Newborn Screening in a Pandemic—Lessons Learned
Abstract
1. Background
2. Methods
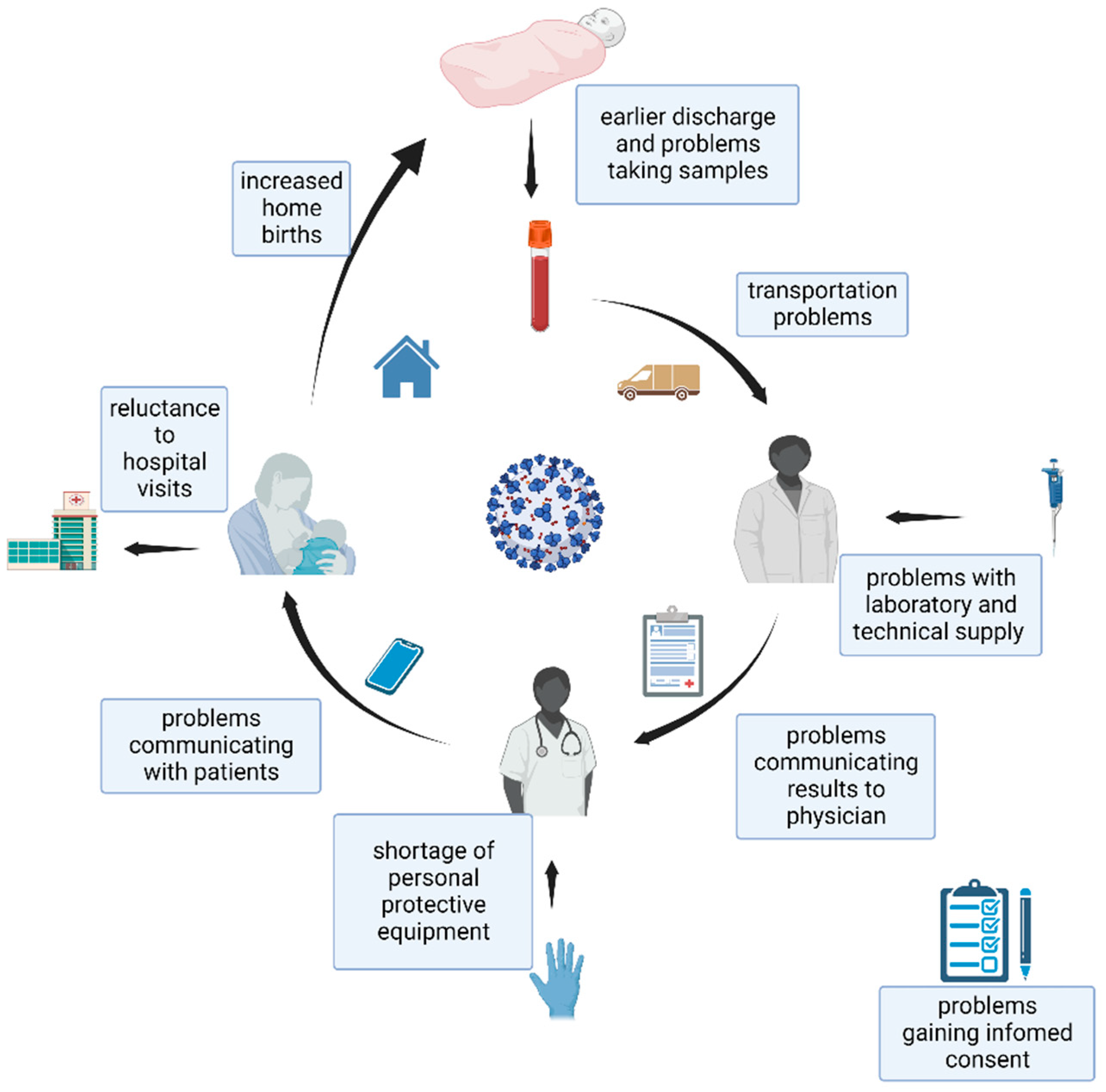
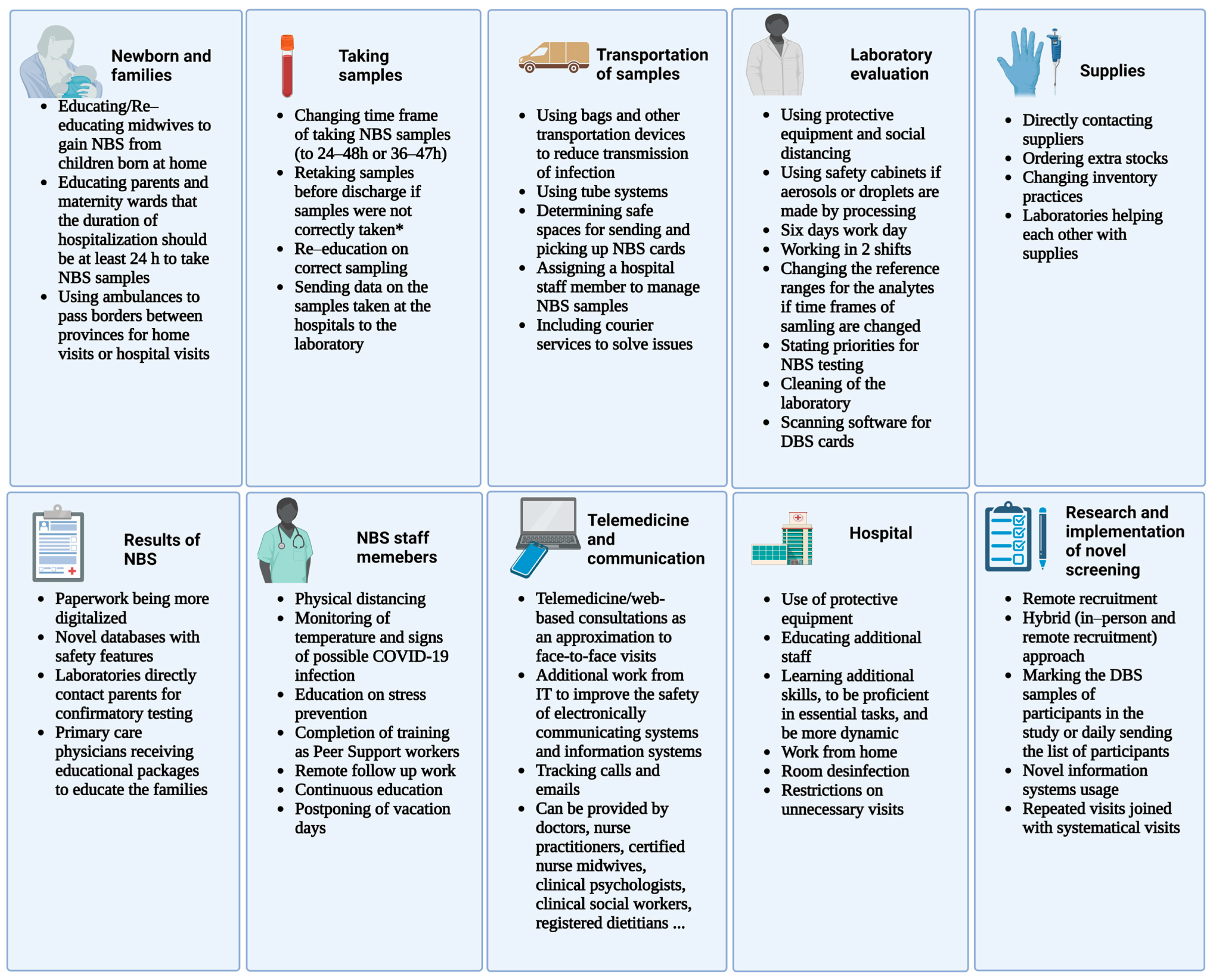
3. Challenges Faced by Staff Working on Newborn Screening Programs
4. Challenges Faced in the Management of Newborns and Families Participating in Newborn Screening
5. Challenges Faced in Collecting Samples for Newborn Screening
6. Challenges Faced in Transportation
7. Challenges Faced in Provision of Supplies for Newborn Screening
8. Challenges Faced with Laboratory Evaluation of Newborn Screening Samples
9. Challenges Faced in Communication of the Team Members and Participants of Newborn Screening
10. Challenges Faced in the Management of Abnormal Newborn Screening Results
11. Challenges Faced at the Hospitals and Maternity Wards to Perform Newborn Screening
12. Challenges Faced in Research and Implementation of Novel Screening Methods for Newborn Screening
13. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lanpher, B.; Brunetti-Pierri, N.; Lee, B. Inborn errors of metabolism: The flux from Mendelian to complex diseases. Nat. Rev. Genet. 2006, 7, 449–459. [Google Scholar] [CrossRef] [PubMed]
- Wilcken, B.; Wiley, V.; Hammond, J.; Carpenter, K. Screening Newborns for Inborn Errors of Metabolism by Tandem Mass Spectrometry. N. Engl. J. Med. 2003, 348, 2304–2312. [Google Scholar] [CrossRef] [PubMed]
- Therrell, B.L.; Padilla, C.D.; Loeber, J.G.; Kneisser, I.; Saadallah, A.; Borrajo, G.J.C.; Adams, J. Current status of newborn screening worldwide: 2015. Semin. Perinatol. 2015, 39, 171–187. [Google Scholar] [CrossRef]
- Loeber, J.G.; Platis, D.; Zetterström, R.H.; Almashanu, S.; Boemer, F.; Bonham, J.R.; Borde, P.; Brincat, I.; Cheillan, D.; Dekkers, E.; et al. Neonatal Screening in Europe Revisited: An ISNS Perspective on the Current State and Developments Since 2010. Int. J. Neonatal Screen. 2021, 7, 15. [Google Scholar] [CrossRef] [PubMed]
- Sikonja, J.; Groselj, U.; Scarpa, M.; la Marca, G.; Cheillan, D.; Kölker, S.; Zetterström, R.H.; Kožich, V.; Le Cam, Y.; Gumus, G.; et al. Towards Achieving Equity and Innovation in Newborn Screening across Europe. Int. J. Neonatal Screen. 2022, 8, 31. [Google Scholar] [CrossRef]
- Greaves, R.F.; Pitt, J.; McGregor, C.; Wall, M.; Christodoulou, J. Newborn bloodspot screening in the time of COVID-19. Genet. Med. 2021, 23, 1143–1150. [Google Scholar] [CrossRef]
- COVID-19 NBLS Response: Continuity of Operations Plans (COOP)|NewSTEPs. Available online: https://www.newsteps.org/resources/covid-19-nbs-response-continuity-operations-plans-coop (accessed on 4 October 2022).
- Tumiene, B.; Peters, H.; Melegh, B.; Peterlin, B.; Utkus, A.; Fatkulina, N.; Pfliegler, G.; Graessner, H.; Hermanns, S.; Scarpa, M.; et al. Rare disease education in Europe and beyond: Time to act. Orphanet J. Rare Dis. 2022, 17, 441. [Google Scholar] [CrossRef]
- Wynn, J.; Tavakoli, N.P.; Armstrong, N.; Gomez, J.; Koval, C.; Lai, C.; Tang, S.; Prince, A.Q.; Quevedo, Y.; Rufino, K.; et al. Improving Recruitment for a Newborn Screening Pilot Study with Adaptations in Response to the COVID-19 Pandemic. Int. J. Neonatal Screen. 2022, 8, 23. [Google Scholar] [CrossRef]
- Koracin, V.; Loeber, J.G.; Mlinaric, M.; Battelino, T.; Bonham, J.R.; Groselj, U. COVID-NBS ISNS global network. Global impact of COVID-19 on newborn screening programmes. BMJ Glob. Health 2022, 7, e007780. [Google Scholar] [CrossRef]
- Sebastião, F.M.; Michelin-Tirelli, K.; Bender, F.; Lopes, F.F.; Moraes, I.; Kubaski, F.; Giugliani, R.; Burin, M. COVID-19 impact on the diagnosis of Inborn Errors of Metabolism: Data from a reference center in Brazil. Genet. Mol. Biol. 2021, 45, e20210253. [Google Scholar] [CrossRef]
- Paneghetti, L.; Bellettato, C.M.; Sechi, A.; Stepien, K.M.; Scarpa, M. One year of COVID-19: Infection rates and symptoms in patients with inherited metabolic diseases followed by MetabERN. Orphanet J. Rare Dis. 2022, 17, 109. [Google Scholar] [CrossRef] [PubMed]
- Mütze, U.; Gleich, F.; Barić, I.; Baumgartner, M.; Burlina, A.; Chapman, K.A.; Chien, Y.; Cortès-Saladelafont, E.; De Laet, C.; Dobbelaere, D.; et al. Impact of the SARS-CoV-2 pandemic on the health of individuals with intoxication-type metabolic diseases—Data from the E-IMD consortium. J. Inherit. Metab. Dis. 2022, 46, 220–231. [Google Scholar] [CrossRef] [PubMed]
- Sechi, A.; Macor, D.; Valent, S.; Da Riol, R.M.; Zanatta, M.; Spinelli, A.; Bianchi, K.; Bertossi, N.; Dardis, A.; Valent, F.; et al. Impact of COVID-19 related healthcare crisis on treatments for patients with lysosomal storage disorders, the first Italian experience. Mol. Genet. Metab. 2020, 130, 170–171. [Google Scholar] [CrossRef]
- Singh, S.; Caggana, M.; Johnson, C.; Lee, R.; Zarbalian, G.; Gaviglio, A.; Keehn, A.; Morrison, M.; Becker, S.J.; Ojodu, J. COVID-19 Pandemic-Related Impacts on Newborn Screening Public Health Surveillance. Int. J. Neonatal Screen. 2022, 8, 28. [Google Scholar] [CrossRef]
- Simon, D.; Broadbridge, E.; Baker, M.; Gaviglio, A.; Gruber, D.; Piper, K.N.; Tavakoli, N.P.; Sullivan, J.; Kennedy, A. Common Challenges and Identified Solutions for State Newborn Screening Programs during COVID-19 Pandemic. Int. J. Neonatal Screen. 2022, 8, 7. [Google Scholar] [CrossRef]
- Loh, T.P.; Greaves, R.F.; Mak, C.M.; Salomons, G.S.; Bonham, J.R.; Lang, T.; IFCC Committee on Emerging Technologies in Paediatric Laboratory Medicine. Impact of COVID-19 on Pediatric Laboratory Medicine: An IFCC C-ETPLM, SSIEM, ISNS Global Survey. EJIFCC 2022, 33, 194–208. [Google Scholar] [PubMed]
- Loh, T.P.; Horvath, A.R.; Wang, C.-B.; Koch, D.; Lippi, G.; Mancini, N.; Ferrari, M.; Hawkins, R.; Sethi, S.; Adeli, K. The International Federation of Clinical Chemistry and Laboratory Medicine Taskforce on COVID-19. Laboratory practices to mitigate biohazard risks during the COVID-19 outbreak: An IFCC global survey. Clin. Chem. Lab. Med. 2020, 58, 1433–1440. [Google Scholar] [CrossRef]
- Ramgoolam, T. The impact of COVID-19 on the GOSH newborn screening service. Arch. Dis. Child. 2020, 105, A35. [Google Scholar]
- Cheng, R.J.; Fisher, A.C.; Nicholson, S.C. Interest in Home Birth during the COVID-19 Pandemic: Analysis of Google Trends Data. J. Midwifery Women’s Health 2022, 67, 427–434. [Google Scholar] [CrossRef]
- Verhoeven, C.J.M.; Boer, J.; Kok, M.; Nieuwenhuijze, M.; de Jonge, A.; Peters, L.L. More home births during the COVID-19 pandemic in the Netherlands. Birth 2022, 49, 792–804. [Google Scholar] [CrossRef]
- Maceda, E.B.G.; Abadingo, M.E.; Panol, K.A.R.; Beltran, F.D.E.; Valdez-Acosta, I.R.C.; Taquiqui, G.D.; Gawigawen, S.B.; Macalino, M.V.L.; Aguirre-Aguinaldo, L.M.S.M.; Flores-Declaro, M.A.; et al. Newborn Screening Long-Term Follow-Up Clinics (Continuity Clinics) in the Philippines during the COVID-19 Pandemic: Continuing Quality Patient Care. Int. J. Neonatal Screen. 2022, 9, 2. [Google Scholar] [CrossRef] [PubMed]
- Aragona, E.; West, D.; Loyal, J. Well-Newborn Unit Director Experiences During the COVID-19 Pandemic: A BORN Study. Hosp. Pediatr. 2021, 11, e170–e181. [Google Scholar] [CrossRef] [PubMed]
- Elmonem, M.A.; Belanger-Quintana, A.; Bordugo, A.; Boruah, R.; Cortès-Saladelafont, E.; Endrakanti, M.; Giraldo, P.; Grünert, S.C.; Gupta, N.; Kabra, M.; et al. The impact of COVID-19 pandemic on the diagnosis and management of inborn errors of metabolism: A global perspective. Mol. Genet. Metab. 2020, 131, 285–288. [Google Scholar] [CrossRef]
- Lippi, G.; Adeli, K.; Ferrari, M.; Horvath, A.R.; Koch, D.; Sethi, S.; Wang, C.-B. Biosafety measures for preventing infection from COVID-19 in clinical laboratories: IFCC Taskforce Recommendations. Clin. Chem. Lab. Med. CCLM 2020, 58, 1053–1062. [Google Scholar] [CrossRef] [PubMed]
- Guidance for General Laboratory Safety Practices during the COVID-19 Pandemic. Available online: https://www.cdc.gov/coronavirus/2019-ncov/lab/lab-safety-practices.html (accessed on 4 October 2022).
- EPA. Cleaning and Disinfecting: Best Practices during the COVID-19 Pandemic. Available online: https://www.epa.gov/coronavirus/cleaning-and-disinfecting-best-practices-during-covid-19-pandemic (accessed on 4 October 2022).
- Colorado. Newborn Screening State Emergency Plan for Time Critical Testing Limited Staff. Available online: https://www.newsteps.org/sites/default/files/covid-19_newborn_screening_state_emergency_plan_for_time_critical_testing_limited_staff_colorado_4.2.20.pdf (accessed on 4 October 2022).
- Heather, N.; Morgan, L.; Knoll, D.; Shore, K.; de Hora, M.; Webster, D. Introduction of a Protocol for Structured Follow-Up and Texting of Inadequate and Borderline-Positive Newborn Metabolic Screening Results. Int. J. Neonatal Screen. 2022, 8, 30. [Google Scholar] [CrossRef]
- Chudleigh, J.; Shakespeare, L.; Holder, P.; Chinnery, H.; Hack, G.; Gill, T.; Gould, R.; Southern, K.W.; Olander, E.K.; Morris, S.; et al. Co-designing improved communication of newborn bloodspot screening results to Parents: Mixed Methods Study. J. Particip. Med. 2022, 14, e33485. [Google Scholar] [CrossRef]
- Gold, J.I.; Campbell, I.M.; Ficicioglu, C. Provider Perspectives on the Impact of the COVID-19 Pandemic on Newborn Screening. Int. J. Neonatal Screen. 2021, 7, 38. [Google Scholar] [CrossRef]
- CMS. Telehealth Services MLN Fact Sheet. Available online: https://www.cms.gov/Outreach-and-Education/Medicare-Learning-Network-MLN/MLNProducts/MLN-Publications-Items/CMS1243327 (accessed on 4 October 2022).
- Lampe, C.; MetabERN collaboration group; Dionisi-Vici, C.; Bellettato, C.M.; Paneghetti, L.; van Lingen, C.; Bond, S.; Brown, C.; Finglas, A.; Francisco, R.; et al. The impact of COVID-19 on rare metabolic patients and healthcare providers: Results from two MetabERN surveys. Orphanet J. Rare Dis. 2020, 15, 341. [Google Scholar] [CrossRef]
- Galle, A.; Semaan, A.; Huysmans, E.; Audet, C.; Asefa, A.; Delvaux, T.; Afolabi, B.B.; El Ayadi, A.M.; Benova, L. A double-edged sword—Telemedicine for maternal care during COVID-19: Findings from a global mixed-methods study of healthcare providers. BMJ Glob. Health 2021, 6, e004575. [Google Scholar] [CrossRef] [PubMed]
- Shur, N.; Atabaki, S.M.; Kisling, M.S.; Tabarani, A.; Williams, C.; Fraser, J.L.S.; Regier, D.S.; Summar, M.; Rare Disease Institute. Rapid deployment of a telemedicine care model for genetics and metabolism during COVID-19. Am. J. Med. Genet. Part A 2021, 185, 68–72. [Google Scholar] [CrossRef]
- Mak, D.; Akinseye, L.; Lahoti, A.; Diaz-Thomas, A. Abnormal Newborn Screen during a Pandemic. Neoreviews 2022, 23, e212–e215. [Google Scholar] [CrossRef] [PubMed]
- Ramanan, A.V.; Modi, N.; de Wildt, S.N.; c4c Learning from COVID-19 Group. Improving clinical paediatric research and learning from COVID-19: Recommendations by the Conect4Children expert advice group. Pediatr. Res. 2022, 91, 1069–1077. [Google Scholar] [CrossRef]
- Osterrieder, A.; Cuman, G.; Pan-Ngum, W.; Cheah, P.K.; Cheah, P.K.; Peerawaranun, P.; Silan, M.; Orazem, M.; Perkovic, K.; Groselj, U.; et al. Economic and social impacts of COVID-19 and public health measures: Results from an anonymous online survey in Thailand, Malaysia, the UK, Italy and Slovenia. BMJ Open 2021, 11, e046863. [Google Scholar] [CrossRef] [PubMed]
- Gemeinsamer Bundesausschuss. Richtlinie des Gemeinsamen Bundesausschusses über die Früher-kennung von Krankheiten bei Kindern (Kinder-Richtlinie). 2022. Available online: https://www.g-ba.de/downloads/62-492-3038/Kinder-RL_2022-12-15_iK-2022-12-15.pdf (accessed on 17 February 2023).
- Abhyankar, S.; Goodwin, R.M.; Sontag, M.; Yusuf, C.; Ojodu, J.; McDonald, C.J. An update on the use of health information technology in newborn screening. Semin. Perinatol. 2015, 39, 188–193. [Google Scholar] [CrossRef]
- Malvagia, S.; Forni, G.; Ombrone, D.; la Marca, G. Development of Strategies to Decrease False Positive Results in Newborn Screening. Int. J. Neonatal Screen. 2020, 6, 84. [Google Scholar] [CrossRef] [PubMed]
- Department of Health and Human Services. Newborn Screening Contingency Plan (CONPLAN). Available online: https://www.cdc.gov/ncbddd/documents/screening-contingency-plan-version-ii.pdf (accessed on 17 February 2023).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mlinaric, M.; Bonham, J.R.; Kožich, V.; Kölker, S.; Majek, O.; Battelino, T.; Torkar, A.D.; Koracin, V.; Perko, D.; Remec, Z.I.; et al. Newborn Screening in a Pandemic—Lessons Learned. Int. J. Neonatal Screen. 2023, 9, 21. https://doi.org/10.3390/ijns9020021
Mlinaric M, Bonham JR, Kožich V, Kölker S, Majek O, Battelino T, Torkar AD, Koracin V, Perko D, Remec ZI, et al. Newborn Screening in a Pandemic—Lessons Learned. International Journal of Neonatal Screening. 2023; 9(2):21. https://doi.org/10.3390/ijns9020021
Chicago/Turabian StyleMlinaric, Matej, James R. Bonham, Viktor Kožich, Stefan Kölker, Ondrej Majek, Tadej Battelino, Ana Drole Torkar, Vanesa Koracin, Dasa Perko, Ziga Iztok Remec, and et al. 2023. "Newborn Screening in a Pandemic—Lessons Learned" International Journal of Neonatal Screening 9, no. 2: 21. https://doi.org/10.3390/ijns9020021
APA StyleMlinaric, M., Bonham, J. R., Kožich, V., Kölker, S., Majek, O., Battelino, T., Torkar, A. D., Koracin, V., Perko, D., Remec, Z. I., Lampret, B. R., Scarpa, M., Schielen, P. C. J. I., Zetterström, R. H., & Groselj, U. (2023). Newborn Screening in a Pandemic—Lessons Learned. International Journal of Neonatal Screening, 9(2), 21. https://doi.org/10.3390/ijns9020021








