Neonatal Jaundice Detection Using a Computer Vision System
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Collection
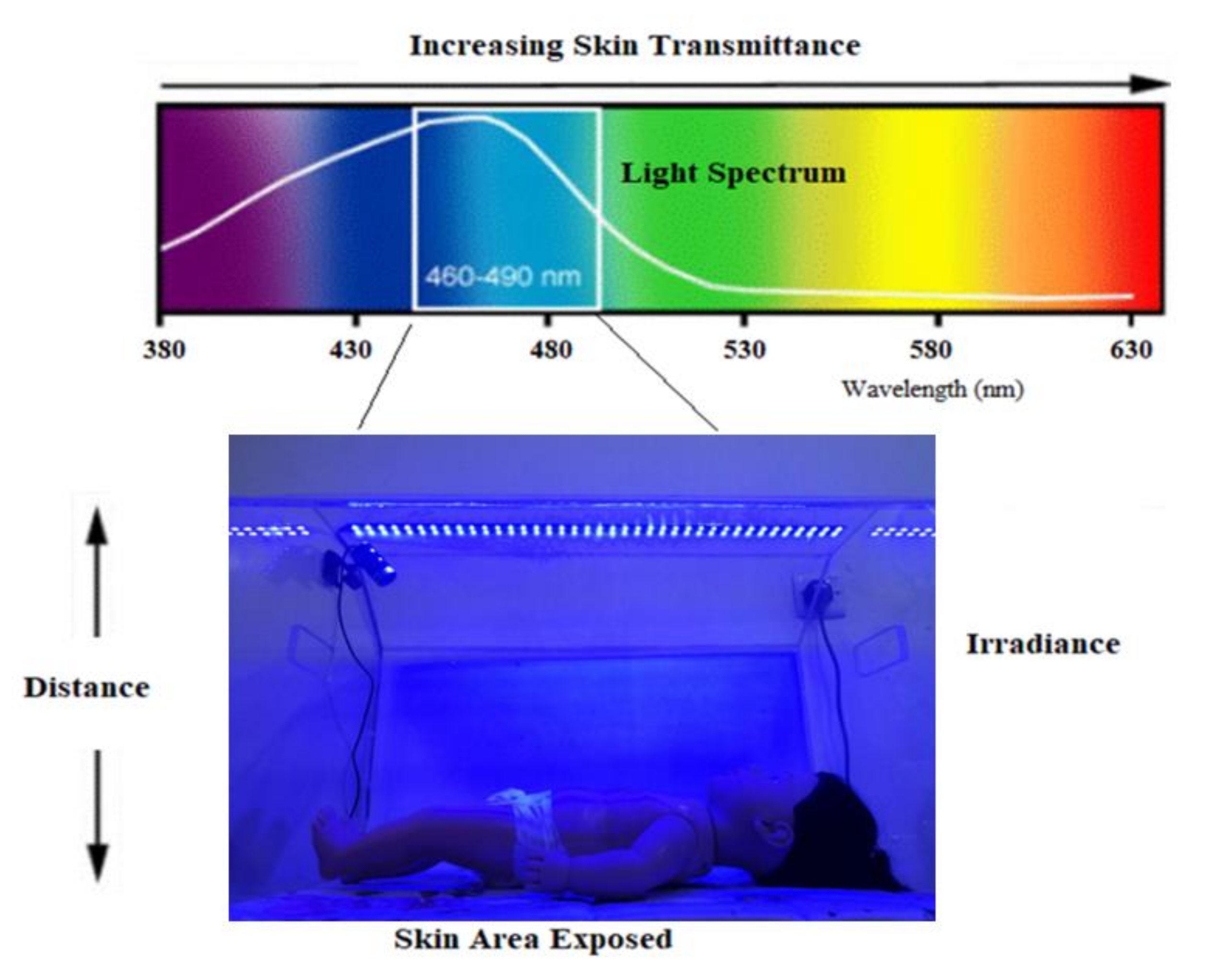
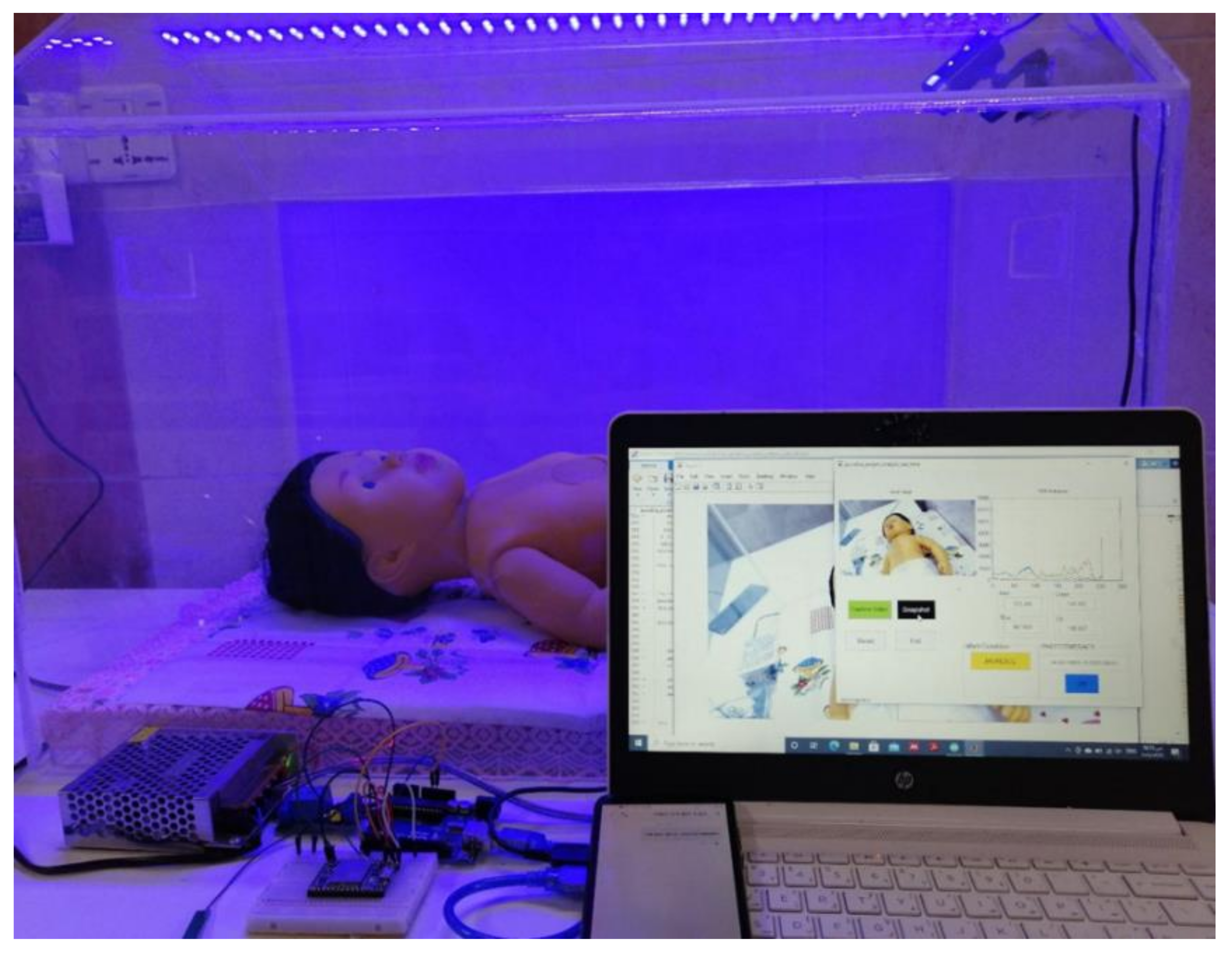
2.2. Experimental Setup
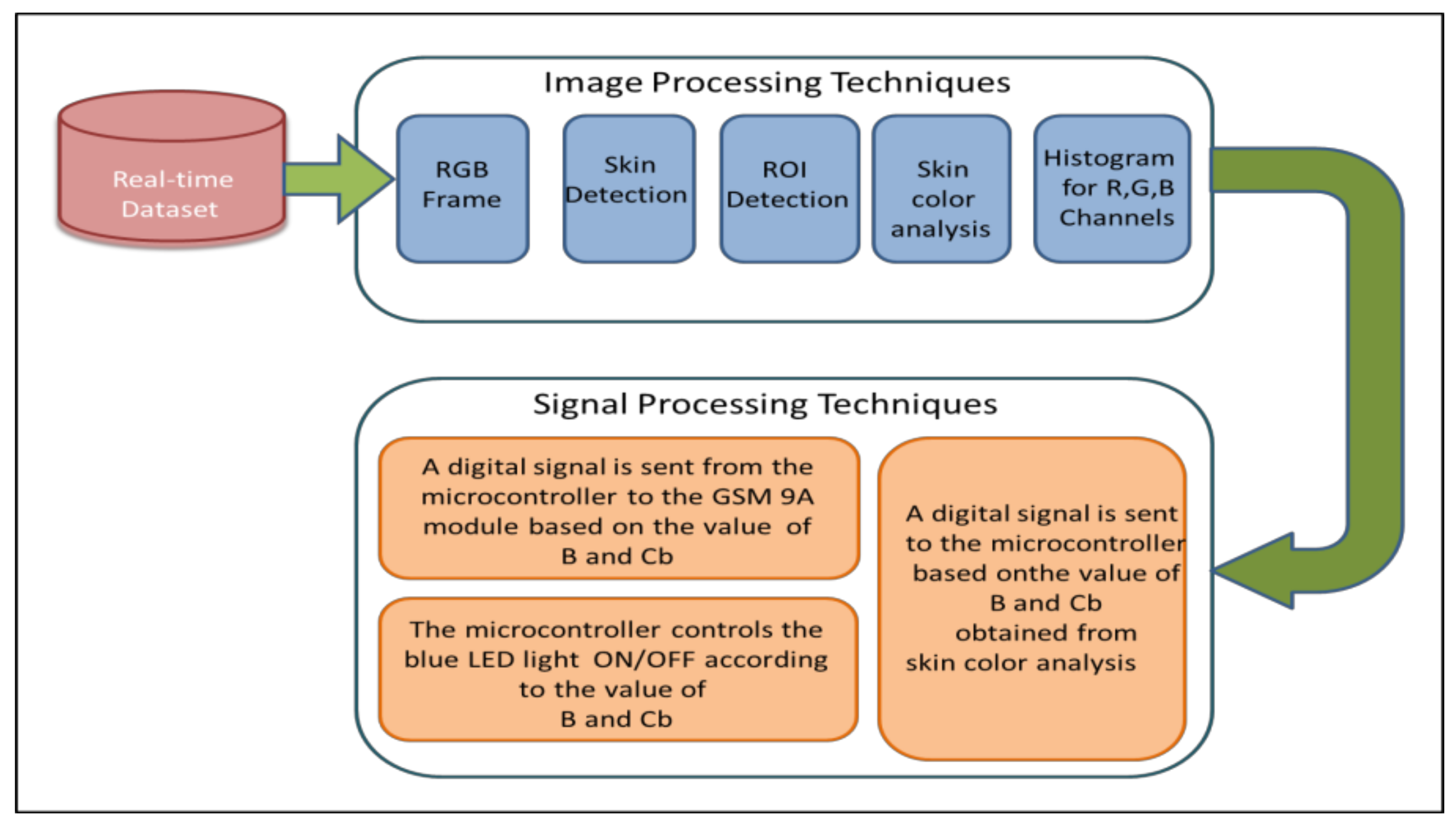
2.3. System Design
- Capturing real-time images using a full HD digital camera.
- Using color transformation and Otsu thresholding techniques to detect the skin in the captured images.
- Specify the ROI (skin) by using image processing techniques.
- Finding the values of the B channel and Cb channel for the ROI from the RGB and YCbCr color spaces, respectively.
- Comparing the values of the B and Cb channels with the predefined threshold value.
- If the value of either B or Cb channel is less than the threshold, the diagnosis result is “Jaundice” and the infant needs phototherapy. Otherwise, the diagnosis result is “Normal”.
- If phototherapy is needed, the system will switch ON the blue LED light. Otherwise, the blue LED will be switched OFF. In both cases, an SMS message is sent to the care provider informing him/her whether the infant needs phototherapy or not.
3. Results
3.1. Scenario 1
3.2. Scenario 2
3.3. Scenario 3
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dzulkifli, F.A.; Mashor, M.Y.; Khalid, K. Methods for determining bilirubin level in neonatal jaundice screening and monitoring: A literature review. J. Eng. Res. Educ. 2018, 10, 1–10. [Google Scholar]
- Brits, H.; Adendorff, J.; Huisamen, D.; Beukes, D.; Botha, K.; Herbst, H.; Joubert, G. The prevalence of neonatal jaundice and risk factors in healthy term neonates at National District Hospital in Bloemfontein. Afr. J. Prim. Health Care Fam. Med. 2018, 10, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Bhutani, V.K.; Zipursky, A.; Blencowe, H.; Khanna, R.; Sgro, M.; Ebbesen, F.; Bell, J.; Mori, R.; Slusher, T.M.; Fahmy, N. Neonatal hyperbilirubinemia and Rhesus disease of the newborn: Incidence and impairment estimates for 2010 at regional and global levels. Pediatric Res. 2013, 74, 86–100. [Google Scholar] [CrossRef] [PubMed]
- Vodret, S. Neonatal Hyperbilirubinemia: In Vivo Characterization of Mechanisms of Bilirubin Neurotoxicity and Pharmacological Treatments; International Centre for Genetic Engineering and Biotechnology: New Delhi, India, 2016. [Google Scholar]
- Mishra, S.; Agarwal, R.; Deorari, A.K.; Paul, V.K. Jaundice in the newborns. Indian J. Pediatrics 2008, 75, 157–163. [Google Scholar] [CrossRef]
- Hyperbilirubinemia, A.A.o.P.S.o. Management of hyperbilirubinemia in the newborn infant 35 or more weeks of gestation. Pediatrics 2004, 114, 297–316. [Google Scholar] [CrossRef]
- Mantagou, L.; Fouzas, S.; Skylogianni, E.; Giannakopoulos, I.; Karatza, A.; Varvarigou, A. Trends of transcutaneous bilirubin in neonates who develop significant hyperbilirubinemia. Pediatrics 2012, 130, e898–e904. [Google Scholar] [CrossRef]
- Mansouri, M.; Mahmoodnejad, A.; Taghizadeh Sarvestani, R.; Gharibi, F. A comparison between transcutaneous bilirubin (TcB) and total serum bilirubin (TSB) measurements in term neonates. Int. J. Pediatrics 2015, 3, 633–641. [Google Scholar]
- Alsaedi, S.A. Transcutaneous bilirubin measurements can be used to measure bilirubin levels during phototherapy. Int. J. Pediatrics 2018, 2018, 4856390. [Google Scholar] [CrossRef]
- Mreihil, K.; Nakstad, B.; Stensvold, H.J.; Benth, J.Š.; Hansen, T.W.R.; Group, N.N.P.S.; Network, N.N.; Scheck, O.; Nordin, S.; Prytz, A. Uniform national guidelines do not prevent wide variations in the clinical application of phototherapy for neonatal jaundice. Acta Paediatr. 2018, 107, 620–627. [Google Scholar] [CrossRef]
- Maisels, M.J.; McDonagh, A.F. Phototherapy for neonatal jaundice. N. Engl. J. Med. 2008, 358, 920–928. [Google Scholar] [CrossRef]
- Mreihil, K.; Benth, J.Š.; Stensvold, H.J.; Nakstad, B.; Hansen, T.W.R.; Group, N.N.P.S.; Network, N.N.; Scheck, O.; Nordin, S.; Prytz, A. Phototherapy is commonly used for neonatal jaundice but greater control is needed to avoid toxicity in the most vulnerable infants. Acta Paediatr. 2018, 107, 611–619. [Google Scholar] [CrossRef]
- Woodgate, P.; Jardine, L.A. Neonatal jaundice: Phototherapy. BMJ Clin. Evid. 2015, 2015. [Google Scholar]
- Donel, J. Bili blanket phototherapy. Int. J. Contemp. Pediatr. 2019, 6, 2231–2234. [Google Scholar] [CrossRef]
- Maisels, M.; Watchko, J.; Bhutani, V.; Stevenson, D. An approach to the management of hyperbilirubinemia in the preterm infant less than 35 weeks of gestation. J. Perinatol. 2012, 32, 660–664. [Google Scholar] [CrossRef]
- Marco, L.; Giovanni, M.F. Computer Vision For Assistive Healthcare; Academic Press: Cambridge, MA, USA, 2018. [Google Scholar]
- Hashim, W.; Al-Naji, A.; Al-Rayahi, I.A.; Oudah, M. Computer Vision for Jaundice Detection in Neonates Using Graphic User Interface. IOP Conf. Ser. Mater. Sci. Eng. 2021, 1105, 012076. [Google Scholar] [CrossRef]
- Leartveravat, S. Transcutaneous bilirubin measurement in full term neonate by digital camera. Med J. Srisaket Surin Buriram Hosp. 2009, 24, 105–118. [Google Scholar]
- Mansor, M.; Yaacob, S.; Hariharan, M.; Basah, S.; Jamil, S.A.; Khidir, M.M.; Rejab, M.; Ibrahim, K.K.; Jamil, A.A.; Junoh, A. Jaundice in newborn monitoring using color detection method. Procedia Eng. 2012, 29, 1631–1635. [Google Scholar] [CrossRef][Green Version]
- Mansor, M.; Hariharan, M.; Basah, S.; Yaacob, S. New newborn jaundice monitoring scheme based on combination of pre-processing and color detection method. Neurocomputing 2013, 120, 258–261. [Google Scholar] [CrossRef]
- Munkholm, S.B.; Krøgholt, T.; Ebbesen, F.; Szecsi, P.B.; Kristensen, S.R. The smartphone camera as a potential method for transcutaneous bilirubin measurement. PLoS ONE 2018, 13, e0197938. [Google Scholar]
- Kawano, S.; Zin, T.T.; Kodama, Y. A Study on Non-contact and Non-invasive Neonatal Jaundice Detection and Bilirubin Value Prediction. In Proceedings of the 2018 IEEE 7th Global Conference on Consumer Electronics (GCCE), Nara, Japan, 9–12 October 2018; pp. 401–402. [Google Scholar]
- Juliastuti, E.; Nadhira, V.; Satwika, Y.W.; Aziz, N.A.; Zahra, N. Risk Zone Estimation of Newborn Jaundice Based on Skin Color Image Analysis. In Proceedings of the 2019 6th International Conference on Instrumentation, Control and Automation (ICA), Bandung, Indonesia, 31 July–2 August 2019; pp. 176–181. [Google Scholar]
- Aune, A.; Vartdal, G.; Bergseng, H.; Randeberg, L.L.; Darj, E. Bilirubin estimates from smartphone images of newborn infants’ skin correlated highly to serum bilirubin levels. Acta Paediatr. 2020, 109, 2532–2538. [Google Scholar] [CrossRef]
- Rong, Z.; Luo, F.; Ma, L.; Chen, L.; Wu, L.; Liu, W.; Du, L.; Luo, X. Evaluation of an automatic image-based screening technique for neonatal hyperbilirubinemia. Zhonghua Er Ke Za Zhi / Chin. J. Pediatrics 2016, 54, 597–600. [Google Scholar]
- Taylor, J.A.; Stout, J.W.; de Greef, L.; Goel, M.; Patel, S.; Chung, E.K.; Koduri, A.; McMahon, S.; Dickerson, J.; Simpson, E.A. Use of a smartphone app to assess neonatal jaundice. Pediatrics 2017, 140, e20170312. [Google Scholar] [CrossRef]
- Aydın, M.; Hardalaç, F.; Ural, B.; Karap, S. Neonatal jaundice detection system. J. Med Syst. 2016, 40, 166. [Google Scholar] [CrossRef]
- Padidar, P.; Shaker, M.; Amoozgar, H.; Khorraminejad-Shirazi, M.; Hemmati, F.; Najib, K.S.; Pourarian, S. Detection of neonatal jaundice by using an android OS-based smartphone application. Iran. J. Pediatrics 2019, 29, e84397. [Google Scholar] [CrossRef]
- Angelico, R.; Liccardo, D.; Paoletti, M.; Pietrobattista, A.; Basso, M.S.; Mosca, A.; Safarikia, S. A Novel Mobile Phone Application for Infant Stool Color Recognition: An Easy and Effective Tool to Identify Acholic Stools in Newborns. J. Med Screen. 2020, 26, 230–237. [Google Scholar]
- Miah, M.M. Non-Invasive Bilirubin Level Quantification and Jaundice Detection by Sclera Image Processing. In Proceedings of the 2019 IEEE Global Humanitarian Technology Conference (GHTC), Seattle, WA, USA, 17–20 October 2019; pp. 1–7. [Google Scholar]
- Outlaw, I.d.; Nixon, M.; Odeyemi, O.; Macdonald, L.W.; Meek, J.; Leung, T.S. Smartphone Screening for Neonatal Jaundice via Ambient-Subtracted Sclera Chromaticity. PLoS ONE 2020, 15, e0216970. [Google Scholar] [CrossRef]
- Viau Colindres, J.; Rountree, C.; Destarac, M.A.; Cui, Y.; Pérez Valdez, M.; Herrera Castellanos, M.; Mirabal, Y.; Spiegel, G.; Richards-Kortum, R.; Oden, M. Prospective randomized controlled study comparing low-cost LED and conventional phototherapy for treatment of neonatal hyperbilirubinemia. J. Trop. Pediatrics 2012, 58, 178–183. [Google Scholar] [CrossRef][Green Version]
- Kolkur, S.; Kalbande, D.; Shimpi, P.; Bapat, C.; Jatakia, J. Human skin detection using RGB, HSV and YCbCr color models. arXiv 2017, arXiv:1708.02694. [Google Scholar]
- Bangare, S.L.; Dubal, A.; Bangare, P.S.; Patil, S. Reviewing otsu’s method for image thresholding. Int. J. Appl. Eng. Res. 2015, 10, 21777–21783. [Google Scholar] [CrossRef]
- Chowdhary, A.; Dutta, S.; Ghosh, R. Neonatal Jaundice Detection using Colour Detection Method. Int. Adv. Res. J. Sci. Eng. Technol. 2017, 4, 197–203. [Google Scholar]

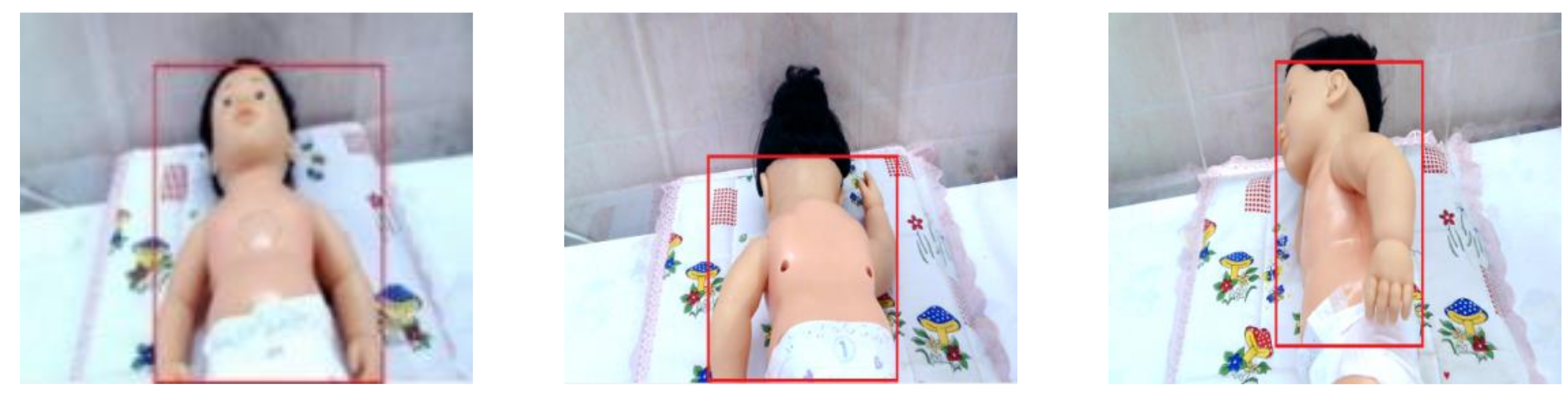
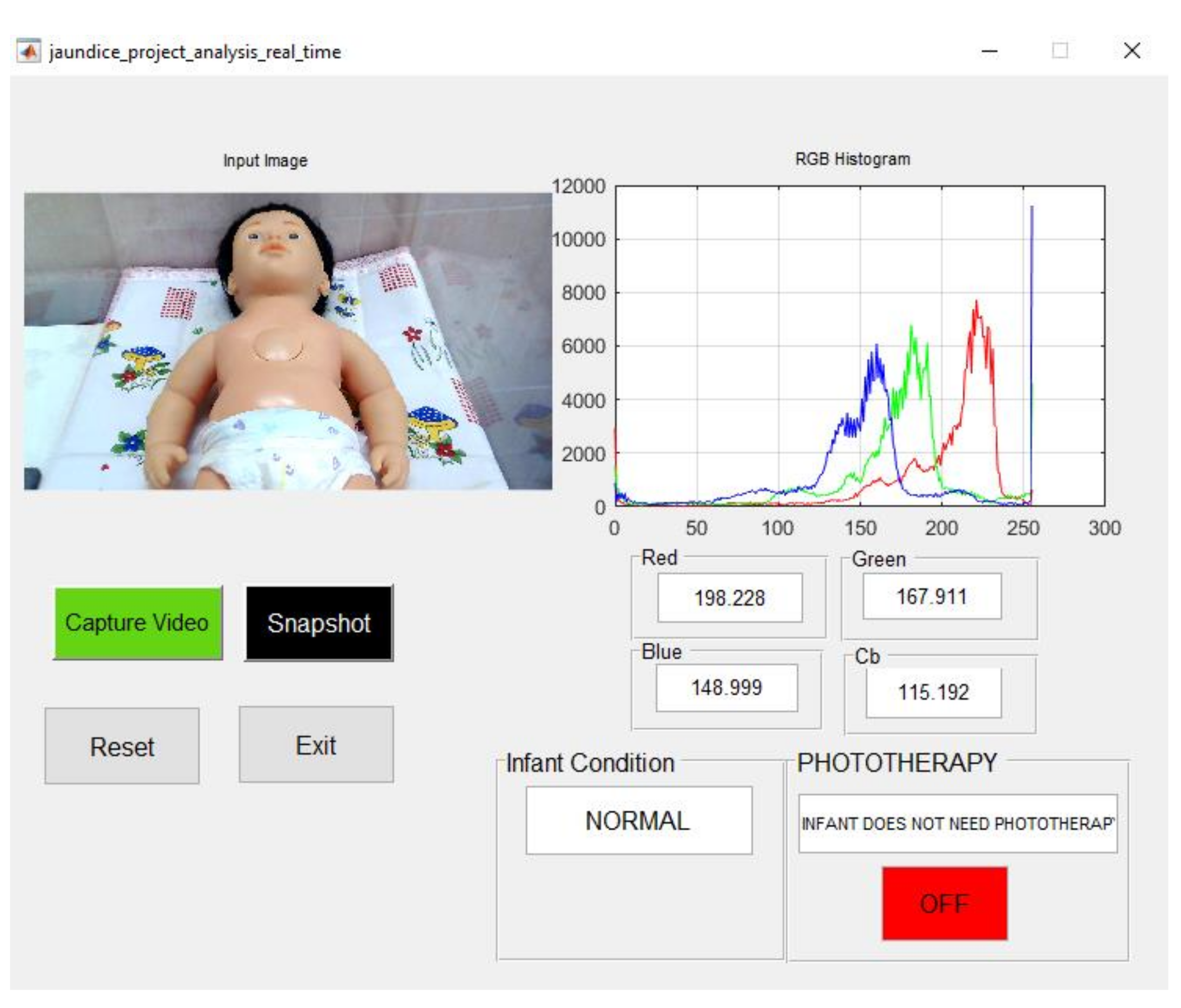
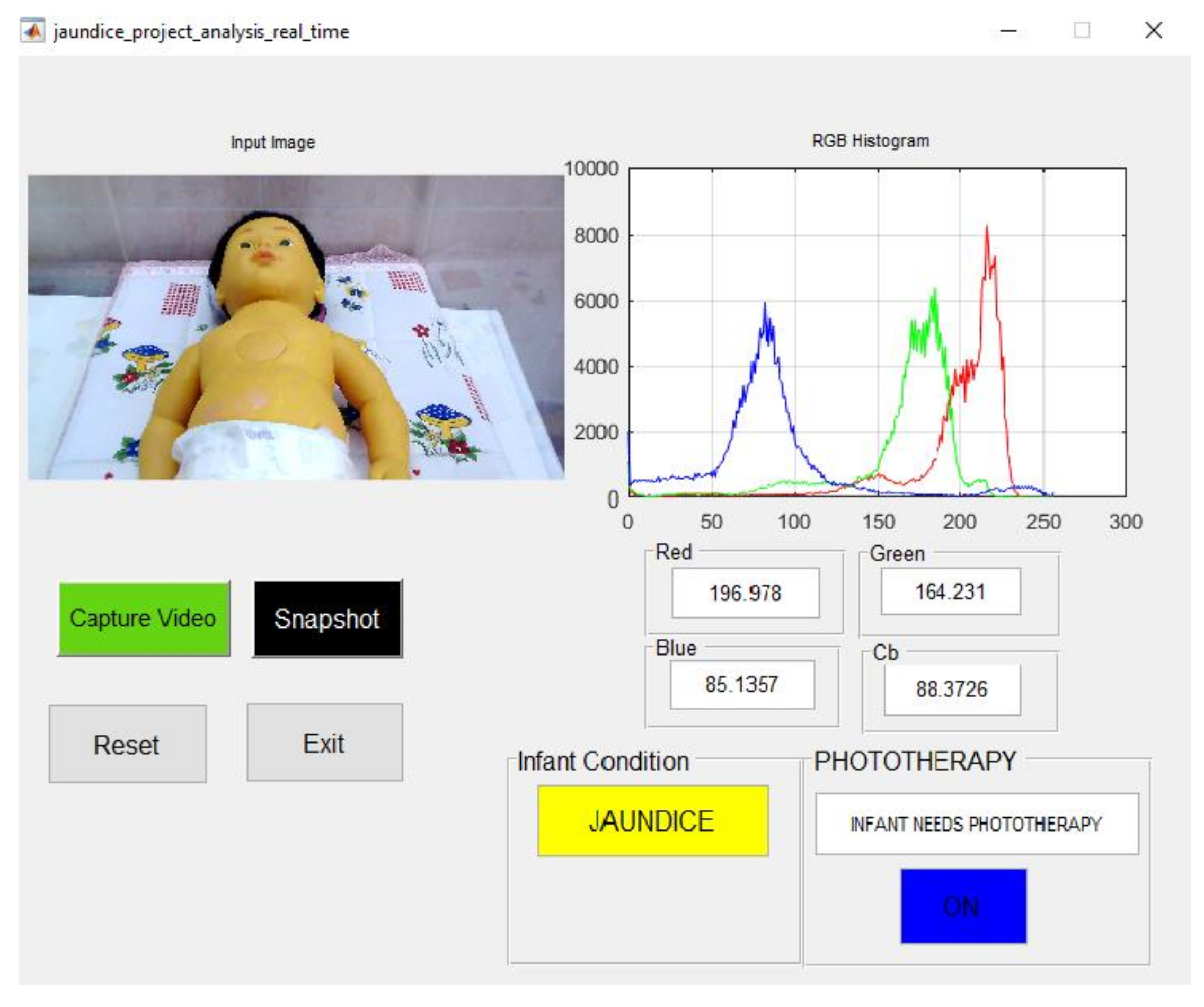

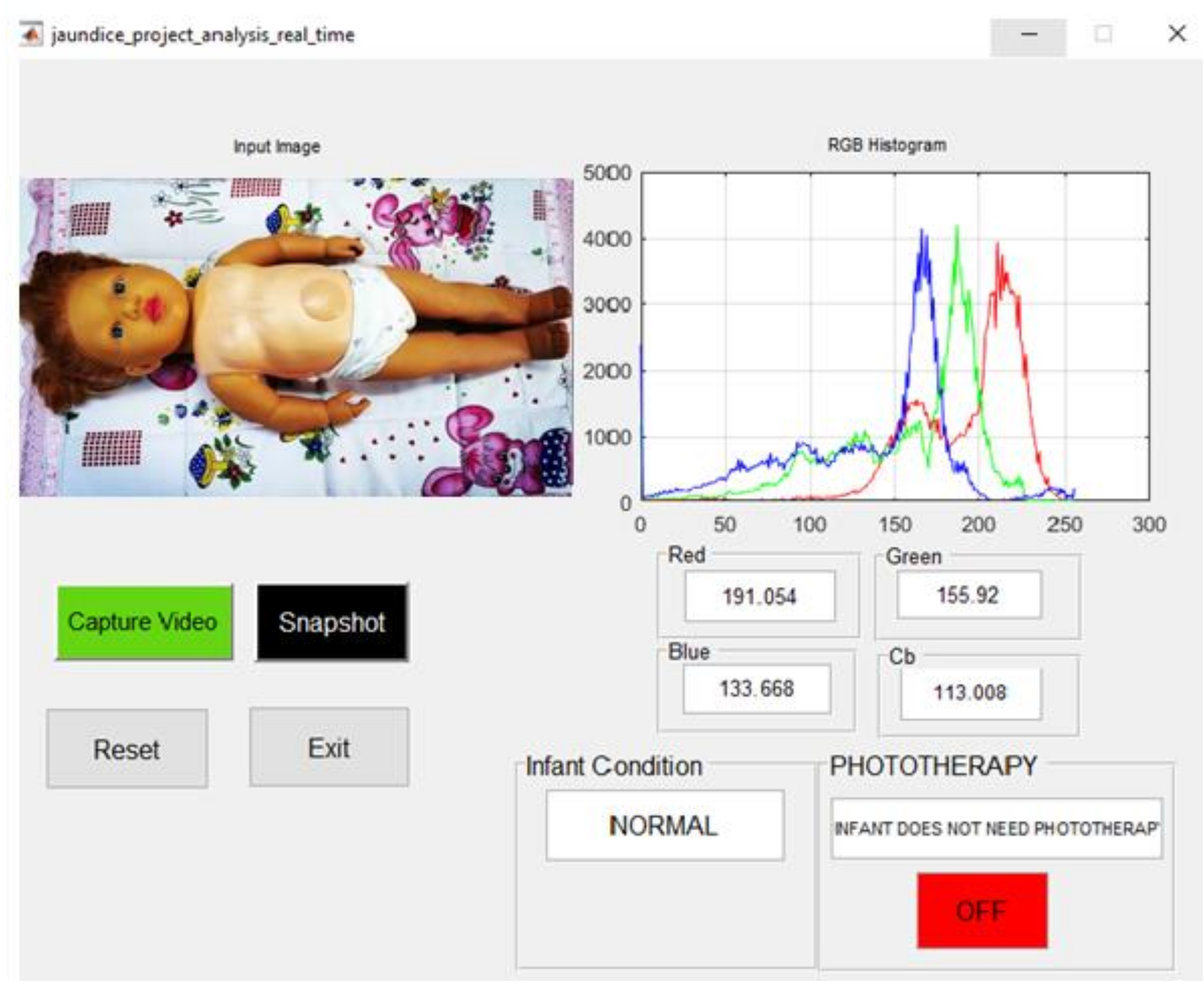
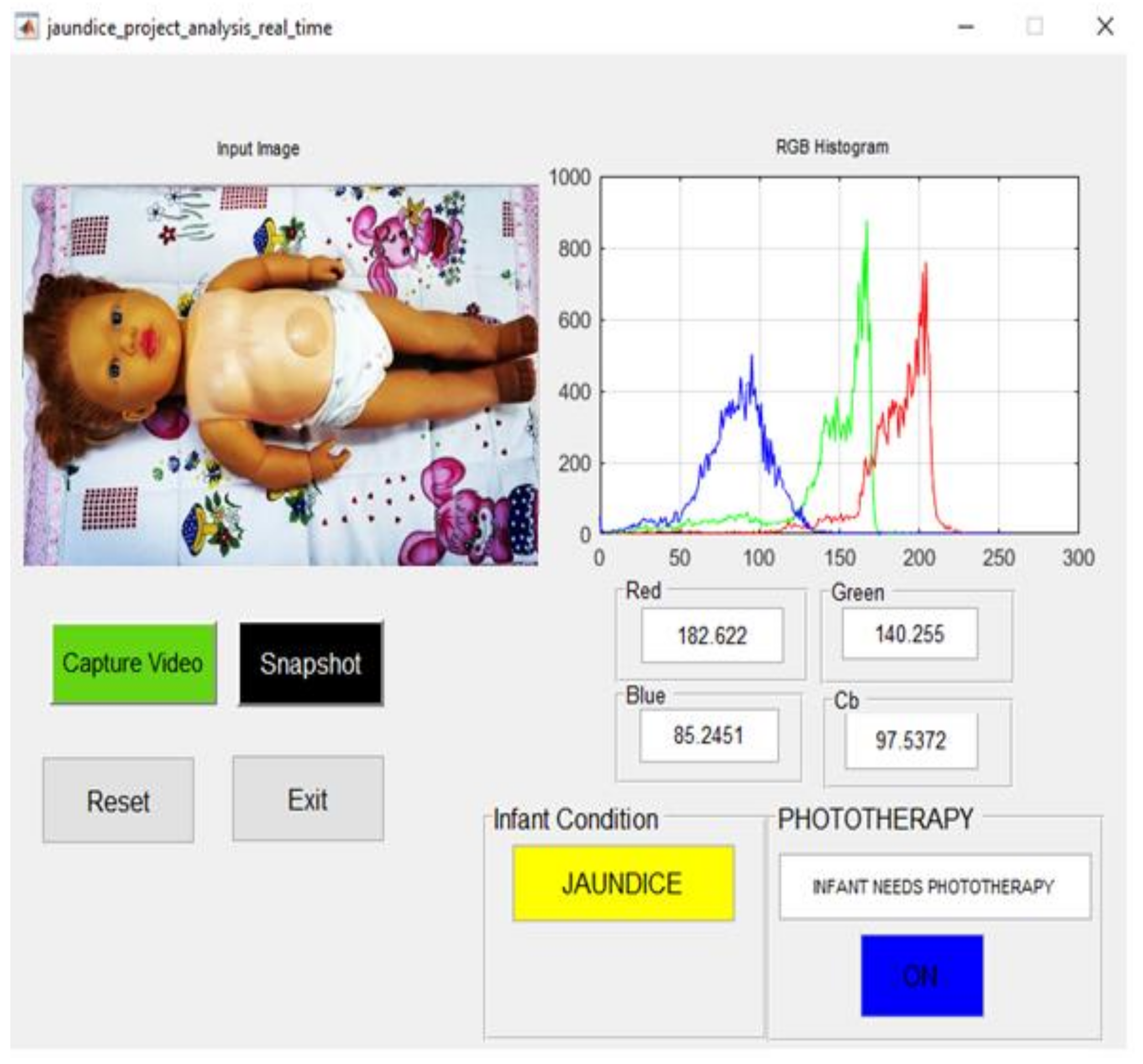
| Manikin Skin Color | B Channel | Cb Channel | Infant Condition | Blue LED Status |
|---|---|---|---|---|
| light complexion | 150.272 | 116.807 | Normal | OFF |
| light and yellow complexion | 46.666 | 99.427 | Jaundice | ON |
| brown complexion | 127.764 | 113.391 | Normal | OFF |
| brown and yellow complexion | 85.245 | 97.537 | Jaundice | ON |
| B Channel | Cb Channel | System Diagnosis | BLUE LED State | Detection Rate | |
|---|---|---|---|---|---|
| Image 1 | 153.689 | 107.847 | Normal | OFF | 100% |
| Image 2 | 82.243 | 71.472 | Jaundice | ON | 100% |
| Image 3 | 78.390 | 93.892 | Jaundice | ON | 100% |
| Image 4 | 96.535 | 94.734 | Jaundice | ON | 100% |
| Image 5 | 139.922 | 105.819 | Normal | OFF | 100% |
| Image 6 | 72.2273 | 87.396 | Jaundice | ON | 100% |
| Image 7 | 74.297 | 86.833 | Jaundice | ON | 100% |
| Image 8 | 174.604 | 122.421 | Normal | OFF | 100% |
| Image 9 | 149.787 | 106.868 | Normal | OFF | 100% |
| Image 10 | 71.236 | 89.514 | Jaundice | ON | 100% |
| Image 11 | 91.926 | 90.523 | Jaundice | ON | 100% |
| Image 12 | 81.257 | 70.999 | Jaundice | ON | 100% |
| Image 13 | 73.113 | 91.935 | Jaundice | ON | 100% |
| Image 14 | 71.236 | 89.514 | Jaundice | ON | 100% |
| Image 15 | 83.224 | 85.680 | Jaundice | ON | 100% |
| Image 16 | 113.996 | 109.049 | Normal | OFF | 100% |
| TSB mg/dl | B Channel | Cb Channel | Diagnosis | Blue LED Status | |
|---|---|---|---|---|---|
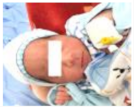 | 7 | 172.586 | 122.225 | Normal | OFF |
 | 8.6 | 165.272 | 112.581 | Normal | OFF |
 | 9.5 | 158.541 | 119.198 | Normal | OFF |
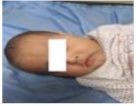 | 9.7 | 159.115 | 113.239 | Normal | OFF |
 | 11 | 142.738 | 120.761 | Normal | OFF |
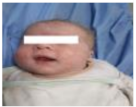 | 12 | 132.405 | 108.44 | Normal | OFF |
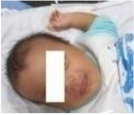 | 12.3 | 128.241 | 101.419 | Normal | OFF |
 | 12.5 | 123.8 | 124.007 | Normal | OFF |
 | 13.3 | 111.418 | 120.743 | Normal | OFF |
 | 13.8 | 108.689 | 107.645 | Normal | OFF |
 | 14.2 | 99.702 | 119.679 | Jaundice | ON |
 | 14.8 | 93.178 | 118.437 | Jaundice | ON |
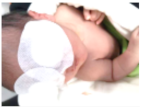 | 15.1 | 91.688 | 107.441 | Jaundice | ON |
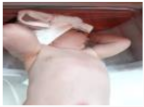 | 16.5 | 86.435 | 94.539 | Jaundice | ON |
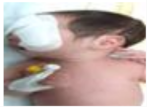 | 16.7 | 83.36 | 93.043 | Jaundice | ON |
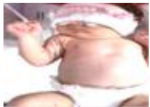 | 17 | 80.576 | 92.693 | Jaundice | ON |
 | 18 | 73.741 | 88.060 | Jaundice | ON |
 | 18.2 | 70.382 | 89.123 | Jaundice | ON |
 | 20.1 | 65.555 | 70.467 | Jaundice | ON |
 | 26 | 48.240 | 67.465 | Jaundice | ON |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hashim, W.; Al-Naji, A.; Al-Rayahi, I.A.; Alkhaled, M.; Chahl, J. Neonatal Jaundice Detection Using a Computer Vision System. Designs 2021, 5, 63. https://doi.org/10.3390/designs5040063
Hashim W, Al-Naji A, Al-Rayahi IA, Alkhaled M, Chahl J. Neonatal Jaundice Detection Using a Computer Vision System. Designs. 2021; 5(4):63. https://doi.org/10.3390/designs5040063
Chicago/Turabian StyleHashim, Warqaa, Ali Al-Naji, Izzat A. Al-Rayahi, Makram Alkhaled, and Javaan Chahl. 2021. "Neonatal Jaundice Detection Using a Computer Vision System" Designs 5, no. 4: 63. https://doi.org/10.3390/designs5040063
APA StyleHashim, W., Al-Naji, A., Al-Rayahi, I. A., Alkhaled, M., & Chahl, J. (2021). Neonatal Jaundice Detection Using a Computer Vision System. Designs, 5(4), 63. https://doi.org/10.3390/designs5040063








