Build Your Own Microscope: Step-By-Step Guide for Building a Prism-Based TIRF Microscope
Abstract
1. Introduction
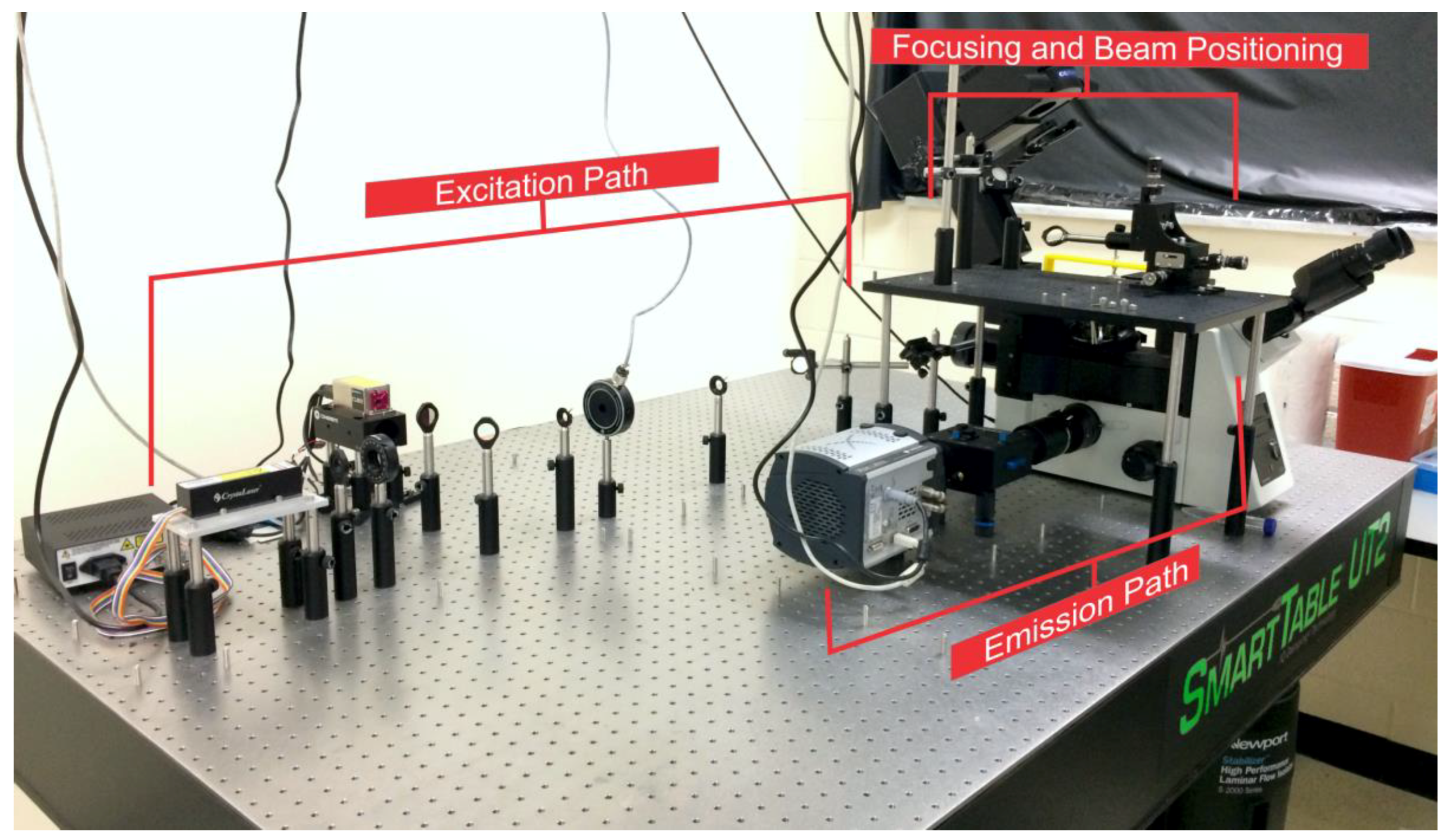
1.1. Excitation Light Source and Filters
1.2. Focusing and Beam Positioning
1.3. Emission Path
2. Space Design
3. Safety Considerations
- Block the surroundings using black laser curtains.
- Use the sign ‘laser in use’ to warn outsiders so that they may take necessary precautions before entering the area.
- Wear the appropriate laser safety goggles when using the laser and entering the laser area.
- Keep the shutter closed when the laser source is not required.
- Never look directly into the light path when the shutter is open.
4. Microscope Assembly
4.1. Optics Installation
- Assemble optics on the laser table in a straight line along a single plane as specified in the instrument optical diagram.
CRITICAL STEP Later steps will require that the lasers are aligned such that the beams are traveling co-linear. Ensuring that the lasers, emitters, and optics are mounted level with one another can prevent headaches down the line.
- Build the elevated platform 90° to the end of the optical path next to the area intended for the microscope.
- The lasers must be adjusted so that both beams pass through both irises. The iris closest to the laser is first narrowed, taking care not to close the iris completely, and the lasers readjusted so that both beams pass through its center. This step is then repeated with the second iris which is narrowed and the lasers adjusted so that the beams now pass through the center of both irises [12].Note: This process may take several repeated steps of narrowing one or both irises and adjusting the lasers to achieve total centering of the beams. Fine tuning of the leveling can be accomplished by allowing the lasers to leave the laser table and making minute adjustments to ensure that the points cast by both lasers hit at the same point on a wall. From this point onwards, the optics should not be adjusted unless something comes out of alignment.
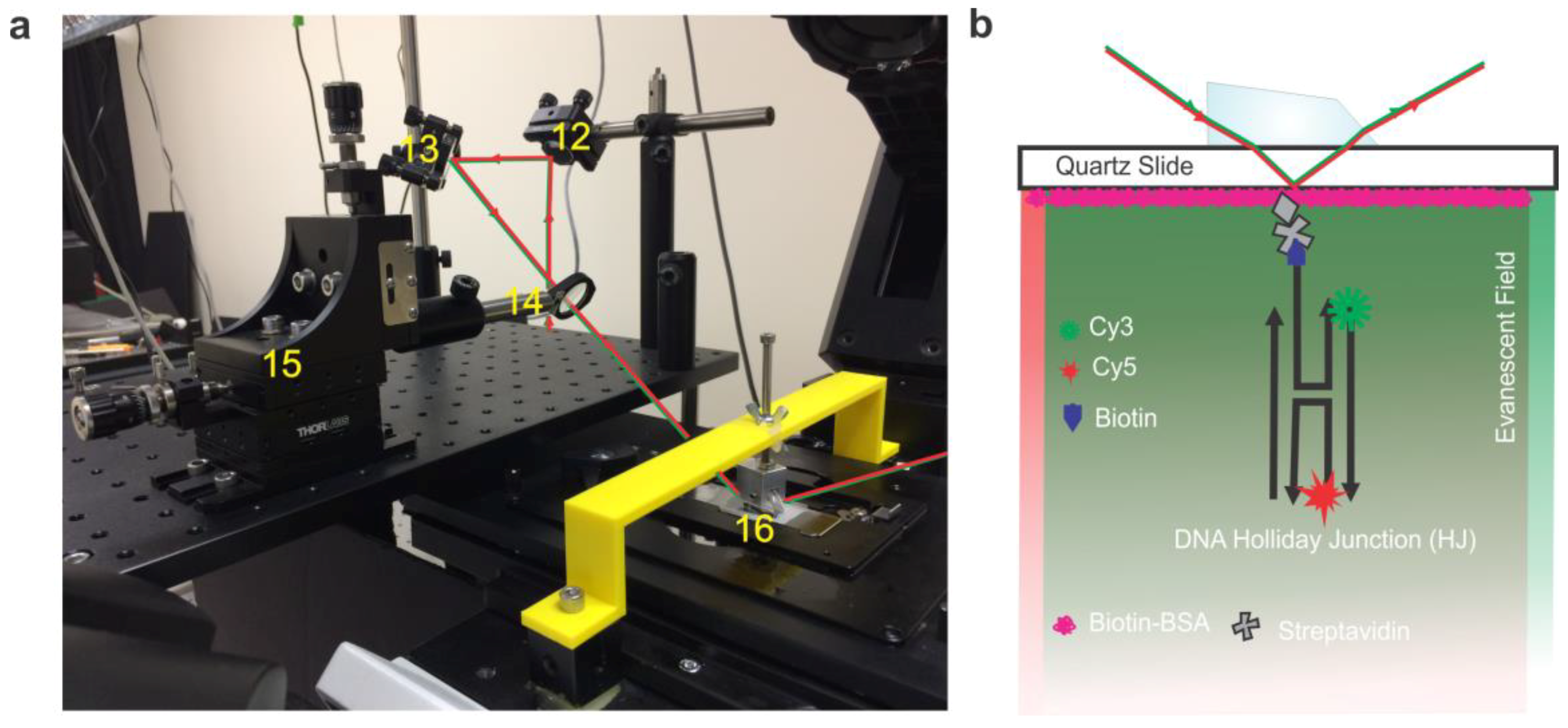
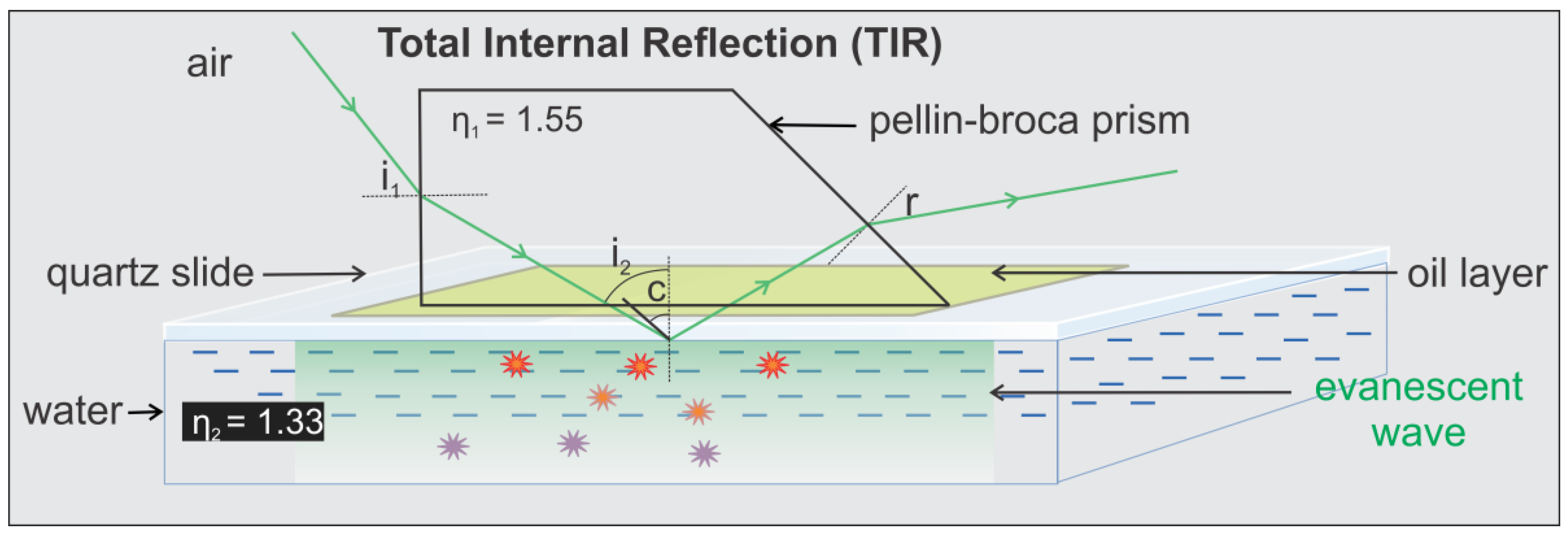
- Position the first mirror to direct the beam toward the elevated platform. The next two mirrors are placed in the line of the laser at roughly 45 degrees relative to the plane of the table and used to guide the beam toward the elevated stage and lift it to a level above the micrometer. The final mirror is used to direct the laser path through the focusing lens with 200 mm focal length (mounted on the micrometer) and into the prism, all adjusted so that the laser path enters into the prism at the appropriate angle to induce total internal reflection (in the case of our setup ~35°, calculated according to method detailed below in the Technical Note section).
CRITICAL STEP It will be necessary to adjust the mirrors such that the focus is not elongated.
- The microscope should be situated to the right of the elevated platform in such a way that the laser can be aimed through the optics and still maintain both the angle of total internal reflection and be within the focusing distance of the focusing lens.
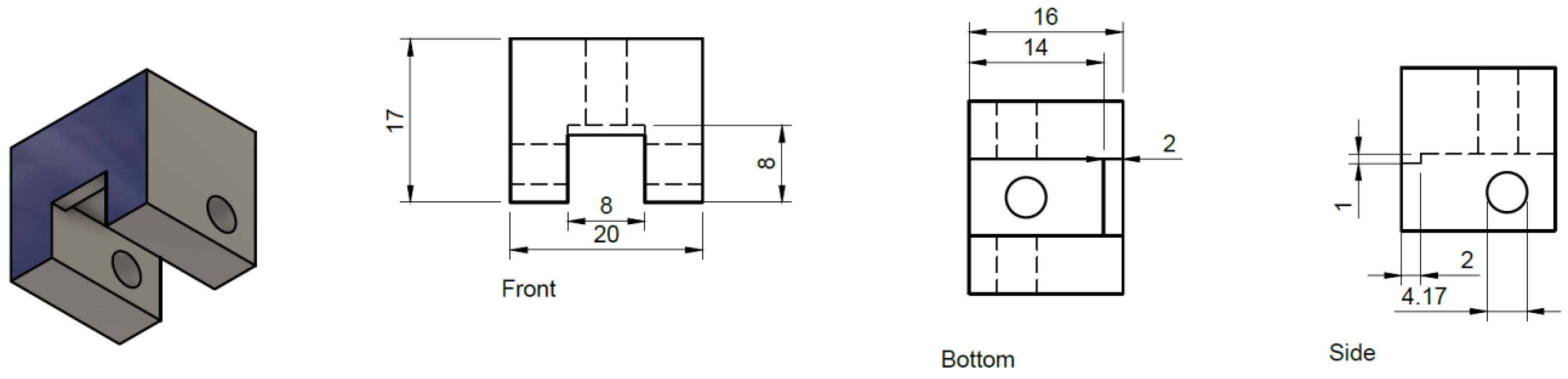
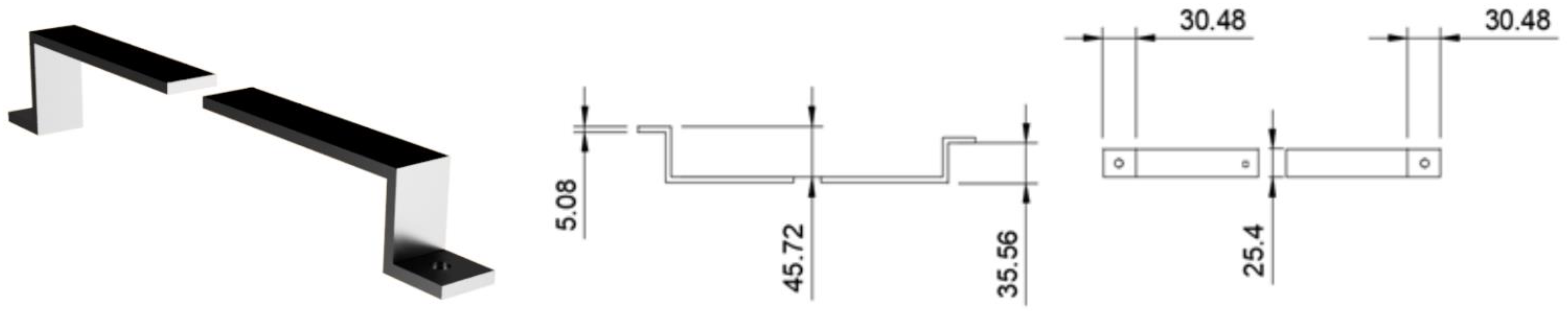
- The prism should be mounted in place over the flow cell by a clamping device affixed to the support arm which is screwed onto mounting hardware fixed to the upper body of the microscope.Note: Our prism clamp and support arm were custom manufactured by machining of aluminum and 3D printing respectively (see Technical Note section below for details). For a given microscope, the dimensions of the support arm and relative height of the prism will change, however the central principle of the design remains the same: to fix the position of the prism directly over the objective. So long as this is accomplished the design can be modified in many ways to accommodate various microscope brands and configurations.
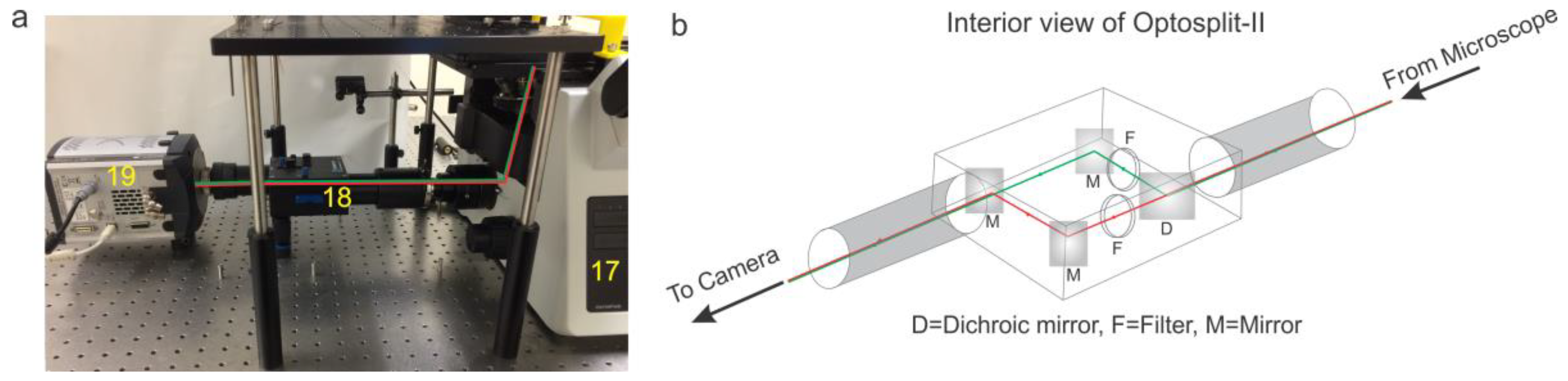
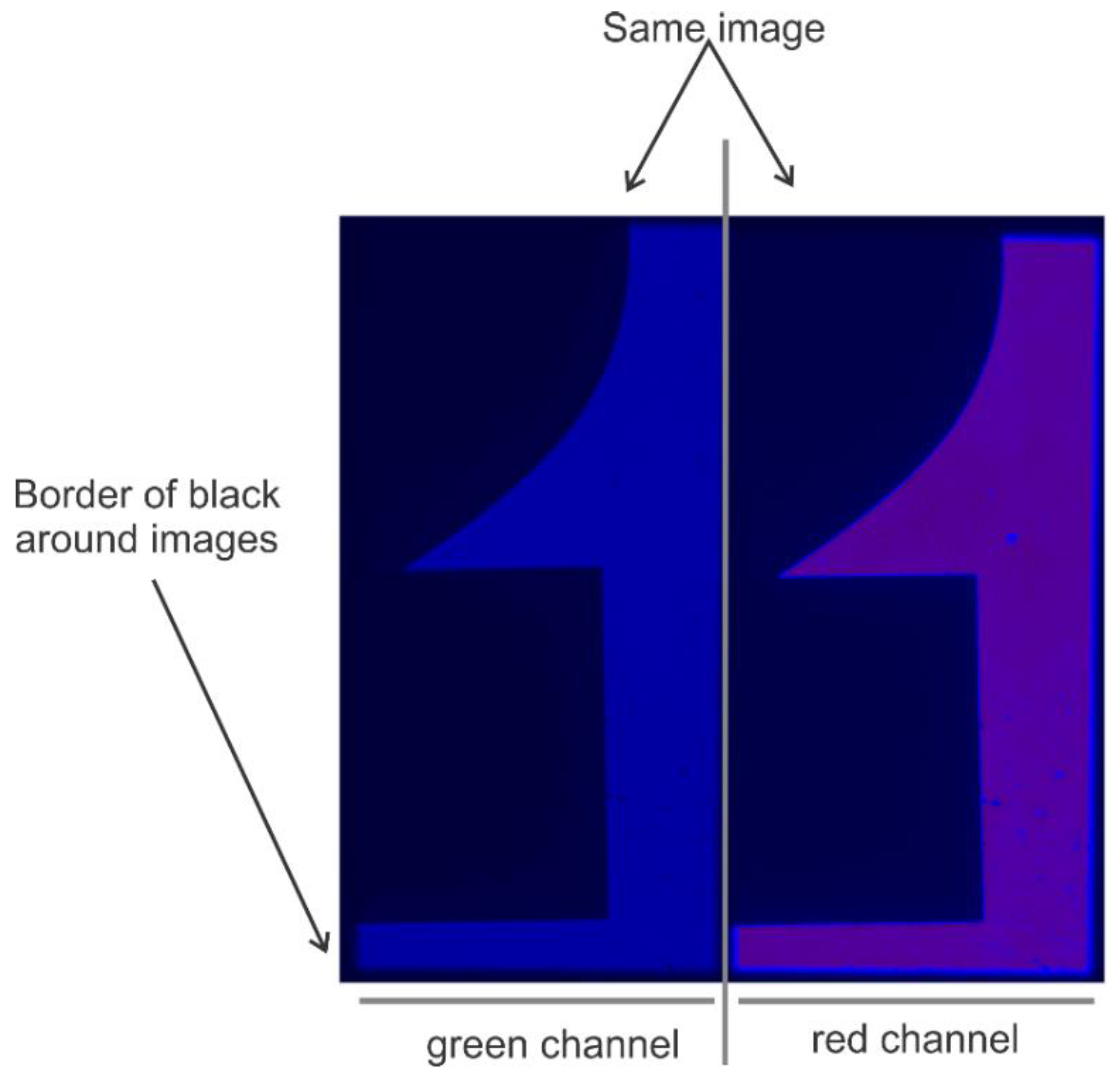
- Install the Optosplit-II (Carin Research, UK) into the imaging port and install the dichroic mirror and filters cube into the Optosplit-II. The aperture of the optosplit can then be tuned using the aperture adjustors to size the incoming light down to fit onto half of the EMCCD camera’s field of view and adjusted to split the channels to show parallel images (see Figure 5 and Figure 6). This can be accomplished by using a slide with some mm sized details (such as etched writing) to align the two channels in a parallel fashion for analysis by some smFRET program.Note: The Optosplit-II manual contains a more in-depth description of this process.
- Install Single.exe, a program that is made available by the TJ Ha group designed to record single molecule fluorescence data (see Data Acquisition and Analysis section below. Instructions on how to configure the EMCCD camera can be found in the Single.exe reference manual but can be summarized as finding the appropriate “atmcd32d.dll” file for the selected camera model and overwriting the existing file in the Single.exe program directory. This dll can be found in the driver install software package that accompanies Andor cameras (https://cplc.illinois.edu/software/).Note: In all cases we used a ×64 Windows PC to run this software. OSX or Linux versions may not be compatible/available for all mentioned software.
- OPTIONAL STEP Set up computer control of one or both lasers. In our case, we used a 639 nm Coherent CUBE Diode Laser (part#1069417) which comes equipped for computer control with an accompanying computer program Coherent Connection (http://cohrdownloads.blob.core.windows.net/file/CUBE%20Connection.zip). This laser’s serial cable connection was routed to our modern computer using a serial to USB connector.Note: When connecting an item to the processing computer with a serial cable, some configuration may be necessary to ensure that the coherent connection is monitoring the same serial port that the USB adaptor is feeding to.
4.2. Laser Alignment and Focusing
- The first step in fine tuning the laser alignment is to ensure that the laser lands immediately over the microscope objective. This can be aided by using a small piece of Scotch magic tape (preferably white in color) affixed on a glass slide to more easily visualize the location of the focus.
CRITICAL STEP It is essential to get the beam as close to the center of the objective lenses as possible as this will save time in adjusting the micrometer later on.
- A flow cell filled with water should be placed onto the microscope, a drop of immersion oil should be placed on the top surface of the flow cell and the prism support arm assembly screwed into place.Note: It is important that the flow cell be assembled using a quartz slide as using glass will result in an unusable background signal.
- The micrometers should be used to adjust the position of the focus on the X and Y axis until it is centered in the field of view through the eyepiece in the lowest objective. Once done, the Single.exe program can be used to track the intensity of the light coming off the focus.
- The Z-axis is then adjusted either up or down to increase the intensity of the light, during which some X and Y adjustment needs to take place to keep the focus at the center of the field of view. This Z axis adjustment should be carried out until the intensity of the signal reaching the camera is at its maximum.Note: The center of the microscope field of view may be offset from the cameras field of view, so adjustments should be made accordingly.
5. Flow Cell Design and Construction for pTIRF Experiments
5.1. Cleaning Procedure
- Briefly, wash the slides in warm soapy water and then scrub thoroughly with a thick paste of Alconox, follow by rinsing the slides in deionized water, acetone, and ethanol successively.
- Then flame the slides for 30 s on each side using a propane torch and immediately transfer them to a boiling base-piranha bath (Solution of 4% hydrogen peroxide and ammonia) for about 15 min and flame again on each side for 30 s with propane torch.
5.2. Design the Flow Cell
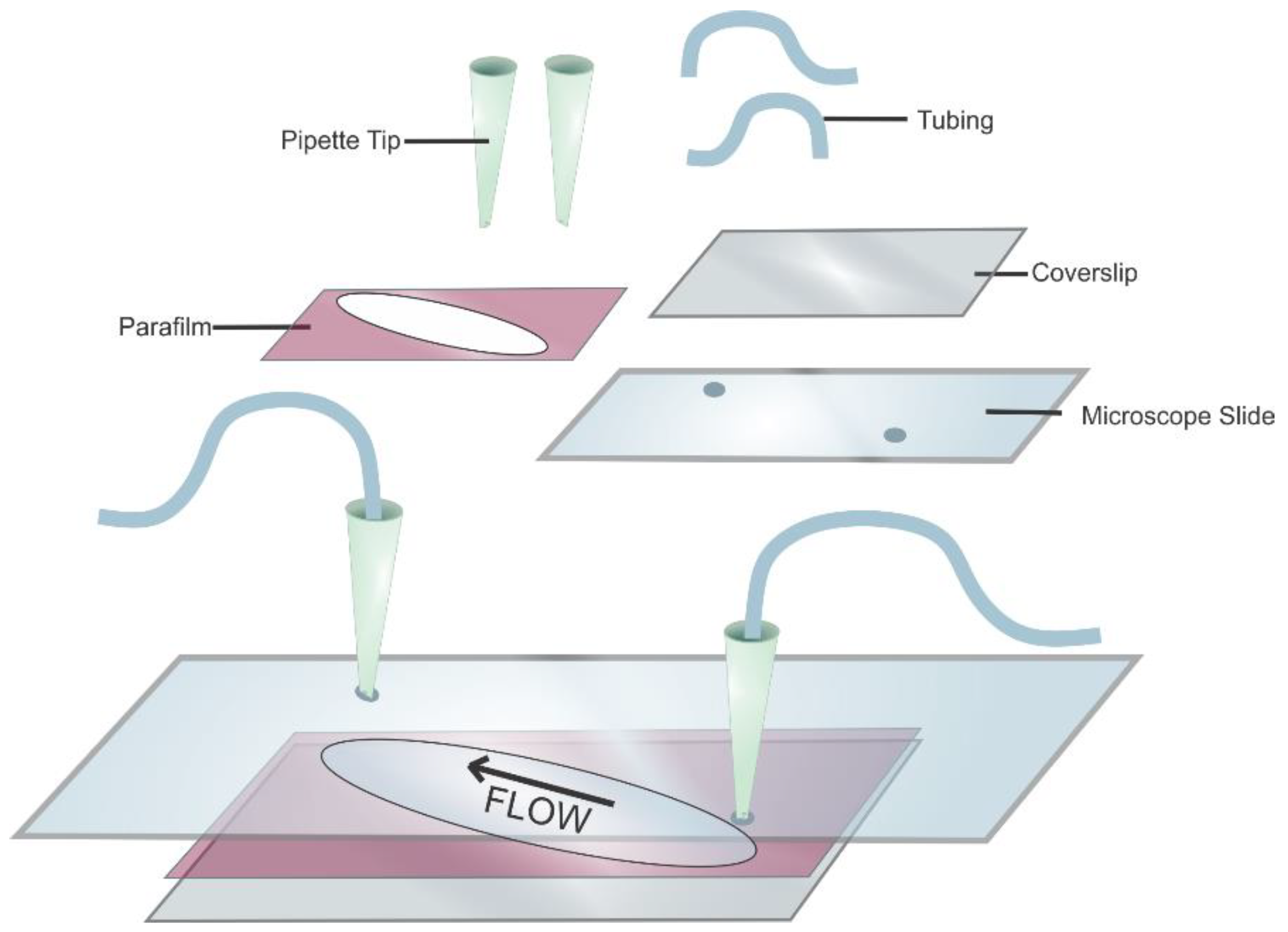
- Take a pre-cleaned standard quartz slide (75 × 26 × 1 mm) with two diagonally drilled holes (drilled using a diamond-coated drill bit (1 mm in diameter) in a Dremel multitool purchased from Walmart, Bentonville, AR, USA) and add parafilm overtop (Figure 7).
- Create a sample chamber by cutting the parafilm diagonally to encompass the drilled holes.
- Cover the sample chamber with a glass coverslip (24 × 60 mm, Fisher Scientific, Waltham, MA, USA) and heat the whole assembly to 120 °C for 5 min on a hot plate in order to melt the parafilm, thus sealing the glass coverslip to the microscope slide.
- Cut two 200 µL plastic pipette tips to about an inch long, insert into the holes and plume with tubing (0.02 in. ID, 0.06 in. OD, Cole-Palmer, Vernon Hills, IL, USA) using Double Bubble Quick-Set epoxy from Hardman Adhesives.Note: The physical obstructions of the pluming in this flow cell design limit the usable space for the experiment to ~1/5 of the slides total surface area.
- Functionalize the flow cell by sequential incubation of 1 mg/mL biotinylated BSA and 0.2 mg/mL streptavidin for 5 min and 2 min, respectively [14].
- Then flush the flow cell with ~300 µL of 1 × TAE-Mg buffer (40 mM tris, 2 mM EDTA, 20 mM acetic acid, 12 mM MgCl2).
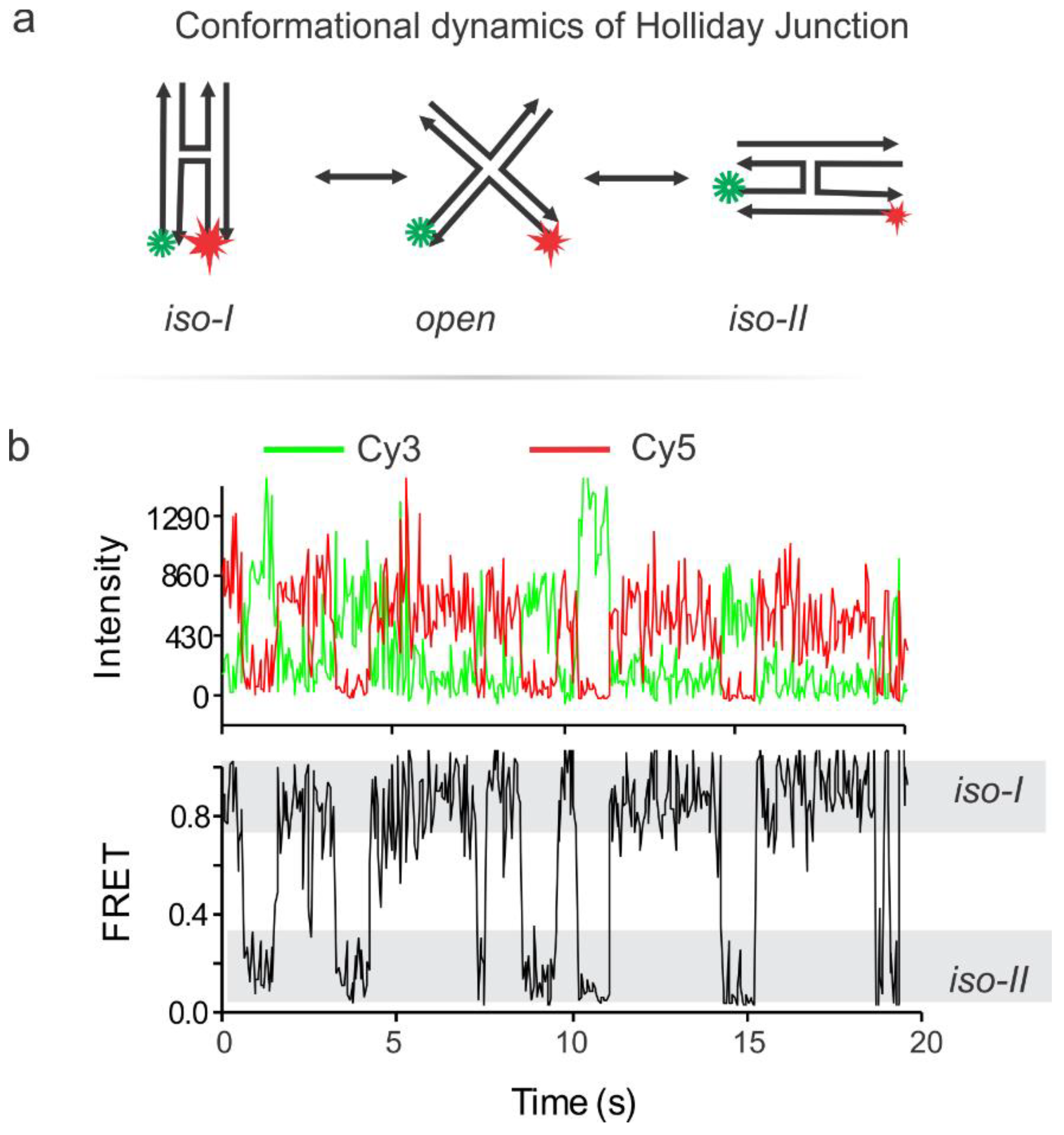
6. Validation of the pTIRF Setup via Typical smFRET Experiment
7. Data Acquisition and Analysis
8. Technical Notes
8.1. Prism Angle Calculation
8.2. Prism Clamp
8.3. Prism Support Arm
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kozuka, J.; Yokota, H.; Arai, Y.; Ishii, Y.; Yanagida, T. Dynamic polymorphism of single actin molecules in the actin filament. Nat. Chem. Biol. 2006, 2, 83–86. [Google Scholar] [CrossRef] [PubMed]
- Ha, T.; Rasnik, I.; Cheng, W.; Babcock, H.P.; Gauss, G.H.; Lohman, T.M.; Chu, S. Initiation and re-initiation of DNA unwinding by the Escherichia coli Rep helicase. Nature 2002, 419, 638–641. [Google Scholar] [CrossRef] [PubMed]
- Poulter, N.S.; Pitkeathly, W.T.E.; Smith, P.J.; Rappoport, J.Z. The physical basis of total internal reflection fluorescence (TIRF) microscopy and its cellular applications. In Advanced Fluorescence Microscopy: Methods and Protocols; Verveer, P.J., Ed.; Methods in Molecular Biology; Springer: New York, NY, USA, 2015; pp. 1–23. ISBN 978-1-4939-2080-8. [Google Scholar]
- Millis, B.A. Evanescent-wave field imaging: An introduction to total internal reflection fluorescence microscopy. In Molecular Profiling: Methods and Protocols; Espina, V., Liotta, L.A., Eds.; Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2012; pp. 295–309. ISBN 978-1-60327-216-2. [Google Scholar]
- Axelrod, D.; Burghardt, T.P.; Thompson, N.L. Total internal reflection fluorescence. Annu. Rev. Biophys. Bioeng. 1984, 13, 247–268. [Google Scholar] [CrossRef] [PubMed]
- Yildiz, A.; Vale, R.D. Total Internal reflection fluorescence microscopy. Cold Spring Harbor Protoc. 2015, 2015, pdb.top086348. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Fu, R.; Xu, J.; Liu, Y. A simple and cost-effective setup for super-resolution localization microscopy. Sci. Rep. 2017, 7, 1542. [Google Scholar] [CrossRef] [PubMed]
- Fish, K.N. Total internal reflection fluorescence (TIRF) microscopy. Curr. Protoc. Cytom 2009. [Google Scholar] [CrossRef]
- Walter, N.G.; Huang, C.-Y.; Manzo, A.J.; Sobhy, M.A. Do-it-yourself guide: How to use the modern single molecule toolkit. Nat. Methods 2008, 5, 475–489. [Google Scholar] [CrossRef] [PubMed]
- Diao, J.; Ishitsuka, Y.; Lee, H.; Joo, C.; Su, Z.; Syed, S.; Shin, Y.-K.; Yoon, T.-Y.; Ha, T. A single vesicle-vesicle fusion assay for in vitro studies of SNAREs and accessory proteins. Nat. Protoc. 2012, 7, 921–934. [Google Scholar] [CrossRef] [PubMed]
- Martin-Fernandez, M.; Tynan, C.; Webb, S. A ‘pocket guide’ to total internal reflection fluorescence. J. Microsc. 2013, 252, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Heintzmann, R. A Appendix: Practical guide to optical alignment. In Fluorescence Microscopy; Wiley-Blackwell: Hoboken, NJ, USA, 2013; pp. 393–401. ISBN 978-3-527-67159-5. [Google Scholar]
- Joo, C.; Ha, T. Preparing sample chambers for single-molecule FRET. Cold Spring Harbor Protoc. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Ha, T.; Zhuang, X.; Kim, H.D.; Orr, J.W.; Williamson, J.R.; Chu, S. Ligand-induced conformational changes observed in single RNA molecules. Proc. Natl. Acad. Sci. USA 1999, 96, 9077–9082. [Google Scholar] [CrossRef] [PubMed]
- Deffeyes, K.S.; Deffeyes, S.E. Nanoscale: Visualizing an Invisible World; MIT Press: Cambridge, MA, USA, 2011; ISBN 978-0-262-32252-2. [Google Scholar]
- Hyeon, C.; Lee, J.; Yoon, J.; Hohng, S.; Thirumalai, D. Hidden complexity in the isomerization dynamics of Holliday junctions. Nat. Chem. 2012, 4, 907–914. [Google Scholar] [CrossRef] [PubMed]
- Joo, C.; McKinney, S.A.; Lilley, D.M.; Ha, T. Exploring rare conformational species and ionic effects in DNA Holliday junctions using single-molecule spectroscopy. J. Mol. Biol. 2004, 341, 739–751. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.R. Mechanism and control of homologous recombination in Escherichia coli. Annu. Rev. Genet. 1987, 21, 179–201. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Heyer, W.-D. Homologous recombination in DNA repair and DNA damage tolerance. Cell Res. 2008, 18, 99–113. [Google Scholar] [CrossRef] [PubMed]
- Kaniecki, K.; De Tullio, L.; Greene, E.C. A change of view: Homologous recombination at single-molecule resolution. Nat. Rev. Genet. 2017, 19, 191–207. [Google Scholar] [CrossRef] [PubMed]
- Chernikova, S.B.; Game, J.C.; Brown, J.M. Inhibiting homologous recombination for cancer therapy. Cancer Biol. Therapy 2012, 13, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Van Gent, D.C.; Kanaar, R.; Kellogg, D. Exploiting DNA repair defects for novel cancer therapies. MBoC 2016, 27, 2145–2148. [Google Scholar] [CrossRef] [PubMed]
- Moynahan, M.E.; Jasin, M. Mitotic homologous recombination maintains genomic stability and suppresses tumorigenesis. Nat. Rev. Mol. Cell Biol. 2010, 11, 196–207. [Google Scholar] [CrossRef] [PubMed]
- Helleday, T.; Petermann, E.; Lundin, C.; Hodgson, B.; Sharma, R.A. DNA repair pathways as targets for cancer therapy. Nat. Rev. Cancer 2008, 8, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Dunham, M.A.; Neumann, A.A.; Fasching, C.L.; Reddel, R.R. Telomere maintenance by recombination in human cells. Nat. Genet. 2000, 26, 447–450. [Google Scholar] [CrossRef] [PubMed]
- Dilley, R.L.; Greenberg, R.A. ALTernative telomere maintenance and cancer. Trends Cancer 2015, 1, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Bryan, T.M.; Englezou, A.; Gupta, J.; Bacchetti, S.; Reddel, R.R. Telomere elongation in immortal human cells without detectable telomerase activity. EMBO J. 1995, 14, 4240–4248. [Google Scholar] [CrossRef] [PubMed]
- Forster, T. Energiewanderung und Fluoreszenz. Naturwissenschaften 1946, 33, 166–175. [Google Scholar] [CrossRef]
- Gordon, G.W.; Berry, G.; Liang, X.H.; Levine, B.; Herman, B. Quantitative fluorescence resonance energy transfer measurements using fluorescence microscopy. Biophys. J. 1998, 74, 2702–2713. [Google Scholar] [CrossRef]
- Wouters, F.S.; Verveer, P.J.; Bastiaens, P.I. Imaging biochemistry inside cells. Trends Cell Biol. 2001, 11, 203–211. [Google Scholar] [CrossRef]
- Jalink, K.; van Rheenen, J. Chapter 7 FilterFRET: Quantitative imaging of sensitized emission. In Laboratory Techniques in Biochemistry and Molecular Biology; Fret and Flim Techniques; Elsevier: Amsterdam, The Netherlands, 2009; Volume 33, pp. 289–349. [Google Scholar]
- McKinney, S.A.; Déclais, A.-C.; Lilley, D.M.J.; Ha, T. Structural dynamics of individual Holliday junctions. Nat. Struct. Biol. 2003, 10, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Ha, T.; Schulten, K. Conformational model of the Holliday junction transition deduced from molecular dynamics simulations. Nucleic Acids Res. 2004, 32, 6683–6695. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, D.R.; Dhakal, S. Single-molecule imaging reveals conformational manipulation of Holliday junction DNA by the junction processing protein RuvA. Biochemistry 2018, 57, 3616–3624. [Google Scholar] [CrossRef] [PubMed]
- Aitken, C.E.; Marshall, R.A.; Puglisi, J.D. An oxygen scavenging system for improvement of dye stability in single-molecule fluorescence experiments. Biophys. J. 2008, 94, 1826–1835. [Google Scholar] [CrossRef] [PubMed]
- Widom, J.R.; Dhakal, S.; Heinicke, L.A.; Walter, N.G. Single-molecule tools for enzymology, structural biology, systems biology and nanotechnology: An update. Arch. Toxicol. 2014, 88, 1965–1985. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Lim, J.; Ha, T. Acidification of the oxygen scavenging system in single-molecule fluorescence studies: In Situ Sensing with a Ratiometric Dual-Emission Probe. Anal. Chem. 2010, 82, 6132–6138. [Google Scholar] [CrossRef] [PubMed]
- Swoboda, M.; Henig, J.; Cheng, H.-M.; Brugger, D.; Haltrich, D.; Plumeré, N.; Schlierf, M. Enzymatic oxygen scavenging for photostability without pH drop in single-molecule experiments. ACS Nano 2012, 6, 6364–6369. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Yang, Y.R.; Dhakal, S.; Zhao, Z.; Liu, M.; Zhang, T.; Walter, N.G.; Yan, H. Assembly of multienzyme complexes on DNA nanostructures. Nat. Protoc. 2016, 11, 2243–2273. [Google Scholar] [CrossRef] [PubMed]
- Suddala, K.C.; Cabello-Villegas, J.; Michnicka, M.; Marshall, C.; Nikonowicz, E.P.; Walter, N.G. Hierarchical mechanism of amino acid sensing by the T-box riboswitch. Nat. Commun. 2018, 9, 1896. [Google Scholar] [CrossRef] [PubMed]
- McKinney, S.A.; Joo, C.; Ha, T. Analysis of single-molecule FRET trajectories using hidden Markov modeling. Biophys. J. 2006, 91, 1941–1951. [Google Scholar] [CrossRef] [PubMed]
- Roy, R.; Hohng, S.; Ha, T. A practical guide to single-molecule FRET. Nat. Methods 2008, 5, 507–516. [Google Scholar] [CrossRef] [PubMed]
- Dhakal, S.; Adendorff, M.R.; Liu, M.; Yan, H.; Bathe, M.; Walter, N.G. Rational design of DNA-actuated enzyme nanoreactors guided by single molecule analysis. Nanoscale 2016, 8, 3125–3137. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Fu, J.; Dhakal, S.; Johnson-Buck, A.; Liu, M.; Zhang, T.; Woodbury, N.W.; Liu, Y.; Walter, N.G.; Yan, H. Nanocaged enzymes with enhanced catalytic activity and increased stability against protease digestion. Nat. Commun. 2016, 7, 10619. [Google Scholar] [CrossRef] [PubMed]


| Catalog/Item # | Item Description | Quantity | Vendor | Optics # |
|---|---|---|---|---|
| p/n 325–1206 | p/n 325–1206 UV FS Pellin-Broca Prism 11 × 20 × 6.4 mm | 2 | Altos Photonics, Inc. (Bozeman, MT, USA ) | 16 |
| Laser Barrier 150” wide X 92” long | 1 | Beamstop’r (Lyndhurst. OH, USA | ||
| 16212 | Immersion Oil Type FF (4 Fl. Oz.) | 2 | Cargille (Cedar Grove, NJ, USA) | |
| zet640/20x | magnetron bandpass clean-up filter (excitation path), 0–5 deg aoi | 1 | Chroma Technology Corp (Bellows Falls, VT, USA) | 5 |
| t565spxxr-uf3 | magnetron shortpass dichroic, 45 deg aoi | 1 | Chroma Technology Corp | 6 |
| 1069417 | SYS: CUBE 640-40 CIRCULAR: 640 nm: 40 mW | 1 | Coherent Inc. (Santa Clara, CA, USA) | 2 |
| 1073840 | ASSY: HEAT SINK: ACCESSORY: CUBE | 1 | Coherent Inc. | |
| 1214333 | Productivity Plus Bronze-CUBE | 3 | Coherent Inc. | |
| EW-06419-01 | Tygon Microbore Autoanalysis Tubing, 0.020” × 0.060”OD, 100 ft/roll | 4 | Cole-Parmer | |
| CL532-050-L | 532 nm Central Wavelength 50 mW CW Power | 1 | CyrstaLaser (Reno, NV, USA) | 1 |
| 4001 | Hardman DOUBLE/BUBBLE Extra-Fast Set Epoxy Red Package 3.5 g Packet | 3 | Ellsworth Adhesive Systems (Germantown, WI, USA) | |
| KBH-5503 | Laser Protective Eyewear for HeNe Alignment and KTP Alignment applications | 2 | Kentek Corporation (Pittsfield, NH, USA) | |
| 9470 | 15 ft USB 3.0 A Male to A Female Active Extension Cable | 2 | Monoprice (Rancho Cucamonga, CA, USA) | |
| 5010 | Cat6 24AWG UTP Ethernet Network Patch Cable, 20 ft Gray | 2 | Monoprice | |
| 2067 | USB to RS232 DB9 male(Serial)/DB25 male Converter Cable | 1 | Monoprice | |
| U-R380 | IX3-D6RES;6-POSITION IX NOSEPIECE CODED, DIC | 1 | Olympus America Inc. (Center Valley, PA, USA) | |
| U-V111C | U-TV1XC;C-MOUNT CAMERA ADAPTER, CENTERABLE | 1 | Olympus America Inc. | |
| 9-U734 | 45FR; 45MM FROSTED DIFFUSION FILTER, IX3 | 1 | Olympus America Inc. | |
| UYCP-11 | UYCP-11;US STYLE 3-PRONG POWER CORD | 1 | Olympus America Inc. | |
| 5-UR403 | IX3-RFA;STRAIGHT ILLUMINATOR | 1 | Olympus America Inc. | 17 |
| 5-UR416-1 | IX3-RFACS-1-2; CODED IX3FLUORESCENCE TURRET | 1 | Olympus America Inc. | 17 |
| OCT-TD7BX3 | TRF59907-OL3; Dual-band ET-532/640 nm laser TIRF set | 1 | Olympus America Inc. | |
| OAT-DU-897U-CS0-#BV | DU-897U-CS0-#BV; IXON ULTRA897 EMCCD, 56FPS, 512 × 512, 16 UM, USB | 1 | Olympus America Inc. | 19 |
| OAT-TR-EMFS-F06 | 532/640 EM SPLITTING 3 PART FILTER SET FOR DPC&OPTOSPLIT | 1 | Olympus America Inc. | |
| OAT-TR-DCIS-CA1-00 | DUAL CAM CASSETTE. REQ(TR-EMF S-F) | 1 | Olympus America Inc. | |
| O89-OptoIILS | OptoSplit II LS–1.0x; Optosplit II system w/cubes and diaphragm | 1 | Olympus America Inc. | 18 |
| OMT-010 | MT-010; LOGO CLOTH DUSTCOVER 11 × 25 × 26”, ANTI-STATIC BX, IX | 1 | Olympus America Inc. | |
| OVP-MSTUT2468 | M-ST-UT2-46-8; Tuned-Damped Table, 1200 × 1800 × 203 mm, M6 Holes | 1 | Olympus America Inc. | |
| OVP-S2000A428 | S-2000A-428; 28” Isolators w/Auto Leveling, Set of 4 | 1 | Olympus America Inc. | |
| OVP-ACWS | ACWS; Air Compressor, Low Noise, 110 V | 1 | Olympus America Inc. | |
| HPZ440WIN7-2 | 2805181;HP Z440, 2 × 1TBHD, RAID1, 32GB DDR3, WIN764, SERIAL, MS OFC | 1 | Olympus America Inc. | |
| D-M27FPW2 | 718668226; 27-INCH 16:9 RATIO FLAT PANEL, 4K UHD, HDMI, D PORT | 1 | Olympus America Inc. | |
| DIB-551.00 | Diamond Coated “Stick” Drills, DIB-551.00 | 10 | Shor International | |
| TR6-P5 | Ø1/2” Optical Post, SS, 8-32 Setscrew, 1/4”−20 Tap, L = 6”, 5 Pack | 3 | Thorlabs, Inc. (Newton, NJ USA) | |
| MB1218 | Aluminum Breadboard 12” × 18” × 1/2”, 1/4”−20 Taps | 1 | Thorlabs, Inc. | |
| SH8S050 | 8-32 Stainless Steel Cap Screw, 1/2” Long | 1 | Thorlabs, Inc. | |
| TR12 | Ø1/2” Optical Post, SS, 8-32 Setscrew, 1/4”−20 Tap, L = 12” | 6 | Thorlabs, Inc. | |
| SH25S038 | 1/4”-20 Stainless Steel Cap Screw, 3/8” Long | 1 | Thorlabs, Inc. | |
| RA90 | Right-Angle Clamp for Ø1/2” Posts, 3/16” Hex | 8 | Thorlabs, Inc. | |
| TR6 | Ø1/2” Optical Post, SS, 8-32 Setscrew, 1/4”−20 Tap, L = 6” | 1 | Thorlabs, Inc. | |
| TR075 | Ø1/2” Optical Post, SS, 8-32 Setscrew, 1/4”−20 Tap, L = 0.75” | 2 | Thorlabs, Inc. | |
| SS25S075 | 1/4”−20 Stainless Steel Setscrew, 3/4” Long, Pack of 25 | 1 | Thorlabs, Inc. | |
| SH25S075 | 1/4”−20 Stainless Steel Cap Screw, 3/4” Long, Pack of 25 | 1 | Thorlabs, Inc. | |
| B3648F | 36” × 48” × 2.4” Imperial Breadboard, 128 × 98 × 23 cm | 1 | Thorlabs, Inc. | |
| PSY313 | 900 × 1200 mm Full Under Shelf, 146 × 95 × 6 cm | 1 | Thorlabs, Inc. | |
| PTA512 | Air Compressor-110/115 V-60 Hz, US Power Plug, 45 × 38 × 46 cm | 1 | Thorlabs, Inc. | |
| RSP1 | Rotation Stage For 1” Optics 2.2”OD 1.062-20 ID | 1 | Thorlabs, Inc. | |
| TR3-P5 | 1/2” Dia. × 3” Length: Pack of 5 Post | 3 | Thorlabs, Inc. | |
| SS6MS25 | M6-1.0 × 25 mm Set Screw, 25 Pack | 2 | Thorlabs, Inc. | |
| SS6MS12 | M6 × 1.0 Stainless Steel Set Screw 12 mm Long Pack of (25) | 1 | Thorlabs, Inc. | |
| FMP1-P5 | Fixed Ø1” Optical Mount 5-Pack | 2 | Thorlabs, Inc. | |
| WP25M-VIS | Mounted Ø25.0 mm Wire Grid Polarizer, 420–700 nM | 1 | Thorlabs, Inc. | 4 |
| ID8 | Mounted Standard Iris, 8.0.mm max. Aper. | 1 | Thorlabs, Inc. | 7 |
| ID12 | Iris Diaphragm 1/2” | 1 | Thorlabs, Inc. | |
| ID15 | Mounted Standard Iris, 15.0.mm max. Aper. | 1 | Thorlabs, Inc. | 9 |
| WPMH05M-532 | Mounted Multi Order 1/2 Waveplate 532 nm | 1 | Thorlabs, Inc. | 3 |
| BB1-E02 | Ø25.4 mm Mirror, Broadband 400–750 nm | 4 | Thorlabs, Inc. | 10–13 |
| LB1904-A-ML | Mounted N-BK7 Bi-Convex Lens, Ø1”, f = 125 mm, -A | 1 | Thorlabs, Inc. | |
| LB1437-A-ML | Mounted N-BK7 Bi-Convex Lens, Ø1”, f = 150 mm, -A | 1 | Thorlabs, Inc. | |
| LB1945-A-ML | Mounted N-BK7 Bi-Convex Lens, Ø1”, f = 200 mm, -A | 1 | Thorlabs, Inc. | 14 |
| ESK01 | MOUNTING SUPPORTS ESSENTIALS KIT #1 | 1 | Thorlabs, Inc. | |
| LG1 | Laser Glasses, 190–400 nm, 808–1090 nm | 1 | Thorlabs, Inc. | |
| ADB-10 | Pellin Broca Prism 10 mm BK7 | 1 | Thorlabs, Inc. | 16 |
| MT3A/M | XYZ Metric Translator Stage | 1 | Thorlabs, Inc. | 15 |
| KS1 | Lockable Kinematic 1” Optic Mount | 4 | Thorlabs, Inc. | |
| SDA90120S | Standing Height Active Science Desk to suit 900 × 1200 mm | 1 | Thorlabs, Inc. | |
| LS6S2T0 | Uni-stabled housed Shutter | 1 | Vincent Associates | 8 |
| VCM-D1 | Shutter Driver | 1 | Vincent Associates |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gibbs, D.R.; Kaur, A.; Megalathan, A.; Sapkota, K.; Dhakal, S. Build Your Own Microscope: Step-By-Step Guide for Building a Prism-Based TIRF Microscope. Methods Protoc. 2018, 1, 40. https://doi.org/10.3390/mps1040040
Gibbs DR, Kaur A, Megalathan A, Sapkota K, Dhakal S. Build Your Own Microscope: Step-By-Step Guide for Building a Prism-Based TIRF Microscope. Methods and Protocols. 2018; 1(4):40. https://doi.org/10.3390/mps1040040
Chicago/Turabian StyleGibbs, Dalton R., Anisa Kaur, Anoja Megalathan, Kumar Sapkota, and Soma Dhakal. 2018. "Build Your Own Microscope: Step-By-Step Guide for Building a Prism-Based TIRF Microscope" Methods and Protocols 1, no. 4: 40. https://doi.org/10.3390/mps1040040
APA StyleGibbs, D. R., Kaur, A., Megalathan, A., Sapkota, K., & Dhakal, S. (2018). Build Your Own Microscope: Step-By-Step Guide for Building a Prism-Based TIRF Microscope. Methods and Protocols, 1(4), 40. https://doi.org/10.3390/mps1040040






