Melioidosis in the Philippines
Abstract
:1. Introduction and History of Melioidosis in the Philippines
2. Materials and Methods
2.1. Review of Publications
2.2. Collection of Locally Published and Unpublished Cases from the Philippines
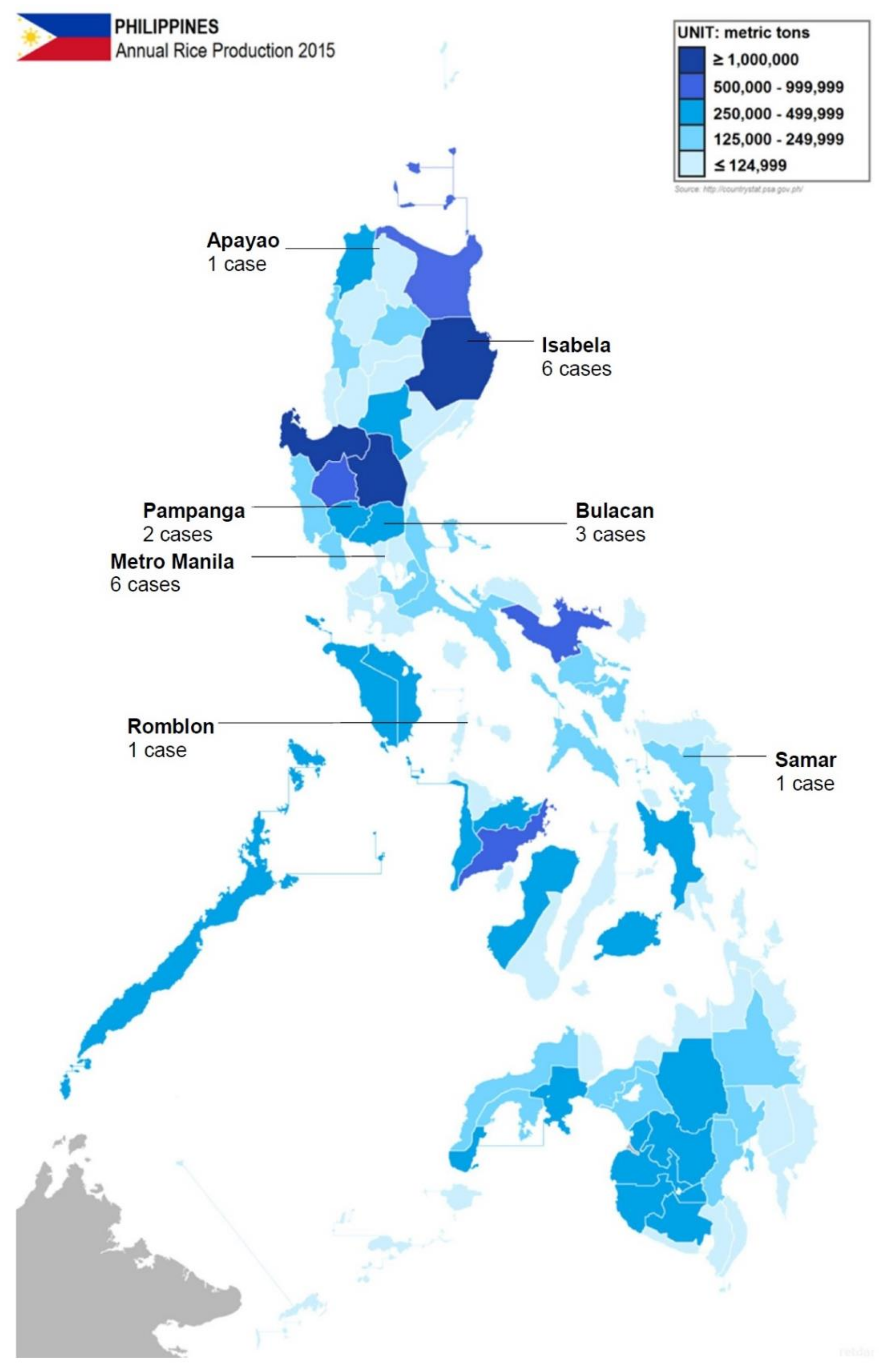
3. Results and Discussion
4. Challenges
5. Conclusions and Recommendations
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cheng, A.C.; Currie, B.J. Melioidosis: Epidemiology, pathophysiology, and management. Clin. Microbiol. Rev. 2005, 18, 383–416. [Google Scholar] [CrossRef] [PubMed]
- Dance, D.A. Melioidosis: The tip of the iceberg? Clin. Microbiol. Rev. 1991, 4, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Whitmore, A. An account of a glanders-like disease occurring in Rangoon. J. Hyg. 1913, 13, 1–34. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.J.; Allen, J.C.; Embi, M.N.; Othman, O.; Razak, N.; Ismail, G. Human melioidosis: An emerging medical problem. MIRCEN J. 1987, 3. [Google Scholar] [CrossRef]
- Limmathurotsakul, D.; Golding, N.; Dance, D.A.B.; Messina, J.P.; Pigott, D.M.; Moyes, C.L.; Rolim, D.B.; Bertherat, E.; Day, N.P.; Peacock, S.J.; et al. Predicted global distribution of Burkholderi pseudomallei and burden of melioidosis. Nat. Microbiol. 2016, 1. [Google Scholar] [CrossRef] [PubMed]
- Gutner, L.B.; Fisher, M.W. Chronic melioidosis: Discussion, case report, and special studies. Ann. Intern. Med. 1948, 28, 1157–1169. [Google Scholar] [CrossRef]
- Pons, R.; Advier, M. Melioidosis in Cochin China. J. Hyg. 1927, 26, 28–30. [Google Scholar] [CrossRef] [PubMed]
- De Moor, C.E.; Soekarnen Walle, N. Melioidosis op Java. Mededeeling uit het Geneeskundig Laboratorium te Weltevreden 1932, 72, 1618–1635. [Google Scholar]
- Chittivej, C.; Buspavanich, S.; Chaovanasai, A. Melioidosis with case report in a Thai. R. Thai Army Med. J. 1955, 68, 11–17. [Google Scholar]
- Prevatt, A.L.; Hunt, J.S. Chronic systemic melioidosis. Am. J. Med. Sci. 1957, 23, 810–823. [Google Scholar] [CrossRef]
- Lee, N.; Wu, J.L.; Lee, C.H.; Tsai, W.C. Pseudomonas pseudomallei infection from drowning: The first reported case in Taiwan. J. Clin. Microbiol. 1985, 22, 352–354. [Google Scholar] [PubMed]
- Wetmore, P.W.; Gochenour, W.S. Comparative studies of the genus Malleomyces and selected Pseudomonas species. J. Bacteriol. 1956, 72, 79–89. [Google Scholar] [PubMed]
- Dance, D.A.; King, C.; Aucken, H.; Knott, C.D.; West, P.G.; Pitt, T.L. An outbreak of melioidosis in imported primates in Britain. Vet. Rec. 1992, 130, 525–529. [Google Scholar] [CrossRef] [PubMed]
- Ereno, I.L.; Mariano, N.; Reyes, J.; Amando, C. Melioidosis: A case report. Philipp. J. Microbiol. Infect. Dis. 2002, 31, 125–133. [Google Scholar]
- Martin, P.F.M.S.; Teh, C.S.C.; Casupang, M.A.J. Melioidosis: A rare cause of liver abscess. Case Rep. Hepatol. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Kingsley, P.V.; Leader, M.; Nagodawithana, N.S.; Tipre, M.; Sathiakumar, N. Melioidosis in Malaysia: A review of case reports. PLoS Negl. Trop. Dis. 2016, 10, e0005182. [Google Scholar] [CrossRef] [PubMed]
- Meumann, E.M.; Cheng, A.C.; Ward, L.; Currie, B.J. Clinical features and epidemiology of melioidosis pneumonia: Results from a 21-year study and review of the literature. Clin. Infect. Dis. 2012, 54, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Churuangsuk, C.; Chusri, S.; Hortiwakul, T.; Charernmak, B.; Silpapojakul, K. Characteristics, clinical outcomes and factors influencing mortality of patients with melioidosis in southern Thailand: A 10-year retrospective study. Asian Pac. J. Trop. Med. 2016, 9, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Mays, E.E.; Ricketts, E.A. Melioidosis: Recrudescence associated with bronchogenic carcinoma twenty-six years following initial geographic exposure. Chest 1975, 68, 261–263. [Google Scholar] [CrossRef] [PubMed]
- John, J.F., Jr. Trimethoprim-sulfamethoxazole therapy of pulmonary melioidosis. Am. Rev. Respir. Dis. 1976, 114, 1021–1025. [Google Scholar] [CrossRef] [PubMed]
- Fuller, P.B.; Fisk, D.E.; Byrd, R.B.; Griggs, G.A.; Smith, M.R. Treatment of pulmonary melioidosis with combination of trimethoprim and sulfamethoxazole. Chest 1978, 74, 222–224. [Google Scholar] [CrossRef] [PubMed]
- Turner, M.O.; Lee, V.T.; FitzGerald, J.M. Melioidosis in a diabetic sailor. Chest 1994, 106, 952–954. [Google Scholar] [CrossRef] [PubMed]
- Falade, O.O.; Antonarakis, E.S.; Kaul, D.R.; Saint, S.; Murphy, P.A. Clinical problem-solving. Beware of first impressions. N. Engl. J. Med. 2008, 359, 628–634. [Google Scholar] [CrossRef] [PubMed]
- Duplessis, C.; Maguire, J.D. Melioidosis masquerading as community-acquired pneumonia: A case report demonstrating efficacy of intrapleural fibrinolytic therapy. J. Travel. Med. 2009, 16, 74–77. [Google Scholar] [CrossRef] [PubMed]
- Velasco, A.B.; Untalan, C.A.; Visperas, J.C. Melioiodisis presenting as severe community acquired pneumonia. Am. J. Respir. Crit. Care 2010, 181. [Google Scholar] [CrossRef]
- Pande, K.C.; Kadir, K.A. Melioidosis of the extremities in Brunei Darussalam. Singap. Med. J. 2011, 52, 346–350. [Google Scholar]
- Chagla, Z.; Aleksova, N.; Quirt, J.; Emery, J.; Kraeker, C.; Haider, S. Melioidosis in a returned traveller. Can. J. Infect. Dis. Med. Microbiol. 2014, 25, 225–226. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.W.; Kwon, G.-Y.; Kim, B.; Kwon, D.; Shin, J.; Bae, G.-R. Imported melioidosis in South Korea: A case series with a literature review. Osong Public Health Res. Perspect. 2015, 6, 363–368. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.F.; Wong, F.L.; Perez, M.L. Splenic abscesses in a returning traveler. Infect. Dis. Rep. 2015, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hemarajata, P.; Baghdadi, J.D.; Hoffman, R.; Humphries, R.M. Burkholderia pseudomallei: Challenges for the clinical microbiology laboratory. J. Clin. Microbiol. 2016, 54, 2866–2873. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Mahmood, M. Melioidosis: The great mimicker. J. Community Hosp. Intern. Med. Perspect. 2017, 7, 245–247. [Google Scholar] [CrossRef] [PubMed]
- Panginikkod, S.; Ramachandran, A.; Bollimunta, P.; Habibi, R.; Kumar Arjal, R.; Gopalakrishnan, V. Burkholderia aortic aneurysm: A case report and review of the literature. Case Rep. Infect. Dis. 2017. [Google Scholar] [CrossRef] [PubMed]
- Hadano, Y. Imported melioidosis in Japan: A review of cases. Infect. Drug Resist. 2018, 11, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Masbang, A.N. Melioidosis in the Philippines—Its Emergence from Obscurity: A Case Series; St. Luke’s Hospital: Quezon City, Philippines, 2015; unpublished work. [Google Scholar]
- Ocampo, L.D.M. Septicemic Melioidosis and Pulmonary Tuberculosis Co-Infection: A Case Report; Chinese General Hospital: Metro Manila, Philippines, 2014; unpublished work. [Google Scholar]
- Yap, E.M.; Rosario, M.; Inductivo-Yu, I. Liver abscess harbors melioidosis: A rare finding in a potentially endemic community. In Proceedings of the APASL Single Topic Conference, Kaohsiung, Taiwan, 10–12 June 2016; National Kidney and Transplant Institute: Metro Manila, Philippines, 2016, unpublished work. [Google Scholar]
- Santos, S.V.; Coronel, R.F.; Bergantin, M.R.G.; Delgado, J.S. Demographic Characteristics and Clinical Profile of Adult Patients with Burkholderia pseudomallei Infection at the University of Santo Tomas Hospital: A Case Series; University of Santo Thomas: Metro Manila, Philippines, 2014; unpublished. [Google Scholar]
- Hassan, M.R.; Pani, S.P.; Peng, N.P.; Voralu, K.; Vijayalakshmi, N.; Mehanderkar, R.; Aziz, N.A.; Michael, E. Incidence, risk factors and clinical epidemiology of melioidosis: A complex socio-ecological emerging infectious disease in the Alor Setar region of Kedah, Malaysia. BMC Infect. Dis. 2010, 10, 302. [Google Scholar] [CrossRef] [PubMed]
- Simpson, A.J.; Newton, P.N.; Chierakul, W.; Chaowagul, W.; White, N.J. Diabetes mellitus, insulin, and melioidosis in Thailand. Clin. Infect. Dis. 2003, 36. [Google Scholar] [CrossRef] [PubMed]
- Jimeno, C.A.; Kho, S.A.; Matawaran, B.J.; Duante, C.A.; Jasul, G.V. Prevalence of diabetes mellitus and pre-diabetes in the Philippines: A sub-study of the 7th National Nutrition and Health Survey (2008). Philipp. J. Intern. Med. 2015, 53, 1–8. [Google Scholar]
- World Health Organization. Philippines: WHO Statistical Profile. Country Statistics and Global Health Estimates by WHO and UN Partners. Available online: http://www.who.int/gho/countries/phl.pdf?ua=1 (accessed on 1 July 2018).
- Dance, D. Treatment and prophylaxis of melioidosis. Int. J. Antimicrob. Agents 2014, 43, 310–318. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Surveillance System in the Philippines—World Health Organization. Available online: www.wpro.who.int/philippines/mediacentre/features/surveillanceresponseevd.pdf (accessed on 1 July 2018).
| Year Published | Age | Sex | Place Diagnosed | Co-Morbidities/Risk Factors | Melioidosis Type | Method of Diagnosis | Antibiotics | Outcome | Reference |
|---|---|---|---|---|---|---|---|---|---|
| 1948 | 25 | M | No data | No data | Pulmonary | Sputum culture (guinea pig inoculation) | No data | No data | Gutner and Fisher [6] |
| 1957 | 32 | M | USA | No data | Cutaneous/soft tissue | Sputum culture (guinea pig inoculation); tissue (rib and femoral mass) culture | PEN, SD, STR, CLT, CHL | Recurrent | Prevatt and Hunt [10] |
| 1975 | 54 | M | USA | Lung carcinoma | Pulmonary | Sputum and bronchial washing cultures | TET, CHL | Died | Mays and Ricketts [19] |
| 1976 | 21 | M | USA | None | Pulmonary | Sputum culture | PEN, KAN, SXT | Improved | John [20] |
| 1978 | 23 | M | USA | None | Pulmonary | Sputum culture | TET, CHL, KAN, SXT | Improved | Fuller et al. [21] |
| 1985 | 46 | F | Taiwan | Drowning | Pulmonary | Blood culture | CEF, AMK | Improved | Lee et al. [11] |
| 1994 | 40 | M | Canada | Diabetes mellitus | Pulmonary | Sputum culture | CAZ | Improved | Turner et al. [22] |
| 2002 | 49 | M | Philippines | Diabetes mellitus | Disseminated (joint, pulmonary) | Blood culture | IPM, AMC, SXT | Improved | Ereno et al. [14] |
| 2008 | 64 | M | USA | Diabetes mellitus, coronary artery disease, hypertension | Pulmonary, osteomyelitis | Paraspinal abscess culture, PCR, isolate sent to CDC for confirmation | MEM, SXT | Improved | Falade et al. [23] |
| 2009 | 61 | M | USA | Metabolic syndrome | Pulmonary | Blood culture and pleural fluid culture, PCR | IMP, TGC, SXT, DOX | Improved | Duplessis and Maguire [24] |
| 2010 | 45 | F | Philippines | Diabetes mellitus | Pulmonary | Blood culture | TZP, CLI, AZM | Died | Velasco et al. [25] |
| 2011 | 50 | M | Brunei | Diabetes mellitus | Liver, spleen, cellulitis | Blood culture | No Data | Improved | Pande et al. [26] 1 |
| 2011 | 41 | M | Brunei | Diabetes mellitus | Cellulitis, pulmonary, liver | Blood culture | No Data | Improved | Pande et al. [26] 1 |
| 2014 | 62 | M | Canada | Diabetes mellitus, Sjogren syndrome, chronic kidney disease, Stevens-Johnson syndrome secondary to amoxicillin, schistosomiasis, warm autoimmune hemolytic anemia | Musculoskeletal | Blood culture and PCR | LVX, MET, DOX, SXT | Improved | Chagla et al. [27] |
| 2015 | 60 | M | South Korea | None | Pulmonary | Tissue (lung) and sputum culture | No data | Improved | Kim et al. [28] |
| 2015 | 68 | M | USA | Diabetes mellitus, coronary artery disease, hypertension, paroxysmal SVT | Splenic | Splenic abscess culture | CAZ, DOX, SXT | Improved | Guo et al. [29] |
| 2016 | 44 | M | Philippines | Diabetes mellitus | Hepatic | Liver abscess culture | MEM, SXT | Improved | San Martin et al. [15] |
| 2016 | 67 | F | USA | Diabetes mellitus, cardiovascular disease, tuberculosis | Mycotic aneurysm | Blood culture, PCR, isolate sent to CDC for confirmation | MEM, SXT | Died | Hemarajata et al. [30] |
| 2017 | 60 | M | USA | Diabetes mellitus, hypertension, dyslipidemia | Fever, loss of appetite, myalgia, weight loss | Blood culture, isolate sent to CDC for confirmation | CAZ, SXT | Improved | Singh and Mahmood [31] |
| 2017 | 82 | M | USA | Hypertension, hyperlipidemia, osteoarthritis | Mycotic aneurysm | Blood culture | CAZ | Improved | Panginnikod et al. [32] |
| 2018 | 41 | M | Japan | Unknown | Pulmonary, septic arthritis (soft tissue) | Blood culture | DOR | Died | Hadano et al. [33] |
| Year Diagnosed | Age | Sex | Co-Morbidities/Risk Factors | Melioidosis Type | Culture Specimen | Antibiotics | Outcome | Reference |
|---|---|---|---|---|---|---|---|---|
| 2010 | 54 | M | Diabetes mellitus | Pulmonary | Sputum | TZP, CLR | Improved | Masbang [34] |
| 2010 | 62 | M | Diabetes mellitus, cardiovascular disease | Disseminated (cutaneous/soft tissue, pulmonary) | Blood | CAZ, SXT | Improved | Masbang [34] |
| 2010 | 55 | M | Heavy alcohol consumption | Pulmonary, neurologic | Bronchial washing | MEM, CAZ, SXT | Improved | Masbang [34] |
| 2011 | 66 | M | Squamous cell lung carcinoma, hypertension, heavy smoker | Pulmonary | Sputum | MEM, AMC | Improved | Masbang [34] |
| 2011 | 54 | F | Diabetes mellitus, hypertension | Pulmonary | Blood | FEP | Improved | Masbang [34] |
| 2012 | 60 | M | Hypertension | Pulmonary | Sputum | MEM, SXT | Improved | Masbang [34] |
| 2013 | 45 | M | Diabetes mellitus | Neurologic | Sputum | MEM, CAZ, DOX | Improved | Masbang [34] |
| 2014 | 57 | F | Lung adenocarcinoma, hypertension | Pulmonary | Blood | CAZ, LVX | Died | Masbang [34] |
| 2014 | 38 | F | Breast cancer | Pulmonary | Endotracheal aspirate | CAZ | Died | Masbang [34] |
| 2014 | 64 | M | Hypertension | Cutaneous/soft tissue | Abscess | CAZ | No data | Masbang [34] |
| 2014 | 47 | M | Cardiovascular disease | Pulmonary | Sputum | CAZ | No data | Masbang [34] |
| 2014 | 59 | M | Pulmonary tuberculosis | Pulmonary, cutaneous/soft tissue | Blood, sputum, and wound swab | FEP, CLI, MEM, SXT, TB antibiotics (Isoniazid, Rifampicin, Ethambutol, Pyrazinamide) | Improved | Ocampo [35] |
| 2013 | 40 | M | Diabetes mellitus, poultry worker | Disseminated (soft tissue, liver, intra-abdominal infection) | Abscess | CAZ, SXT, DOX | Improved | Yap et al. [36] |
| 2009 | 34 | M | Diabetes mellitus | Pulmonary | Blood | CAZ, SXT | Improved | Santos [37] |
| 2008 | 36 | M | Diabetes mellitus | Hepatic | Abscess | CAZ, SXT | Improved | Santos [37] |
| 2012 | 59 | M | Diabetes mellitus | Disseminated (cutaneous, joint, pulmonary) | Synovial fluid, wound swab | CAZ, SXT, DOX | Improved | Santos [37] |
| 2009, 2012 | 44 | M | Diabetes mellitus | Soft tissue (parotid), neurologic (chronic and recurrent) | Abscess | 2009: CAZ, CIP, 2012: CAZ DOX, CHL 1 | Recurrent | Santos [37] |
| 2009 | 50 | M | Diabetes mellitus, hypertension, coronary artery disease | Pulmonary | Pleural fluid | CAZ, SXT, DOX | Improved | Santos [37] |
| 2011 | 43 | M | Diabetes mellitus, hypertension | Neurologic | Frontal cortex abscess | CAZ, SXT | Improved | Santos [37] |
| 2009 | 58 | M | Diabetes mellitus, hypertension | Urinary tract, CKD | Urine | CAZ, SXT | Improved | Santos [37] |
| Clinical Characteristics | Number of Cases |
|---|---|
| Mean Age | 50.2 |
| Sex | |
| Male | 35 (85.4%) |
| Female | 6 (14.6%) |
| Comorbidities | |
| Diabetes mellitus | 24 (58.5%) |
| Cardiovascular disease | 11 (26.82%) |
| Cancer | 4 (9.76%) |
| Pulmonary tuberculosis | 2 (4.9%) |
| Chronic kidney disease | 1 (2.4%) |
| Drowning | 1 (2.4%) |
| Heavy alcohol consumption | 1 (2.4%) |
| Hyperlipidemia | 1 (2.4%) |
| Osteoarthritis | 1 (2.4%) |
| Organs involved | |
| Pulmonary | 22 (53.7%) |
| Soft tissue | 12 (29.3%) |
| Hepatic | 5 (12.2%) |
| Neurologic | 4 (9.76%) |
| Splenic | 2 (4.9%) |
| Mycotic aneurysm | 2 (4.9%) |
| Osteomyelitis | 1 (2.4%) |
| Positive cultures | |
| Blood | 17 (41.5%) |
| Respiratory (sputum) | 16 (39.0%) |
| Abscess pus | 7 (17.1%) |
| Wound swab/Tissue | 3 (7.3%) |
| Synovial fluid | 1 (2.4%) |
| Outcome | |
| Improved | 22 (53.7%) |
| Relapsed | 2 (4.9%) |
| Died | 6 (14.6%) |
| No Data | 5 (12.2%) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
San Martin, P.F.M.; Chua, J.C.; Bautista, R.L.P.; Nailes, J.M.; Panaligan, M.M.; Dance, D.A.B. Melioidosis in the Philippines. Trop. Med. Infect. Dis. 2018, 3, 99. https://doi.org/10.3390/tropicalmed3030099
San Martin PFM, Chua JC, Bautista RLP, Nailes JM, Panaligan MM, Dance DAB. Melioidosis in the Philippines. Tropical Medicine and Infectious Disease. 2018; 3(3):99. https://doi.org/10.3390/tropicalmed3030099
Chicago/Turabian StyleSan Martin, Peter Franz M., Joseph C. Chua, Ralph Louie P. Bautista, Jennifer M. Nailes, Mario M. Panaligan, and David A. B. Dance. 2018. "Melioidosis in the Philippines" Tropical Medicine and Infectious Disease 3, no. 3: 99. https://doi.org/10.3390/tropicalmed3030099
APA StyleSan Martin, P. F. M., Chua, J. C., Bautista, R. L. P., Nailes, J. M., Panaligan, M. M., & Dance, D. A. B. (2018). Melioidosis in the Philippines. Tropical Medicine and Infectious Disease, 3(3), 99. https://doi.org/10.3390/tropicalmed3030099






