1. Introduction
Haemophilia is a disorder of bleeding, in which most of the bleeding occurs into muscles and joints, leading to severe destructive arthritis. There is currently no known cure, but as a result of medical advancements, many people with haemophilia (PWH) are living longer. In fact, the most recent review [
1] concluded that the current life expectancy of haemophilia patients has approached that of males in the general population. Increasingly, age-related co-morbidities including cardiovascular disease, hypertension, diabetes, cancer, renal disease and overweight/ obesity have been described in the PWH [
2,
3,
4,
5,
6], but joint arthropathy remains the main co-morbidity [
3,
6].
Among older people without haemophilia (PWOH), falls and fall-related injuries are a common and serious problem. Approximately, sixty per cent of non-spinal fractures are the result of a fall, with the highest incidence in men occurring in the sixth decade of life. Data comparing falls incidence in the general population from 2000 with that of 2010 show that the incidence of fractures following a fall are on the increase, possibly due to the increase in diabetes and obesity [
7].
It has been reported that older people with osteoarthritis of the knee have a two-fold increase in their likelihood of having a fall and those with osteoarthritis of the ankle have significantly poorer balance than those without osteoarthritis of the ankle joint [
8,
9]. Although the underlying mechanism is not known, it is thought that muscle weakness and altered proprioception may play a role. PWH who have not benefited from prophylaxis treatment using recombinant clotting factor concentrates during childhood show signs of haemophilic arthritis, an end-stage pattern of multiple joint impairment and disability, including reduced joint range of motion, muscle weakness and atrophy, in young adulthood, suggesting they may be at risk of falls earlier than unaffected people and before the onset of age-related co-morbidities [
10]. Recent studies have shown that one in three men with haemophilia aged over the age of 40 years has suffered a fall [
11,
12]. Of concern is that PWH have been shown to have a higher risk of having low bone mineral density [
13] and therefore the consequences of increased rates of falling with age may have marked detrimental effects on bone fracture rates, mobilityand healthcare costs in terms of coagulation factor treatment and subsequent social care costs This in comparison to PWOH, who have been shown to have on fall event at a rate of between 33–40% after 65 years of age [
14,
15].
For a person to maintain balance, coordination of the vestibular, somatosensory, and visual systems is required. Measurement of postural sway, the horizontal movement of the body around the centre of gravity, is commonly used to evaluate postural stability, and disturbance of input from the sensory systems will affect postural sway [
16,
17,
18,
19,
20,
21]. Studies of postural sway in young adults also suggest that fatigue of the hip and ankle muscles increases postural sway [
16,
17]. Furthermore fatigue time and the length of time standing increases postural sway and velocity [
18,
19]. The presence of lower limb joint arthropathy together with the associated muscle atrophy and reduced strength in PWH suggests that balance stability may decrease the longer they stand.
Despite increasing age of PWH and the early incidence of arthropathy, a recent systematic review of the orthopaedic co-morbidities in the older haemophilic population revealed few studies have investigated balance and the risk of falls [
20]. Gallach et al. [
21] compared standing balance in a group of 50 adult PWH and 25 age-matched healthy non-haemophilic men and observed greater amplitude and velocity of postural sway in men with haemophilia during standing. In a study of twenty PWH aged 22–58 years, Fearn et al. [
22] reported that half of haemophilic men had experienced a fall within the previous 12 months. The underlying cause of the alterations in balance and greater frequency and risk of falling together with how best to identify those at risk of falling are not clear from the studies.
Thabti and colleagues [
23] recently studied the effect of vision and floor surface on balance performance in adolescent boys with haemophilia, compared to their unaffected peers. Using a forceplate to record the speed of displacement of the body’s centre of pressure with the floor, they found that both groups increased the speed of postural sway when they closed their eyes and to a greater extent when they stood on a 60 mm thick foam rubber mat. When compared to their healthy peers, significantly higher speeds of postural sway for the eyes closed and foam conditions, but not when standing on a hard floor surface with eyes open, were seen in the adolescents with haemophilia. It is not known if the speed of postural sway increased uniformly in the sagittal and coronal planes.
The aim of this study is to evaluate postural stability, together with lower limb joint function and mobility in a group of older men with haemophilia. On the basis of the above we hypothesised that postural stability, joint function and mobility would be significantly worse in men with haemophilia when compared to aged-matched men without haemophilia. Additionally, we hypothesised that postural stability would significantly deteriorate in men with haemophilia the longer they stood.
2. Materials and Methods
2.1. Participants
At a single UK Haemophilia Centre, patients with severe haemophilia A or B and aged more than 40 years and that were fearful of falling were provided with an information sheet and letter at their routine clinic appointment inviting them to participate in the study. Eight males aged between 4982 years old with severe haemophilia (PWH) were recruited. The study was approved by HRA NRES Committee London—Camden & Islington (Ref: 14/LO/2094) and complied with the tenets of the Declaration of Helsinki. In addition, eight matched males without haemophilia (PWOH) aged between 49–63 years old were recruited voluntarily as controls via poster and online staff network advertisements at the study institution. Written informed consent was obtained from all participants in both groups who volunteered to participate. Participants who had a past history of acquired brain injury or any other disturbance of the central nervous system or who were unable to fully comply with verbal instructions were excluded. Independent samples t-tests were completed demonstrating there were no significant difference in age (mean age: PWH 60.5 ± 13.5 years, PWOH 57.3 ± 4.8 years, t (8.768) = 0.641, p > 0.05), height (mean height: PWH 177.1 ± 10.1 cm, PWOH 178.3 ±6.3 cm, t (14) = −0.289, p > 0.05), or body mass (mean body mass: PWH 86.1 ± 11.1 kg, PWOH 88.8 ± 7.4 kg, t (14) = −0.575, p > 0.05) between the two groups.
2.2. Procedures
Participants’ age, height, mass, and shoe size were recorded prior to data collection. Haemophilia Joint Health Score was performed by a qualified physiotherapist according to the protocol described by Hilliard and colleagues [
24], and reported as total lower limb, knee joint, and ankle joint scores.
Musculoskeletal ultrasounds were taken of participants’ ankle and knee joints using a Logiq-e system (GE Medical Systems Co., Ltd., Wuxi, China) and 8L-RS transducer probe, and these images were categorised using the Haemophilia Early Arthropathy Detection with Ultrasound (HEAD-US) score. This grading was performed by a qualified physiotherapist who had previously undertaken HEAD-US training, and reported as total lower limb, knee joint, and ankle joint scores.
Ankle and knee joint passive Range of Motion (RoM—flexion, extension, and total motion) was assessed via a dual arm goniometer by the same physiotherapist (DS).
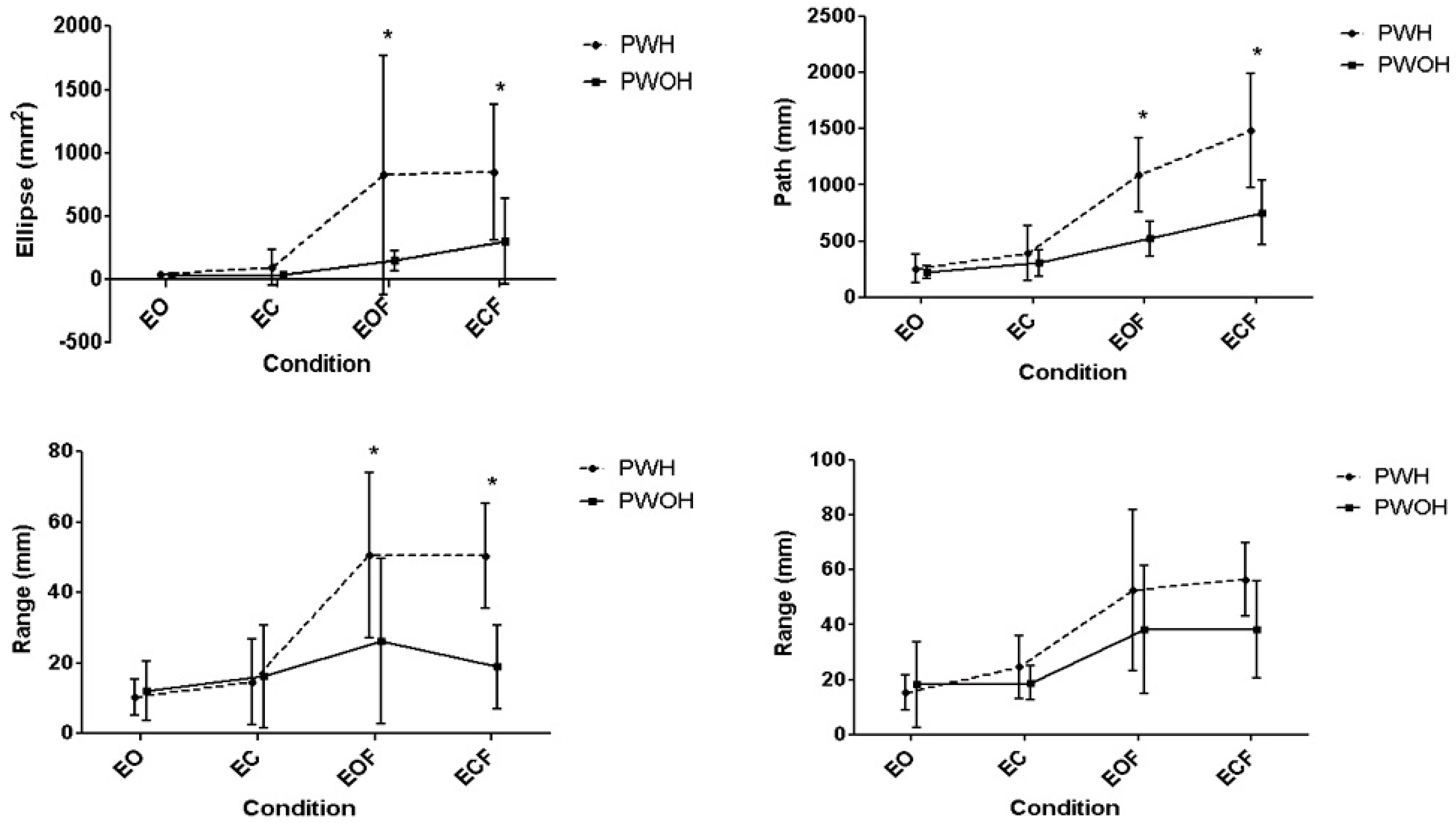
Postural Stability was assessed by asking participants to stand barefoot in a bilateral standing position on a 1 metre RSscan FootScan pressure plate (RSscan International, Paal, Belgium) for a period of 60 s quiet stance, whilst plantar pressure measurements (8 Hz, footscan Balance software, v7.7, RSscan International) and qualitative observations were recorded. This was performed four times, each in a different condition challenging different somatosensory contributions to balance stability. The participants completed the task with their eyes open (EO), focusing on a point on the wall in front of them. This was followed by three perturbed conditions combining visual and physical perturbations. Firstly, they repeated the same task but with a visual perturbation, their eyes closed (EC). Secondly, they completed the eyes open task (EO) with the addition of a physical perturbation, a 0.075 m thick high density foam block (Eyes Open Foam, EOF). Finally, they completed the task with both visual and physical perturbations (Eyes Closed Foam, ECF). Foot position (including foot rotation and stance width) was noted in condition one (eyes open) and the same stance position was used for subsequent conditions. An investigator was positioned behind the participant at all times to minimise the risk of falling. Data on the centre of pressure (CoP) path during each of the test conditions were extracted and reported as maximal mediolateral (ML) excursion, maximal antero-posterior (AP) excursion, total excursion (total path), considered to be indicative of effort to maintain stability, and CoP ellipse, considered to be a direct indication of stability. Average velocity of the path of the CoP was also calculated.
Participants were asked to walk barefoot for two minutes around two cones placed 15 m apart for a Timed 2-min walk test (2MWT) which has been shown to correlate with muscle strength and joint arthropathy in children with haemophilia [
25]. They were instructed to walk as fast as they could maintain for two minutes without running, maintaining a period of double support, until they were asked to stop. The distance walked was recorded by counting the number of shuttles completed, multiplied by 15 m, then adding the additional distance from the final uncompleted shuttle measured with a tape measure. This measure has been reported to have excellent test-retest reliability (0.98 CI) [
26,
27].
2.3. Statistical Analysis
Normality was assessed using Shapiro-Wilk tests using SPSS (v23.0.0.3, IBM Corporation, Armonk, NY, USA). Where data were non-parametric they were normalised using Log10 transformations [
28,
29]. Mixed-design ANOVA were used for condition × group (fixed effect factor), and for time × condition (beginning, middle and end; duration 20 s) × group (random effect factor) comparisons. Independent samples pairwise comparisons with Bonferroni correction was used for all post-hoc. Sphericity was assessed using Mauchley’s test, with any violations of the test assumptions being corrected with a Greenhouse-Geisser. For all statistical tests, effect sizes were calculated, Cohen’s
d for the pairwise comparisons and partial Eta
2 (
) for the mixed design ANOVA’s, and were interpreted as small (
d = 0.2;
= 0.01), medium (
d = 0.5;
= 0.09), or large (
d = 0.8;
= 0.25) effects [
30].
3. Results
PWH were associated with significantly worse scores for joint health and HEAD-US score. Exhibited significantly lower total dorsiflexion (DF) and plantarflexion (PF), ankle range of motion (RoM), extension across both knee joints, and significantly lower total RoM and flexion in the left knee joint than PWOH (see
Table 1). PWH walked significantly lower distance during the 2MWT than PWOH. As Cohen’s
d values were all above 0.8, the statistically significant differences were deemed to be large effects [
26].
Increases in CoP ellipse and path were evident as level of perturbation increased within both groups (Ellipse
p < 0.001,
= 0.79; Path
p < 0.001,
= 0.86), with post-hoc pairwise comparisons establishing the physical intervention (foam) as the source of the significant increases across participants (
Figure 1). Furthermore, a significant interaction between condition and group (Ellipse
p = 0.028,
= 0.19; Path
p = 0.004,
= 0.27) indicated different responses to perturbation between groups. Post hoc testing identified significantly larger increases in CoP ellipse and path in the PWH group under both foam conditions (
p < 0.001).
Deconstructing CoP measures into AP and ML components identified significant effects from condition (AP
p < 0.001,
= 0.70, ML
p < 0.001,
= 0.64). Bonferroni post-hoc analysis identified significant differences between EO and EOF (
p = 0.001), EO and ECF (
p < 0.001), EC and EOF (
p = 0.003) and EC and ECF (
p < 0.001) in the ML and AP range of CoP (
Table 2). Furthermore, the AP measure exhibited a significant interaction between condition and group (
p = 0.001,
= 0.34). Post hoc testing identified that the magnitude of the increases in the PWH group were significantly larger than the PWOH in the physical perturbation conditions (EOF
p = 0.018; ECF
p < 0.001).
The average velocity of the CoP path significantly increased across conditions for all participants (
p = 0.001,
= 0.80). Bonferroni post-hoc analysis identified significant (
p < 0.05) increases in velocity between across all comparisons (
Table 2). There was also significant interaction between condition and group (
p = 0.001,
= 0.43), with PWH experiencing significant increases in CoP velocity across all conditions (
p < 0.001–0.036), whilst PWOH only experienced significant increases in CoP velocity between the EO and the foam conditions (EO and EOF
p = 0.022; EO and ECF
p = 0.015). There was a significant differences between the groups for the two foam conditions (EOF
p < 0.001; ECF
p = 0.003), with PWH exhibiting higher CoP velocities.
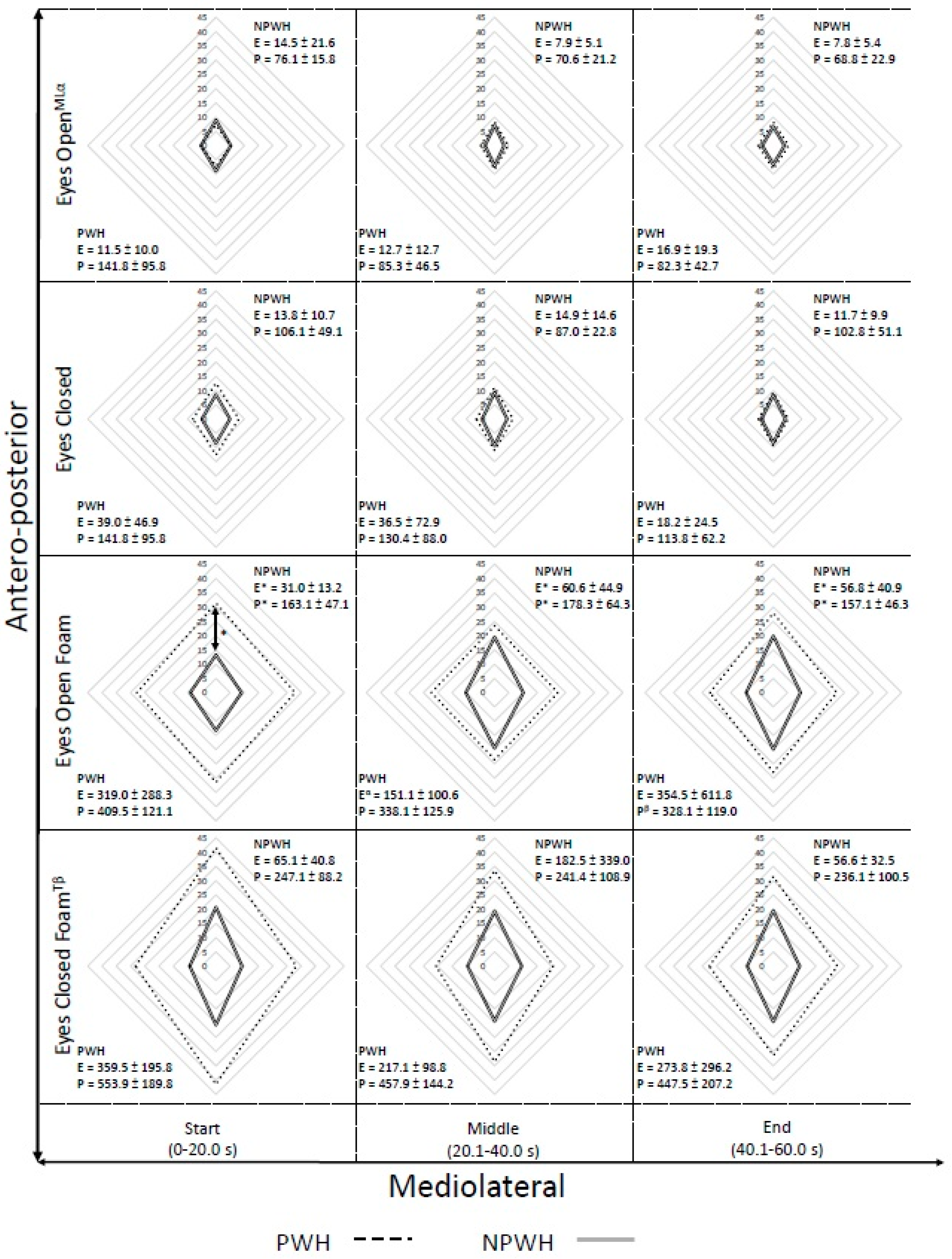
Secondary analysis, within-condition, was conducted assessing postural stability across the duration of the task. Comparisons across group were made between the first, middle and last 20 s period, within each condition, to determine if there was a temporal influence in the postural control (
Figure 2).
Significant effects of time and group by time were clustered when the physical perturbation was introduced. A statistically significant interaction of group and time was observed under EOF in the CoP Ellipse (f(2,28) = 5.319, p = 0.011, = 0.28), CoP path(f(2,28) = 3.790, p = 0.038, = 0.21) and CoP AP range (f(2,28) = 4.467, p = 0.021, = 0.24). Bonferroni post-hoc assessment identified that significant differences were apparent between groups at all time points (start, p < 0.001; middle, p = 0.023; end, p = 0.016) for CoP ellipse, with the PWH exhibiting a significantly larger ellipse, but only the PWH demonstrated significant difference between the start and middle 20 s (p = 0.030) under the EOF condition. Similarly, the CoP path demonstrated significant differences between groups at all time points (start, p < 0.001; middle, p = 0.006; end, p = 0.001), however only PWH demonstrated significant difference across time (start vs. end p = 0.013). The final significant interaction for the EOF condition, in the AP CoP range, was driven by a significant difference between groups during the first 20 s of the trial. ECF condition demonstrated a significant effect of time (f(2,28) = 3.680, p = 0.038, = 0.21) in the CoP Path measure, however Bonferroni post hoc correction were unable to identify the source of this finding. A significant effect was demonstrated by time in the ML CoP range under the EO condition (f(2,28) = 4.756, p = 0.017, = 0.25). Post hoc identified that there was a significantly greater ML range at the start of the trial than the middle (p = 0.019). When CoP velocity was assessed for changes over the duration of the balance task, the EO and EC conditions displayed no significant differences, but EOF demonstrated a significant interaction of Group*Time (p = 0.042, = 0.20), with velocity decreasing significantly across the task duration, and Bonferroni post-hoc tests revealing differences between PWH and PWOH groups at all time periods (start p < 0.001; middle p = 0.006; end p = 0.002). Additionally, there was a significant reduction of CoP velocity over time in the ECF condition (p = 0.047, = 0.20), with post-hoc least significance differences tests identifying the difference to be between the start and end periods (p = 0.046), as Bonferroni was too conservative to be able to identify where the differences lay.
4. Discussion
Few studies have compared postural stability, joint function and mobility between older men with and without haemophilia. We hypothesised that due to the presence of haemophilic arthropathy, the postural stability, joint function and mobility would be significantly worse in men with haemophilia. During quiet standing under normal and mildly perturbed conditions (EO and EC), postural stability was broadly comparable between the two groups as indicated by the CoP excursions observed. These findings suggest that static balance on a level surface is not sufficient to identify balance impairments in men with haemophilia. A more challenging measure of stability may be necessary to determine the difference between those with, and those without, haemophilia. Moreover, results indicate that all participants are able to maintain stability under visual restriction regardless of standing surface, as visual restriction did not demonstrate significant alterations to CoP motion regardless of standing surface. This is in contrast to Thabti, Besusoleil and Hue’s [
23] study using adolescents, who did find a difference between their EO and EC conditions.
The introduction of the physical perturbation (foam) caused both groups to significantly increase CoP motion. However the magnitude of these increases were not equal and PWH exhibited significantly greater changes in CoP motion and velocity, similar to those found by Thabti, Besusoleil and Hue [
23]. Specifically, the physical perturbation significantly increased the effort required (total path), and reduced the stability (larger ellipse), and increased the CoP velocity of the PWH when compared with the age matched control group. Further differentiation was apparent when the CoP was separated into AP and ML directions. ML CoP range did not differ significantly with perturbation condition. Instead, the overall increase in CoP motion, caused by the physical perturbation, was attributed to the AP CoP excursion. This study is unable to determine the underlying mechanisms that contributed to the increases along with the altered patterns of postural sway seen in the men with haemophilia. Marked arthropathy of the ankle and knee joints as indicated by the HJHS (both separately at the ankle and knee joints, and when combined together with gait score for the total lower limb score), the HEAD-US scores (total lower limb score, and at the ankle joint separately) and sagittal plane RoM exhibited by the PWH group and the joint stiffness and muscle weakness associated with this arthropathy may have contributed. In addition, disruption of the joint mechanoreceptors as a result of joint bleeding may impair the proprioceptive input as indicated by the large increase in postural sway in the foam conditions. Further research is required to determine the contribution of mechanical and sensorimotor impairment to postural stability in this population.
It has been proposed that maintenance of balance is accomplished by two distinct strategies, namely, the ankle strategy and the hip strategy [
31]. These strategies might be used separately or together in varying degrees to produce optimal and adaptable balance control, depending on the difficulty of the balance task and amount of perturbation [
32,
33,
34]. It has been suggested that the ankle strategy is responsible for small corrections of balance and the hip strategy, larger corrections. The larger CoP displacement of PWH in the current study suggest a reliance on the hip strategy. The chronic ankle joint arthropathy observed in PWH, resulting in significantly less ankle joint dorsi and plantarflexion range of motion compared to the age-matched controls (PWOH), coupled with the muscle atrophy and weakness associated with arthropathy may have contributed to the dominant hip strategy observed. Moreover, findings indicate that the presence of early arthropathy can be linked to instability in PWH, as well as limiting the functional performance, supported by the significantly shorter distance covered during the 2MWT protocol and the reduced ankle and knee joint RoM.
Secondary analysis of changes over time, within condition, indicated a significant difference between groups. However temporal alterations were sporadic without discernible consistency. This indicates that duration of standing time has no influence on CoP motion, although task duration was relatively short (60 s), and prolonged standing protocols may identify changes in CoP motion over time. There was, however, an influence of time on the CoP velocity, with both groups able to decrease the velocity as the task progressed. Further consideration of overall findings may require some caution, due to the small sample size and the influence of body mass on the compression of the foam block may be important to consider. As those with greater body mass were able to compress the foam to a greater degree than lighter participants, this may have affected the level of proprioception received during this condition. Supplementary assessment of this was conducted comparing participants heavier and lighter than the sample average, from which no significant differences were observed. However this variation in compression may account for the large variance displayed across subjects.
Overall, findings allow the cautious acceptance of the primary hypothesis, namely that older PWH have significantly worse postural stability, joint function and mobility when compared to older men without haemophilia under perturbed conditions. However only the physical perturbation accounted for this, with removal of visual information having no measurable effect. The secondary hypothesis that time would decrease the postural stability of the PWH was not supported. Whilst it was clear that there were significant decreases compared to men without haemophilia across all time intervals, the significant differences within the PWH group across the time intervals indicated some improvements in postural stability as time progressed, although a short time period of 60 s was used overall.
5. Conclusions
This study has provided pilot data on significant differences between older PWH and PWOH, which can provide a focus for further studies seeking to measure and improve balance and falls risk in PWH. Ability to maintain postural stability in both PWH and PWOH is greatly decreased when a physical perturbation is introduced, but not affected by removal of visual reference, which was in contrast to adolescents [
23]. This effect is magnified in PWH, as participants exhibited significantly greater increases in CoP path, ellipse, and velocity, the cause of which is believed to be the haemophilic arthropathy identified in the joint health and HEAD-US scores. Furthermore, the presence of haemophilic arthropathy and reduced ankle and knee mobility does not appear to significantly affect the PWH ability to maintain ML stability, rather it is the AP motion that is compromised, something that previous studies have not specified [
23]. The reduced postural stability in PWH may increase their risk of falls and musculoskeletal injuries. Currently, there is little information on how to identify PWH who are at risk of losing their balance and falling, and some current measures may not accurately identify fallers [
35]. Moving forward, if we are to identify those at risk, the results of this study suggest that dynamic tests incorporating physical or external perturbation may be more effective than static balance tests on a level surface. Assessment of postural stability over a longer period of time may be needed to determine whether fatigue plays any role in the ability of PWH to correct their balance. In addition, the possible dominance of a hip strategy to achieve balance suggests falls prevention programs might need to focus on increasing hip strength and retraining ankle strategy movement to allow PWH to improve balance stability.