Remote Sensing of the Subtropical Front in the Southeast Pacific and the Ecology of Chilean Jack Mackerel Trachurus murphyi
Abstract
1. Introduction
2. Data and Methods
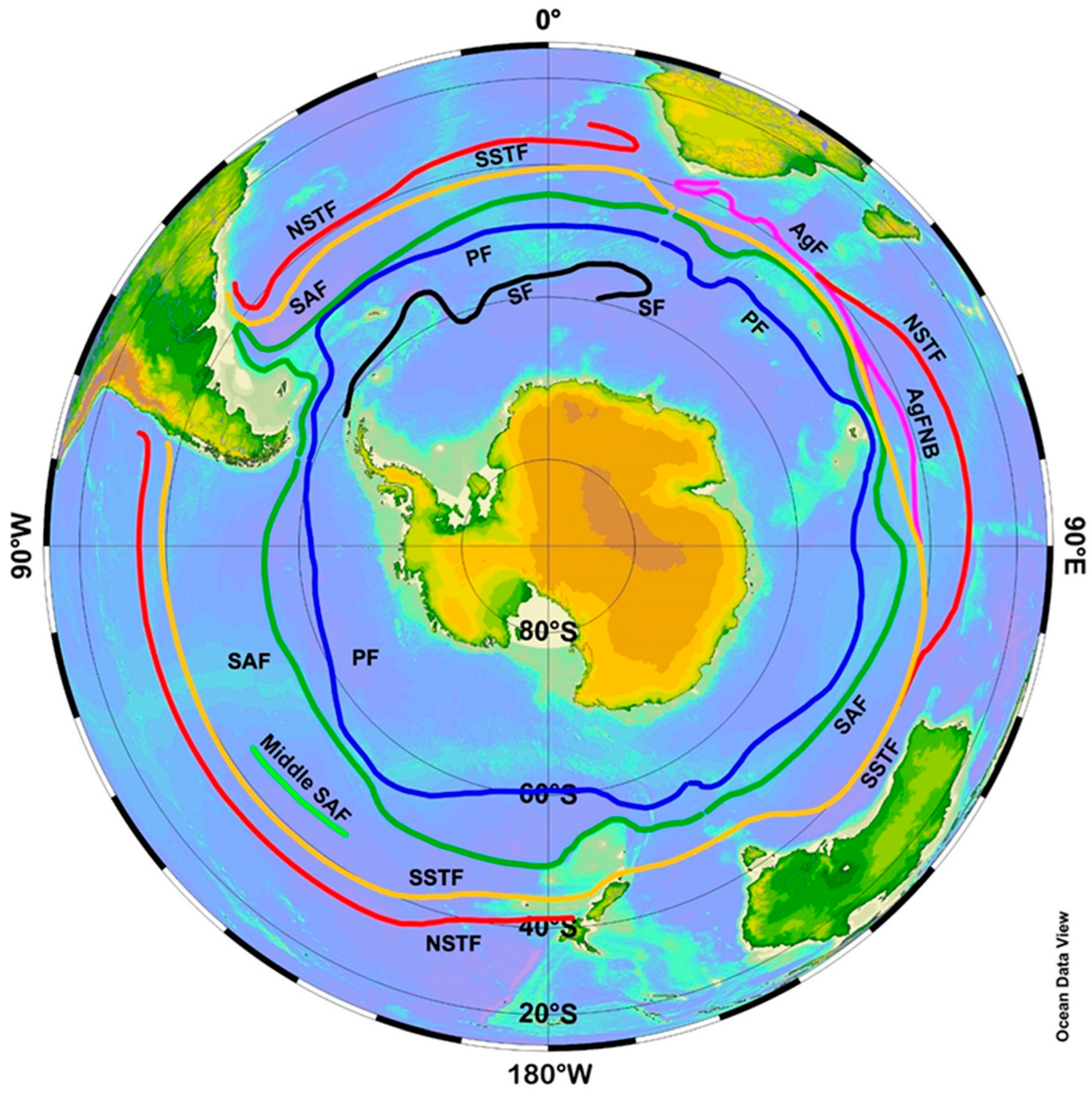
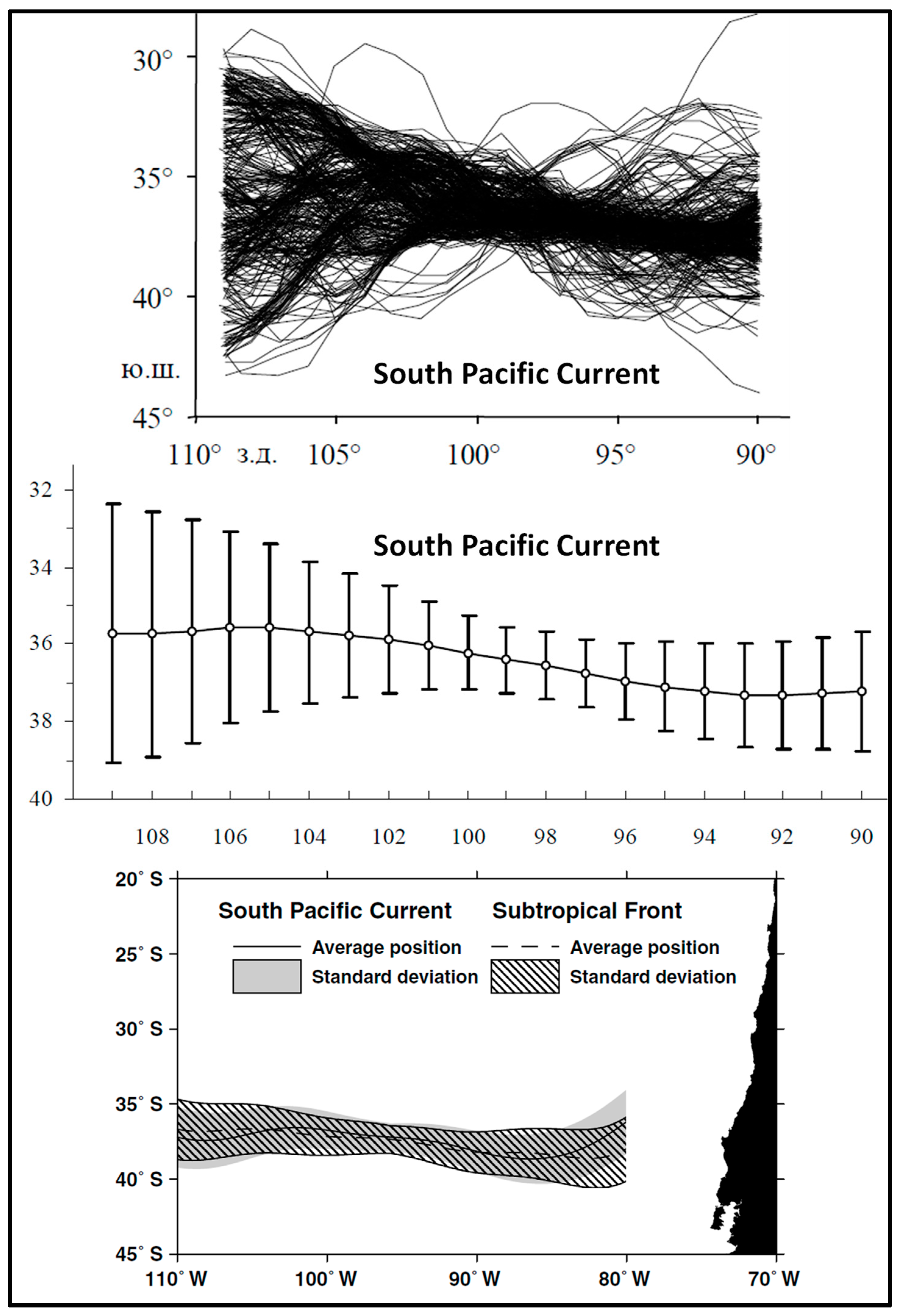
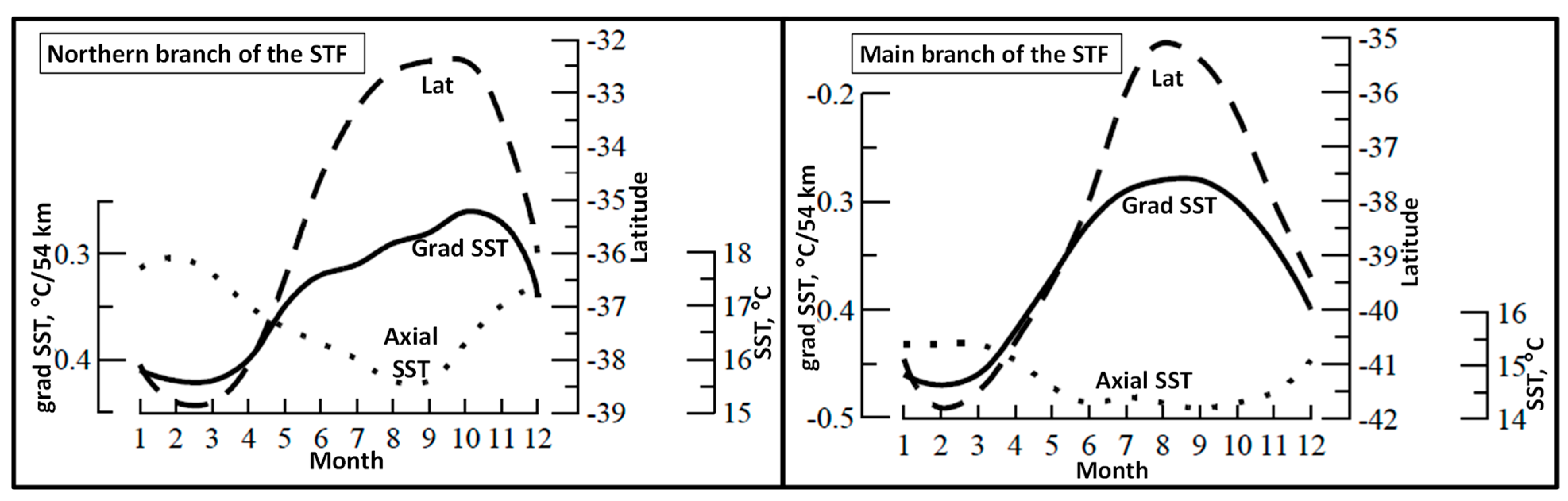
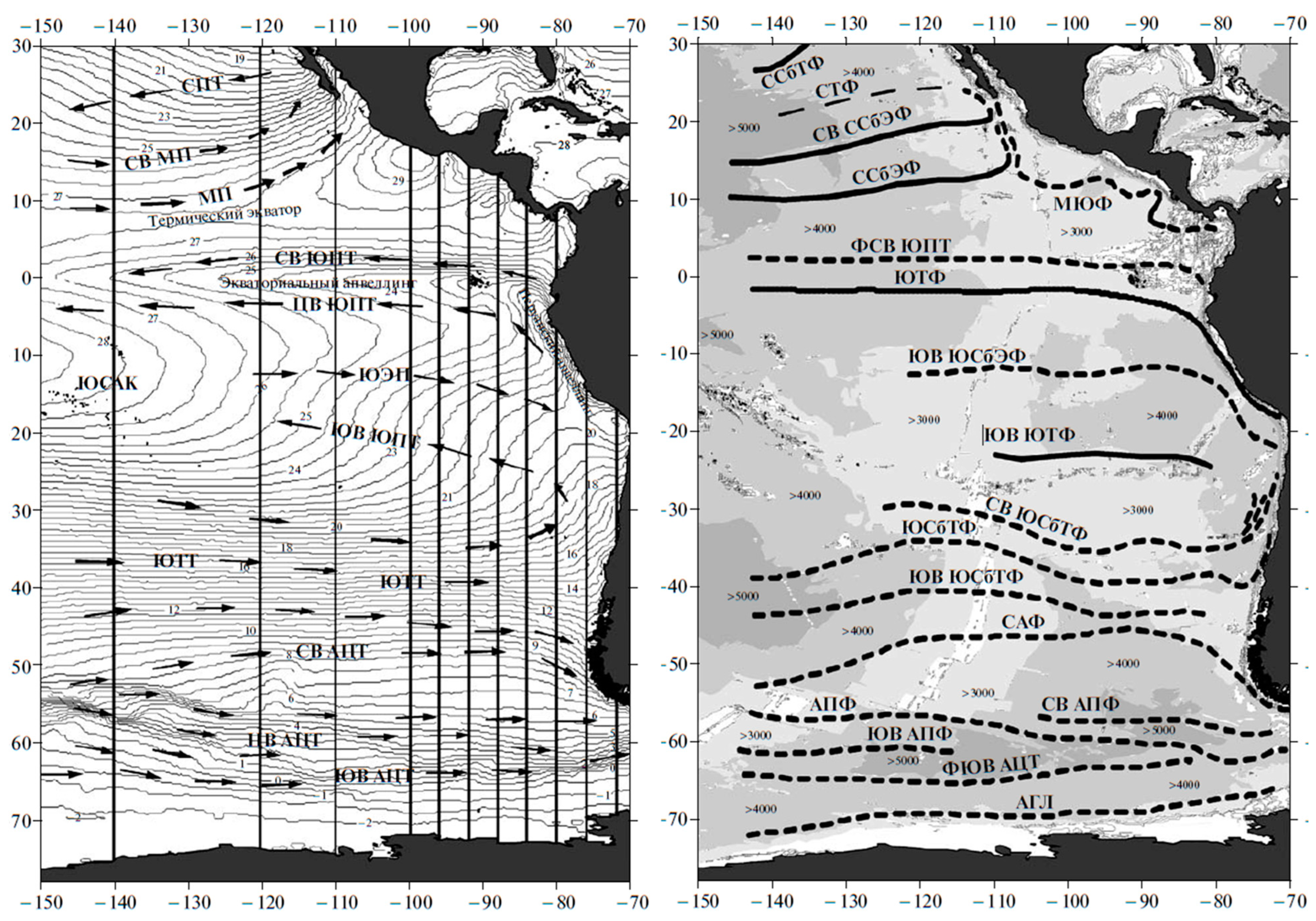
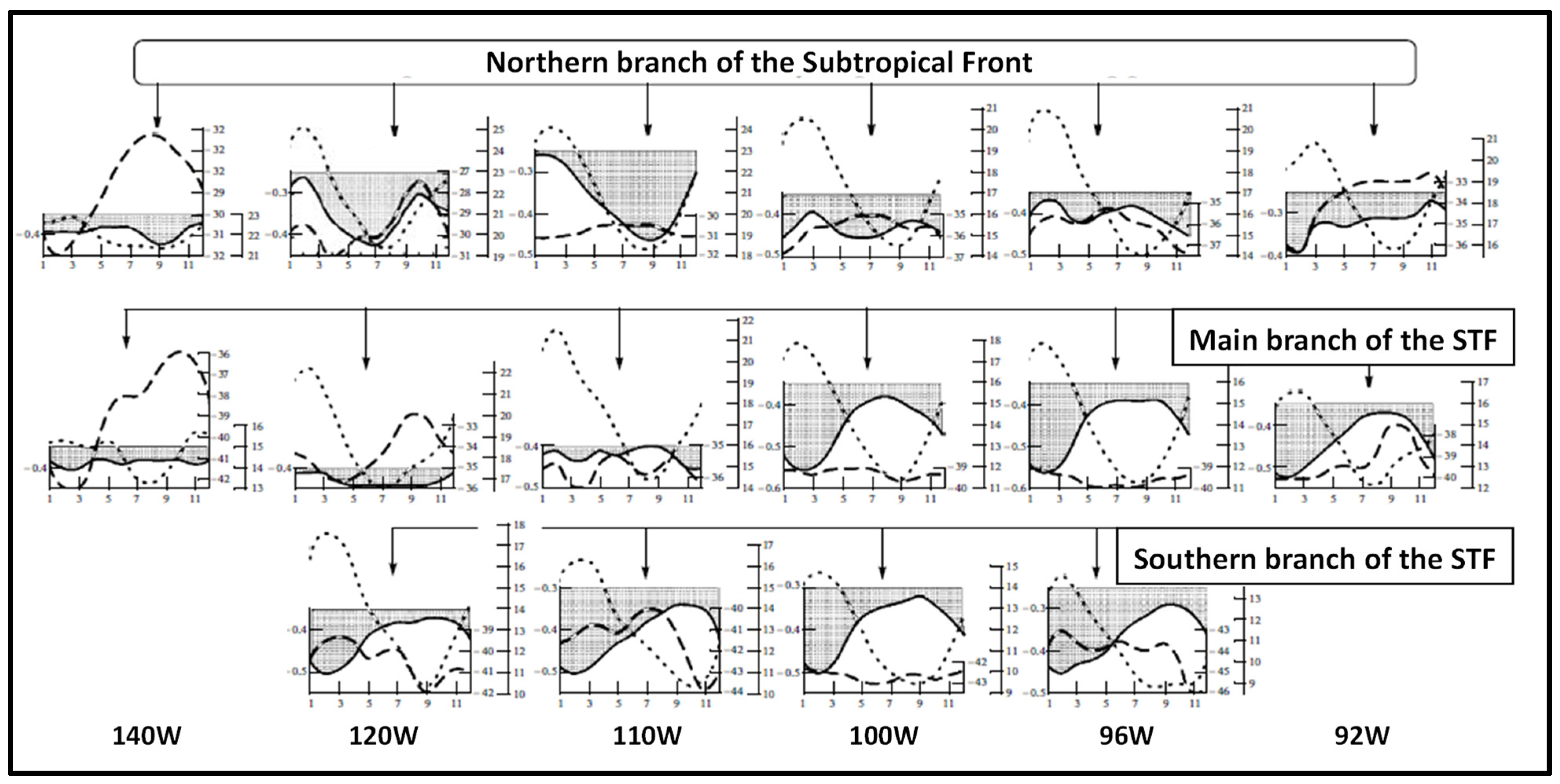
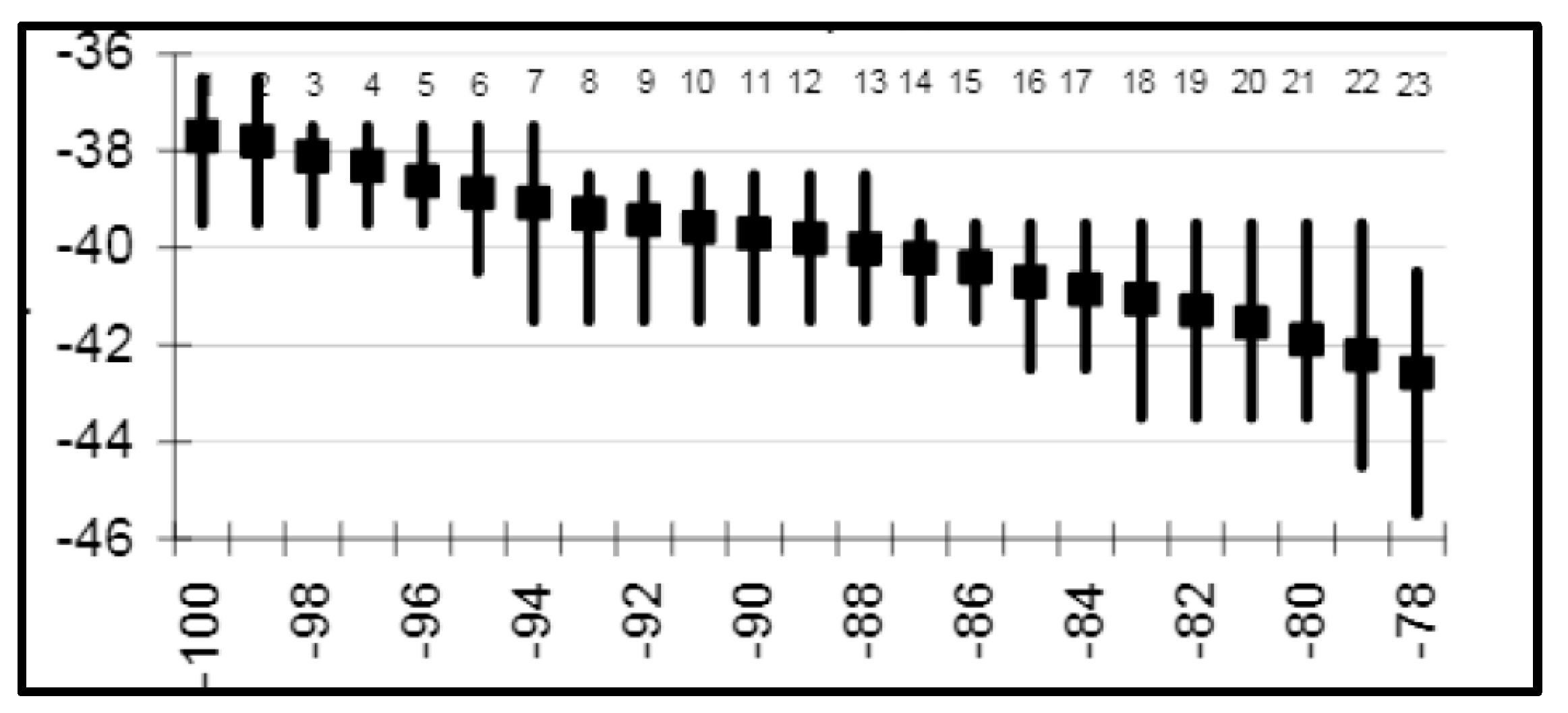
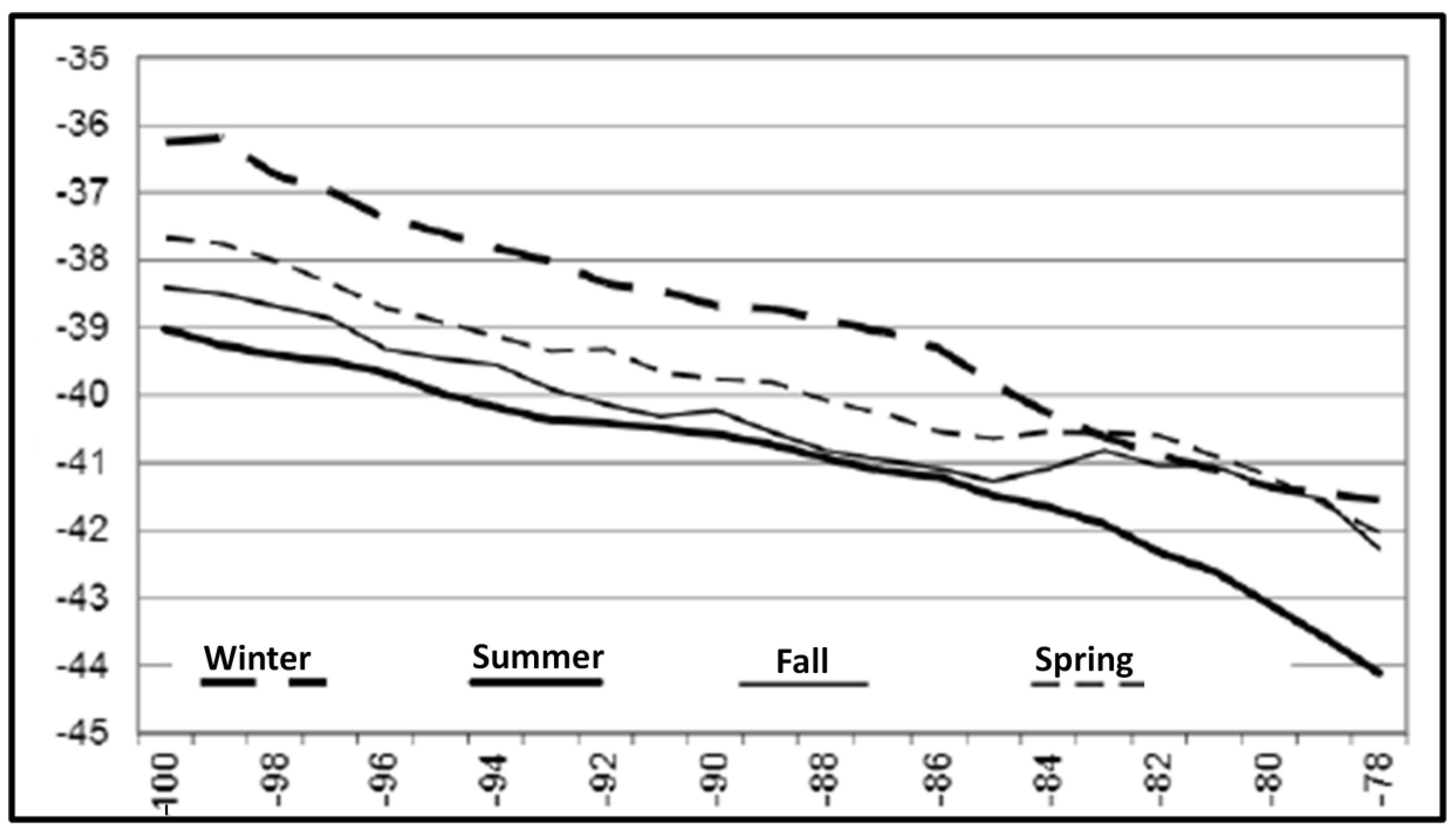
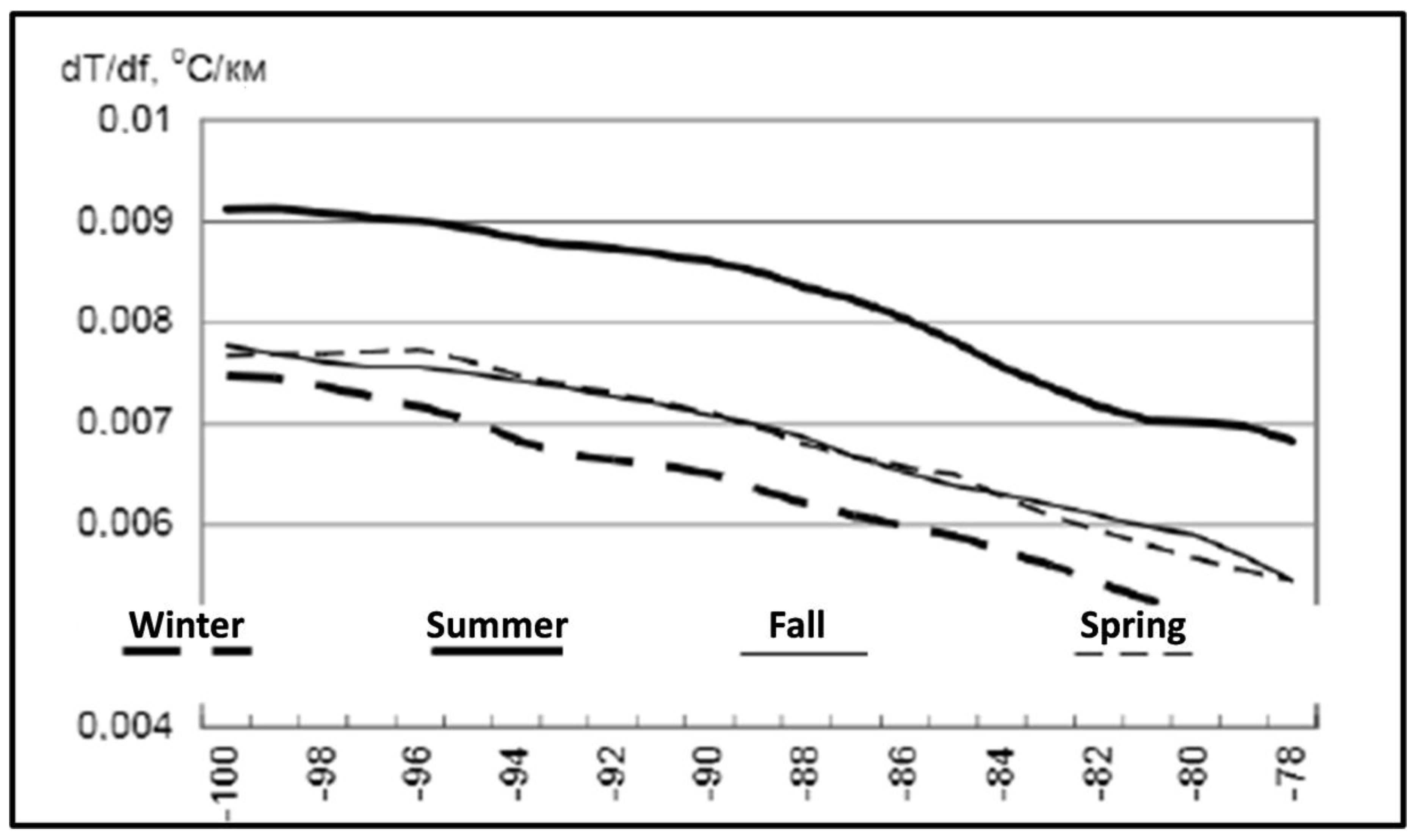
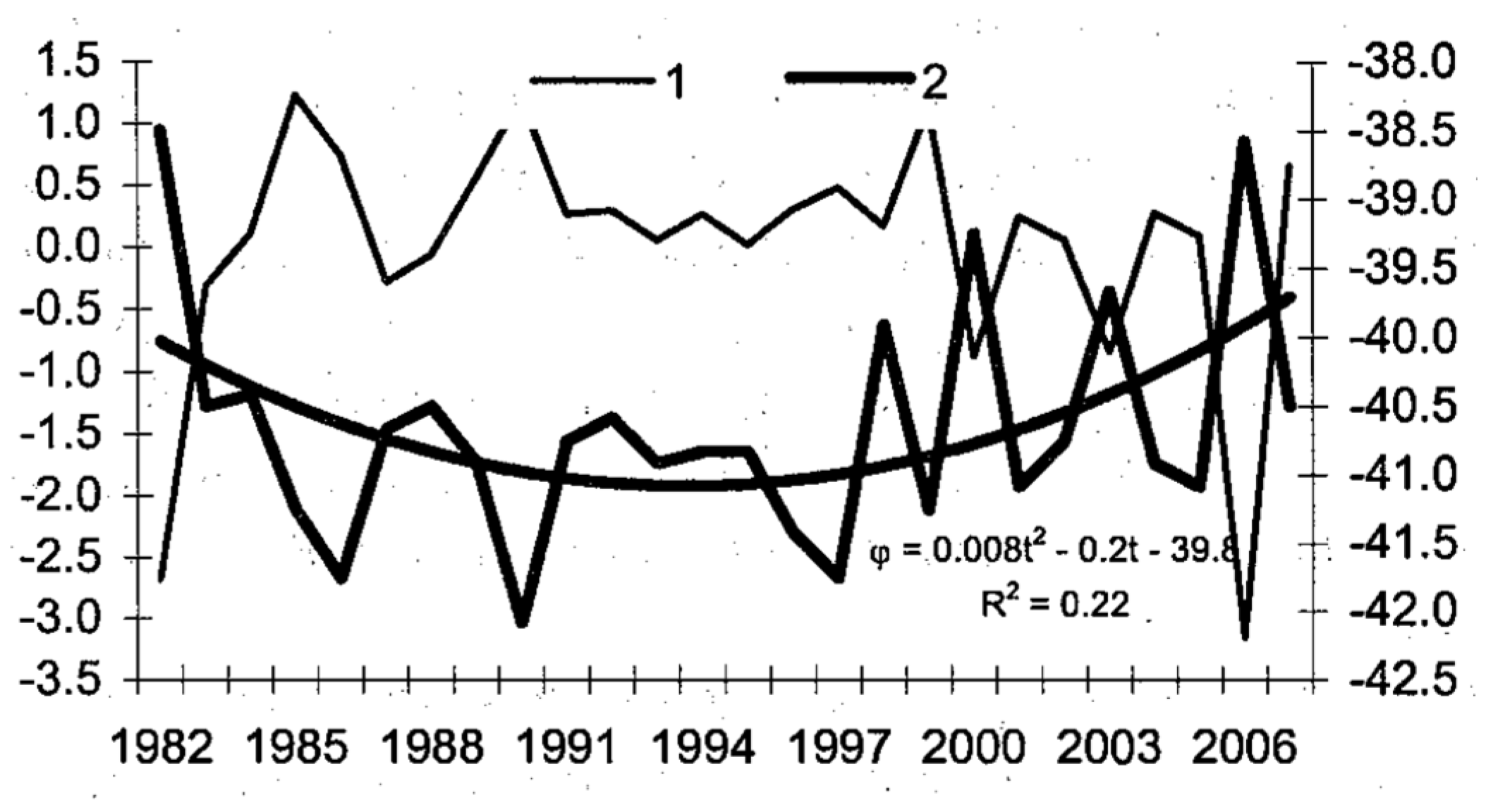
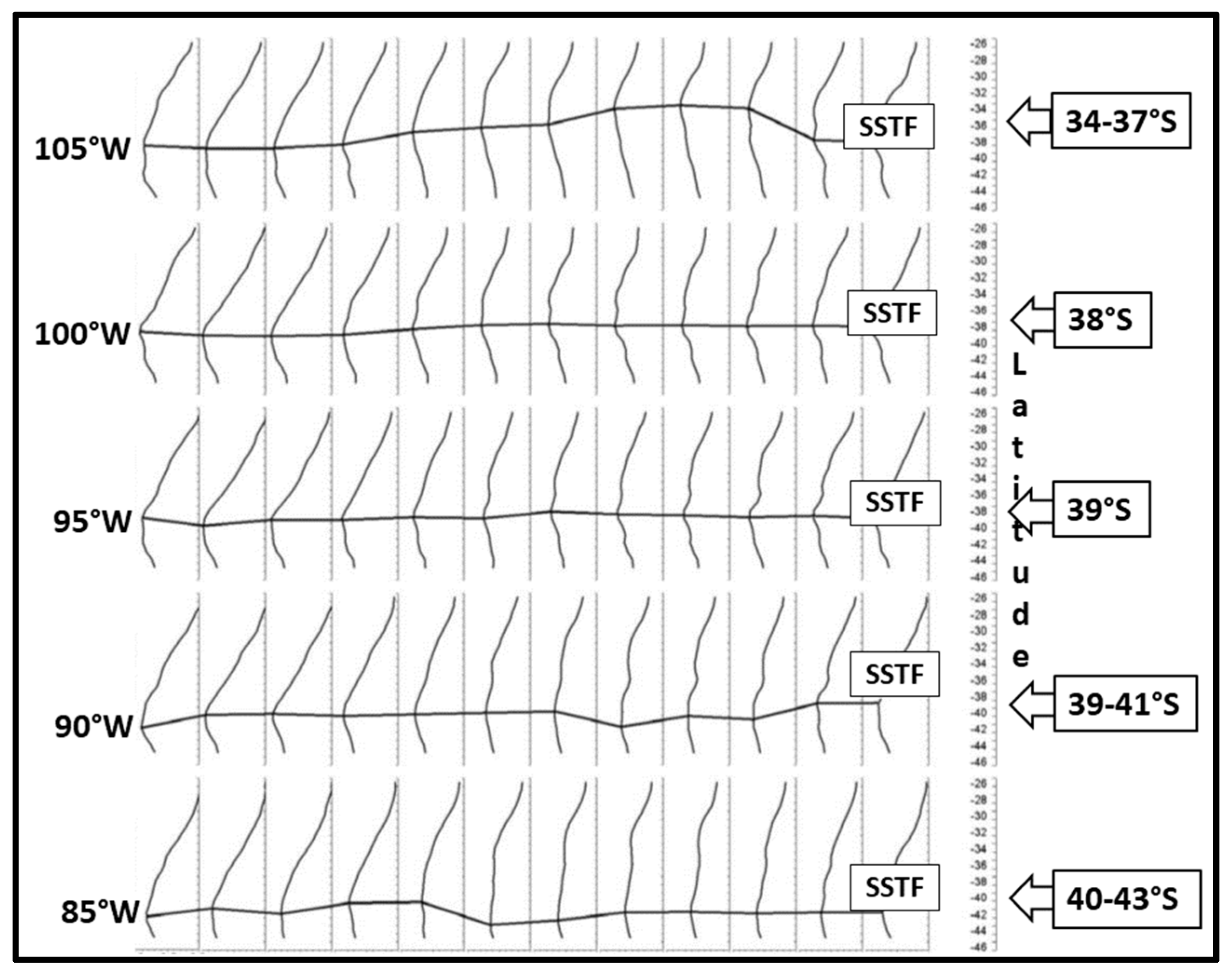
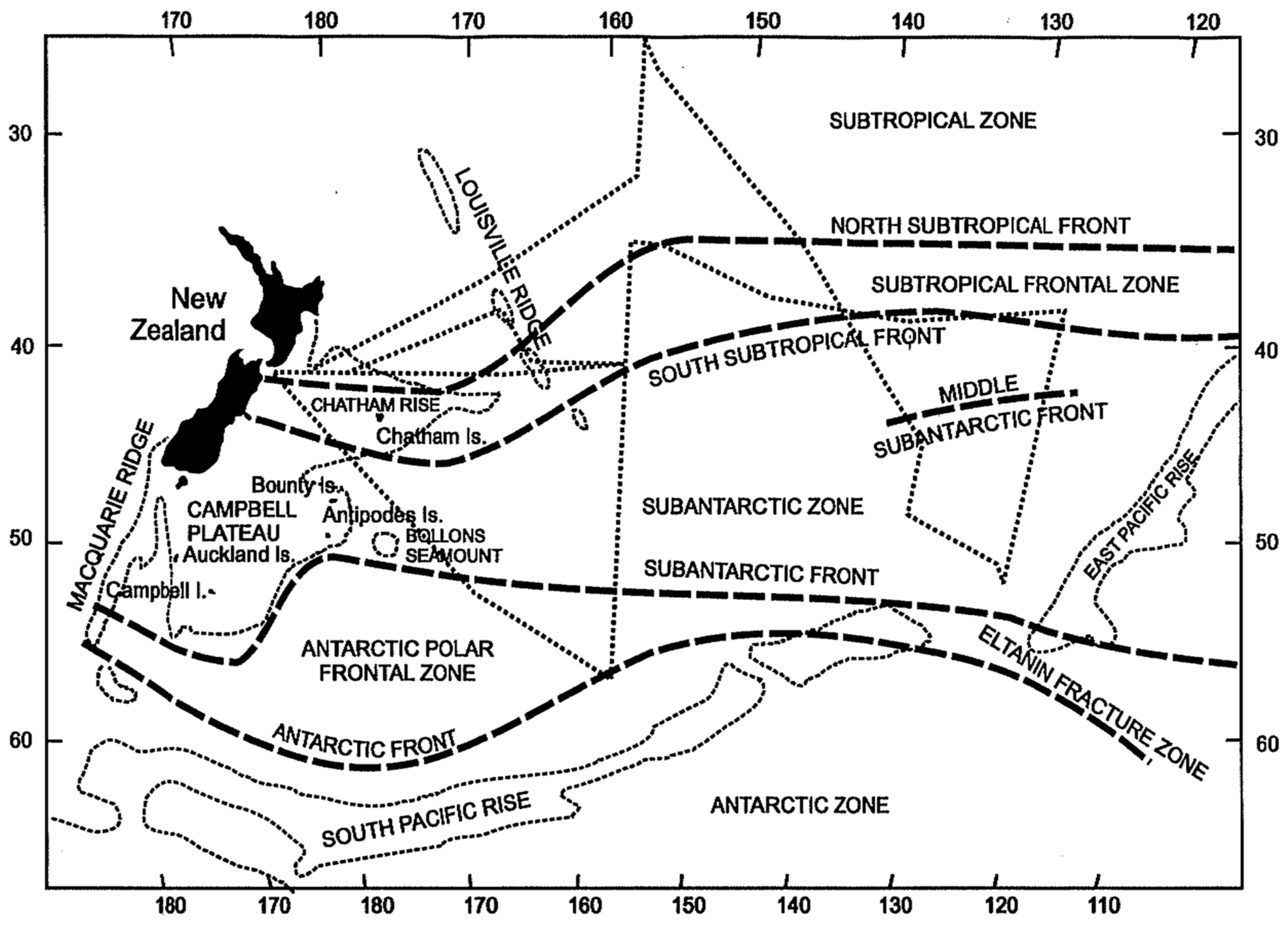
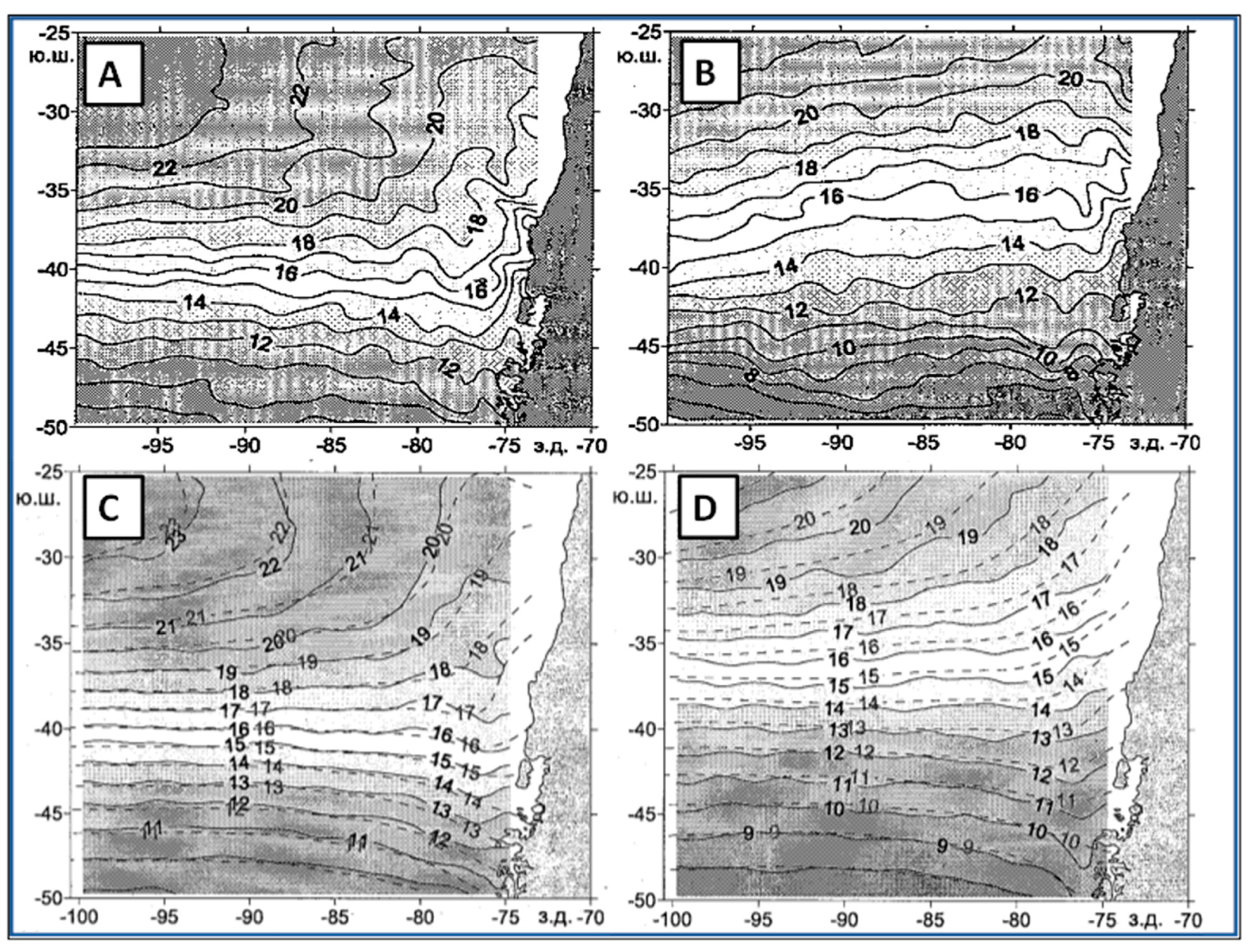
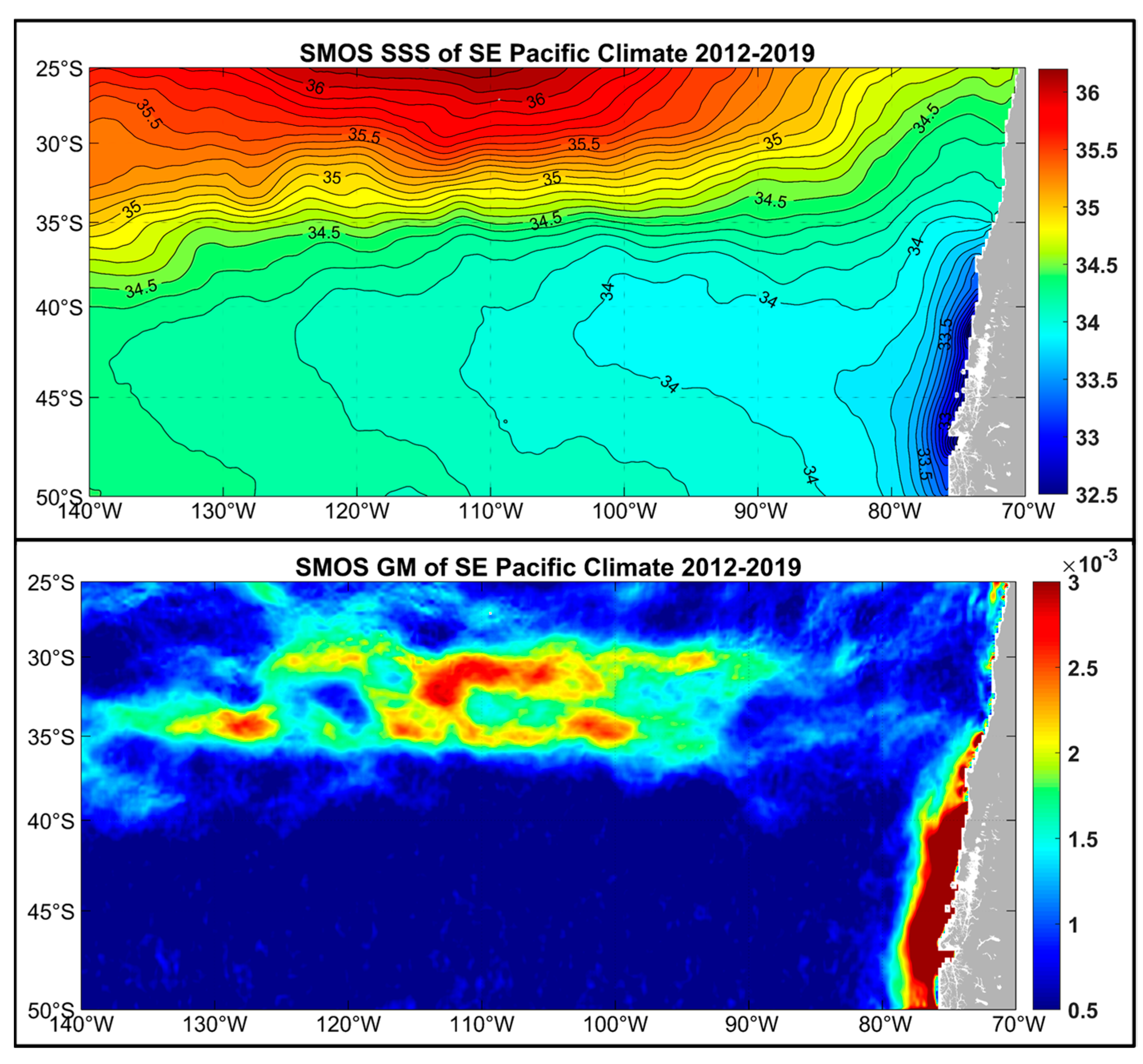
3. Satellite Oceanography of the Subtropical Front in the Southeast Pacific
4. Subtropical Front and the Ecology of Chilean Jack Mackerel Trachurus murphyi
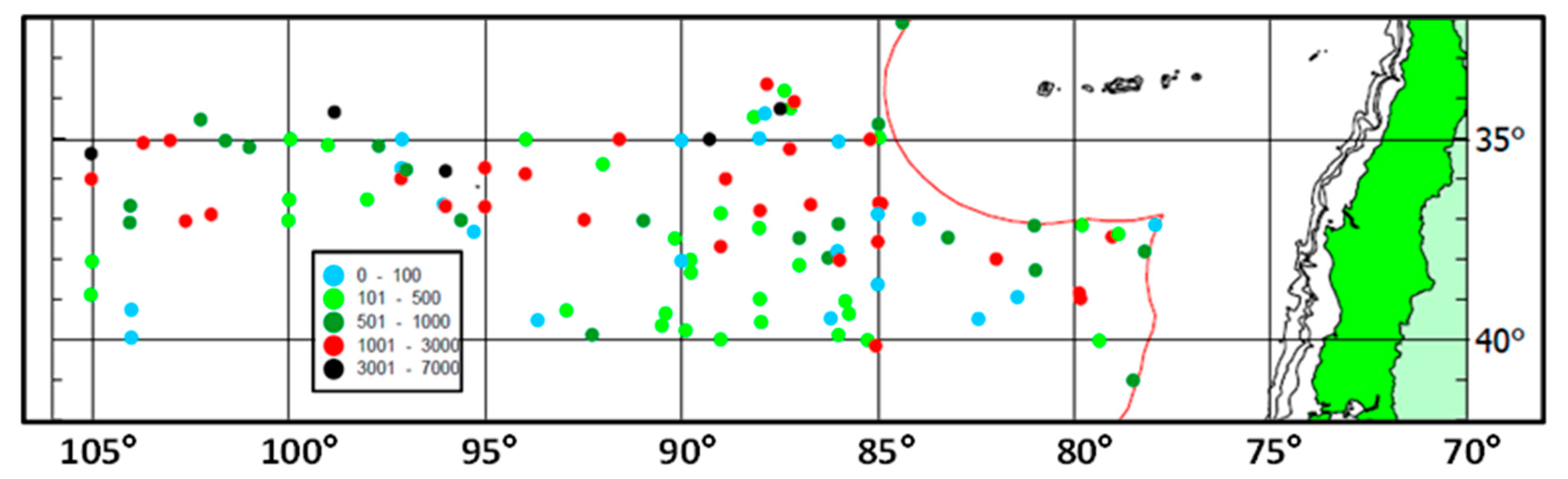
4.1. Jack Mackerel Spawning in the Subtropical Frontal Zone
4.2. Population Structure of Jack Mackerel and along-STF Connectivity
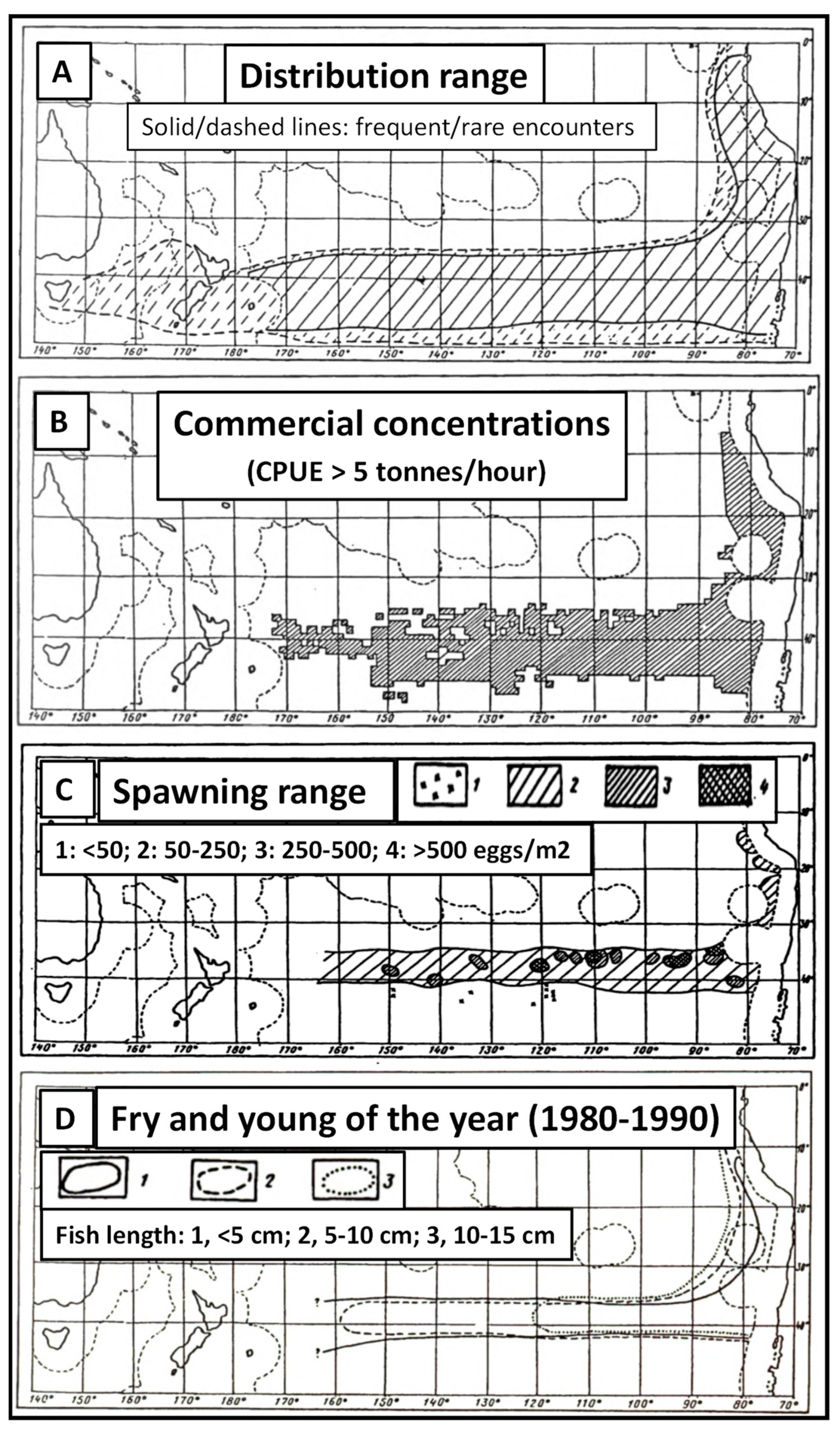
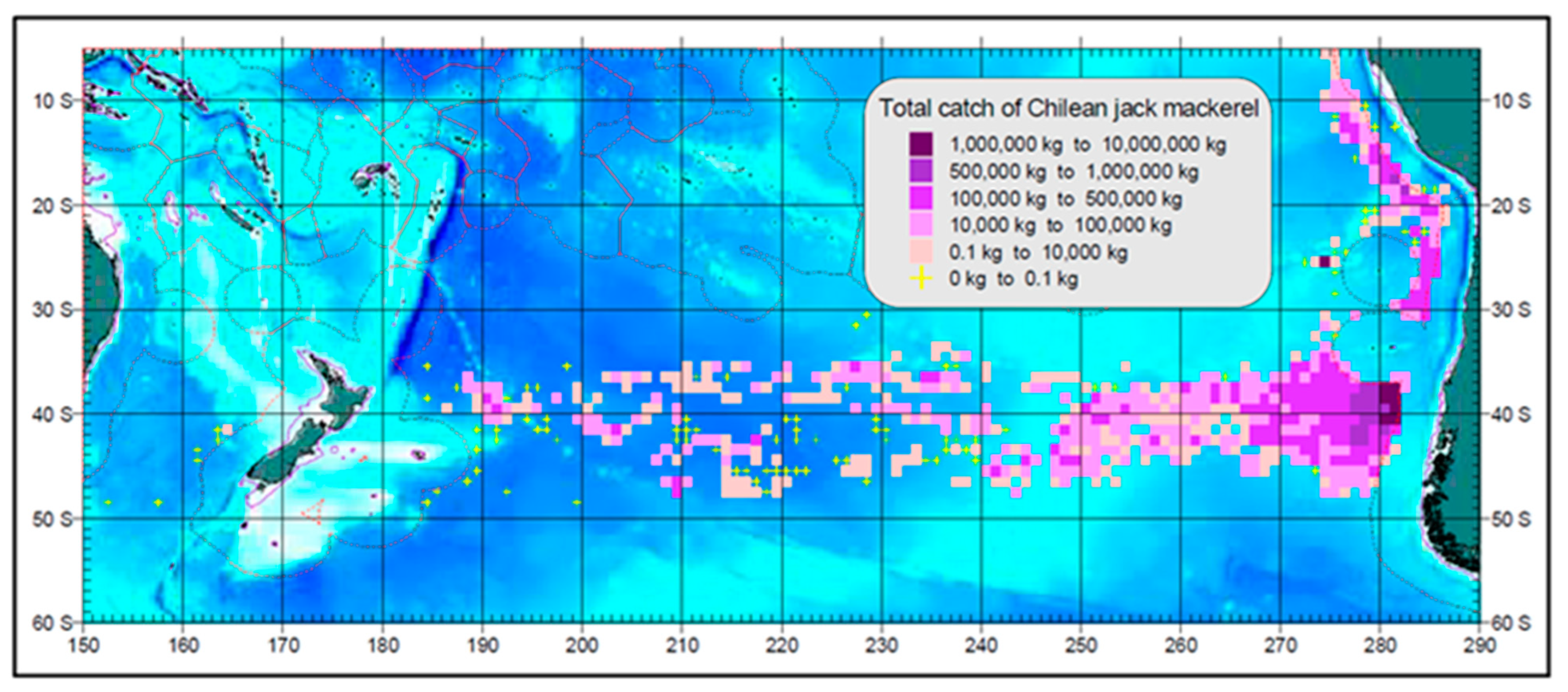
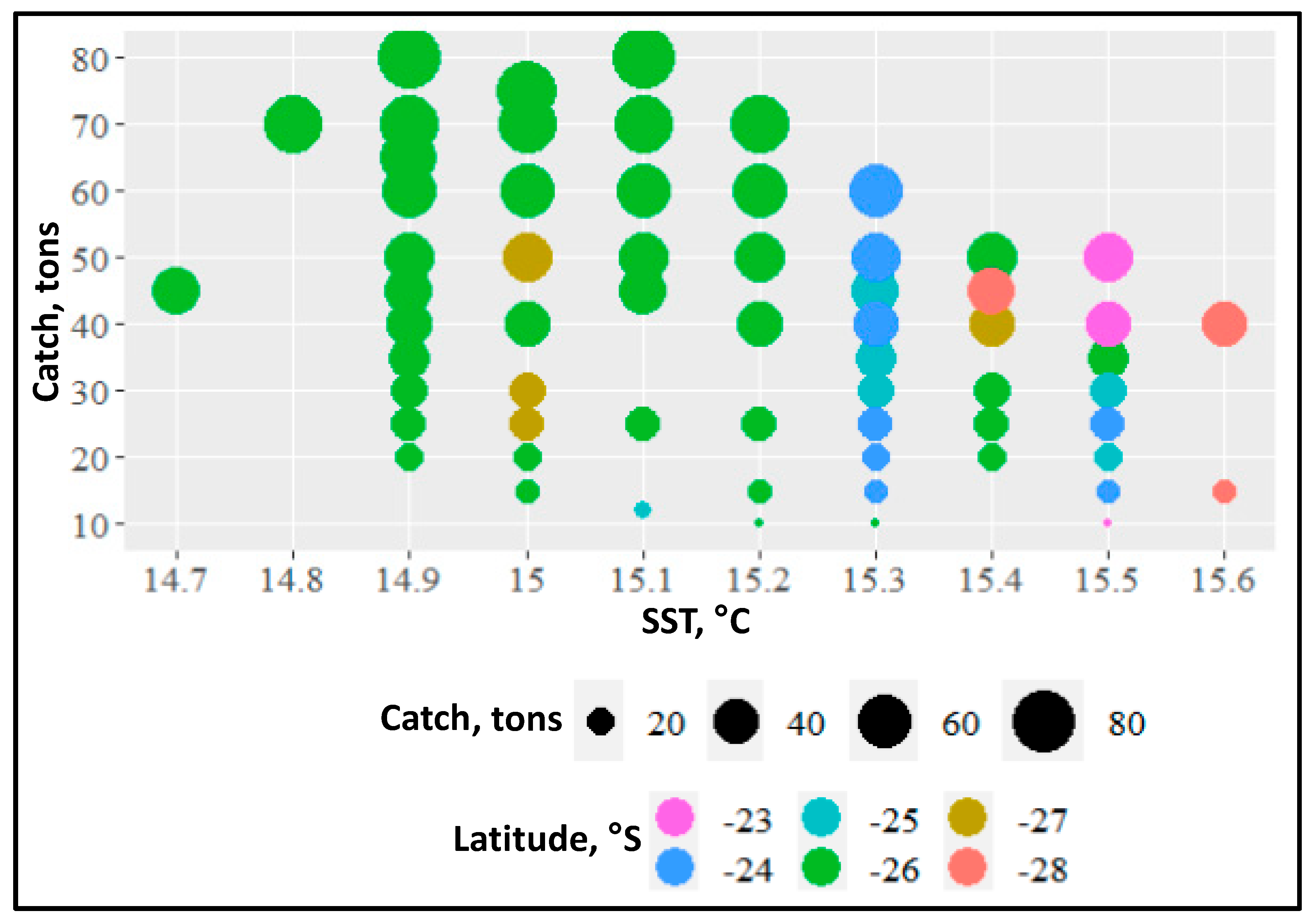
4.3. Jack Mackerel Distribution and Subtropical Front
4.4. Subtropical Front and Spawning Grounds of Jack Mackerel
4.5. Adult Fishes Leave the Subtropical Frontal Zone to Forage in Subantarctic Waters
4.6. Jack Mackerel’s Affinity to the Subtropical Front
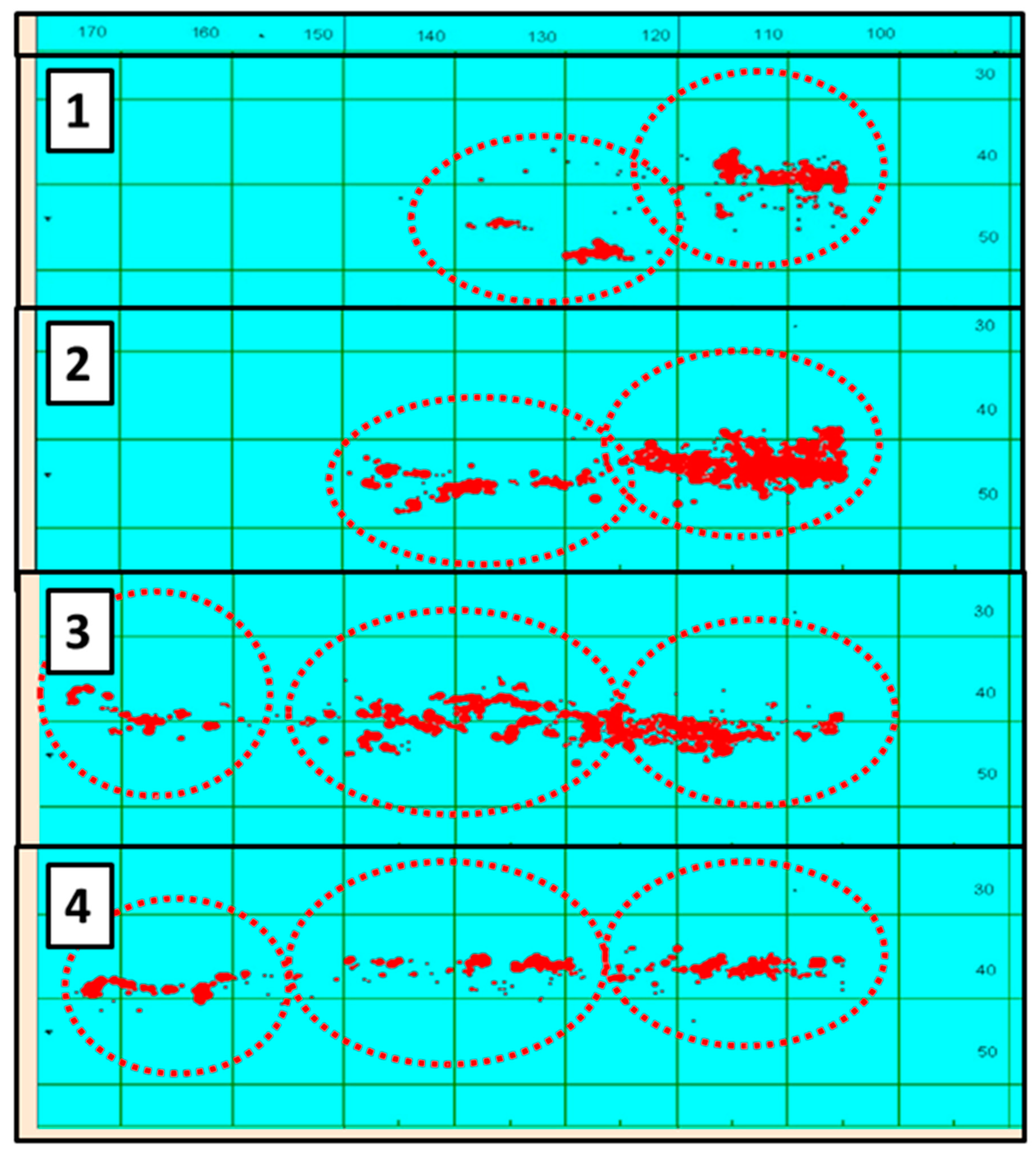
4.7. Migration of Jack Mackerel along the Subtropical Front
5. Discussion
5.1. Spatial Discordance between Temperature and Salinity Manifestations of the STF
5.2. Salinity Data in Fish Ecology
5.3. Fronts (Gradients) in Population Models
5.4. Front Data in Marine Ecology and Fisheries
6. Data Gaps, Knowledge Gaps, and Key Next Steps
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lima, M.; Canales, T.M.; Wiff, R.; Montero, J. The interaction between stock dynamics, fishing and climate caused the collapse of the jack mackerel stock at Humboldt Current ecosystem. Front. Mar. Sci. 2020, 7, 123. [Google Scholar] [CrossRef]
- Stramma, L.; Peterson, R.G.; Tomczak, M. The South Pacific Current. J. Phys. Oceanogr. 1995, 25, 77–91. [Google Scholar] [CrossRef]
- Belkin, I.M. Main hydrological features of the Central South Pacific. In Ecosystems of the Subantarctic Zone of the Pacific Ocean; Vinogradov, M.E., Flint, M.V., Eds.; Nauka: Moscow, Russia, 1988; pp. 21–28, English translation: Pacific Subantarctic Ecosystems; New Zealand Translation Centre Ltd.: Wellington, New Zealand, 1997; pp. 12–17. [Google Scholar]
- Belkin, I.M. Frontal structure of the South Atlantic. In Pelagic Ecosystems of the Southern Ocean; Voronina, N.M., Ed.; Nauka: Moscow, Russia, 1993; pp. 40–53. (In Russian) [Google Scholar]
- Belkin, I.M.; Gordon, A.L. Southern Ocean fronts from the Greenwich meridian to Tasmania. J. Geophys. Res. Ocean. 1996, 101, 3675–3696. [Google Scholar] [CrossRef]
- Schlitzer, R. Ocean Data View. 2022. Available online: https://odv.awi.de (accessed on 4 December 2022).
- Chaigneau, A.; Pizarro, O. Eddy characteristics in the eastern South Pacific. J. Geophys. Res. Ocean. 2005, 110, C06005. [Google Scholar] [CrossRef]
- Chaigneau, A.; Pizarro, O. Mean surface circulation and mesoscale turbulent flow characteristics in the eastern South Pacific from satellite tracked drifters. J. Geophys. Res. Ocean. 2005, 110, C05014. [Google Scholar] [CrossRef]
- Chaigneau, A.; Pizarro, O. Surface circulation and fronts of the South Pacific Ocean, east of 120° W. Geophys. Res. Lett. 2005, 32, L08605. [Google Scholar] [CrossRef]
- Belkin, I.M.; Gritsenko, A.M.; Kryukov, V.V. Thermohaline structure and hydrological fronts. In Ecosystems of the Subantarctic Zone of the Pacific Ocean; Vinogradov, M.E., Flint, M.V., Eds.; Nauka: Moscow, Russia, 1988; pp. 28–36. (In Russian); English translation: Pacific Subantarctic Ecosystems; New Zealand Translation Centre Ltd.: Wellington, New Zealand, 1997; pp. 18–23 [Google Scholar]
- Belkin, I.M.; Gusev, Y.M.; Levin, L.A. Surface thermohaline fronts of the South Pacific. In Ecosystems of the Subantarctic Zone of the Pacific Ocean; Vinogradov, M.E., Flint, M.V., Eds.; Nauka: Moscow, Russia, 1988; pp. 36–47. (In Russian); English translation: Pacific Subantarctic Ecosystems; New Zealand Translation Centre Ltd.: Wellington, New Zealand; 1997; pp. 24–29 [Google Scholar]
- Vinogradov, M.E.; Flint, M.V. (Eds.) Ecosystems of the Subantarctic Zone of the Pacific Ocean; Nauka: Moscow, Russia; p. 304. (In Russian); English translation: Pacific Subantarctic Ecosystems; New Zealand Translation Centre Ltd.: Wellington, New Zealand, 1997, p. 243.
- Tsuchiya, M.; Talley, L.D. Water-property distributions along an eastern Pacific hydrographic section at 135W. J. Mar. Res. 1996, 54, 541–564. [Google Scholar] [CrossRef]
- Tsuchiya, M.; Talley, L.D. A Pacific hydrographic section at 88° W: Water-property distribution. J. Geophys. Res. Ocean. 1998, 103, 12899–12918. [Google Scholar] [CrossRef]
- González Carman, V.; Piola, A.; O’Brien, T.D.; Tormosov, D.D.; Acha, E.M. Circumpolar frontal systems as potential feeding grounds of Southern Right whales. Prog. Oceanogr. 2019, 176, 102123. [Google Scholar] [CrossRef]
- Clay, T.A.; Phillips, R.A.; Manica, A.; Jackson, H.A.; de Brooke, M.L. Escaping the oligotrophic gyre? The year-round movements, foraging behaviour and habitat preferences of Murphy’s petrels. Mar. Ecol. Prog. Ser. 2017, 579, 139–155. [Google Scholar]
- Evseenko, S.A. On the reproduction of the Peruvian jack mackerel, Trachurus symmetricus murphyi (Nichols), in the Southern Pacific. J. Ichthyol. 1987, 27, 151–160. [Google Scholar]
- Bailey, K. Description and surface distribution of juvenile Peruvian jack mackerel, Trachurus murphyi, Nichols from the subtropical convergence zone of the central South Pacific. Fish. Bull. 1989, 87, 273–278. [Google Scholar]
- Gerlotto, F.; Dioses, T. Bibliographical Synopsis on the Main Traits of life of Trachurus murphyi in the South Pacific Ocean. SPRFMO 2013, SC-01-INF-17. In Proceedings of the 1st Meeting of the Scientific Committee, La Jolla, CA, USA, 21–27 October 2013; p. 217. [Google Scholar]
- Haddaway, N.R.; Macura, B.; Whaley, P.; Pullin, A.S. ROSES RepOrting standards for Systematic Evidence Syntheses: Pro forma, flow-diagram and descriptive summary of the plan and conduct of environmental systematic reviews and systematic maps. Environ. Evid. 2018, 1998, 7. [Google Scholar] [CrossRef]
- Artamonov, Y.V.; Skripaleva, E.A. Variability of hydrological fronts in the Peru-Chilean sector from satellite data. Ukr. Antarct. J. 2006, 5, 109–116. (In Russian) [Google Scholar] [CrossRef]
- Artamonov, Y.V.; Skripaleva, E.A. Seasonal variability of the large-scale fronts in the Eastern Pacific based on satellite data. Earth Stud. Space 2008, 4, 45–61. (In Russian) [Google Scholar]
- Artamonov, Y.V.; Skripaleva, E.A.; Babiy, M.V.; Galkovskaya, L.K. Seasonal and interannual variability of the hydrological fronts in the Southern Ocean. Ukr. Antarct. J. 2009, 8, 205–216. (In Russian) [Google Scholar] [CrossRef]
- Gordeeva, V.N.; Malinin, V.N. Large-scale variability of the South Subtropical Front in the south-eastern part of the Pacific Ocean. Vestn. Immanuel Kant Balt. Fed. Univ. 2006, 2, 160–169. (In Russian) [Google Scholar]
- Krasnoborodko, O.Y. Formation of local frontal zones and mesoscale dynamics of waters in the southern part of Pacific Ocean. Tr. VNIRO 2017, 169, 136–141. (In Russian) [Google Scholar]
- Lebedev, S.A.; Sirota, A.M. Oceanographic investigation in the Southeastern Pacific Ocean by satellite radiometry and altimetry data. Adv. Space Res. 2007, 39, 203–208. [Google Scholar] [CrossRef]
- Malinin, V.N.; Gordeeva, S.M. Fisheries Oceanography of the Southeast Pacific: Variability of Habitat Factors; St. Petersburg State University Publishing House: St. Petersburg, Russia, 2009; Volume 1, p. 278. (In Russian) [Google Scholar]
- Sirota, A.M.; Lebedev, S.A.; Timokhin, E.N.; Chernyshkov, P.P. Application of Satellite Altimetry for Diagnostics of Fisheries Oceanography Conditions in the Atlantic Ocean and Southeast Pacific; AtlantNIRO: Kaliningrad, Russia, 2004; p. 68. (In Russian) [Google Scholar]
- Graham, R.M.; De Boer, A.M. The Dynamical Subtropical Front. J. Geophys. Res. Ocean. 2013, 118, 5676–5685. [Google Scholar] [CrossRef]
- Tippins, D.; Tomczak, M. Meridional Turner angles and density compensation in the upper ocean. Ocean Dyn. 2003, 53, 332–342. [Google Scholar] [CrossRef]
- Núñez, S.; Letelier, J.; Donoso, D.; Sepúlveda, A.; Arcos, D. Relating spatial distribution of Chilean jack mackerel and environmental factors in the oceanic waters off Chile. Gayana 2004, 68, 444–449. [Google Scholar] [CrossRef]
- Cubillos, L.A.; Paramo, J.; Ruiz, P.; Núñez, S.; Sepúlveda, A. The spatial structure of the oceanic spawning of jack mackerel (Trachurus murphyi) off central Chile (1998–2001). Fish. Res. 2008, 90, 261–270. [Google Scholar] [CrossRef]
- Núñez, S.; Vásquez, S.; Ruiz, P.; Sepúlveda, A. Distribution of Early Developmental Stages of Jack Mackerel in the Southeastern Pacific Ocean. Technical Report, Chilean Jack Mackerel Workshop Paper #2, 2010; p. 11.
- Vásquez, S.; Correa-Ramírez, M.; Parada, C.; Sepúlveda, A. The influence of oceanographic processes on jack mackerel (Trachurus murphyi) larval distribution and population structure in the southeastern Pacific Ocean. ICES J. Mar. Sci. 2013, 70, 1097–1107. [Google Scholar] [CrossRef]
- Elizarov, A.A.; Grechina, A.S.; Kotenev, B.N.; Kuznetsov, A.N. Peruvian jack mackerel, Trachurus symmetricus murphyi in the open waters of the South Pacific. J. Ichthyol. 1993, 33, 86–104. [Google Scholar]
- Serra, R. Important life history aspects of the Chilean jack mackerel, Trachurus symmetricus murphyi. Investig. Pesq. 1991, 36, 67–83. [Google Scholar]
- Taylor, P.R. Stock structure and population biology of the Peruvian jack mackerel, Trachurus symmetricus murphyi. New Zealand Fish. Assess. Rep. 2002, 2002, 78. [Google Scholar]
- Cárdenas, L.; Silva, A.X.; Magoulas, A.; Cabezas, J.; Poulin, E.; Ojeda, F.P. Genetic population structure in the Chilean jack mackerel, Trachurus murphyi (Nichols) across the South-eastern Pacific Ocean. Fish. Res. 2009, 100, 109–115. [Google Scholar] [CrossRef]
- Ashford, J.; Serra, R.; Saavedra, J.C.; Letelier, J. Otolith chemistry indicates large-scale connectivity in Chilean jack mackerel (Trachurus murphyi), a highly mobile species in the Southern Pacific Ocean. Fish. Res. 2011, 107, 291–299. [Google Scholar] [CrossRef]
- Gerlotto, F.; Gutiérrez, M.; Bertrand, A. Insight on population structure of the Chilean jack mackerel (Trachurus murphyi). Aquat. Living Resour. 2012, 25, 341–355. [Google Scholar] [CrossRef]
- Zhu, G.P.; Zhang, M.; Ashford, J.; Zou, X.R.; Chen, X.J.; Zhou, Y.Q. Does life history connectivity explain distributions of Chilean jack mackerel Trachurus murphyi caught in international waters prior to decline of the southeastern Pacific fishery? Fish. Res. 2014, 151, 20–25. [Google Scholar] [CrossRef]
- Dragon, A.-C.; Senina, I.; Hintzen, N.T.; Lehodey, P. Modelling South Pacific jack mackerel spatial population dynamics and fisheries. Fish. Oceanogr. 2017, 27, 97–113. [Google Scholar] [CrossRef]
- Parada, C.; Gretchina, A.; Vásquez, S.; Belmadani, A.; Combes, V.; Ernst, B.; Di Lorenzo, E.; Porobic, J.; Sepúlveda, A. Expanding the conceptual framework of the spatial population structure and life history of jack mackerel in the eastern South Pacific: An oceanic seamount region as potential spawning/nursery habitat. ICES J. Mar. Sci. 2017, 74, 2398–2414. [Google Scholar] [CrossRef]
- Gerlotto, F.; Bertrand, A.; Hintzen, N.; Gutiérrez, M. Adapting the concept of metapopulations to large scale pelagic habitats. SPRFMO, SC9-HM01, 2021; p. 35.
- Bertrand, A.; Habasque, J.; Hattab, T.; Hintzen, N.T.; Oliveros-Ramos, R.; Gutiérrez, M.; Demarcq, H.; Gerlotto, F. 3-D habitat suitability of jack mackerel Trachurus murphyi in the Southeastern Pacific, a comprehensive study. Prog. Oceanogr. 2016, 146, 199–211. [Google Scholar] [CrossRef]
- Li, G.; Cao, J.; Zou, X.R.; Chen, X.J.; Runnebaum, J. Modeling habitat suitability index for Chilean jack mackerel (Trachurus murphyi) in the South East Pacific. Fish. Res. 2016, 178, 47–60. [Google Scholar] [CrossRef]
- Vinogradov, M.E.; Shushkina, E.K.; Evseyenko, S.A. Plankton biomass and potential stocks of the Peruvian jack mackerel in the southeastern Pacific subantarctic zone. J. Ichthyol. 1991, 31, 146–151. [Google Scholar]
- Trotsenko, B.G.; Kukharev, N.N.; Romanov, E.V. Database on Ukrainian research of Chilean jack mackerel Trachurus murphyi in the high seas of the southern Pacific. In Proceedings of the International Conference “The Humboldt Current System: Climate, Ocean Dynamics, Ecosystem Processes and Fisheries”, Lima, Peru, 27 November–1 December 2006; p. 195. [Google Scholar]
- Timokhin, I.G.; Kukharev, N.N. Stock status of the Peruvian horse mackerel Trachurus murphyi in the open waters of the southeastern Pacific Ocean (FAO areas 81, 87) and prospects for the resumption of the Ukrainian fishery. Proc. YugNIRO 2008, 46, 162–173. (In Russian) [Google Scholar]
- Soldat, V.T.; Kolomeiko, F.V.; Glubokov, A.I.; Nesterov, A.A.; Chernyshkov, P.P.; Timokhin, E.N. Jack Mackerel (Trachurus murphyi) Distribution Peculiarities in the High Seas of the South Pacific in Relation to the Population Structure. In Proceedings of the 8th Chilean Jack Mackerel Workshop, Santiago, Chile, 30 June–4 July 2008. [Google Scholar]
- Li, G.; Zou, X.R.; Chen, X.J.; Zhou, Y.Q.; Zhang, M. Standardization of CPUE for Chilean jack mackerel (Trachurus murphyi) from Chinese trawl fleets in the high seas of the Southeast Pacific Ocean. J. Ocean Univ. China 2013, 12, 441–451. [Google Scholar] [CrossRef]
- Feng, Z.P.; Yu, W.; Chen, X.J.; Zou, X.R. Distribution of Chilean jack mackerel (Trachurus murphyi) habitats off Chile based on a maximum entropy model. J. Fish. Sci. China 2021, 28, 431–441. [Google Scholar]
- Corten, A. The Fishery for Jack Mackerel in the Eastern Central Pacific by European Trawlers in 2008 and 2009. In Proceedings of the SPRFMO, SP-08-SWG-JM-01, 2009, Santiago, Chile, 30 June–4 July 2008; p. 10. [Google Scholar]
- Vinogradov, V.I.; Arkhipov, A.G.; Kozlov, D.A. Nutrition of Peruvian horse mackerel Trachurus symmetricus murphyi and Peruvian mackerel Scomber japonicus peruanus in the southeast Pacific. KSTU News 2013, 28, 49–69. (In Russian) [Google Scholar]
- Konchina, Y.V.; Nesin, A.V.; Onishchik, N.A.; Pavlov, Y.P. On the migration and feeding of the jack mackerel Trachurus symmetricus murphyi in the Eastern Pacific. J. Ichthyol. 1996, 36, 753–766. [Google Scholar]
- Zhang, H.; Zhang, S.-M.; Cui, X.-S.; Yang, S.-L.; Hua, C.-J.; Ma, H.-Y. Spatio-temporal dynamics in the location of the fishing grounds and catch per unit effort (CPUE) for Chilean jack mackerel (Trachurus murphyi Nichols, 1920) from Chinese trawl fleets on the high seas of the Southeast Pacific Ocean, 2001–2010. J. Appl. Ichthyol. 2015, 31, 646–656. [Google Scholar] [CrossRef]
- Arcos, D.F.; Cubillos, L.A.; Núñez, S.P. The jack mackerel fishery and El Niño 1997-98 effects off Chile. Prog. Oceanogr. 2001, 49, 597–617. [Google Scholar] [CrossRef]
- Dubishchuk, M.M. Peculiarities of the jack mackerel Trachurus murphyi biological traits and fishery in the open waters of the central subarea of South-East Pacific in August–October 2020. Tr. AtlantNIRO 2021, 5, 122–135. (In Russian) [Google Scholar]
- Horn, P.L.; Maolagáin, C. The growth and age structure of Chilean jack mackerel (Trachurus murphyi) following its influx to New Zealand waters. J. Fish Biol. 2021, 98, 1144–1154. [Google Scholar] [CrossRef] [PubMed]
- Konchina, Y.V.; Pavlov, Y.P. On the strength of generations of the Pacific jack mackerel Trachurus symmetricus murphyi. J. Ichthyol. 1999, 39, 748–754. [Google Scholar]
- Grechina, A.S. Historia de investigaciones y aspectos básicos de la ecología del jurel Trachurus symmetricus murphyi (Nichols) en alta mar del Pacífico Sur. In Biología y Ecología del Jurel en Aguas Chilenas; Arcos, D., Ed.; Instituto de Investigación Pesquera, Talcahuano: Santiago, Chile, 1998; pp. 11–34. [Google Scholar]
- McGinnis, R.F. Counterclockwise circulation in the Pacific subantarctic sector of the Southern Ocean. Science 1974, 186, 736–738. [Google Scholar] [CrossRef]
- Deacon, G.E.R. Comments on a counterclockwise circulation in the Pacific subantarctic sector of the Southern Ocean suggested by McGinnis. Deep-Sea Res. 1977, 24, 927–930. [Google Scholar] [CrossRef]
- Dávila, P.M.; Figueroa, D.; Müller, E. Freshwater input into the coastal ocean and its relation with the salinity distribution off austral Chile (35–55° S). Cont. Shelf Res. 2002, 22, 521–534. [Google Scholar] [CrossRef]
- Saldías, G.S.; Sobarzo, M.; Quiñones, R. Freshwater structure and its seasonal variability off western Patagonia. Prog. Oceanogr. 2019, 174, 143–153. [Google Scholar] [CrossRef]
- Karstensen, J. Formation of the South Pacific shallow salinity minimum: A Southern Ocean pathway to the Tropical Pacific. J. Phys. Oceanogr. 2004, 34, 2398–2412. [Google Scholar] [CrossRef]
- Neshyba, S.; Fonseca, T.R. Evidence for counterflow to the West Wind Drift off South America. J. Geophys. Res. 1980, 85, 4888–4892. [Google Scholar] [CrossRef]
- Uribe, E.; Neshyba, S.; Fonseca, T. Phytoplankton community composition across the West Wind Drift off South America. Deep Sea Res. 1982, 29, 1229–1243. [Google Scholar] [CrossRef]
- Belmadani, A.; Concha, E.; Donoso, D.; Chaigneau, A.; Colas, F.; Maximenko, N.; Di Lorenzo, E. Striations and preferred eddy tracks triggered by topographic steering of the background flow in the eastern South Pacific. J. Geophys. Res. Ocean. 2017, 122, 2847–2870. [Google Scholar] [CrossRef]
- Belmadani, A.; Auger, P.-A.; Maximenko, N.; Gomez, K.; Cravatte, S. Similarities and contrasts in time-mean striated surface tracers in Pacific eastern boundary upwelling systems: The role of ocean currents in their generation. Fluids 2021, 6, 455. [Google Scholar] [CrossRef]
- Belkin, I.M. Remote sensing of ocean fronts in marine ecology and fisheries. Remote Sens. 2021, 13, 883. [Google Scholar] [CrossRef]
- Koshlyakov, M.N.; Tarakanov, R.Y. Intermediate water masses in the southern part of the Pacific Ocean. Oceanology 2005, 45, 455–473. [Google Scholar]
- Stramma, L.; Peterson, R.G. The South Atlantic Current. J. Phys. Oceanogr. 1990, 20, 846–859. [Google Scholar] [CrossRef]
- Stramma, L. The South Indian Ocean Current. J. Phys. Oceanogr. 1992, 22, 421–430. [Google Scholar] [CrossRef]
- Stramma, L.; Lutjeharms, J.R.E. The flow field of the subtropical gyre of the South Indian Ocean. J. Geophys. Res. Ocean. 1997, 102, 5513–5530. [Google Scholar] [CrossRef]
- James, C.; Tomczak, M.; Helmond, I.; Pender, L. Summer and winter surveys of the Subtropical Front of the southeastern Indian Ocean 1997-1998. J. Mar. Syst. 2002, 37, 129–149. [Google Scholar] [CrossRef]
- Wong, A.P.S.; Johnson, G.C. South Pacific Eastern Subtropical Mode Water. J. Phys. Oceanogr. 2003, 33, 1493–1509. [Google Scholar] [CrossRef]
- Hamilton, L.J. Structure of the Subtropical Front in the Tasman Sea. Deep-Sea Res. Part I 2006, 53, 1989–2009. [Google Scholar] [CrossRef]
- Rudnick, D.L.; Ferrari, R. Compensation of horizontal temperature and salinity gradients in the ocean mixed layer. Science 1999, 283, 526–529. [Google Scholar] [CrossRef]
- Vásquez, S.; Salas, C.; Sepulveda, A.; Pennino, M. Estimation and prediction of the spatial occurrence of jack mackerel (Trachurus murphyi) using Bayesian hierarchical spatial models. In Proceedings of the 8th Meeting of the South Pacific Regional Fisheries Management Organisation, Port Vila, Vanuatu, 14–18 February 2020; p. 14. [Google Scholar]
- Feng, Z.; Yu, W.; Zhang, Y.; Li, Y.; Chen, X. Habitat variations of two commercially valuable species along the Chilean waters under different-intensity El Niño events. Front. Mar. Sci. 2022, 9, 919620. [Google Scholar] [CrossRef]
- Gordeeva, V.N.; Zharova, A.D. Operational assessment of fisheries in the Southeast Pacific. Proc. Russ. State Hydrometeorol. Univ. 2016, 44, 96–103. (In Russian) [Google Scholar]
- Belkin, I.M.; Hunt, G.L.; Hazen, E.L.; Zamon, J.E.; Schick, R.S.; Prieto, R.; Brodziak, J.; Teo, S.L.H.; Thorne, L.; Bailey, H.; et al. Fronts, fish, and predators (editorial). Deep. Sea Res. Part II 2014, 107, 1–2. [Google Scholar] [CrossRef]
- Belkin, I.M. Subtropical Frontal Zone of the Southern Ocean (in preparation). Preprints 2023, 2021, 2021060183. [Google Scholar]
| Authors, Year, Reference Number | Var. * | Sensor; Satellite | Period | Region | Resolution |
|---|---|---|---|---|---|
| Artamonov, Skripaleva 2006 [21] | SST | AVHRR | 1985–2002 | 84° W | 54 km, 1 mo. |
| Artamonov, Skripaleva 2008 [22] | SST | AVHRR | 1985–2002 | 140° W–72° W | 54 km, 1 mo. |
| Artamonov et al. 2009 [23] | SST | AVHRR | 1985–2002 | S. Ocean | 54 km, 1 mo. |
| Gordeeva, Malinin 2006 [24] | SST | Misc. | 1982–2003 | 100° W–77° W | 111 km, 1 mo. |
| Krasnoborodko 2017 [25] | SST | Misc. | 1971–2000 | 170° W–85° W | 111 km, 1 mo. |
| Lebedev, Sirota 2007 [26] | SSH | TOPEX/Poseidon | 1992–2003 | 110° W–80° W | 111 km, 1 day |
| Lebedev, Sirota 2007 [26] | SST | MCSST * | 1992–2003 | 110° W–80° W | 18 km, 1 week |
| Malinin, Gordeeva 2009 [27] | SST | Misc. | 1982–2006 | 100° W–77° W | 111 km, 1 mo. |
| Sirota et al. 2004 [28] | SSH | TOPEX/Poseidon | 1992–2003 | 109° W–90° W | 111 km, 1 day |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Belkin, I.M.; Shen, X.-T. Remote Sensing of the Subtropical Front in the Southeast Pacific and the Ecology of Chilean Jack Mackerel Trachurus murphyi. Fishes 2023, 8, 29. https://doi.org/10.3390/fishes8010029
Belkin IM, Shen X-T. Remote Sensing of the Subtropical Front in the Southeast Pacific and the Ecology of Chilean Jack Mackerel Trachurus murphyi. Fishes. 2023; 8(1):29. https://doi.org/10.3390/fishes8010029
Chicago/Turabian StyleBelkin, Igor M., and Xin-Tang Shen. 2023. "Remote Sensing of the Subtropical Front in the Southeast Pacific and the Ecology of Chilean Jack Mackerel Trachurus murphyi" Fishes 8, no. 1: 29. https://doi.org/10.3390/fishes8010029
APA StyleBelkin, I. M., & Shen, X.-T. (2023). Remote Sensing of the Subtropical Front in the Southeast Pacific and the Ecology of Chilean Jack Mackerel Trachurus murphyi. Fishes, 8(1), 29. https://doi.org/10.3390/fishes8010029







