1. Introduction
Salinity is an important feature of aquatic environments, and maintaining ionic homeostasis is crucial for optimal function of cellular and physiological processes [
1]. Ionic stress, induced by increases in salinity, therefore has the potential to disrupt these processes. Increasing the salinity of freshwater environments is exacerbated by rising sea levels as a consequence of global climate change [
2] and by road salt run-off [
3], and poses an important threat to aquatic ecosystems. For instance, a long-term monitoring study found that salt concentrations of freshwater streams in Milwaukee, USA, frequently exceed 1000 mg/L (1 ppt) and extended to nearly 7000 mg/L (7 ppt) during road salt application periods in February–March, a range which included concentrations that were lethal to adult fathead minnows in a chronic bioassay [
4]. Freshwater habitats face an additional source of increased salinity in the form of saltwater intrusion as a consequence of intensive aquaculture, which is especially prominent in southeast Asia as a consequence of intensive shrimp farming [
5]. Therefore, it is important to characterise the potential effects of increasing environmental salinity on aquatic organisms.
Fully-developed animals are able to respond adaptively to unfavourable chemical environments via physiological or behavioural responses (e.g., escaping from the adverse environment), however, animals at earlier stages of development—particularly embryos—may be far more sensitive to adverse environments. Osmoregulation in teleost fishes is carried out by the coordination of several organs and organ systems (e.g., gills, kidneys, urinary system), but these structures are not differentiated in early embryos, which instead rely on ion pumping activity by ionocytes covering the skin [
6]. Importantly, developing embryos are immobile while confined to the egg casing and thus cannot easily escape from unfavourable conditions as adult animals can.
The zebrafish (
Danio rerio), a freshwater fish, is an ideal model system for the effects of physiological stress on the early embryo, given that their eggs are fertilised externally and can be easily observed throughout early development. Their rapid development into free-swimming larvae within the first five days of life also makes them suitable for high-throughput experiments. Zebrafish are thought to be tolerant to a range of salinities in their natural environments, which have been recorded to range from 0.1–0.6 ppt, thus technically extending to brackish conditions (>0.5 ppt) [
7]. However, this tolerance range has not been well-characterised. Increasing rearing salinity has previously been shown to induce higher mortality on the embryos of non-marine fish, including zebrafish [
8,
9] and medaka [
10], but the propensity of embryos for plastic responses under ionic stress conditions has not been investigated. One such trait which may be subject to plasticity is hatching—an important life history event in egg-laying species which exhibits plasticity in response to biotic and abiotic stress [
11]. In zebrafish, hatching time is mostly determined by the rate of development (largely controlled by temperature [
12]), but can also be influenced by light regimes, suggesting a role of circadian chemistry in the regulation of hatching [
13]. Overall, however, hatching plasticity has not been extensively studied in zebrafish and has not been evaluated in the context of salinity.
This study first aimed to (1) characterise the effect of salinity on the mortality of zebrafish embryos; and (2) assess the effects of different salt concentrations on the timing of hatching.
2. Results and Discussion
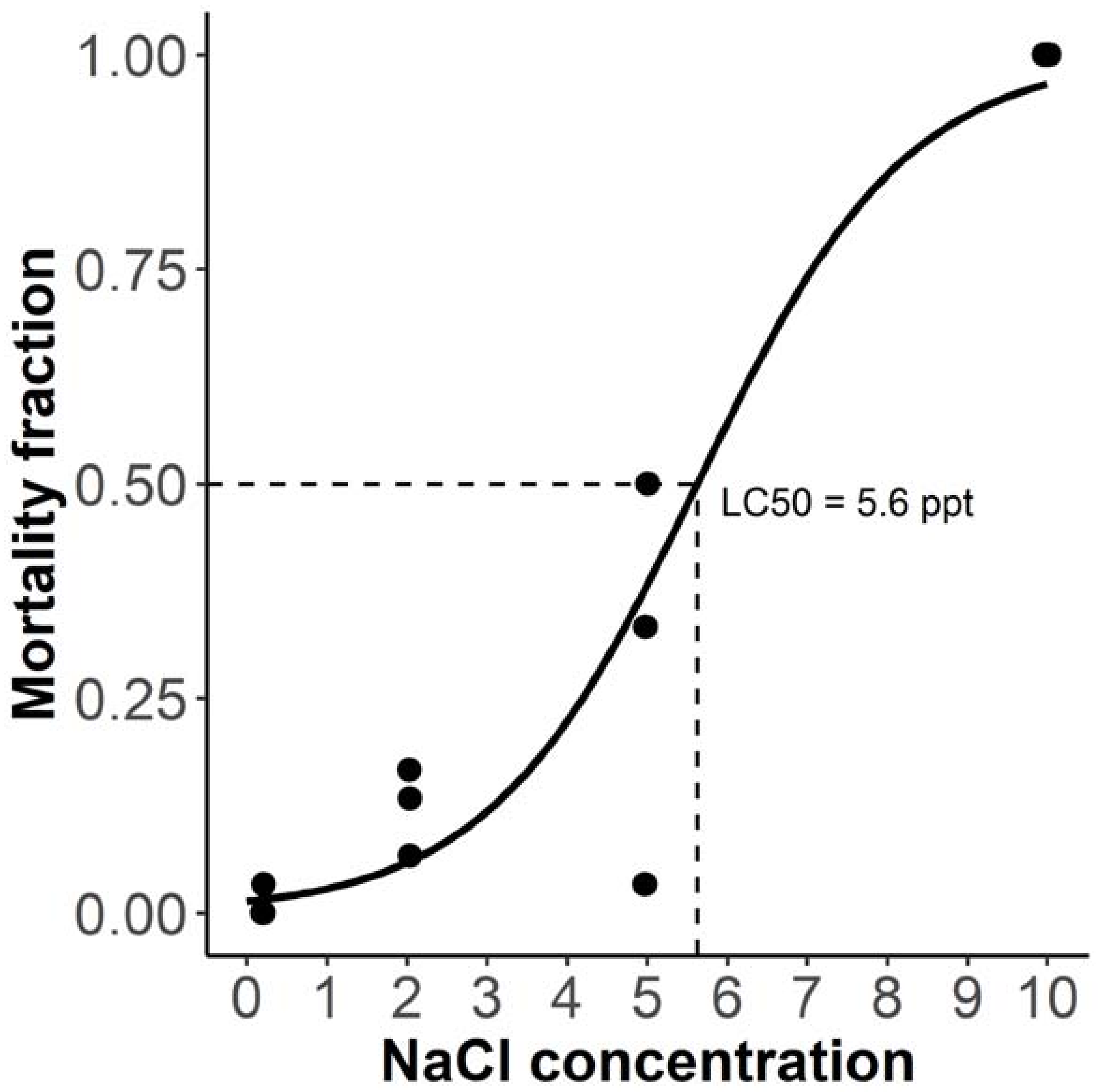
Initially, the 24-h mortality of embryos was assessed using four NaCl concentrations (0.2, 2, 5, and 10 parts per thousand), the lowest of which was the concentration of aquarium water. Exposure commenced <2 h postfertilisation (hpf), when embryos were in the 4–64-cell stage. The relationship between concentration and mortality was modelled using a generalised linear model, for which the goodness of fit was assessed using pseudo R
2, calculated as the inverse of the ratio of the residual deviance to the null deviance. The model had a pseudo R
2 of 0.87 and showed a significant effect of concentration on 24-h mortality (
p < 0.001). The 10 ppt concentration was lethal to all embryos, 5 ppt induced moderate mortality (up to 50%), while mortality among 2 ppt embryos was noticeable but did not exceed 20% (
Figure 1). As predicted from the model, the LC50 of NaCl was approximately 5.6 parts per thousand.
The results are generally concordant with previously published experiments, e.g., Haque et al. [
9] showed that 21% of embryos survived to hatching in 6 ppt salinity, while none survived in 10 ppt. One brood exposed to 5 ppt did not exhibit appreciable mortality at 24 h, which may be reflective of genetic differences in the osmoregulatory capacity of early embryos. However, as within-brood replication was not performed, the reason for this discrepancy is unclear.
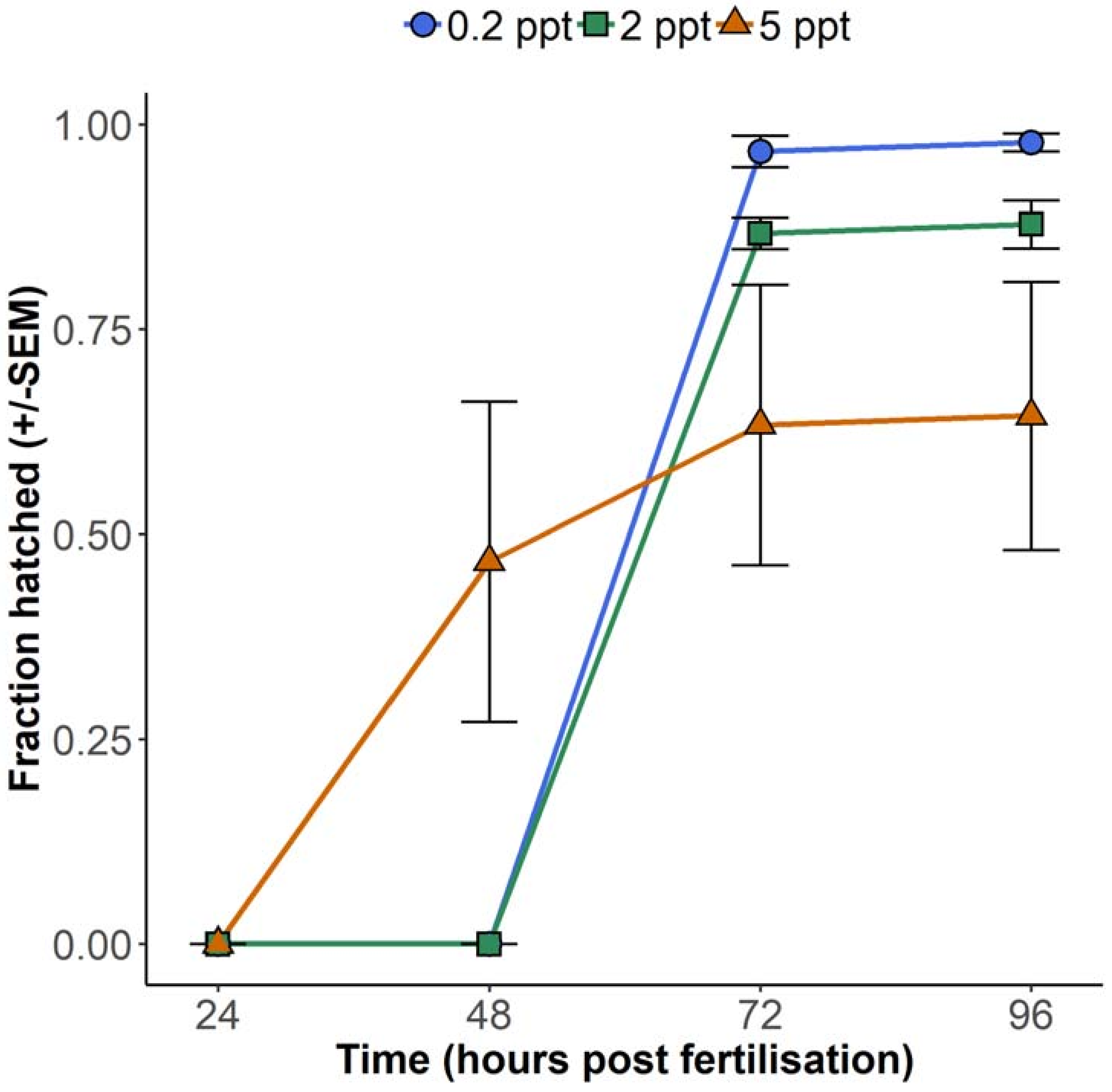
Although 5 ppt resulted in fewer embryos hatched at 72 hpf, embryos reared at this concentration hatched earlier than their lower salinity counterparts. Hatching at 0.2 and 2 ppt was not observed until 72 hpf, while most of the surviving embryos exposed to 5 ppt hatched 24 h earlier than at these lower concentrations, at 48 hpf (
Figure 2). For statistical evaluation of the interaction between concentration and hatching time, a linear mixed model was fitted, which accounted for repeated measures from the same dishes. This model showed a significant interaction between 5 ppt salinity and time (
t-test with Kenward Roger approximation of degrees of freedom, T
24 = 2.1,
p = 0.045).
A previously study demonstrated that the lethality of salinity to zebrafish embryos depended on the developmental stage at which they are exposed [
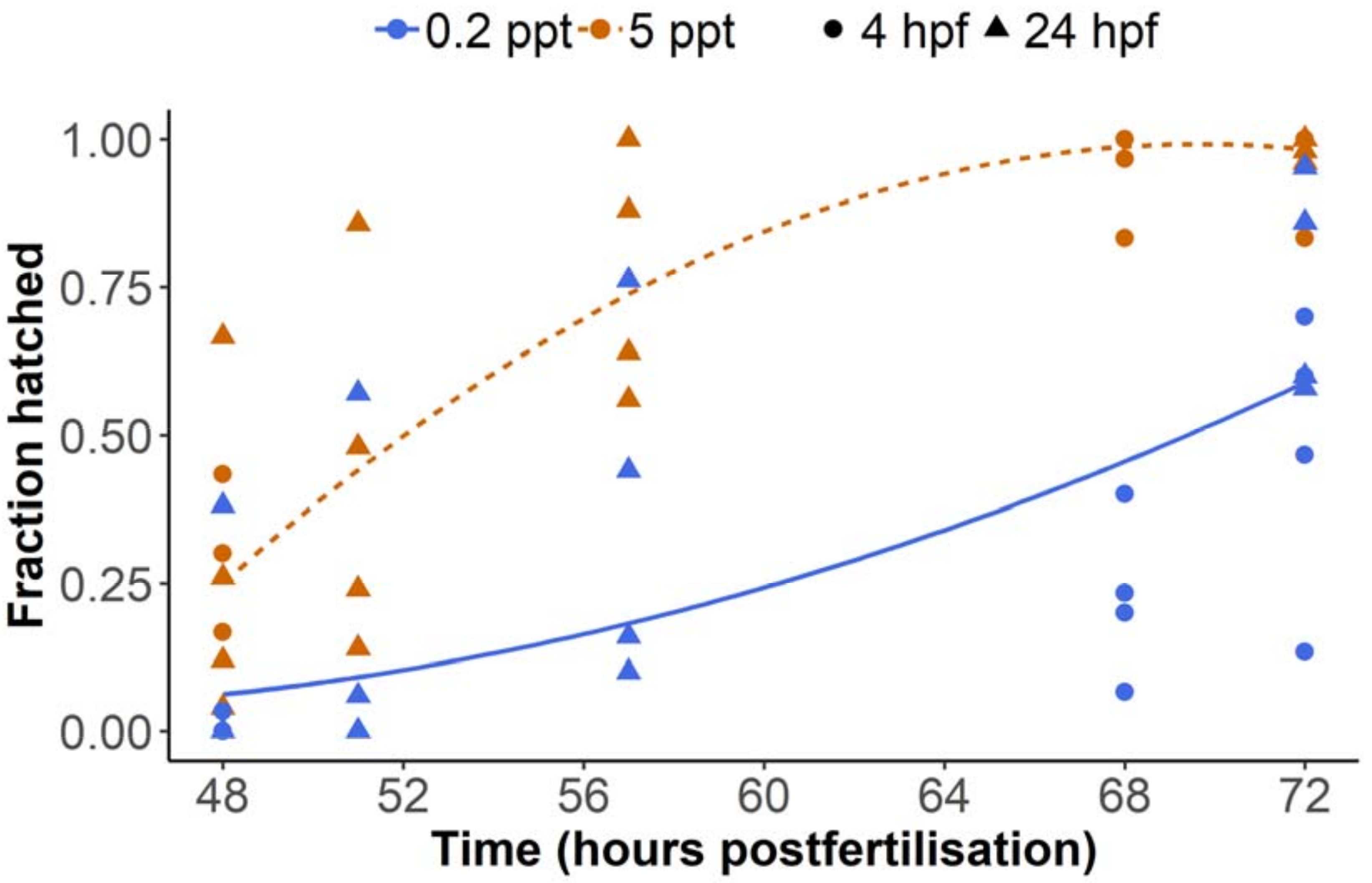
8]. Embryos become more tolerant of salinity from the blastula and gastrula stages compared to the cleavage stage, presumably due to more advanced osmoregulatory capacities. Therefore, the present study also examined hatching data from two additional experiments, in which groups of embryos were exposed to 0.2 or 5 ppt salinity beginning from the late blastula stage or later (4 or 24 hpf depending on the experiment), and hatching was monitored between 48 and 72 h. Concordant with the first experiment (
Figure 2), 5 ppt embryos showed increased hatching from 48 hpf (
Figure 3). A linear mixed model with time as a quadratic term revealed that hatching % was significantly higher at 5 ppt throughout the observation period (F-test with Kenward Roger approximation of degrees of freedom, F
1,13.3 = 18.4,
p < 0.001) and that there was a significant interaction between treatment and time (F
2,37.1 = 7.6,
p = 0.002). Five ppt salinity did not induce appreciable mortality, as overall hatching success was not impeded (close to 100% for most 5 ppt dishes at 72 hpf). This suggests that later stage embryos are resilient to a concentration that is lethal to earlier stage embryos and thus supports previously published observations [
8]. Hatching at 0.2 ppt was noticeably lower at 72 hpf compared to the first experiment (
Figure 2), which may be attributable to other environmental differences relating to the embryos having been transferred from egg collection dishes to petri dishes at a later time (see Materials and Methods). Importantly, these data indicate that increased salinity still prompts earlier hatching even at later stages, when embryos are supposedly more physiologically tolerant.
Overall, the results of the present study demonstrate that zebrafish embryo hatching is plastic in response to potentially stressful or lethal changes in salinity. Premature hatching has previously been observed in tree frog embryos upon snake attacks [
11,
14] and in zebrafish embryos in response to environmental toxicants [
15], suggesting that earlier hatching is an adaptive response which allows the animal an opportunity to escape unfavourable conditions. As the salt concentration which induced earlier hatching induced mortality of early embryos, the results of this study are in alignment with this paradigm. It could be inferred that as external ion concentrations threaten to exceed the capacity of ionocytes to regulate internal osmolarity, ‘emergency’ pathways are triggered, which accelerate the hatching process. Hatching is triggered following induction of hatching enzyme synthesis [
16], and thus such emergency response pathways would likely involve mechanisms by which hatching enzyme synthesis is induced earlier or at greater levels than normal. Given their higher tolerance to salinity, this response would be less urgent for later stage embryos, however it would still provide an advantage, assuming heightened salinity negatively affects other aspects of fitness.
It must be conceded that this study did not ascertain whether earlier hatching in response to increased salinity was driven by accelerated development rate (as is the case for temperature [
12]) or whether embryos hatched at an earlier developmental stage. As zebrafish hatching time appears to be controlled partially by light, via the circadian clock [
13], it is also possible that ionic stress induces earlier hatching by disruption of circadian clock chemistry.
It must also be conceded that the present study has examined the effect of salinity in only one strain of zebrafish (London wildtype). Under common husbandry practices, laboratory zebrafish are reared in low-salinity conditions (0.25–0.75 ppt) [
7], and it is not known whether domestication has affected salinity tolerance relative to wild varieties, which may be tolerant of a wider range of salinities. From comparative studies of different laboratory strains and wild varieties, insight could therefore be gained into the effect of domestication on salinity tolerance. Furthermore, as zebrafish are thought to be tolerant of a range of salinities in the wild [
7], the embryo responses observed in this study may not be entirely representative of stenohaline species, and it would therefore be pertinent to compare salinity-induced hatching plasticity across a range of stenohaline and euryhaline species.
In conclusion, the results of this study both support and expand upon previously published findings on the effects of salinity on early zebrafish embryos, showing that marked plasticity occurs in hatch timing in response to a concentration that is close to the LC50 for early embryos. The phenomenon warrants more detailed characterisation, as salinity-induced hatching plasticity could serve as a useful phenotypic reference for studies of osmoregulatory and stress response capacities of the early embryo. Furthermore, the results suggest that embryonic adaptive plasticity may be an important component of the response of aquatic organisms to changes in environmental salinity.
3. Materials and Methods
Adult London wildtype zebrafish were maintained in a UK Home Office approved facility in the Department of Animal and Plant Sciences, University of Sheffield. The collection of embryos from adult zebrafish was carried out under the UK Home Office project license number 40/3704. As no NaCl exposures were carried out on animals older than 5 days post-fertilisation and no exposed embryos were kept beyond 5 dpf, the exposure of embryos in the experiment did not comprise a regulated procedure as specified under the Animals (Scientific Procedures) Act (1986), and thus did not require specific approval.
Embryos were derived from adult London wildtype
Danio rerio which were reared and housed on a recirculatory system with water heated to approx. 26 °C. Animals were fed twice daily with live
Artemia nauplii or commercial flake food. Egg collection dishes filled with marbles were placed in mixed sex tanks at approx. 1600 and were collected the following morning. Three clutches of fertilised eggs were derived from three separate sets of mixed-sex adult zebrafish, and each clutch was considered to be a biological replicate. Although the exact time of fertilisation could not be determined, zebrafish typically spawn at first light [
17], which was between 08.00 and 08.30 in the aquarium where the present study was carried out. At less than 2 h post-fertilisation (before 10.00), four sets of 30 embryos at the 4–64-cell stage (identified under light microscope) were taken from each clutch and were placed in lidded 90 mm petri dishes containing each of the four salt concentrations (0.2, 2, 5, and 10 parts per thousand). The lowest was 0.2 ppt, as this was the salinity of aquarium water with no additional salt. Higher concentrations were prepared by dissolving ‘Instant Ocean’ salt in aquarium water and concentrations were checked using a digital salinity meter. Embryo dishes were kept in a temperature-controlled room at 25 °C on a 12:12 light/dark cycle, and were checked for mortality and hatching after 24, 48, and 72 h of exposure.
For the experiments in which embryos were exposed to increased salinity at later developmental stages, embryos were collected in the same manner as described above, again with each set of embryos deriving from a separate set of adult zebrafish. Marbles were removed from the egg collection dishes and water was changed, and embryos remained in these dishes until being transferred to petri dishes for exposure to either 0.2 or 5 ppt NaCl. For one experiment, embryos were transferred at approx. 4 hpf (around 12.00 on day of egg collection), while for another experiment they were transferred at 24 hpf. The mixed model fit to this data did not reveal a significant effect of the experiment on the response variable (F1,13.9 = 2.6, p = 0.13).
Graphical generation and statistical analyses were carried out in ‘R’ version 3.5.1 [
18]. The generalised linear model for 24-h mortality was fitted using the glm function from the ‘stats’ package and LC50 value predicted form the model using the predict function from the same package. Mixed effects models fit using the lmer function from the ‘lme4′ package [
19] were used to model the interaction between concentration and time on hatching, with ‘Dish ID’ entered as a random factor. For the analysis of hatching data of later stage embryos, time was included as a second-degree polynomial (quadratic) term. Model results were derived using the summary and anova functions with significance values (
t-tests and F-tests with Kenward Roger approximation of degrees of freedom) obtained using the ‘lmerTest’ [
20] and ‘pbkrtest’ [
21] packages. Curve fits shown in
Figure 2 were derived using the ‘effects’ package [
22] and a linear mixed model.