Directed Evolution and Engineering of Gallium-Binding Phage Clones—A Preliminary Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Handling of Phage Display Library Clones
2.2. Site-Directed Mutagenesis Experiments
2.3. Biosorption Experiments
3. Results and Discussion
3.1. Experimental Context
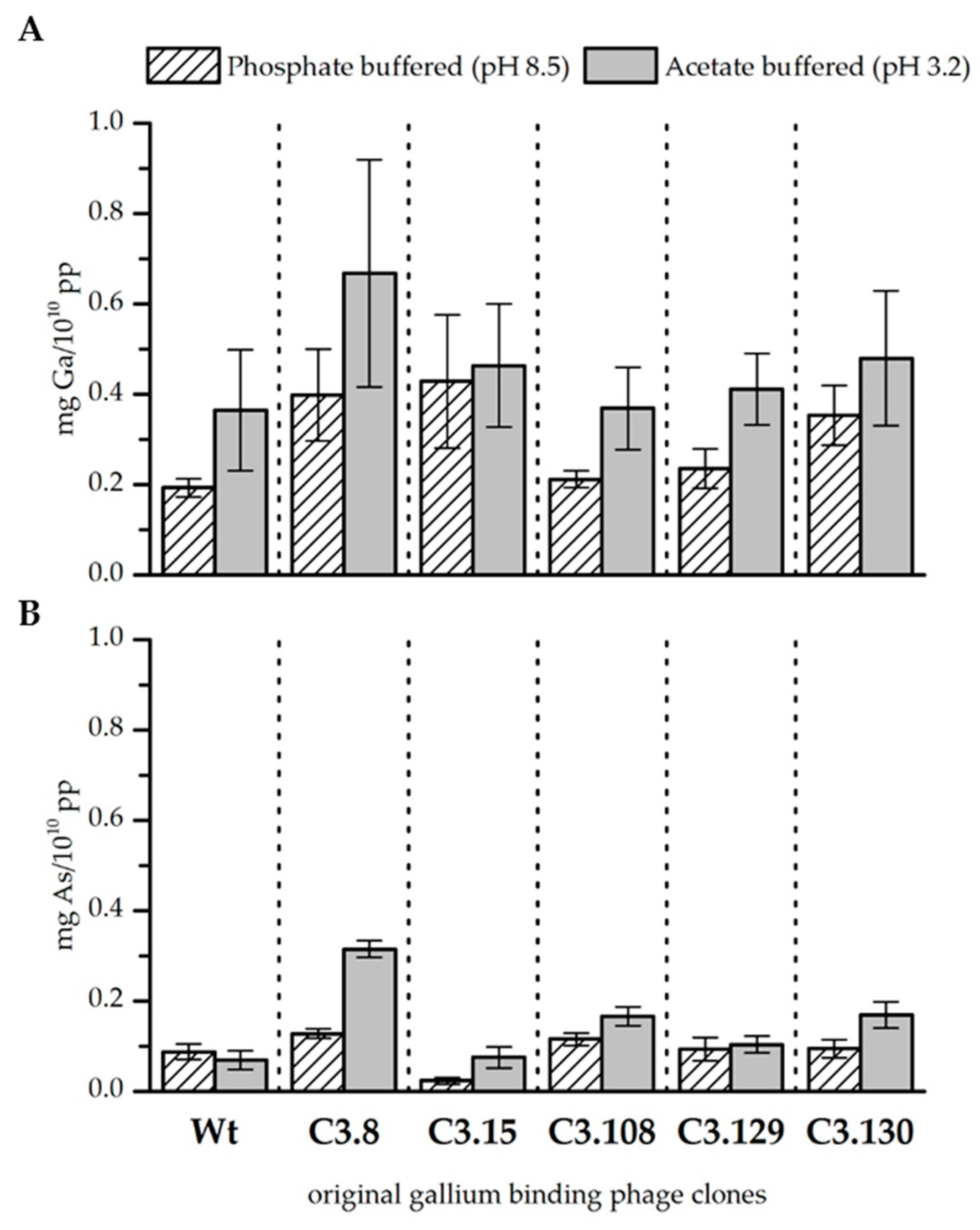
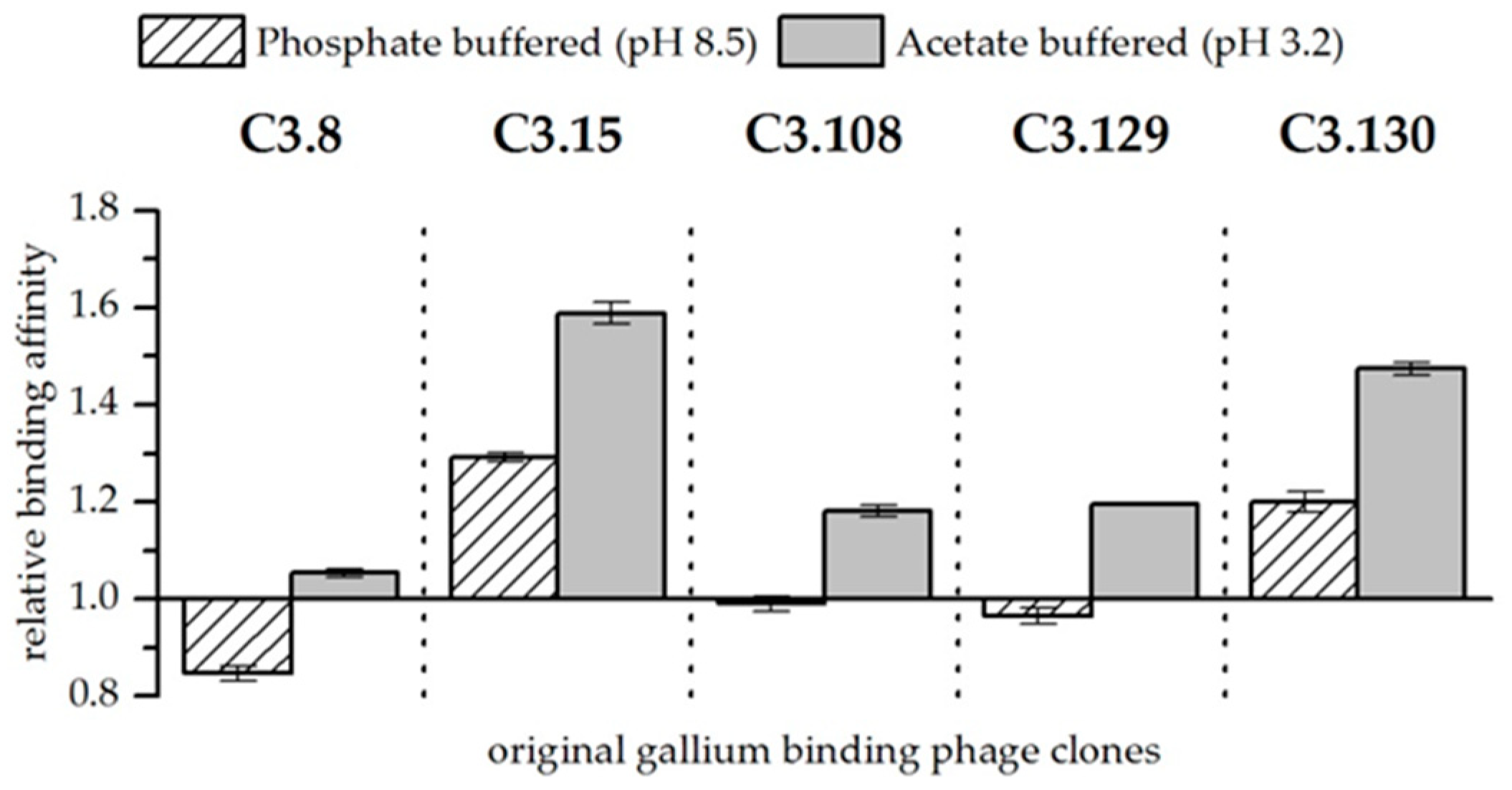
3.2. Original Phage Clone Characterization
3.3. Site-Directed Mutagenesis Experiments
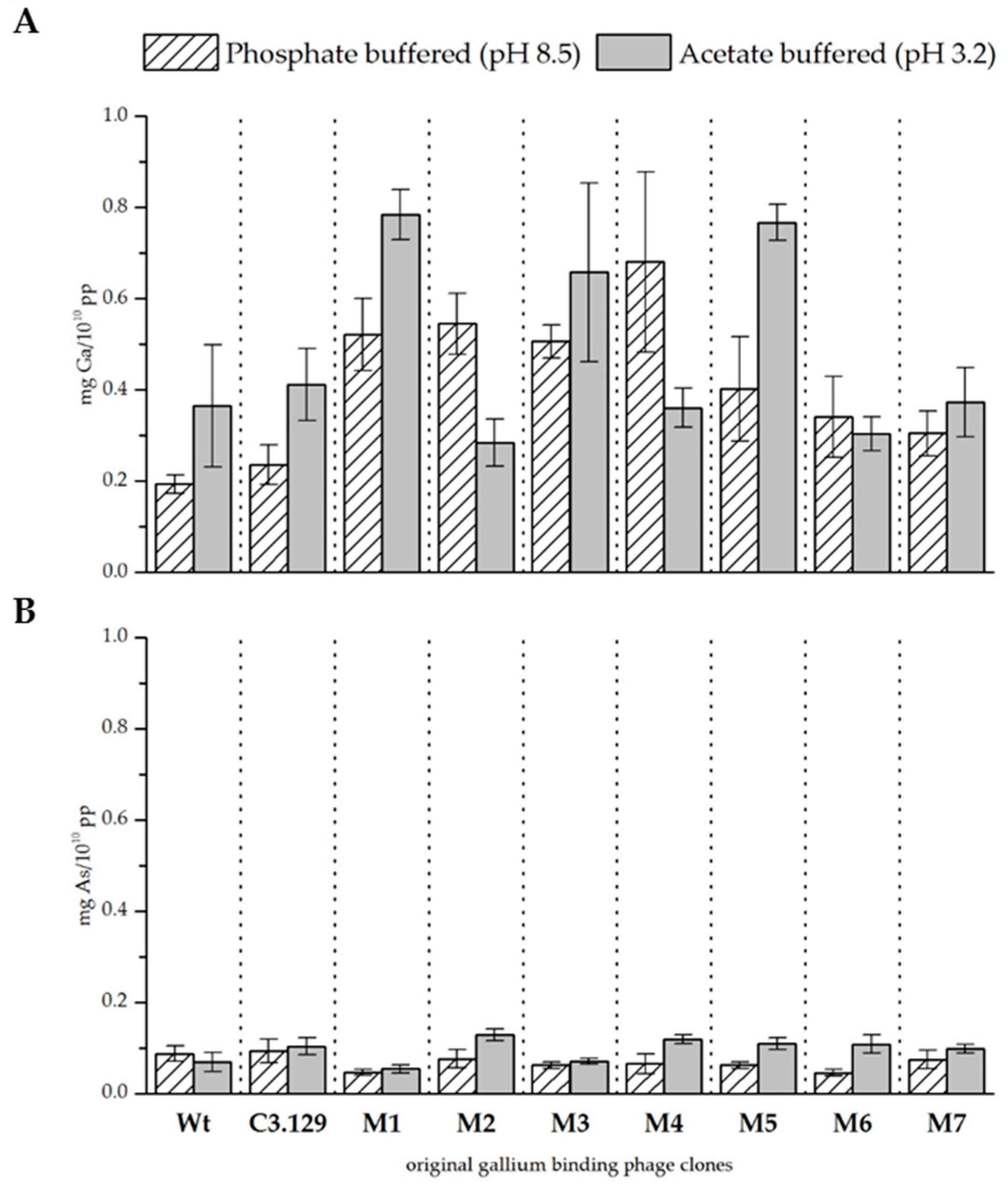
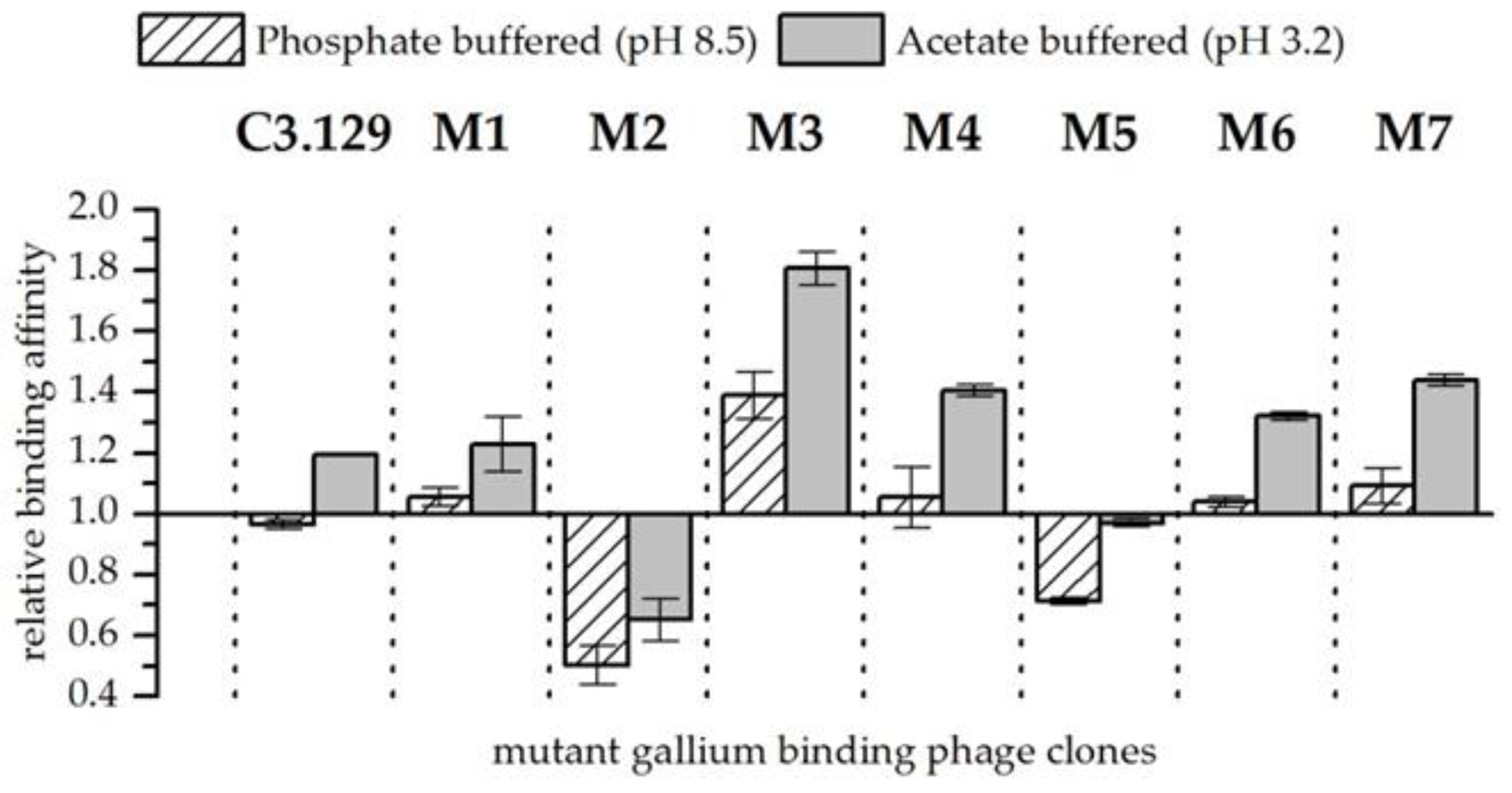
3.4. Mutant Phage Clone Characterization
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Farrell, N. Biomedical uses and applications of inorganic chemistry. An overview. Coord. Chem. Rev. 2002, 232, 1–4. [Google Scholar] [CrossRef]
- Benson, D.E.; Wisz, M.S.; Hellinga, H.W. The development of new biotechnologies using metalloprotein design. Curr. Opin. Biotechnol. 1998, 9, 370–376. [Google Scholar] [CrossRef]
- Happe, T.; Hemschemeier, A. Metalloprotein mimics–old tools in a new light. Trends Biotechnol. 2014, 32, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Fushinobu, S. A new face for biomass breakdown. Nat. Methods 2013, 10, 88–89. [Google Scholar] [CrossRef] [PubMed]
- Care, A.; Bergquist, P.L.; Sunna, A. Solid-binding peptides: smart tools for nanobiotechnology. Trends Biotechnol. 2015, 33, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Cohavi, O.; Corni, S.; De Rienzo, F.; Di Felice, R.; Gottschalk, K.E.; Hoefling, M.; Kokh, D.; Molinari, E.; Schreiber, G.; Vaskevich, A.; et al. Protein-surface interactions: challenging experiments and computations. J. Mol. Recognit. 2010, 23, 259–262. [Google Scholar] [CrossRef] [PubMed]
- Drummy, L.F.; Jones, S.E.; Pandey, R.B.; Farmer, B.L.; Vaia, R.A.; Naik, R.R. Bioassembled Layered Silicate-Metal Nanoparticle Hybrids. ACS Appl. Mater. Interfaces 2010, 2, 1492–1498. [Google Scholar] [CrossRef] [PubMed]
- Tamerler, C.; Sarikaya, M. Molecular biomimetics: nanotechnology and bionanotechnology using genetically engineered peptides. Philos. Trans. Soc. A: Math. Phys. Eng. Sci. 2009, 367, 1705–1726. [Google Scholar] [CrossRef] [PubMed]
- Dunbar, W.S. Biotechnology and the Mine of Tomorrow. Trends Biotechnol. 2017, 35, 79–89. [Google Scholar] [CrossRef]
- Pollmann, K.; Kutschke, S.; Matys, S.; Raff, J.; Hlawacek, G.; Lederer, F.L. Bio-recycling of metals: Recycling of technical products using biological applications. Biotechnol. Adv. 2018, 36, 1048–1062. [Google Scholar] [CrossRef]
- Hellinga, H.W. Metalloprotein design. Curr. Opin. Biotechnol. 1996, 7, 437–441. [Google Scholar] [CrossRef]
- Sarikaya, M.; Tamerler, C.; Jen, A.K.-Y.; Schulten, K.; Baneyx, F. Molecular biomimetics: nanotechnology through biology. Nat. Mater. 2003, 2, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Nian, R.; Kim, D.S.; Nguyen, T.; Tan, L.; Kim, C.W.; Yoo, I.K.; Choe, W.S. Chromatographic biopanning for the selection of peptides with high specificity to Pb2+ from phage displayed peptide library. J. Chromatogr. A 2010, 1217, 5940–5949. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Zhang, X.-Y.; Zhang, X.-X.; Chen, M.-L.; Wang, J.-H. Chromium(III) Binding Phage Screening for the Selective Adsorption of Cr(III) and Chromium Speciation. ACS Appl. Mater. Interfaces 2015, 7, 21287–21294. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Report on Critical Raw Materials and the Circular Economy; European Union: Brussels, Belgium, 2018. [Google Scholar]
- Frenzel, M.; Mikolajczak, C.; Reuter, M.A.; Gutzmer, J. Quantifying the relative availability of high-tech by-product metals—The cases of gallium, germanium and indium. Resour. Policy 2017, 52, 327–335. [Google Scholar] [CrossRef]
- Schönberger, N.; Braun, R.; Matys, S.; Lederer, F.L.; Lehmann, F.; Flemming, K.; Pollmann, K. Chromatopanning for the identification of gallium binding peptides. J. Chromatogr. A 2019. [Google Scholar] [CrossRef] [PubMed]
- Vallet, V.; Wahlgren, U.; Grenthe, I. Chelate Effect and Thermodynamics of Metal Complex Formation in Solution: A Quantum Chemical Study. J. Am. Chem. Soc. 2003, 125, 14941–14950. [Google Scholar] [CrossRef]
- Hancock, R.D.; Martell, A.E. The Chelate, Cryptate and Macrocyclic Effects. Comments Inorg. Chem. 1988, 6, 237–284. [Google Scholar] [CrossRef]
- Smith, G.P.; Scott, J.K. Libraries of peptides and proteins displayed on filamentous phage. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 1993; Volume 217, pp. 228–257. [Google Scholar]
- Barbas, C.F.; Burton, D.R.; Scott, J.K.; Silverman, G.J. Phage Display: A Laboratory Manual; Cold Spring Harbor Labratory Press: New York, NY, USA, 2001. [Google Scholar]
- Center for Cocoa Biotechnology Research and Development. Preparation of Competent E. Coli Cells Using Calcium Chloride. Available online: http://www.koko.gov.my/CocoaBioTech/DNA%20Cells23.html#procedure (accessed on 21 September 2018).
- Wood, S.A.; Samson, I.M. The aqueous geochemistry of gallium, germanium, indium and scandium. Ore Geol. Rev. 2006, 28, 57–102. [Google Scholar] [CrossRef]
- Cullen, W.R.; Reimer, K.J. Arsenic speciation in the environment. Chem. Rev. 1989, 89, 713–764. [Google Scholar] [CrossRef]
- Schmidtke, A.; Läppchen, T.; Weinmann, C.; Bier-Schorr, L.; Keller, M.; Kiefer, Y.; Holland, J.P.; Bartholomä, M.D. Gallium Complexation, Stability, and Bioconjugation of 1,4,7-Triazacyclononane Derived Chelators with Azaheterocyclic Arms. Inorg. Chem. 2017, 56, 9097–9110. [Google Scholar] [CrossRef] [PubMed]
- Kubíček, V.; Havlíčková, J.; Kotek, J.; Tircsó, G.; Hermann, P.; Tóth, É.; Lukeš, I. Gallium(III) Complexes of DOTA and DOTA−Monoamide: Kinetic and Thermodynamic Studies. Inorg. Chem. 2010, 49, 10960–10969. [Google Scholar] [CrossRef] [PubMed]
- Hacht, B. Gallium(III) Ion Hydrolysis under Physiological Conditions. Bull. Korean Chem. Soc. 2008, 29, 5. [Google Scholar] [CrossRef]
- Cabbiness, D.K.; Margerum, D.W. Macrocyclic effect on the stability of copper(II) tetramine complexes. J. Am. Chem. Soc. 1969, 91, 6540–6541. [Google Scholar] [CrossRef]
- Lederer, F.L.; Curtis, S.B.; Bachmann, S.; Dunbar, W.S.; MacGillivray, R.T.A. Identification of lanthanum-specific peptides for future recycling of rare earth elements from compact fluorescent lamps. Biotechnol. Bioeng. 2017, 114, 1016–1024. [Google Scholar] [CrossRef] [PubMed]
- Sarikaya, M.; Dinçer, S.; Schwartz, D.T.; Tamerler, C.; Cebeci, A.; Karaguler, N.G.; Oren, E.E.; Cetinel, S.; Whitaker, J.D. Peptides to bridge biological-platinum materials interface. Bioinspired Biomim. Nanobiomat. 2012, 1, 143–153. [Google Scholar]
- Stöhrer, G. Arsenic: Opportunity for risk assessment. Arch. Toxicol. 1991, 65, 525–531. [Google Scholar] [CrossRef] [PubMed]
| Name | Sequence | pI | Origin |
|---|---|---|---|
| C3.8 | TMHHAAIAHPPH | 6.82 | [17] |
| C3.15 | NYPLHQSSSPSR | 5.08 | |
| C3.108 | SQALSTSRQDLR | 9.31 | |
| C3.129 | HTQHIQSDDHLA | 5.70 | |
| C3.130 | NDLQRHRLTAP | 9.61 | |
| M1: H1C1/Q6C2 | CTQHICSDDHLA | 5.05 | This work; SDM experiment of clone C3.129 |
| M2: T2C1/S7C2 | HCQHIQCDDHLA | 5.70 | |
| M3: Q3C1/D8C2 | HTCHIQSCDHLA | 6.25 | |
| M4: H4C1/D9C2 | HTQCIQSDCHLA | 5.97 | |
| M5: I5C1/H10C2 | HTQHCQSDDCLA | 5.05 | |
| M6: Q6C1/L11C2 | HTQHICSDDHCA | 5.70 | |
| M7: S7C1/A12C2 | HTQHIQCDDHLC | 5.70 |
| Clone | TA | Pol | SDM Primer_Forward (5’-3’) & SDM Primer_Reverse (5’-3’) |
|---|---|---|---|
| M1 | 59 °C | Ph | CATATTTGTAGTGATGATCATCTTGCG/ CTGCGTACAAGAGTGAGAATAGAAAGGTAC |
| M2 | 59 °C | Ph | ATTCAGTGTGATGATCATCTTGCGGGTG/ATGCTGACAATGAGAGTGAGAATAGAAAGG |
| M3 | 61 °C | Q5 | CAGAGTTGTGATCATCTTGCGGGTGGA/ AATATGACACGTATGAGAGTGAGAATAGAAAG |
| M4 | 61 °C | Q5 | AGTGATTGTCATCTTGCGGGTGGAGGT/CTGAATACACTGCGTATGAGAGTGAGAATAG |
| M5 | 64 °C | Q5 | GATGATTGTCTTGCGGGTGGAGGTTCG/ACTCTGACAATGCTGCGTATGAGAGTGAG |
| M6 | 65 °C | Q5 | GATCATTGTGCGGGTGGAGGTTCGGCC/ATCACTACAAATATGCTGCGTATGAGAGTGAGAATAGAAAGGTAC |
| M7 | 62 °C | Ph | CATCTTTGTGGTGGAGGTTCGGCCGAA/ATCATCACACTGAATATGCTGCGTATGAGAGTG |
| Metal Concentration | Concentration of Buffer Components | pH |
|---|---|---|
| 2.8 mM Ga | 0.0947 M Na2HPO4 0.0053 M NaH2PO4 | 8.5 |
| 2.7 mM Ga | 0.0995 M CH3COOH 0.0005 M NaCH3COO | 3.2 |
| 3.1 mM As | 0.0947 M Na2HPO4 0.0053 M NaH2PO4 | 8.5 |
| 2.6 mM As | 0.0995 M CH3COOH 0.0005 M NaCH3COO | 3.2 |
| 1.2 mM Ga 1.3 mM As | 0.0947 M Na2HPO4 0.0053 M NaH2PO4 | 8.5 |
| 1.5 mM Ga 1.4 mM As | 0.0995 M CH3COOH 0.0005 M NaCH3COO | 3.2 |
| Clone | Relative Biosorption of Ga (Increase above Wt) | Relative Biosorption of As (Increase above Wt) | ||||||
|---|---|---|---|---|---|---|---|---|
| Phosphate Buffered (pH 8.5) | Acetate Buffered (pH 3.2) | Phosphate Buffered (pH 8.5) | Acetate Buffered (pH 3.2) | |||||
| C3.8 | 2.06 ± | 0.52 | 1.83 ± | 0.69 | 1.46 ± | 0.12 | 4.58 ± | 0.27 |
| C3.15 | 2.21 ± | 0.76 | 1.27 ± | 0.37 | 0.27 ± | 0.08 | 1.09 ± | 0.34 |
| C3.108 | 1.10 ± | 0.09 | 1.01 ± | 0.25 | 1.32 ± | 0.15 | 2.41 ± | 0.3 |
| C3.129 | 1.22 ± | 0.22 | 1.13 ± | 0.22 | 1.06 ± | 0.3 | 1.51 ± | 0.28 |
| C3.130 | 1.83 ± | 0.34 | 1.31 ± | 0.41 | 1.08 ± | 0.23 | 2.46 ± | 0.43 |
| Clone | Relative Biosorption of Ga (Increase above Wt) | Relative Biosorption of As (Increase above Wt) | ||||||
|---|---|---|---|---|---|---|---|---|
| Phosphate Buffered (pH 8.5) | Acetate Buffered (pH 3.2) | Phosphate Buffered (pH 8.5) | Acetate Buffered (pH 3.2) | |||||
| C3.129 | 1.22 ± | 0.22 | 1.13 ± | 0.22 | 1.06 ± | 0.3 | 1.51 ± | 0.28 |
| M1 | 2.69 ± | 0.41 | 2.15 ± | 0.15 | 0.53 ± | 0.07 | 0.78 ± | 0.13 |
| M2 | 2.81 ± | 0.34 | 0.78 ± | 0.14 | 0.87 ± | 0.24 | 1.87 ± | 0.19 |
| M3 | 2.62 ± | 0.19 | 1.80 ± | 0.54 | 0.71 ± | 0.08 | 1.04 ± | 0.09 |
| M4 | 3.52 ± | 1.02 | 0.99 ± | 0.12 | 0.74 ± | 0.25 | 1.74 ± | 0.13 |
| M5 | 2.08 ± | 0.59 | 2.10 ± | 0.11 | 0.71 ± | 0.08 | 1.59 ± | 0.18 |
| M6 | 1.76 ± | 0.46 | 0.83 ± | 0.1 | 0.52 ± | 0.07 | 1.57 ± | 0.29 |
| M7 | 1.58 ± | 0.25 | 1.02 ± | 0.21 | 0.85 ± | 0.23 | 1.43 ± | 0.14 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schönberger, N.; Zeitler, C.; Braun, R.; Lederer, F.L.; Matys, S.; Pollmann, K. Directed Evolution and Engineering of Gallium-Binding Phage Clones—A Preliminary Study. Biomimetics 2019, 4, 35. https://doi.org/10.3390/biomimetics4020035
Schönberger N, Zeitler C, Braun R, Lederer FL, Matys S, Pollmann K. Directed Evolution and Engineering of Gallium-Binding Phage Clones—A Preliminary Study. Biomimetics. 2019; 4(2):35. https://doi.org/10.3390/biomimetics4020035
Chicago/Turabian StyleSchönberger, Nora, Christina Zeitler, Robert Braun, Franziska L. Lederer, Sabine Matys, and Katrin Pollmann. 2019. "Directed Evolution and Engineering of Gallium-Binding Phage Clones—A Preliminary Study" Biomimetics 4, no. 2: 35. https://doi.org/10.3390/biomimetics4020035
APA StyleSchönberger, N., Zeitler, C., Braun, R., Lederer, F. L., Matys, S., & Pollmann, K. (2019). Directed Evolution and Engineering of Gallium-Binding Phage Clones—A Preliminary Study. Biomimetics, 4(2), 35. https://doi.org/10.3390/biomimetics4020035





