The Role of Exercise in Pediatric and Adolescent Cancers: A Review of Assessments and Suggestions for Clinical Implementation
Abstract
1. Introduction
2. Types of Childhood Cancer
3. Treatments for Childhood Cancer
4. Late Adverse Events (LAE) in Childhood Cancer Patients
5. Complications after Childhood/Adolescent Cancer Treatment
6. Guidelines for Assessment
7. Echocardiographic Assessment
7.1. Cardiopulmonary Exercise Testing (CPET)
7.2. Power Work Capacity (PWC150)
7.3. Pulmonary Function Testing (PFT)
7.4. Motor and Skill Assessment
7.5. Quality of Life (QoL)
7.6. Nutrition
8. Guidelines for Exercise Prescription in Pediatric Cancer Patients
9. Recommendations
10. Conclusions
Conflicts of Interest
References
- Steliarova-Foucher, E.; Colombet, M.; Ries, L.A.G.; Moreno, F.; Dolya, A.; Bray, F.; Hesseling, P.; Shin, H.Y.; Stiller, C.A. International incidence of childhood cancer, 2001–10: A population-based registry study. Lancet Oncol. 2017, 18, 719–731. [Google Scholar] [CrossRef]
- American Cancer Society. Cancer Facts & Figures 2016; American Cancer Society: Atlanta, GA, USA, 2016. [Google Scholar]
- Gatta, G.; Botta, L.; Rossi, S.; Aareleid, T.; Bielska-Lasota, M.; Clavel, J.; Dimitrov, N.; Jakab, Z.; Kaatsch, P.; Lacour, B.; et al. The EUROCARE Working Group Childhood cancer survival in Europe 1999–2007: Results of EUROCARE-5—A population based study. Lancet Oncol. 2014, 15, 35–47. [Google Scholar] [CrossRef]
- Howlader, N.; Noone, A.M.; Krapcho, M.; Miller, D.; Bishop, K.; Kosary, C.L.; Yu, M.; Ruhl, J.; Tatalovich, Z.; Mariotto, A.; et al. (Eds.) SEER Cancer Statistics Review, 1975–2014; National Cancer Institute: Bethesda, MD, USA. Available online: https://seer.cancer.gov/csr/1975_2014/ (accessed on 4 April 2017).
- Mosso, M.L.; Colombo, R.; Giordano, L.; Pastore, G.; Terracini, B.; Magnani, C. Childhood cancer registry of the province of Torino, Italy: Survival, incidence and mortality over 20 years. Cancer 1992, 69, 1300–1306. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, G.T.; Chow, E.J.; Sklar, C.A. Alterations in pubertal timing following therapy for childhood malignancies. In Endocrinopathy after Childhood Cancer Treatment; Wallace, W.H.B., Kelnar, C.J.H., Eds.; Karger Publishers: Basel, Switzerland, 2009; Volume 15, pp. 25–39. [Google Scholar]
- Armstrong, G.T.; Stovall, M.; Robison, L.L. Long-term effects of radiation exposure among adult survivors of childhood cancer: Results from the Childhood Cancer Survivor Study. Radiat. Res. 2010, 174, 840–850. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, S.; Davies, S.M.; Baker, S.K.; Pulsipher, M.A.; Hansen, J.A. NCI, NHLBI first international consensus conference on late effects after pediatric hematopoietic cell transplantation: Etiology and pathogenesis of late effects after HCT performed in childhood—Methodologic challenges. Biol. Blood Marrow Transplant. 2011, 17, 1428–1435. [Google Scholar] [CrossRef] [PubMed]
- Clanton, N.R.; Klosky, J.L.; Li, C.; Jain, N.; Srivastava, D.K.; Mulrooney, D.; Zeltzer, L.; Stovall, M.; Robison, L.; Krull, K. Fatigue, vitality, sleep, and neurocognitive functioning in adult survivors of childhood cancer: A report from the childhood cancer survivor study. Cancer 2011, 117, 2559–2568. [Google Scholar] [CrossRef] [PubMed]
- Diller, L.; Chow, E.J.; Gurney, J.G.; Hudson, M.M.; Kadin-Lottick, N.S.; Kawashima, T.I.; Leisenring, W.M.; Meacham, L.R.; Mertens, A.C.; Mulrooney, D.A.; et al. Chronic disease in the childhood cancer survivor study cohort: A review of published findings. J. Clin. Oncol. 2009, 27, 2339–2355. [Google Scholar] [CrossRef] [PubMed]
- Feijen, E.A.M.; Font-Gonzalez, A.; van Dalen, E.C.; van der Pal, H.J.H.; Reulen, R.C.; Winter, D.L.; Kuehni, C.E.; Haupt, R.; Alessi, D.; Byrne, J.; et al. Late cardiac events after childhood cancer: Methodological aspects of the Pan-European study PanCareSurFup. PLoS ONE 2016, 11, e0162778. [Google Scholar] [CrossRef] [PubMed]
- Friedman, D.L.; Whitton, J.; Leisenring, W.; Mertens, A.C.; Hammond, S.; Stoval, M.; Donaldson, S.S.; Meadows, A.T.; Robison, L.L.; Neglia, J.P. Subsequent neoplasms in 5-year survivors of childhood cancer: The childhood cancer survivor study. J. Natl. Cancer Inst. 2010, 102, 1083–1095. [Google Scholar] [CrossRef] [PubMed]
- Gibson, T.M.; Robison, L.L. Impact of cancer therapy-related exposures on late mortality in childhood cancer survivors. Chem. Res. Toxicol. 2015, 28, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Haupt, R.; Jankovic, M.; Hjorth, L.; Skinner, R. Late effects in childhood cancer survivors and survivorship issues. Epidemiol. Prev. 2013, 37 (Suppl. 1), 1–296. [Google Scholar]
- Lipshultz, E.R.; Holt, G.E.; Ramasamy, R.; Yechieli, R.; Lipshultz, S.E. Fertility, cardiac, and orthopedic challenges in survivors of adult and childhood sarcoma. Am. Soc. Clin. Oncol. Educ. Book 2017, 37, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Mulrooney, D.A.; Yeazel, M.W.; Kawashima, T.; Mertens, A.C.; Mitby, P.; Stovall, M.; Donaldson, S.S.; Green, D.M.; Sklar, C.A.; Robison, L.L.; et al. Cardiac outcomes in a cohort of adult survivors of childhood and adolescent cancer: Retrospective analysis of the Childhood Cancer Survivor Study cohort. BMJ 2009, 339, b4606. [Google Scholar] [CrossRef] [PubMed]
- Oeffinger, K.C.; Mertens, A.C.; Sklar, C.A.; Kawashima, T.; Hudson, M.M.; Meadows, A.T.; Friedman, D.L.; Marina, N.; Hobbie, W.; Kadan-Lottick, N.S.; et al. Childhood Cancer Survivor Study. Chronic health conditions in adult survivors of childhood cancer. N. Engl. J. Med. 2006, 355, 1572–1582. [Google Scholar] [CrossRef] [PubMed]
- Bhakta, N.; Liu, Q.; Yeo, F.; Baassiri, M.; Ehrhardt, M.J.; Srivastava, D.K.; Metzger, M.L.; Krasin, M.J.; Ness, K.K.; Hudson, M.M.; et al. Cumulative burden of cardiovascular morbidity in paediatric, adolescent, and young adult survivors of Hodgkin’s lymphoma: An analysis from the St Jude Lifetime Cohort Study. Lancet Oncol. 2016, 17, 1325–1334. [Google Scholar] [CrossRef]
- Carneiro Teixeira, J.F.; Maia-Lemos, P.D.S.; Cypriano, M.D.S.; Pellegrini, P.L. Obesity in survivors of childhood cancer: A review. Pediatr. Endocrinol. Rev. 2017, 15, 33–39. [Google Scholar] [PubMed]
- Ligibel, J.A.; Alfano, C.M.; Courneya, K.S.; Demark-Wahnefried, W.; Burger, R.A.; Chlebowski, R.T.; Fabian, C.J.; Gucalp, A.; Hershman, D.L.; Hudson, M.M.; et al. American Society of Clinical Oncology position statement on obesity and cancer. J. Clin. Oncol. 2014, 32, 3568–3574. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Pineda, I.; Hudson, M.M.; Pappo, A.S.; Bishop, M.W.; Klosky, J.L.; Brinkman, T.M.; Srivastava, D.K.; Neel, M.D.; Rao, B.N.; Davidoff, A.M.; et al. Long-term functional outcomes and quality of life in adult survivors of childhood extremity sarcomas: A report from the St. Jude lifetime cohort study. J. Cancer Surv. 2017, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Gerber, L.H.; Hoffman, K.; Chaudhry, U.; Augustine, E.; Parks, R.; Bernad, M.; Mackall, C.; Steinberg, S.; Mansky, P. Functional outcomes and life satisfaction in long-term survivors of pediatric sarcomas. Arch. Phys. Med. Rehabil. 2006, 87, 1611–1617. [Google Scholar] [CrossRef] [PubMed]
- Ridola, V.; Fawaz, O.; Aubier, F.; Bergeron, C.; de Vathaire, F.; Orbach, D.; Gentet, J.C.; Schmitt, C.; Dufour, C.; Oberlin, O. Testicular function of survivors of childhood cancer: A comparative study between ifosfamide and cyclophosphamide-based regimens. Eur. J. Cancer 2009, 45, 814–818. [Google Scholar]
- Rose, S.R.; Danish, R.K.; Kearney, N.S.; Schreiber, R.E.; Lustig, R.H.; Burghen, G.A.; Hudson, M.M. ACTH deficiency in childhood cancer survivors. Pediatr. Blood Cancer 2005, 45, 808–813. [Google Scholar] [CrossRef] [PubMed]
- Siviero-Miachon, A.A.; Spinola-Castro, A.M.; Guerra-Junior, G. Detection of metabolic syndrome features among childhood cancer survivors: A target to prevent disease. Vasc. Health Risk Manag. 2008, 4, 825–836. [Google Scholar] [PubMed]
- Skinner, R.; Mulder, R.L.; Kremer, L.C.; Hudson, M.M.; Constine, L.S.; Bardi, E.; Boekhout, A.; Borgmann-Staudt, A.; Brown, M.C.; Cohn, R.; et al. Recommendations for gonadotoxicity surveillance in male childhood, adolescent, and young adult cancer survivors: A report from the International Late Effects of Childhood Cancer Guideline Harmonization Group in collaboration with the PanCareSurFup Consortium. Lancet Oncol. 2017, 18, e75–e90. [Google Scholar]
- Massimino, M.; Gandola, L.; Mattavelli, F.; Pizzi, N.; Seregni, E.; Pallotti, F.; Spreafico, F.; Marchianò, A.; Terenziani, M.; Cefalo, G.; et al. Radiation-induced thyroid changes: A retrospective and a prospective view. Eur. J. Cancer 2009, 45, 2546–2551. [Google Scholar] [CrossRef] [PubMed]
- Hartman, A.; te Winkel, M.L.; van Beek, R.D.; de Muinck Keizer-Schrama, S.M.P.F.; Kemper, H.C.G.; Hop, W.C.J.; van den Heuvel-Eibrink, M.M.; Pieters, R. A randomized trial investigating an exercise program to prevent reduction of bone mineral density and impairment of motor performance during treatment for childhood acute lymphoblastic leukemia. Pediatr. Blood Cancer 2009, 53, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Hudson, M.M.; Mertens, A.C.; Yasui, Y.; Hobbie, W.; Chen, H.; Gurney, J.G.; Yeazel, M.; Recklitis, C.J.; Marina, N.; Robison, L.R.; et al. Health status of adult long-term survivors of childhood cancer: A report from the childhood cancer survivor study. JAMA 2003, 290, 1583–1592. [Google Scholar] [CrossRef] [PubMed]
- Hudson, M.M.; Ness, K.K.; Gurney, J.G.; Mulrooney, D.A.; Chemaitilly, W.; Krull, K.R.; Green, D.M.; Armstrong, G.T.; Nottage, K.A.; Jones, K.E.; et al. Clinical ascertainment of health outcomes among adults treated for childhood cancer. JAMA 2013, 309, 2371–2381. [Google Scholar] [CrossRef] [PubMed]
- Skinner, R. Nephrotoxicity—What do we know and what don’t we know? J. Pediatr. Hematol. Oncol. 2011, 33, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Sklar, C.A.; Mertens, A.C.; Mitby, P.; Whitton, J.; Stovall, M.; Kasper, C.; Mulder, J.; Green, D.; Nicholson, H.S.; Yasui, Y.; et al. Premature menopause in survivors of childhood cancer: A report from the childhood cancer survivor study. J. Natl. Cancer Inst. 2006, 98, 890–896. [Google Scholar] [CrossRef] [PubMed]
- Interiano, R.B.; Kaste, S.C.; Li, C.; Srivastava, D.K.; Rao, B.N.; Warner, W.C.Jr.; Green, D.M.; Krasin, M.J.; Robison, L.L.; Davidoff, A.M.; Hudson, M.M.; et al. Associations between treatment, scoliosis, pulmonary function, and physical performance in long-term survivors of sarcoma. J. Cancer Surv. 2017, 11. [Google Scholar] [CrossRef] [PubMed]
- Ness, K.K.; Armstrong, G.T.; Kundu, M.; Wilson, C.L.; Tchkonia, T.; Kirkland, J.L. Frailty in childhood cancer survivors. Cancer 2015, 121, 1540–1547. [Google Scholar] [CrossRef] [PubMed]
- Ness, K.K.; Krull, K.R.; Jones, K.E.; Mulrooney, D.A.; Armstrong, G.T.; Green, D.M.; Chemaitilly, W.; Smith, W.A.; Wilson, C.L.; Sklar, C.A.; et al. Physiologic frailty as a sign of accelerated aging among adult survivors of childhood cancer: A report from the St Jude Lifetime Cohort Study. J. Clin. Oncol. 2013, 31, 4496–4503. [Google Scholar] [CrossRef] [PubMed]
- Winther, J.F.; Kenborg, L.; Byrne, J.; Hjorth, L.; Kaatsch, P.; Kremer, L.C.; Kuehni, C.E.; Auquier, P.; Michel, G.; de Vathaire, F.; et al. Childhood cancer survivor cohorts in Europe. Acta Oncol. 2015, 54, 655–668. [Google Scholar] [CrossRef] [PubMed]
- Wilson, C.L.; Chemaitilly, W.; Jones, K.E.; Kaste, S.C.; Srivastava, D.K.; Ojha, R.P.; Yasui, Y.; Pui, C.-H.; Robison, L.L.; Hudson, M.M.; et al. Modifiable factors associated with aging phenotypes among adult survivors of childhood acute lymphoblastic leukemia. J. Clin. Oncol. 2016, 34, 2509–2515. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, K.H.; Courneya, K.S.; Matthews, C.; Demark-Wahnefried, W.; Galvão, D.A.; Pinto, B.M.; Irwin, M.L.; Wolin, K.Y.; Segal, R.J.; Lucia, A.; et al. American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Med. Sci. Sports Exerc. 2010, 42, 1409–1426. [Google Scholar] [CrossRef] [PubMed]
- Braam, K.I.; van der Torre, P.; Takken, T.; Veening, M.A.; van Dulmen-den Broeder, E.; Kaspers, G.J.L. Physical exercise training interventions for children and young adults during and after treatment for childhood cancer. Cochrane Database Syst. Rev. 2013, 4. [Google Scholar] [CrossRef]
- Huang, T.-T.; Ness, K.K. Exercise interventions in children with cancer: A review. Int. J. Pediatr. 2011, 1–11. [Google Scholar] [CrossRef]
- Deisenroth, A.; Söntgerath, R.; Schuster, A.J.; von Busch, C.; Huber, G.; Eckert, K.; Kulozik, A.E.; Wiskemann, J. Muscle strength and quality of life in patients with childhood cancer at early phase of primary treatment. Pediatr. Hematol. Oncol. 2016, 33, 393–407. [Google Scholar] [CrossRef] [PubMed]
- Esbenshade, A.J.; Friedman, D.L.; Smith, W.A.; Jeha, S.; Pui, C.H.; Robison, L.L.; Ness, K.K. Feasibility and initial effectiveness of home exercise during maintenance therapy for childhood acute lymphoblastic leukemia. Pediatr. Phys. Ther. 2014, 26, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Keats, M.R.; Culos-Reed, S.N. A community-based physical activity program for adolescents with cancer (project TREK): Program feasibility and preliminary findings. J. Pediatr. Hematol./Oncol. 2008, 30, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Rueegg, C.S.; Michel, G.; Wengenroth, L.; von der Weid, N.X.; Bergstraesser, E.; Kuehni, C.E. Physical performance limitations in adolescent and adult survivors of childhood cancer and their siblings. PLoS ONE 2012, 7, e47944. [Google Scholar] [CrossRef] [PubMed]
- Wurz, A.; Brunet, J. The effects of physical activity on health and quality of life in adolescent cancer survivors: A systematic review. JMIR Cancer 2016, 2, e6. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.F.; Kelly, M.J.; Aviva, M. Early nutrition and physical activity interventions in childhood cancer survivors. Curr. Obes. Rep. 2017, 6, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.F.; Meagher, S.; Scheurer, M.; Folta, S.; Finnan, E.; Criss, K.; Economos, C.; Dreyer, Z.; Kelly, M. Developing a web-based weight management program for childhood cancer survivors: Rationale and methods. JMIR Res. Protoc. 2016, 5, e214. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.F.; Parsons, S.K. Obesity in childhood cancer survivors: Call for early weight management. Adv. Nutr. 2015, 6, 611–619. [Google Scholar] [CrossRef] [PubMed]
- Fairey, A.S.; Courneya, K.S.; Field, C.J.; Mackey, J.R. Physical exercise and immune system function in cancer survivors: A comprehensive review and future direction. Cancer 2002, 94, 539–551. [Google Scholar] [CrossRef] [PubMed]
- Malina, R.M.; Bouchard, C.; Bar-Or, O. Growth, Maturation, and Physical Activity, 2nd ed.; Human Kinetics: Champaign, IL, USA, 2004; pp. 3–18. [Google Scholar]
- Li, T.; Wei, S.; Shi, Y.; Pang, S.; Qin, Q.; Yin, J.; Deng, Y.; Chen, Q.; Wei, S.; Nie, S.; et al. The dose-response effect of physical activity on cancer mortality: Findings from 71 prospective cohort studies. Br. J. Sports Med. 2016, 50, 339. [Google Scholar] [CrossRef] [PubMed]
- Hjorth, L.; Haupt, R.; Skinner, R.; Grabow, D.; Byrne, J.; Karner, S.; Levitt, G.; Michel, G.; van Der Pal, H.; Bárdi, E.; et al. Survivorship after childhood cancer: PanCare: A European network to promote optimal long-term care. Eur. J. Cancer 2015, 1, 1203–1211. [Google Scholar] [CrossRef] [PubMed]
- Wallace, W.H.; Blacklay, A.; Eiser, C.; Davies, H.; Hawkins, M.; Levitt, G.A.; Jenney, M.E.M. Regular review: Developing strategies for long term follow up of survivors of childhood cancer. BMJ 2001, 323, 271–274. [Google Scholar] [CrossRef] [PubMed]
- American Academy of Pediatrics Section on Hematology/Oncology, Children’s Oncology Group Long-Term Follow-Up Care for Pediatric Cancer Survivors. Pediatrics 2009, 123, 906–915. [CrossRef]
- Castellino, S.M.; Ullrich, N.J.; Whelen, M.J.; Lange, B.J. Developing interventions for cancer-related cognitive dysfunction in childhood cancer survivors. J. Natl. Cancer Inst. 2014, 106, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Langeveld, N.E.; Grootenhuis, M.A.; Voûte, P.A.; de Haan, R.J.; van den Bos, C. Quality of life, self-esteem and worries in young adult survivors of childhood cancer. Psychooncology 2004, 13, 867–881. [Google Scholar] [CrossRef] [PubMed]
- Huang, I.-C.; Brinkman, T.M.; Armstrong, G.T.; Leisenring, W.; Robison, L.L.; Krull, K.R. Emotional distress impacts quality of life evaluation: A report from the Childhood Cancer Survivor Study. J. Cancer Surv. 2017, 11, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Varni, J.W.; Katz, E.R.; Seid, M.; Quiggins, D.J.L.; Friedman-Bender, A. The pediatric cancer quality of life inventory-32 (PCQL-32). Cancer 1998, 82, 1184–1196. [Google Scholar] [CrossRef]
- The Children’s Oncology Group. Long-Term Follow-Up Guidelines for Survivors of Childhood, Adolescent, and Young Adult Cancers. Available online: http://www.survivorshipguidelines.org/ (accessed on 20 December 2017).
- Lohman, T.G.; Roche, A.F.; Martorell, R. (Eds.) Anthropometric Standardization Reference Manual; Human Kinetics; Information Systems Division, Naional Agricultural Library: Champaign, IL, USA, 1988.
- Stewart, A.; Marfell-Jones, M. International Standards for Anthropometric Assessment; International Society for the Advancement of Kinanthropometry: Lower Hutt, New Zealand, 2011. [Google Scholar]
- Roche, A.F.; Malina, R.M. Manual of Physical Status and Performance in Childhood; Plenum Press: New York, NY, USA, 1983; p. 1. [Google Scholar]
- Bachrach, L.K.; Sills, I.N. Clinical report-bone densitometry in children and adolescents. Pediatrics 2011, 127, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Cox, C.l.; Zuh, L.; Ojha, R.P.; Steen, B.D.; Ogg, S.; Robison, L.L.; Hudson, M.M. Factors supporting cardiomyopathy screening among at-risk adult survivors of pediatric malignancies. Support Care Cancer 2017, 25, 1307–1316. [Google Scholar] [CrossRef] [PubMed]
- Golden, E.; Beach, B.; Hastings, C. The pediatrician and medical care of the child with cancer. Pediatr. Clin. N. Am. 2002, 49, 1319–1338. [Google Scholar] [CrossRef]
- Stefani, L.; Pedrizzetti, G.; Galanti, G. Clinical application of 2D speckle tracking strain for assessing cardio-toxicity in oncology. J. Funct. Morphol. Kinesiol. 2016, 1, 343–354. [Google Scholar] [CrossRef]
- Voigt, J.U.; Pedrizzetti, G.; Lysyansky, P.; Marwick, T.M.; Houle, H.; Baumann, R.; Pedri, S.; Ito, Y.; Abe, Y.; Metz, S.; et al. Definitions for a common standard for 2D Speckle tracking echocardiography: Consensus document of the EACVI/ASE/industry task force to standardize deformation imaging. Eur. Heart J. Cardiovasc. Imaging 2015, 16, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Yu, A.F.; Raikhelkar, J.; Zabor, E.C.; Tonorezos, E.S.; Moskowitz, C.S.; Adsuar, R.; Mara, E.; Huie, K.; Oeffinger, K.C.; Steingart, R.M.; et al. Two-dimensional speckle tracking echocardiography detects subclinical left ventricular systolic dysfunction among adult survivors of childhood, adolescent, and young adult cancer. Cancer Biomed. Res. Int. 2016. [Google Scholar] [CrossRef] [PubMed]
- Leger, L. Aerobic performance. In Measurement in Pediatric Exercise Science; Dougherty, D., Ed.; Human Kinetics: Champaign, IL, USA, 1996; pp. 183–223. [Google Scholar]
- Borg, G.; Hassmen, P.; Whipp, B.J. Perceived exertion in relation to heart rate and blood lactate 12 during and arm and leg exercise. Eur. J. Appl. Physiol. 1985, 65, 679–685. [Google Scholar]
- Bayley, N. Bayley scales of infant and toddler development: Administration manual. In Harcourt Assessment; Pearson: San Antonio, TX, USA, 2006. [Google Scholar]
- Naumann, F.L.; Hunt, M.; Ali, D.; Wakefield, C.E.; Moultrie, K.; Cohn, R.J. Assessment of fundamental motor skills in childhood cancer patients. Pediatr. Blood Cancer 2015, 62, 2211–2215. [Google Scholar] [CrossRef] [PubMed]
- Moultrie, K.; Cohn, R.J. Assessment of gross motor skills and phenotype profile in children 9-11 years of age in survivors of acute lymphoblastic leukemia. Pediatr. Blood Cancer 2015, 1, 46–52. [Google Scholar] [CrossRef]
- Perry, J.; Davids, J.R. Gait analysis: Normal and pathological function. J. Pediatr. Orthop. 1992, 126, 815. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services. Physical Activity Guidelines for Americans; Department of Health and Human Services: Washington, DC, USA, 2008.
- McNeely, M.L.; Peddle, C.J.; Parliament, M.; Courneya, K.S. Cancer rehabilitation: Recommendations for integrating exercise programming in a clinical setting. Curr. Cancer Ther. Rev. 2006, 2, 251–260. [Google Scholar] [CrossRef]
- Malina, R.M. Movement proficiency in childhood: Implications for physical activity and youth sport. Kinesiol. Slov. 2012, 18, 19–34. [Google Scholar]
| Domain Being Measured | Item |
|---|---|
| Body size and nutritional status | Height (cm) |
| Weight (kg) | |
| BMI | |
| Head Circumference | |
| Skinfolds (mm) | |
| Motor Skill | Posture |
| Gait | |
| Balance | |
| Motor skill proficiency | |
| Flexibility | Sit and Reach |
| Ankle dorsiflexion | |
| Performance Level | Hand grip dynamometry (kg) |
| TUG-3 m (s) | |
| 30 s Chair stand (# of successful stands) | |
| Cardiovascular Fitness | PWC150 |
| Echocardiography | |
| CPET (VO2 peak) (mL∙kg−1∙min−1) | |
| Pulmonary Function | Spirometry (FEV, FEV1) |
| Quality of Life | PCQL Inventory |
| Considerations | Contraindications to Exercise Testing and Training | Precautions Requiring Modification and/or Physician Approval |
|---|---|---|
| Factors Related to Cancer Treatment | No exercise on days of intravenous chemotherapy (recommendation changing) | Caution if on treatments that affect the lung and/or heart: recommend medically supervised exercise testing and training |
| No exercise before blood draw | Mouth sores/ulcerations: avoid mouthpiece for maximal testing: use face mask | |
| Severe tissue reaction to radiation therapy | Lymphedema: wear appropriate compression garments | |
| Hematologic | Platelet Count <50,000 | Platelets >50,000–150,000: avoid tests or exercise (contact sports) that increase risk of bleeding |
| Hemoglobin level <10.0 g/dL | White blood cells >3000–4000: ensure proper sterilization of equipment | |
| Absolute Neutrophil Count <0.5 × 109/L | Hemoglobin >10 g/dL−11.5–13.5 g/dL: caution with maximal tests | |
| Avoid activities that may increase the risk of bacterial infection (swimming) | ||
| Musculoskeletal | Extreme fatigue/muscle weakness | Any pain or cramping: investigate |
| Bone, back or neck pain | Osteopenia: avoid high-impact exercise if risk of fracture | |
| Severe cachexia (loss of >35% premorbid weight) | Loss of muscle mass limits exercise to mild intensity | |
| Karnofsky performance status score <60%; Poor functional status: avoid exercise testing | Cachexia: multidisciplinary approach to exercise | |
| Systemic | Acute infections | May indicate systemic infection and should be investigated. Avoid high intensity exercise |
| Febrile illness: fever >100 F | Avoid exercise until asymptomatic for >48 h | |
| General Malaise | ||
| Gastrointestinal | Severe Nausea | Compromised fluid and/or food intake: recommend multidisciplinary approach/consultation with nutritionist |
| Dehydration | Ensure adequate nutrition with electrolyte drinks and water (avoid hyponatremia) | |
| Vomiting or diarrhea within 24–36 h | ||
| Poor nutrition: inadequate fluid and/or intake | ||
| Cardiovascular | Chest pain | Exercise is contraindicated (refer to physician) |
| Resting HR >100 bpm or <50 bpm | Caution: recommend medically supervised exercise testing and training | |
| Resting SBP >145 mm Hg and/or DBP >95 mm Hg | ||
| Resting SBP <85 mm Hg | ||
| Irregular HR | Exercise is contraindicated (refer to physician) | |
| Swelling of ankles | Lymphedema: wear appropriate compression garments | |
| Pulmonary | Dyspnea | Mild to moderate dyspnea: avoid maximal tests |
| Cough, wheezing | Avoid activities that require significant oxygen transport (high intensity X) | |
| Chest pain increased by deep breath | ||
| Neurologic | Ataxia/Dizziness/peripheral Sensory Neuropathy | Avoid activities that require significant balance and coordination (treadmill) |
| Significant decline in cognitive performance | Ensure patient is able to understand and follow instructions | |
| Disorientation | Use well supported positions for exercise | |
| Blurred vision |
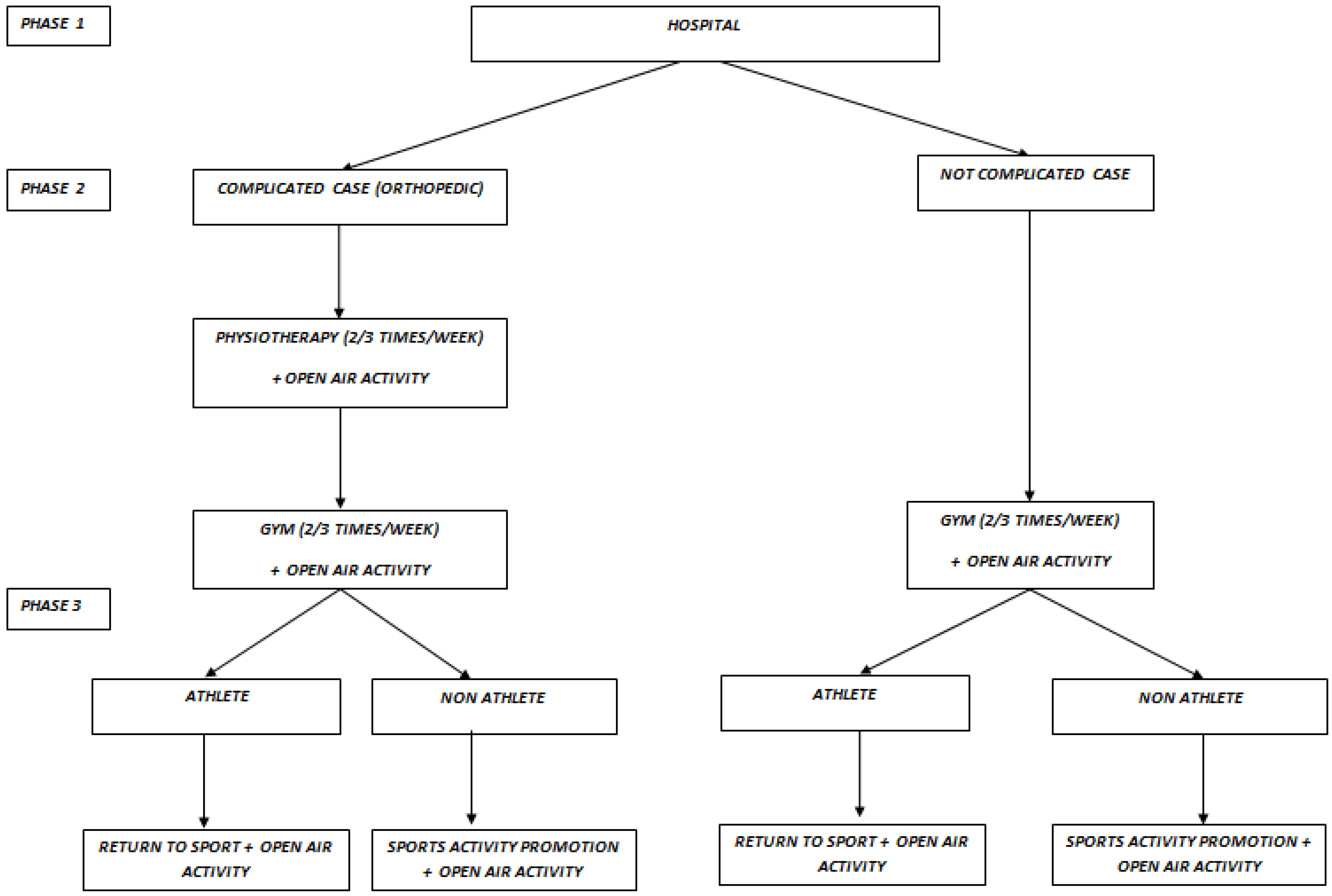
| Phase | Where | Assessment | Exercise Plan | Comments |
|---|---|---|---|---|
| I | Hospital Based | Anthropometry: height, weight, BMI, head circumference, skinfolds and muscle circumferences. Consult with dietitian and oncology team if patient appears malnourished. | Hospital based under supervision. | Training plan: Emphasis on fundamental movements patterns (motor skill) and fun |
| Orthopedic evaluation: Abnormal: refer to pediatric orthopedic team. | ||||
| Look for any obvious swellings or surgical scars. | Modality: play (if possible), walking, cycle ergometer, stretching. | |||
| Assess for deformity: scoliosis, kyphosis, loss of lumbar lordosis or hyperlordosis of the lumbar spine. Look for shoulder asymmetry and pelvic tilt. Postural scoliosis resolves on bending, structural scoliosis does not resolve. | Frequency 2–5x per week | |||
| Observe the patient walking to assess for any abnormalities of gait. | Intensity: Low-to moderate HR | |||
| Cardiopulmonary Exercise Testing (CPET)—Cycle ergometry, ECG, O2 saturation, HR, RPE. | Duration: 5–60 min per session depending on status | |||
| Power work capacity: Work capacity at Heart rate of 150 bpm | ||||
| Pulmonary function testing: Spirometry | Play: balance, agility, hopping, skipping, throwing | |||
| Motor skill: Gait, Time up and go | Routine: Warm up exercises with body weight, play or aerobic exercise, strength exercises, cool down. | |||
| Flexibility: sit and reach | Monitor HR, O2 saturation if needed, dyspnea, RPE, fatigue level | |||
| Strength: Hand grip dynamometry, 30 s chair stand | ||||
| Quality of Life: PedQoL | ||||
| II | Transition to Home based | Assessment: Nurse, physical therapist, psychologist, cancer specialist in addition to medical team | Home-based under parental supervision. | Training plan: Emphasis on aerobic fitness, fundamental movements patterns (motor skill) and fun |
| Anthropometry: height, weight, BMI, head circumference, skinfolds and muscle circumferences. Consult with dietitian and oncology team if patient appears malnourished. | Goal: return to normal activities and sport activities | |||
| Power work capacity: Work capacity at Heart rate of 150 bpm | ||||
| Pulmonary function testing: Spirometry | Modality: play (if possible), walking, cycle ergometer, stretching. | |||
| Motor skill: Gait, TUG | Frequency: 3–5x per week | |||
| Flexibility: sit and reach | Education materials for parents and siblings. | Intensity: Low-to moderate HR | ||
| Strength: Hand grip dynamometry, 30 s chair stand | Duration: 20–60 min per session depending on status | |||
| Play: balance, agility, hopping, skipping, throwing | ||||
| Routine: Warm up exercises with body weight, play or aerobic exercise, strength exercises, cool down. | ||||
| Monitor HR and complete home exercise log. | ||||
| III | Home based and Independent | Assessment at 6 months and 12 months, 5, 10, 15 years (in addition to medical team) | Home-based no supervision | Emphasis on strength, proficiency, postural control, cardiovascular conditioning and fun |
| Anthropometry: height, weight, BMI, head circumference, skinfolds and muscle circumferences. | Goal: return to normal activities and sport activities | |||
| Power work capacity: Work capacity at Heart rate of 150 bpm | ||||
| Pulmonary function testing: Spirometry | Modality: play (if possible), walking, cycle ergometer, stretching, sports activities. | |||
| Motor skill: Gait, TUG | Frequency: 3–5x per week | |||
| Flexibility: sit and reach | Intensity: Low, moderate and vigorous HR | |||
| Strength: Hand grip dynamometry, 30-s chair stand | Duration: 30–60 min per session depending on status | |||
| Play: team sports (football), swimming. | ||||
| Routine: Warm up exercises with body weight, play or aerobic exercise, strength exercises, cool down. | ||||
| Monitor HR and complete home exercise log. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klika, R.; Tamburini, A.; Galanti, G.; Mascherini, G.; Stefani, L. The Role of Exercise in Pediatric and Adolescent Cancers: A Review of Assessments and Suggestions for Clinical Implementation. J. Funct. Morphol. Kinesiol. 2018, 3, 7. https://doi.org/10.3390/jfmk3010007
Klika R, Tamburini A, Galanti G, Mascherini G, Stefani L. The Role of Exercise in Pediatric and Adolescent Cancers: A Review of Assessments and Suggestions for Clinical Implementation. Journal of Functional Morphology and Kinesiology. 2018; 3(1):7. https://doi.org/10.3390/jfmk3010007
Chicago/Turabian StyleKlika, Riggs, Angela Tamburini, Giorgio Galanti, Gabriele Mascherini, and Laura Stefani. 2018. "The Role of Exercise in Pediatric and Adolescent Cancers: A Review of Assessments and Suggestions for Clinical Implementation" Journal of Functional Morphology and Kinesiology 3, no. 1: 7. https://doi.org/10.3390/jfmk3010007
APA StyleKlika, R., Tamburini, A., Galanti, G., Mascherini, G., & Stefani, L. (2018). The Role of Exercise in Pediatric and Adolescent Cancers: A Review of Assessments and Suggestions for Clinical Implementation. Journal of Functional Morphology and Kinesiology, 3(1), 7. https://doi.org/10.3390/jfmk3010007








