Management of Persistent Erectile Dysfunction after COVID-19 Infection: An 18-Month Follow-Up Case Report
Abstract
1. Introduction
2. Patients and Methods
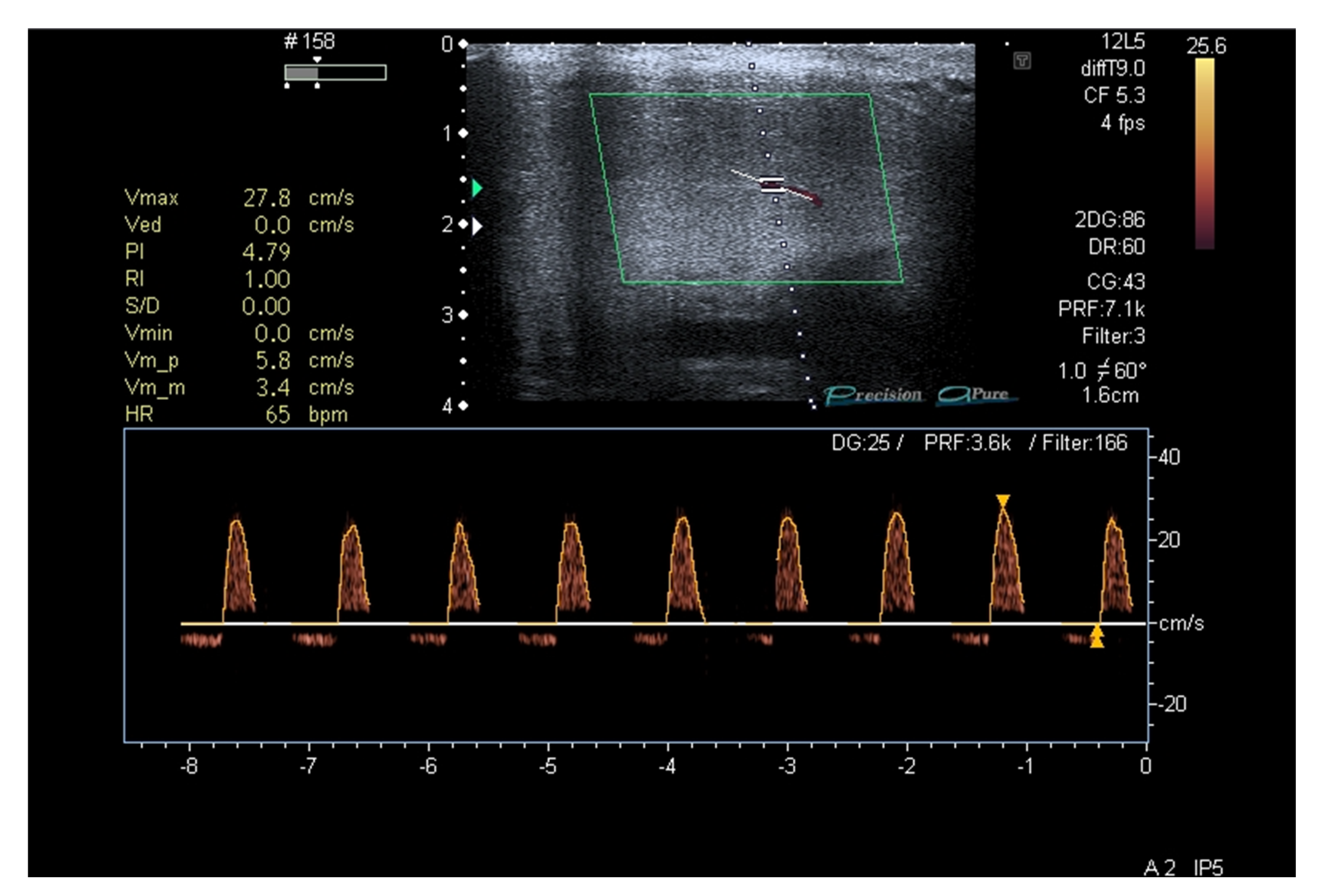
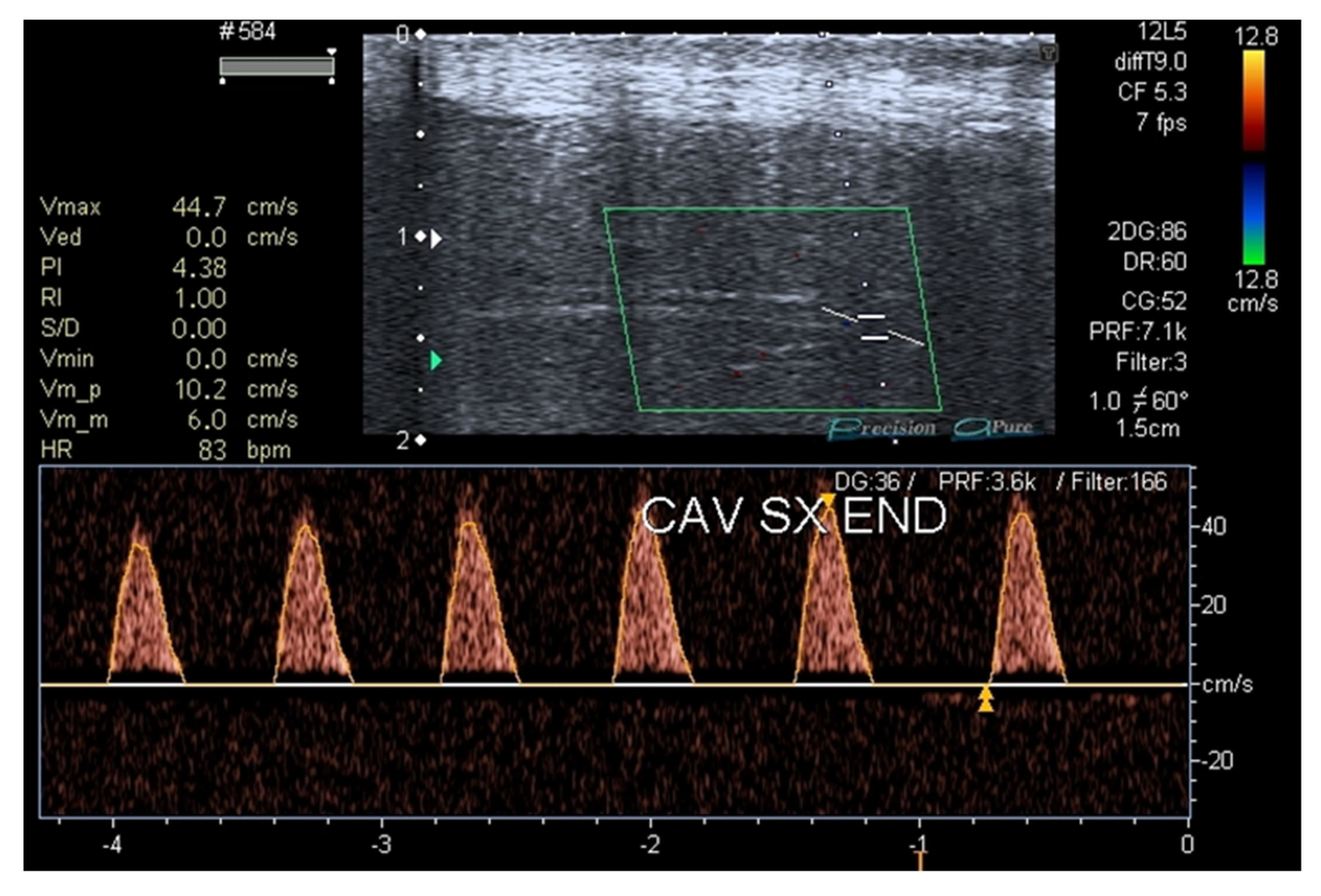
3. Results
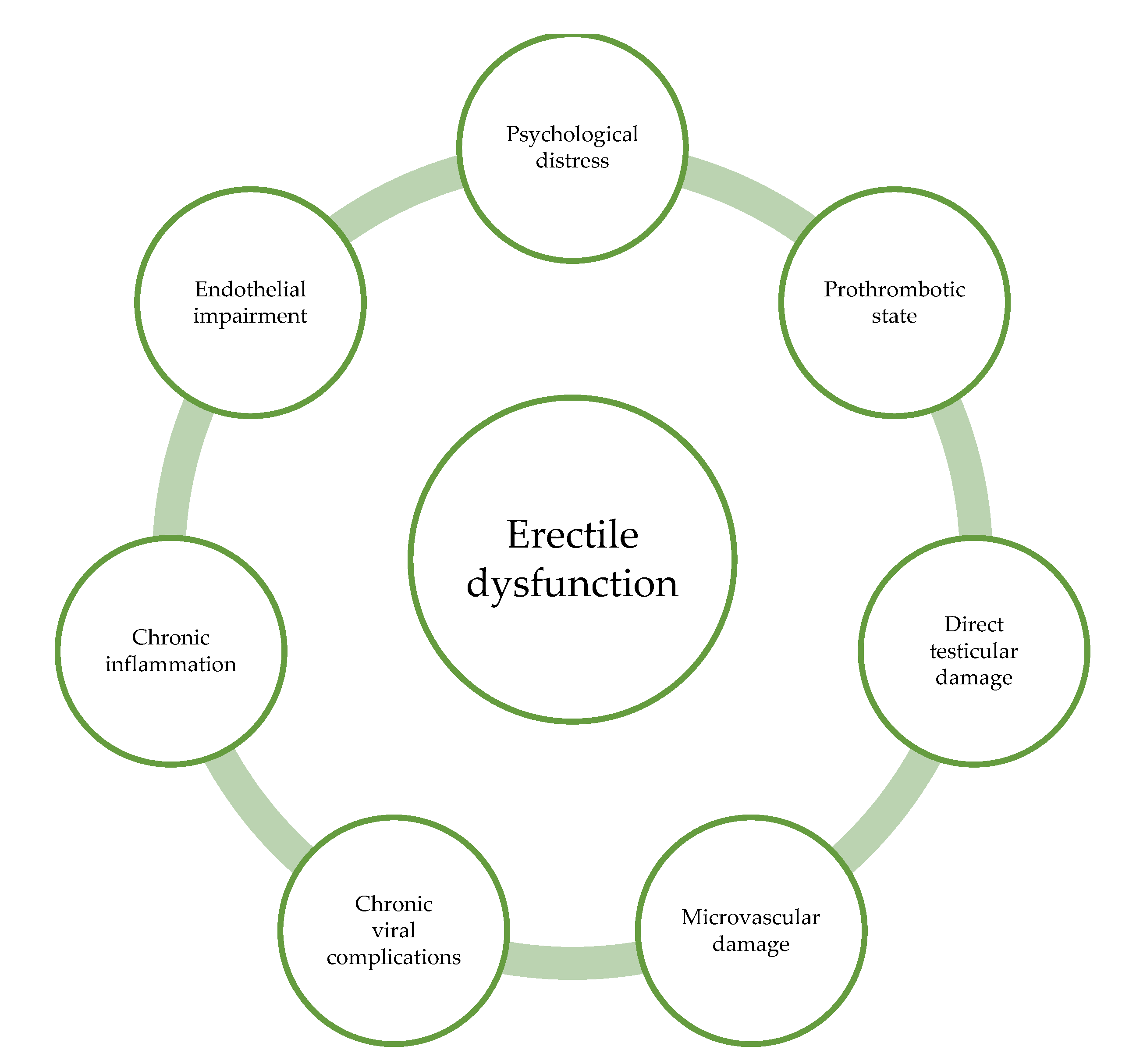
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wiersinga, W.J.; Rhodes, A.; Cheng, A.C.; Peacock, S.J.; Prescott, H.C. Pathophysiology, Transmission, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19): A Review. JAMA 2020, 324, 782–793. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 507–513. [Google Scholar] [CrossRef] [PubMed]
- Bernard, I.; Limonta, D.; Mahal, L.K.; Hobman, T.C. Endothelium Infection and Dysregulation by SARS-CoV-2: Evidence and Caveats in COVID-19. Viruses 2020, 13, 29. [Google Scholar] [CrossRef] [PubMed]
- Harrison, A.G.; Lin, T.; Wang, P. Mechanisms of SARS-CoV-2 Transmission and Pathogenesis. Trends Immunol. 2020, 41, 1100–1115. [Google Scholar] [CrossRef]
- La Vignera, S.; Cannarella, R.; Condorelli, R.A.; Torre, F.; Aversa, A.; Calogero, A.E. Sex-Specific SARS-CoV-2 Mortality: Among Hormone-Modulated ACE2 Expression, Risk of Venous Thromboembolism and Hypovitaminosis D. Int. J. Mol. Sci. 2020, 21, 2948. [Google Scholar] [CrossRef]
- Oran, D.P.; Topol, E.J. Prevalence of Asymptomatic SARS-CoV-2 Infection: A Narrative Review. Ann. Intern. Med. 2020, 173, 362–367. [Google Scholar] [CrossRef]
- Crocerossa, F.; Visser, W.; Carbonara, U.; Falagario, U.G.; Pandolfo, S.D.; Loizzo, D.; Imbimbo, C.; Klausner, A.P.; Porpiglia, F.; Damiano, R.; et al. The impact the COVID-19 pandemic on urology literature: A bibliometric analysis. Cent. Eur. J. Urol. 2022, 75, 102–109. [Google Scholar] [CrossRef]
- Sansone, A.; Mollaioli, D.; Ciocca, G.; Limoncin, E.; Colonnello, E.; Vena, W.; Jannini, E.A. Addressing male sexual and reproductive health in the wake of COVID-19 outbreak. J. Endocrinol. Invest. 2021, 44, 223–231. [Google Scholar] [CrossRef]
- Hu, B.; Ruan, Y.; Liu, K.; Wei, X.; Wu, Y.; Feng, H.; Deng, Z.; Liu, J.; Wang, T. A Mid-to-Long Term Comprehensive Evaluation of Psychological Distress and Erectile Function in COVID-19 Recovered Patients. J. Sex Med. 2021, 18, 1863–1871. [Google Scholar] [CrossRef]
- Vaccaro, M.G.; Izzo, G.; Sarica, A.; La Vignera, S.; Aversa, A. Cluster Analysis Method Reveals Gender Attitudes in Sociosexual Orientation of a Southern Italy Population During the COVID-19 Lockdown. Sex Res. Social Policy 2022, 24, 1–14. [Google Scholar] [CrossRef]
- Salonia, A.; Bettocchi, C.; Boeri, L.; Capogrosso, P.; Carvalho, J.; Cilesiz, N.C.; Cocci, A.; Corona, G.; Dimitropoulos, K.; Gül, M.; et al. European Association of Urology Guidelines on Sexual and Reproductive Health-2021 Update: Male Sexual Dysfunction. Eur. Urol. 2021, 80, 333–357. [Google Scholar] [CrossRef]
- Yafi, F.A.; Jenkins, L.; Albersen, M.; Corona, G.; Isidori, A.M.; Goldfarb, S.; Maggi, M.; Nelson, C.J.; Parish, S.; Salonia, A.; et al. Erectile dysfunction. Nat. Rev. Dis. Prim. 2016, 2, 16003. [Google Scholar] [CrossRef]
- Sansone, A.; Mollaioli, D.; Ciocca, G.; Colonnello, E.; Limoncin, E.; Balercia, G.; Jannini, E.A. “Mask up to keep it up”: Preliminary evidence of the association between erectile dysfunction and COVID-19. Andrology 2021, 9, 1053–1059. [Google Scholar] [CrossRef]
- Kresch, E.; Achua, J.; Saltzman, R.; Khodamoradi, K.; Arora, H.; Ibrahim, E.; Kryvenko, O.N.; Almeida, V.W.; Firdaus, F.; Hare, J.M.; et al. COVID-19 Endothelial Dysfunction Can Cause Erectile Dysfunction: Histopathological, Immunohistochemical, and Ultrastructural Study of the Human Penis. World J. Mens Health 2021, 39, 466–469. [Google Scholar] [CrossRef]
- Kaynar, M.; Gomes, A.L.Q.; Sokolakis, I.; Gül, M. Tip of the iceberg: Erectile dysfunction and COVID-19. Int. J. Impot. Res. 2022, 34, 152–157. [Google Scholar] [CrossRef]
- Pizzol, D.; Shin, J.I.; Trott, M.; Ilie, P.C.; Ippoliti, S.; Carrie, A.M.; Ghayda, R.A.; Lozano, J.M.O.; Muyor, J.M.; Butler, L.; et al. Social environmental impact of COVID-19 and erectile dysfunction: An explorative review. J. Endocrinol. Invest. 2022, 45, 483–487. [Google Scholar] [CrossRef]
- Adeyemi, D.H.; Odetayo, A.F.; Hamed, M.A.; Akhigbe, R.E. Impact of COVID 19 on erectile function. Aging Male 2022, 25, 202–216. [Google Scholar] [CrossRef]
- Jannini, E.A.; McCabe, M.P.; Salonia, A.; Montorsi, F.; Sachs, B.D. Organic vs. psychogenic? The Manichean diagnosis in sexual medicine. J. Sex. Med. 2010, 7, 1726–1733. [Google Scholar] [CrossRef]
- De Leonardis, F.; Colalillo, G.; Finazzi Agrò, E.; Miano, R.; Fuschi, A.; Asimakopoulos, A.D. Endothelial Dysfunction, Erectile Deficit and Cardiovascular Disease: An Overview of the Pathogenetic Links. Biomedicines 2022, 10, 1848. [Google Scholar] [CrossRef]
- Barthelmes, J.; Nägele, M.P.; Ludovici, V.; Ruschitzka, F.; Sudano, I.; Flammer, A.J. Endothelial dysfunction in cardiovascular disease and Flammer syndrome—Similarities and differences. EPMA J. 2017, 8, 99–109. [Google Scholar] [CrossRef]
- Neumann, P.; Gertzberg, N.; Johnson, A. TNF-alpha induces a decrease in eNOS promoter activity. Am. J. Physiol. Lung Cell. Mol. Physiol. 2004, 286, L452–L459. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Xie, W.; Li, D.; Shi, L.; Mao, Y.; Xiong, Y.; Zhang, Y.; Zhang, M. Effect of SARS-CoV-2 infection upon male gonadal function: A single center-based study. medRxiv 2020. [Google Scholar] [CrossRef]
- Okçelik, S. COVID-19 pneumonia causes lower testosterone levels. Andrologia 2021, 53, e13909. [Google Scholar] [CrossRef] [PubMed]
- Magadum, A.; Kishore, R. Cardiovascular Manifestations of COVID-19 Infection. Cells 2020, 9, 2508. [Google Scholar] [CrossRef] [PubMed]
- Graney, B.A.; Wamboldt, F.S.; Baird, S.; Churney, T.; Fier, K.; Korn, M.; McCormick, M.; Vierzba, T.; Swigris, J.J. Looking ahead and behind at supplemental oxygen: A qualitative study of patients with pulmonary fibrosis. Heart Lung 2017, 46, 387–393. [Google Scholar] [CrossRef]
- Sansone, A.; Mollaioli, D.; Limoncin, E.; Ciocca, G.; Bắc, N.H.; Cao, T.N.; Hou, G.; Yuan, J.; Zitzmann, M.; Giraldi, A.; et al. The Sexual Long COVID (SLC): Erectile Dysfunction as a Biomarker of Systemic Complications for COVID-19 Long Haulers. Sex Med. Rev. 2022, 10, 271–285. [Google Scholar] [CrossRef]
- Romano, L.; Pellegrino, R.; Sciorio, C.; Barone, B.; Gravina, A.G.; Santonastaso, A.; Mucherino, C.; Astretto, S.; Napolitano, L.; Aveta, A.; et al. Erectile and sexual dysfunction in male and female patients with celiac disease: A cross-sectional observational study. Andrology 2022, 10, 910–918. [Google Scholar] [CrossRef]
- Apolone, G.; Mosconi, P. The Italian SF-36 Health Survey: Translation, validation and norming. J. Clin. Epidemiol. 1998, 51, 1025–1036. [Google Scholar] [CrossRef]
- Cappelleri, J.C.; Rosen, R.C.; Smith, M.D.; Mishra, A.; Osterloh, I.H. Diagnostic evaluation of the erectile function domain of the International Index of Erectile Function. Urology 1999, 54, 346–351. [Google Scholar] [CrossRef]
- Rosen, R.C.; Cappelleri, J.C.; Smith, M.D.; Lipsky, J.; Peña, B.M. Development and evaluation of an abridged, 5-item version of the International Index of Erectile Function (IIEF-5) as a diagnostic tool for erectile dysfunction. Int. J. Impot. Res. 1999, 11, 319–326. [Google Scholar] [CrossRef]
- Steidle, C.P.; McCullough, A.R.; Kaminetsky, J.C.; Crowley, A.R.; Siegel, R.L.; Deriesthal, H.; Tseng, L.J. Early sildenafil dose optimization and personalized instruction improves the frequency, flexibility, and success of sexual intercourse in men with erectile dysfunction. Int. J. Impot. Res. 2007, 19, 154–160. [Google Scholar] [CrossRef]
- Aversa, A.; Crafa, A.; Alessandra Greco, E.; Chiefari, E.; Brunetti, A.; La Vignera, S. The Penile Duplex Ultrasound: How and when to perform it? Andrology 2021, 9, 1457–1466. [Google Scholar] [CrossRef]
- Vena, W.; Vaccalluzzo, L.; Morenghi, E.; D’Agostino, C.; Perri, A.; Giammusso, B.; Lania, A.G.; Aversa, A.; Pizzocaro, A. Low-intensity shockwave treatment (liswt) improves penile rigidity in eugonadal subjects with erectile dysfunction: A pilot study. Minerva Endocrinol. 2021. [Google Scholar] [CrossRef]
- Groff, D.; Sun, A.; Ssentongo, A.E.; Ba, D.M.; Parsons, N.; Poudel, G.R.; Lekoubou, A.; Oh, J.S.; Ericson, J.E.; Ssentongo, P.; et al. Short-term and Long-term Rates of Postacute Sequelae of SARS-CoV-2 Infection: A Systematic Review. JAMA Netw. Open 2021, 4, e2128568. [Google Scholar] [CrossRef]
- Moncada, I.; Martinez-Salamanca, J.; Ruiz-Castañe, E.; Romero, J. Combination therapy for erectile dysfunction involving a PDE5 inhibitor and alprostadil. Int. J. Impot. Res. 2018, 30, 203–208. [Google Scholar] [CrossRef]
- McMahon, C.N.; Smith, C.J.; Shabsigh, R. Treating erectile dysfunction when PDE5 inhibitors fail. BMJ Clin. Res. Ed. 2006, 332, 589–592. [Google Scholar] [CrossRef]
- Herrero, A.; Marcos, M.; Galindo, P.; Miralles, J.M.; Corrales, J.J. Clinical and biochemical correlates of male hypogonadism in type 2 diabetes. Andrology 2018, 6, 58–63. [Google Scholar] [CrossRef]
- Aversa, A.; Basciani, S.; Visca, P.; Arizzi, M.; Gnessi, L.; Frajese, G.; Fabbri, A. Platelet-derived growth factor (PDGF) and PDGF receptors in rat corpus cavernosum: Changes in expression after transient in vivo hypoxia. J. Endocrinol. 2001, 170, 395–402. [Google Scholar] [CrossRef]
- Hellstrom, W.J.; Elhilali, M.; Homering, M.; Taylor, T.; Gittleman, M. Vardenafil in patients with erectile dysfunction: Achieving treatment optimization. J. Androl. 2005, 26, 604–609. [Google Scholar] [CrossRef]
- Albersen, M.; Mwamukonda, K.B.; Shindel, A.W.; Lue, T.F. Evaluation and treatment of erectile dysfunction. Med. Clin. North Am. 2011, 95, 201–212. [Google Scholar] [CrossRef]
- Hackett, G.; Kirby, M.; Wylie, K.; Heald, A.; Ossei-Gerning, N.; Edwards, D.; Muneer, A. British Society for Sexual Medicine Guidelines on the Management of Erectile Dysfunction in Men-2017. J. Sex Med. 2018, 15, 430–457. [Google Scholar] [CrossRef] [PubMed]
- Eardley, I.; Cartledge, J. Tadalafil (Cialis) for men with erectile dysfunction. Int. J. Clin. Pract. 2002, 56, 300–304. [Google Scholar] [PubMed]
- Porst, H.; Padma-Nathan, H.; Giuliano, F.; Anglin, G.; Varanese, L.; Rosen, R. Efficacy of tadalafil for the treatment of erectile dysfunction at 24 and 36 hours after dosing: A randomized controlled trial. Urology 2003, 62, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Li, H.J.; Bai, W.J.; Dai, Y.T.; Xu, W.P.; Wang, C.N.; Li, H.Z. An analysis of treatment preferences and sexual quality of life outcomes in female partners of Chinese men with erectile dysfunction. Asian J. Androl. 2016, 18, 773–779. [Google Scholar] [CrossRef]
- McMahon, C. Efficacy and safety of daily tadalafil in men with erectile dysfunction previously unresponsive to on-demand tadalafil. J. Sex. Med. 2004, 1, 292–300. [Google Scholar] [CrossRef]
- Albersen, M.; Joniau, S.; Claes, H.; Van Poppel, H. Preclinical Evidence for the Benefits of Penile Rehabilitation Therapy following Nerve-Sparing Radical Prostatectomy. Adv. Urol. 2008, 2008, 594868. [Google Scholar] [CrossRef]
- Aversa, A.; Greco, E.; Bruzziches, R.; Pili, M.; Rosano, G.; Spera, G. Relationship between chronic tadalafil administration and improvement of endothelial function in men with erectile dysfunction: A pilot study. Int. J. Impot. Res. 2007, 19, 200–207. [Google Scholar] [CrossRef]
- Frairia, R.; Berta, L. Biological effects of extracorporeal shock waves on fibroblasts. A review. Muscles Ligaments Tendons J. 2011, 1, 138–147. [Google Scholar]
- Pons, S.; Fodil, S.; Azoulay, E.; Zafrani, L. The vascular endothelium: The cornerstone of organ dysfunction in severe SARS-CoV-2 infection. Crit. Care 2020, 24, 353. [Google Scholar] [CrossRef]
- Smilowitz, N.R.; Kunichoff, D.; Garshick, M.; Shah, B.; Pillinger, M.; Hochman, J.S.; Berger, J.S. C-reactive protein and clinical outcomes in patients with COVID-19. Eur. Heart J. 2021, 42, 2270–2279. [Google Scholar] [CrossRef]
- Willems, L.H.; Nagy, M.; Ten Cate, H.; Spronk, H.M.H.; Groh, L.A.; Leentjens, J.; Janssen, N.A.F.; Netea, M.G.; Thijssen, D.H.J.; Hannink, G.; et al. Sustained inflammation, coagulation activation and elevated endothelin-1 levels without macrovascular dysfunction at 3 months after COVID-19. Thromb. Res. 2022, 209, 106–114. [Google Scholar] [CrossRef]
- Perri, A.; Bossio, S.; Rago, V.; Greco, E.A.; Lofaro, D.; Aversa, A. NLRP3-inflammasome activation in male reproductive system diseases. Minerva Endocrinol. 2022, 14, 5323. [Google Scholar] [CrossRef]
- Bikdeli, B.; Madhavan, M.V.; Jimenez, D.; Chuich, T.; Dreyfus, I.; Driggin, E.; Nigoghossian, C.; Ageno, W.; Madjid, M.; Guo, Y.; et al. COVID-19 and Thrombotic or Thromboembolic Disease: Implications for Prevention, Antithrombotic Therapy, and Follow-Up: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2020, 75, 2950–2973. [Google Scholar] [CrossRef]
- Harirugsakul, K.; Wainipitapong, S.; Phannajit, J.; Paitoonpong, L.; Tantiwongse, K. Erectile dysfunction after COVID-19 recovery: A follow-up study. PLoS ONE 2022, 17, e0276429. [Google Scholar] [CrossRef]
- Ottaviano, G.; Zuccarello, D.; Frasson, G.; Scarpa, B.; Nardello, E.; Foresta, C.; Marioni, G.; Staffieri, A. Olfactory sensitivity and sexual desire in young adult and elderly men: An introductory investigation. Am. J. Rhinol. Allergy 2013, 27, 157–161. [Google Scholar] [CrossRef]
- Siegel, J.K.; Kung, S.Y.; Wroblewski, K.E.; Kern, D.W.; McClintock, M.K.; Pinto, J.M. Olfaction Is Associated With Sexual Motivation and Satisfaction in Older Men and Women. J. Sex. Med. 2021, 18, 295–302. [Google Scholar] [CrossRef]
- Bertolo, R.; Cipriani, C.; Bove, P. Anosmia and ageusia: A piece of the puzzle in the etiology of COVID-19-related transitory erectile dysfunction. J. Endocrinol. Invest. 2021, 44, 1123–1124. [Google Scholar] [CrossRef]
- Crafa, A.; Cannarella, R.; Condorelli, R.A.; La Vignera, S.; Calogero, A.E. Is There an Association Between Vitamin D Deficiency and Erectile Dysfunction? A Systematic Review and Meta-Analysis. Nutrients 2020, 12, 1411. [Google Scholar] [CrossRef]
- Crafa, A.; Cannarella, R.; Condorelli, R.A.; Mongioì, L.M.; Barbagallo, F.; Aversa, A.; La Vignera, S.; Calogero, A.E. Influence of 25-hydroxy-cholecalciferol levels on SARS-CoV-2 infection and COVID-19 severity: A systematic review and meta-analysis. Eclinical Med. 2021, 37, 100967. [Google Scholar] [CrossRef]
- Mirhosseini, N.; Rainsbury, J.; Kimball, S.M. Vitamin D Supplementation, Serum 25(OH)D Concentrations and Cardiovascular Disease Risk Factors: A Systematic Review and Meta-Analysis. Front. Cardiovasc. Med. 2018, 5, 87. [Google Scholar] [CrossRef]
- Jamali, N.; Sorenson, C.M.; Sheibani, N. Vitamin D and regulation of vascular cell function. Am. J. Physiol. -Heart Circ. Physiol. 2018, 314, H753–H765. [Google Scholar] [CrossRef]
- Rastogi, A.; Bhansali, A.; Khare, N.; Suri, V.; Yaddanapudi, N.; Sachdeva, N.; Puri, G.D.; Malhotra, P. Short term, high-dose vitamin D supplementation for COVID-19 disease: A randomised, placebo-controlled, study (SHADE study). Postgrad. Med. J. 2022, 98, 87–90. [Google Scholar] [CrossRef]
- Krajewska, M.; Witkowska-Sędek, E.; Rumińska, M.; Stelmaszczyk-Emmel, A.; Sobol, M.; Majcher, A.; Pyrżak, B. Vitamin D Effects on Selected Anti-Inflammatory and Pro-Inflammatory Markers of Obesity-Related Chronic Inflammation. Front. Endocrinol. 2022, 13, 920340. [Google Scholar] [CrossRef]
- Yang, C.-Y.; Leung, P.S.C.; Adamopoulos, I.E.; Gershwin, M.E. The Implication of Vitamin D and Autoimmunity: A Comprehensive Review. Clin. Rev. Allergy Immunol. 2013, 45, 217–226. [Google Scholar] [CrossRef]
- Zhang, J.; Shi, W.; Zou, M.; Zeng, Q.; Feng, Y.; Luo, Z.; Gan, H. Prevalence and risk factors of erectile dysfunction in COVID-19 patients: A systematic review and meta-analysis. J. Endocrinol. Investig. 2022, 1–10. [Google Scholar] [CrossRef] [PubMed]
| Hormone | Normal Range (Age Related) | T0 | T1 (6 Months) | T2 (12 Months) | T3 (18 Months) |
|---|---|---|---|---|---|
| Total Testosterone | 13–33 nmol/L | 21.1 | 19.9 | 19.8 | 20.2 |
| Free Testosterone | 0.17–0.66 nmol/L | 0.65 | 0.60 | 0.60 | 0.62 |
| Luteinizing Hormone (LH) | 1.8–12.0 mUI/mL | 6.3 | 7.1 | 6.9 | 7.6 |
| Follicle-stimulating Hormone (FSH) | 1.4–15.4 UI/L | 7.2 | 6.3 | 6.2 | 6.7 |
| Estradiol | 10–45 pg/mL | 12.0 | 13.2 | 12.7 | 13.2 |
| Sex-hormon binding-globulin (SHBG) | 10–57 nmol/L | 15.0 | 16.2 | 15.9 | 15.4 |
| Vitamin D | 30–150 ng/dL | 14.2 | 23.2 | 33.4 | 47.2 |
| Marker | Normal Range | T0 | T1 (6 Months) | T2 (12 Months) | T3 (18 Months) |
|---|---|---|---|---|---|
| Endothelin-1 | 0.87–1.61 pg/ml | 1.8 | 1.6 | 1.2 | 1.0 |
| C-Reactive Protein (CRP) | <5.0 mg/L | 8.8 | 6.2 | <5.0 | <5.0 |
| Questionnaire | Normal Scores (%) | T0 | T1 (6 Months) | T2 (12 Months) | T3 (18 Months) | |
|---|---|---|---|---|---|---|
| SF-36 | Vitality | 100 | 70 | 70 | 80 | 85 |
| Social functioning | 100 | 37.5 | 62.5 | 100 | 100 | |
| Role-emotional | 100 | 100 | 100 | 100 | 100 | |
| Mental Health | 100 | 72 | 76 | 88 | 96 | |
| Physical functioning | 100 | 90 | 95 | 100 | 100 | |
| Role-Physical | 100 | 75 | 100 | 100 | 100 | |
| Bodily pain | 100 | 77,5 | 77,5 | 100 | 100 | |
| General Health | 100 | 65 | 70 | 75 | 90 | |
| IEEF-5 | ≥22 | 13 | 17 | 20 | 23 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iuliano, S.; Seminara, G.; Zagari, M.C.; Di Luigi, L.; Aversa, A. Management of Persistent Erectile Dysfunction after COVID-19 Infection: An 18-Month Follow-Up Case Report. Sexes 2023, 4, 55-64. https://doi.org/10.3390/sexes4010006
Iuliano S, Seminara G, Zagari MC, Di Luigi L, Aversa A. Management of Persistent Erectile Dysfunction after COVID-19 Infection: An 18-Month Follow-Up Case Report. Sexes. 2023; 4(1):55-64. https://doi.org/10.3390/sexes4010006
Chicago/Turabian StyleIuliano, Stefano, Giuseppe Seminara, Maria Carmela Zagari, Luigi Di Luigi, and Antonio Aversa. 2023. "Management of Persistent Erectile Dysfunction after COVID-19 Infection: An 18-Month Follow-Up Case Report" Sexes 4, no. 1: 55-64. https://doi.org/10.3390/sexes4010006
APA StyleIuliano, S., Seminara, G., Zagari, M. C., Di Luigi, L., & Aversa, A. (2023). Management of Persistent Erectile Dysfunction after COVID-19 Infection: An 18-Month Follow-Up Case Report. Sexes, 4(1), 55-64. https://doi.org/10.3390/sexes4010006







