Feasibility of Utilizing Waste Natural Rubber Gloves as a Primary Rubber Matrix: Aspect of Vulcanization Systems
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation Continuous WNRG Sheet
2.3. Compound Preparation
2.4. Characterizations
2.4.1. Curing Characteristics
2.4.2. Swelling Equilibrium Test
2.4.3. Mechanical Properties
2.4.4. Wide-Angle X-Ray Scattering
2.4.5. Dynamic Mechanical Analysis
2.4.6. Morphological Properties
3. Results
3.1. Curing Characteristics
3.2. Swelling Properties and Total Crosslink Density
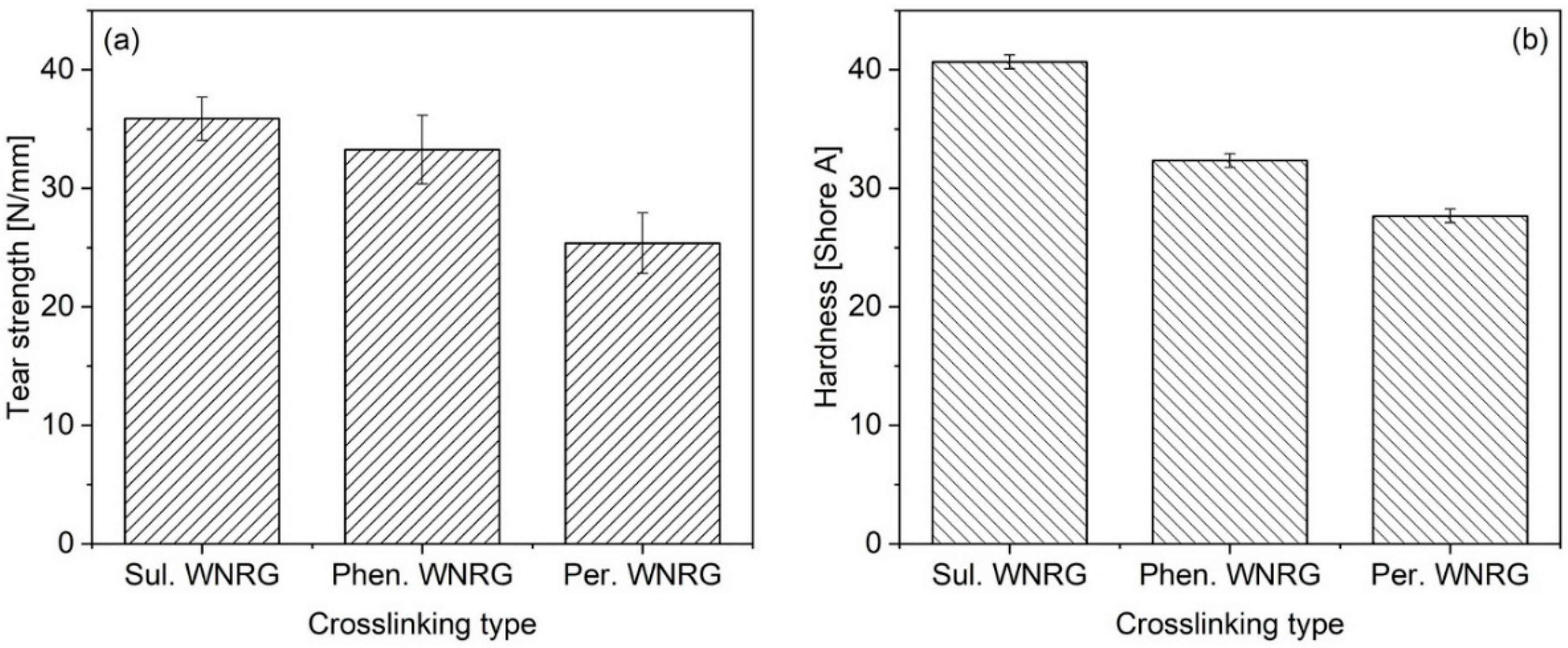
3.3. Mechanical Properties
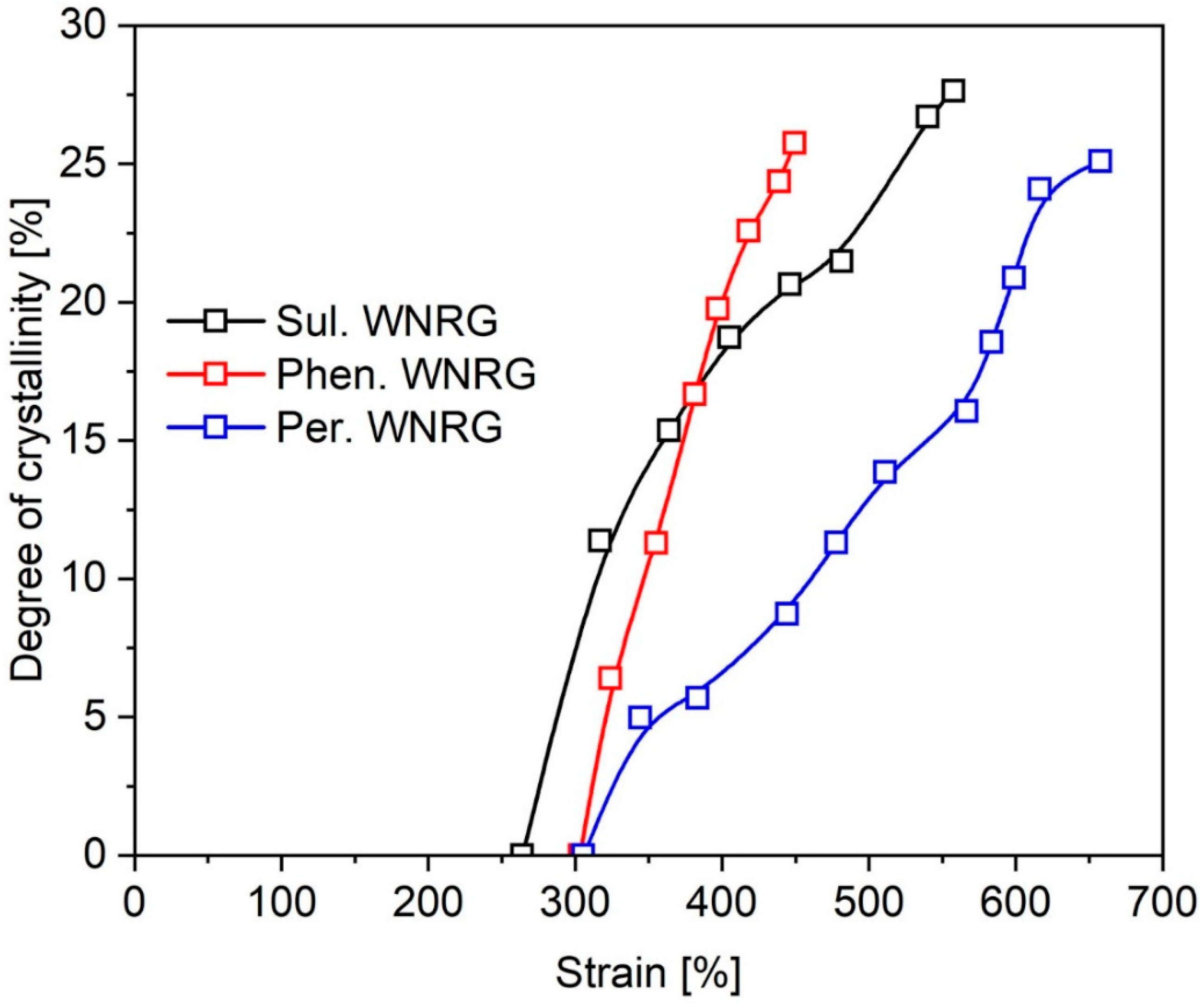
3.4. Strain-Induced Crystallization
3.5. Dynamic Mechanical Properties
3.6. Morphological Properties
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| WNRG | Waste natural rubber glove |
| NR | Natural rubber |
| TGA | Thermogravimetric analysis |
| ts1 | Scorch time |
| t90 | Cure time |
| CRI | Cure rate index |
| ML | Minimum torque |
| MH | Maximum torque |
| MH − ML | Torque difference |
| 100% Mod | 100% modulus |
| 300% Mod | 300% modulus |
| TS | Tensile strength |
| EB | Elongation at break |
| E′ | Storage modulus |
| Tg | Glass transition temperature |
| WAXS | Wide-angle X-ray scattering |
| Xc | Degree of crystallinity |
References
- Nuzaimah, M.; Sapuan, S.M.; Nadlene, R.; Jawaid, M. Recycling of waste rubber as fillers: A review. IOP Conf. Ser. Mater. Sci. Eng. 2018, 368, 012016. [Google Scholar] [CrossRef]
- Chittella, H.; Yoon, L.W.; Ramarad, S.; Lai, Z.W. Rubber waste management: A review on methods, mechanism, and prospects. Polym. Degrad. Stab. 2021, 194, 109761. [Google Scholar] [CrossRef]
- Yip, E.; Cacioli, P. The manufacture of gloves from natural rubber latex. J. Allergy Clin. Immunol. 2002, 110, S3–S14. [Google Scholar] [CrossRef]
- Hayeemasae, N.; Ismail, H.; Khoon, T.B.; Husseinsyah, S.; Harahap, H. Effect of carbon black on the properties of polypropylene/recycled natural rubber glove blends. Prog. Rubber Plast. Recycl. Technol. 2016, 32, 241–252. [Google Scholar] [CrossRef]
- Gedik, A.; Ozcan, O.; Ozcanan, S. Recycling COVID-19 health care wastes in bitumen modification: A case of disposable medical gloves. Environ. Sci. Pollut. Res. 2023, 30, 74977–74990. [Google Scholar] [CrossRef] [PubMed]
- Patarapaiboolchai, O.; Tulayapitak, T.; Sriwang, I. Recycled gloves as a filler. Songklanakarin J. Sci. Technol. 2002, 24, 701–706. [Google Scholar]
- Nuzaimah, M.; Sapuan, S.M.; Nadlene, R.; Jawaid, M. Microstructure and mechanical properties of unsaturated polyester composites filled with waste rubber glove crumbs. Fibers Polym. 2019, 20, 1290–1300. [Google Scholar] [CrossRef]
- Saiwari, S.; Lohyi, E.; Nakason, C. Application of NR gloves reclaim: Cure and mechanical properties of NR/reclaim rubber blends. Adv. Mater. Res. 2014, 844, 437–440. [Google Scholar] [CrossRef]
- Saiwari, S.; Yusoh, B.; Thitithammawong, A. Recycled rubber from waste of natural rubber gloves blending with polypropylene for preparation of thermoplastic vulcanizates compatibilized by maleic anhydride. J. Polym. Environ. 2019, 27, 1141–1149. [Google Scholar] [CrossRef]
- Phummor, P.; Riyajan, S.A.; Tangboriboonrat, P. Physical properties of polymer blend: Natural rubber glove waste/polystyrene foam waste/cellulose fiber via brabender. Adv. Mater. Res. 2014, 844, 57–60. [Google Scholar] [CrossRef]
- Riyajan, S.A.; Intharit, I.; Tangboriboonrat, P. Physical properties of polymer composite: Natural rubber glove waste/polystyrene foam waste/cellulose. Ind. Crops Prod. 2012, 36, 376–382. [Google Scholar] [CrossRef]
- Mousavi, S.S.; Dehestani, M. Influence of latex and vinyl disposable gloves as recycled fibers in 3D printing sustainable mortars. Sustainability 2022, 14, 9908. [Google Scholar] [CrossRef]
- Masa, A.; Soontaranon, S.; Hayeemasae, N. Influence of sulfur/accelerator ratio on tensile properties and structural inhomogeneity of natural rubber. Polym. Korea 2020, 44, 519–526. [Google Scholar] [CrossRef]
- Lopattananon, N.; Julyanon, J.; Masa, A.; Kaesaman, A.; Thongpin, C.; Sakai, T. The role of nanofillers on (natural rubber)/(ethylene vinyl acetate)/clay nanocomposite in blending and foaming. J. Vinyl Addit. Technol. 2015, 21, 134–146. [Google Scholar] [CrossRef]
- Masa, A.; Saito, R.; Saito, H.; Sakai, T.; Kaesaman, A.; Lopattananon, N. Phenolic resin-crosslinked natural rubber/clay nanocomposites: Influence of clay loading and interfacial adhesion on strain-induced crystallization behavior. J. Appl. Polym. Sci. 2016, 133, 43214. [Google Scholar] [CrossRef]
- ASTM D5289-19; Standard Test Method for Rubber Property—Vulcanization Using Rotorless Cure Meters. ASTM International: West Conshohocken, PA, USA, 2019.
- Flory, P.J. Statistical mechanics of swelling of network structures. J. Chem. Phys. 1950, 18, 108–111. [Google Scholar] [CrossRef]
- ISO 37:2017; Rubber, Vulcanized or Thermoplastic—Determination of Tensile Stress–Strain Properties. ISO: Geneva, Switzerland, 2017.
- ISO 34-1:2022; Rubber, Vulcanized or Thermoplastic—Determination of Tear Strength—Part 1: Trouser, Angle and Crescent Test Pieces. ISO: Geneva, Switzerland, 2022.
- ASTM D2240-03; Standard Test Method for Rubber Property—Durometer Hardness. ASTM International: West Conshohocken, PA, USA, 2003.
- Hayeemasae, N.; Soontaranon, S.; Rasidi, M.S.M.; Masa, A. Tensile and structural properties of natural rubber vulcanizates with different mastication times. Polímeros 2021, 31, e2021003. [Google Scholar] [CrossRef]
- Chenal, J.-M.; Chazeau, L.; Guy, L.; Bomal, Y.; Gauthier, C. Molecular weight between physical entanglements in natural rubber: A critical parameter during strain-induced crystallization. Polymer 2007, 48, 1042–1046. [Google Scholar] [CrossRef]
- Kruzelak, J.; Sykora, R.; Hudec, I. Sulphur and peroxide vulcanisation of rubber compounds—Overview. Chem. Pap. 2016, 70, 1533–1555. [Google Scholar] [CrossRef]
- Ismail, H.; Kamal, S.K.; Mark, S.E. The comparison effects of palm oil fatty acid and stearic acid in various natural rubber compounds. J. Elastomers Plast. 2001, 33, 100–113. [Google Scholar] [CrossRef]
- Mayasari, H.E.; Wirapraja, A.Y. The curing characteristics and tear properties of phenolic resin on chloroprene rubber vulcanizate. IOP Conf. Ser. Mater. Sci. Eng. 2020, 732, 012008. [Google Scholar] [CrossRef]
- Nakason, C.; Nakaramontri, Y.; Kaesaman, A.; Kangwansukpamonkon, W.; Kiatkamjornwong, S. Synthesis and characterization of water swellable natural rubber vulcanizates. Eur. Polym. J. 2013, 49, 1098–1110. [Google Scholar] [CrossRef]
- Du, M.; Liu, D. Synthesis and characterization of a novel aldehyde teroctyl phenolic resin vulcanizing agent for isoprene rubber processing. J. Polym. Res. 2023, 30, 249. [Google Scholar] [CrossRef]
- Hayeemasae, N.; Soontaranon, S.; Masa, A. Influence of different vulcanizing agents on structures and properties of sepiolite-filled natural rubber composites. Express Polym. Lett. 2023, 17, 181–195. [Google Scholar] [CrossRef]
- Bulbul, S. Improving the crosslink density and the mechanical properties after vulcanization for an iron oxide layer (scale) and SBR/rubber masterbatch. Mater. Technol. 2020, 54, 71–78. [Google Scholar] [CrossRef]
- Chudzik, J.; Bielinski, D.M.; Bratychak, M.; Demchuk, Y.; Astakhova, O.; Jedrzejczyk, M.; Celichowski, G. Influence of modified epoxy resins on peroxide curing, mechanical properties and adhesion of SBR, NBR and XNBR to silver wires. Part I: Application of monoperoxy derivative of epoxy resin (PO). Materials 2021, 14, 1320. [Google Scholar] [CrossRef] [PubMed]
- Tosaka, M.; Murakami, S.; Poompradub, S.; Kohjiya, S.; Ikeda, Y.; Toki, S.; Sics, I.; Hsiao, B.S. Orientation and crystallization of natural rubber network as revealed by WAXD using synchrotron radiation. Macromolecules 2004, 37, 3299–3309. [Google Scholar] [CrossRef]
- Che, J.; Burger, C.; Toki, S.; Rong, L.; Hsiao, B.S.; Amnuaypornsri, S.; Sakdapipanich, J. Crystal and crystallites structure of natural rubber and peroxide-vulcanized natural rubber by a two-dimensional wide-angle X-ray diffraction simulation method. II. Strain-induced crystallization versus temperature-induced crystallization. Macromolecules 2013, 46, 9712–9721. [Google Scholar] [CrossRef]
- Candau, N.; Oguz, O.; Federico, C.E.; Stoclet, G.; Tahon, J.F.; Maspoch, M.L. Strain induced crystallization in vulcanized natural rubber containing ground tire rubber particles with reinforcement and nucleation abilities. Polym. Test. 2021, 101, 107313. [Google Scholar] [CrossRef]
- Toki, S.; Sics, I.; Ran, S.; Liu, L.; Hsiao, B.S. Molecular orientation and structural development in vulcanized polyisoprene rubbers during uniaxial deformation by in situ synchrotron X-ray diffraction. Polymer 2003, 44, 6003–6011. [Google Scholar] [CrossRef]
- Tosaka, M. Strain-induced crystallization of crosslinked natural rubber as revealed by X-ray diffraction using synchrotron radiation. Polym. J. 2007, 39, 1207–1220. [Google Scholar] [CrossRef]
- Osaka, N.; Kato, M.; Saito, H. Mechanical properties and network structure of phenol resin crosslinked hydrogenated acrylonitrile-butadiene rubber. J. Appl. Polym. Sci. 2013, 129, 3396–3403. [Google Scholar] [CrossRef]
- Tosaka, M.; Senoo, K.; Kohjiya, S.; Ikeda, Y. Crystallization of stretched network chains in cross-linked natural rubber. J. Appl. Phys. 2007, 101, 084909. [Google Scholar] [CrossRef]
- Bashir, M.A. Use of dynamic mechanical analysis (DMA) for characterizing interfacial interactions in filled polymers. Solids 2021, 2, 108–120. [Google Scholar] [CrossRef]
- Bakosova, D.; Bakosova, A. Testing of rubber composites reinforced with carbon nanotubes. Polymers 2022, 14, 3039. [Google Scholar] [CrossRef] [PubMed]
- Paczkowski, P.; Puszka, A.; Gawdzik, B. Green composites based on unsaturated polyester resin from recycled poly (ethylene terephthalate) with wood flour as filler-Synthesis, characterization and aging effect. Polymers 2020, 12, 2966. [Google Scholar] [CrossRef] [PubMed]
- Bandzierz, K.; Reuvekamp, L.; Dryzek, J.; Dierkes, W.; Blume, A.; Bielinski, D. Influence of network structure on glass transition temperature of elastomers. Materials 2016, 9, 607. [Google Scholar] [CrossRef]

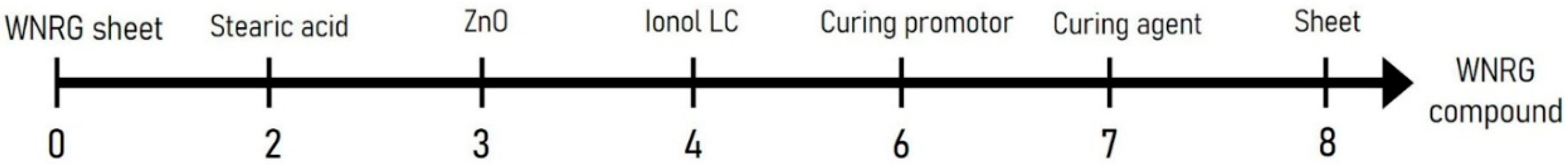







| Ingredient\Vulcanization System | Quantity (phr 1) | ||
|---|---|---|---|
| Sulfur | Phenolic Resin | Peroxide | |
| WNRG sheet | 100 | 100 | 100 |
| Stearic acid | 1 | - | - |
| ZnO | 3 | - | - |
| Ionol LC | 1 | 1 | 1 |
| CBS | 1.5 | - | - |
| Sulfur | 1.5 | - | - |
| SnCl2.2H2O | - | 1 | - |
| Phenolic resin | - | 10 | - |
| TMPTMA | - | - | 4 |
| DCP | - | - | 0.7 |
| Sample Name | Curing Parameter | |||||
|---|---|---|---|---|---|---|
| ML (dN·m) | MH (dN·m) | MH − ML (dN·m) | ts1 (min) | t90 (min) | CRI (min−1) | |
| Pure WNRG | 2.53 ± 0.29 | N/A | N/A | N/A | N/A | N/A |
| Sul. WNRG | 0.65 ± 0.00 | 10.45 ± 0.00 | 9.80 ± 0.00 | 0.83 ± 0.05 | 1.53 ± 0.02 | 142.86 ± 5.88 |
| Phen. WNRG | 0.85 ± 0.00 | 6.15 ± 0.00 | 6.15 ± 0.00 | 1.99 ± 0.05 | 17.59 ± 0.29 | 6.41 ± 0.18 |
| Per. WNRG | 0.91 ± 0.00 | 4.47 ± 0.00 | 3.56 ± 0.00 | 1.58 ± 0.09 | 9.99 ± 0.02 | 11.89 ± 0.17 |
| Sample Type | 100% Mod (MPa) | 300% Mod (MPa) | TS (MPa) | EB (%) |
|---|---|---|---|---|
| Sul. WNRG | 1.34 ± 0.10 | 3.98 ± 0.86 | 16.23 ± 1.88 | 569 ± 80 |
| Phen. WNRG | 0.73 ± 0.08 | 2.63 ± 0.49 | 12.32 ± 0.45 | 585 ± 52 |
| Per. WNRG | 0.61 ± 0.04 | 1.96 ± 0.60 | 10.74 ± 1.31 | 636 ± 76 |
| Neat WNRG | 1.22 ± 0.13 | 3.26 ± 0.22 | 21.03 ± 0.37 | 662 ± 18 |
| Sample Type | E′ at 25 °C (MPa) | Tg (°C) | tan delta Peak Width | tan delta Peak Height |
|---|---|---|---|---|
| Sul. WNRG | 4.53 | −43.04 | 19.02 | 1.48 |
| Phen. WNRG | 2.16 | −45.52 | 20.74 | 1.57 |
| Per. WNRG | 1.29 | −49.17 | 16.44 | 1.57 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Masa, A.; Mesa, N.; Soontaranon, S.; Hayeemasae, N. Feasibility of Utilizing Waste Natural Rubber Gloves as a Primary Rubber Matrix: Aspect of Vulcanization Systems. Sci 2026, 8, 67. https://doi.org/10.3390/sci8030067
Masa A, Mesa N, Soontaranon S, Hayeemasae N. Feasibility of Utilizing Waste Natural Rubber Gloves as a Primary Rubber Matrix: Aspect of Vulcanization Systems. Sci. 2026; 8(3):67. https://doi.org/10.3390/sci8030067
Chicago/Turabian StyleMasa, Abdulhakim, Nurulhuda Mesa, Siriwat Soontaranon, and Nabil Hayeemasae. 2026. "Feasibility of Utilizing Waste Natural Rubber Gloves as a Primary Rubber Matrix: Aspect of Vulcanization Systems" Sci 8, no. 3: 67. https://doi.org/10.3390/sci8030067
APA StyleMasa, A., Mesa, N., Soontaranon, S., & Hayeemasae, N. (2026). Feasibility of Utilizing Waste Natural Rubber Gloves as a Primary Rubber Matrix: Aspect of Vulcanization Systems. Sci, 8(3), 67. https://doi.org/10.3390/sci8030067








