A Rapid Bacteriophage DNA Extraction Method
Abstract
:1. Introduction
2. Experimental Design
3. Procedure
3.1. Propagation of Streptococcal Phages by the Plate Method. Time for Completion: 2–3 days
- Day 1: Determine which dilution of the phage lysate that will produce confluent lysis. Mix 100 µL of this dilution with 4 mL of molten top layer containing 80 µL of an overnight broth culture of the propagation strain. This mix is then poured over the bottom layer. Once the agarose had solidified, plates are incubated upside down at 37 °C overnight.
- Day 2: Add 5 mL of SM buffer to the plate and incubate at 4 °C overnight with shaking (50 rpm) to elute phages from the top layer.
- Day 3. Recover the eluted phage by removing the SM buffer to 13 mL centrifuge tubes. Centrifuge at 5 min at 5000× g to remove residual bacteria and filter lysate with a 0.22 µm syringe filter.
 PAUSE STEP Lysates can be kept at 4 °C until further use.
PAUSE STEP Lysates can be kept at 4 °C until further use.3.2. DNA Extraction. Time for Completion: 4 Hours
3.2.1. Removal of Bacterial DNA and RNA, and Digestion of Phage Capsid [4]. Time for Completion: 3 Hours
- To remove any residual bacterial DNA and RNA present in the lysate, 450 µL of the filter-sterilized lysate is incubated with 50 µL DNase I 10x buffer, 1 µL DNase I (1 U/µL), and 1 µL RNase A (10 mg/mL) for 1.5 h at 37 °C without shaking. Thereafter, 20 µL of 0.5 M EDTA (final concentration 20 mM) is added to inactivate DNase I and RNase A.
- To digest the phage protein capsid, 1.25 µL Proteinase K (20 mg/mL) is then added and incubated for 1.5 h at 56 °C without shaking.
3.2.2. DNA Purification with the DNeasy Blood & Tissue Kit (Qiagen, [6]). Time for Completion: 1 Hour for 12 Samples)
- Add 200 µL of lysed phage lysate to 200 µL of AL buffer, vortex to mix thoroughly and incubate for 10 min at 70 °C. Optional: to achieve a higher DNA yield, a larger volume of the lysed phage lysate can be used and add the equivalent volume of AL buffer.
- Add 200 µL of 99.9% EtOH (or the equivalent volume if using a larger volume), vortex to mix thoroughly.
- Transfer the mixture to the DNeasy Mini spin column placed in a 2 mL collection tube (provided with the kit) and centrifuge for 1 min at 6000× g. If larger volumes are used, repeat this step several times. Do not load more than 750 µL at a time onto the column. Discard the flow-through and the collection tube.
- Place the DNeasy Mini spin column in a new 2 mL collection tube (provided), add 500 µL Buffer AW1, and centrifuge for 1 min at 6000× g. Discard the flow-through and the collection tube.
- Place the DNeasy Mini spin column in a new 2 mL collection tube (provided), add 500 µL Buffer AW2, and centrifuge for 3 min at 20,000× g to dry the DNeasy membrane. Discard the flow-through and the collection tube. Place the DNeasy Mini spin column in a new 2 mL collection tube (provided) and centrifuge for an additional 1 min at 20,000× g to make sure there is no carryover of ethanol.
- Place the DNeasy Mini spin column in a sterile 1.5 mL or 2 mL microcentrifuge tube (not provided in the kit), and pipette 30 µL AE Buffer directly onto the DNeasy membrane.
- Incubate at room temperature for 1 min, and then centrifuge for 1 min at 6000× g to elute the DNA.OPTIONAL STEP Repeat the elution step using the eluate from the first elution.
4. Expected Results
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sulakvelidze, A. Safety by nature: Potential bacteriophage applications. Microbe Mag. 2011, 6, 122–126. [Google Scholar] [CrossRef]
- Vandenheuvel, D.; Lavigne, R.; Ussow, H.B. Bacteriophage therapy: Advances in formulation strategies and human clinical trials. Annu. Rev. Virol. 2015, 2, 599–618. [Google Scholar] [CrossRef] [PubMed]
- Pickard, D.J.J. Preparation of Bacteriophage Lysates and Pure DNA. In Bacteriophages; Clokie, M.R.J., Kropinski, A.M., Eds.; Methods in Molecular Biology; Humana Press: New York, NY, USA, 2009; Volume 2, pp. 3–10. ISBN 978-1-58829-682-5. [Google Scholar]
- Sambrook, J.; Russell, D.W. Molecular Cloning: A Laboratory Manual; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2001; ISBN 9780879695774. [Google Scholar]
- Kropinski, A.M.; Mazzocco, A.; Waddell, T.E.; Lingohr, E.; Johnson, R.P. Enumeration of Bacteriophages by Double Agar Overlay Plaque Assay. In Bacteriophages; Clokie, M.R.J., Kropinski, A.M., Eds.; Humana Press: New York, NY, USA, 2009; Volume 1, pp. 69–76. ISBN 978-1-58829-682-5. [Google Scholar]
- DNeasy® Blood & Tissue Handbook. July 2006. Available online: http://www.webcitation.org/query?url=http%3A%2F%2Fdiagnostics1.com%2FMANUAL%2FGeneral_Qiagen.pdf&date=2018-06-01 (accessed on 9 September 2017).
- Kot, W.; Vogensen, F.K.; Sørensen, S.J.; Hansen, L.H. DPS—A rapid method for genome sequencing of DNA-containing bacteriophages directly from a single plaque. J. Virol. Methods 2014, 196, 152–156. [Google Scholar] [CrossRef] [PubMed]
- MiSeq Support—Documentation Literature. Available online: http://emea.support.illumina.com/sequencing/sequencing_instruments/miseq/documentation.html (accessed on 26 July 2018).
- PacBio. Available online: www.pacb.com (accessed on 24 July 2018).
- Oxford Nanopore. Available online: https://nanoporetech.com (accessed on 24 July 2018).
| Phage ID | Starting PFU/mL | Nanodrop (ng/µL) | Qubit (ng/µL) | 260/280 a |
|---|---|---|---|---|
| SU1 | 2.1 × 1011 | 225.67 | 216 | 1.86 |
| SU2 | 9.0 × 1010 | 81.15 | 80.8 | 1.82 |
| SU3 | 9.0 × 1010 | 66.87 | 56.4 | 1.88 |
| SU4 | 6.0 × 1010 | 26.85 | 17.5 | 1.65 |
| SU5 | 1.6 × 1011 | 268.01 | 224 | 1.83 |
| SU6 | 3.0 × 1011 | 69.41 | 62.4 | 1.82 |
| SU7 | 6.0 × 1010 | 95.73 | 89.6 | 1.86 |
| SU8 | 1.0 × 1011 | 105.76 | 78.4 | 1.80 |
| SU9 | 1.1 × 1011 | 59.35 | 55.2 | 1.84 |
| SU10 | 2.0 × 1011 | 287.35 | 216 | 1.84 |
| SU11 | 1.0 × 1010 | 12.67 | 8.56 | 1.81 |
| SU12 | 1.4 × 1010 | 4.56 | 1.22 | 1.57 |
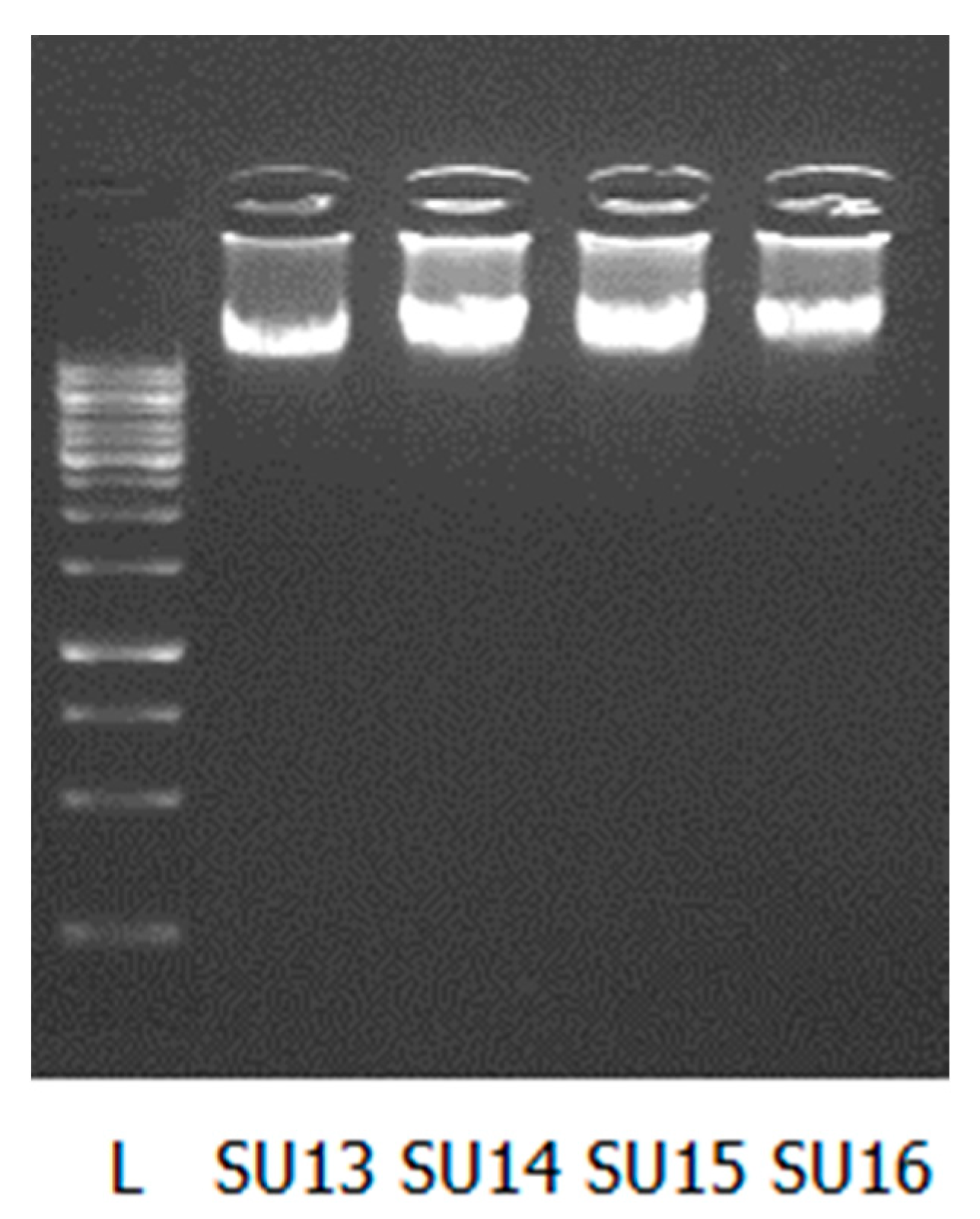
| SU13 | 2.0 × 1011 | 85.73 | 67.6 | 1.82 |
| SU14 | 1.9 × 1011 | 93.16 | 80.8 | 1.84 |
| SU15 | 1.3 × 1011 | 115.78 | 91.6 | 1.83 |
| SU16 | 1.0 × 1012 | 73.95 | 59.6 | 1.88 |
| *S112 | 1.0 × 1010 | 57.60 | 52 | 1.60 |
| *EC11 | 2.8 × 1010 | 30.91 | 35 | 1.80 |
| *EC80 | 2.0 × 1011 | 33.55 | 24.8 | 1.84 |
| Phage ID | Number of Contigs >1000 bp | Max Contig Length | Min Contig Length | N50 | Reads Assembled to Max Contig (%) | Number of Paired Reads | Reads with Phred Score Above 30 (%) |
|---|---|---|---|---|---|---|---|
| SU1 | 1 | 34,770 | 209 | 34,770 | 99.85 | 575,584 | 96.93 |
| SU2 | 3 | 34,819 | 200 | 34,819 | 99.96 | 547,242 | 97.58 |
| SU3 | 1 | 34,808 | 201 | 34,808 | 99.86 | 560,430 | 96.76 |
| SU4 | 1 | 34,819 | 204 | 34,819 | 99.94 | 422,742 | 95.65 |
| SU5 | 1 | 34,800 | 98 | 34,800 | 99.86 | 238,880 | 96.95 |
| SU6 | 1 | 34,799 | 205 | 34,799 | 99.95 | 483,526 | 96.61 |
| SU7 | 1 | 34,784 | 218 | 34,784 | 99.98 | 391,000 | 93.43 |
| SU8 | 1 | 34,802 | 206 | 34,802 | 99.92 | 354,100 | 96.86 |
| SU9 | 1 | 34,771 | 204 | 34,771 | 99.85 | 245,804 | 96.83 |
| SU10 | 1 | 34,873 | 204 | 34,873 | 99.99 | 266,872 | 95.73 |
| SU11 | 2 | 37,919 | 214 | 37,919 | 99.94 | 247,626 | 95.79 |
| SU12 | 1 | 34,507 | 221 | 34,507 | 99.99 | 337,214 | 95.70 |
| SU13 | 1 | 34,804 | 201 | 34,804 | 99.93 | 304,082 | 92.69 |
| SU14 | 1 | 35,029 | 200 | 35,029 | 99.91 | 294,802 | 96.27 |
| SU15 | 1 | 34,974 | 205 | 34,974 | 99.98 | 371,720 | 94.62 |
| SU16 | 1 | 34,819 | 201 | 34,819 | 99.99 | 552,896 | 94.31 |
| S112 | 1 | 52,664 | 210 | 52,664 | 99.97 | 100,884 | 93.28 |
| EC11 | 1 | 169,478 | 213 | 169,478 | 99.98 | 206,448 | 97.31 |
| EC80 | 1 | 52,706 | 202 | 52,706 | 99.96 | 5902 | 95.20 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jakočiūnė, D.; Moodley, A. A Rapid Bacteriophage DNA Extraction Method. Methods Protoc. 2018, 1, 27. https://doi.org/10.3390/mps1030027
Jakočiūnė D, Moodley A. A Rapid Bacteriophage DNA Extraction Method. Methods and Protocols. 2018; 1(3):27. https://doi.org/10.3390/mps1030027
Chicago/Turabian StyleJakočiūnė, Džiuginta, and Arshnee Moodley. 2018. "A Rapid Bacteriophage DNA Extraction Method" Methods and Protocols 1, no. 3: 27. https://doi.org/10.3390/mps1030027
APA StyleJakočiūnė, D., & Moodley, A. (2018). A Rapid Bacteriophage DNA Extraction Method. Methods and Protocols, 1(3), 27. https://doi.org/10.3390/mps1030027





