Evaluating Compressed SENSE (CS) MRI Metal Artifact Reduction Using Pig L-Spine Phantom and Transplant Patients: Focused on the CS-SEMAC (SPIR), mDixon(O-MAR) and STIR Techniques
Abstract
:1. Introduction
2. Subjects and Methods
2.1. Subjects
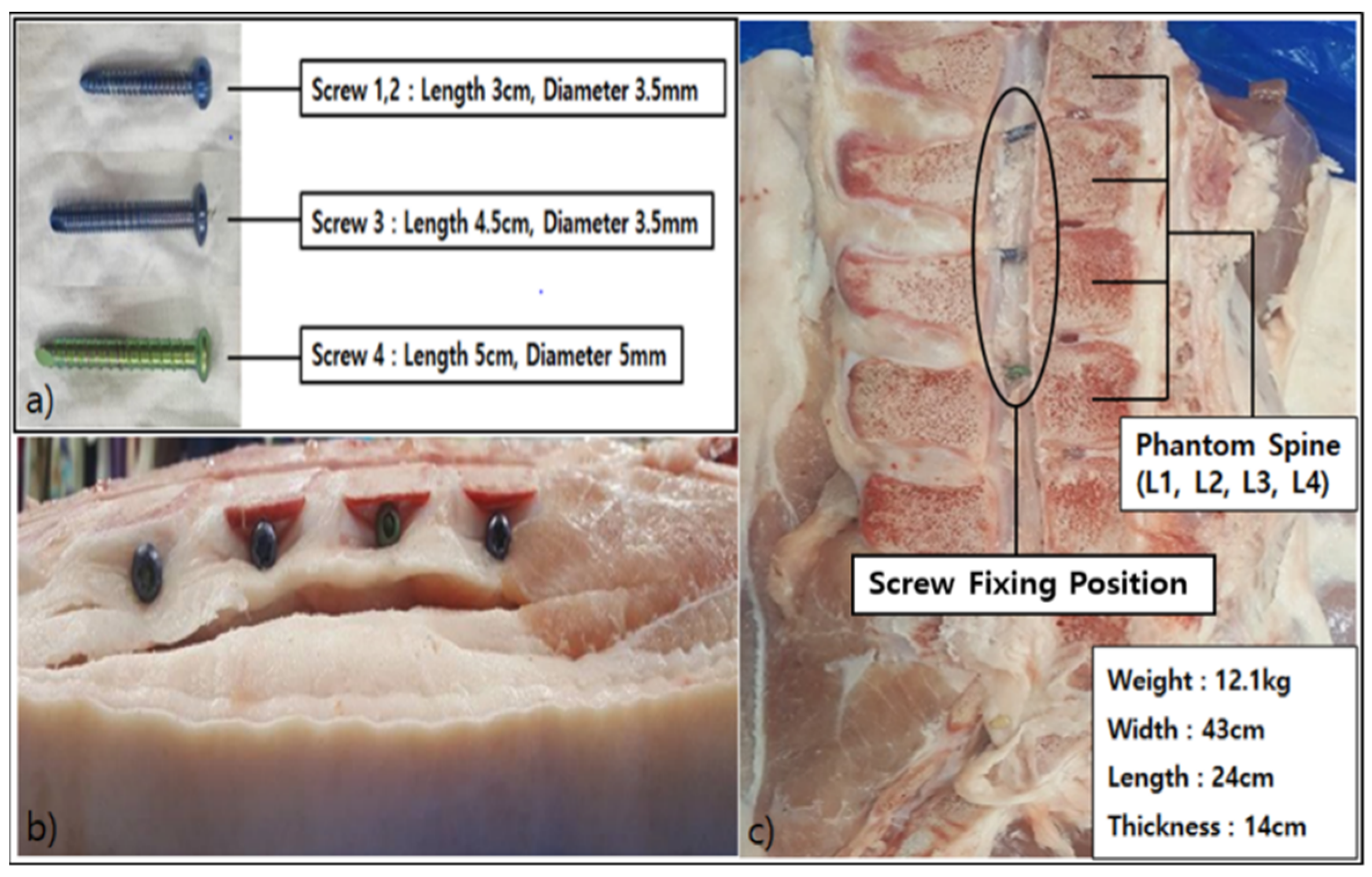
2.2. Process of Producing Phantom
2.3. Analysis Method
2.3.1. Quantitative Analysis Method
2.3.2. Qualitative Analysis Method
2.4. Statistical Analysis
3. Results
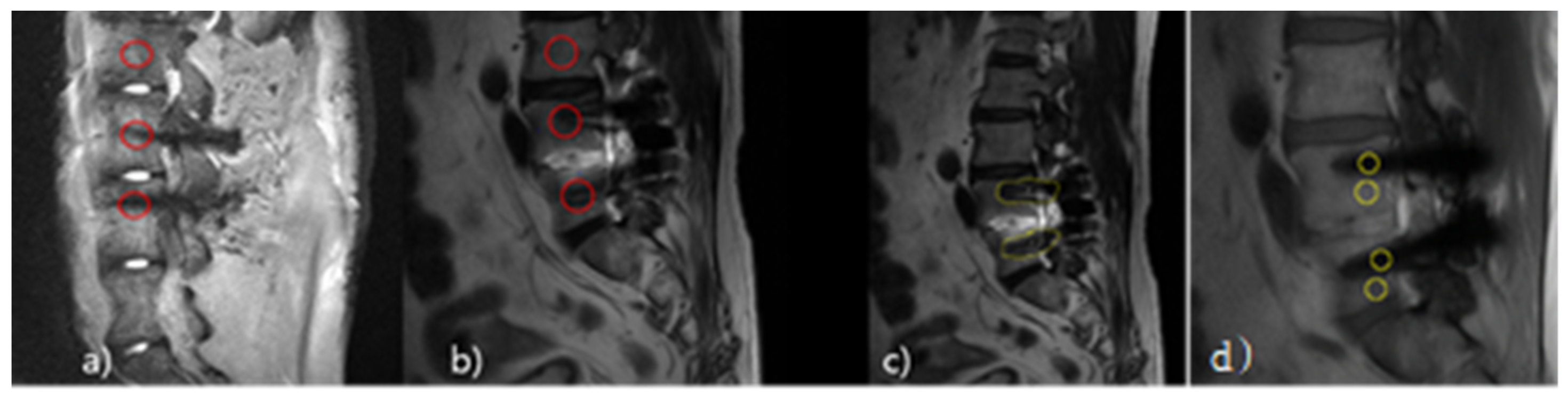
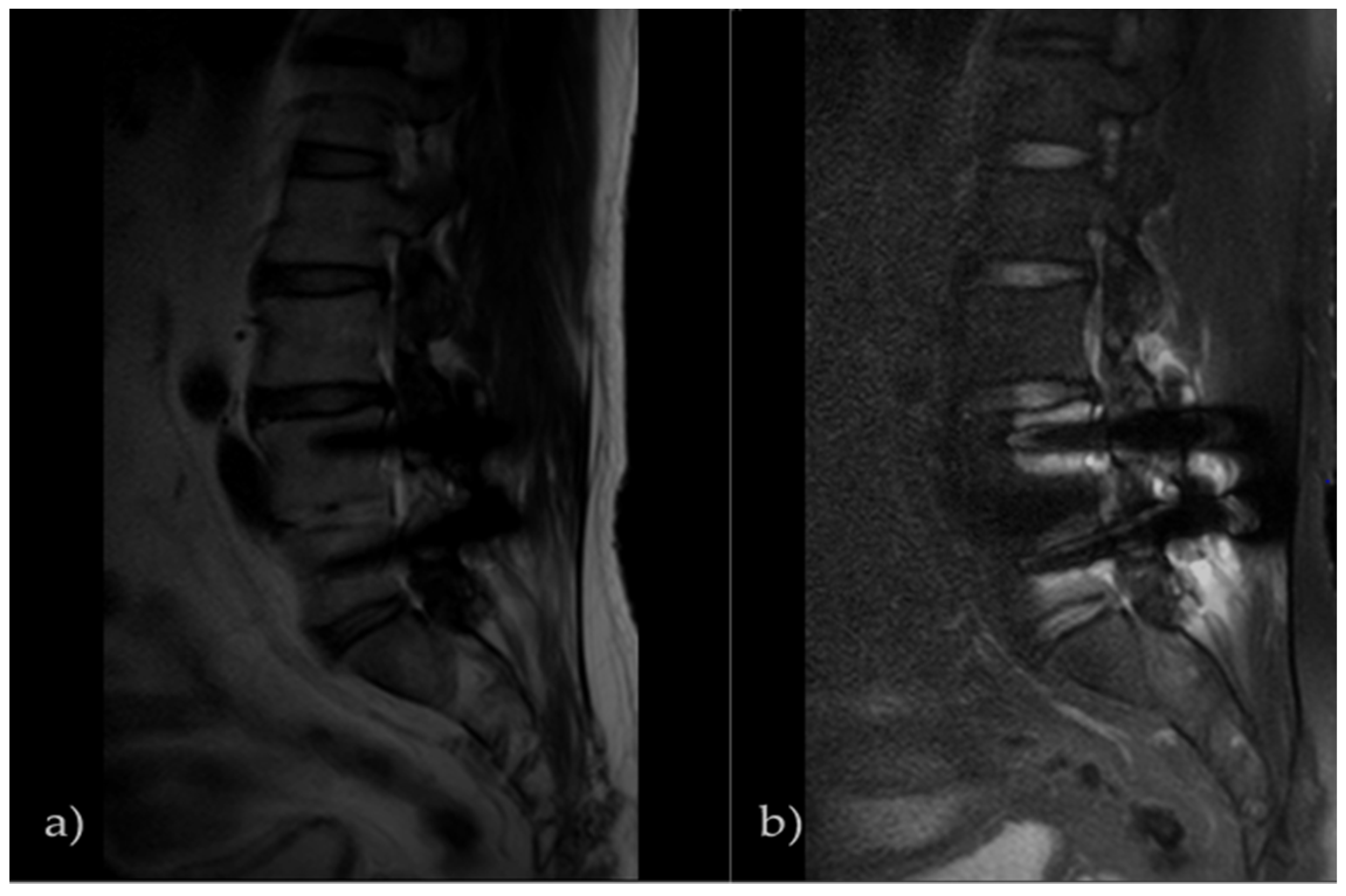
3.1. SNR Result of Pig L-Spine Phantom
3.2. SNR Result of Transplant Patients
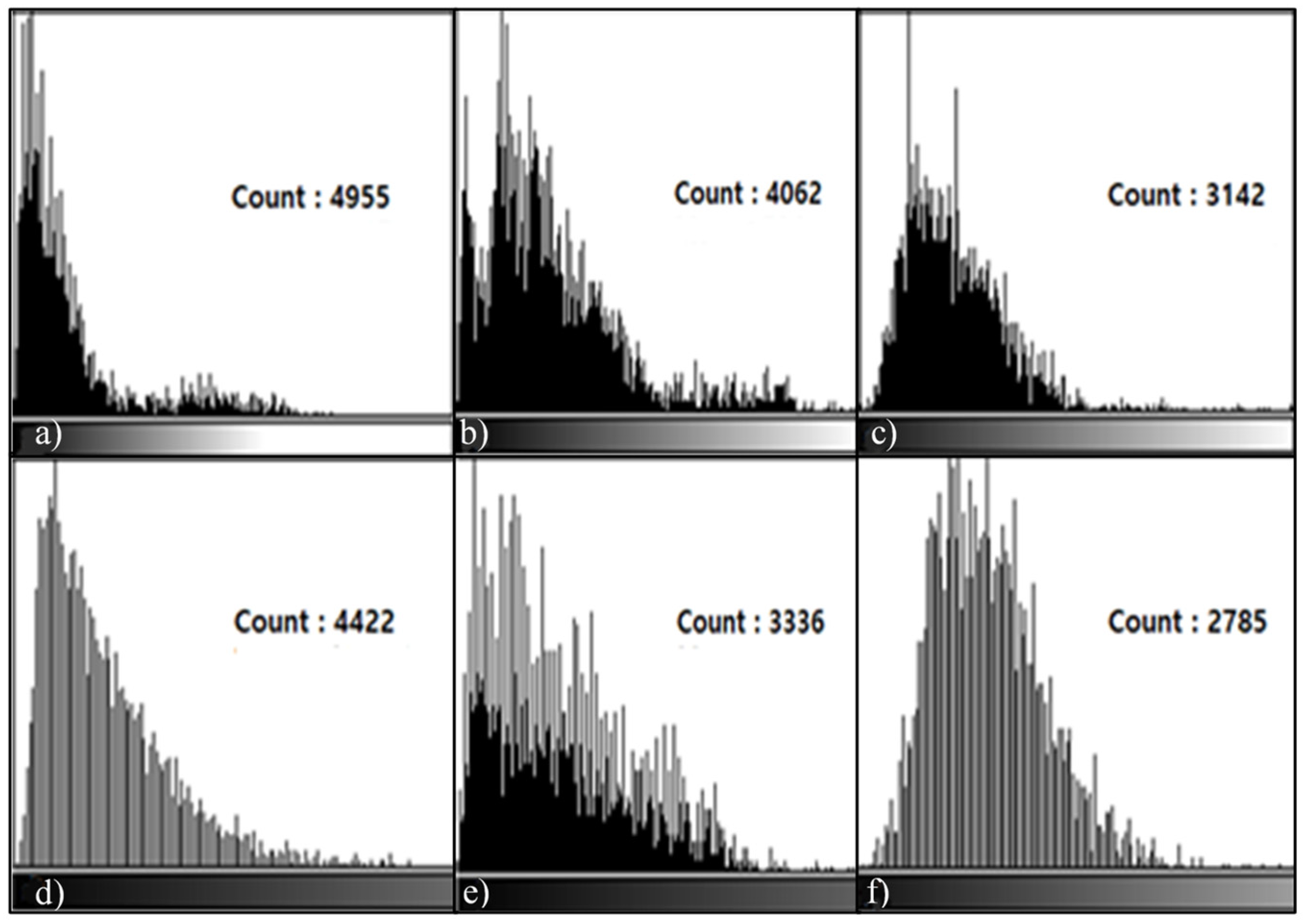
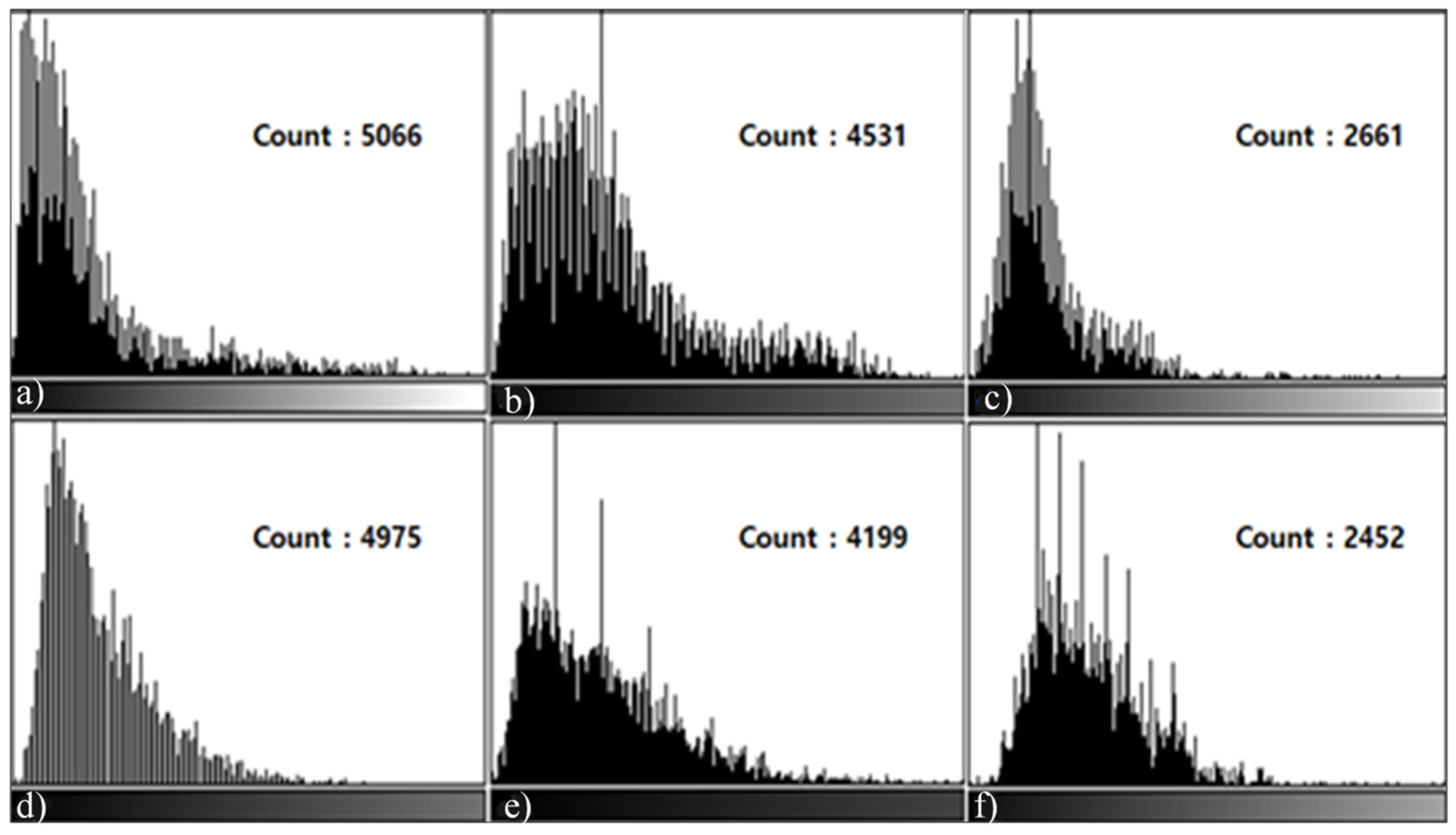
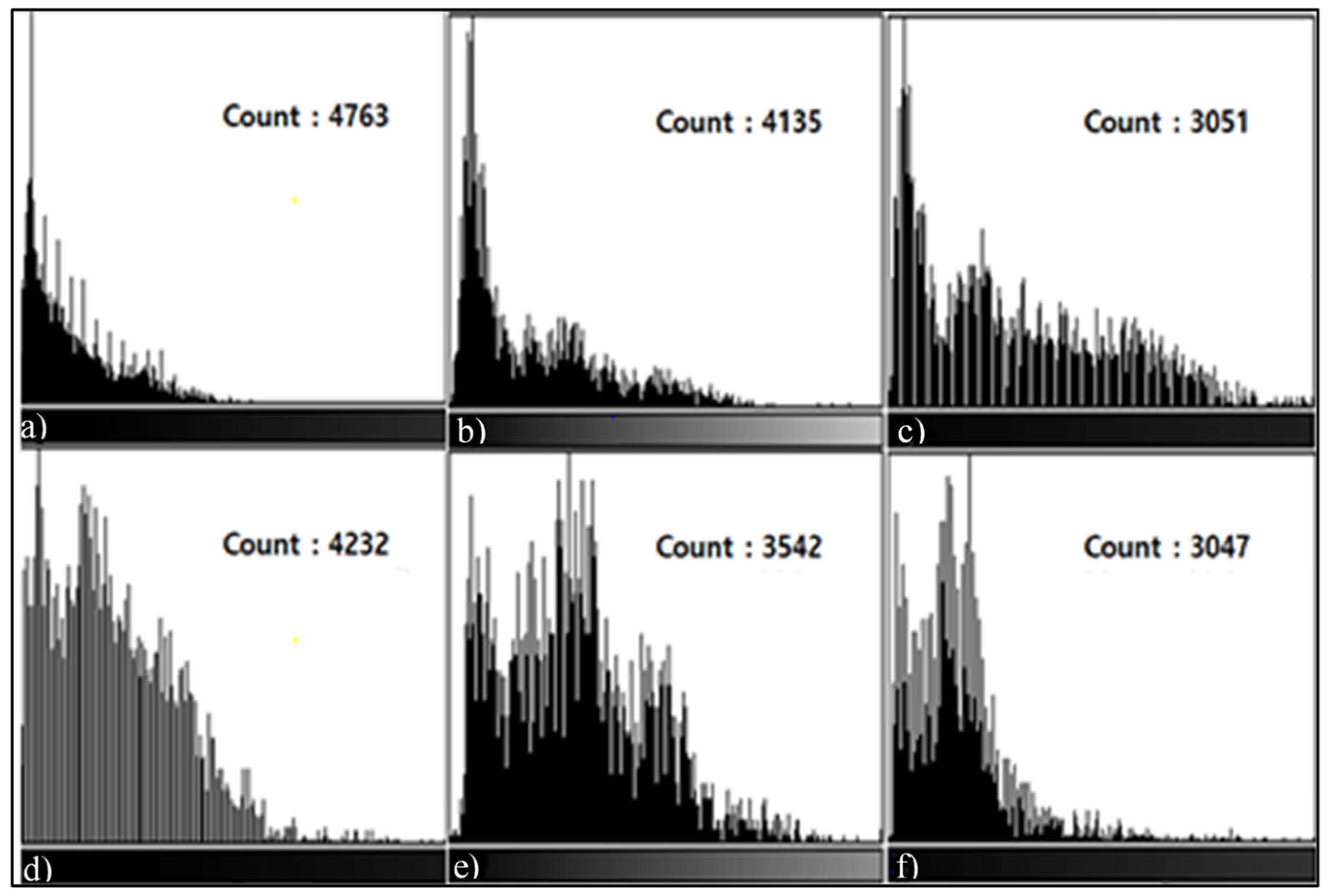
3.3. Histogram Measurement Results
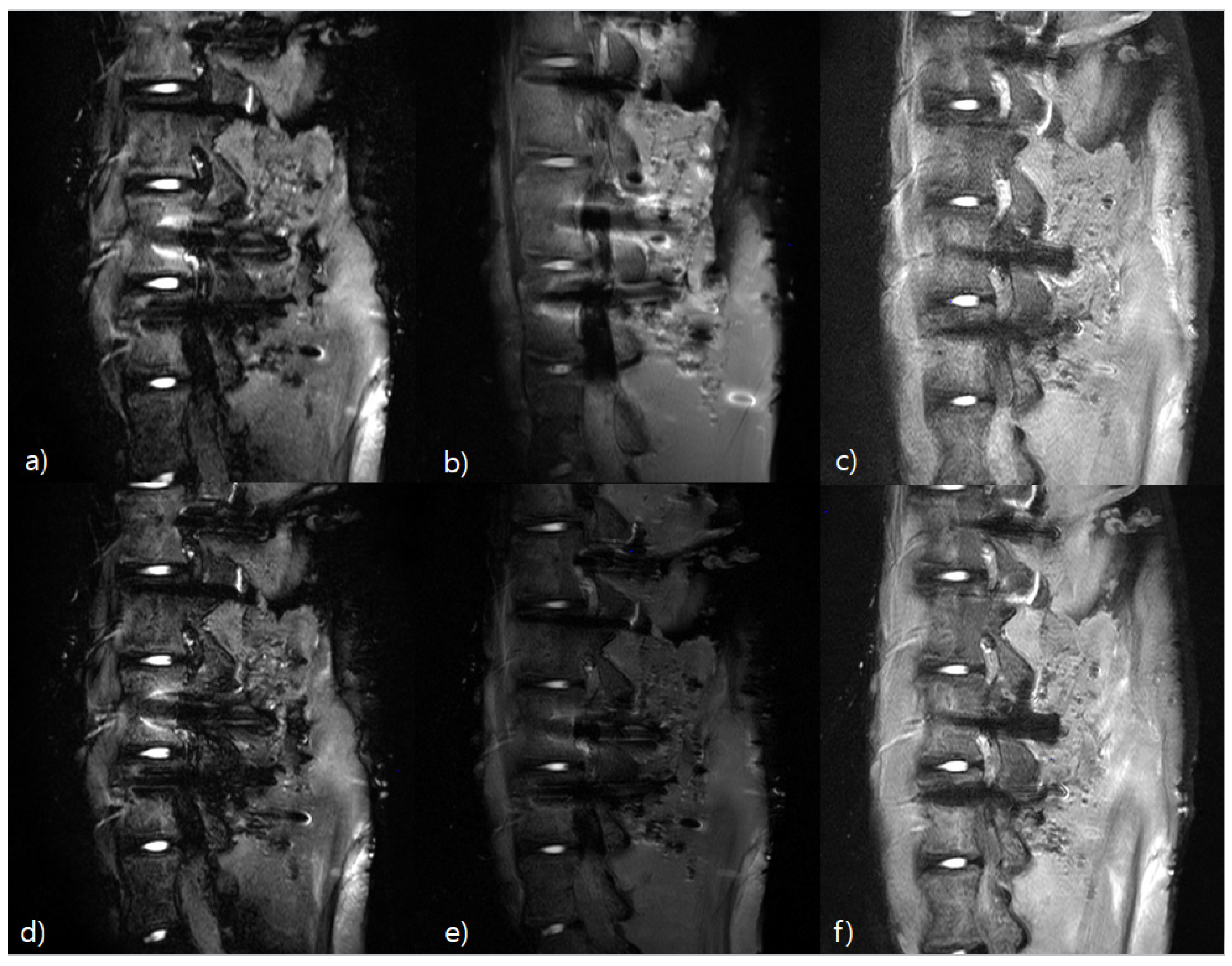
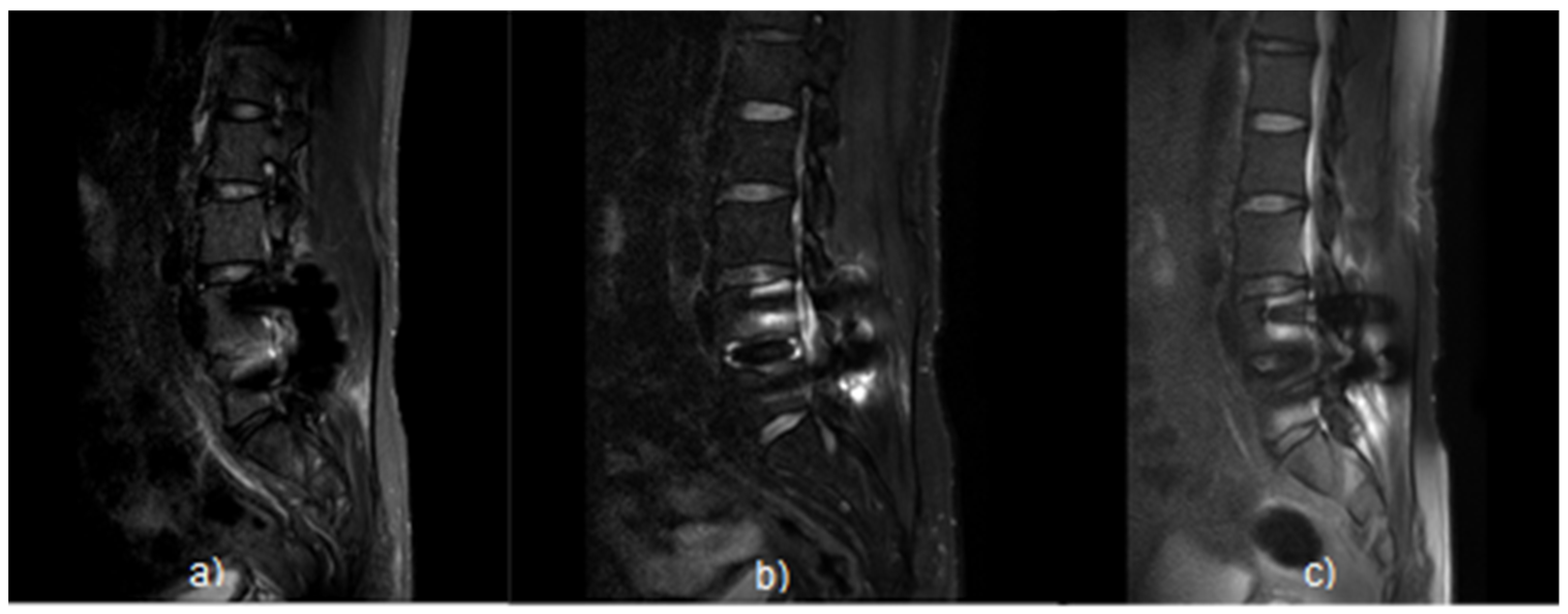
3.4. Qualitative Analysis Results
3.5. Results of Post-Hoc Analysis and Cohen’s Kappa Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Marques, J.P.; Simonis, F.F.J.; Webb, A.G. Low-field MRI: An MR physics perspective. J. Magn. Reson. Imaging 2019, 49, 1528–1542. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, P.M.; Law, M.; Schweitzer, M.E. High-Field MRI. Orthop. Clin. 2006, 37, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhou, Z.; Wang, C.; Cheng, X.; Wang, L.; Duanmu, Y.; Zhang, C.; Veronese, N.; Guglielmi, G. Reliability of measuring the fat content of the lumbar vertebral marrow and paraspinal muscles using MRI mDIXON-Quant sequence. Diagn. Interv. Radiol. 2018, 24, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H. Evaluation of O-MAR XD Technique for Reduction of Magnetic Susceptibility Artifact of Knee Implant. J. Radiol. Sci. Technol. 2018, 41, 413–419. [Google Scholar] [CrossRef]
- Talbot, B.S.; Weinberg, E.P. MR Imaging with Metal-suppression Sequences for Evaluation of Total Joint Arthroplasty. Radiographics 2016, 36, 209–225. [Google Scholar] [CrossRef]
- Kim, H. Quantitative evaluation of MRI distortion using orthopedic prosthetic metal. J. Radiol. Sci. Technol. 2013, 7, 57–62. [Google Scholar] [CrossRef]
- Cho, Y.B.; Sung, J.G.; Young, Y.; Yang, S.W.; Seo, D.G. Study to reduce the swap artifact occurring fat suppression Dixon technique used in the MRI scans through self-correction material. J. Korean Soc. MR Technol. 2015, 25, 77–86. [Google Scholar]
- Ma, J. Dixon techniques for water and fat imaging. J. Magn. Reson. Imaging 2008, 28, 543–558. [Google Scholar] [CrossRef]
- Jung, D.B.; Lee, H.K.; Heo, Y.C. Comparison of mDixon, T2 TSE, and T2 SPIR Images in Magnetic Resonance Imaging of Lumbar Sagittal Plane. J. Korean Soc. Radiol. 2021, 15, 927–933. [Google Scholar]
- Park, M.C.; Lee, J.H.; Kim, K.J.; Bae, S.H. Evaluation of Usefulness of an m-DIXON Technique during an Abdomen MRI Examination: A Comparison with an e-THRIVE Technique. J. Digit. Converg. 2014, 12, 385–390. [Google Scholar] [CrossRef]
- Perkins, T.G.; Duijndam, A.; Eggers, H.; de Weerdt, E.; Rijckaert, Y.H.E. mDIXON XD—The Next Generation Fat-Free Imaging. PHILIPS 2015; MR Software: Madhya Pradesh, India, 2015. [Google Scholar]
- Back, I.H.; Pee, W.H.; Kim, J.D.; Lee, S.K. Usability Evaluation of mDixon Technique by Comparing with Fat-suppression Techniques Metal Artifacts. J. Korean Soc. MR Technol. 2015, 25, 71–76. [Google Scholar]
- Lee, Y.H.; Hahn, S.; Kim, E.J.; Suh, J.S. Fat-suppressed MR Imaging of the Spine for Metal Artifact Reduction at 3T: Comparison of STIR and Slice Encoding for Metal Artifact Correction Fat-suppressed T2-weighted Images. Magn. Reson. Med. Sci. 2016, 15, 371–378. [Google Scholar] [CrossRef] [PubMed]
- de Cesar Netto, C.; Fonseca, L.F.; Fritz, B.; Stern, S.E.; Raithel, E.; Nittka, M.; Schon, L.C.; Fritz, J. Metal artifact reduction MRI of total ankle arthroplasty implants. Eur. Radiol. 2018, 28, 2216–2227. [Google Scholar] [CrossRef]
- Reichert, M.; Ai, T.; Morelli, J.N.; Nittka, M.; Attenberger, U.; Runge, V.M. Metal artefact reduction in MRI at both 1.5 and 3.0 T using slice encoding for metal artefact correction and view angle tilting. Br. J. Radiol. 2015, 88, 20140601. [Google Scholar] [CrossRef]
- Geerts Ossevoort, L.; de Weerdt, E.; Duijndam, A.; van Yperen, G.; Peeters, H.; Doneva, M.; Nijenhuis, M.; Huang, A. Compressed SENSE, Speed Done Right. Every Time. Available online: https://philipsproductcontent.blob.core.windows.net/assets/20180109/619119731f2a42c4acd4a863008a46c7.pdf (accessed on 6 July 2022).
- Lustig, M.; Donoho, D.L.; Santos, J.M.; Pauly, J.M. Compressed Sensing MRI[A look at how CS can improve on current imaging techniques]. IEEE Signal Process. Mag. 2008, 25, 72–82. [Google Scholar] [CrossRef]
- Rani, M.; DHOK, S.B.; DESHMUKH, R.B. A Systematic Review of Compressive Sensing: Concepts, Implementations and Applications. IEEE Access 2018, 6, 4875–4894. [Google Scholar] [CrossRef]
- Lu, W.; Pauly, K.B.; Gold, G.E.; Pauly, J.M.; Hargreaves, B.A. SEMAC: Slice encoding for metal artifact correction in MRI. Magn. Reson. Med. 2009, 62, 66–76. [Google Scholar] [CrossRef]
- Filli, L.; Jud, L.; Luechinger, R.; Nanz, D.; Andreisek, G.; Runge, V.M.; Kozerke, S.; Farshad-Amacker, N.A. Material-Dependent Implant Artifact Reduction Using SEMAC-VAT and MAVRIC: A Prospective MRI Phantom Study. Investig. Radiol. 2017, 52, 381–387. [Google Scholar] [CrossRef]
- Gutierrez, L.B.; Do, B.H.; Gold, G.E.; Hargreaves, B.A.; Koch, K.M.; Worters, P.W.; Stevens, K.J. MR Imaging Near Metallic Implants Using MAVRIC SL: Initial Clinical Experience at 3T. Acad. Radiol. 2015, 22, 370–379. [Google Scholar] [CrossRef]
- Kishida, Y.; Koyama, H.; Seki, S.; Yoshikawa, T.; Kyotani, K.; Okuaki, T.; Sugimura, K.; Ohno, Y. Comparison of fat suppression capability for chest MR imaging with Dixon, SPAIR and STIR techniques at 3 Tesla MR system. Magn. Reson. Imaging 2018, 47, 89–96. [Google Scholar] [CrossRef]
- Toms, A.P.; Smith-Bateman, C.; Malcolm, P.N.; Cahir, J.; Graves, M. Optimization of metal artefact reduction (MAR) sequences for MRI of total hip prostheses. Clin. Radiol. 2010, 65, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.Y.; Seethamraju, R.T.; Patel, P.; Hahn, P.F.; Kirsch, J.E.; Guimaraes, A.R. Body MR Imaging: Artifacts, k-Space, and Solutions. Radiographics 2015, 35, 1439–1460. [Google Scholar] [CrossRef] [PubMed]
- Olsen, R.V.; Munk, P.L.; Lee, M.J.; Janzen, D.L.; MacKay, A.L.; Xiang, Q.-S.; Masri, B. Metal Artifact Reduction Sequence: Early Clinical Applications. Radiographics 2000, 20, 699–712. [Google Scholar] [CrossRef] [PubMed]
- Toossi, A.; Bergin, B.; Marefatallah, M.; Parhizi, B.; Tyreman, N.; Everaert, D.G.; Rezaei, S.; Seres, P.; Gatenby, J.C.; Perlmutter, S.I.; et al. Comparative neuroanatomy of the lumbosacral spinal cord of the rat, cat, pig, monkey, and human. Sci. Rep. 2021, 11, 1955. [Google Scholar] [CrossRef]
- Morita, K.; Nakaura, T.; Maruyama, N.; Iyama, Y.; Oda, S.; Utsunomiya, D.; Namimoto, T.; Kitajima, M.; Yoneyama, M.; Yamashita, Y. Hybrid of Compressed Sensing and Parallel Imaging Applied to Three-dimensional Isotropic T(2)-weighted Turbo Spin-echo MR Imaging of the Lumbar Spine. Magn. Reson. Med. Sci. 2020, 19, 48–55. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, J.; He, B. Magnetic-Resonance-Based Electrical Properties Tomography: A Review. IEEE Rev. Biomed. Eng. 2014, 7, 87–96. [Google Scholar] [CrossRef]
- Tokue, H.; Tokue, A.; Tsushima, Y. Unexpected magnetic resonance imaging burn injuries from jogging pants. Radiol. Case Rep. 2019, 14, 1348–1351. [Google Scholar] [CrossRef]
- Qi, S.; Wu, Z.G.; Mu, Y.F.; Gao, L.L.; Yang, J.; Zuo, P.L.; Nittka, M.; Liu, Y.; Wang, H.Q.; Yin, H. SEMAC-VAT MR Imaging Unravels Peri-instrumentation Lesions in Patients With Attendant Symptoms After Spinal Surgery. Medicine 2016, 95, e3184. [Google Scholar] [CrossRef]
- Pokorney, A.L.; Chia, J.M.; Pfeifer, C.M.; Miller, J.H.; Hu, H.H. Improved fat-suppression homogeneity with mDIXON turbo spin echo (TSE) in pediatric spine imaging at 3.0 T. Acta Radiol. 2017, 58, 1386–1394. [Google Scholar] [CrossRef]
- Molière, S.; Dillenseger, J.-P.; Ehlinger, M.; Kremer, S.; Bierry, G. Comparative study of fat-suppression techniques for hip arthroplasty MR imaging. Skelet. Radiol. 2017, 46, 1209–1217. [Google Scholar] [CrossRef]
| Features | Mean ± SD/(%) |
|---|---|
| Age (years) | 64.23 ± 12.98 |
| Males/Females | 22 (73.33%)/8 (26.66) |
| Height (m) | 1.6 ± 0.2 |
| Weight (kg) | 68.25 ± 15.32 |
| Osteoporosis | 13 (43.33%) |
| DDD | 15 (50%) |
| IS | 2 (6.66%) |
| LT | 30 (100%) |
| Parameters | T1 mDixon | T1 STIR | T1 SEMAC | T2 mDixon | T2 STIR | T2 SEMAC (d) |
|---|---|---|---|---|---|---|
| TR (a) (ms) | 558 | 3110 | 589 | 1980 | 3215 | 2100 |
| TE (b) (ms) | 18 | 60 | 50 | 120 | 110 | 110 |
| ST (mm) | 3 | 3 | 3 | 3 | 3 | 3 |
| Gap (mm) | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 |
| Matrix | 416 × 250 | 416 × 233 | 416 × 250 | 416 × 239 | 416 × 250 | 416 × 250 |
| NEX | 2 | 2 | 2 | 2 | 2 | 2 |
| FOV (mm) | 250 | 250 | 250 | 250 | 250 | 250 |
| ST (c) | 02 : 36 | 06 : 19 | 10 : 55 | 04 : 06 | 07 : 24 | 14 : 23 |
| Units | mDixon (O-MAR) a | STIR b | CS-SEMAC (SPIR) c | p-Value | |
|---|---|---|---|---|---|
| L-2 | T1 Sagittal | 19.32 ± 0.79 | 11.20 ± 0.48 | 28.13 ± 1.24 | <0.001, (c > a > b) |
| T2 Sagittal | 18.49 ± 0.81 | 9.52 ± 0.42 | 33.15 ± 1.52 | <0.001, (c > a > b) | |
| L3 | T1 Sagittal | 17.52 ± 0.61 | 9.59 ± 0.34 | 22.81 ± 0.96 | <0.001, (c > a > b) |
| T2 Sagittal | 14.23 ± 0.62 | 6.82 ± 0.27 | 23.25 ± 1.04 | <0.001, (c > a > b) | |
| L4 | T1 Sagittal | 10.31 ± 0.41 | 7.46 ± 0.32 | 10.92 ± 0.45 | <0.001, (c > a > b) |
| T2 Sagittal | 8.51 ± 0.38 | 4.84 ± 0.17 | 12.77 ± 0.47 | <0.001, (c > a > b) | |
| Units | mDixon (O-MAR) a | STIR b | CS-SEMAC (SPIR) c | p-Value | |
|---|---|---|---|---|---|
| L3 | T1 Sagittal | 18.75 ± 0.91 | 7.80 ± 0.27 | 32.55 ± 1.12 | <0.001, (c > a > b) |
| T2 Sagittal | 12.20 ± 0.24 | 9.01 ± 0.58 | 22.27 ± 0.84 | <0.001, (c > a > b) | |
| L4 | T1 Sagittal | 8.93 ± 0.44 | 7.58 ± 0.21 | 14.2 ± 0.65 | <0.001, (c > a > b) |
| T2 Sagittal | 10.42 ± 0.90 | 8.09 ± 0.52 | 12.17 ± 0.44 | <0.001, (c > a > b) | |
| L5 | T1 Sagittal | 11.40 ± 0.49 | 7.69 ± 0.28 | 22.17 ± 0.89 | <0.001, (c > a > b) |
| T2 Sagittal | 11.09 ± 0.27 | 8.24 ± 0.47 | 18.13 ± 0.61 | <0.001, (c > a > b) | |
| Units | mDixon (O-MAR) a | STIR b | CS-SEMAC (SPIR) c | p-Value | |
|---|---|---|---|---|---|
| L4 | T1 Sagittal | 7.42 ± 0.20 | 4.61 ± 0.11 | 13.28 ± 0.74 | <0.001, (c > a > b) |
| T2 Sagittal | 9.59 ± 0.84 | 8.34 ± 0.12 | 19.25 ± 0.48 | <0.001, (c > a > b) | |
| L5 | T1 Sagittal | 10.25 ± 0.50 | 6.33 ± 0.14 | 15.13 ± 0.43 | <0.001, (c > a > b) |
| T2 Sagittal | 15.01 ± 0.40 | 7.47 ± 0.15 | 17.03 ± 0.30 | <0.001, (c > a > b) | |
| mDixon (O-MAR) a | STIR b | CS_SEMAC (SPIR) c | p-Value | ||
|---|---|---|---|---|---|
| Overall Image Qualities | T1 Sagittal | (2.2–5) | (2–4) | (3.5–5) | <0.001, (c > a > b) |
| T2 Sagittal | (2.5–5) | (2.5–4) | (3.5–5) | <0.001, (c > a > b) | |
| Susceptibility Artifact | T1 Sagittal | (2.5–5) | (2.5–4) | (3.8–5) | <0.001, (c > a > b) |
| T2 Sagittal | (2.5–5) | (2.5–4) | (3.2–5) | <0.001, (c > a > b) | |
| Pedicle Visualization | T1 Sagittal | (2–5) | (2–4) | (3–5) | <0.001, (c > a > b) |
| T2 Sagittal | (2–5) | (2.1–4) | (3–5) | <0.001, (c > a > b) | |
| T2 mDixon (O-MAR) a | T2 CS-SEMAC (SPIR) b | T2 STIR c | p-Value | |
|---|---|---|---|---|
| Overall Image Qualities | (2.5–5) | (3.5–5) | (2.5–4) | <0.001, (b > a > c) |
| Susceptibility Artifact | (2.5–4) | (3–5) | (2–4) | <0.001, (b > a > c) |
| Pedicle Visualization | (3–5) | (3.5–5) | (2.4–5) | <0.001, (b > a > c) |
| Spine Body | Artifact | Diff. | SRR(%) | ||
|---|---|---|---|---|---|
| mDixon (O-MAR) | L4 | 59.20 ± 7.25 | 22.45 ± 4.20 | 36.75 ± 2.15 | 65.58 |
| L5 | 54.21 ± 5.02 | 20.10 ± 2.08 | 33.76 ± 2.07 | 62.27 | |
| STIR | L4 | 45.10 ± 4.60 | 13.84 ± 2.03 | 31.26 ± 1.81 | 72.45 |
| L5 | 48.27 ± 3.10 | 16.25 ± 2.87 | 32.02 ± 0.16 | 66.33 | |
| CS_SEMAC (SPIR) | L4 | 67.74 ± 8.57 | 29.23 ± 3.57 | 38.51 ± 3.53 | 56.84 |
| L5 | 70.56 ± 7.41 | 32.04 ± 2.30 | 38.52 ± 3.61 | 54.59 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goo, E.-H.; Kim, S.-S. Evaluating Compressed SENSE (CS) MRI Metal Artifact Reduction Using Pig L-Spine Phantom and Transplant Patients: Focused on the CS-SEMAC (SPIR), mDixon(O-MAR) and STIR Techniques. Tomography 2022, 8, 2298-2312. https://doi.org/10.3390/tomography8050192
Goo E-H, Kim S-S. Evaluating Compressed SENSE (CS) MRI Metal Artifact Reduction Using Pig L-Spine Phantom and Transplant Patients: Focused on the CS-SEMAC (SPIR), mDixon(O-MAR) and STIR Techniques. Tomography. 2022; 8(5):2298-2312. https://doi.org/10.3390/tomography8050192
Chicago/Turabian StyleGoo, Eun-Hoe, and Sung-Soo Kim. 2022. "Evaluating Compressed SENSE (CS) MRI Metal Artifact Reduction Using Pig L-Spine Phantom and Transplant Patients: Focused on the CS-SEMAC (SPIR), mDixon(O-MAR) and STIR Techniques" Tomography 8, no. 5: 2298-2312. https://doi.org/10.3390/tomography8050192
APA StyleGoo, E.-H., & Kim, S.-S. (2022). Evaluating Compressed SENSE (CS) MRI Metal Artifact Reduction Using Pig L-Spine Phantom and Transplant Patients: Focused on the CS-SEMAC (SPIR), mDixon(O-MAR) and STIR Techniques. Tomography, 8(5), 2298-2312. https://doi.org/10.3390/tomography8050192






