Reproductive Biology of Puntius sophore in Bangladesh
Abstract
:1. Introduction
2. Results and Discussion
2.1. Calculation of Total Length, Body Weight, and Ovary Weight of P. sophore
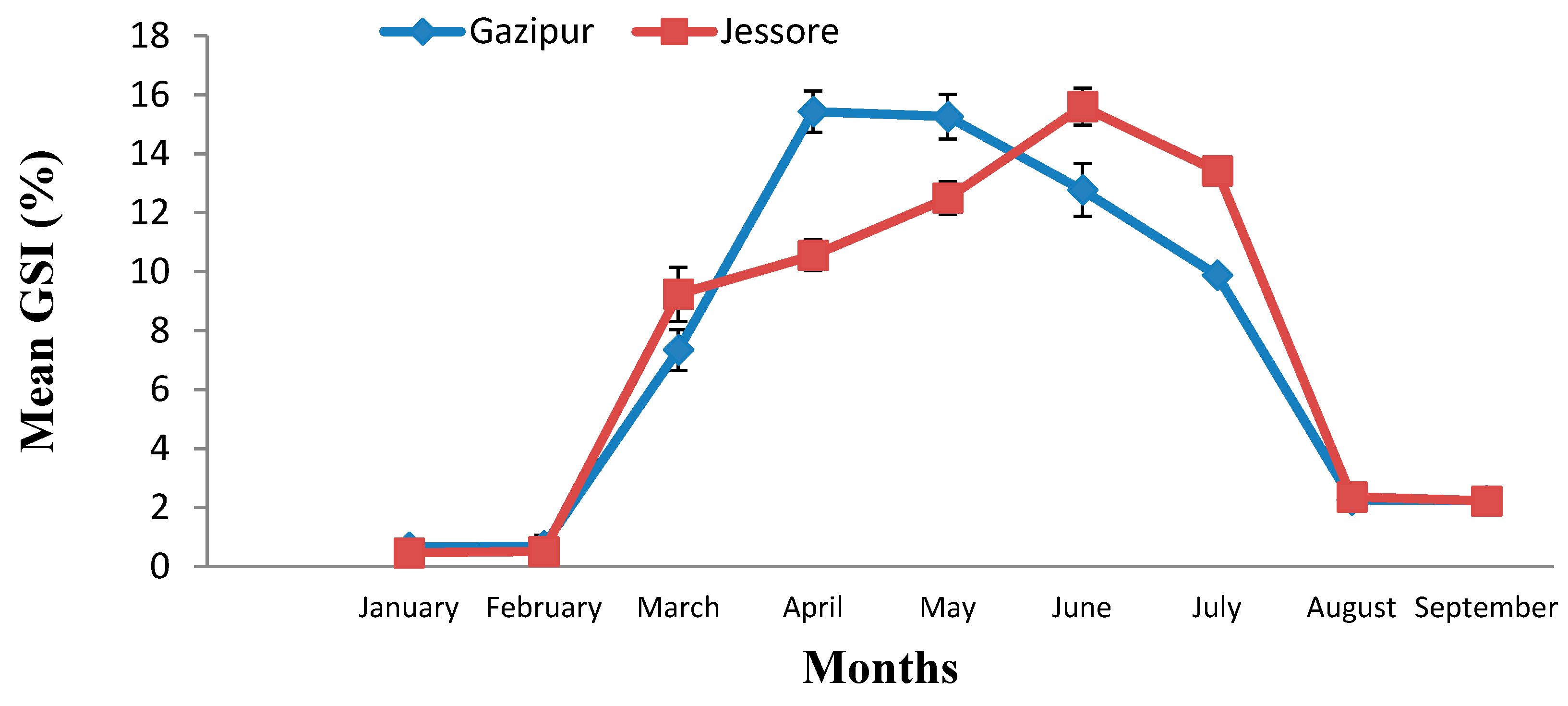
2.2. Analysis of Gonadosomatic Index of P. sophore
2.3. Calculation of Fecundity of P. sophore
2.4. Oocyte Diameter of P. sophore
2.5. Annual Changes in the Ovary
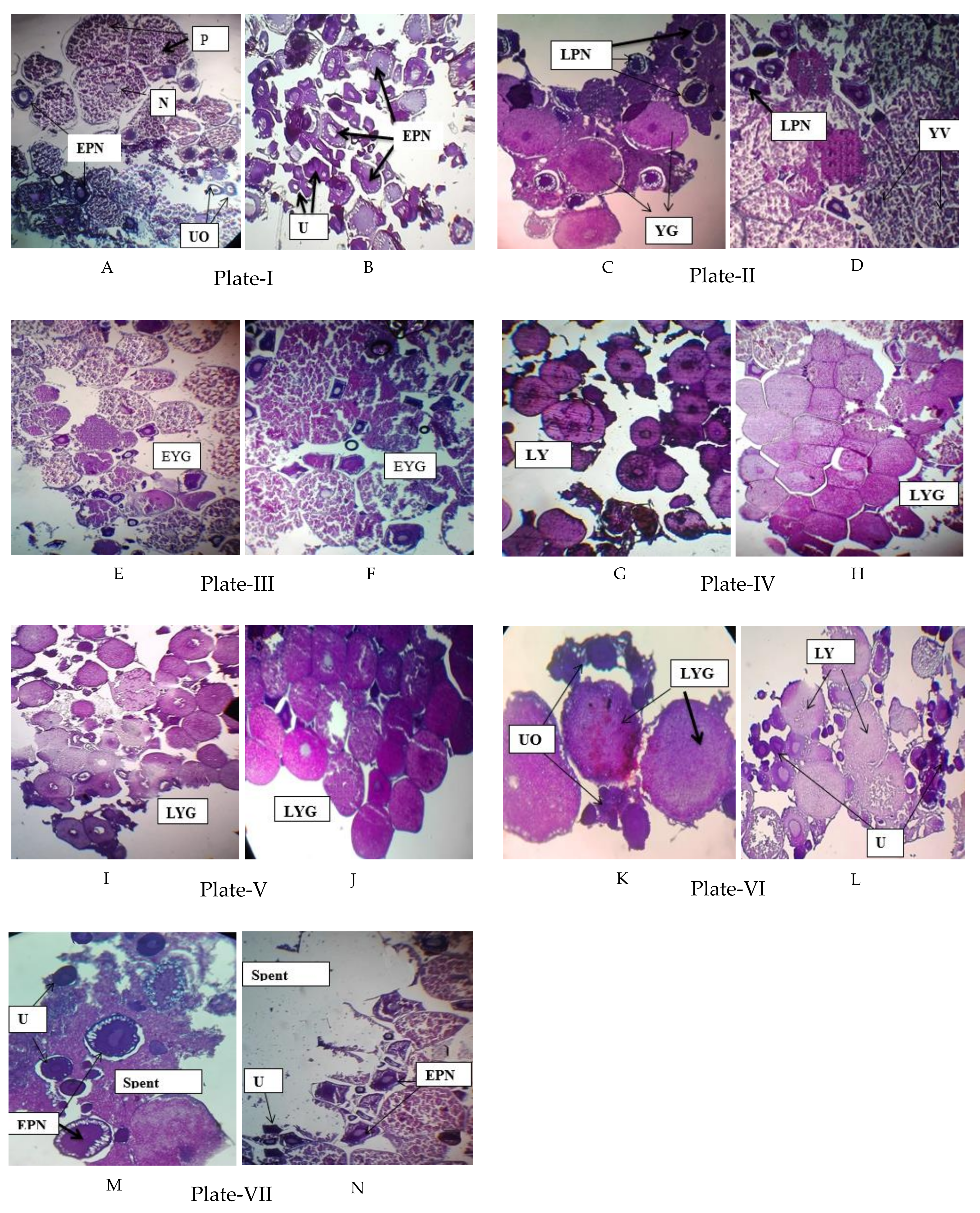
2.6. Histological Observation of Oocyte
2.6.1. Undeveloped Oocyte
2.6.2. Early Perinucleolar Stage
2.6.3. Late Perinucleolar Stage
2.6.4. Yolk Vesicle Stage
2.6.5. Early Yolk Granule Stage
2.6.6. Late Yolk Granule Stage
2.6.7. Spent Phase
2.6.8. Maturity Stage of Female Gonads in P. sophore
2.7. Meteorological Information (Rainfall and Temperature)
3. Materials and Methods
3.1. Sample and Data Collection
3.2. Gonadosomatic Index (GSI)
3.3. Estimation of Fecundity
3.4. Histology of Gonad
3.5. Collection of Meteorological Information (Rainfall and Temperature)
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- IUCN Bangladesh. Red List of Bangladesh: A Brief on Assessment Result 2015; International Union for Conservation of Nature (IUCN), Bangladesh Country Office: Dhaka, Bangladesh, 2015; p. 24. [Google Scholar]
- IUCN Bangladesh. Red List of Bangladesh Volume 5: Freshwater Fishes; International Union for Conservation of Nature (IUCN), Bangladesh Country Office: Dhaka, Bangladesh, 2015; p. xvi+360. [Google Scholar]
- Talwar, P.K.; Jhingran, A.G. Inland Fishes of India and Adjacent Countries; Oxford and IBH Publishing Co. Pvt. Ltd.: New Delhi, India, 1991; Volume 2. [Google Scholar]
- Petr, T. Coldwater fish and fisheries in Bhutan. In Fish and Fisheries at Higher Altitudes: Asia; Petr, T., Ed.; FAO Fisheries Technical Paper No. 385; FAO: Rome, Italy, 1999; pp. 6–12. [Google Scholar]
- Menon, A.G.K. Checklist—Fresh Water Fishes of India; Zoological Survey of India, Occasional Paper No. 175; Miscellaneous Publications: Delhi, India, 1999; 366p. [Google Scholar]
- Craig, J.F.; Halls, A.S.; Barr, J.J.F.; Bean, C.W. The Bangladesh floodplain fisheries. Fish. Res. 2004, 66, 271–286. [Google Scholar] [CrossRef]
- Food and Agriculture Organization (FAO). Management Guideline for Asian Floodplain River Fisheries. Part 2: Summary of DFID Research; FAO: Rome, Italy, 1996; pp. 32–34. [Google Scholar]
- Samad, M.A.; Galib, S.M.; Flowra, F.A. Fish drying in Chalan Beel areas, Bangladesh. J. Sci. Ind. Res. 2009, 44, 461–466. [Google Scholar]
- Shafi, M.; Quddus, M.M.A. Bangladesher Matshya Sampad (Fishery Resources of Bangladesh); Bangla Academy: Dhaka, Bangladesh, 1982; 444p. [Google Scholar]
- Rahman, A.K.A. Freshwater fishes of Bangladesh, 2nd ed.; Zoological Society of Bangladesh, Department of Zoology, University of Dhaka: Dhaka, Bangladesh, 2005; pp. 66–67. [Google Scholar]
- Roos, N.; Leth, T.; Jakobsen, J.; Thilsted, S.H. High vitamin A content in some small indigenous fish species in Bangladesh: Perspectives for food-based strategies to reduce vitamin A deficiency. Int. J. Food Sci. Nutr. 2002, 53, 425–437. [Google Scholar] [CrossRef] [PubMed]
- Thilsted, S.H. The importance of small indigenous fish species for improved human nutrition in rural Bangladesh. In Small Indigenous Species of Fish in Bangladesh; Wahab, M.A., Thilsted, S.H., Hoq, M.E., Eds.; Bangladesh Agricultural University: Mymensingh, Bangladesh, 2003; 116p. [Google Scholar]
- Thilsted, S.H.; Roos, N.; Hassan, N. The role of small indigenous fish species in food and nutrition security in Bangladesh. Naga ICLARM Q. 1997, 20, 82–84. [Google Scholar]
- Froese, R.; Pauly, D. FishBase 2011. World Wide Web Electronic Publication. 2011. Available online: http://www.fishbase.org (accessed on 12 December 2016).
- Lagler, K.F. Enumeration of fish eggs. In Freshwater Fishery Biology, 2nd ed.; W.M.C. Brown Co.: Dubuque, IA, USA, 1956; pp. 106–110. [Google Scholar]
- Wootton, R.J. Ecology of Teleost Fishes; Chapman and Hall: London, UK, 1995. [Google Scholar]
- Choudhury, T.G.; Singh, S.K.; Baruah, A.; Das, A.; Parhi, A.; Bhattacharjee, P.; Biswas, P. Reproductive Features of Puntius sophore (Hamilton 1822) from Rivers of Tripura, India. Fish. Technol. 2015, 52, 140–144. [Google Scholar]
- Chandrasoma, J.; De-Silva, S.S. Reproductive biology of Puntius sarana, an indigenous species and Tilapia rendalli, an exotic, in an ancient man-made lake in Sri Lanka. Fish. Manag. 1981, 12, 17–28. [Google Scholar] [CrossRef]
- Hossain, M.A.R.; Wahab, M.A. Bangladesher Nadir Machher Jibaboichitra (in Bengali); Matshya Saptaha Songkolan (Proceeding published in Fisheries Week, 2012); Department of Fisheries (DoF): Dhaka, Bangladesh, 2012. [Google Scholar]
- Agarwal, A. Studies on the Karyotype and Histological Changes in Gonads of Some Fishes of Jammu and Kashmir. Ph.D. Thesis, University of Jammu, Jammu, India, 1982. [Google Scholar]
- Kant, K.J.; Gupta, K.; Langer, S. Fecundity in fish Puntius sophore and relationship of fecundity with fish length, fish weight and ovary weight from Jammu water bodies J and K (India). Int. J. Fish. Aquac. Sci. 2016, 6, 99–110. [Google Scholar]
- Tracey, S.R.; Lyle, J.; Haddon, M. Reproductive biology and per-recruit analyses of striped trumpeter (Latris lineata) from Tasmania, Australia: Implications for management. Fish. Res. 2007, 84, 358–368. [Google Scholar] [CrossRef]
- Tareque, A.M.H.B.; Biswas, B.; Hossain, M.S.; Rahman, M.M.; Rahman, M.M. Some aspects of Puntius sophore (Hamilton) collected from the Mouri River, Khula, Bangladesh. Bangladesh Res. Publ. J. 2009, 2, 406–422. [Google Scholar]
- Bithy, K.; Miah, M.I.; Haque, M.S.; Hasan, K.R.; Islam, M.F. Estimation of the Fecundity of Jat Puti, Puntius sophore (Hamilton). J. Environ. Sci. Nat. Resour. 2012, 5, 295–300. [Google Scholar] [CrossRef]
- Das, D.R. A Study on the Fecundity of Featherback, Notopterus notopterus (pallas) from two Different Sources (Pond and Beel). Master’s Thesis, Department of Aquaculture, Bangladesh Agricultural University, Mymensingh, Bangladesh, 1998; 70p. [Google Scholar]
- Chakraborty, B.K.; Mirza, Z.A.; Miah, M.I.; Habib, M.A.B.; Chakraborty, A. Reproductive Cycle of the Endangered Sarpunti, Puntius sarana (Hamilton, 1822) in Bangladesh. Asian Fish. Sci. 2007, 20, 145–164. [Google Scholar]
- Alam, M.M.; Ahmad, S.; Hussain, M.A. Ovarian development and reproductive cycle of two freshwater barbs, Puntius sophore and Puntius ticto. Front. Biol. Life Sci. 2014, 2, 12–15. [Google Scholar] [CrossRef]
- Mollah, M.F.A. Cyclic changes in the ovary of freshwater catfish, Clarias macrocephalus (Gunther). Indian J. Fish. 1986, 33, 54–65. [Google Scholar]

| No. of Fish Examined | Mean and Standard Deviation (±SD) | |||
|---|---|---|---|---|
| Total Length (cm) | Body Mass (g) | Ovary Mass (g) | ||
| January | 20 | 7.43 ± 0.29 | 7.83 ± 0.49 | 0.0104 ± 0.01 |
| February | 20 | 7.42 ± 0.30 | 7.89 ± 0.58 | 0.0352 ± 0.01 |
| March | 20 | 8.72 ± 0.39 | 9.23 ± 0.77 | 0.6758 ± 0.09 |
| April | 20 | 10.06 ± 0.59 | 13.0 ± 1.84 | 2.0183 ± 0.46 |
| May | 20 | 9.42 ± 0.46 | 12.06 ± 0.87 | 1.842 ± 0.24 |
| June | 20 | 8.96 ± 0.58 | 10.07 ± 1.68 | 1.153 ± 0.24 |
| July | 20 | 8.81 ± 0.31 | 9.54 ± 0.84 | 0.9478 ± 0.22 |
| August | 20 | 7.14 ± 0.44 | 7.53 ± 0.47 | 0.1697 ± 0.03 |
| September | 20 | 6.9 ± 0.65 | 7.355 ± 1.64 | 0.1717 ± 0.08 |
| No. of Fish Examined | Mean ± SD | |||
|---|---|---|---|---|
| Total Length (cm) | Body Weight (g) | Ovary Weight (g) | ||
| January | 20 | 7.08 ± 0.17 | 7.2 ± 0.39 | 0.033 ± 0.02 |
| February | 20 | 6.99 ± 0.46 | 7.19 ± 0.58 | 0.034 ± 0.02 |
| March | 20 | 7.22 ± 0.38 | 7.55 ± 0.86 | 0.71 ± 0.16 |
| April | 20 | 7.98 ± 0.57 | 8.19 ± 0.63 | 0.88 ± 0.26 |
| May | 20 | 8.46 ± 0.30 | 8.99 ± 0.33 | 1.126 ± 0.15 |
| June | 20 | 9.93 ± 0.23 | 10.32 ± 0.85 | 1.61 ± 0.28 |
| July | 20 | 8.88 ± 0.42 | 9.28 ± 0.48 | 1.24 ± 0.11 |
| August | 20 | 7.18 ± 0.49 | 7.34 ± 1.29 | 0.178 ± 0.07 |
| September | 20 | 7.07 ± 0.38 | 7.2 ± 1.15 | 0.166 ± 0.05 |
| Month | Fecundity (Mean ± SD) | |
|---|---|---|
| Gazipur | Jessore | |
| March | 2601 ± 333.37 | 1870 ± 272.84 |
| April | 5053 ± 878.26 | 2328 ± 193.00 |
| May | 4550 ± 345.86 | 3550 ± 547.2 |
| June | 2659 ± 570.52 | 5433 ± 968.25 |
| July | 2764 ± 394.25 | 4032 ± 622.68 |
| August | 523 ± 169.23 | 735 ± 87.18 |
| September | 616 ± 77.56 | 516 ± 175.23 |
| Months | Mean ± SD | |||
|---|---|---|---|---|
| Ovary Weight (g) in Jessore | Oocyte Diameter (mm) in Jessore | Ovary Weight (g) in Gazipur | Oocyte Diameter (mm) in Gazipur | |
| January | 0.033 ± 0.02 | - | 0.0104 ± 0.01 | - |
| February | 0.034 ± 0.02 | - | 0.0352 ± 0.01 | - |
| March | 0.71 ± 0.16 | 0.61 ± 0.03 | 0.6758 ± 0.09 | 0.71 ± 0.05 |
| April | 0.88 ± 0.26 | 0.65 ± 0.04 | 2.0183 ± 0.46 | 0.79 ± 0.03 |
| May | 1.126 ± 0.25 | 0.678 ± 0.04 | 1.842 ± 0.24 | 0.72 ± 0.04 |
| June | 1.61 ± 0.28 | 0.72 ± 0.04 | 1.153 ± 0.24 | 0.69 ± 0.03 |
| July | 1.24 ± 0.29 | 0.66 ± 0.04 | 0.9478 ± 0.22 | 0.65 ± 0.05 |
| August | 0.178 ± 0.07 | 0.54 ± 0.03 | 0.1697 ± 0.04 | 0.61 ± 0.05 |
| September | 0.166 ± 0.05 | 0.51 ± 0.03 | 0.1717 ± 0.08 | 0.51 ± 0.04 |
| Maturity Stage | Period Name | Description |
|---|---|---|
| I | Immature | Chromatin threads with no visible eggs. Growth oocyte begins, nucleus increases in size, and multiple nucleoli become located. |
| II | Previtellogenesis | A follicular layer is formed around the oocyte. Beginning of the formation of the chorion. |
| III | Vitellogenesis | Formation of yolk vesicle in the periphery of the oocytes. Yolk vesicles developed as minute bodies but gradually increased in size and number. |
| IV | Maturation | The number of yolk granules was sharply increasing. They are densely packed and occupy almost the total volume of the oocyte. |
| V | Spent | At this stage, the oocyte enters the ovulatory phase. Different stages of ova were found, mostly immature ones with a few ripe ones. Polar bodies are produced and oocytes are released to the exterior body of fish. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hasan, T.; Hossain, M.F.; Mamun, M.; Alam, M.J.; Salam, M.A.; Rafiquzzaman, S.M. Reproductive Biology of Puntius sophore in Bangladesh. Fishes 2018, 3, 22. https://doi.org/10.3390/fishes3020022
Hasan T, Hossain MF, Mamun M, Alam MJ, Salam MA, Rafiquzzaman SM. Reproductive Biology of Puntius sophore in Bangladesh. Fishes. 2018; 3(2):22. https://doi.org/10.3390/fishes3020022
Chicago/Turabian StyleHasan, Tajmira, Md. Foysul Hossain, Md. Mamun, Md. Jahangir Alam, Mohammad Abdus Salam, and S. M. Rafiquzzaman. 2018. "Reproductive Biology of Puntius sophore in Bangladesh" Fishes 3, no. 2: 22. https://doi.org/10.3390/fishes3020022
APA StyleHasan, T., Hossain, M. F., Mamun, M., Alam, M. J., Salam, M. A., & Rafiquzzaman, S. M. (2018). Reproductive Biology of Puntius sophore in Bangladesh. Fishes, 3(2), 22. https://doi.org/10.3390/fishes3020022





