Assessing the Effects of Dietary Tea Polyphenols on the Gut Microbiota of Loaches (Paramisgurnus dabryanus) under Chronic Ammonia Nitrogen Stress
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Fish
2.2. Ammonia Stress Test
2.3. Sampling
2.4. Growth Performance Measurement
2.5. DNA Extraction and 16S rRNA High-Throughput Sequencing
2.6. Bioinformatics
2.7. Statistical Analysis
3. Results
3.1. Effects of Tea Polyphenols on Growth Performance
3.2. Pyrosequencing of the Gut Bacterial Community
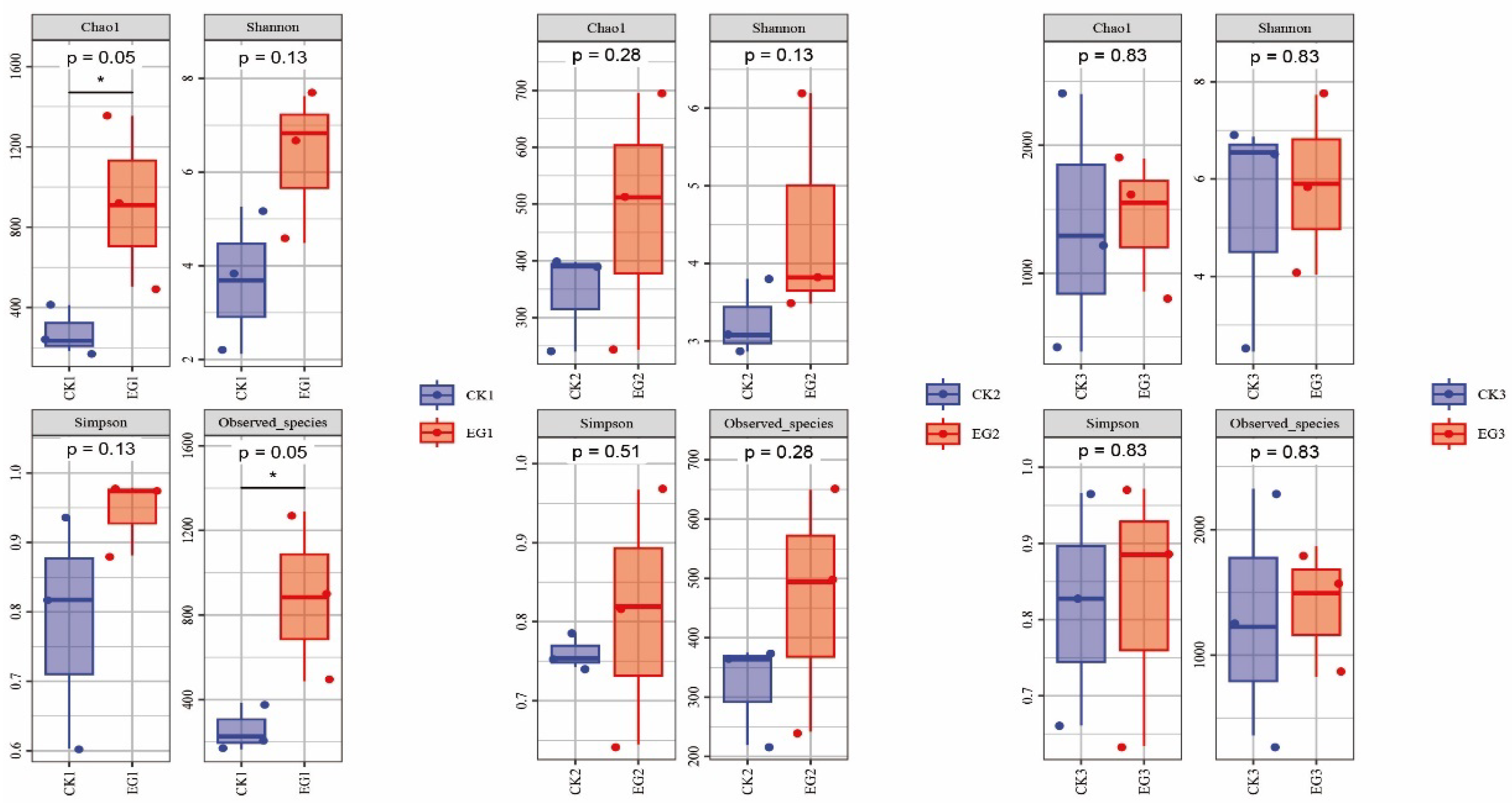
3.3. Alpha Diversity Reveals Alterations in Gut Bacterial Community Structure
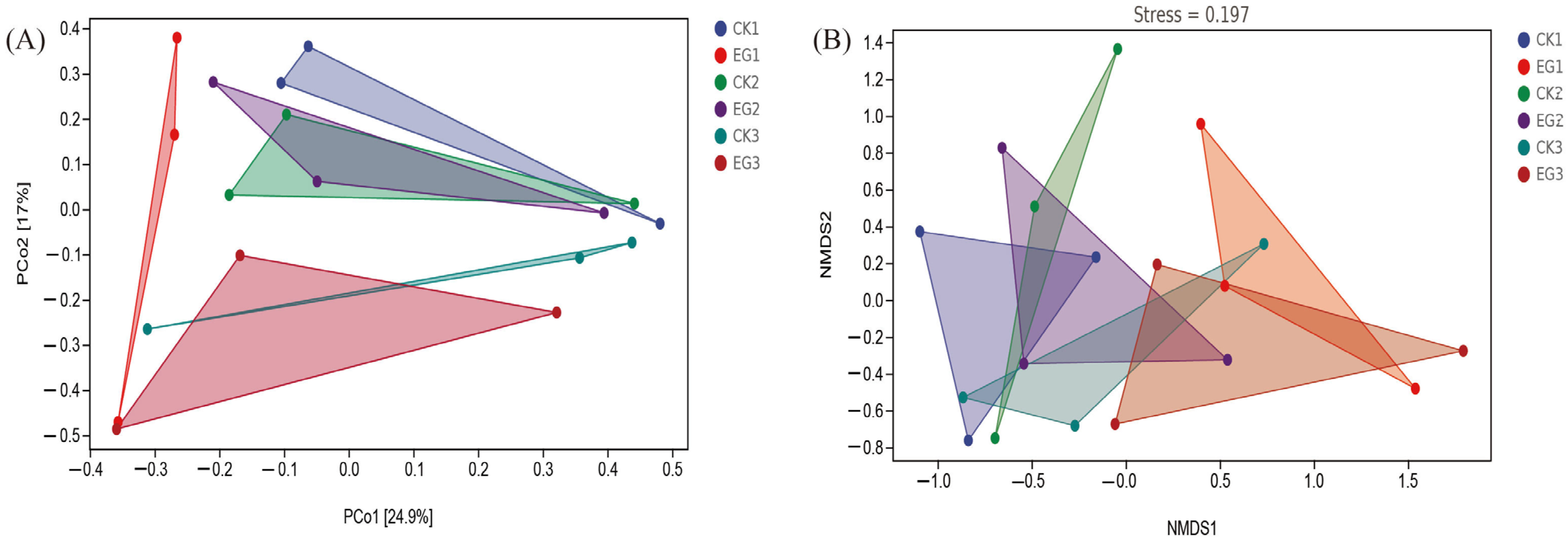
3.4. Beta-Diversity Analysis Reveals Shifts in Gut Bacterial Community Structure
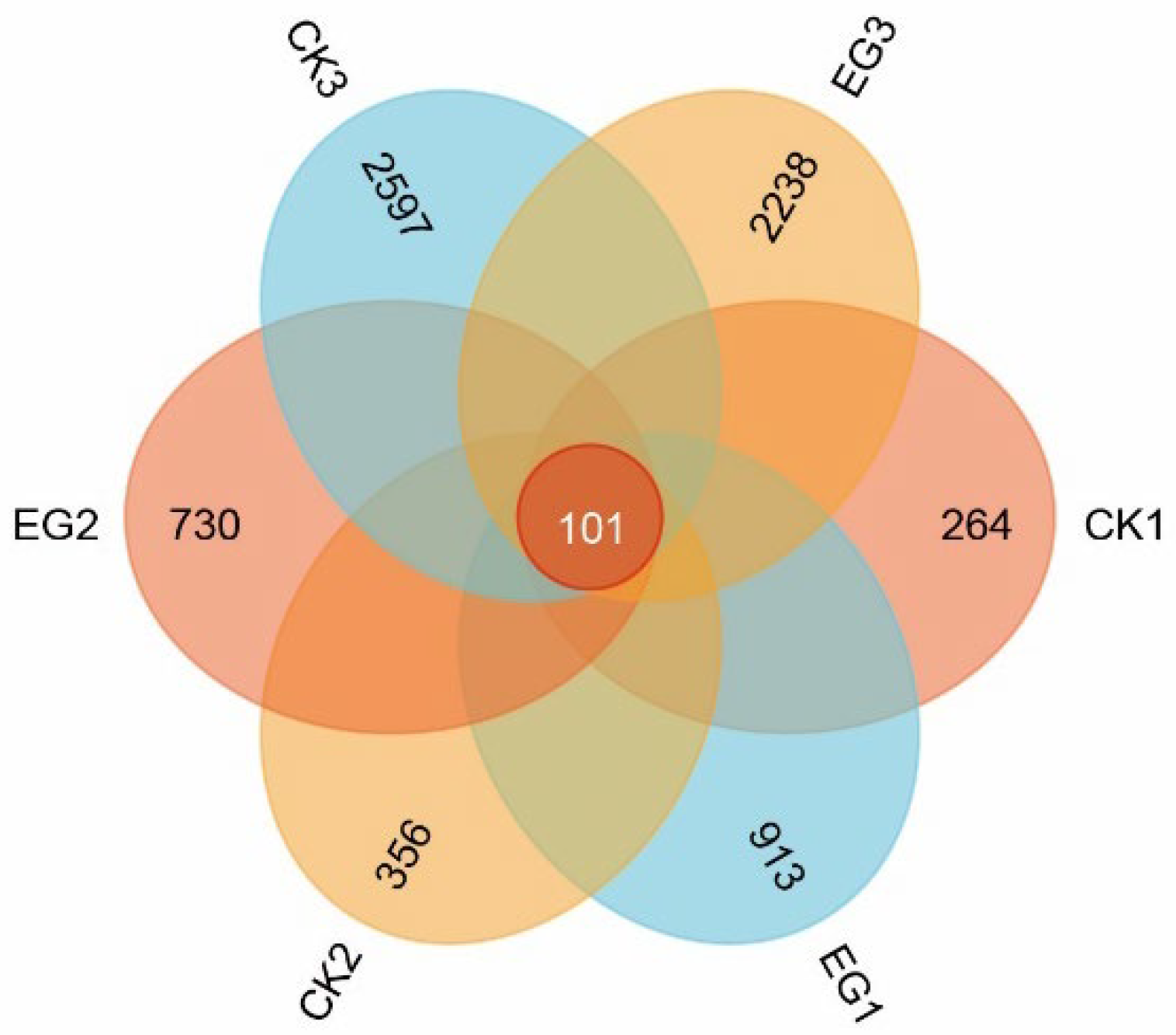
3.5. Venn Diagram Analysis
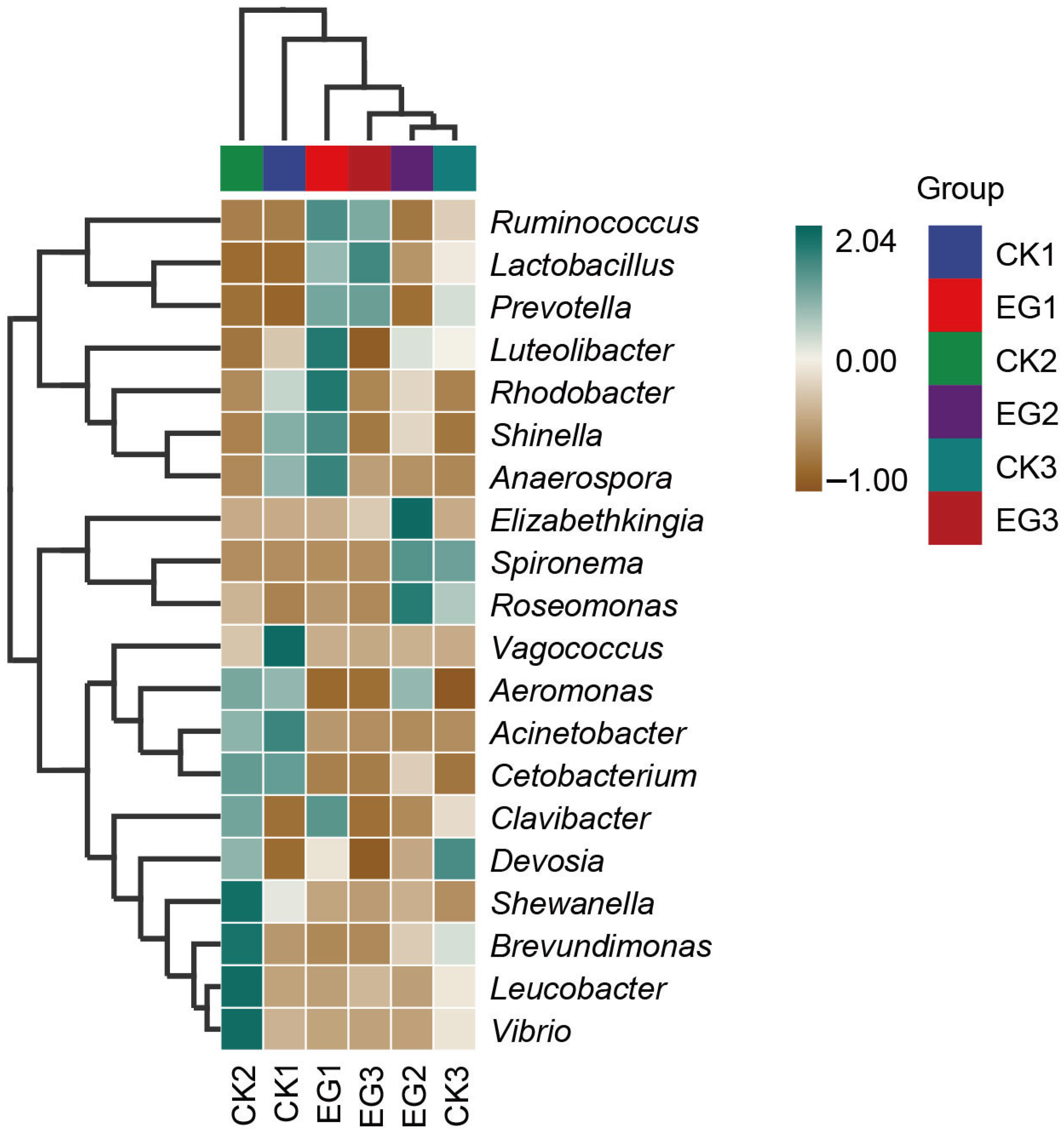
3.6. Taxonomic Differences and Marker Species
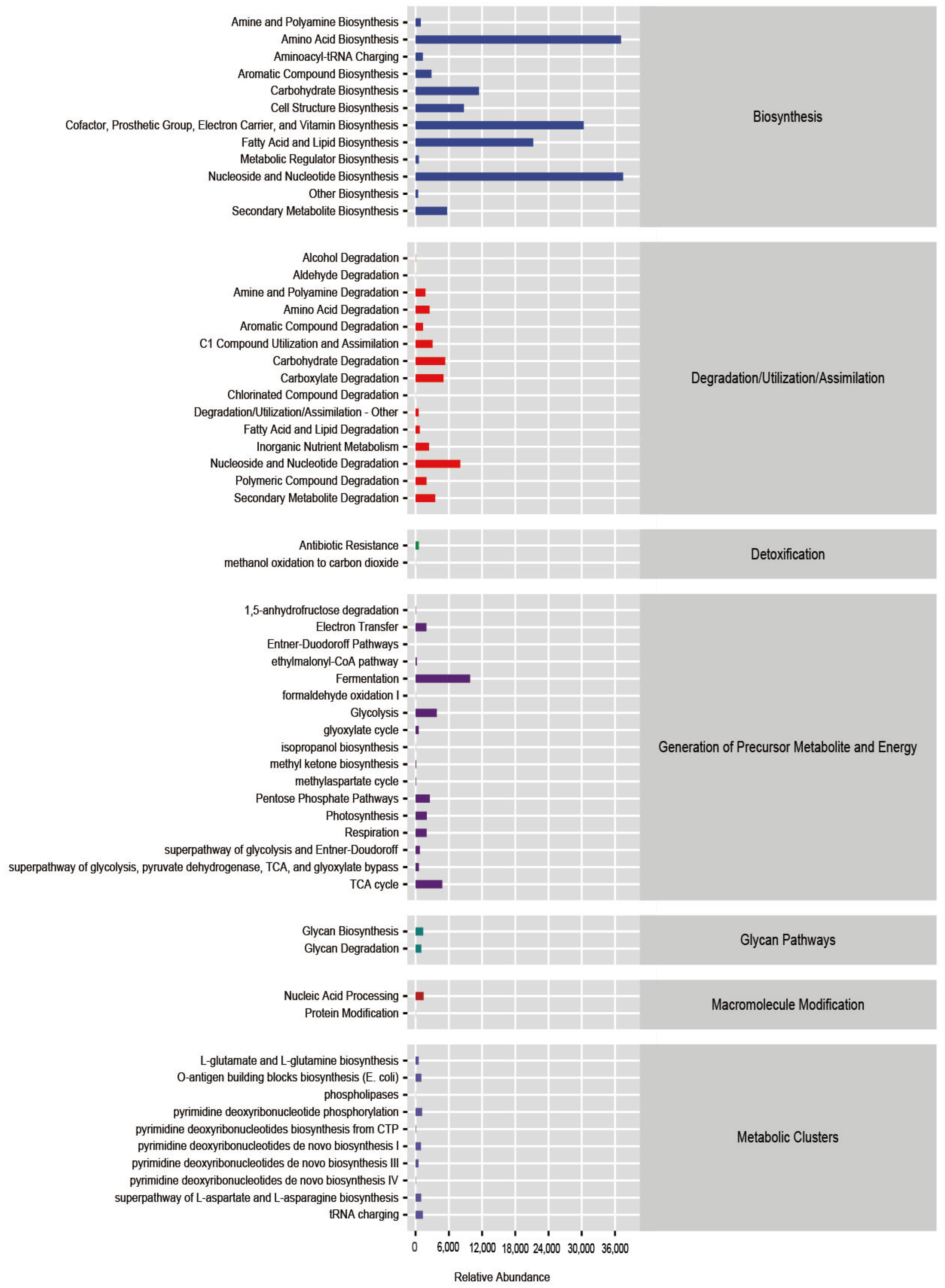
3.7. Functional Prediction of Intestinal Microbiota
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhao, J.H.; Lam, T.J.; Guo, J.Y. Acute toxicity of ammonia to the early stage-larvae and juveniles of Eriocheir sinensis H. Milne-Edwards, 1853 (Decapoda: Grapsidae) reared in the laboratory. Aquac. Res. 1997, 28, 517–525. [Google Scholar] [CrossRef]
- Dutra, F.M.; Forneck, S.C.; Brazão, C.C.; Freire, C.A.; Ballester, E.L.C. Acute toxicity of ammonia to various life stages of the Amazon river prawn, Macrobrachium amazonicum, Heller, 1862. Aquaculture 2016, 453, 104–109. [Google Scholar] [CrossRef]
- Ip, Y. Ammonia production, excretion, toxicity, and defense in fish: A review. Front. Physiol. 2011, 1, 134. [Google Scholar] [CrossRef]
- Mangang, Y.A.; Pandey, P.K. Hemato-biochemical responses and histopathological alterations in the gill and kidney tissues of Osteobrama belangeri (Valenciennes, 1844) exposed to different sub-lethal unionized ammonia. Aquaculture 2021, 542, e736887. [Google Scholar] [CrossRef]
- Silva, M.; Costa, F.F.B.D.; Leme, F.P.; Takata, R.; Miranda-Filho, K.C. Biological responses of Neotropical freshwater fish Lophiosilurus alexandri exposed to ammonia and nitrite. Sci. Total Environ. 2017, 616, 1566–1575. [Google Scholar] [CrossRef] [PubMed]
- Tsui, H.T.; Randall, A.D. Ammonia toxicity in fish. Mar. Pollut. Bull. 2001, 45, 17–23. [Google Scholar] [CrossRef]
- Yue, F.; Pan, L.; Xie, P.; Zheng, D.; Li, J. Immune responses and expression of immune-related genes in swimming crab Portunus trituberculatus exposed to elevated ambient ammonia-N stress. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2010, 157, 246–251. [Google Scholar] [CrossRef]
- Shan, H.; Wang, T.; Dong, Y.; Ma, S. Effects of dietary Ampithoe sp. supplementation on the growth, energy status, antioxidant capacity, and ammonia-N tolerance of the shrimp Litopenaeus vannamei: Continuous versus interval feeding. Aquaculture 2019, 509, 32–39. [Google Scholar] [CrossRef]
- Salem, M.E.S.; Abdel-Ghany, H.M.; Sallam, A.E.; El-Feky, M.M.M.; Almisherfi, H.M. Effects of dietary orange peel on growth performance, antioxidant activity, intestinal microbiota and liver histology of Gilthead sea bream (Sparus aurata) larvae. Aquac. Nutr. 2019, 25, 1087–1097. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, Y.; Li, J.; Fu, C.; Zhang, X. The neuroprotective effect of tea polyphenols on the regulation of intestinal flora. Molecules 2021, 26, 3692. [Google Scholar] [CrossRef]
- Li, H.; Christman, L.M.; Li, R.; Gu, L. Synergic interactions between polyphenols and gut microbiota in mitigating inflammatory bowel diseases. Food Funct. 2020, 11, 4878–4891. [Google Scholar] [CrossRef]
- Pérez-Burillo, S.; Navajas-Porras, B.; López-Maldonado, A.; Hinojosa-Nogueira, D.; Pastoriza, S.; Rufián-Henares, J.Á. Green tea and its relation to human gut microbiome. Molecules 2021, 26, 3907. [Google Scholar] [CrossRef]
- Wang, S.; Li, X.; Zhang, M.; Jiang, H.; Wang, R.; Qian, Y.; Li, M. Ammonia stress disrupts intestinal microbial community and amino acid metabolism of juvenile yellow catfish (Pelteobagrus fulvidraco). Ecotoxicol. Environ. Saf. 2021, 227, e112932. [Google Scholar] [CrossRef]
- Yang, C.S.; Kim, S.; Yang, G.Y.; Lee, M.J.; Liao, J.; Chung, J.Y.; Ho, C.T. Inhibition of Carcinogenesis by Tea: Bioavailability of Tea Polyphenols and Mechanisms of Actions. Exp. Biol. Med. 1999, 220, 213–217. [Google Scholar] [CrossRef]
- Ciraj, A.M.; Sulaim, J.; Mamatha, B.; Gopalkrishna, B.K.; Shivananda, P.G. Antibacterial activity of black tea (Camelia sinensis) extract against Salmonella serotypes causing enteric fever. Indian J. Med. Sci. 2001, 55, 376. [Google Scholar]
- Zhang, H.; Lu, X.; Zhang, Y.; Ma, X.; Wang, S.; Ni, Y.; Chen, J. Bioaccumulation of organochlorine pesticides and polychlorinated biphenyls by loaches living in rice paddy fields of Northeast China. Environ. Pollut. 2016, 216, 893–901. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; He, K.; Xiao, Q.; Liu, Q.; Luo, W.; Luo, J.; Fu, H.; Li, J.; Wu, X.; Du, J.; et al. Comparative transcriptome profiles of large and small bodied large-scale loaches cultivated in paddy fields. Sci. Rep. 2021, 11, e4936. [Google Scholar] [CrossRef]
- Gao, J.; Koshio, S.; Nguyen, B.T.; Wang, W.; Cao, X. Comparative studies on lipid profiles and amino acid composition of wild and cultured Dojo loach Misgurnus anguillicaudatus obtained from southern Japan. Fish. Sci. 2012, 78, 1331–1336. [Google Scholar] [CrossRef]
- Qian, Y.-C.; Wang, X.; Ren, J.; Wang, J.; Limbu, S.M.; Li, R.-X.; Zhou, W.-H.; Qiao, F.; Zhang, M.-L.; Du, Z.-Y. Different effects of two dietary levels of tea polyphenols on the lipid deposition, immunity and antioxidant capacity of juvenile GIFT tilapia (Oreochromis niloticus) fed a high-fat diet. Aquaculture 2021, 542, e736896. [Google Scholar] [CrossRef]
- Guo, H.; Lin, W.; Hou, J.; Wang, L.; Zhang, D.; Wu, X.; Li, L.; Li, D. The protective roles of dietary selenium yeast and tea polyphenols on growth performance and ammonia tolerance of juvenile Wuchang bream (Megalobrama amblycephala). Front. Physiol. 2018, 9, 377933. [Google Scholar] [CrossRef]
- Ji, R.; Li, Y.; Li, X.; Xiang, X.; Li, Y.; Zhu, S.; Yang, B.; Zhang, Y.; Mai, K.; Ai, Q. Effects of dietary tea polyphenols on growth, biochemical and antioxidant responses, fatty acid composition and expression of lipid metabolism related genes of large yellow croaker (Larimichthys crocea). Aquac. Res. 2018, 49, 1210–1218. [Google Scholar] [CrossRef]
- Li, D.; Xu, X.; Li, Z.; Wang, T.; Wang, C. Detection methods of ammonia nitrogen in water: A review. TrAC Trends Anal. Chem. 2020, 127, e115890. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, J.; Zhou, Q.; Wang, Z.; Zhang, W. Analysis of the Intestinal Flora in Male Versus Female Swamp Eels (Monopterus albus). Front. Microbiol. 2020, 11, e699. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Xiao, X.H.; Zhang, Q.; Mao, L.L.; Yu, M.; Xu, J.P.; Wang, T. Correlation of placental microbiota with fetal macrosomia and clinical characteristics in mothers and newborns. Oncotarget 2017, 8, e82314. [Google Scholar] [CrossRef] [PubMed]
- Bokulich, N.A.; Subramanian, S.; Faith, J.J.; Gevers, D.; Gordon, J.I.; Knight, R.; Mills, D.A.; Caporaso, J.G. Quality-filtering vastly improves diversity estimates from Illumina amplicon sequencing. Nat. Methods 2013, 10, 57–59. [Google Scholar] [CrossRef]
- Xing, Y.; Liu, J.; Lu, F.; Wang, L.; Li, Y.; Ouyang, C.; Shin, J.H. Dynamic distribution of gallbladder microbiota in rabbit at different ages and health states. PLoS ONE 2019, 14, e0211828. [Google Scholar] [CrossRef]
- Callahan, B.J.; Mcmurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed]
- Bokulich, N.A.; Kaehler, B.D.; Ram, R.J.; Matthew, D.; Evan, B.; Rob, K.; Huttley, G.A.; Gregory, C.J. Optimizing taxonomic classification of marker-gene amplicon sequences with QIIME 2′s q2-feature-classifier plugin. Microbiome 2018, 6, 90. [Google Scholar] [CrossRef]
- DeSantis, T.Z.; Hugenholtz, P.; Larsen, N.; Rojas, M.; Brodie, E.L.; Keller, K.; Huber, T.; Dalevi, D.; Hu, P.; Andersen, G.L. Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl. Environ. Microbiol. 2006, 72, 5069–5072. [Google Scholar] [CrossRef]
- Bray, J.R.; Curtis, J.T. An ordination of the upland forest communities of Southern Wisconsin. Ecol. Monogr. 1957, 27, 326–349. [Google Scholar] [CrossRef]
- Zaura, E.; Keijser, B.J.F.; Huse, S.M.; Crielaard, W. Defining the healthy “core microbiome” of oral microbial communities. BMC Microbiol. 2009, 9, 259. [Google Scholar] [CrossRef] [PubMed]
- Shang, Z.H.; Huang, M.; Wu, M.X.; Mi, D.; You, K.; Zhang, Y.I. Transcriptomic analyses of the acute aerial and ammonia stress response in the gill and liver of large-scale loach (Paramisgurnus dabryanus). Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2021, 250, e109185. [Google Scholar] [CrossRef] [PubMed]
- Chai, Y.; Peng, R.; Jiang, M.; Jiang, X.; Han, Z. Effects of ammonia nitrogen stress on the blood cell immunity and liver antioxidant function of Sepia pharaonis. Aquaculture 2021, 546, 737417. [Google Scholar] [CrossRef]
- Li, Y.; Pan, L.; Tong, R.; Li, Y.; Li, Z.; Chen, Y. Effects of ammonia-N stress on molecular mechanisms associated with immune behavior changes in the haemocytes of Litopenaeus vannamei. Mol. Immunol. 2022, 149, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.F.; Sun, B.; Chen, D.D.; Yi, C.; Teng, J.; Yu, J.L.; Wang, S.Q.; Ru, Y.Y.; Wang, H. Characterization of a novel ADP-ribosylation factor gene from Macrobrachium nipponense and its response to ammonia nitrogen stress. Aquac. Environ. Interact. 2021, 13, 165–175. [Google Scholar] [CrossRef]
- Zhao, C.; Xu, J.; Xu, X.; Wang, Q.; Kong, Q.; Xu, F.; Du, Y. Organ-specific responses to total ammonia nitrogen stress on juvenile grass carp (Ctenopharyngodon idellus). Environ. Sci. Pollut. Res. Int. 2019, 26, 10826–10834. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Gao, J.; Zhang, R.; Zhang, R. Ammonia stress on nitrogen metabolism in tolerant aquatic plant-Myriophyllum aquaticum. Ecotoxicol. Environ. Saf. 2017, 143, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Burr, G.; Gatlin III, D.; Ricke, S. Microbial ecology of the gastrointestinal tract of fish and the potential application of prebiotics and probiotics in finfish aquaculture. J. World Aquac. Soc. 2005, 36, 425–436. [Google Scholar] [CrossRef]
- Welker, T.L.; Wan, X.C.; Zhou, Y.B.; Yang, Y.O.; Overturf, K.; Barrows, F.; Liu, K. Effect of dietary green tea supplementation on growth, fat content, and muscle fatty acid profile of rainbow trout (Oncorhynchus mykiss). Aquac. Int. 2016, 25, 1073–1094. [Google Scholar] [CrossRef]
- Hwang, J.H.; Lee, S.W.; Rha, S.J.; Yoon, H.S.; Kim, S.J. Dietary green tea extract improves growth performance, body composition, and stress recovery in the juvenile black rockfish, Sebastes schlegeli. Aquac. Int. 2013, 21, 525–538. [Google Scholar] [CrossRef]
- Zhong, L.; Hu, Y.; Hu, Y.; Li, J.; Tian, Y.; Chen, J.; Ai, Q.; Xiao, T. Effects of dietary tea polyphenols on growth, immunity and lipid metabolism of juvenile black carp Mylopharyngodon piceus. Aquac. Res. 2020, 51, 569–576. [Google Scholar] [CrossRef]
- Kormas, K.A.; Meziti, A.; Mente, E.; Frentzos, A. Dietary differences are reflected on the gut prokaryotic community structure of wild and commercially reared sea bream (Sparus aurata). Microbiol. Open 2014, 3, 718–728. [Google Scholar] [CrossRef]
- Wu, S.; Wang, G.; Angert, E.R.; Wang, W.; Li, W.; Zou, H.; Wu, S.; Wang, G.; Angert, E.R.; Wang, W. Composition, Diversity, and Origin of the Bacterial Community in Grass Carp Intestine. PLoS ONE 2012, 7, e30440. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.Z.; Huang, J.S.; Zhang, J.D.; Wang, Z.L.; Chen, G. Effects of hypoxia stress on the intestinal microflora of juvenile of cobia (Rachycentron canadum). Aquaculture 2021, 536, 736419. [Google Scholar] [CrossRef]
- Lyons, P.P.; Turnbull, J.F.; Dawson, K.A.; Crumlish, M. Phylogenetic and functional characterization of the distal intestinal microbiome of rainbow trout Oncorhynchus mykiss from both farm and aquarium settings. J. Appl. Microbiol. 2017, 122, 347–363. [Google Scholar] [CrossRef]
- Shin, N.R.; Whon, T.W.; Bae, J.W. Proteobacteria: Microbial signature of dysbiosis in gut microbiota. Trends Biotechnol. 2015, 33, 496–503. [Google Scholar] [CrossRef] [PubMed]
- Cotter, P. Beneficial modulation of the gut microbiota. Int. J. Infect. Dis. 2016, 45, 38. [Google Scholar] [CrossRef][Green Version]
- Zhao, Y.; Hao, Q.; Zhang, Q.; Yang, Y.; Ran, C.; Xu, Q.; Wu, C.; Liu, W.; Li, S.; Zhang, Z.; et al. Nuclease treatment enhanced the ameliorative effect of yeast culture on epidermal mucus, hepatic lipid metabolism, inflammation response and gut microbiota in high-fat diet-fed zebrafish. Fish Shellfish Immunol. 2022, 131, 1182–1191. [Google Scholar] [CrossRef]
- Walczak, N.; Puk, K.; Guz, L. Bacterial Flora Associated with Diseased Freshwater Ornamental Fish. J. Vet. Res. 2017, 61, 445–449. [Google Scholar] [CrossRef]
- Butt, R.L.; Volkoff, H. Gut Microbiota and Energy Homeostasis in Fish. Front. Endocrinol. 2019, 10, 9. [Google Scholar] [CrossRef]
- Xiang, Q.; Liu, Y.; Wu, Z.; Wang, R.; Zhang, X. New hints for improving sleep: Tea polyphenols mediate gut microbiota to regulate circadian disturbances. Food Front. 2023, 4, 47–59. [Google Scholar] [CrossRef]
- Li, Y.; Rahman, S.U.; Huang, Y.; Zhang, Y.; Ming, P.; Zhu, L.; Chu, X.; Li, J.; Feng, S.; Wang, X.; et al. Green tea polyphenols decrease weight gain, ameliorate alteration of gut microbiota, and mitigate intestinal inflammation in canines with high-fat-diet-induced obesity. J. Nutr. Biochem. 2020, 78, e108324. [Google Scholar] [CrossRef] [PubMed]
- Hong, M.Y.; Zhang, R.L.; Liu, Y.A.; Wu, Z.F.; Weng, P.F. The interaction effect between tea polyphenols and intestinal microbiota: Role in ameliorating neurological diseases. J. Food Biochem. 2022, 46, e13870. [Google Scholar] [CrossRef] [PubMed]
- Semova, I.; Carten, J.D.; Stombaugh, J.; Mackey, L.C.; Knight, R.; Farber, S.A.; Rawls, J.F. Microbiota regulate intestinal absorption and metabolism of fatty acids in the zebrafish. Cell Host Microbe 2012, 12, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.-Z.; Yu, Y.-J.; Adeli, K. Role of Gut Microbiota in Neuroendocrine Regulation of Carbohydrate and Lipid Metabolism via the Microbiota-Gut-Brain-Liver Axis. Microorganisms 2020, 8, 527. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Tang, L.; Shen, C.L.; Wang, J.S. Green Tea Polyphen. Modify gut-microbiota dependent metabolisms of energy, bile constituents and micronutrients in female Sprague-Dawley rats. J. Nutr. Biochem. 2018, 61, 68–81. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Li, L.; Jin, J.; Zhang, B.; Wei, H.; Zhao, Y.; Li, X.; Li, Y. Comparative analysis of gut bacterial community composition during a single day cycle in Chinese mitten crab (Eriocheir sinensis). Aquac. Rep. 2021, 21, e100907. [Google Scholar] [CrossRef]
- Lee, S.M.L.; Wong, W.P.; Loong, A.M.; Hiong, K.C.; Chew, S.F.; Ip, Y.K. Postprandial increases in nitrogenous excretion and urea synthesis in the Chinese soft-shelled turtle, Pelodiscus sinensis. J. Comp. Physiol. B 2007, 177, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Sinha, A.K.; Giblen, T.; Abdelgawad, H.; De Rop, M.; Asard, H.; Blust, R.; De Boeck, G. Regulation of amino acid metabolism as a defensive strategy in the brain of three freshwater teleosts in response to high environmental ammonia exposure. Aquat. Toxicol. 2013, 130, 86–96. [Google Scholar] [CrossRef]
- John, R.; Rajan, A.P. Effective sequestration of chromium by bacterial biosorption: A review. Prep. Biochem. Biotechnol. 2021, 51, 738–748. [Google Scholar] [CrossRef]
- Yan, Z.M.; Zhong, Y.Z.; Duan, Y.H.; Chen, Q.H.; Li, F.N. Antioxidant mechanism of tea polyphenols and its impact on health benefits. Anim. Nutr. 2020, 6, 115–123. [Google Scholar] [CrossRef] [PubMed]

| Ingredient | No Tea Polyphenols | Contains Tea Polyphenols |
|---|---|---|
| Fish meal | 20 | 20 |
| Dextrin | 5 | 5 |
| Soybean meal | 32 | 31.99 |
| Flour | 10 | 10 |
| Wheat bran | 11 | 11 |
| Corn gluten powder | 15 | 15 |
| Fish oil | 5 | 5 |
| Calcium phosphate | 1 | 1 |
| Vitamin premix | 1 | 1 |
| Tea polyphenols | 0 | 0.01 |
| Total ratio | 100 | 100 |
| Control Group | Tea Polyphenol Group | p-Values | |
|---|---|---|---|
| Initial body weight/g | 8.29 ± 0.05 | 8.53 ± 0.32 | 0.284 |
| Day 10 weight/g | 9.68 ± 1.08 | 9.06 ± 0.12 | 0.38 |
| Day 20 weight/g | 9.11 ± 0.89 | 9.67 ± 0.25 | 0.358 |
| Final weight/g | 10.8 ± 0.53 | 9.93 ± 0.13 | 0.054 |
| Weight gain/% | 30.24 ± 5.98 a | 16.59 ± 5.67 b | 0.045 |
| Specific growth rate/% | 8.36 ± 1.68 a | 4.68 ± 1.45 b | 0.046 |
| Survival/% | 71.7 | 83.3 | 0.206 |
| Sample | Chao | Coverage | Species | Shannon | Simpson | Group |
|---|---|---|---|---|---|---|
| CK10_1 | 234.588 | 0.999705 | 226.6 | 3.68927 | 0.817233 | CK1 |
| CK10_2 | 184.037 | 0.999535 | 165.8 | 2.12276 | 0.603577 | CK1 |
| CK10_3 | 411.753 | 0.999451 | 386.0 | 5.26191 | 0.936348 | CK1 |
| EG10_1 | 909.117 | 0.999194 | 884.3 | 6.83122 | 0.973744 | EG1 |
| EG10_2 | 503.742 | 0.999483 | 487.4 | 4.48923 | 0.881454 | EG1 |
| EG10_3 | 1354.28 | 0.998181 | 1288.9 | 7.62177 | 0.978703 | EG1 |
| CK20_1 | 240.27 | 0.999409 | 219.5 | 2.86509 | 0.753720 | CK2 |
| CK20_2 | 390.41 | 0.999178 | 363.9 | 3.07832 | 0.742806 | CK2 |
| CK20_3 | 398.164 | 0.999338 | 374.8 | 3.79994 | 0.785523 | CK2 |
| EG20_1 | 511.733 | 0.999210 | 494.5 | 6.19355 | 0.967188 | EG2 |
| EG20_2 | 695.119 | 0.998515 | 649.4 | 3.48330 | 0.644694 | EG2 |
| EG20_3 | 243.425 | 0.999840 | 241.6 | 3.82030 | 0.819175 | EG2 |
| CK30_1 | 387.626 | 0.999210 | 362.7 | 2.45514 | 0.661230 | CK3 |
| CK30_2 | 1292.57 | 0.997923 | 1225.6 | 6.86703 | 0.966195 | CK3 |
| CK30_3 | 2402.26 | 0.996557 | 2324.1 | 6.54731 | 0.827503 | CK3 |
| EG30_1 | 1550.90 | 0.997167 | 1493.7 | 5.90112 | 0.885300 | EG3 |
| EG30_2 | 858.088 | 0.999107 | 828.8 | 4.03164 | 0.634363 | EG3 |
| EG30_3 | 1896.01 | 0.997834 | 1868.3 | 7.73055 | 0.971686 | EG3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chai, Y.; Sun, S.; Li, Y. Assessing the Effects of Dietary Tea Polyphenols on the Gut Microbiota of Loaches (Paramisgurnus dabryanus) under Chronic Ammonia Nitrogen Stress. Fishes 2024, 9, 180. https://doi.org/10.3390/fishes9050180
Chai Y, Sun S, Li Y. Assessing the Effects of Dietary Tea Polyphenols on the Gut Microbiota of Loaches (Paramisgurnus dabryanus) under Chronic Ammonia Nitrogen Stress. Fishes. 2024; 9(5):180. https://doi.org/10.3390/fishes9050180
Chicago/Turabian StyleChai, Yuqiao, Shuhao Sun, and Yingdong Li. 2024. "Assessing the Effects of Dietary Tea Polyphenols on the Gut Microbiota of Loaches (Paramisgurnus dabryanus) under Chronic Ammonia Nitrogen Stress" Fishes 9, no. 5: 180. https://doi.org/10.3390/fishes9050180
APA StyleChai, Y., Sun, S., & Li, Y. (2024). Assessing the Effects of Dietary Tea Polyphenols on the Gut Microbiota of Loaches (Paramisgurnus dabryanus) under Chronic Ammonia Nitrogen Stress. Fishes, 9(5), 180. https://doi.org/10.3390/fishes9050180









