Quantification of Left Atrial Size and Function in Cardiac MR in Correlation to Non-Gated MR and Cardiovascular Risk Factors in Subjects without Cardiovascular Disease: A Population-Based Cohort Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population and Design
- -
- History of cardiovascular disease (myocardial disease, stroke and revascularization therapy);
- -
- Age over 72 years;
- -
- A non-MRI-suitable implant device (cardiac pacemaker or implantable defibrillator, cerebral aneurysm clip, neural stimulator, any type of ear implant, an ocular foreign body, or any implanted device);
- -
- Breast-feeding;
- -
- Claustrophobia;
- -
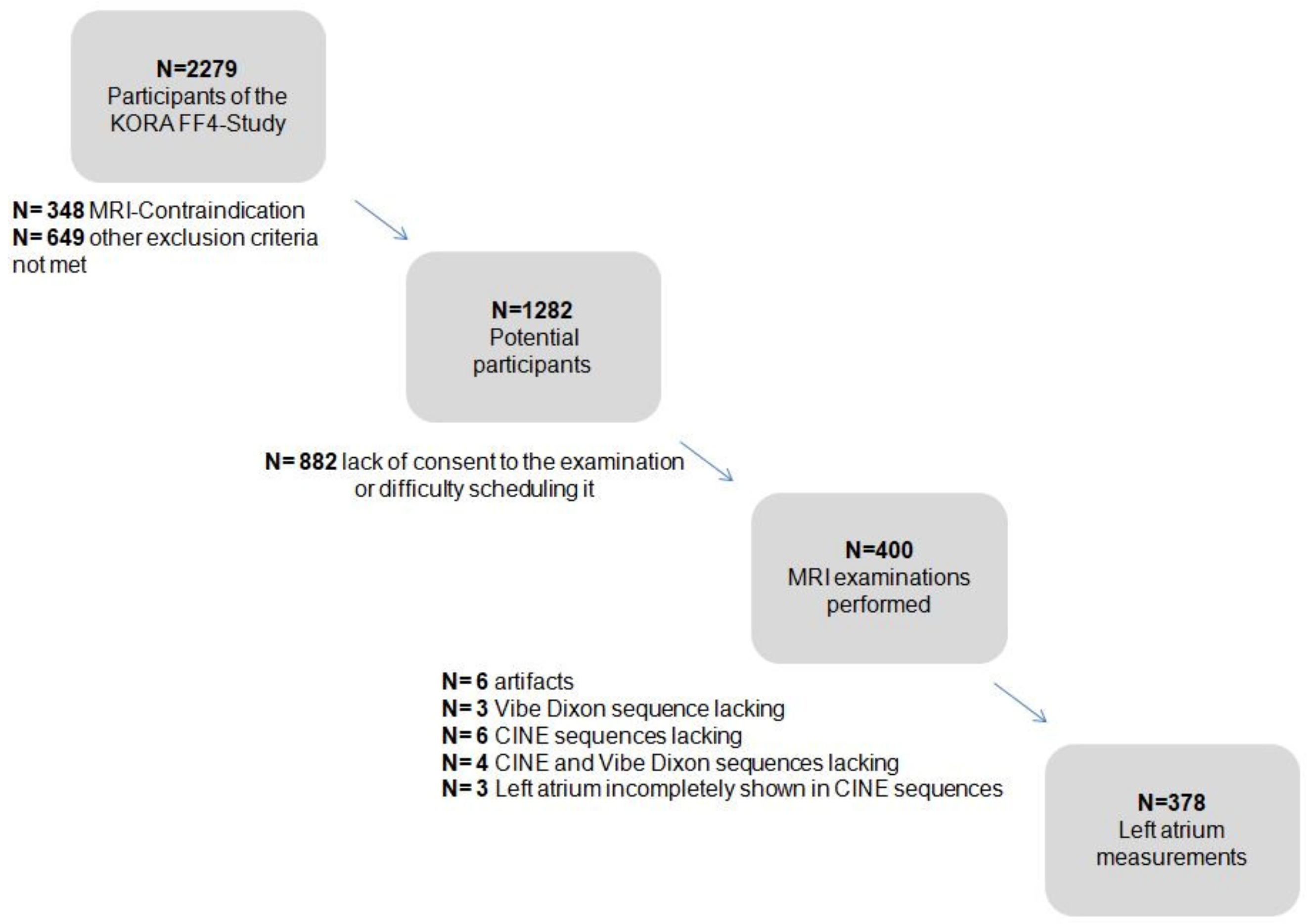
- A known allergy to gadolinium compounds or renal insufficiency. A detailed flowchart is presented in Appendix A Figure A1.
2.2. Covariates
2.3. Magnetic Resonance Imaging
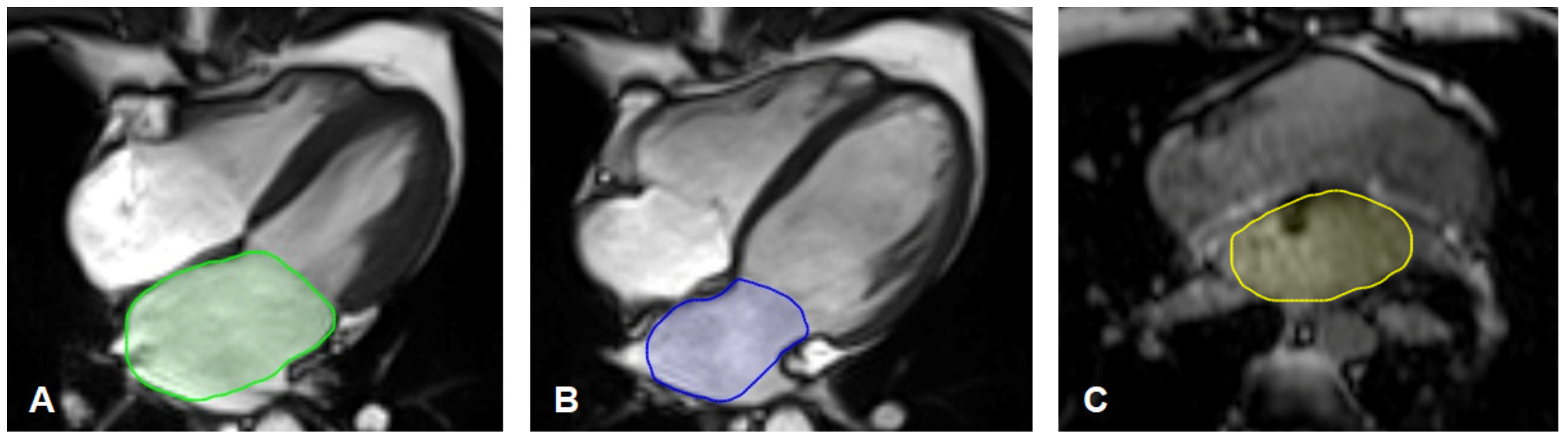
2.4. MR-Image Analysis for Left Atrium Size
2.5. Statistical Analysis
3. Results
3.1. Study Population
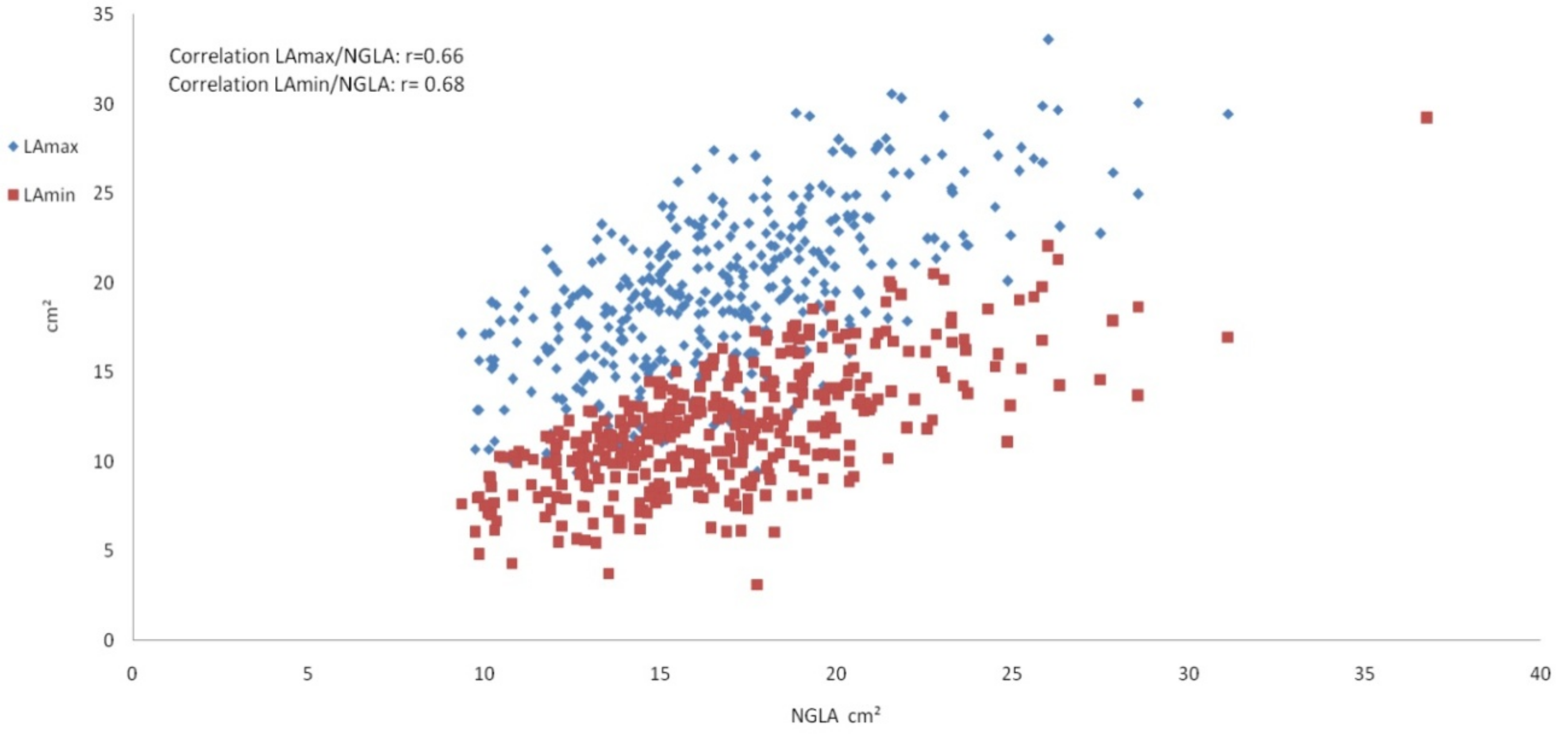
3.2. Left Atrium Measurements
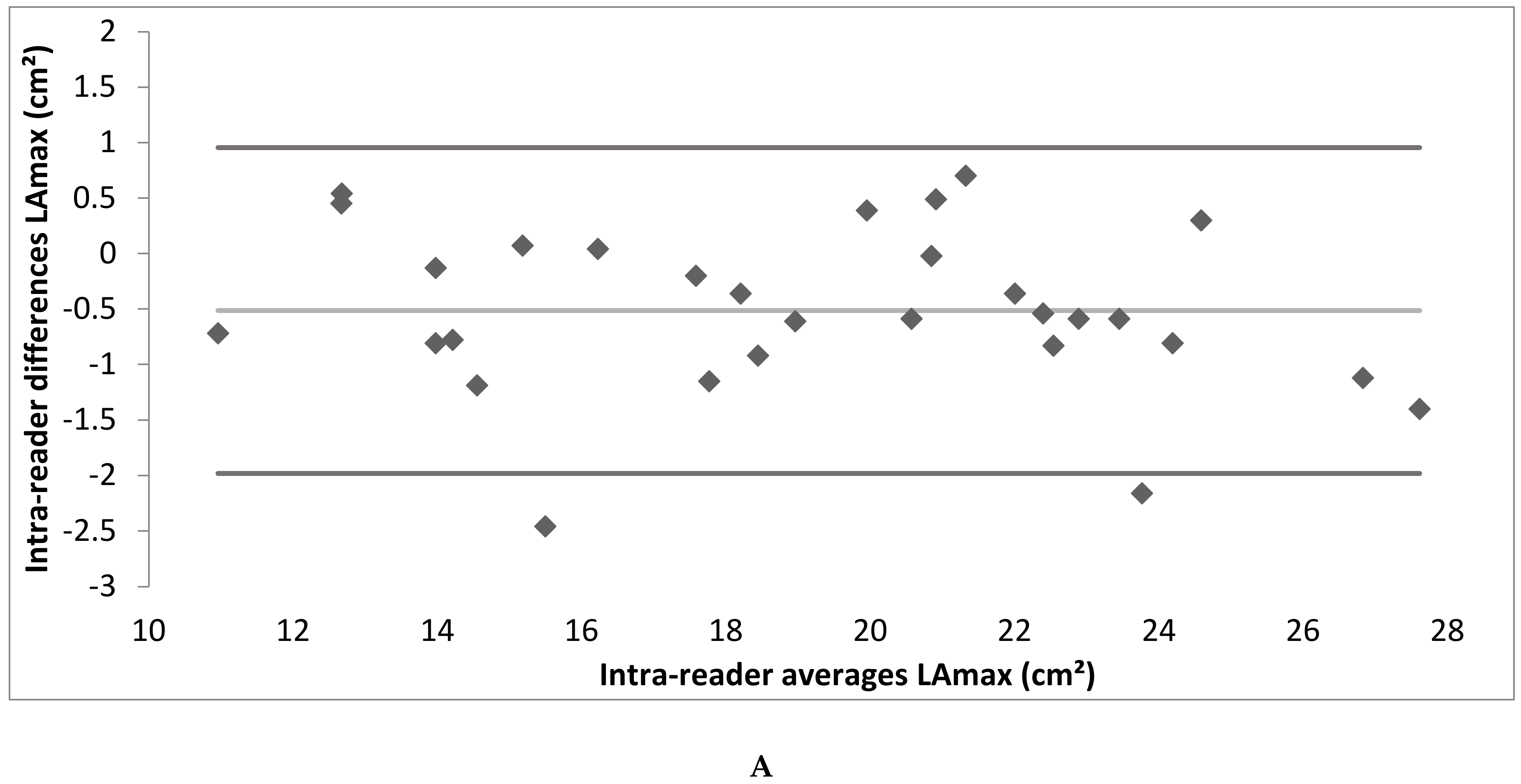
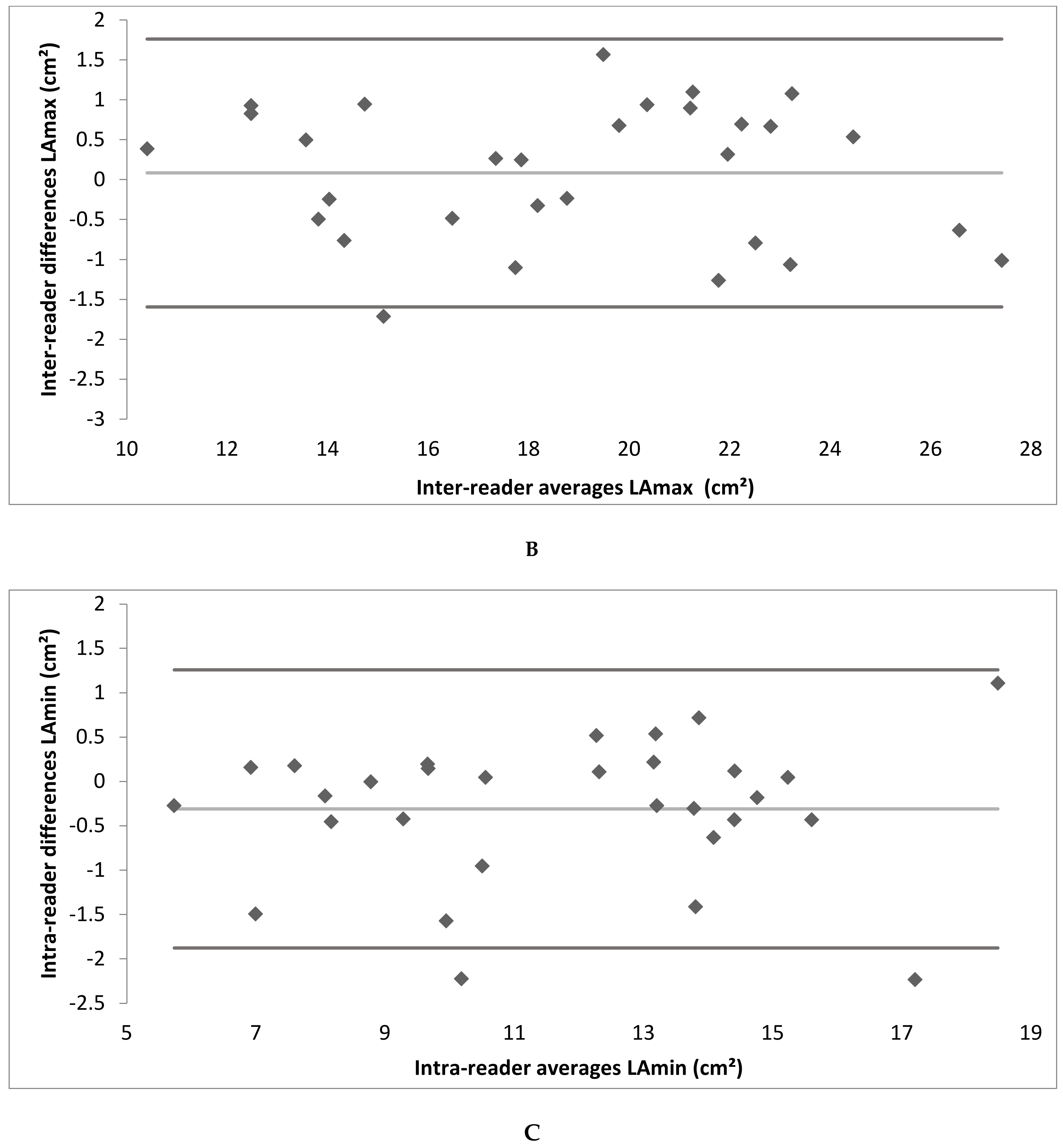
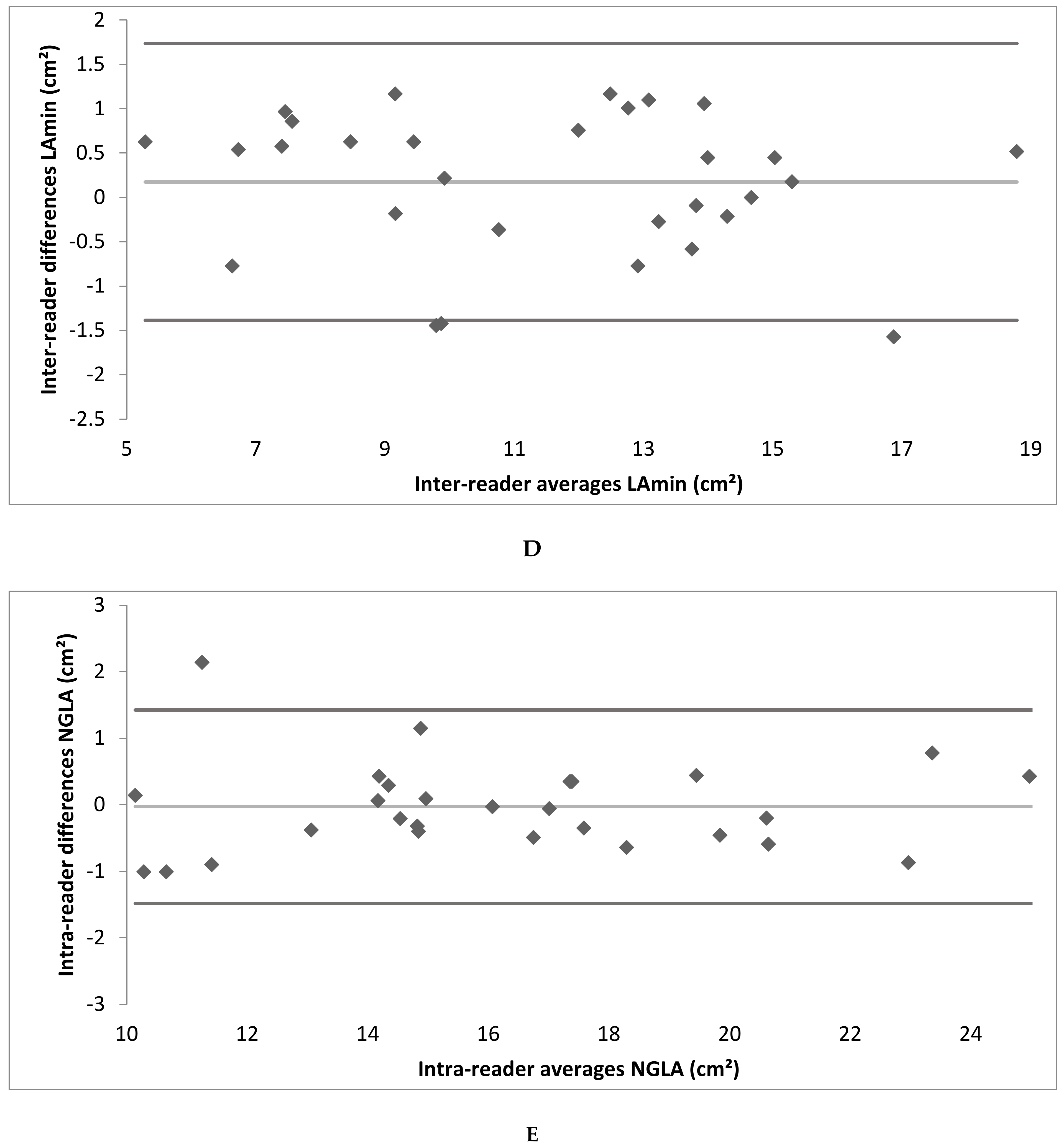
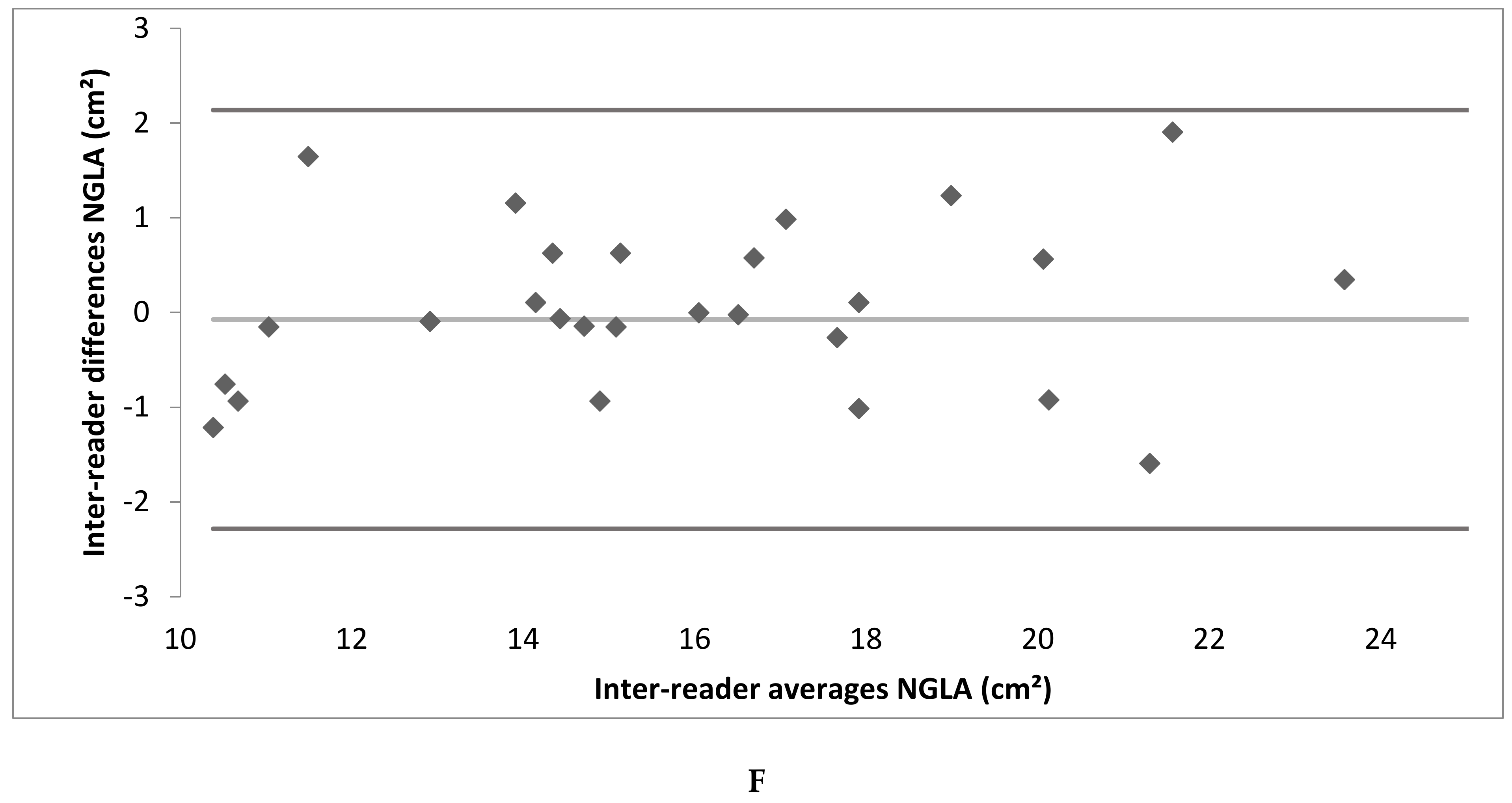
3.3. Intra- and Inter-Reader Reliability
3.4. Left Atrium Size in Association with Demographic Data and Cardiovascular Risk Factors
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Tsang, T.S.M.; Barnes, M.E.; Bailey, K.R.; Leibson, C.L.; Montgomery, S.C.; Takemoto, Y.; Diamond, P.M.; Marra, M.A.; Gersh, B.J.; Wiebers, D.O.; et al. Left Atrial Volume: Important Risk Marker of Incident Atrial Fibrillation in 1655 Older Men and Women. Mayo Clin. Proc. 2001, 76, 467–475. [Google Scholar] [CrossRef]
- Martin, O.; Kaniz, F.; Fatema, Q.; Ahmed, A.-S.; Barnes Marion, E.; Bailey Kent, R.; Gersh Bernard, J.; Tsang Teresa, S.M.; Zehr Kenton, J.; Seward James, B. Left Atrial Volume Predicts the Risk of Atrial Fibrillation After Cardiac Surgery. J. Am. Coll. Cardiol. 2006, 48, 779–786. [Google Scholar] [CrossRef]
- Thomas, L.; Abhayaratna, W.P. Left Atrial Reverse Remodeling: Mechanisms, Evaluation, and Clinical Significance. JACC Cardiovasc. Imaging 2017, 10, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Wojciech, K.; Makoto, S.; Gerry, K.; Kazuaki, N.; Nick, L.; Michael, G.; Marwick Thomas, H. Incremental Value of Left Atrial Structural and Functional Characteristics for Prediction of Atrial Fibrillation in Patients Receiving Cardiac Pacing. Circ. Cardiovasc. Imaging 2015, 8, e002942. [Google Scholar] [CrossRef]
- Rossi, A.; Cicoira, M.; Florea, V.G.; Golia, G.; Florea, N.D.; Khan, A.A.; Murray, S.T.M.; Nguyen, J.T.; O’Callaghan, P.; Anand, I.S.; et al. Chronic Heart Failure with Preserved Left Ventricular Ejection Fraction: Diagnostic and Prognostic Value of Left Atrial Size. Int. J. Cardiol. 2006, 110, 386–392. [Google Scholar] [CrossRef] [PubMed]
- Rossi, A.; Cicoira, M.; Zanolla, L.; Sandrini, R.; Golia, G.; Zardini, P.; Enriquez-Sarano, M. Determinants and Prognostic Value of Left Atrial Volume in Patients with Dilated Cardiomyopathy. J. Am. Coll. Cardiol. 2002, 40, 1425–1430. [Google Scholar] [CrossRef]
- Melenovsky, V.; Borlaug, B.A.; Rosen, B.; Hay, I.; Ferruci, L.; Morell, C.H.; Lakatta, E.G.; Najjar, S.S.; Kass, D.A. Cardiovascular Features of Heart Failure With Preserved Ejection Fraction Versus Nonfailing Hypertensive Left Ventricular Hypertrophy in the Urban Baltimore Community: The Role of Atrial Remodeling/Dysfunction. J. Am. Coll. Cardiol. 2007, 49, 198–207. [Google Scholar] [CrossRef]
- Pellicori, P.; Zhang, J.; Lukaschuk, E.; Joseph, A.C.; Bourantas, C.V.; Loh, H.; Bragadeesh, T.; Clark, A.L.; Cleland, J.G.F. Left Atrial Function Measured by Cardiac Magnetic Resonance Imaging in Patients with Heart Failure: Clinical Associations and Prognostic Value. Eur. Heart J. 2015, 36, 733–742. [Google Scholar] [CrossRef]
- Dini, F.L.; Cortigiani, L.; Baldini, U.; Boni, A.; Nuti, R.; Barsotti, L.; Micheli, G. Prognostic Value of Left Atrial Enlargement in Patients with Idiopathic Dilated Cardiomyopathy and Ischemic Cardiomyopathy. Am. J. Cardiol. 2002, 89, 518–523. [Google Scholar] [CrossRef]
- Møller Jacob, E.; Hillis Graham, S.; Oh Jae, K.; Seward James, B.; Reeder Guy, S.; Wright, R.S.; Park Seung, W.; Bailey Kent, R.; Pellikka Patricia, A. Left Atrial Volume. Circulation 2003, 107, 2207–2212. [Google Scholar] [CrossRef] [Green Version]
- Welles, C.C.; Ku, I.A.; Kwan, D.M.; Whooley, M.A.; Schiller, N.B.; Turakhia, M.P. Left Atrial Function Predicts Heart Failure Hospitalization in Subjects With Preserved Ejection Fraction and Coronary Heart Disease: Longitudinal Data From the Heart and Soul Study. J. Am. Coll. Cardiol. 2012, 59, 673–680. [Google Scholar] [CrossRef]
- Kizer, J.R.; Bella, J.N.; Palmieri, V.; Liu, J.E.; Best, L.G.; Lee, E.T.; Roman, M.J.; Devereux, R.B. Left Atrial Diameter as an Independent Predictor of First Clinical Cardiovascular Events in Middle-Aged and Elderly Adults: The Strong Heart Study (SHS). Am. Heartj. 2006, 151, 412–418. [Google Scholar] [CrossRef]
- Nistri, S.; Olivotto, I.; Betocchi, S.; Losi, M.A.; Valsecchi, G.; Pinamonti, B.; Conte, M.R.; Casazza, F.; Galderisi, M.; Maron, B.J.; et al. Prognostic Significance of Left Atrial Size in Patients with Hypertrophic Cardiomyopathy (from the Italian Registry for Hypertrophic Cardiomyopathy). Am. J. Cardiol. 2006, 98, 960–965. [Google Scholar] [CrossRef] [PubMed]
- Maron, B.J.; Haas, T.S.; Maron, M.S.; Lesser, J.R.; Browning, J.A.; Chan, R.H.; Olivotto, I.; Garberich, R.F.; Schwartz, R.S. Left Atrial Remodeling in Hypertrophic Cardiomyopathy and Susceptibility Markers for Atrial Fibrillation Identified by Cardiovascular Magnetic Resonance. Am. J. Cardiol. 2014, 113, 1394–1400. [Google Scholar] [CrossRef]
- Benjamin Emelia, J.; D’Agostino Ralph, B.; Belanger Albert, J.; Wolf Philip, A. Levy Daniel Left Atrial Size and the Risk of Stroke and Death. Circulation 1995, 92, 835–841. [Google Scholar] [CrossRef] [PubMed]
- Laukkanen, J.A.; Kurl, S.; Eränen, J.; Huttunen, M.; Salonen, J.T. Left Atrium Size and the Risk of Cardiovascular Death in Middle-Aged Men. Arch. Intern. Med. 2005, 165, 1788–1793. [Google Scholar] [CrossRef]
- Cereda, A.F.; De Luca, F.; Lanzone, A.M.; Cottini, M.; Pastori, L.; Sangiorgi, G. Case Report and Systematic Review of Iatrogenic Left Atrial Dissection in Different Cardiovascular Specialties: A Common Treatment for an Uncommon Complication? Catheter. Cardiovasc. Interv. 2020, 95, E30–E36. [Google Scholar] [CrossRef]
- Raisi-Estabragh, Z.; McCracken, C.; Condurache, D.; Aung, N.; Vargas, J.D.; Naderi, H.; Munroe, P.B.; Neubauer, S.; Harvey, N.C.; Petersen, S.E. Left Atrial Structure and Function Are Associated with Cardiovascular Outcomes Independent of Left Ventricular Measures: A UK Biobank CMR Study. Eur. Heartj. Cardiovasc. Imaging 2022, 23, 1191–1200. [Google Scholar] [CrossRef]
- Tops, L.F.; van der Wall, E.E.; Schalij, M.J.; Bax, J.J. Multi-modality Imaging to Assess Left Atrial Size, Anatomy and Function. Heart 2007, 93, 1461–1470. [Google Scholar] [CrossRef]
- Soufer, A.; Peters, D.C.; Henry, M.L.; Baldassarre, L.A. Advanced Imaging of the Left Atrium with Cardiac Magnetic Resonance: A Review of Current and Emerging Methods and Clinical Applications. Curr. Radiol. Rep. 2018, 6, 44. [Google Scholar] [CrossRef]
- Kramer, C.M.; Barkhausen, J.; Bucciarelli-Ducci, C.; Flamm, S.D.; Kim, R.J.; Nagel, E. Standardized Cardiovascular Magnetic Resonance Imaging (CMR) Protocols: 2020 Update. J. Cardiovasc. Magn. Reson. 2020, 22, 17. [Google Scholar] [CrossRef] [PubMed]
- Maceira, A.M.; Cosin-Sales, J.; Prasad, S.K.; Pennell, D.J. Characterization of Left and Right Atrial Function in Healthy Volunteers by Cardiovascular Magnetic Resonance. J. Cardiovasc. Magn. Reson. 2016, 18, 64. [Google Scholar] [CrossRef]
- Mahabadi, A.A.; Lehmann, N.; Sonneck, N.C.; Kälsch, H.; Bauer, M.; Kara, K.; Geisel, M.H.; Moebus, S.; Jöckel, K.-H.; Erbel, R.; et al. Left Atrial Size Quantification Using Non-Contrast-Enhanced Cardiac Computed Tomography—Association with Cardiovascular Risk Factors and Gender-Specific Distribution in the General Population: The Heinz Nixdorf Recall Study. Acta Radiol. 2014, 55, 917–925. [Google Scholar] [CrossRef] [PubMed]
- Bamberg, F.; Hetterich, H.; Rospleszcz, S.; Lorbeer, R.; Auweter, S.D.; Schlett, C.L.; Schafnitzel, A.; Bayerl, C.; Schindler, A.; Saam, T.; et al. Subclinical Disease Burden as Assessed by Whole-Body MRI in Subjects With Prediabetes, Subjects With Diabetes, and Normal Control Subjects From the General Population: The KORA-MRI Study. Diabetes 2017, 66, 158–169. [Google Scholar] [CrossRef]
- Lorbeer, R.; Rospleszcz, S.; Schlett, C.L.; Rado, S.D.; Thorand, B.; Meisinger, C.; Rathmann, W.; Heier, M.; Vasan, R.S.; Bamberg, F.; et al. Association of Antecedent Cardiovascular Risk Factor Levels and Trajectories with Cardiovascular Magnetic Resonance-Derived Cardiac Function and Structure. J. Cardiovasc. Magn. Reson. 2021, 23, 2. [Google Scholar] [CrossRef]
- Holle, R.; Happich, M.; Löwel, H.; Wichmann, H.E.; The MONICA/KORA Study Group. KORA–A Research Platform for Population Based Health Research. Gesundheitswesen 2005, 67, 19–25. [Google Scholar] [CrossRef]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; de Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the Management of Arterial Hypertension: The Task Force for the Management of Arterial Hypertension of the European Society of Cardiology (ESC) and the European Society of Hypertension (ESH). Eur. Heartj. 2018, 39, 3021–3104. [Google Scholar] [CrossRef]
- World Health Organization; International Diabetes Federation. Definition and Diagnosis of Diabetes Mellitus and Intermediate Hyperglycaemia: Report of a WHO/IDF Consultation; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- Mahabadi, A.A.; Samy, B.; Seneviratne, S.K.; Toepker, M.H.; Bamberg, F.; Hoffmann, U.; Truong, Q.A. Quantitative Assessment of Left Atrial Volume by Electrocardiographic-Gated Contrast-Enhanced Multidetector Computed Tomography. J. Cardiovasc. Comput. Tomogr. 2009, 3, 80–87. [Google Scholar] [CrossRef]
- Maceira, A.M.; Cosín-Sales, J.; Roughton, M.; Prasad, S.K.; Pennell, D.J. Reference Left Atrial Dimensions and Volumes by Steady State Free Precession Cardiovascular Magnetic Resonance. J. Cardiovasc. Magn. Reson. 2010, 12, 65. [Google Scholar] [CrossRef]
- Kawel-Boehm, N.; Hetzel, S.J.; Ambale-Venkatesh, B.; Captur, G.; Francois, C.J.; Jerosch-Herold, M.; Salerno, M.; Teague, S.D.; Valsangiacomo-Buechel, E.; van der Geest, R.J.; et al. Reference Ranges (“Normal Values”) for Cardiovascular Magnetic Resonance (CMR) in Adults and Children: 2020 Update. J. Cardiovasc. Magn. Reson. 2020, 22, 87. [Google Scholar] [CrossRef]
- Petersen, S.E.; Aung, N.; Sanghvi, M.M.; Zemrak, F.; Fung, K.; Paiva, J.M.; Francis, J.M.; Khanji, M.Y.; Lukaschuk, E.; Lee, A.M.; et al. Reference Ranges for Cardiac Structure and Function Using Cardiovascular Magnetic Resonance (CMR) in Caucasians from the UK Biobank Population Cohort. J. Cardiovasc. Magn. Reson. 2017, 19, 18. [Google Scholar] [CrossRef] [PubMed]
- Mohammadali, H.; Sanaz, S.; Bharath, A.V.; Anders, O.; Helle-Valle Thomas, M.; Mytra, Z.; Almeida Andre, L.C.; Eui-Young, C.; Colin, W.; Alvaro, A.; et al. Cardiac Magnetic Resonance–Measured Left Atrial Volume and Function and Incident Atrial Fibrillation. Circ. Cardiovasc. Imaging 2016, 9, e004299. [Google Scholar] [CrossRef]
- Truong, Q.A.; Bamberg, F.; Mahabadi, A.A.; Toepker, M.; Lee, H.; Rogers, I.S.; Seneviratne, S.K.; Schlett, C.L.; Brady, T.J.; Nagurney, J.T.; et al. Left Atrial Volume and Index by Multi-Detector Computed Tomography: Comprehensive Analysis from Predictors of Enlargement to Predictive Value for Acute Coronary Syndrome (ROMICAT Study). Int. J. Cardiol. 2011, 146, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Pritchett Allison, M.; Jacobsen Steven, J.; Mahoney Douglas, W.; Rodeheffer Richard, J.; Bailey Kent, R.; Redfield Margaret, M. Left Atrial Volume as an Index Ofleft Atrial Size: A Population-Based Study. J. Am. Coll. Cardiol. 2003, 41, 1036–1043. [Google Scholar] [CrossRef]
- Vasan Ramachandran, S.; Larson Martin, G.; Daniel, L.; Evans Jane, C.; Benjamin Emelia, J. Distribution and Categorization of Echocardiographic Measurements in Relation to Reference Limits. Circulation 1997, 96, 1863–1873. [Google Scholar] [CrossRef]
- Lang, R.M.; Bierig, M.; Devereux, R.B.; Flachskampf, F.A.; Foster, E.; Pellikka, P.A.; Picard, M.H.; Roman, M.J.; Seward, J.; Shanewise, J.S.; et al. Recommendations for Chamber Quantification: A Report from the American Society of Echocardiography’s Guidelines and Standards Committee and the Chamber Quantification Writing Group, Developed in Conjunction with the European Association of Echocardiography, a Branch of the European Society of Cardiology. J. Am. Soc. Echocardiogr. 2005, 18, 1440–1463. [Google Scholar] [CrossRef]
- Wang, Y.; Gutman, J.M.; Heilbron, D.; Wahr, D.; Schiller, N.B. Atrial Volume in a Normal Adult Population by Two-Dimensional Echocardiography. Chest 1984, 86, 595–601. [Google Scholar] [CrossRef]
- Knutsen, K.M.; Stugaard, M.; Michelsen, S.; Otterstad, J.E. M-Mode Echocardiographic Findings in Apparently Healthy, Non-Athletic Norwegians Aged 20−70 Years. Influence of Age, Sex and Body Surface Area. J. Intern. Med. 1989, 225, 111–115. [Google Scholar] [CrossRef]
- Filip, Z.; Bharath, A.-V.; Gabriella, C.; Jonathan, C.; Ela, C.; Mohammadali, H.; Saman, N.; Mohiddin Saidi, A.; Moon James, C.; Petersen Steffen, E.; et al. Left Atrial Structure in Relationship to Age, Sex, Ethnicity, and Cardiovascular Risk Factors. Circ. Cardiovasc. Imaging 2017, 10, e005379. [Google Scholar] [CrossRef]
- Liza, T.; Kate, L.; Anita, B.; Leung Dominic, Y.C.; Schiller Nelson, B.; Ross David, L. Compensatory Changes in Atrial Volumes with Normal Aging: Is Atrial Enlargement Inevitable? J. Am. Coll. Cardiol. 2002, 40, 1630–1635. [Google Scholar] [CrossRef] [Green Version]
- Fredgart, M.H.; Lindholt, J.S.; Brandes, A.; Steffensen, F.H.; Frost, L.; Lambrechtsen, J.; Karon, M.; Busk, M.; Urbonaviciene, G.; Egstrup, K.; et al. Association of Left Atrial Size Measured by Non-Contrast Computed Tomography with Cardiovascular Risk Factors—The Danish Cardiovascular Screening Trial (DANCAVAS). Diagnostics 2022, 12, 244. [Google Scholar] [CrossRef]
- Singh, A.; Carvalho Singulane, C.; Miyoshi, T.; Prado, A.D.; Addetia, K.; Bellino, M.; Daimon, M.; Gutierrez Fajardo, P.; Kasliwal, R.R.; Kirkpatrick, J.N.; et al. Normal Values of Left Atrial Size and Function and the Impact of Age: Results of the World Alliance Societies of Echocardiography Study. J. Am. Soc. Echocardiogr. Off. Publ. Am. Soc. Echocardiogr. 2022, 35, 154–164.e3. [Google Scholar] [CrossRef]
- Boyd, A.C.; Schiller, N.B.; Leung, D.; Ross, D.L.; Thomas, L. Atrial Dilation and Altered Function Are Mediated by Age and Diastolic Function But Not Before the Eighth Decade. Jacc Cardiovasc. Imaging 2011, 4, 234–242. [Google Scholar] [CrossRef]
- D’Andrea, A.; Riegler, L.; Rucco, M.A.; Cocchia, R.; Scarafile, R.; Salerno, G.; Martone, F.; Vriz, O.; Caso, P.; Calabrò, R.; et al. Left Atrial Volume Index in Healthy Subjects: Clinical and Echocardiographic Correlates. Echocardiography 2013, 30, 1001–1007. [Google Scholar] [CrossRef]
- Nikitin, N.P.; Witte, K.K.A.; Thackray, S.D.R.; Goodge, L.J.; Clark, A.L.; Cleland, J.G.F. Effect of Age and Sex on Left Atrial Morphology and Function. Eur. J. Echocardiogr. 2003, 4, 36–42. [Google Scholar] [CrossRef]
- Spencer, K.T.; Mor-Avi, V.; Gorcsan, J.; DeMaria, A.N.; Kimball, T.R.; Monaghan, M.J.; Perez, J.E.; Weinert, L.; Bednarz, J.; Edelman, K.; et al. Effects of Aging on Left Atrial Reservoir, Conduit, and Booster Pump Function: A Multi-Institution Acoustic Quantification Study. Heart 2001, 85, 272–277. [Google Scholar] [CrossRef]
- Triposkiadis, F.; Tentolouris, K.; Androulakis, A.; Trikas, A.; Toutouzas, K.; Kyriakidis, M.; Gialafos, J.; Toutouzas, P. Left Atrial Mechanical Function in the Healthy Elderly: New Insights from a Combined Assessment of Changes in Atrial Volume and Transmitral Flow Velocity. J. Am. Soc. Echocardiogr. 1995, 8, 801–809. [Google Scholar] [CrossRef]
- Thomas, L.; Levett, K.; Boyd, A.; Leung, D.Y.C.; Schiller, N.B.; Ross, D.L. Changes in Regional Left Atrial Function with Aging: Evaluation by Doppler Tissue Imaging. Eur. Heartj. Cardiovasc. Imaging 2003, 4, 92–100. [Google Scholar] [CrossRef]
- Gupta, S.; Matulevicius, S.A.; Ayers, C.R.; Berry, J.D.; Patel, P.C.; Markham, D.W.; Levine, B.D.; Chin, K.M.; de Lemos, J.A.; Peshock, R.M.; et al. Left Atrial Structure and Function and Clinical Outcomes in the General Population. Eur. Heartj. 2013, 34, 278–285. [Google Scholar] [CrossRef]
- Abhayaratna, W.P.; Seward, J.B.; Appleton, C.P.; Douglas, P.S.; Oh, J.K.; Tajik, A.J.; Tsang, T.S.M. Left Atrial Size: Physiologic Determinants and Clinical Applications. J. Am. Coll. Cardiol. 2006, 47, 2357–2363. [Google Scholar] [CrossRef] [Green Version]
- Suzanne, E.; Ross David, L.; Liza, T. Impact of Mild Hypertension on Left Atrial Size and Function. Circ. Cardiovasc. Imaging 2009, 2, 93–99. [Google Scholar] [CrossRef]
- Tymon, P.; Claes, H.; Johan, W.; Johan, L.; Alexander John, H.; Marco, A.; Cetin, E.; Shinya, G.; Sigrun, H.; Kurt, H.; et al. Dyslipidemia and Risk of Cardiovascular Events in Patients with Atrial Fibrillation Treated with Oral Anticoagulation Therapy: Insights from the ARISTOTLE (Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation) Trial. J. Am. Heart Assoc. 2018, 7, e007444. [Google Scholar] [CrossRef]
- Berliner, J.A.; Navab, M.; Fogelman, A.M.; Frank, J.S.; Demer, L.L.; Edwards, P.A.; Watson, A.D.; Lusis, A.J. Atherosclerosis: Basic Mechanisms. Oxidation, Inflammation, and Genetics. Circulation 1995, 91, 2488–2496. [Google Scholar] [CrossRef] [PubMed]
- McManus David, D.; Vanessa, X.; Sullivan Lisa, M.; Justin, Z.; Jayashri, A.; Larson Martin, G.; Benjamin Emelia, J.; Vasan Ramachandran, S. Longitudinal Tracking of Left Atrial Diameter Over the Adult Life Course: Clinical Correlates in the Community. Circulation 2010, 121, 667–674. [Google Scholar] [CrossRef] [Green Version]
| n | 378 |
|---|---|
| Age (years) | 56.3 ± 9.2 |
| Male sex | 218 (57.7%) |
| BMI (kg/m2) | 28.1 ± 4.9 |
| Body surface area (m2) | 1.95 ± 0.22 |
| Systolic blood pressure (mmHg) | 120.8 ± 16.8 |
| Diastolic blood pressure (mmHg) | 75.3 ± 10.1 |
| Hypertension | 131 (34.7%) |
| Diabetes status | |
| Normoglycemia | 228 (60.3%) |
| Prediabetes | 99 (26.2%) |
| Diabetes | 51 (13.5%) |
| Smoking status | |
| Smoker | 136 (36%) |
| Ex-smoker | 163 (43.1%) |
| Never-smoker | 79 (20.9%) |
| Total cholesterol (mg/dL) | 218.5 ± 36.8 |
| HDL (mg/dL) | 61.8 ± 17.7 |
| LDL (mg/dL) | 140.1 ± 33.3 |
| Triglycerides (mg/dL) | 133.6 ± 86.2 |
| Lipid-lowering medications | 41 (10.9%) |
| Antihypertensive medications | 98 (25.9%) |
| Left ventricle mass (g) * | 141.0 ± 35.3 |
| Left ventricle end-diastolic volume (mL) * | 129.6 ± 32.8 |
| Left ventricle end-systolic volume (mL) * | 41.0 ± 18.2 |
| Left ventricle stroke volume (mL) * | 88.7 ± 20.4 |
| Left ventricle ejection fraction (%) * | 69.2 ± 7.8 |
| Maximal left atrium area (cm2) | 19.6 ± 4.5 |
| Minimal left atrium area (cm2) | 11.9 ± 3.5 |
| Non-gated left atrium area (cm2) | 16.8 ± 4 |
| Left atrium area fraction | 40 ± 9 % |
| Inter-Reader Reliability | Intra-Reader Reliability | |||||
|---|---|---|---|---|---|---|
| ICC | Relative Difference | Mean Difference | ICC | Relative Difference | Mean Difference | |
| LAmax | 0.99 | 0.6% | 0.09 | 0.99 | 2.7% | −0.51 |
| LAmin | 0.99 | 2.0% | 0.18 | 0.98 | 3.0% | −0.31 |
| NGLA | 0.99 | 0.3% | −0.07 | 0.99 | 0.3% | −0.03 |
| LAmax | LAmin | LAaf(%) | NGLA | |||||
|---|---|---|---|---|---|---|---|---|
| β (95% CI) | p | β (95% CI) | p | β (95% CI) | p | β (95% CI) | p | |
| Body surface area (m2) | 0.85 (0.40; 1.30) | <0.001 | 0.82 (0.47; 1.16) | <0.001 | −1.72 (−2.63; −0.81) | <0.001 | 1.59 (1.21; 1.96) | <0.001 |
| Male sex | 0.43 (−0.5; 1.35) | 0.37 | 0.9 (0.19; 1.6) | 0.013 | −3.74 (−5.58; −1.9) | <0.001 | 2.28 (1.5; 3.07) | <0.001 |
| Age (years) | −0.18 (−0.64; 0.28) | 0.44 | 0.35 (0.01; 0.7) | 0.047 | −2.20 (−3.10; −1.30) | <0.001 | 0.40 (0.00; 0.80) | 0.05 |
| Prediabetes | −0.14 (−1.21; 0.94) | 0.80 | 0.56 (−0.26; 1.38) | 0.18 | −3.42 (−5.53; −1.3) | 0.002 | 0.57 (−0.38; 1.51) | 0.24 |
| Diabetes | 0.06 (−1.32; 1.44) | 0.93 | 1.01 (−0.04; 2.06) | 0.06 | −5.14 (−7.87; −2.42) | <0.001 | 1.41 (0.2; 2.62) | 0.023 |
| Hypertension | 1.28 (0.32; 2.23) | 0.009 | 1.56 (0.84; 2.28) | <0.001 | −3.88 (−5.79; −1.97) | <0.001 | 1.78 (0.95; 2.61) | <0.001 |
| Ex-smoker | 0.56 (−0.48; 1.59) | 0.29 | 0.6 (−0.19; 1.39) | 0.14 | −1.28 (−3.37; 0.8) | 0.23 | 0.41 (−0.51; 1.32) | 0.38 |
| Smoker | −0.12 (−1.38; 1.14) | 0.86 | −0.25 (−1.21; 0.72) | 0.62 | 1.23 (−1.31; 3.77) | 0.34 | −0.5 (−1.61; 0.61) | 0.38 |
| Total cholesterol (mg/dL) | −0.39 (−0.84; 0.07) | 0.10 | −0.21 (−0.56; 0.14) | 0.23 | −0.06 (−0.99; 0.87) | 0.90 | −0.30 (−0.70; 0.11) | 0.15 |
| HDL (mg/dL) | 0.20 (−0.26; 0.66) | 0.40 | −0.05 (−0.4; 0.3) | 0.79 | 1.16 (0.24; 2.08) | 0.014 | −0.49 (−0.89; −0.09) | 0.017 |
| LDL (mg/dL) | 0.37 (−0.49; 1.23) | 0.40 | −0.09 (−0.75; 0.57) | 0.79 | 2.17 (0.44; 3.9) | 0.014 | −0.92 (−1.67; −0.17) | 0.017 |
| Triglycerides (mg/dL) | −0.16 (−0.61; 0.3) | 0.51 | 0.10 (−0.25; 0.45) | 0.57 | −1.08 (−2.00; −0.15) | 0.022 | 0.30 (−0.11; 0.70) | 0.15 |
| Lipid-lowering medications | 0.81 (−0.66; 2.28) | 0.28 | 0.92 (−0.20; 2.05) | 0.11 | −2.84 (−5.81; 0.13) | 0.06 | 0.93 (−0.36; 2.23) | 0.16 |
| LAmax | LAmin | LAaf(%) | NGLA | |||||
|---|---|---|---|---|---|---|---|---|
| β (95% CI) | p | β (95% CI) | p | β (95% CI) | p | β (95% CI) | p | |
| Body surface area (m2) | 1.43 (0.76; 2.10) | <0.001 | 1.09 (0.58; 1.59) | <0.001 | −1.01 (−2.33; 0.31) | 0.13 | 1.78 (1.24; 2.33) | <0.001 |
| Male sex | −0.86 (−2.08; 0.36) | 0.17 | −0.23 (−1.15; 0.69) | 0.63 | −1.93 (−4.35; 0.48) | 0.12 | 0.29 (−0.71; 1.3) | 0.56 |
| Age (years) | −0.13 (−0.66; 0.39) | 0.62 | 0.34 (−0.05; 0.74) | 0.09 | −1.93 (−2.97; −0.89) | <0.001 | 0.59 (0.16; 1.02) | 0.008 |
| Prediabetes | −0.86 (−2.02; 0.30) | 0.14 | −0.34 (−1.21; 0.54) | 0.45 | −1.18 (−3.48; 1.11) | 0.31 | −0.88 (−1.83; 0.07) | 0.07 |
| Diabetes | −0.71 (−2.32; 0.90) | 0.39 | −0.10 (−1.32; 1.11) | 0.87 | −1.84 (−5.03; 1.35) | 0.26 | −0.22 (−1.54; 1.11) | 0.75 |
| Hypertension | 1.30 (0.22; 2.37) | 0.018 | 1.07 (0.26; 1.88) | 0.010 | −1.20 (−3.32; 0.93) | 0.27 | 0.94 (0.06; 1.82) | 0.037 |
| Ex-smoker | 0.38 (−0.64; 1.39) | 0.47 | 0.34 (−0.43; 1.11) | 0.39 | −0.45 (−2.47; 1.56) | 0.66 | −0.08 (−0.92; 0.75) | 0.84 |
| Smoker | 0.32 (−0.93; 1.57) | 0.61 | 0.17 (−0.78; 1.11) | 0.73 | 0.72 (−1.75; 3.2) | 0.57 | −0.11 (−1.14; 0.92) | 0.83 |
| Total cholesterol (mg/dL) | −0.20 (−0.73; 0.33) | 0.46 | −0.14 (−0.54; 0.26) | 0.51 | −0.08 (−1.13; 0.97) | 0.88 | −0.10 (−0.53; 0.34) | 0.66 |
| HDL (mg/dL) | 0.79 (0.19; 1.40) | 0.011 | 0.50 (0.04; 0.96) | 0.034 | 0.31 (−0.89; 1.52) | 0.61 | 0.36 (−0.14; 0.86) | 0.16 |
| Triglycerides (mg/dL) | 0.05 (−0.58; 0.68) | 0.88 | 0.05 (−0.43; 0.53) | 0.83 | 0.15 (−1.10; 1.40) | 0.81 | −0.03 (−0.55; 0.49) | 0.91 |
| Lipid-lowering medications | 0.67 (−0.95; 2.28) | 0.42 | 0.31 (−0.91; 1.53) | 0.62 | −0.52 (−3.72; 2.67) | 0.75 | 0.30 (−1.03; 1.62) | 0.66 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kulka, C.; Lorbeer, R.; Askani, E.; Kellner, E.; Reisert, M.; von Krüchten, R.; Rospleszcz, S.; Hasic, D.; Peters, A.; Bamberg, F.; et al. Quantification of Left Atrial Size and Function in Cardiac MR in Correlation to Non-Gated MR and Cardiovascular Risk Factors in Subjects without Cardiovascular Disease: A Population-Based Cohort Study. Tomography 2022, 8, 2202-2217. https://doi.org/10.3390/tomography8050185
Kulka C, Lorbeer R, Askani E, Kellner E, Reisert M, von Krüchten R, Rospleszcz S, Hasic D, Peters A, Bamberg F, et al. Quantification of Left Atrial Size and Function in Cardiac MR in Correlation to Non-Gated MR and Cardiovascular Risk Factors in Subjects without Cardiovascular Disease: A Population-Based Cohort Study. Tomography. 2022; 8(5):2202-2217. https://doi.org/10.3390/tomography8050185
Chicago/Turabian StyleKulka, Charlotte, Roberto Lorbeer, Esther Askani, Elias Kellner, Marco Reisert, Ricarda von Krüchten, Susanne Rospleszcz, Dunja Hasic, Annette Peters, Fabian Bamberg, and et al. 2022. "Quantification of Left Atrial Size and Function in Cardiac MR in Correlation to Non-Gated MR and Cardiovascular Risk Factors in Subjects without Cardiovascular Disease: A Population-Based Cohort Study" Tomography 8, no. 5: 2202-2217. https://doi.org/10.3390/tomography8050185
APA StyleKulka, C., Lorbeer, R., Askani, E., Kellner, E., Reisert, M., von Krüchten, R., Rospleszcz, S., Hasic, D., Peters, A., Bamberg, F., & Schlett, C. L. (2022). Quantification of Left Atrial Size and Function in Cardiac MR in Correlation to Non-Gated MR and Cardiovascular Risk Factors in Subjects without Cardiovascular Disease: A Population-Based Cohort Study. Tomography, 8(5), 2202-2217. https://doi.org/10.3390/tomography8050185