Cytoreductive Surgical Treatment of Pleural Mesothelioma in a Porcine Model Using Magnetic-Resonance-Guided Focused Ultrasound Surgery (MRgFUS) and Radiofrequency Ablation (RFA)
Abstract
1. Introduction
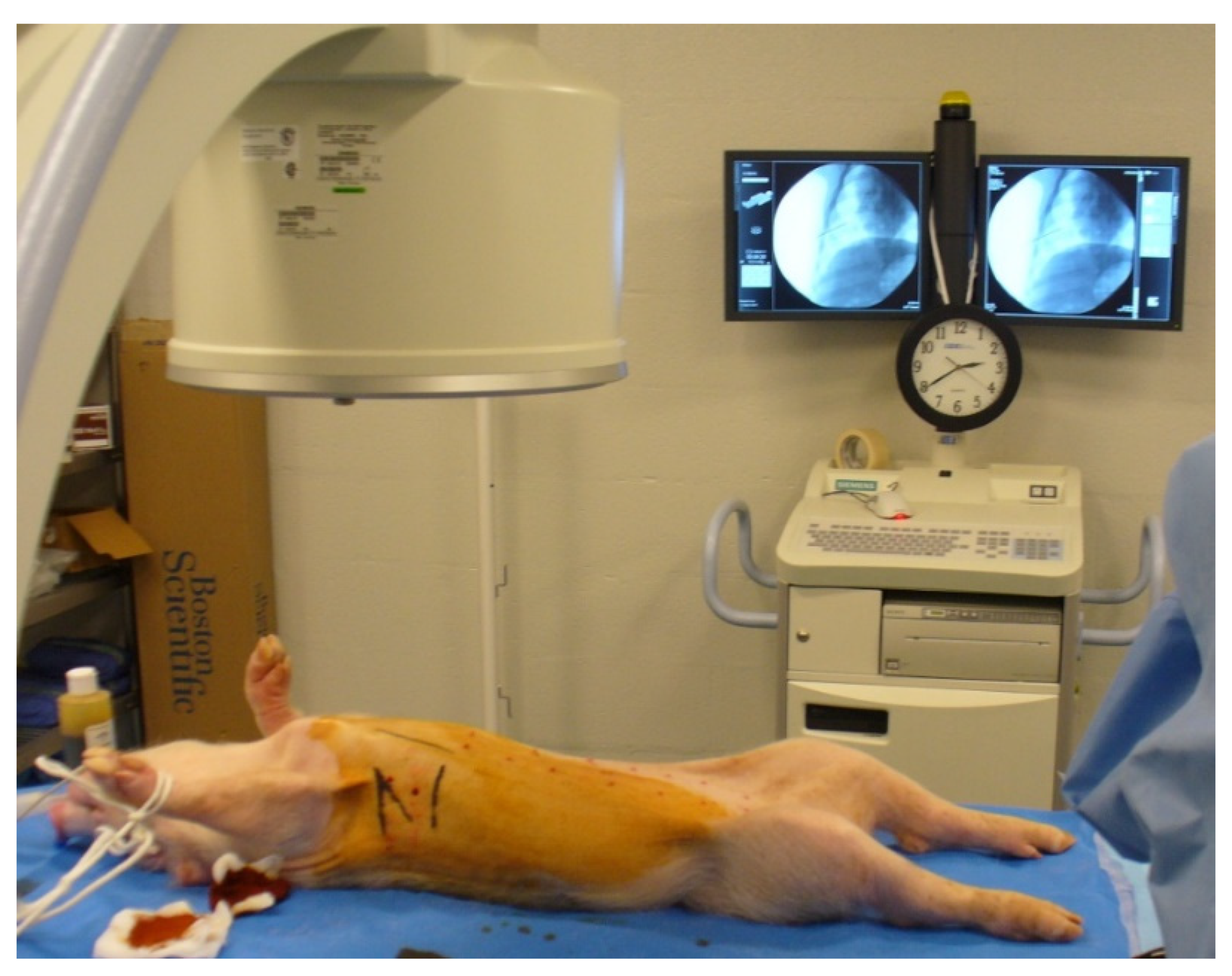
2. Materials and Methods
3. Results
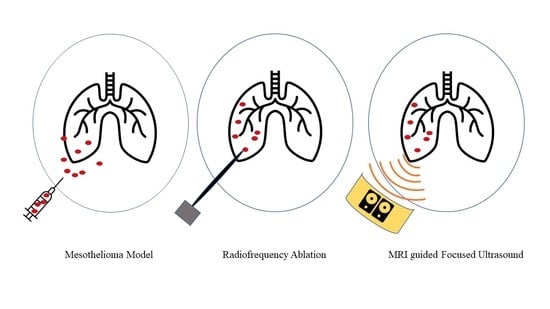
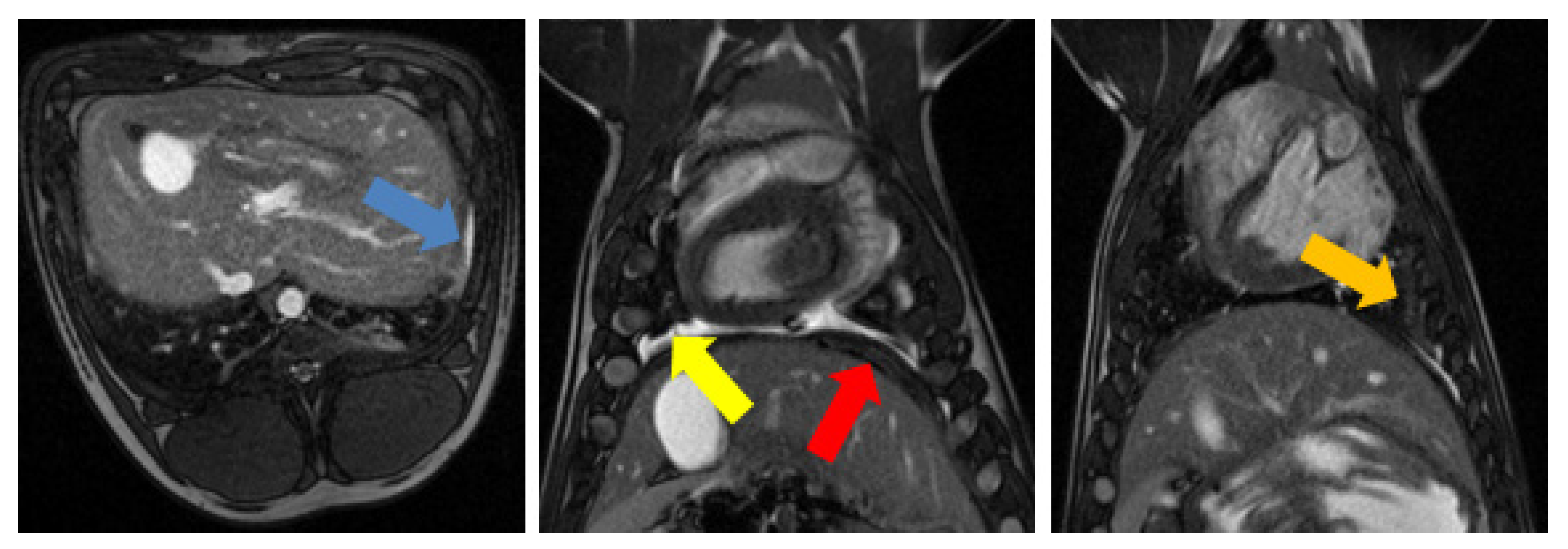
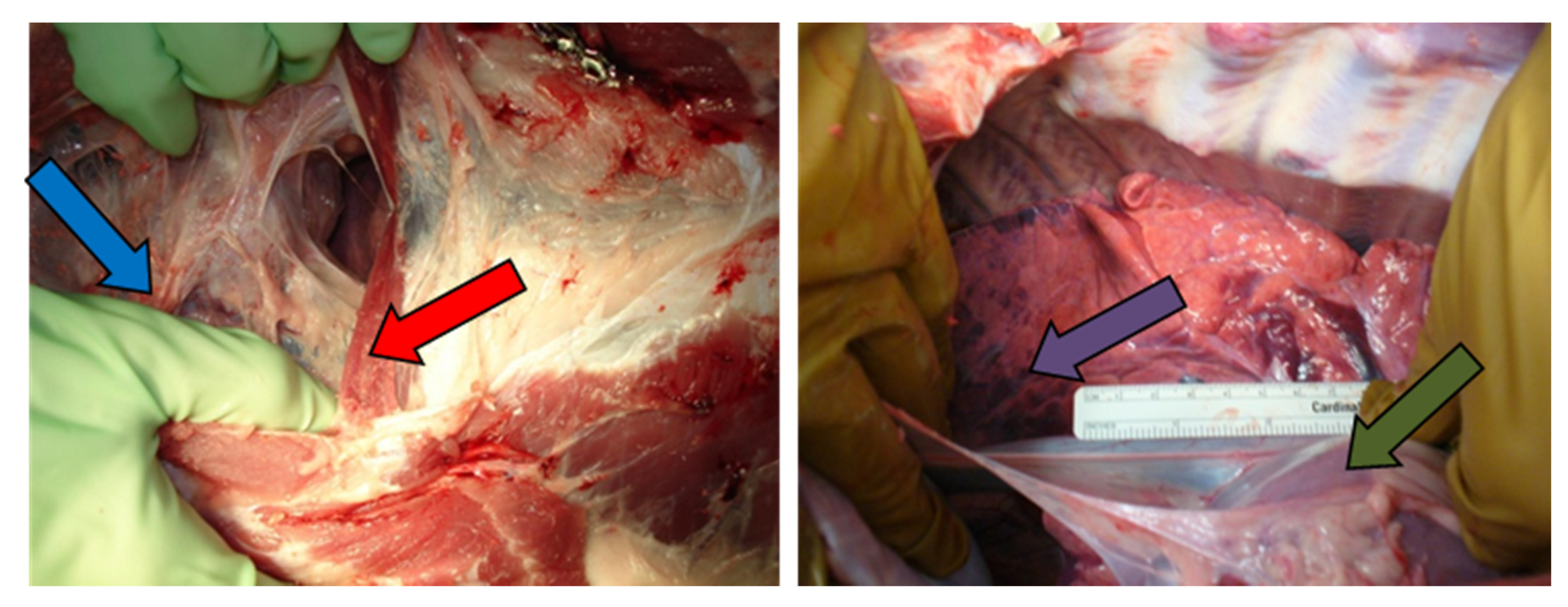
3.1. Tumor Model
3.2. Radiofrequency Ablation
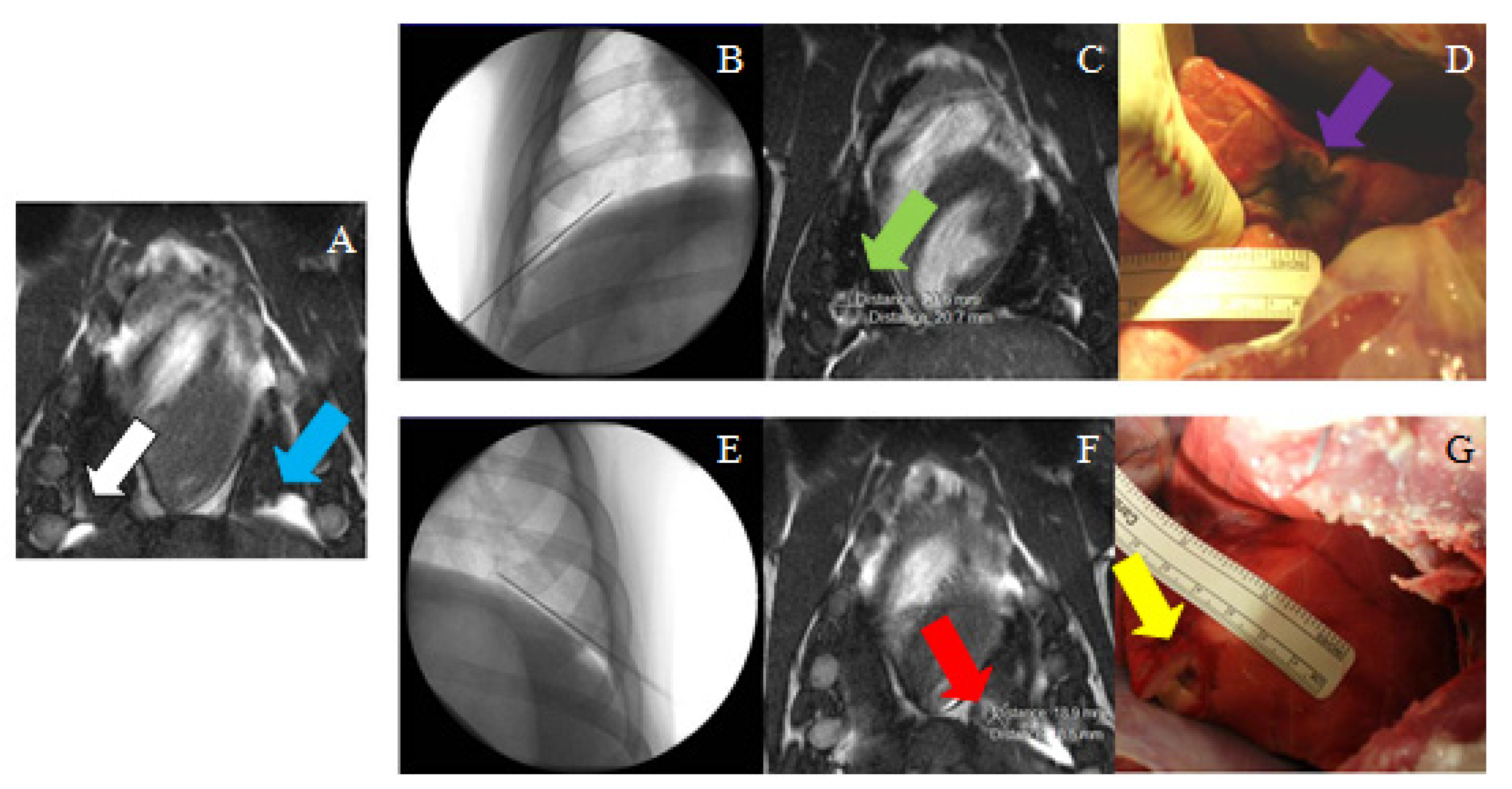
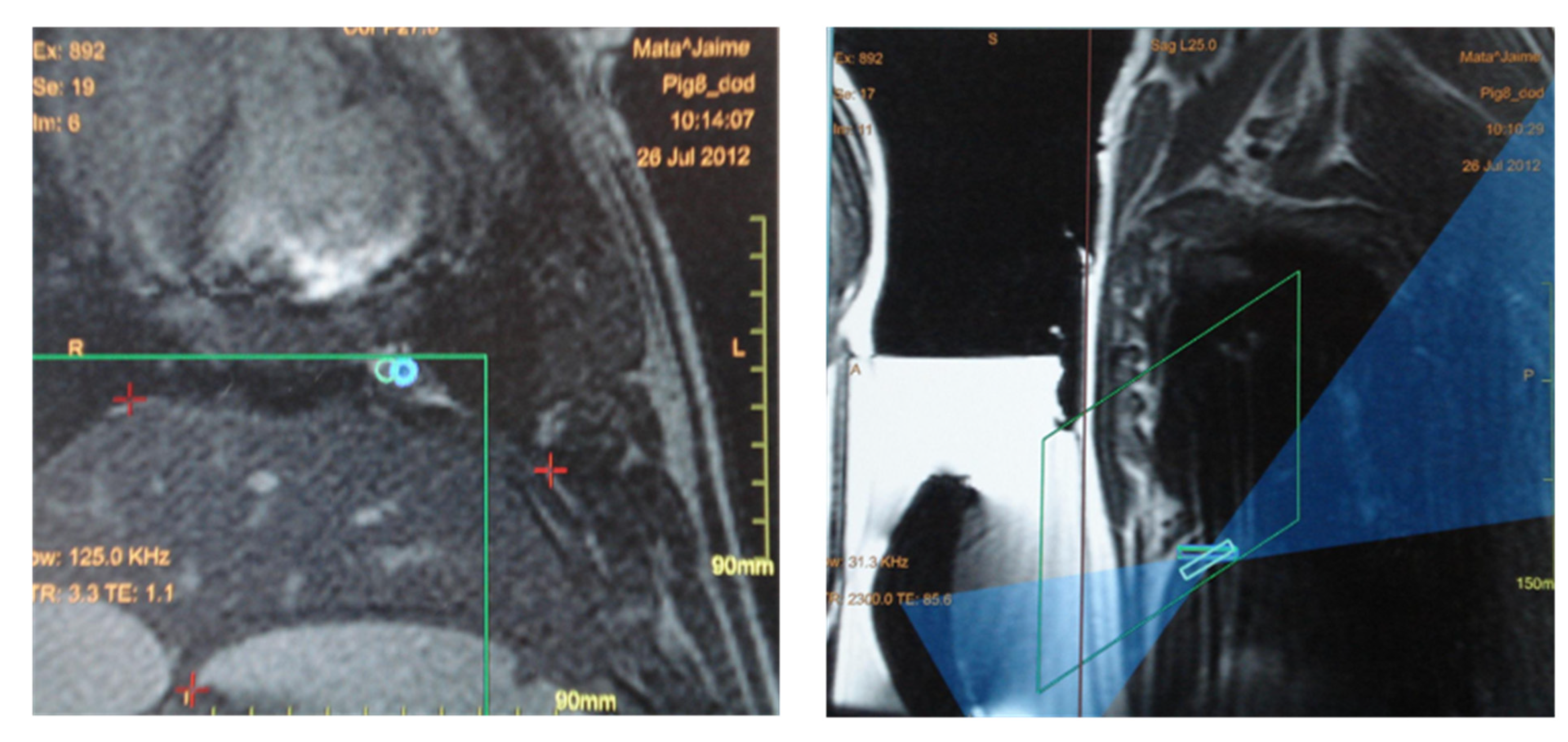
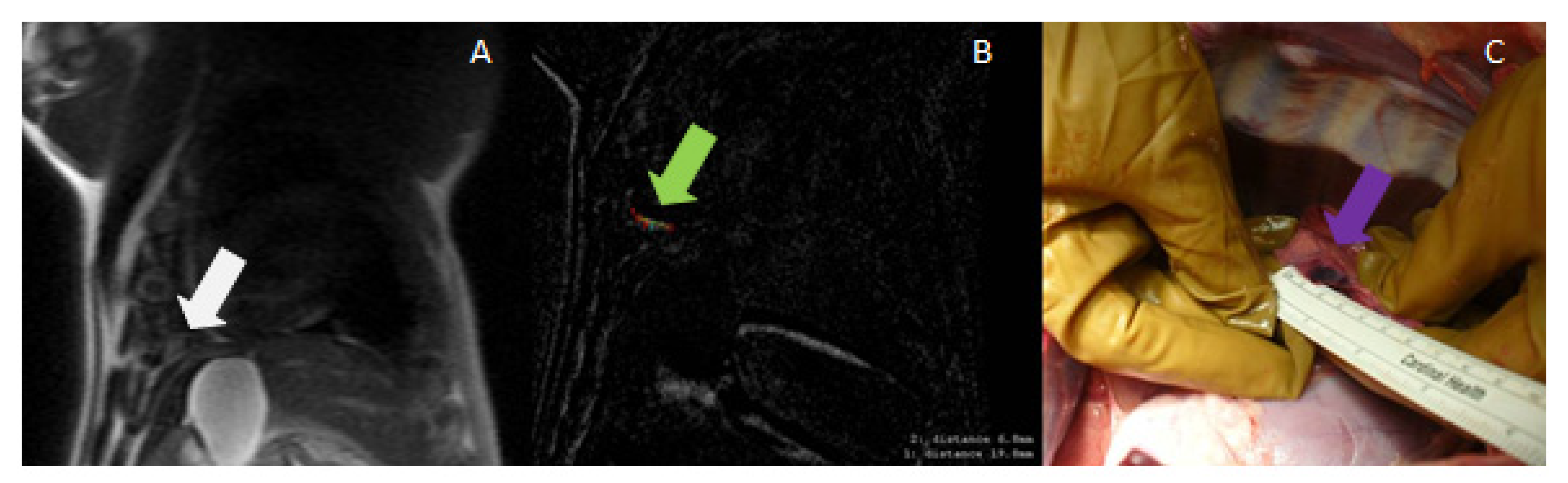
3.3. Magnetic-Resonance-Guided Focused Ultrasound
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Robinson, B.W.S.; Lake, R.A. Advances in Malignant Mesothelioma. N. Engl. J. Med. 2005, 353, 1591–1603. [Google Scholar] [CrossRef] [PubMed]
- Scott, B.; Mukherjee, S.; Lake, R.; Robinson, B.W.S. Malignant mesothelioma. In Textbook of Lung Cancer; Hanson, H., Ed.; Martin Dunitz: London, UK, 2000; pp. 273–293. [Google Scholar]
- Pass, H.I.; Kranda, K.; Temeck, B.K.; Feuerstein, I.; Steinberg, S.M. Surgically debulked malignant pleural mesothelioma: Results and prognostic factors. Ann. Surg. Oncol. 1997, 4, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Stewart, D.J.; Martin-Ucar, A.; Pilling, J.E.; Edwards, J.G.; O’Byrne, K.J.; Waller, D.A. The effect of extent of local resection on patterns of disease progression in malignant pleural mesothelioma. Ann. Thorac. Surg. 2004, 78, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Sugarbaker, D.J.; Jaklitsch, M.T.; Bueno, R.; Richards, W.; Lukanich, J.; Mentzer, S.J.; Colson, Y.; Linden, P.; Chang, M.; Capalbo, L.; et al. Prevention, early detection, and management of complications after 328 consecutive extrapleural pneumonectomies. J. Thorac. Cardiovasc. Surg. 2004, 128, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.C.G.; Light, R.W.; Musk, A.W. Management of malignant pleural mesothelioma: A critical review. Curr. Opin. Pulm. Medicine. 2000, 6, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Abtin, F.G.; Eradat, J.; Gutierrez, A.J.; Lee, C.; Fishbein, M.C.; Suh, R.D. Radiofrequency Ablation of Lung Tumors: Imaging Features of the Postablation Zone. Radio Graph. 2012, 32, 947–969. [Google Scholar] [CrossRef] [PubMed]
- Izadifar, Z.; Izadifar, Z.; Chapman, D.; Babyn, P. An Introduction to High Intensity Focused Ultrasound: Systematic Review on Principles, Devices, and Clinical Applications. JCM 2020, 9, 460. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Orsi, F.; Arnone, P.; Chen, W. High intensity focused ultrasound ablation: A new therapeutic option for solid tumors. J. Can. Res. Ther. 2010, 6, 414. [Google Scholar] [CrossRef]
- Tailly, T.; Goeman, L.; Gontero, P.; Joniau, S. The safety and oncologic efficacy of radio-frequency ablation for the treatment of small renal masses: Comprehensive review of the current literature. Panminerva Med. 2010, 52, 319–329. [Google Scholar]
- Prud’homme, C.; Deschamps, F.; Moulin, B.; Hakime, A.; Al-Ahmar, M.; Moalla, S.; Roux, C.; Teriitehau, C.; de Baere, T.; Tselikas, L. Image-guided lung metastasis ablation: A literature review. Int. J. Hyperthermia. 2019, 36, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.K.; Chou, H.P.; Sheu, M.H. Image-guided lung tumor ablation: Principle, technique, and current status. J. Chin. Med. Assoc. 2013, 76, 303–311. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Frey, B.M.; Sieber, M.; Mettler, D.; Gänger, H.; Frey, F.J. Marked interspecies differences between humans and pigs in cyclosporine and prednisolone disposition. Drug Metab. Dispos. 1988, 16, 285–289. [Google Scholar] [PubMed]
- Cibulskyte, D.; Pedersen, M.; Jakobsen, P.; Hansen, H.; Mortensen, J. Pharmacokinetic characterization of a pig model of ciclosporin A nephrotoxicity following intravenous administration. Pharmacol. Res. 2007, 56, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Crick, S.J.; Sheppard, M.N.; Ho, S.Y.; Gebstein, L.; Anderson, R.H. Anatomy of the pig heart: Comparisons with normal human cardiac structure. J. Anat. 1998, 193 Pt 1, 105–119. [Google Scholar] [CrossRef] [PubMed]
- Iazzio, P. Handbook of Cardiac Anatomy, Physiology, and Devices, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Naimark, W.A.; Lee, J.M.; Limeback, H.; Cheung, D.T. Correlation of structure and viscoelastic properties in the pericardia of four mammalian species. Am. J. Physiology-Heart Circ. Physiol. 1992, 263, H1095–H1106. [Google Scholar] [CrossRef] [PubMed]
- Gorny, K.R.; Chen, S.; Hangiandreou, N.J.; Hesley, G.K.; Woodrum, D.A.; Brown, D.L.; Felmlee, J.P. Initial evaluation of acoustic reflectors for the preservation of sensitive abdominal skin areas during MRgFUS treatment. Phys. Med. Biol. 2009, 54, N125–N133. [Google Scholar] [CrossRef] [PubMed]
- Salomir, R.; Petrusca, L.; Auboiroux, V.; Muller, A.; Vargas, M.I.; Morel, D.R.; Goget, T.; Breguet, R.; Terraz, S.; Hopple, J.; et al. Magnetic Resonance–Guided Shielding of Prefocal Acoustic Obstacles in Focused Ultrasound Therapy: Application to Intercostal Ablation in Liver. Investig. Radiol. 2013, 48, 366–380. [Google Scholar] [CrossRef] [PubMed]
- Wolfram, F.; Güllmar, D.; Böttcher, J.; Schubert, H.; Bischoff, S.; Reichenbach, J.R.; Lesser, T.G. Assessment of MR imaging during one-lung flooding in a large animal model. Magn Reson Mater. Phy. 2019, 32, 581–590. [Google Scholar] [CrossRef] [PubMed]
- Bitton, R.R.; Webb, T.D.; Pauly, K.B.; Ghanouni, P. Improving Thermal Dose Accuracy in Magnetic Resonance-Guided Focused Ultrasound Surgery: Long-Term Thermometry Using a Prior Baseline as a Reference: Improving Thermal Dose Accuracy in MRgFUS. J. Magn. Reson. Imaging 2016, 43, 181–189. [Google Scholar] [CrossRef] [PubMed][Green Version]
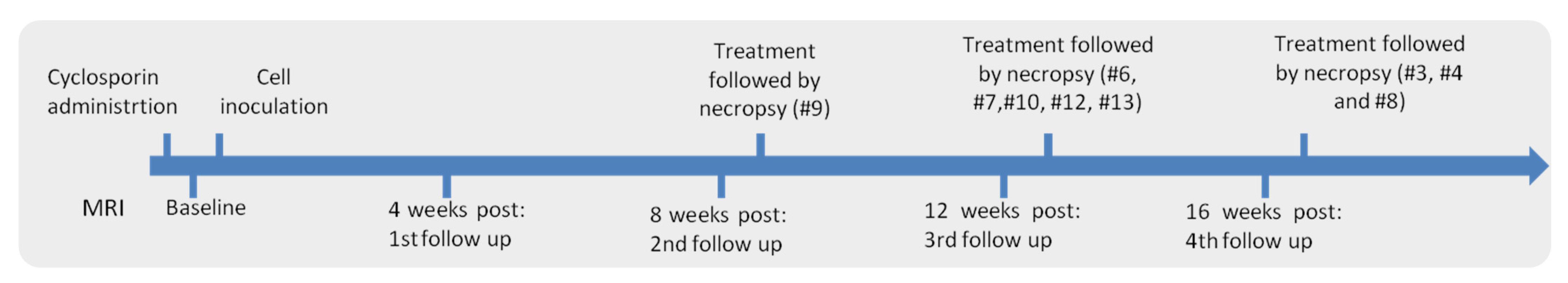
| Animal # | Cell Solution Injected (mL) | Cyclosporine Dose (mg/kg/day) | Type of Treatment Received |
|---|---|---|---|
| 1 | 6.0 | 10 (until euthanasia) | Test (none) |
| 2 | 13.0 | 10 (until euthanasia) | Test (none) |
| 3 | 11.0 | 20 (until 12 weeks post-inoculation) | RFA |
| 4 | 5.0 | 20 (until 12 weeks post-inoculation) | RFA |
| 5 | 5.5 | 20 (died after inoculation) | -- |
| 6 | 9.0 | 20 (until 12 weeks post-inoculation) | RFA |
| 7 | 7.0 | 20 (until 8 weeks post-inoculation) | RFA |
| 8 | 7.0 | 20 (until 8 weeks post-inoculation) | MRgFUS |
| 9 | 10.0 | 20 (until 8 weeks post-inoculation) | RFA |
| 10 | 5.0 | 20 (until 4 weeks post-inoculation) | MRgFUS |
| 11 | 6.0 | 20 (died after inoculation) | -- |
| 12 | 8.0 | 20 (until 4 weeks post-inoculation) | MRgFUS |
| 13 | 8.0 | 20 (until 4 weeks post-inoculation) | MRgFUS |
| Pericardium Thickness (mm) | Pig #3 | Pig #4 | Pig #6 | Pig #7 | Pig #8 | Pig #9 | Pig #10 | Pig #13 | Average | p-Value |
|---|---|---|---|---|---|---|---|---|---|---|
| Baseline | 3.17 | 3.00 | 2.54 | 2.88 | 3.00 | 3.90 | 3.35 | 3.50 | 3.17 | - |
| 4 weeks | 3.24 | 3.93 | 2.34 | 3.14 | 3.65 | 6.29 | 5.73 | 5.19 | 4.19 | 0.0255 |
| 8 weeks | 3.20 | 5.11 | 5.51 | 4.76 | 4.80 | 7.42 | 6.20 | 6.21 | 5.40 | 0.0006 |
| 12 weeks | 5.17 | 5.00 | 7.02 | 5.01 | 8.98 | - | - | - | 6.24 | 0.0153 |
| 16 weeks | 6.21 | 6.06 | - | - | - | - | - | - | 6.14 * | - * |
| Baseline-final % change | 96% | 102% | 176% | 74% | 200% | 90% | 85% | 77% | 94% | - |
| Diaphragm thickness (mm) | ||||||||||
| Baseline | 2.84 | 3.01 | 5.30 | 5.02 | 4.27 | 4.12 | 3.40 | 5.01 | 4.12 | - |
| 4 weeks | 6.00 | 4.00 | 6.13 | 5.89 | 7.43 | 5.72 | 5.04 | 5.93 | 5.77 | 0.0021 |
| 8 weeks | 7.67 | 5.21 | 6.10 | 6.74 | 8.55 | 7.47 | 5.33 | 6.48 | 6.69 | 0.0014 |
| 12 weeks | 7.06 | 4.80 | 7.57 | 7.48 | 9.62 | - | - | - | 7.31 | 0.0087 |
| 16 weeks | 9.52 | 5.85 | - | - | - | - | - | - | 7.68 * | - * |
| Baseline-final % change | 236% | 94% | 43% | 49% | 125% | 81% | 57% | 29% | 86% | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Costa, M.; Fernandes, C.; Eames, M.; Hananel, A.; Mugler, J.P., III; Huaromo, J.; Yang, J.B.; Mata, J. Cytoreductive Surgical Treatment of Pleural Mesothelioma in a Porcine Model Using Magnetic-Resonance-Guided Focused Ultrasound Surgery (MRgFUS) and Radiofrequency Ablation (RFA). Tomography 2022, 8, 2232-2242. https://doi.org/10.3390/tomography8050187
Costa M, Fernandes C, Eames M, Hananel A, Mugler JP III, Huaromo J, Yang JB, Mata J. Cytoreductive Surgical Treatment of Pleural Mesothelioma in a Porcine Model Using Magnetic-Resonance-Guided Focused Ultrasound Surgery (MRgFUS) and Radiofrequency Ablation (RFA). Tomography. 2022; 8(5):2232-2242. https://doi.org/10.3390/tomography8050187
Chicago/Turabian StyleCosta, Marcia, Carolina Fernandes, Matt Eames, Arik Hananel, John P. Mugler, III, Jhosep Huaromo, Jack B. Yang, and Jaime Mata. 2022. "Cytoreductive Surgical Treatment of Pleural Mesothelioma in a Porcine Model Using Magnetic-Resonance-Guided Focused Ultrasound Surgery (MRgFUS) and Radiofrequency Ablation (RFA)" Tomography 8, no. 5: 2232-2242. https://doi.org/10.3390/tomography8050187
APA StyleCosta, M., Fernandes, C., Eames, M., Hananel, A., Mugler, J. P., III, Huaromo, J., Yang, J. B., & Mata, J. (2022). Cytoreductive Surgical Treatment of Pleural Mesothelioma in a Porcine Model Using Magnetic-Resonance-Guided Focused Ultrasound Surgery (MRgFUS) and Radiofrequency Ablation (RFA). Tomography, 8(5), 2232-2242. https://doi.org/10.3390/tomography8050187