Comparative Assessment and Deployment of Zeolites, MOFs, and Activated Carbons for CO2 Capture and Geological Sequestration Applications
Abstract
1. Introduction
2. Background on Porous Materials for CO2 Capture
2.1. Methodology and Evaluation Criteria
2.2. Definition of Properties
2.2.1. Surface Area (m2/g)
2.2.2. CO2 Adsorption Capacity (mmol/g)
2.2.3. Regeneration Ease
2.2.4. Cost (Inverse Scale, USD/kg)
2.2.5. Moisture Resistance
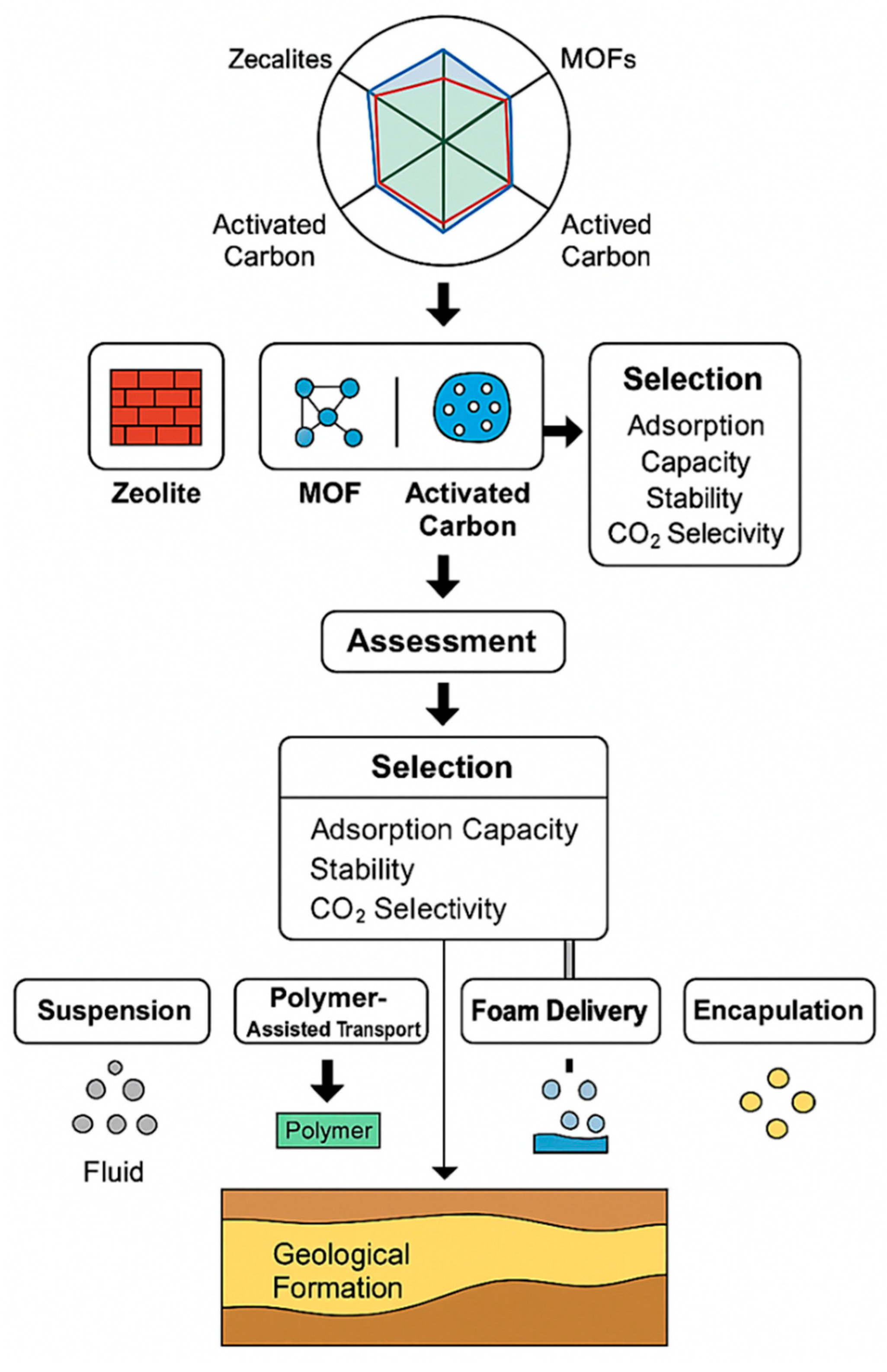
3. Comparative Performance of Zeolites, MOFs, and Activated Carbons
3.1. Zeolites
3.2. Metal–Organic Frameworks (MOFs)
3.3. Activated Carbons
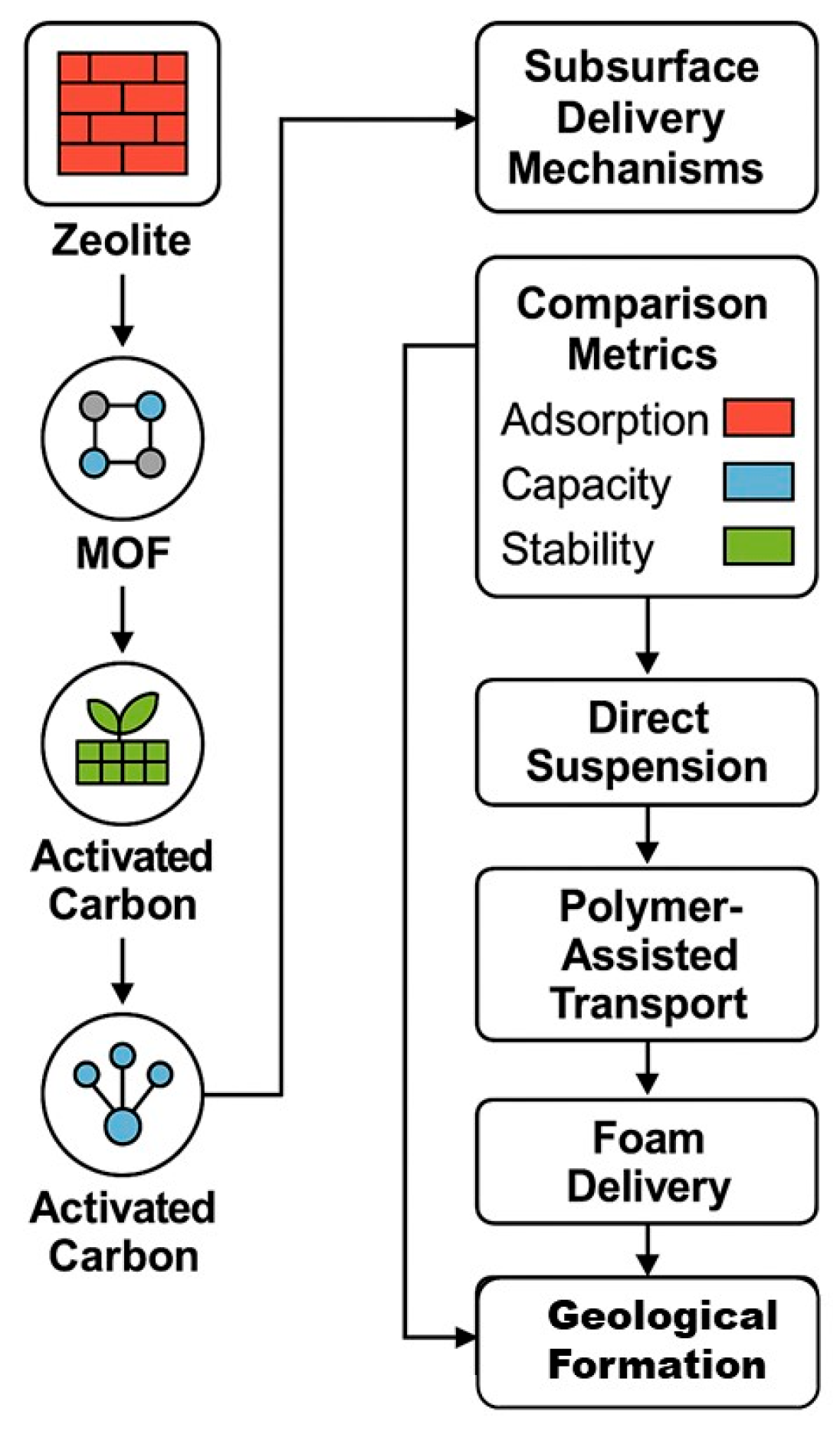
4. Delivery Mechanisms for Subsurface Sequestration
5. Hybrid Systems and Advanced Materials for CO2 Sequestration
5.1. MOF–Zeolite and MOF–Carbon Composites
5.2. Polymer-Integrated Smart Adsorbents
5.3. AI-Guided Materials Design and Screening
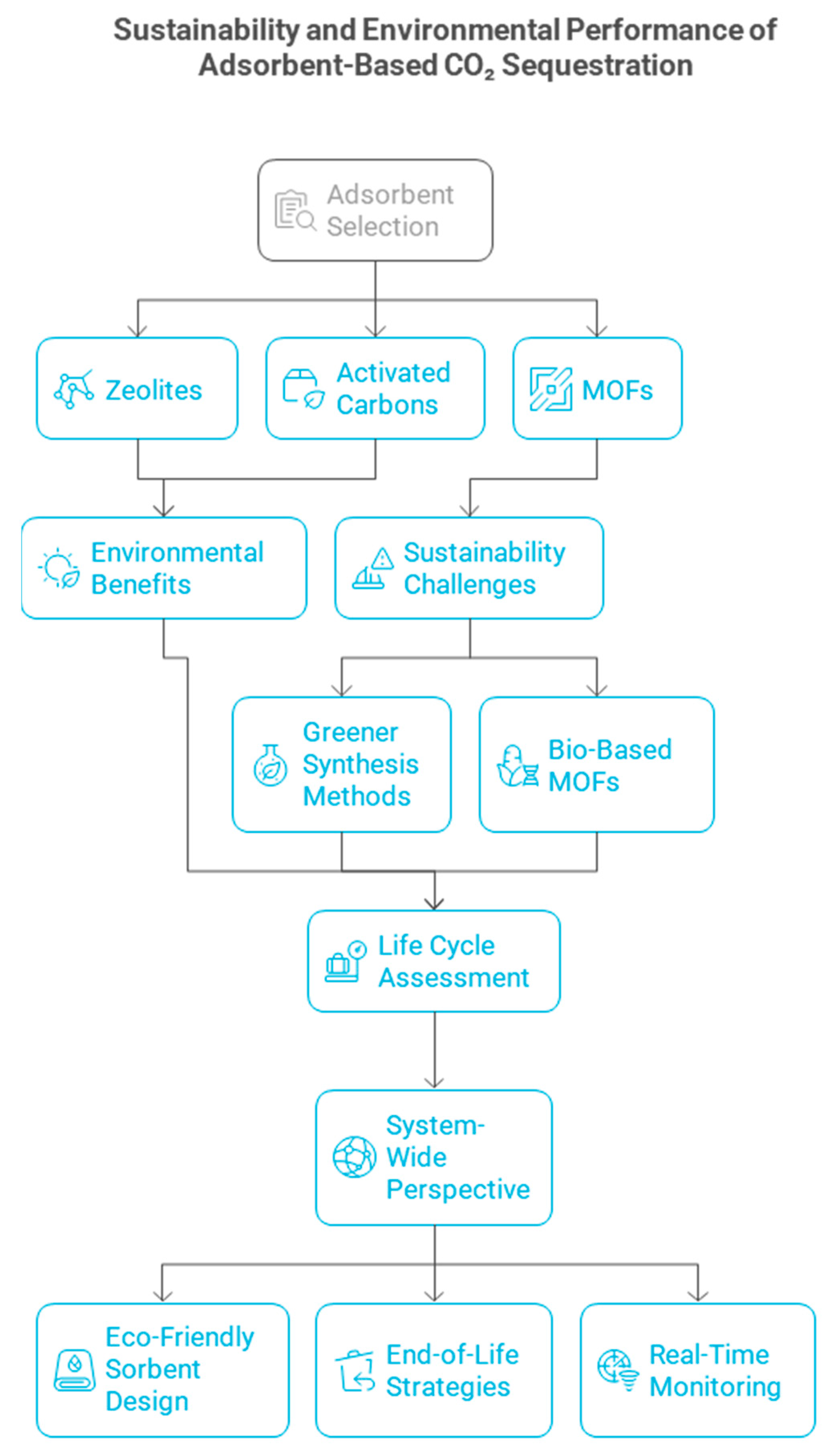
5.4. Environmental and Economic Considerations
5.5. Smart Adsorbents for Saline and Harsh Subsurface Environments
5.6. AI-Assisted Discovery and Deployment of Hybrid Materials
6. Discussion: Deployment Challenges and Climate Policy Alignment
6.1. Infrastructure and Field Compatibility
6.2. Monitoring and Verification Technologies
6.3. Lifecycle and Economic Viability
6.4. Policy Incentives and International Commitments
6.5. Toward Integrated CCS Ecosystems
6.6. Extrapolating Laboratory Data to Field Conditions
7. Sustainability and Environmental Performance of Adsorbent-Based CO2 Sequestration
Life Cycle Assessment (LCA) of Adsorbent Materials
8. Limitations and Recommendations for Future Research
8.1. Data Gaps and Standardization Deficiencies
8.2. Scaling and Integration Challenges of Porous Materials
8.3. Field Deployment Uncertainty and Environmental Sensitivity
9. Conclusions, Challenges, and Opportunities
9.1. Conclusions
9.2. Challenges and Opportunities in Advancing Adsorbent-Based Carbon Storage
- Lack of Large-Scale Field Validation: While many adsorbent materials and delivery techniques have shown great promise in laboratory settings or small-scale pilot studies, there is still a significant gap in demonstrating their performance in real-world, large-scale environments. Subsurface geological formations are highly complex and variable, and without full-scale field trials, it is difficult to fully understand how these systems will behave. Key questions remain around how well materials can be injected, how long they will stay in place, and how effectively they can capture and retain CO2 in different geological conditions. Long-term field validation is crucial to prove these technologies are not only effective but also reliable and safe over time;
- Uncertainty in Long-Term Material Stability: Another major concern is the durability of the materials used—especially porous adsorbents like MOFs (metal–organic frameworks), zeolites, and activated carbons. These materials may be exposed to harsh subsurface conditions, including high pressures, fluctuating temperatures, and varying salinity levels. Over time, this can lead to chemical degradation, changes in mechanical strength, or loss of adsorption capacity. Currently, we lack a clear understanding of how these materials behave over the long term, particularly over periods of several years or decades. Research is needed to explore their degradation pathways, resilience under cyclical loading, and potential interactions with native reservoir fluids;
- Integration with Carbon Capture and Utilization (CCU): Beyond simply storing CO2, there is a growing interest in turning captured carbon into useful products—what is known as Carbon Capture and Utilization (CCU). For this vision to be realized, adsorbents must not only capture CO2 efficiently but also allow for its recovery and regeneration so that it can be fed into catalytic processes or industrial value chains. Designing materials that can be regenerated repeatedly and are compatible with downstream applications is a critical next step toward circular and economically viable carbon management;
- Environmental and Economic Assessments: To ensure that these technologies are both sustainable and practical, we need a better understanding of their environmental and economic impacts. This includes analyzing the full life cycle of adsorbent materials—from raw material extraction and manufacturing to deployment and disposal—as well as considering their supply chain emissions and overall cost-effectiveness. This is especially important for newer materials like MOFs and hybrids, which may involve rare or energy-intensive components. Robust life cycle assessment (LCA) and techno-economic analysis (TEA) can help guide responsible development and inform policy and investment decisions;
- Smart Monitoring and Data-Driven Optimization: One of the most exciting frontiers lies in using smart sensing and data analytics to improve the performance and safety of CCS systems. For example, adsorbents could be functionalized with tracers or responsive sensors that enable real-time tracking of CO2 movement and material behavior underground. These innovations would not only enhance the reliability of the storage process but also allow for adaptive control strategies, reducing risk and improving efficiency. Integrating such tools into deployment strategies could be a game-changer for field operations.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Intergovernmental Panel On Climate Change (IPCC). Climate Change 2021—The Physical Science Basis: Working Group I Contribution to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change, 1st ed.; Cambridge University Press: Cambridge, UK, 2023; ISBN 978-1-009-15789-6. [Google Scholar] [CrossRef]
- Pardakhti, M.; Jafari, T.; Tobin, Z.; Dutta, B.; Moharreri, E.; Shemshaki, N.S.; Suib, S.; Srivastava, R. Trends in Solid Adsorbent Materials Development for CO2 Capture. ACS Appl. Mater. Interfaces 2019, 11, 34533–34559. [Google Scholar] [CrossRef]
- Sumida, K.; Rogow, D.L.; Mason, J.A.; McDonald, T.M.; Bloch, E.D.; Herm, Z.R.; Long, J.R. Carbon Dioxide Capture in Metal—Organic Frameworks. Chem. Rev. 2012, 112, 724–781. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Chaowamalee, S.; Pitayachinchot, H.; Kaewngam, C.; Chotirattanachote, A.; Thiensuwan, N.; Poompradub, S.; Yokoi, T.; Ngamcharussrivichai, C. Bifunctionality of Amine-Modified Metal-Organic Frameworks for CO2 Capture and Selective Utilization in Cycloaddition. Carbon Capture Sci. Technol. 2024, 13, 100262. [Google Scholar] [CrossRef]
- Jedli, H.; Almonnef, M.; Rabhi, R.; Mbarek, M.; Abdessalem, J.; Slimi, K. Activated Carbon as an Adsorbent for CO2 Capture: Adsorption, Kinetics, and RSM Modeling. ACS Omega 2024, 9, 2080–2087. [Google Scholar] [CrossRef]
- Adegoke, K.A.; Akpomie, K.G.; Okeke, E.S.; Olisah, C.; Malloum, A.; Maxakato, N.W.; Ighalo, J.O.; Conradie, J.; Ohoro, C.R.; Amaku, J.F.; et al. UiO-66-Based Metal-Organic Frameworks for CO2 Catalytic Conversion, Adsorption and Separation. Sep. Purif. Technol. 2024, 331, 125456. [Google Scholar] [CrossRef]
- Wang, B.; Feng, Y.; Blears, J.; Thompson, K.; Hughes, R. Streamline-Based Simulation of Nanoparticle Transport in Field-Scale Heterogeneous Subsurface Systems. Adv. Water Resour. 2021, 148, 103842. [Google Scholar] [CrossRef]
- Breck, D.W. Zeolite Molecular Sieves: Structure, Chemistry, and Use; John Wiley & Sons: New York, NY, USA, 1974. [Google Scholar] [CrossRef]
- Coasne, B.; Farrusseng, D. Gas Oversolubility in Nanoconfined Liquids: Review and Perspectives for Adsorbent Design. Microporous Mesoporous Mater. 2019, 288, 109561. [Google Scholar] [CrossRef]
- Bahmanzadegan, F.; Ghaemi, A. Modification and Functionalization of Zeolites to Improve the Efficiency of CO2 Adsorption: A Review. Case Stud. Chem. Environ. Eng. 2024, 9, 100564. [Google Scholar] [CrossRef]
- Zhang, Z.; Ding, H.; Pan, W.; Ma, J.; Zhang, K.; Zhao, Y.; Song, J.; Wei, C.; Lin, F. Research Progress of Metal–Organic Frameworks (MOFs) for CO2 Conversion in CCUS. J. Energy Inst. 2023, 108, 101226. [Google Scholar] [CrossRef]
- Kong, M.; Song, L.; Liao, H.; Zhang, S.; Wang, Y.; Deng, X.; Feng, W. A Review on Development of Post-Combustion CO2 Capture Technologies: Performance of Carbon-Based, Zeolites and MOFs Adsorbents. Fuel 2024, 371, 132103. [Google Scholar] [CrossRef]
- Chen, C.; Park, D.-W.; Ahn, W.-S. CO2 Capture Using Zeolite 13X Prepared from Bentonite. Appl. Surf. Sci. 2014, 292, 63–67. [Google Scholar] [CrossRef]
- Jedli, H.; Almoneef, M.M.; Mbarek, M.; Jbara, A.; Slimi, K. Adsorption of CO2 onto Zeolite ZSM-5: Kinetic, Equilibrium and Thermodynamic Studies. Fuel 2022, 321, 124097. [Google Scholar] [CrossRef]
- Panda, D.; Kumar, E.A.; Singh, S.K. Introducing Mesoporosity in Zeolite 4A Bodies for Rapid CO2 Capture. J. CO2 Util. 2020, 40, 101223. [Google Scholar] [CrossRef]
- Usman, M.; Ghanem, A.S.; Niaz Ali Shah, S.; Garba, M.D.; Yusuf Khan, M.; Khan, S.; Humayun, M.; Laeeq Khan, A. A Review on SAPO-34 Zeolite Materials for CO2 Capture and Conversion. Chem. Rec. 2022, 22, e202200039. [Google Scholar] [CrossRef]
- Díaz-García, M.; Mayoral, Á.; Díaz, I.; Sánchez-Sánchez, M. Nanoscaled M-MOF-74 Materials Prepared at Room Temperature. Cryst. Growth Des. 2014, 14, 2479–2487. [Google Scholar] [CrossRef]
- Lin, K.-S.; Adhikari, A.K.; Ku, C.-N.; Chiang, C.-L.; Kuo, H. Synthesis and Characterization of Porous HKUST-1 Metal Organic Frameworks for Hydrogen Storage. Int. J. Hydrog. Energy 2012, 37, 13865–13871. [Google Scholar] [CrossRef]
- Saha, D.; Deng, S. Hydrogen Adsorption on Metal-Organic Framework MOF-177. Tsinghua Sci. Technol. 2010, 15, 363–376. [Google Scholar] [CrossRef]
- Demir, H.; Aksu, G.O.; Gulbalkan, H.C.; Keskin, S. MOF Membranes for CO2 Capture: Past, Present and Future. Carbon Capture Sci. Technol. 2022, 2, 100026. [Google Scholar] [CrossRef]
- Tavagh Mohammadi, B.; Jahanbani Ghahfarokhi, A.; Grimstad, A.-A. A Review of Modeling Approaches for CO2 Injection into Depleted Gas Reservoirs: Coupling Transient Wellbore and Reservoir Dynamics. Int. J. Greenh. Gas Control 2025, 145, 104406. [Google Scholar] [CrossRef]
- Zhao, G.; Li, Z.; Cheng, B.; Zhuang, X.; Lin, T. Hierarchical Porous Metal Organic Framework Aerogel for Highly Efficient CO2 Adsorption. Sep. Purif. Technol. 2023, 315, 123754. [Google Scholar] [CrossRef]
- Bello, A.; Dorhjie, D.B.; Ivanova, A.; Cheremisin, A. A Numerical Feasibility Study of CO2 Foam for Carbon Utilization and Storage in a Depleted, High Salinity, Carbonate Oil Reservoir. Sci. Rep. 2024, 14, 20585. [Google Scholar] [CrossRef] [PubMed]
- Neubertová, V.; Švorčík, V.; Kolská, Z. Amino-Modified ZIF-8 for Enhanced CO2 Capture: Synthesis, Characterization and Performance Evaluation. Microporous Mesoporous Mater. 2024, 366, 112956. [Google Scholar] [CrossRef]
- Salunkhe, B.; Schuman, T.; Al Brahim, A.; Bai, B. Ultra-High Temperature Resistant Preformed Particle Gels for Enhanced Oil Recovery. Chem. Eng. J. 2021, 426, 130712. [Google Scholar] [CrossRef]
- Choi, H.L.; Jeong, Y.; Lee, H.; Bae, T.-H. High-Performance Mixed-Matrix Membranes Using a Zeolite@MOF Core–Shell Structure Synthesized via Ion-Exchange-Induced Crystallization and Post-Synthetic Conversion. JACS Au 2024, 4, 253–262. [Google Scholar] [CrossRef]
- Ullah, S.; Rehman, A.U.; Najam, T.; Hossain, I.; Anjum, S.; Ali, R.; Shahid, M.U.; Shah, S.S.A.; Nazir, M.A. Advances in Metal-Organic Framework@activated Carbon (MOF@AC) Composite Materials: Synthesis, Characteristics and Applications. J. Ind. Eng. Chem. 2024, 137, 87–105. [Google Scholar] [CrossRef]
- Park, H.; Yan, X.; Zhu, R.; Huerta, E.A.; Chaudhuri, S.; Cooper, D.; Foster, I.; Tajkhorshid, E. A Generative Artificial Intelligence Framework Based on a Molecular Diffusion Model for the Design of Metal-Organic Frameworks for Carbon Capture. Commun. Chem. 2024, 7, 21. [Google Scholar] [CrossRef]
- Dalla Longa, F.; Detz, R.; Van Der Zwaan, B. Integrated Assessment Projections for the Impact of Innovation on CCS Deployment in Europe. Int. J. Greenh. Gas Control 2020, 103, 103133. [Google Scholar] [CrossRef]
- Silveira, T.; Awano, C.M.; Donatti, D.A.; Vicente, F.S.; Vollet, D.R. About the thermal stability and pore elimination in the ordered hexagonal mesoporous silica SBA-15. Microporous Mesoporous Mater. 2014, 190, 227–233. [Google Scholar] [CrossRef]
- Ioannidou, O.; Zabaniotou, A. Agricultural residues as precursors for activated carbon production—A review. Renew. Sustain. Energy Rev. 2007, 11, 1966–2005. [Google Scholar] [CrossRef]
- Karan, H.; Roles, J.; Ross, I.L.; Ebrahimi, M.; Rackemann, D.; Rainey, T.; Hankamer, B. Solar biorefinery concept for sustainable co-production of microalgae-based protein and renewable fuel. J. Clean. Prod. 2022, 368, 132981. [Google Scholar] [CrossRef]
- Zhang, Q.; Yan, S.; Yan, X.; Lv, Y. Recent advances in metal-organic frameworks: Synthesis, application and toxicity. Sci. Total Environ. 2023, 902, 165944. [Google Scholar] [CrossRef] [PubMed]
- Pan, S.Y.; Chen, Y.H.; Fan, L.S.; Kim, H.; Gao, X.; Ling, T.C.; Chiang, P.C.; Pei, S.; Gu, G. CO2 mineralization and utilization by alkaline solid wastes for potential carbon reduction. Nat Sustain 2020, 3, 399–405. [Google Scholar] [CrossRef]
- Zhang, K.; Nalaparaju, A.; Jiang, J. CO2 Capture in Rht Metal–Organic Frameworks: Multiscale Modeling from Molecular Simulation to Breakthrough Prediction. J. Mater. Chem. A 2015, 3, 16327–16336. [Google Scholar] [CrossRef]
- Indira, V.; Abhitha, K. A Review on Polymer Based Adsorbents for CO2 Capture. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2021; Volume 1114, p. 012081. [Google Scholar] [CrossRef]
- Gajda, A.; Pajdak, A.; Skoczylas, N.; Kudasik, M.; Kozieł, K.; Kurowski, G.; Hyjek, K.; Dymek, K.; Sitarz, M.; Jodłowski, P. Effective Heat Transfer Associated with CO2 and CH4 Adsorption on HKUST-1 Metal-Organic Framework Deposited in Situ on Heat Exchanger. Energy 2024, 307, 132525. [Google Scholar] [CrossRef]
- Kim, J.S.; Lee, S.; Kim, T.; Oh, K.H.; Choi, Y.; Byun, H.; Lee, C.-H.; Ravi, S.; Bae, Y.-S. Enhanced Hydrogen Storage via Microporous Defects and Cu(I) Sites in HKUST-1. Int. J. Hydrog. Energy 2024, 90, 941–949. [Google Scholar] [CrossRef]


| Property | Unit | Zeolites | MOFs | Activated Carbons | Normalization Notes | References |
|---|---|---|---|---|---|---|
| Surface Area | m2/g | 300–800 | 1000–7000 | 500–2500 | Normalized from 300 (1) to 7000 (5) | [3,5,13] |
| Moisture Resistance | Qualitative | Low to Moderate | Variable (depends on MOF type) | High | Assigned ordinal scale (1–5) based on range | [3,12,14] |
| Regeneration Ease | Qualitative | High (via TSA/PSA) | Moderate to High (depends on MOF) | High | Based on regeneration temperature/load | [3,11,12] |
| Cost (Inverse) | USD/kg | ~2–10 | ~100–500 | ~1–5 | Inverted scale: higher cost = lower score | [2,12,14] |
| CO2 Adsorption Cap. | mmol/g | 3.5–5.0 | 5.0–8.0 | 3.0–5.0 | Normalized from 3 (1) to 8 (5) | [2,5,11] |
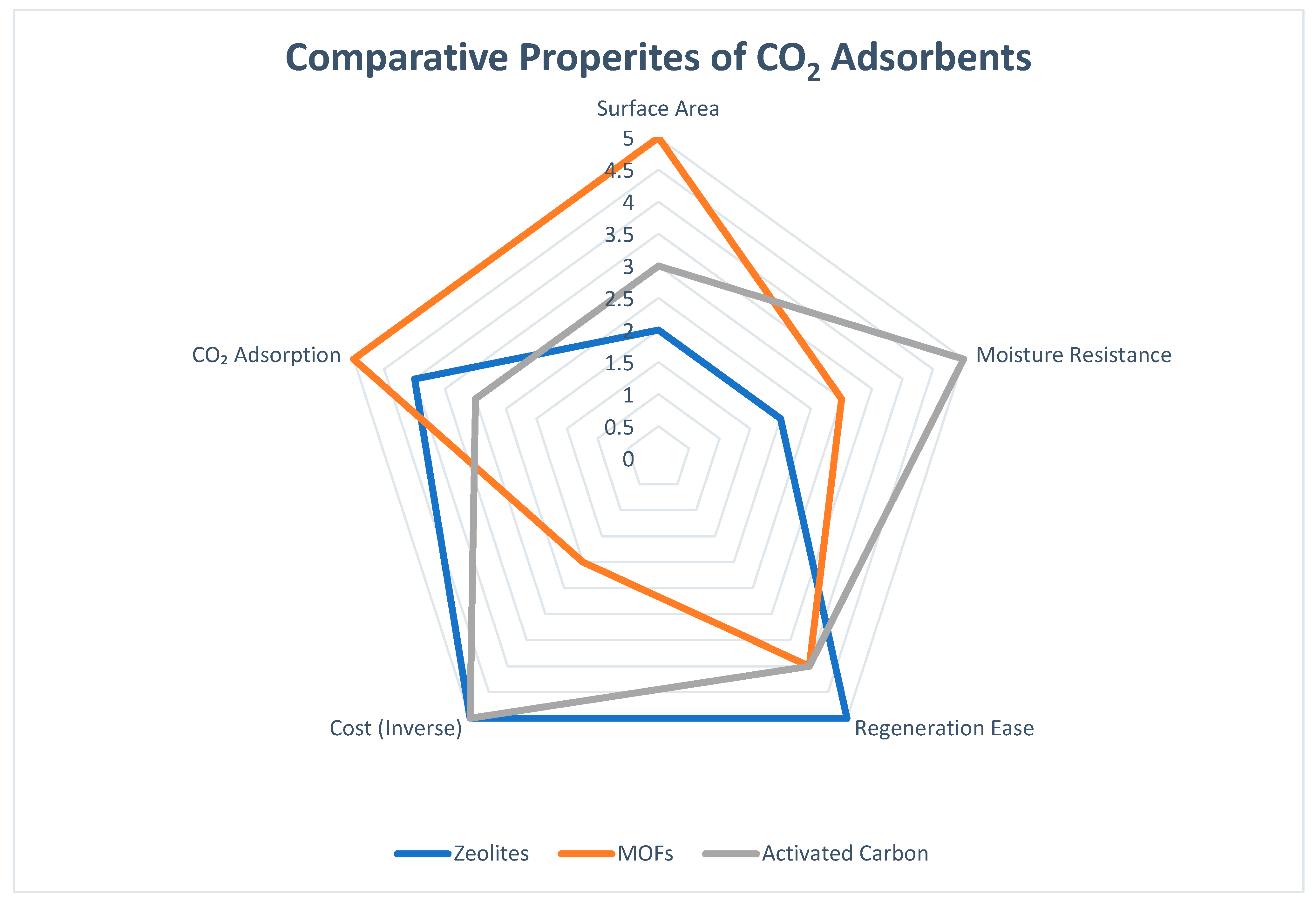
| Normalized Scores (Used in Radar Chart Figure 1) | ||||||
| Material | Surface Area | Moisture Resistance | Regeneration Ease | Cost (Inverse) | CO2 Adsorption | |
| Zeolites | 2 | 2 | 5 | 5 | 4 | |
| MOFs | 5 | 3 | 4 | 2 | 5 | |
| Activated Carbon | 3 | 5 | 4 | 5 | 3 | |
| Parameter | Zeolite 13X | Zeolite 4A (Na-A) | ZSM-5 (MFI-Type) | Zeolite Y | SAPO-34 |
|---|---|---|---|---|---|
| BET Surface Area (m2/g) | ~600–700 | ~400–500 | ~350–450 | ~600–800 | ~550–650 |
| CO2 Adsorption Capacity (mmol/g) | ~4.5–5.0 | ~3.0–3.5 | ~2.5–3.5 | ~4.0–4.5 | ~3.5–4.0 |
| Regenerability | Very High | Very High | Very High | Very High | High |
| Stability in Humid Conditions | Excellent | Excellent | Good | Excellent | Moderate |
| Thermal Stability | Up to ~700 °C | ~500–600 °C | ~800 °C | ~700 °C | ~600 °C |
| Chemical Stability | Excellent (alkaline-sensitive) | Excellent | Excellent | Moderate-to-Good | Moderate |
| Regeneration Energy (kJ/mol CO2) | ~20–30 | ~25–30 | ~30–40 | ~25–35 | ~25–35 |
| Regeneration Method | TSA, PSA, VSA | TSA, PSA, VSA | TSA, PSA, VSA | TSA, PSA | TSA, PSA |
| Production Cost | Low | Very Low | Low to Moderate | Moderate | Moderate |
| Scalability | High | High | High | High | Moderate |
| Industrial Viability | Excellent | Excellent | Excellent | High | Good |
| References | [13] | [15] | [14] | [10] | [16] |
| Parameter | MOF-74 | MOF-199 (HKUST-1) | MOF-177 | UiO-66 |
|---|---|---|---|---|
| BET Surface Area (m2/g) | 1100–1500 | 1400–1800 | 4500–5100 | 1000–1500 |
| CO2 Adsorption Capacity (mmol/g) | Up to 8.0 | ~5.0–6.0 | ~6.0–7.5 | ~2.5–4.0 |
| Regenerability | High | High | High (dry only) | Very High |
| Stability in Humid Conditions | Moderate to Poor | Poor | Poor | Excellent |
| Thermal Stability (°C) | ~300–400 | ~240–300 | ~350 | ~500–550 |
| Chemical Stability | Moderate | Limited | Limited | Very High |
| Regeneration Energy (kJ/mol CO2) | ~30–40 | ~25–35 | ~30–40 | ~20–30 |
| Regeneration Method | TSA, PSA, VSA | TSA, PSA, VSA | TSA, PSA, VSA | TSA, PSA, VSA |
| Production Cost (USD /kg) | Moderate–High | Moderate | Moderate–High | Moderate |
| Scalability | Moderate | High | Limited | High |
| Industrial Viability | Promising (dry gases) | Promising (dry gases) | Limited | Excellent |
| References | [17] | [18] | [19] | [6] |
| Property/Feature | Zeolites | MOFs | Activated Carbons |
|---|---|---|---|
| Structure | Crystalline aluminosilicates | Crystalline MOFs | Amorphous carbon structures |
| Surface Area (m2/g) | 300–800 | Up to 7000 | 500–2500 |
| Thermal Stability | High (>700 °C) | Moderate to High | Very High (>900 °C) |
| Hydrothermal Stability | Good | Often Limited | Excellent |
| Moisture Sensitivity | High | Varies | Low |
| Regenerability | Easy | Moderate | Easy |
| Cost | Low to Moderate | High | Low |
| Pore Size Range (nm) | 0.3–1.2 | 0.3–3+ | 0.5–50 |
| Hydrogen Storage | Low to Moderate | High | Moderate |
| Methane Storage | Moderate | High | High |
| CO2 Selectivity | High | Very High | Moderate |
| CO2/N2 Selectivity | High | Very High | Moderate |
| Regeneration Ease | High | Moderate | High |
| Scalability | High | Moderate | Very High |
| Reference | [12] | [12] | [12] |
| Delivery Method | Compatible Material(s) | Reservoir Compatibility | Advantages | Challenges | Technology Readiness Level (TRL) |
|---|---|---|---|---|---|
| Direct Suspension Injection | Zeolites, Activated Carbons | High-permeability sandstone, depleted reservoirs | Simple implementation, compatible with existing infrastructure | Particle settling, clogging risks | 7–9 |
| Polymer-Assisted Transport | MOFs, Zeolites | Low-permeability formations, heterogeneous zones | Improves sweep efficiency, protects sensitive materials | Polymer retention, formation damage | 5–7 |
| Foam-Assisted Delivery | Zeolites, MOFs | Fractured formations, high-permeability layers | Mobility control, deep penetration | Foam stability under salinity/temperature | 5–6 |
| Encapsulation with Controlled Release | MOFs | Saline aquifers, variable pH zones | Targeted release, material protection | High complexity, cost | 4–5 |
| Preformed Particle Gels (PPGs) | MOFs, Zeolites | Fractured or bypassed zones | Dual function: flow diversion and adsorption | Swelling risks, limited reusability | 4–6 |
| Parameter | MOFs (Conventional) | MOFs (Green Synthesis) | Activated Carbon | Zeolites (Synthetic/Natural) |
|---|---|---|---|---|
| Energy Input (MJ/kg) | 200–400 MJ/kg [34] | 80–150 MJ/kg [32] | 30–70 MJ/kg [31] | 80–150 MJ/kg [30] |
| GHG Emissions (kg CO2-eq/kg) | 10–30 kg CO2-eq/kg [34] | 4–12 kg CO2-eq/kg [32] | 2–5 kg CO2-eq/kg [31,35] | 4–7 kg CO2-eq/kg [30,35] |
| Solvent Use | High (e.g., DMF, ethanol) [34] | Low (e.g., water, mechanochemical) [32] | Low [31] | Low to Moderate [30,36] |
| Circularity Potential | Low–Moderate [34] | Moderate [32] | High [31,36] | Moderate–High [30] |
| End-of-Life Management | Limited recyclability [34] | Emerging (e.g., bio-MOFs, recyclable linkers) [32] | Mature (reusable, degradable) [31,36] | High reusability and mineral stability [30,36] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mouctar, M.H.; Hassan, M.G.; Bimbo, N.; Abbas, S.Z.; Shigidi, I. Comparative Assessment and Deployment of Zeolites, MOFs, and Activated Carbons for CO2 Capture and Geological Sequestration Applications. Inventions 2025, 10, 78. https://doi.org/10.3390/inventions10050078
Mouctar MH, Hassan MG, Bimbo N, Abbas SZ, Shigidi I. Comparative Assessment and Deployment of Zeolites, MOFs, and Activated Carbons for CO2 Capture and Geological Sequestration Applications. Inventions. 2025; 10(5):78. https://doi.org/10.3390/inventions10050078
Chicago/Turabian StyleMouctar, Mohamadou Hamadama, Mohamed G Hassan, Nuno Bimbo, Syed Zaheer Abbas, and Ihab Shigidi. 2025. "Comparative Assessment and Deployment of Zeolites, MOFs, and Activated Carbons for CO2 Capture and Geological Sequestration Applications" Inventions 10, no. 5: 78. https://doi.org/10.3390/inventions10050078
APA StyleMouctar, M. H., Hassan, M. G., Bimbo, N., Abbas, S. Z., & Shigidi, I. (2025). Comparative Assessment and Deployment of Zeolites, MOFs, and Activated Carbons for CO2 Capture and Geological Sequestration Applications. Inventions, 10(5), 78. https://doi.org/10.3390/inventions10050078









