Abstract
The first documented case of melioidosis in the Philippines occurred in 1948. Since then, there have been sporadic reports in the literature about travelers diagnosed with melioidosis after returning from the Philippines. Indigenous cases, however, have been documented rarely, and under-reporting is highly likely. This review collated all Philippine cases of melioidosis published internationally and locally, as well as unpublished case series and reports from different tertiary hospitals in the Philippines. In total, 25 papers and 41 cases were identified. Among these, 23 were indigenous cases (of which 20 have not been previously reported in the literature). The most common co-morbidity present was diabetes mellitus, and the most common presentations were pulmonary and soft tissue infections. Most of the cases received ceftazidime during the intensive phase, while trimethoprim-sulfamethoxazole was given during the eradication phase. The known mortality rate was 14.6%, while 4.9% of all cases were reported to have had recurrence. The true burden of melioidosis in the country is not well defined. A lack of awareness among clinicians, a dearth of adequate laboratories, and the absence of a surveillance system for the disease are major challenges in determining the magnitude of the problem.
1. Introduction and History of Melioidosis in the Philippines
Melioidosis, or Whitmore’s disease, is a potentially lethal infection caused by the Gram-negative bacterium Burkholderia pseudomallei. Its clinical presentation is so diverse that the diagnosis relies heavily on laboratory culture.
During the past few decades, melioidosis has emerged as an important public health concern, especially in the South East Asian (SEA) region and northern Australia [1]. The increase in global interest in the disease was a consequence of the rising number of reported cases, both within endemic areas and among travelers, the occurrence of cases in areas not previously known to be endemic; and also, its potential use as an agent of bioterrorism.
After the disease was first described by Alfred Whitmore and C.S. Krishnaswami in 1912 [1,2,3], initially the majority of cases reported were from the SEA region. Reviews of the global distribution of the disease showed that cases have been found in the tropical and subtropical regions, mainly between 20° north and 20° south of the equator—which includes the Philippines [2,4]. A recent modelling study suggested that there could be as many as 9000 cases and 4500 deaths, due to melioidosis occurring in the Philippines each year [5]. Certainly, nowhere near that number are being diagnosed, so how likely is this to be true?
The first documented case of human melioidosis in the Philippines occurred in 1948 in a 25-year-old American soldier who presented with weight loss and symptoms of pneumonia. Diagnosis was established through guinea pig inoculation of sputum and lymph node pus [6]. This was not long after the disease had first been recognized in countries like Vietnam [7] and Indonesia [8], and before the first indigenous case was described in Thailand [9]. However, in contrast to other SEA countries, there has been little development with regards to defining the epidemiology of the disease in the Philippines. A further case in an American soldier was reported in 1957, although his illness had started many years earlier, while he was stationed in the Philippines [10]. Sporadic cases were reported thereafter in people who had either transited or stayed in the country. Some of these individuals also had a history of travel to other parts of SEA, hence the evidence that the infection was acquired in the Philippines was inconclusive [2]. However, in others, such as the first case diagnosed in Taiwan in 1985, there was strong circumstantial evidence that infection was acquired in the Philippines during a near-drowning incident in a river near Manila [11].
Cases have not been limited to humans: For example, an isolate from a horse from the Philippines was included in a taxonomic study published in 1956, although no further clinical details are available about this case [12]. Then in 1992, a number of primates imported from the Philippines to the United Kingdom were involved in an outbreak of melioidosis [13]. However, there have been no other cases among animals from the Philippines reported in the literature thereafter.
Indigenous cases, meaning those with no apparent travel history to other endemic areas, and who were diagnosed and treated in the Philippines, had never been reported in the literature until 2002, when a 49-year-old farmer from Bulacan in the Philippines presented with pain and swelling of the left shoulder and was found to have melioidosis after Burkholderia pseudomallei was isolated from blood [14]. The second indigenous case, and the first case published in an international journal, was reported as recently as 2016 [15]. The patient was a farmer from Isabela with no history of travel outside the country, who was diagnosed and treated in a hospital in Metro Manila.
The preponderance of reports of melioidosis among travelers from the Philippines, and the paucity of indigenous cases, leads us to suspect that the disease is being grossly under-reported in the country. This article describes the evidence for, and distribution of, melioidosis in the Philippines by reviewing reports published in international and local publications and unpublished case reports that were obtained from tertiary hospitals and local subspecialty organizations. These cases were examined to determine common patient profiles, disease presentations, modes of diagnosis, treatment received, and outcomes.
2. Materials and Methods
2.1. Review of Publications
Published reports citing cases of culture-confirmed melioidosis from the Philippines were collected. The melioidosis website (www.melioidosis.info) served as the main starting point for the search, as it already contains a database of published case reports by country. In addition, PubMed and Google Scholar were searched, using various combinations of keywords (and MESH terms, where applicable) such as Philippines, Filipino, Southeast Asia, melioidosis, Whitmore’s disease, Burkholderia pseudomallei, and Pseudomonas pseudomallei. The bibliography of each reference was searched for further cases. Finally, the personal EndNote database of one of us (DABD) was searched for references that had been missed. From each reference, relevant information about each case was extracted, such as age, sex, co-morbidities, risk factors, type of infection, mode of diagnosis, treatment received, outcome, country where diagnosed, and year of diagnosis.
2.2. Collection of Locally Published and Unpublished Cases from the Philippines
Local medical journals and university papers available in the National Library of the Philippines, different universities, and hospitals were searched for any mention of local cases of melioidosis. Inquiries regarding cases of melioidosis were made to the Philippine Society for Microbiology and Infectious Diseases, the Research Institute for Tropical Medicine, and other medical centers. Consent to use the data was obtained from the authors of unpublished case series and reports that were collected.
3. Results and Discussion
A total of 25 articles, either single case reports or case series, describing culture-confirmed melioidosis in the Philippines were collected (Table 1 and Table 2). Of these 25 papers, only 11 were listed in the melioidosis.org database—although those not included had either been published or presented at international conferences recently (between 2015 and 2017). This also included four unpublished local reports which were obtained from different tertiary hospitals in Metro Manila. Only one of these reports related to a case of melioidosis in an animal [13].

Table 1.
Clinical characteristics of culture confirmed cases of melioidosis in the Philippines from published case reports.

Table 2.
Clinical characteristics of confirmed cases of melioidosis in the Philippines from unpublished case reports and series.
A total of 41 human cases was included in these articles, with 18 cases involving travelers and 23 indigenous cases. Among the indigenous cases, 20 were from the four previously unpublished reports. Of these four unpublished papers, two were case series derived from a 3- to 5-year chart review of patients that were culture-positive for B. pseudomallei from their respective institution, and the other two were single case reports. Diagnosis was established via culture-based technique and VITEK among indigenous cases. No additional molecular confirmatory tests were performed for these specimens.
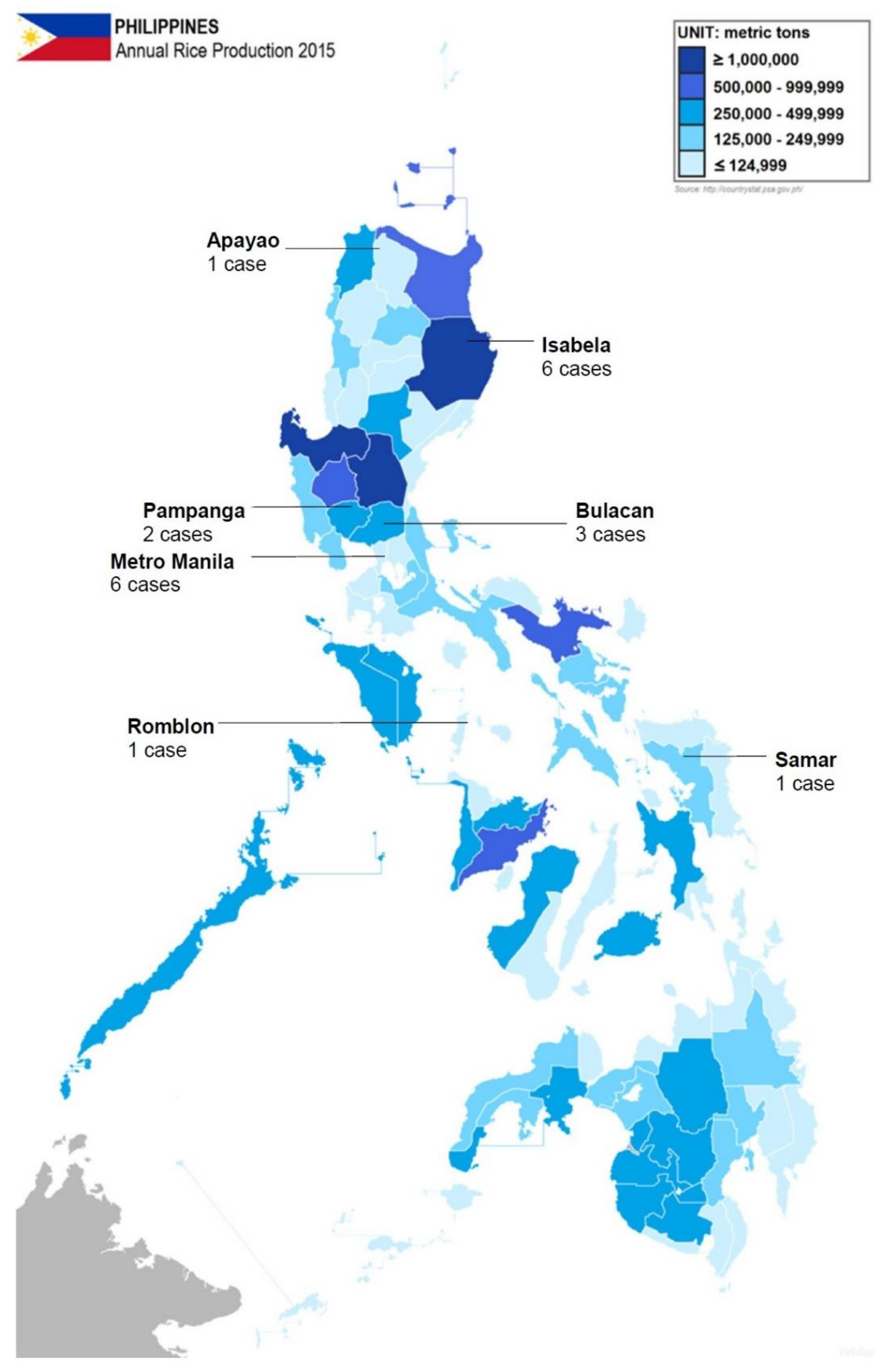
The patients were between the ages of 21 and 82 years, with a mean of 50.2 years (Table 3). The patients were predominantly (85.4%) male. This is consistent with the epidemiologic data from some other endemic areas [16,17,18]. Data on risk factors were limited, but only six patients were known to be working in the agricultural sector. Geographical data were available for 20 indigenous cases, where 11 were from areas with moderate to high rice production in the Philippines (Isabela, Pampanga, and Bulacan). Figure 1 shows the geographical distribution of the reported cases in the Philippines, and annual rice production per province. Although the number of cases in Metro Manila is inconsistent with the expected high incidence of melioidosis among rice farmers, the authors suspected that these patients had previous exposure to agricultural areas. In addition, the proximity and access to diagnostic facility could have caused the relatively higher proportion of cases in Manila. All the indigenous cases were diagnosed in tertiary hospitals in Metro Manila, which generally have better-equipped laboratory and diagnostic facilities than most provincial hospitals, and it is likely that many patients are going undiagnosed outside the city.

Table 3.
Summary of clinical characteristics of culture-confirmed cases of melioidosis in the Philippines.
Figure 1.
Distribution of indigenous cases of melioidosis in the Philippines (reported from 2002 to 2016) and the amount of rice production per province. Original image imported from: https://commons.wikimedia.org/wiki/File:Philippine_provinces_Annual_Rice_Production_2015.png.
The most common co-morbidity was diabetes mellitus, comprising 58.5% of cases, followed by cardiovascular disease (which includes hypertension and coronary artery disease) at 26.82%. Diabetes mellitus is the most common co-morbidity and risk factor found in the literature [16,38,39]. With the steadily increasing prevalence of type 2 diabetes in the Philippines [40], it is likely that the incidence of melioidosis will increase correspondingly.
Pneumonia and soft tissue infection were present in 53.7% and 29.3% of cases respectively, which again is consistent with the melioidosis literature from elsewhere [17]. Pulmonary diseases, particularly pneumonia, chronic lower respiratory tract diseases, and tuberculosis, are among the top 10 causes of mortality in the Philippines [41]. Since melioidosis may be difficult to differentiate from other respiratory diseases, it is possible that many cases labelled simply as ‘pneumonia’ or other respiratory infections may actually have melioidosis. A high index of suspicion, and good laboratory support, are necessary to make a specific diagnosis of melioidosis. Furthermore, co-infection with B. pseudomallei and Mycobacterium tuberculosis was seen in two patients in this review, which presents a further diagnostic and therapeutic challenge to clinicians. Other than pulmonary cases, four neurological cases were reported among indigenous cases. Although, conclusions could not be drawn, due to limited number of cases, this should be subject for further investigation if this type of melioidosis occur more frequently in the country.
All cases were diagnosed by culture studies. Bacteremia was found in 41.5% of cases. The most common antibiotic used were ceftazidime and trimethoprim-sulfamethoxazole for the intensive phase and eradication phase, respectively. Most of the cases received the recommended antibiotic treatment for melioidosis [42] such as ceftazidime (43.59%), meropenem (12.82%), trimethoprim-sulfamethoxazole (53.84%), and amoxicillin-clavulanic acid (5.13%). Those treated with different antibiotics were cases diagnosed earlier than 1990s, and those indigenous cases which followed the result of the antibiotic sensitivity test. The reported recurrence rate among all cases were 4.9%, while the mortality rate was 12.6%. This, however, could be an underestimate due to lack of data and follow-up.
Failure to consider melioidosis, which requires specific treatment, may be contributing to the continued high mortality from pulmonary diseases in the Philippines. Local clinical guidelines do not include ceftazidime and meropenem as empiric treatment unless classified as high-risk community-acquired pneumonia, healthcare-associated pneumonia, or those at risk for multidrug-resistant organisms.
4. Challenges
The greatest challenges for the Philippines are to raise awareness of melioidosis amongst clinicians and laboratory staff, and to improve the availability of facilities capable of making a laboratory diagnosis of the disease. During our enquiries with colleagues and local institutions about case reports, many of those who responded admitted to being unfamiliar with melioidosis. Also, melioidosis is rarely included in the curriculum of medical schools within the Philippines. It is notable that the majority of the earlier cases in this series were diagnosed in travelers to other countries, albeit only 21 cases occurring over 70 years, implying considerable under-diagnosis of indigenous cases. However, there are some encouraging signs, particularly, the fact that four unpublished reports, comprising 20 indigenous cases seen over a relatively short span of seven years, were identified. This suggests an increasing awareness of the disease among physicians, resulting in improved diagnosis and reporting. However, these were all diagnosed in Metro Manila, which is unlikely to be the area of highest incidence in a country where the environment is predicted to be widely suitable for B. pseudomallei [5], and the incidence of melioidosis in more rural areas requires further investigation. Areas such as those in Mindanao, which are a highly rural area, post additional challenge since access to health care facilities is another existing issue. However, this will require the strengthening of medical laboratories in these areas, and education of staff in how to detect and identify B. pseudomallei.
Although the most common presentation of B. pseudomallei infection is pneumonia, the 2016 community-acquired pneumonia (CAP) guidelines in the Philippines do not even mention melioidosis as a differential diagnosis. There is no national surveillance system for melioidosis, nor is the disease included in the list of notifiable disease in the Philippines [43].
5. Conclusions and Recommendations
This report highlights the fact that melioidosis is indeed endemic in the Philippines but is likely to be grossly under-reported. The clinical and epidemiological characteristics of the disease in the Philippines, albeit based on limited data, are consistent with those described in published literature from other countries. Furthermore, the data show that this condition has been successfully diagnosed and treated in the country with increasing frequency over the past few years.
Further studies should be done on the extent and impact of this disease within the country. It is recommended that a program of education of clinicians and laboratory staff about melioidosis should be initiated, ideally starting at medical schools, and especially targeted at rural areas that are likely to have the highest incidence of melioidosis. Furthermore, consideration should be given to making the disease statutorily notifiable. This would provide more accurate information about the clinical characteristics and distribution of the disease within the Philippines. Soil sampling studies should also be undertaken in order to establish the geographic distribution of this microorganism, as well as enable analysis of the population structure. Genotyping of bacterial isolates are in progress in order to determine if indigenous strains were different than those identified elsewhere in Southeast Asia or Australia. Studies should also be undertaken to describe the current knowledge and attitudes of Filipino clinicians regarding melioidosis.
Author Contributions
Conceptualization, P.F.M.S.M. and D.A.B.D.; Data Curation, P.F.M.S.M. and J.C.C.; Methodology, P.F.M.S.M., J.C.C., R.L.P.B.; Investigation, P.F.M.S.M., J.C.C., R.L.P.B.; Resources, J.M.N. and M.M.P.; Supervision: D.A.B.D. and M.M.P.; Visualization: J.C.C., R.L.P.B.; Project administration, P.F.M.S.M. and J.M.N.; Writing original draft, P.F.M.S.M., J.C.C., R.L.P.B.; Writing review and editing, P.F.M.S.M., D.A.B.D., M.M.P.
Funding
This research received no external funding.
Acknowledgments
Authors would like to extend their sincerest gratitude to Armin Masbang, Lisette Ocampo, Suzanne Santos, and Emily Yap for sharing their unpublished case reports and case series.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Cheng, A.C.; Currie, B.J. Melioidosis: Epidemiology, pathophysiology, and management. Clin. Microbiol. Rev. 2005, 18, 383–416. [Google Scholar] [CrossRef] [PubMed]
- Dance, D.A. Melioidosis: The tip of the iceberg? Clin. Microbiol. Rev. 1991, 4, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Whitmore, A. An account of a glanders-like disease occurring in Rangoon. J. Hyg. 1913, 13, 1–34. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.J.; Allen, J.C.; Embi, M.N.; Othman, O.; Razak, N.; Ismail, G. Human melioidosis: An emerging medical problem. MIRCEN J. 1987, 3. [Google Scholar] [CrossRef]
- Limmathurotsakul, D.; Golding, N.; Dance, D.A.B.; Messina, J.P.; Pigott, D.M.; Moyes, C.L.; Rolim, D.B.; Bertherat, E.; Day, N.P.; Peacock, S.J.; et al. Predicted global distribution of Burkholderi pseudomallei and burden of melioidosis. Nat. Microbiol. 2016, 1. [Google Scholar] [CrossRef] [PubMed]
- Gutner, L.B.; Fisher, M.W. Chronic melioidosis: Discussion, case report, and special studies. Ann. Intern. Med. 1948, 28, 1157–1169. [Google Scholar] [CrossRef]
- Pons, R.; Advier, M. Melioidosis in Cochin China. J. Hyg. 1927, 26, 28–30. [Google Scholar] [CrossRef] [PubMed]
- De Moor, C.E.; Soekarnen Walle, N. Melioidosis op Java. Mededeeling uit het Geneeskundig Laboratorium te Weltevreden 1932, 72, 1618–1635. [Google Scholar]
- Chittivej, C.; Buspavanich, S.; Chaovanasai, A. Melioidosis with case report in a Thai. R. Thai Army Med. J. 1955, 68, 11–17. [Google Scholar]
- Prevatt, A.L.; Hunt, J.S. Chronic systemic melioidosis. Am. J. Med. Sci. 1957, 23, 810–823. [Google Scholar] [CrossRef]
- Lee, N.; Wu, J.L.; Lee, C.H.; Tsai, W.C. Pseudomonas pseudomallei infection from drowning: The first reported case in Taiwan. J. Clin. Microbiol. 1985, 22, 352–354. [Google Scholar] [PubMed]
- Wetmore, P.W.; Gochenour, W.S. Comparative studies of the genus Malleomyces and selected Pseudomonas species. J. Bacteriol. 1956, 72, 79–89. [Google Scholar] [PubMed]
- Dance, D.A.; King, C.; Aucken, H.; Knott, C.D.; West, P.G.; Pitt, T.L. An outbreak of melioidosis in imported primates in Britain. Vet. Rec. 1992, 130, 525–529. [Google Scholar] [CrossRef] [PubMed]
- Ereno, I.L.; Mariano, N.; Reyes, J.; Amando, C. Melioidosis: A case report. Philipp. J. Microbiol. Infect. Dis. 2002, 31, 125–133. [Google Scholar]
- Martin, P.F.M.S.; Teh, C.S.C.; Casupang, M.A.J. Melioidosis: A rare cause of liver abscess. Case Rep. Hepatol. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Kingsley, P.V.; Leader, M.; Nagodawithana, N.S.; Tipre, M.; Sathiakumar, N. Melioidosis in Malaysia: A review of case reports. PLoS Negl. Trop. Dis. 2016, 10, e0005182. [Google Scholar] [CrossRef] [PubMed]
- Meumann, E.M.; Cheng, A.C.; Ward, L.; Currie, B.J. Clinical features and epidemiology of melioidosis pneumonia: Results from a 21-year study and review of the literature. Clin. Infect. Dis. 2012, 54, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Churuangsuk, C.; Chusri, S.; Hortiwakul, T.; Charernmak, B.; Silpapojakul, K. Characteristics, clinical outcomes and factors influencing mortality of patients with melioidosis in southern Thailand: A 10-year retrospective study. Asian Pac. J. Trop. Med. 2016, 9, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Mays, E.E.; Ricketts, E.A. Melioidosis: Recrudescence associated with bronchogenic carcinoma twenty-six years following initial geographic exposure. Chest 1975, 68, 261–263. [Google Scholar] [CrossRef] [PubMed]
- John, J.F., Jr. Trimethoprim-sulfamethoxazole therapy of pulmonary melioidosis. Am. Rev. Respir. Dis. 1976, 114, 1021–1025. [Google Scholar] [CrossRef] [PubMed]
- Fuller, P.B.; Fisk, D.E.; Byrd, R.B.; Griggs, G.A.; Smith, M.R. Treatment of pulmonary melioidosis with combination of trimethoprim and sulfamethoxazole. Chest 1978, 74, 222–224. [Google Scholar] [CrossRef] [PubMed]
- Turner, M.O.; Lee, V.T.; FitzGerald, J.M. Melioidosis in a diabetic sailor. Chest 1994, 106, 952–954. [Google Scholar] [CrossRef] [PubMed]
- Falade, O.O.; Antonarakis, E.S.; Kaul, D.R.; Saint, S.; Murphy, P.A. Clinical problem-solving. Beware of first impressions. N. Engl. J. Med. 2008, 359, 628–634. [Google Scholar] [CrossRef] [PubMed]
- Duplessis, C.; Maguire, J.D. Melioidosis masquerading as community-acquired pneumonia: A case report demonstrating efficacy of intrapleural fibrinolytic therapy. J. Travel. Med. 2009, 16, 74–77. [Google Scholar] [CrossRef] [PubMed]
- Velasco, A.B.; Untalan, C.A.; Visperas, J.C. Melioiodisis presenting as severe community acquired pneumonia. Am. J. Respir. Crit. Care 2010, 181. [Google Scholar] [CrossRef]
- Pande, K.C.; Kadir, K.A. Melioidosis of the extremities in Brunei Darussalam. Singap. Med. J. 2011, 52, 346–350. [Google Scholar]
- Chagla, Z.; Aleksova, N.; Quirt, J.; Emery, J.; Kraeker, C.; Haider, S. Melioidosis in a returned traveller. Can. J. Infect. Dis. Med. Microbiol. 2014, 25, 225–226. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.W.; Kwon, G.-Y.; Kim, B.; Kwon, D.; Shin, J.; Bae, G.-R. Imported melioidosis in South Korea: A case series with a literature review. Osong Public Health Res. Perspect. 2015, 6, 363–368. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.F.; Wong, F.L.; Perez, M.L. Splenic abscesses in a returning traveler. Infect. Dis. Rep. 2015, 7. [Google Scholar] [CrossRef] [PubMed]
- Hemarajata, P.; Baghdadi, J.D.; Hoffman, R.; Humphries, R.M. Burkholderia pseudomallei: Challenges for the clinical microbiology laboratory. J. Clin. Microbiol. 2016, 54, 2866–2873. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Mahmood, M. Melioidosis: The great mimicker. J. Community Hosp. Intern. Med. Perspect. 2017, 7, 245–247. [Google Scholar] [CrossRef] [PubMed]
- Panginikkod, S.; Ramachandran, A.; Bollimunta, P.; Habibi, R.; Kumar Arjal, R.; Gopalakrishnan, V. Burkholderia aortic aneurysm: A case report and review of the literature. Case Rep. Infect. Dis. 2017. [Google Scholar] [CrossRef] [PubMed]
- Hadano, Y. Imported melioidosis in Japan: A review of cases. Infect. Drug Resist. 2018, 11, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Masbang, A.N. Melioidosis in the Philippines—Its Emergence from Obscurity: A Case Series; St. Luke’s Hospital: Quezon City, Philippines, 2015; unpublished work. [Google Scholar]
- Ocampo, L.D.M. Septicemic Melioidosis and Pulmonary Tuberculosis Co-Infection: A Case Report; Chinese General Hospital: Metro Manila, Philippines, 2014; unpublished work. [Google Scholar]
- Yap, E.M.; Rosario, M.; Inductivo-Yu, I. Liver abscess harbors melioidosis: A rare finding in a potentially endemic community. In Proceedings of the APASL Single Topic Conference, Kaohsiung, Taiwan, 10–12 June 2016; National Kidney and Transplant Institute: Metro Manila, Philippines, 2016, unpublished work. [Google Scholar]
- Santos, S.V.; Coronel, R.F.; Bergantin, M.R.G.; Delgado, J.S. Demographic Characteristics and Clinical Profile of Adult Patients with Burkholderia pseudomallei Infection at the University of Santo Tomas Hospital: A Case Series; University of Santo Thomas: Metro Manila, Philippines, 2014; unpublished. [Google Scholar]
- Hassan, M.R.; Pani, S.P.; Peng, N.P.; Voralu, K.; Vijayalakshmi, N.; Mehanderkar, R.; Aziz, N.A.; Michael, E. Incidence, risk factors and clinical epidemiology of melioidosis: A complex socio-ecological emerging infectious disease in the Alor Setar region of Kedah, Malaysia. BMC Infect. Dis. 2010, 10, 302. [Google Scholar] [CrossRef] [PubMed]
- Simpson, A.J.; Newton, P.N.; Chierakul, W.; Chaowagul, W.; White, N.J. Diabetes mellitus, insulin, and melioidosis in Thailand. Clin. Infect. Dis. 2003, 36. [Google Scholar] [CrossRef] [PubMed]
- Jimeno, C.A.; Kho, S.A.; Matawaran, B.J.; Duante, C.A.; Jasul, G.V. Prevalence of diabetes mellitus and pre-diabetes in the Philippines: A sub-study of the 7th National Nutrition and Health Survey (2008). Philipp. J. Intern. Med. 2015, 53, 1–8. [Google Scholar]
- World Health Organization. Philippines: WHO Statistical Profile. Country Statistics and Global Health Estimates by WHO and UN Partners. Available online: http://www.who.int/gho/countries/phl.pdf?ua=1 (accessed on 1 July 2018).
- Dance, D. Treatment and prophylaxis of melioidosis. Int. J. Antimicrob. Agents 2014, 43, 310–318. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Surveillance System in the Philippines—World Health Organization. Available online: www.wpro.who.int/philippines/mediacentre/features/surveillanceresponseevd.pdf (accessed on 1 July 2018).
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).