Anodic Potential and Conversion Chemistry of Anhydrous Iron (II) Oxalate in Na-Ion Batteries
Abstract
1. Introduction
2. Computational Method
2.1. Non-Periodic Calculations
2.2. Periodic Calculations
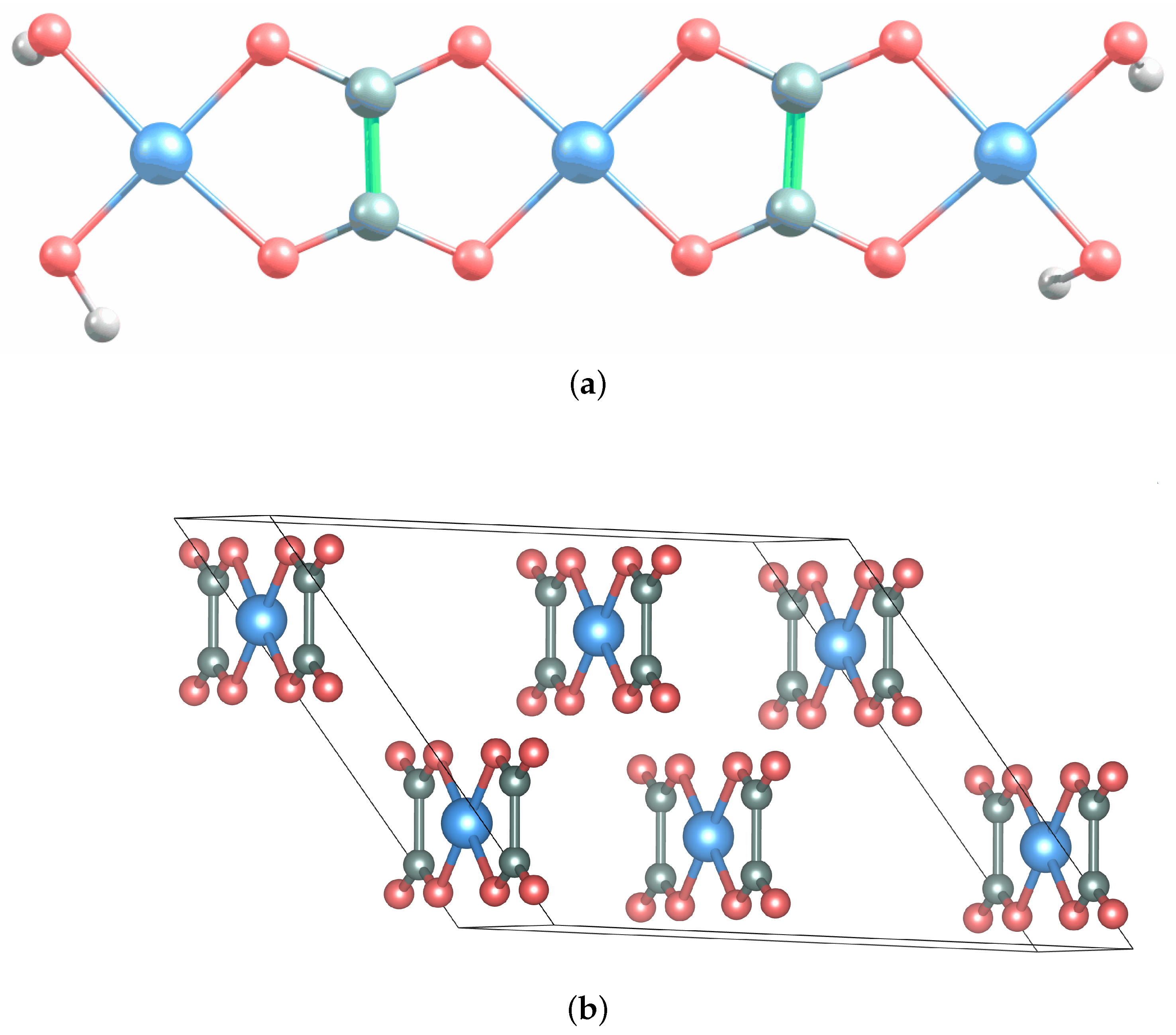
3. Results and Discussion
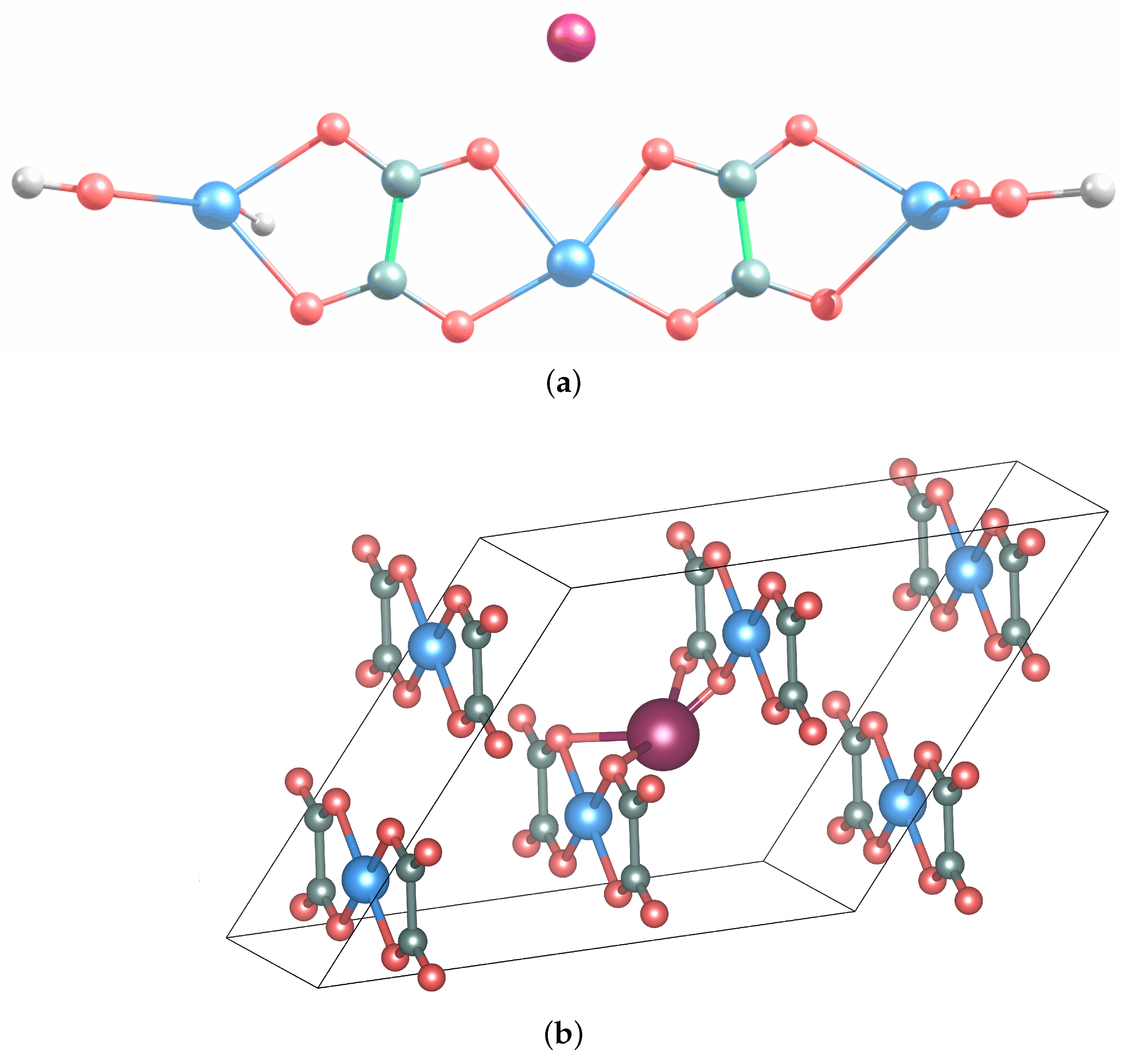
3.1. Na Interaction with AFO
3.2. Conversion Mechanism
3.3. Electrochemical Potential
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hoke, A.; Brissette, A.; Smith, K.; Pratt, A.; Maksimovic, D. Accounting for lithium-ion battery degradation in electric vehicle charging optimization. IEEE J. Emerg. Sel. Top. Power Electron. 2014, 2, 691–700. [Google Scholar] [CrossRef]
- Whittingham, M.S. Ultimate limits to intercalation reactions for lithium batteries. Chem. Rev. 2014, 114, 11414–11443. [Google Scholar] [CrossRef]
- Jaguemont, J.; Boulon, L.; Dubé, Y. A comprehensive review of lithium-ion batteries used in hybrid and electric vehicles at cold temperatures. Appl. Energy 2016, 164, 99–114. [Google Scholar] [CrossRef]
- Panchal, S.; Dincer, I.; Agelin-Chaab, M.; Fraser, R.; Fowler, M. Design and simulation of a lithium-ion battery at large C-rates and varying boundary conditions through heat flux distributions. Measurement 2018, 116, 382–390. [Google Scholar] [CrossRef]
- Lipu, M.H.; Hussain, A.; Saad, M.; Ayob, A.; Hannan, M. Improved recurrent NARX neural network model for state of charge estimation of lithium-ion battery using pso algorithm. In Proceedings of the 2018 IEEE Symposium on Computer Applications and Industrial Electronics (ISCAIE), Penang, Malaysia, 28–29 April 2018; pp. 354–359. [Google Scholar]
- Hwang, J.Y.; Myung, S.T.; Sun, Y.K. Sodium-ion batteries: Present and future. Chem. Soc. Rev. 2017, 46, 3529–3614. [Google Scholar] [CrossRef]
- Kim, D.; Kang, S.H.; Slater, M.; Rood, S.; Vaughey, J.T.; Karan, N.; Balasubramanian, M.; Johnson, C.S. Enabling sodium batteries using lithium-substituted sodium layered transition metal oxide cathodes. Adv. Energy Mater. 2011, 1, 333–336. [Google Scholar] [CrossRef]
- Liu, Y.; He, D.; Han, R.; Wei, G.; Qiao, Y. Nanostructured potassium and sodium ion incorporated Prussian blue frameworks as cathode materials for sodium-ion batteries. Chem. Commun. 2017, 53, 5569–5572. [Google Scholar] [CrossRef]
- Jafta, C.J. Grid scale energy storage: The alkali-ion battery systems of choice. Curr. Opin. Electrochem. 2022, 36, 101130. [Google Scholar] [CrossRef]
- Geng, G.; Keyu, Z.; Qianwen, W.; Libo, Z.; Dingfang, C.; Yaochun, Y. Metal oxalate-based anode materials: A new choice for energy storage materials applied in metal ion batteries. Prog. Chem. 2022, 34, 434. [Google Scholar]
- Yeoh, J.S.; Armer, C.F.; Lowe, A. Transition metal oxalates as energy storage materials. A review. Mater. Today Energy 2018, 9, 198–222. [Google Scholar] [CrossRef]
- Jo, C.H.; Yashiro, H.; Yuan, S.; Shi, L.; Myung, S.T. Conversion chemistry of cobalt oxalate for sodium storage. ACS Appl. Mater. Interfaces 2018, 10, 40523–40530. [Google Scholar] [CrossRef]
- Zhang, K.; Li, Y.; Hu, X.; Liang, F.; Wang, L.; Xu, R.; Dai, Y.; Yao, Y. Inhibitive role of crystal water on lithium storage for multilayer FeC2O4· xH2O anode materials. Chem. Eng. J. 2021, 404, 126464. [Google Scholar] [CrossRef]
- Poizot, P.; Laruelle, S.; Grugeon, S.; Dupont, L.; Tarascon, J. Nano-sized transition-metal oxides as negative-electrode materials for lithium-ion batteries. Nature 2000, 407, 496–499. [Google Scholar] [CrossRef] [PubMed]
- Cabana, J.; Monconduit, L.; Larcher, D.; Palacin, M.R. Beyond intercalation-based Li-ion batteries: The state of the art and challenges of electrode materials reacting through conversion reactions. Adv. Mater. 2010, 22, E170–E192. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.H.; Lee, S.H.; Lee, D.J.; Sung, Y.E.; Hyeon, T. Conversion reaction-based oxide nanomaterials for lithium ion battery anodes. Small 2016, 12, 2146–2172. [Google Scholar] [CrossRef] [PubMed]
- Grugeon, S.; Laruelle, S.; Dupont, L.; Tarascon, J.M. An update on the reactivity of nanoparticles Co-based compounds towards Li. Solid State Sci. 2003, 5, 895–904. [Google Scholar] [CrossRef]
- Vehmaanperä, P.; Gong, B.; Sit, P.H.L.; Salmimies, R.; Barbiellini, B.; Häkkinen, A. Formation of humboldtine during the dissolution of hematite in oxalic acid–Density functional theory (DFT) calculations and experimental verification. Clays Clay Miner. 2021, 69, 655–662. [Google Scholar] [CrossRef]
- Fan, X.; Zhang, L.; Li, M.; Wang, M.; Zhou, X.; Cheng, R.; Zhou, Y.; Shi, J. α-Ferrous oxalate dihydrate: A simple coordination polymer featuring photocatalytic and photo-initiated Fenton oxidations. Sci. China Mater. 2016, 59, 574–580. [Google Scholar] [CrossRef]
- Liu, Z.J.; Liu, W.; Wang, Y.; Guo, M.L. Preparation of β-ferrous oxalate dihydrate layered nanosheets by mechanochemical method and its visible-light-driven photocatalytic performance. Mater. Lett. 2016, 178, 83–86. [Google Scholar] [CrossRef]
- Yamada, T.; Sadakiyo, M.; Kitagawa, H. High proton conductivity of one-dimensional ferrous oxalate dihydrate. J. Am. Chem. Soc. 2009, 131, 3144–3145. [Google Scholar] [CrossRef]
- Chen, N.; Zhao, Y.; Li, M.; Wang, X.; Peng, X.; Sun, H.; Zhang, L. FeC2O4·H2O enables sustainable conversion of hydrogen peroxide to hydroxyl radical for promoted mineralization and detoxification of sulfadimidine. J. Hazard. Mater. 2022, 436, 129049. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; Mu, Y.; Yang, L.; Mei, Y.; Wu, M.; Zou, J.P.; Dionysiou, D.D.; Luo, S. Hydroxyl radical streaming from molecular oxygen activation by β-FeC2O4·2H2O for efficiently degrading Microcystin-LR. Appl. Catal. B 2023, 321, 121970. [Google Scholar] [CrossRef]
- Müller, H.; Bourcet, L.; Hanfland, M. Iron (II) oxalate dihydrate—Humboldtine: Synthesis, spectroscopic and structural properties of a versatile precursor for high pressure research. Minerals 2021, 11, 113. [Google Scholar] [CrossRef]
- Li, M.; Wang, W.; Yang, M.; Lv, F.; Cao, L.; Tang, Y.; Sun, R.; Lu, Z. Large-scale fabrication of porous carbon-decorated iron oxide microcuboids from Fe–MOF as high-performance anode materials for lithium-ion batteries. RSC Adv. 2015, 5, 7356–7362. [Google Scholar] [CrossRef]
- Lin, X.; Gao, J.; Zhong, K.; Huang, Y.; Yao, H.; Lin, Y.; Zheng, Y.; Huang, Z.; Li, J. Fe/Fe3C modification to effectively achieve high-performance Si-C anode. J. Mater. Chem. A 2022, 10, 23103–23112. [Google Scholar] [CrossRef]
- Aragón, M.J.; León, B.; Perez Vicente, C.; Tirado, J.L. Synthesis and electrochemical reaction with lithium of mesoporous iron oxalate nanoribbons. Inorg. Chem. 2008, 47, 10366–10371. [Google Scholar] [CrossRef]
- Zhang, K.; Li, Y.; Wang, Y.; Zhao, J.; Chen, X.; Dai, Y.; Yao, Y. Enhanced electrochemical properties of iron oxalate with more stable Li+ ions diffusion channels by controlling polymorphic structure. Chem. Eng. J. 2020, 384, 123281. [Google Scholar] [CrossRef]
- Keshavarz, F.; Kadek, M.; Barbiellini, B.; Bansil, A. Anodic activity of hydrated and anhydrous Iron (II) oxalate in Li-ion batteries. Condens. Matter 2022, 7, 8. [Google Scholar] [CrossRef]
- Butera, V.; Massaro, A.; Muñoz-García, A.B.; Pavone, M.; Detz, H. d-Glucose Adsorption on the TiO2 anatase (100) surface: A direct comparison between cluster-based and periodic approaches. Front. Chem. 2021, 9, 716329. [Google Scholar] [CrossRef]
- Soto, F.A.; Ma, Y.; Martinez de la Hoz, J.M.; Seminario, J.M.; Balbuena, P.B. Formation and growth mechanisms of solid-electrolyte interphase layers in rechargeable batteries. Chem. Mater. 2015, 27, 7990–8000. [Google Scholar] [CrossRef]
- Fasulo, F.; Muñoz-García, A.B.; Massaro, A.; Crescenzi, O.; Huang, C.; Pavone, M. Vinylene carbonate reactivity at lithium metal surface: First-principles insights into the early steps of SEI formation. J. Mater. Chem. A 2023, 11, 5660–5669. [Google Scholar] [CrossRef]
- Frisch, M.; Trucks, G.; Schlegel, H.; Scuseria, G.; Robb, M.; Cheeseman, J.; Scalmani, G.; Barone, V.; Petersson, G.; Nakatsuji, H.; et al. Gaussian 16 revision a. 03. 2016; Gaussian Inc.: Wallingford, CT, USA, 2016; Volume 2. [Google Scholar]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 1996, 77, 3865. [Google Scholar] [CrossRef] [PubMed]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 2010, 132, 154104. [Google Scholar] [CrossRef]
- Vidal Vidal, Á.; de Vicente Poutás, L.C.; Nieto Faza, O.; Silva López, C. On the use of popular basis sets: Impact of the intramolecular basis set superposition error. Molecules 2019, 24, 3810. [Google Scholar] [CrossRef]
- Giannozzi, P.; Baroni, S.; Bonini, N.; Calandra, M.; Car, R.; Cavazzoni, C.; Ceresoli, D.; Chiarotti, G.L.; Cococcioni, M.; Dabo, I.; et al. QUANTUM ESPRESSO: A modular and open-source software project for quantum simulations of materials. J. Phys. Condens. Matter 2009, 21, 395502. [Google Scholar] [CrossRef] [PubMed]
- Tominaka, S.; Cheetham, A. Intrinsic and extrinsic proton conductivity in metal-organic frameworks. RSC Adv. 2014, 4, 54382–54387. [Google Scholar] [CrossRef]
- Yuan, Y.x. A modified BFGS algorithm for unconstrained optimization. IMA J. Numer. Anal. 1991, 11, 325–332. [Google Scholar] [CrossRef]
- Weber, M.H.; Lynn, K.G.; Barbiellini, B.; Sterne, P.A.; Denison, A.B. Direct observation of energy-gap scaling law in CdSe quantum dots with positrons. Phys. Rev. B 2002, 66, 041305. [Google Scholar] [CrossRef]
- Tang, Q.; Zhou, Z.; Shen, P. Are MXenes promising anode materials for Li ion batteries? Computational studies on electronic properties and Li storage capability of Ti3C2 and Ti3C2X2 (X= F, OH) monolayer. J. Am. Chem. Soc. 2012, 134, 16909–16916. [Google Scholar] [CrossRef]
- Lv, X.; Li, F.; Gong, J.; Gu, J.; Lin, S.; Chen, Z. Metallic FeSe monolayer as an anode material for Li and non-Li ion batteries: A DFT study. Phys. Chem. Chem. Phys. 2020, 22, 8902–8912. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, G.; Deng, S.; Wu, Q.; Meng, Z.; Huang, X.; Bi, L.; Yang, Z.; Lu, R. Lithium and sodium decorated graphdiyne as a candidate for hydrogen storage: First-principles and grand canonical Monte Carlo study. Appl. Surf. Sci. 2020, 509, 144855. [Google Scholar] [CrossRef]
- Zhou, F.; Cococcioni, M.; Marianetti, C.A.; Morgan, D.; Ceder, G. First-principles prediction of redox potentials in transition-metal compounds with LDA+ U. Phys. Rev. B 2004, 70, 235121. [Google Scholar] [CrossRef]
- Callaway, J.; Zou, X.; Bagayoko, D. Total energy of metallic lithium. Phys. Rev. B 1983, 27, 631. [Google Scholar] [CrossRef]
- Qian, L.; Yu, T.; Wei, Z.; Chang, B.; Huang, G.; Wang, Z.; Liu, Y.; Sun, H.; Bai, L.; Huang, W. Lower-voltage plateau Zn-substituted Co3O4 submicron spheres anode for Li-ion half and full batteries. J. Alloys Compd. 2022, 890, 161888. [Google Scholar] [CrossRef]
- Ang, W.A.; Gupta, N.; Prasanth, R.; Madhavi, S. High-performing mesoporous iron oxalate anodes for lithium-ion batteries. ACS Appl. Mater. Interfaces 2012, 4, 7011–7019. [Google Scholar] [CrossRef]
- Pramanik, A.; Bradford, A.J.; Lee, S.L.; Lightfoot, P.; Armstrong, A.R. Na2Fe(C2O4)(HPO4): A promising new oxalate-phosphate based mixed polyanionic cathode for Li/Na ion batteries. J. Phys. Mater. 2021, 4, 024004. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, C.; Dong, Y.; Wei, R.; Zhang, J. Understanding the high-performance anode material of CoC2O4·2H2O microrods wrapped by reduced graphene oxide for lithium-ion and sodium-ion batteries. Chem. Eur. J. 2021, 27, 993–1001. [Google Scholar] [CrossRef]
- Kim, H.; Kim, D.I.; Yoon, W.S. Challenges and design strategies for conversion-based anode materials for lithium-and sodium-ion batteries. J. Electrochem. Sci. Technol. 2022, 13, 32–53. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gromov, V.; Noubir, A.; Keshavarz, F.; Laakso, E.; Barbiellini, B.; Bansil, A. Anodic Potential and Conversion Chemistry of Anhydrous Iron (II) Oxalate in Na-Ion Batteries. Condens. Matter 2023, 8, 38. https://doi.org/10.3390/condmat8020038
Gromov V, Noubir A, Keshavarz F, Laakso E, Barbiellini B, Bansil A. Anodic Potential and Conversion Chemistry of Anhydrous Iron (II) Oxalate in Na-Ion Batteries. Condensed Matter. 2023; 8(2):38. https://doi.org/10.3390/condmat8020038
Chicago/Turabian StyleGromov, Vasilii, Atlas Noubir, Fatemeh Keshavarz, Ekaterina Laakso, Bernardo Barbiellini, and Arun Bansil. 2023. "Anodic Potential and Conversion Chemistry of Anhydrous Iron (II) Oxalate in Na-Ion Batteries" Condensed Matter 8, no. 2: 38. https://doi.org/10.3390/condmat8020038
APA StyleGromov, V., Noubir, A., Keshavarz, F., Laakso, E., Barbiellini, B., & Bansil, A. (2023). Anodic Potential and Conversion Chemistry of Anhydrous Iron (II) Oxalate in Na-Ion Batteries. Condensed Matter, 8(2), 38. https://doi.org/10.3390/condmat8020038






