The Impact of Marine Resource-Free Diets on Quality Attributes of Atlantic Salmon
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Material
2.2. Assessment of Surface Area, Eyes and Fins
2.3. Texture and Color Determination
2.4. Sensory Evaluation
2.5. Authenticity of Fillets
2.6. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- OECD. OECD Review of Fisheries 2022; OECD Publishing: Paris, France, 2022. [Google Scholar] [CrossRef]
- McLean, E. Feed ingredients for sustainable aquaculture. In Reference Module in Food Science; Ferranti, P., Ed.; Sustainable Food Science: A Comprehensive Approach; Elsevier Inc.: Amsterdam, The Netherlands, 2023. [Google Scholar] [CrossRef]
- Næve, I.; Korsvoll, S.A.; Santi, N.; Medina, M.; Aunsmo, A. The power of genetics: Past and future contribution of balanced genetic selection to sustainable growth and productivity of the Norwegian Atlantic salmon (Salmo salar) industry. Aquaculture 2022, 553, 738061. [Google Scholar] [CrossRef]
- Lekang, O.-I. Feeding equipment and control systems. In Feed and Feeding Practices in Aquaculture; Davis, D.A., Ed.; Woodhead Publishing: Cambridge, MA, USA, 2022; pp. 399–426. [Google Scholar]
- Bogomolov, I.S.; Afanasyev, V.A.; Ostrikov, A.N.; Startseva, S.V. The choice of rational modes of vacuum deposition of fatty components in the preparation of feed for valuable fish species. IOP Conf Ser. Earth Environ. Sci. 2022, 1052, 012090. [Google Scholar] [CrossRef]
- Barrows, F.T.; Campbell, K.B.; Gaylord, T.G.; Sanchez, R.C.M.; Castillo, S.A.; McLean, E. Influence of Krill Meal on the Performance of post-smolt Atlantic salmon that are fed plant-based and animal-based fishmeal and fish oil-free diets. Fishes 2023, 8, 590. [Google Scholar] [CrossRef]
- Doney, S.C.; Ruckelshaus, M.; Duffy, J.E.; Barry, J.P.; Chan, F.; English, C.A.; Galindo, H.M.; Grebmeier, J.M.; Hollowed, A.B.; Knowlton, N.; et al. Climate change impacts on marine ecosystems. Ann. Rev. Mar. Sci. 2012, 5, 11–37. [Google Scholar] [CrossRef] [PubMed]
- Pacoureau, N.; Rigby, C.L.; Kyne, P.M.; Sherley, R.B.; Winker, H.; Carlson, J.K.; Fordham, S.V.; Barreto, R.; Fernando, D.; Francis, M.P.; et al. Half a century of global decline in oceanic sharks and rays. Nature 2021, 589, 567–571. [Google Scholar] [CrossRef]
- Smith, K.E.; Burrows, M.T.; Hobday, A.J.; King, N.G.; Moore, P.J.; Gupta, A.S.; Thomsen, M.S.; Wernberg, T.; Smale, D.A. Biological impacts of marine heatwaves. Ann. Rev. Mar. Sci. 2023, 15, 119–145. [Google Scholar] [CrossRef] [PubMed]
- Zeller, D.; Palomares, M.L.D.; Pauly, D. Global fisheries science documents human impacts on oceans: The Sea Around Us serves civil society in the twenty-first century. Ann. Rev. Mar. Sci. 2023, 15, 147–165. [Google Scholar] [CrossRef]
- Georgian, S.; Hameed, S.; Morgan, L.; Amon, D.J.; Sumaila, U.R.; Johns, D.; Ripple, W.J. Scientists’ warning of an imperiled ocean. Biol. Cons. 2022, 272, 109595. [Google Scholar] [CrossRef]
- Lewison, R.L.; Crowder, L.B.; Wallace, B.P.; Moore, J.E.; Cox, T.; Zydelis, R.; McDonald, S.; DiMatteo, A.; Dunn, D.C.; Kot, C.Y.; et al. Global patterns of marine mammal, seabird, and sea turtle bycatch reveal taxa-specific and cumulative megafauna hotspots. Proc. Natl. Acad. Sci. USA 2014, 111, 5271–5276. [Google Scholar] [CrossRef]
- O’Hara, C.C.; Frazier, M.; Halpern, B.S. At-risk marine biodiversity faces extensive, expanding, and intensifying human impacts. Science 2021, 372, 84–87. [Google Scholar] [CrossRef]
- Ramos, J.A.; Pereira, L. (Eds.) Seabird Biodiversity and Human Activities; CRC Press: Boca Raton, FL, USA, 2022; p. 270. [Google Scholar]
- Mente, E.; Deguara, S.; Santos, M.B.; Houlihan, D. White muscle free amino acid concentrations following feeding a maize gluten dietary protein in Atlantic salmon (Salmo salar L.). Aquaculture 2003, 225, 133–147. [Google Scholar] [CrossRef]
- Espe, M.; Lemme, A.; Petri, A.; El-Mowafi, A. Can Atlantic salmon (Salmo salar) grow on diets devoid of fish meal? Aquaculture 2006, 255, 255–262. [Google Scholar] [CrossRef]
- Torstensen, B.; Espe, M.; Sanden, M.; Stubhaug, I.; Waagbø, R.; Hemre, G.-I.; Olsvik, P. Novel production of Atlantic salmon (Salmo salar) protein based on combined replacement of fish meal and fish oil with plant meal and vegetable oil blends. Aquaculture 2008, 285, 193–200. [Google Scholar] [CrossRef]
- Øverland, M.; Sørensen, M.; Storebakken, T.; Penn, M.; Krogdahl, Å.; Skrede, A. Pea protein concentrate substituting fish meal or soybean meal in diets for Atlantic salmon (Salmo salar)—Effect on growth performance, nutrient digestibility, carcass composition, gut health, and physical feed quality. Aquaculture 2009, 288, 305–311. [Google Scholar] [CrossRef]
- Burr, G.S.; Wolters, W.R.; Barrows, F.T.; Hardy, R.W. Replacing fishmeal with blends of alternative proteins on growth performance of rainbow trout (Oncorhynchus mykiss), and early or late stage juvenile Atlantic salmon (Salmo salar). Aquaculture 2012, 334–337, 110–116. [Google Scholar] [CrossRef]
- Davidson, J.; Barrows, F.T.; Kenney, P.B.; Good, C.; Schroyer, K.; Summerfelt, S.T. Effects of feeding a fishmeal-free versus a fishmeal-based diet on post-smolt Atlantic salmon Salmo salar performance, water quality, and waste production in recirculation aquaculture systems. Aquacult. Eng. 2016, 74, 38–51. [Google Scholar] [CrossRef]
- Metochis, C.P.; Crampton, V.O.; Ruohonen, K.; El-Mowafi, A.; Bell, J.G.; Adams, A.; Thompson, K.D. Effects of marine protein-, marine oil- and marinefree diets on the growth performance and innate immune responses of Atlantic salmon (Salmo salar, L. ) post-smolts. Aquacult. Res. 2017, 48, 2495–2515. [Google Scholar] [CrossRef]
- Egerton, S.; Wan, A.; Murphy, K.; Ahern, G.; Sugrue, I.; Busca, K.; Egan, F.; Muller, J.; Whooley, J.; McGinniyy, P.; et al. Replacing fishmeal with plant protein in Atlantic salmon (Salmo salar) diets by supplementation with fish protein hydrolysate. Sci. Rep. 2020, 10, 4194. [Google Scholar] [CrossRef]
- Mikołajczak, Z.; Mazurkiewicz, J.; Rawski, M.; Kierończyk, B.; Józefiak, A.; Świątkiewicz, S.; Józefiak, D. Black soldier fly full-fat meal in Atlantic salmon nutrition—Part B: Effects on growth performance, feed utilization, selected nutriphysiological traits and production sustainability in pre-smolts. Ann. Anim. Sci. 2023, 23, 239–251. [Google Scholar] [CrossRef]
- Torstensen, B.E.; Bell, J.G.; Sargent, J.R.; Rosenlund, G.; Henderson, R.J.; Graff, I.E.; Lie, Ø.; Tocher, D.R. Tailoring of Atlantic salmon (Salmo salar L.) flesh lipid composition and sensory quality by replacing fish oil with a vegetable oil blend. J. Agric. Food Chem. 2005, 53, 10166–10178. [Google Scholar] [CrossRef]
- Jordal, A.-E.O.; Lie, Ø.; Torstensen, B.E. Complete replacement of dietary fish oil with a vegetable oil blend affect liver lipid and plasma lipoprotein levels in Atlantic salmon (Salmo salar L.). Aquacult. Nutr. 2007, 13, 114–130. [Google Scholar] [CrossRef]
- Schmidt, V.; Amaral-Zettler, L.; Davidson, J.; Summerfelt, S.; Good, C. The influence of fishmeal-free diets on microbial communities in Atlantic salmon Salmo salar recirculation aquaculture systems. Appl. Environ. Microbiol. 2016, 82, 4470–4481. [Google Scholar] [CrossRef] [PubMed]
- Belghit, I.; Liland, N.S.; Gjesdal, P.; Biancarosa, I.; Menchetti, E.; Li, Y.; Waagbø, R.; Krogdahl, Å.; Lock, E.-J. Black soldier fly larvae meal can replace fish meal in diets of sea-water phase Atlantic salmon (Salmo salar). Aquaculture 2019, 503, 609–619. [Google Scholar] [CrossRef]
- Tibbetts, S.M.; Scaife, M.A.; Armenta, R.E. Apparent digestibility of proximate nutrients, energy and fatty acids in nutritionally-balanced diets with partial or complete replacement of dietary fish oil with microbial oil from a novel Schizochytrium sp. (T18) by juvenile Atlantic salmon (Salmo salar L.). Aquaculture 2020, 520, 735003. [Google Scholar]
- Santigosa, E.; Olsen, R.E.; Madaro, A.; Trichet, V.V.; Carr, I. Algal oil gives control of long-chain omega-3 levels in full-cycle production of Atlantic salmon, without detriment to zootechnical performance and sensory characteristics. J. World Aquacult. Soc. 2023, 2023, 1–21. [Google Scholar] [CrossRef]
- Wei, M.; Parrish, C.C.; Guerra, N.I.; Armenta, R.E.; Colombo, S.M. Extracted microbial oil from a novel Schizochytrium sp. (T18) as a sustainable high DHA source for Atlantic salmon feed: Impacts on growth and tissue lipids. Aquaculture 2021, 534, 736249. [Google Scholar] [CrossRef]
- Bolivar, R.B.; Vera Cruz, E.M.; Jimenez, E.B.T.; Sayco, R.M.V.; Argueza, R.L.B.; Ferket, P.R.; Stark, C.R.; Malheiros, R.; Ayoola, A.A.; Johnstone, W.M., III; et al. Feeding Reduction Strategies and Alternative Feeds to Reduce Production Costs of Tilapia Culture; Tech Rep Invest 20007–2009; AquaFish CRSP, Oregon State University: Corvallis, OR, USA, 2010. [Google Scholar]
- Skiba, S.; Médale, F.; Kaushik, S.; Lemarié, S.; Gaunand, A. Replacement of Marine Ingredients by Plant Products in Fish Diets; [Technical Report] Inconnu. 2015, p. 25; hal-01901445. Available online: hal.science/hal-01901445/document (accessed on 10 November 2023).
- Savonitto, G.; Barkan, R.; Harpaz, S.; Neori, A.; Chernova, H.; Terlizzi, A.; Guttman, L. Fishmeal replacement by periphyton reduces the fish in fish out ratio and alimentation cost in gilthead sea bream Sparus aurata. Sci. Rep. 2021, 11, 20990. [Google Scholar] [CrossRef]
- Risius, A.; Janssen, M.; Hamm, U. Consumer preferences for sustainable aquaculture products: Evidence from in-depth interviews, think aloud protocols and choice experiments. Appetite 2017, 113, 246–254. [Google Scholar] [CrossRef]
- Zander, K.; Feucht, Y. Consumers’ WTP for sustainable seafood made in Europe. J. Int. Food Agribus. Mark 2018, 30, 251–275. [Google Scholar] [CrossRef]
- Hynes, S.; Ravagnan, E.; Gjerstad, B. Do concerns for the environmental credentials of salmon aquaculture translate into WTP a price premium for sustainably farmed fish? A contingent valuation study in Ireland and Norway. Aquacult. Int. 2019, 27, 1709–1723. [Google Scholar] [CrossRef]
- Asche, F.; Larsen, T.; Smith, M.; Sogn-Grundvåg, G.; Young, J. Pricing of eco-labels with retailer heterogeneity. Food Policy 2015, 53, 82–93. [Google Scholar] [CrossRef]
- Ankamah-Yeboah, I.; Nielsen, M.; Nielsen, R. Price premium of organic salmon in Danish retail sale. Ecol. Econ. 2016, 122, 54–60. [Google Scholar] [CrossRef]
- Olesen, I.; Alfnes, F.; Rora, M.B.; Kolstad, K. Eliciting consumers’ WTP for organic and welfare-labelled salmon in a non-hypothetical choice experiment. Livest. Sci. 2010, 127, 218–226. [Google Scholar] [CrossRef]
- Ferfolja, M.; Cerjak, M.; Matulić, D.; Tomić Maksan, M. Consumer knowledge and perception about fresh fish from organic farming in Croatia. Cro. J. Fish 2022, 80, 7–16. [Google Scholar] [CrossRef]
- Chen, X.; Alfnes, F.; Rickertsen, K. Consumer preferences, ecolabels, and effects of negative environmental information. AgBioForum 2015, 18, 327–336. [Google Scholar]
- Hardy, R.W.; Scott, T.M.; Harrell, L.W. Replacement of herring oil with menhaden oil, soybean oil, or tallow in the diets of Atlantic salmon raised in marine net-pens. Aquaculture 1987, 65, 267–277. [Google Scholar] [CrossRef]
- Davidson, J.; Kenney, P.B.; Barrows, F.T.; Good, C.; Summerfelt, S.T. Fillet quality and processing attributes of postsmolt Atlantic salmon, Salmo salar, fed a fishmeal-free diet and a fishmeal-based diet in recirculation aquaculture systems. J. World Aquacult. Soc. 2018, 49, 183–196. [Google Scholar] [CrossRef]
- Kousoulaki, K.; Østbye, T.K.K.; Krasnov, A.; Torgersen, J.S.; Mørkøre, T.; Sweetman, J. Metabolism, health and fillet nutritional quality in Atlantic salmon (Salmo salar) fed diets containing n-3-rich microalgae. J. Nutr. Sci. 2015, 4, e24. [Google Scholar] [CrossRef]
- Kousoulaki, K.; Gerd, M.B.; Mørkøre, T.; Krasnov, A.; Baeverfjord, G.; Ytrestøyl, T.; Carlehög, M.; Sweetman, J.; Ruyter, B. Microalgal Schizochytrium limacinum biomass improves growth and filet quality when used long-term as a replacement for fish oil, in modern salmon diets. Front. Mar. Sci. 2020, 7, 57. [Google Scholar] [CrossRef]
- Zatti, K.M.; Ceballos, M.J.; Vega, V.V.; Denstadli, V. Full replacement of fish oil with algae oil in farmed Atlantic salmon (Salmo salar)—Debottlenecking omega 3. Aquaculture 2023, 574, 739653. [Google Scholar] [CrossRef]
- Einen, O.; Mørkøre, T.; Rørå, A.M.B.; Thomassen, M.S. Feed ration prior to slaughter—A potential tool for managing product quality of Atlantic salmon (Salmo salar). Aquaculture 1999, 178, 149–169. [Google Scholar] [CrossRef]
- Frederick, C.; Brady, D.C.; Bricknell, I. Landing strips: Model development for estimating body surface area of farmed Atlantic salmon (Salmo salar). Aquaculture 2017, 473, 299–302. [Google Scholar] [CrossRef]
- MacLean, A. Compensatory Growth, Life-History Decisions and Welfare of Farmed Atlantic Salmon (Salmo salar L.) parr. Ph.D. Thesis, Division of Environmental and Evolutionary Biology, Institute of Biomedical and Life Sciences, University of Glasgow, Glasgow, Scotland, 1999; 165p. [Google Scholar]
- Bass, N.; Wall, T. A Standard Procedure for the Field Monitoring of Cataracts in Farmed Atlantic Salmon and Other Species; BIM, Irish Sea Fisheries Board, Dun Laoghaire, Co.: Dublin, Ireland, 2008; p. 3. [Google Scholar]
- Kindschi, G.A.; Shaw, H.T.; Bruhn, D.S. Effect of diet on performance, fin quality and dorsal skin lesions in steelhead. J. Appl. Aquacult. 1991, 1, 113–120. [Google Scholar] [CrossRef]
- MacLean, A.; Metcalfe, N.B.; Mitchell, D. Alternative competitive strategies in juvenile Atlantic salmon (Salmo salar): Evidence from fin damage. Aquaculture 2000, 184, 291–302. [Google Scholar] [CrossRef]
- Browning, H. Improving welfare assessment in aquaculture. Front. Vet Sci Sec. Anim. Behav. Welf. 2023, 10, 1060720. [Google Scholar] [CrossRef] [PubMed]
- Savoca, M.S.; Brodie, S.; Welch, H.; Hoover, A.; Benaka, L.R.; Bograd, S.J.; Hazen, E.L. Comprehensive bycatch assessment in US fisheries for prioritizing management. Nat Sust. 2020, 3, 472–480. [Google Scholar] [CrossRef]
- Huntington, T. Best Practice Framework for the Management of Aquaculture gear; Global Ghost Gear Initiative—Ocean Conservancy: Washington, DC, USA, 2021; p. 81. [Google Scholar]
- HRAT. Does It Do What It Says on the Tin? Fisheries and Aquaculture Certification, Standards and Ratings Ecosystem: An Independent Review; Human Rights at Sea: Havant, UK, 2023; p. 33. [Google Scholar]
- US Department of Labor-International Bureau of Labor Affairs. List of Goods Produced by Child Labor or Forced Labor. 2020. Available online: https://www.dol.gov/agencies/ilab/reports/child-labor/list-of-goods (accessed on 10 November 2023).
- Nakamura, K.; Ota, Y.; Blaha, F. A practical take on the duty to uphold human rights in seafood workplaces. Mar. Policy 2022, 135, 104844. [Google Scholar] [CrossRef]
- Gephart, J.A.; Henriksson, P.J.G.; Parker, R.W.R.; Shepon, A.; Gorospe, K.D.; Bergman, K.; Eshel, G.; Golden, C.D.; Halpern, B.S.; Hornborg, S.; et al. Environmental performance of blue foods. Nature 2021, 507, 360–366. [Google Scholar] [CrossRef]
- Ersdal, C.; Midtlyng, P.J.; Jarp, J. An epidemiological study of cataracts in seawater farmed Atlantic salmon Salmo salar. Dis. Aquat. Org. 2001, 45, 229–236. [Google Scholar] [CrossRef]
- Bjerkås, E.; Holst, J.C.; Bjerkås, I. Cataract in farmed and wild Atlantic salmon (Salmo salar L.). Anim. Eye Res. 2004, 23, 3–13. [Google Scholar]
- Sissener, N.H.; Hamre, K.; Fjelldal, P.G.; Philip, A.J.P.; Espe, M.; Miao, L.; Høglund, E.; Sørensen, C.; Skjærven, K.H.; Holen, E.; et al. Can improved nutrition for Atlantic salmon in freshwater increase fish robustness, survival and growth after seawater transfer? Aquaculture 2021, 542, 736852. [Google Scholar] [CrossRef]
- Tröße, C. Mechanisms of Cataract Development in Adult Atlantic Salmon Growers Relative to Dietary Histidine and Plant Feed Ingredients. Ph.D. Thesis, University of Bergen, Bergen, Norway, 2010. [Google Scholar]
- Remø, S.C.; Hevroy, E.M.; Olsvik, P.A.; Fontanillas, R.; Breck, O.; Waagbo, R. Dietary histidine requirement to reduce the risk and severity of cataracts is higher than the requirement for growth in Atlantic salmon smolts, independently of the dietary lipid source. Br. J. Nutr. 2014, 111, 1759–1772. [Google Scholar] [CrossRef]
- Cañon Jones, H.A.; Noble, C.; Damsgård, B.; Pearce, G.P. Social network analysis of the behavioural interactions that in uence the development of n damage in Atlantic salmon parr (Salmo salar) held at different stocking densities. Appl. Anim. Behv. Sci. 2011, 113, 117–126. [Google Scholar] [CrossRef]
- Soderberg, R.W.; Meade, J.W. Effects of rearing density on growth, survival, and fin condition of Atlantic salmon. Prog. FishCult. 1987, 49, 280–283. [Google Scholar] [CrossRef]
- Ellis, T.; Oidtmann, B.; St-Hilaire, S.; Turnbull, J.F.; North, B.P.; MacIntyre, C.M.; Nikolaidis, J.; Hoyle, I.; Kestin, S.C.; Knowles, T.G. Fin erosion in farmed fish. In Fish Welfare; Branson, E.J., Ed.; Blackwell Publishing Ltd.: Oxford, UK, 2008; pp. 121–149, 300. [Google Scholar]
- Larsen, M.H.; Nemitz, A.; Steinheuer, M.; Lysdale, J.; Thomassen, S.T.; Holdensgaard, G. Effects of Hatchery Feeding Practices on Fin and Operculum Condition of Juvenile Atlantic Salmon Salmo salar. 2018, p. 22. Available online: https://danmarksvildlaks.dk/wp-content/uploads/2018/04/Technical-report_feeding_opercula.pdf (accessed on 3 November 2023).
- Ellis, M.A. Fin Damage in Juvenile Atlantic Salmon: Farm and Experimental, Causes and Consequences. Ph.D. Thesis, University of Stirling, Institute of Aquaculture, Stirling, Scotland, 2019; p. 265. [Google Scholar]
- Timmerhaus, G.; Lazado, C.C.; Cabillon, N.A.R.; Reiten, B.K.M.; Johansen, L.-H. The optimum velocity for Atlantic salmon post-smolts in RAS is a compromise between muscle growth and fish welfare. Aquaculture 2021, 532, 7360765. [Google Scholar] [CrossRef]
- Lemm, C.A.; Rottiers, D.S.; Dropkin, D.S.; Dennison, B.A. Growth, composition, and fin quality of Atlantic Salmon fed different diets at seasonal temperatures in a laboratory and hatchery. US Fish Wildl. Serv. Biol. Rep. 1988, 88, 1–12. [Google Scholar]
- Bourne, M. Texture profile analysis. Food Technol. 1978, 32, 62–66. [Google Scholar]
- Latremouille, D.N. Fin erosion in aquaculture and natural environments. Rev. Fish Sci. 2003, 11, 315–335. [Google Scholar] [CrossRef]
- Cañon Jones, H.A.; Noble, C.; Damsgård, B.; Pearce, G.P. Evaluating the effects of a short-term feed restriction period on the behaviour and welfare of Atlantic salmon (Salmo salar) parr using social network analysis and fin damage. J. World Aquacult. Soc. 2017, 48, 35–45. [Google Scholar] [CrossRef]
- Hoyle, I.; Oidtmann, B.; Ellis, T.; Turnbull, J.; North, B.; Nikolaidis, J.; Knowles, T.G. A validated macroscopic key to assess fin damage in farmed rainbow trout (Onchorynchus mykiss). Aquaculture 2007, 270, 142–148. [Google Scholar] [CrossRef]
- Noble, C.; Kadri, S.; Mitchell, D.F.; Huntingford, F.A. The effect of feed regime on the growth and behaviour of 1+ Atlantic salmon post-smolts (Salmo salar L.) in semi-commercial sea cages. Aquacul. Res. 2007, 38, 1686–1691. [Google Scholar] [CrossRef]
- Noble, C.; Kadri, S.; Mitchell, D.F.; Huntingford, F.A. Growth, production and fin damage in cage-held 0+ Atlantic salmon pre-smolts (Salmo salar L.) fed either (a) on-demand, or (b) to a fixed satiation–restriction regime: Data from a commercial farm. Aquaculture 2008, 275, 163–168. [Google Scholar] [CrossRef]
- Amoroso, G.; Nguyen, C.D.H.; Vo, T.T.M.; Ventura, T.; Elizur, A. Understanding Flesh Colour Variation in Atlantic Salmon: Molecular Mechanisms and Genetic Effect. 2020, p. 201. Available online: https://www.frdc.com.au/project/2014-248 (accessed on 2 November 2023).
- Sylvia, G.; Morrisey, M.T.; Graham, T.; Garcia, S. Organoleptic qualities of farmed and wild salmon. J. Aquat. Food Prod. Technol. 1995, 4, 51–64. [Google Scholar] [CrossRef]
- Storebakken, T.; Foss, P.; Schiedt, K.; Austreng, E.; Liaaen-Jensen, S.; Mans, U. Carotenoids in diets for salmonids. IV. Pigmentation of Atlantic salmon with astaxanthin, astaxanthin dipalmitate and canthaxanthin. Aquaculture 1987, 65, 279–292. [Google Scholar] [CrossRef]
- Bjerkeng, B.; Refstie, S.; Fjalestad, K.T.; Storebakken, T.; Rodbotten, M.; Roem, A.J. Quality parameters of the flesh of Atlantic salmon (Salmo salar) as affected by dietary fat content and full-fat soybean meal as a partial substitute for fish meal in the diet. Aquaculture 1997, 157, 297–309. [Google Scholar] [CrossRef]
- Buttle, L.G.; Crampton, V.O.; Williams, P.D. The effect of feed pigment type on flesh pigment deposition and colour in farmed Atlantic salmon, Salmo salar L. Aquacult. Res. 2001, 32, 103–111. [Google Scholar] [CrossRef]
- Young, A.; Morris, P.C.; Huntingford, F.A.; Sinnott, R. The effects of diet, feeding regime and catch-up growth on fl esh quality attributes of large (1 + sea winter) Atlantic salmon, Salmo salar. Aquaculture 2005, 248, 59–73. [Google Scholar] [CrossRef]
- Sinnott, R. Carcass quality monitoring at the farm and factory. In Farmed Fish Quality; Kestin, S.C., Warrriss, P.D., Eds.; Blackwell Science: Osney Mead, UK, 2001; pp. 318–334, 430. [Google Scholar]
- de Francesco, M.; Parisi, G.; Medale, F.; Lupi, P.; Kaushik, S.J.; Poli, B.M. Effect of long-term feeding with a plant protein mixture based diet on growth and body/fillet quality traits of large rainbow trout Oncorhynchus mykiss. Aquaculture 2004, 236, 413–429. [Google Scholar] [CrossRef]
- D’Souza, N.; Skonberg, D.I.; Stone, D.A.J.; Brown, P.B. Effect of soybean meal-based diets on the product quality of rainbow trout fillets. J. Food Sci. 2006, 71, S337–S342. [Google Scholar] [CrossRef]
- Johnsen, C.A.; Hagen, Ø.; Bendiksen, E.Å. Long-term effects of high-energy, low-fishmeal feeds on growth and flesh characteristics of Atlantic salmon (Salmo salar L.). Aquaculture 2011, 312, 109–116. [Google Scholar] [CrossRef]
- Liu, K.K.M.; Barrows, F.T.; Hardy, R.W.; Dong, F.M. Body composition, growth performance, and product quality of rainbow trout (Oncorhynchus mykiss) fed diets containing poultry fat, soybean/corn lecithin, or menhaden oil. Aquaculture 2004, 238, 309–328. [Google Scholar] [CrossRef]
- Kwan, D.; Dai, L.; Liu, D.; Liu, H.; Du, W. Efficient biodiesel conversion from microalgae oil of Schizochytrium sp. Catalysts 2019, 9, 341. [Google Scholar] [CrossRef]
- Casas, C.; Martinez, O.; Guillen, M.D.; Pin, C.; Salmeron, J. Textural properties of raw Atlantic salmon (Salmo salar) at three points along the fillet, determined by different methods. Food Control 2006, 17, 511–515. [Google Scholar] [CrossRef]
- Sigurgisladottir, S.; Hafsteinsson, H.; Jonsson, A.; Lie, Ø.; Nortvedt, R.; Thomassen, M.S.; Torrissen, O. Textural properties of raw salmon fillets as related to sampling method. J. Food Sci. 1999, 64, 99–104. [Google Scholar] [CrossRef]
- Jonsson, A.; Sigurgisladottir, S.; Hafsteinson, H.; Kristbergsson, K. Textural properties of raw Atlantic salmon (Salmo salar) fillets measured by different methods in comparison to expressible moisture. Aquacult. Nutr. 2001, 7, 81–89. [Google Scholar] [CrossRef]
- Merkin, G.V.; Stein, L.H.; Pittman, K.; Nortvedt, R. The effect of stunning methods and season on muscle texture hardness in Atlantic salmon (Salmo salar L.). J. Food Sci. 2014, 79, E1137–E1141. [Google Scholar] [CrossRef]
- Larsson, T.; Koppang, E.O.; Espe, M.; Terjesen, B.F.; Krasnov, A.; Moreno, H.M.; Rørvik, K.-A.; Thomassen, M.; Mørkøre, T. Fillet quality and health of Atlantic salmon (Salmo salar L.) fed a diet supplemented with glutamate. Aquaculture 2014, 426, 288–295. [Google Scholar] [CrossRef]
- Ostbye, T.K.K.; Ruyter, B.; Standal, I.B.; Stien, L.H. Functional amino acids stimulate muscle development and improve fillet texture of Atlantic salmon. Aquacult. Nutr. 2018, 24, 14–26. [Google Scholar] [CrossRef]
- Azm, F.R.A.; Kong, F.; Wang, X.; Zhu, W.; Yu, H.; Long, X.; Tan, Q. The interaction of dried distillers grains with solubles (DDGS) type and level on growth performance, health, texture, and muscle-related gene expression in grass carp (Ctenopharyngodon idellus). Front. Nutr. Sec. Nutr. Sustain. Diets 2022, 28, 832651. [Google Scholar] [CrossRef]
- Hu, Y.; Hu, Y.; Wu, T.; Chu, W. Effects of high dietary levels of cottonseed meal and rapeseed meal on growth performance, muscle texture, and expression of muscle-related genes in grass carp. N. Am. J. Aquacult. 2019, 81, 235–241. [Google Scholar] [CrossRef]
- Kong, F.; Azm, F.R.A.; Wang, X.; Zhu, Y.; Yu, H.; Yao, J.; Luo, Z.; Tan, Q. Effects of replacement of dietary cottonseed meal by distiller’s dried grains with solubles on growth performance, muscle texture, health and expression of muscle-related genes in grass carp (Ctenopharyngodon idellus). Aquacult Nutr. 2021, 27, 13266. [Google Scholar] [CrossRef]
- Wu, F.; Tian, J.; Yu, L.; Wen, H.; Jiang, M.; Lu, X. Effects of dietary rapeseed meal levels on growth performance, biochemical indices and flesh quality of juvenile genetically improved farmed tilapia. Aquacult. Rep. 2021, 20, 100679. [Google Scholar] [CrossRef]
- Mørkøre, T.; Moreno, H.M.; Borderías, J.; Larsson, T.; Hellberg, H.; Hatlen, B.; Romarheim, O.H.; Ruyter, B.; Lazado, C.C.; Jiménez-Guerrero, R.; et al. Dietary inclusion of Antarctic krill meal during the finishing feed period improves health and fillet quality of Atlantic salmon (Salmo salar L.). Br. J. Nutr. 2020, 124, 418–431. [Google Scholar] [CrossRef] [PubMed]
- Sigurgisladottir, S.; Torrissen, O.; Lie, Ø.; Thomassen, M.; Hafsteinsson, H. Salmon quality: Methods to determine the quality parameters. Rev. Fish Sci. 1997, 5, 223–252. [Google Scholar] [CrossRef]
- Zhu, F.; Peng, J.; Gao, J.; Zhao, Y.; Yu, K.; He, Y. Determination and visualization of fat contents in salmon fillets based on visible and near-infrared hyperspectral imagery. Trans. Chin. Soc. Ag. Eng. 2014, 30, 314–323. [Google Scholar]
- Einen, O.; Waagan, B.; Thomassen, M.S. Starvation prior to slaughter in Atlantic salmon (Salmo salar) I. Effects on weight loss, body shape, slaughter- and fillet-yield, proximate and fatty acid composition. Aquaculture 1998, 166, 85–104. [Google Scholar] [CrossRef]
- Nøstbakken, O.J.; Reksten, A.M.; Hannisdal, R.; Dahl, L.; Duinker, A. Sampling of Atlantic salmon using the Norwegian Quality cut (NQC) vs. whole fillet; differences in contaminant and nutrient contents. Food Chem. 2023, 418, 136056. [Google Scholar] [CrossRef]
- Ares, G.; Varela, P. Trained vs. consumer panels for analytical testing: Fueling a long lasting debate in the field. Food Qual. Prefer 2017, 61, 79–86. [Google Scholar] [CrossRef]
- Calanche, J.B.; Beltrán, J.B.; Arias, A.J.H. Aquaculture and sensometrics: The need to evaluate sensory attributes and the consumers’ preferences. Rev. Aquacult. 2020, 12, 805–821. [Google Scholar] [CrossRef]
- Chambers, E.I.V.; Smith, E.A. Effects of testing experience on performance of trained sensory panelists. J. Sens. Stud. 1993, 8, 155–166. [Google Scholar]
- Elgaard, L.; Jensen, S.; Mielby, L.A.; Byrne, D.V. Performance of beer sensory panels: A comparison of experience level, product knowledge, and responsiveness to feedback calibration. J. Sens. Stud. 2019, 34, e12540. [Google Scholar] [CrossRef]
- Roberts, A.K.; Vickers, Z.M. A comparison of trained and untrained judges, evaluation of sensory attribute intensities and liking of Cheddar cheeses. J. Sens. Stud. 1994, 9, 1–20. [Google Scholar] [CrossRef]
- Carlucci, D.; Nocella, G.; De Devitiis, B.; Viscecchia, R.; Bimbo, F.; Nardone, G. Consumer purchasing behaviour towards fish and seafood products. Patterns and insights from a sample of international studies. Appetite 2015, 84, 212–227. [Google Scholar] [CrossRef] [PubMed]
- Altintzoglou, T.; Cordeiro, C.M.; Honkanen, P.; Onozaka, Y. “It gives me peace of mind”. A new perspective on the identification of quality cues on salmon fillet products in Japan and the USA. Aquaculture 2022, 554, 728112. [Google Scholar] [CrossRef]
- Tomić, M.; Kovačićek, T.; Matulić, D. Attitudes as basis for segmenting Croatian fresh fish consumers. New Medit. 2016, 4, 63–71. [Google Scholar]
- Verbeke, W.; Vackier, I. Individual determinants of fish consumption: Application of the theory of planned behaviour. Appetite 2005, 44, 67–82. [Google Scholar] [CrossRef]
- Al-Saghir, S.; Thurner, K.; Wagner, K.-H.; Frisch, G.; Luf, W.; Razzazi-Fazell, E.; Elmadfa, I. Effects of different cooking procedures on lipid quality and cholesterol oxidation of farmed salmon fish (Salmo salar). J. Agric. Food Chem. 2004, 52, 5290–5296. [Google Scholar] [CrossRef]
- Gladyshev, M.I.; Sushchik, N.N.; Gubanenko, G.A.; Demirchieva, S.M.; Kalachova, G.S. Effect of way of cooking on content of essential polyunsaturated fatty acids in muscle tissue of humpback salmon (Oncorhynchus gorbuscha). Food Chem. 2006, 96, 446–451. [Google Scholar] [CrossRef]
- Larsen, D.; Quek, S.Y.; Eyres, L. Effect of cooking method on the fatty acid profile of New Zealand King Salmon (Oncorhynchus tshawytscha). Food Chem. 2010, 119, 785–790. [Google Scholar] [CrossRef]
- Shearer, K.D. Factors affecting the proximate composition of cultured fishes with emphasis on salmonids. Aquaculture 1994, 119, 63–88. [Google Scholar] [CrossRef]
- Einen, O.; Skrede, G. Quality characteristics in raw and smoked fillets of Atlantic salmon, Salmo salar, fed high-energy diets. Aquacult. Nutr. 1998, 4, 99–108. [Google Scholar] [CrossRef]
- Turchini, G.M.; Francis, D.S.; Du, Z.-Y.; Olsen, R.E.; Ringø, E.; Tocher, D.R. The lipids. In Fish Nutrition; Hardy, R.W., Kaushik, S., Eds.; Academic Press: San Diego, CA, USA, 2022; pp. 303–467. [Google Scholar]
- Wang, Z.; Qiao, F.; Zhang, W.-B.; Parisi, G.; Du, Z.-Y.; Zhang, M.-L. The flesh texture of teleost fish: Characteristics and interventional strategies. Rev. Aquac. 2023, 16, 12849. [Google Scholar] [CrossRef]
- Drewnowski, A. Taste preferences and food intake. Annu. Rev. Nutr. 1997, 17, 237–253. [Google Scholar] [CrossRef] [PubMed]
- Methven, L.; Allen, V.J.; Withers, C.A.; Gosney, M.A. Ageing and taste. Proc. Nutr. Soc. 2012, 71, 556–565. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, T.; Annear, M.J.; Ikebe, K.; Maeda, Y. Taste-related sensations in old age. J. Oral Rehabil. 2017, 44, 626–635. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-J.; Liang, K.-L.; Lin, W.-J.; Chen, C.-Y.; Jiang, R.S. Influence of age and sex on taste function of healthy subjects. PLoS ONE 2020, 15, e0227014. [Google Scholar] [CrossRef] [PubMed]
- Gidhini, S.; Varrà, M.O.; Zanardi, E. Approaching authenticity issues in fish and seafood products by qualitative spectroscopy and chemometrics. Molecules 2019, 24, 1812. [Google Scholar] [CrossRef]
- Hikima, J.I.; Ando, M.; Hamaguchi, H.O.; Sakai, M.; Maita, M.; Yazawa, K.; Takeyama, H.; Aoki, T. On-site direct detection of astaxanthin from salmon fillet using Raman spectroscopy. Mar. Biotech. 2017, 19, 157–163. [Google Scholar] [CrossRef]
- Fiorino, G.M.; Losito, I.; De Angelis, E.; Arlorio, M.; Logrieco, A.F.; Monaci, L. Assessing fish authenticity by direct analysis in real time-high resolution mass spectrometry and multivariate analysis: Discrimination between wild-type and farmed salmon. Food Res. Int. 2019, 116, 1258–1265. [Google Scholar] [CrossRef]
- Chen, Z.; Wu, T.; Xiang, C.; Xu, X.; Tian, X. Rapid identification of rainbow trout adulteration in Atlantic salmon by Raman spectroscopy combined with machine learning. Molecules 2019, 24, 2851. [Google Scholar] [CrossRef]
- Han, C.; Dong, S.L.; Li, L.; Gao, Q. Efficacy of using stable isotopescoupled with chemometrics to differentiate the production method and geographical origin of farmed salmonids. Food Chem. 2021, 364, 130364. [Google Scholar] [CrossRef] [PubMed]
- Han, C.; Li, L.; Dong, X.; Gao, Q.; Dong, S. Current progress in the authentication of fishery and aquatic products using multi-element and stable isotope analyses combined with chemometrics. Rev. Aquacult. 2022, 14, 2023–2037. [Google Scholar] [CrossRef]
- Wang, Y.V.; Wan, A.H.L.; Lock, E.-J.; Andersen, N.; Winter-Schuh, C.; Larsen, T. Know your fish: A novel compound-specific isotope approach for tracing wild and farmed salmon. Food Chem. 2018, 256, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Bi, H. Super-fast seafood authenticity analysis by One-step pretreatment and comparison of mass spectral patterns. Food Control 2021, 123, 107751. [Google Scholar] [CrossRef]
- McLean, E.; Fredriksen, L.; Alfrey, K.; Tlusty, M.F.; Barrows, F.T. Growth, integrity, and consumer acceptance of largemouth bass, Micropterus salmoides (Lacépède, 1802), fed marine resource-free diets. Int. J. Fish. Aquat. Stud. 2020, 8, 365–369. [Google Scholar] [CrossRef]

| Female | Male | ||
|---|---|---|---|
| Ethnicity | Caucasian | 4 | 15 |
| African/Afro-American | 2 | 2 | |
| (Eur)Asian | 7 | 1 | |
| Latino/Hispanic | 3 | ||
| Age group | 18–24 | 2 | 1 |
| 25–34 | 2 | 6 | |
| 35–44 | 3 | 6 | |
| 45–54 | 2 | 2 | |
| 55–64 | 2 | 4 | |
| 65+ | 2 | 2 |
| Diet | C1 | P1 | P2 | P3 | A1 | A2 | A3 |
|---|---|---|---|---|---|---|---|
| Weight (g) | 603.13 ± 76.95 | 568.75 ± 47.49 | 610.63 ± 58.76 | 606.88 ± 67.03 | 613.75 ± 36.52 | 626.88 ± 61.76 | 605.63 ± 57.54 |
| Length (cm) | 36.81 ± 1.93 | 35.63 ± 0.79 | 36.81 ± 1.31 | 36.19 ± 1.10 | 36.88 ± 0.64 | 36.69 ± 1.25 | 36.56 ± 1.05 |
| K | 1.21 ± 0.05 | 1.26 ± 0.09 | 1.22 ± 0.08 | 1.28 ± 0.10 | 1.22 ± 0.03 | 1.27 ± 0.06 | 1.24 ± 0.04 |
| Surface area (cm2) | 689.16 ± 53.90 | 665.57 ± 34.21 | 694.88 ± 41.16 | 692.08 ± 46.55 | 697.48 ± 25.12 | 706.08 ± 42.67 | 691.45 ± 39.78 |
| Gutted wt | 550.00 ± 73.29 | 503.13 ± 42.25 | 547.50 ± 57.63 | 553.75 ± 60.58 | 552.50 ± 37.51 | 564.38 ± 56.28 | 545.63 ± 51.30 |
| HG wt (g) | 501.88 ± 66.44 | 455.50 ± 40.44 | 487.50 ± 54.31 | 500.00 ± 55.16 | 496.88 ± 33.48 | 509.38 ± 50.25 | 493.13 ± 48.10 |
| HG Yield (%) | 83.00 ± 1.51 a | 81.13 ± 1.13 b | 79.88 ± 1.64 b | 82.50 ± 0.76 a | 80.88 ± 0.84 b | 81.25 ± 1.28 a | 81.38 ± 0.74 a |
| Fillet wt (g) | 302.50 ± 43.01 | 273.75 ± 27.22 | 293.75 ± 42.74 | 311.25 ± 46.73 | 305 ± 40.00 | 316.25 ± 39.62 | 303.75 ± 36.23 |
| Fillet yield (%) | 50.19 ± 4.13 | 48.19 ± 3.52 | 48.02 ± 4.30 | 51.26 ± 4.96 | 49.58 ± 4.53 | 50.42 ± 3.39 | 50.08 ± 2.22 |
| Color back | 20.88 ± 1.13 a | 23.88 ± 0.84 b | 22.75 ± 1.17 a,b | 23.00 ± 1.07 b | 22.38 ± 1.19 a,b | 22.63 ± 1.41 a,b | 23.00 ± 1.77 b |
| Color belly | 20.38 ± 0.74 a | 22.88 ± 0.84 b | 21.75 ± 1.17 a,b | 22.00 ± 1.07 a,b | 21.38 ± 1.19 a,b | 21.63 ± 1.41 a,b | 22.00 ± 1.51 a,b |
| Color NQC | 20.88 ± 1.13 a | 23.88 ± 0.84 b | 22.63 ± 1.19 a,b | 23.00 ± 1.07 b | 22.38 ± 1.19 a,b | 22.50 ± 1.51 a,b | 23.00 ± 1.77 b |
| Diet | C1 | P1 | P2 | P3 | A1 | A2 | A3 |
|---|---|---|---|---|---|---|---|
| SL | 33.57 ± 17.9 a | 31.18 ± 10.0 b | 31.46 ± 11.7 a,b | 32.09 ± 12.6 a,b | 33.08 ± 11.3 a,b | 31.78 ± 17.0 a,b | 31.46 ± 11.73 b |
| RFW | 45.18 ± 3.49 | 42.44 ± 2.83 | 42.75 ± 1.67 | 43.69 ± 3.74 | 45.69 ± 3.14 | 43.69 ± 3.70 | 42.44 ± 2.47 |
| RFH | 33.88 ± 2.12 | 29.81 ± 3.77 | 30.81 ± 1.83 | 30.94 ± 4.01 | 33.63 ± 3.14 | 30.56 ± 4.25 | 30.63 ± 3.71 |
| RFI | 13.48 ± 1.08 | 13.60 ± 0.68 | 13.59 ± 0.50 | 13.64 ± 1.35 | 13.80 ± 0.63 | 13.73 ± 0.49 | 13.40 ± 0.71 |
| FW | 3.50 ± 2.69 | 1.63 ± 2.15 | 2.00 ± 1.69 | 1.81 ± 2.07 | 0.75 ± 1.49 | 2.81 ± 4.00 | 2.31 ± 2.74 |
| #F | 2.75 ± 2.25 | 0.88 ± 0.99 | 2.00 ± 2.39 | 1.00 ± 1.69 | 1.25 ± 3.15 | 2.00 ± 2.56 | 1.00 ± 1.31 |
| Diet | C1 | P1 | P2 | P3 | A1 | A2 | A3 |
|---|---|---|---|---|---|---|---|
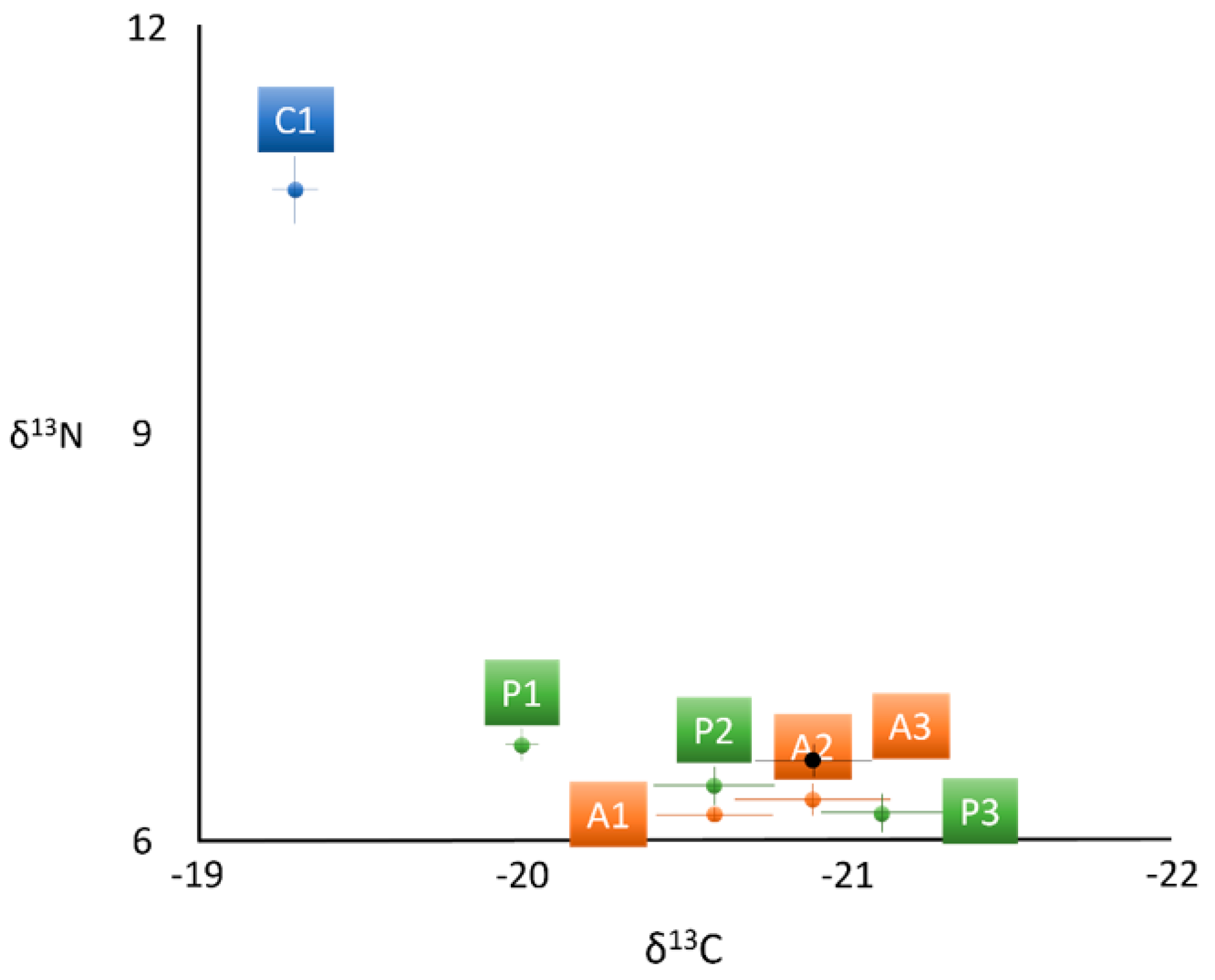
| δ13C | −19.3 ± 0.3 a | −20.0 ± 0.2 b | −20.6 ± 0.4 b | −21.1 ± 0.4 c | −20.6 ± 0.3 b | −20.9 ± 0.4 c | −20.9 ± 0.3 c |
| δ15N | 10.8 ± 0.4 a | 6.7 ± 0.2 b | 6.4 ± 0.3 b | 6.2 ± 0.3 b | 6.2 ± 0.1 b | 6.3 ± 0.2 b | 6.6 ± 0.2 b |
| δ13C: δ15N | −1.8 ± 0.1 a | −3.0 ± 0.1 b | −3.2 ± 0.2 b | −3.4 ± 0.2 b | −3.3 ± 0.1 b | −3.3 ± 0.1 b | −3.2 ± 0.1 b |
| Diet | Hardness | Cohesiveness | Springiness | Resilience |
|---|---|---|---|---|
| C1 | 638.0 ± 34.6 | 40.9 ± 1.4 | 99.9 ± 0.1 | 13.7 ± 0.7 |
| P1 | 610.5 ± 41.6 | 37.2 ± 1.2 | 99.9 ± 0.0 | 11.5 ± 0.5 |
| P2 | 608.5 ± 40.4 | 41.1 ± 1.5 | 100.2 ± 0.3 | 13.4 ± 1.3 |
| P3 | 536.0 ± 25.1 | 38.9 ± 1.5 | 100.0 ± 0.0 | 12.2 ± 0.8 |
| A1 | 633.4 ± 17.2 | 38.9 ± 0.8 | 100.6 ± 0.5 | 14.4 ± 1.3 |
| A2 | 552.8 ± 45.4 | 39.7 ± 1.8 | 100.1 ± 0.2 | 13.3 ± 0.8 |
| A3 | 537.8 ± 45.4 | 39.2 ± 1.4 | 101.6 ± 1.3 | 16.1 ± 2.7 |
| Diet | Hardness | Cohesiveness | Springiness | Resilience |
|---|---|---|---|---|
| C1 | 764.3 ± 133.5 | 38.3 ± 1.2 | 100.2 ± 0.3 b | 13.3 ± 1.0 a,b |
| P1 | 691.5 ± 62.8 | 35.6 ± 1.6 | 99.8 ± 0.0 b | 10.9 ± 0.5 b |
| P2 | 681.5 ± 63.6 | 38.3 ± 1.5 | 99.9 ± 0.0 b | 12.8 ± 0.9 a,b |
| P3 | 619.8 ± 73.9 | 36.2 ± 1.2 | 99.9 ± 0.0 b | 11.9 ± 0.7 b |
| A1 | 711.7 ± 41.1 | 36.4 ± 1.2 | 102.4 ± 1.0 a | 17.8 ± 2.6 a |
| A2 | 729.1 ± 63.2 | 37.0 ± 1.4 | 99.9 ± 0.0 b | 12.3 ± 0.6 b |
| A3 | 659.3 ± 38.5 | 36.1 ± 0.9 | 99.0 ± 0.0 b | 11.7 ± 0.6 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McLean, E.; Campbell, K.B.; Kuhn, D.D.; Tlusty, M.F.; Barrows, F.T. The Impact of Marine Resource-Free Diets on Quality Attributes of Atlantic Salmon. Fishes 2024, 9, 37. https://doi.org/10.3390/fishes9010037
McLean E, Campbell KB, Kuhn DD, Tlusty MF, Barrows FT. The Impact of Marine Resource-Free Diets on Quality Attributes of Atlantic Salmon. Fishes. 2024; 9(1):37. https://doi.org/10.3390/fishes9010037
Chicago/Turabian StyleMcLean, Ewen, Kelly B. Campbell, David D. Kuhn, Michael F. Tlusty, and Frederick T. Barrows. 2024. "The Impact of Marine Resource-Free Diets on Quality Attributes of Atlantic Salmon" Fishes 9, no. 1: 37. https://doi.org/10.3390/fishes9010037
APA StyleMcLean, E., Campbell, K. B., Kuhn, D. D., Tlusty, M. F., & Barrows, F. T. (2024). The Impact of Marine Resource-Free Diets on Quality Attributes of Atlantic Salmon. Fishes, 9(1), 37. https://doi.org/10.3390/fishes9010037







