Oral ivermectin | Yes | No | No | Avoid blackfly bites, MDAs, vector control, etc. |
| Leprosy | Multiple drug therapy (MDT): Oral rifampicin + dapsone + clofazimine for 6 to 12 months | Yes | Yes | Yes | Contact tracing and early detection; prophylaxis with one-dose rifampicin in trial |
| Mycetoma | Antibiotics or antifungals depending on species for long-term | Yes | Yes | Yes | Footwear |
| Podoconiosis | N/A | Yes | Yes | Yes | Footwear |
| Scabies | Oral ivermectin, 1–2 doses 1 week apart | No | No | No | Early diagnosis and treatment of contacts, possible MDAs in endemic communities |
| Tungiasis | None (primary treatment: hygienic mechanical removal of fleas), antibiotics if secondary infection is indicated | No | Yes | No | Footwear |
| Yaws | Single oral azithromycin or injectable benzathine penicillin | No | Yes | No | Contact tracing and early detection, possible MDAs in endemic communities |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yotsu, R.R. Integrated Management of Skin NTDs—Lessons Learned from Existing Practice and Field Research. Trop. Med. Infect. Dis. 2018, 3, 120. https://doi.org/10.3390/tropicalmed3040120
Yotsu RR. Integrated Management of Skin NTDs—Lessons Learned from Existing Practice and Field Research. Tropical Medicine and Infectious Disease. 2018; 3(4):120. https://doi.org/10.3390/tropicalmed3040120
Chicago/Turabian StyleYotsu, Rie R. 2018. "Integrated Management of Skin NTDs—Lessons Learned from Existing Practice and Field Research" Tropical Medicine and Infectious Disease 3, no. 4: 120. https://doi.org/10.3390/tropicalmed3040120
APA StyleYotsu, R. R. (2018). Integrated Management of Skin NTDs—Lessons Learned from Existing Practice and Field Research. Tropical Medicine and Infectious Disease, 3(4), 120. https://doi.org/10.3390/tropicalmed3040120




