Biosafety of Mesoporous Silica Nanoparticles
Abstract
:1. Introduction
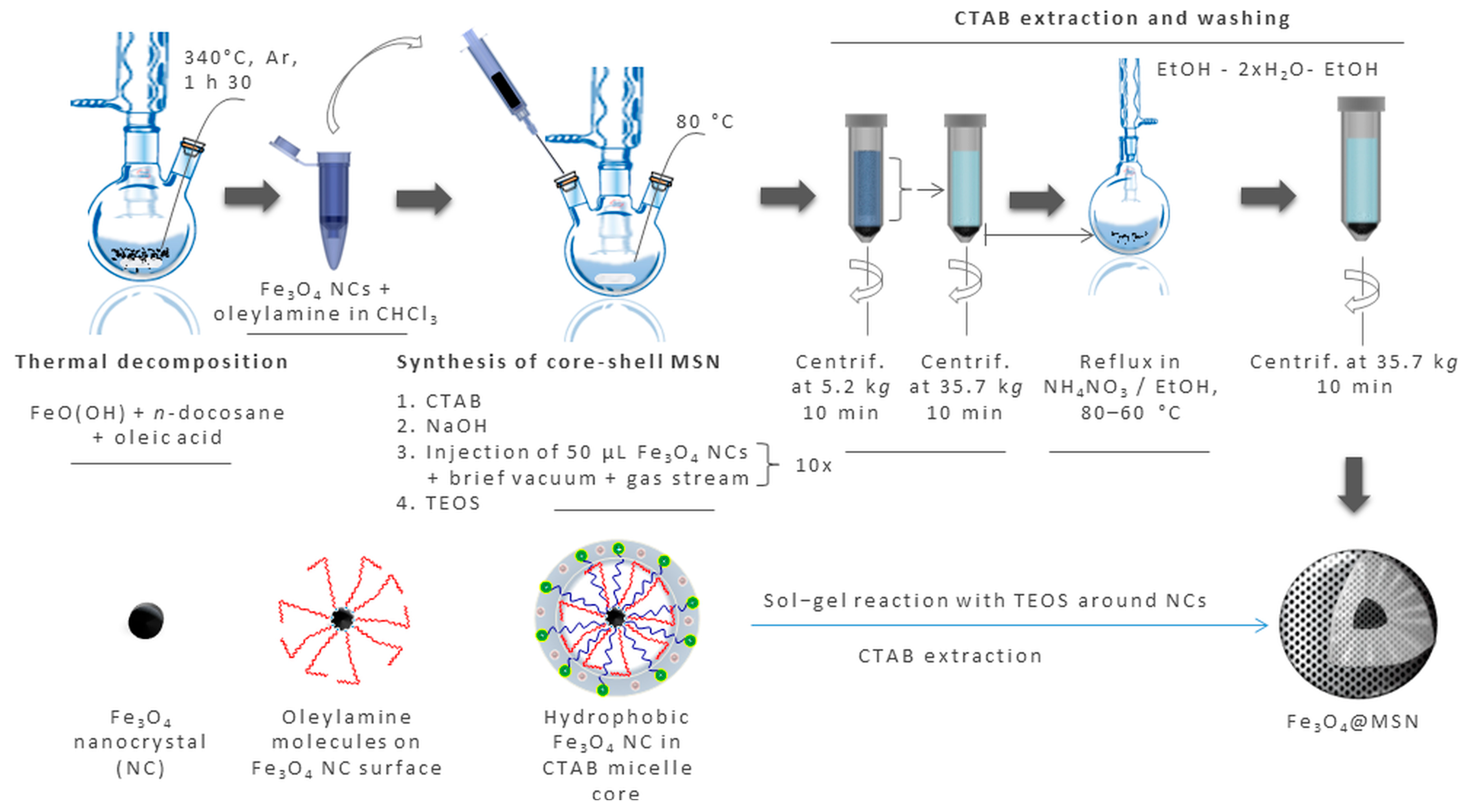
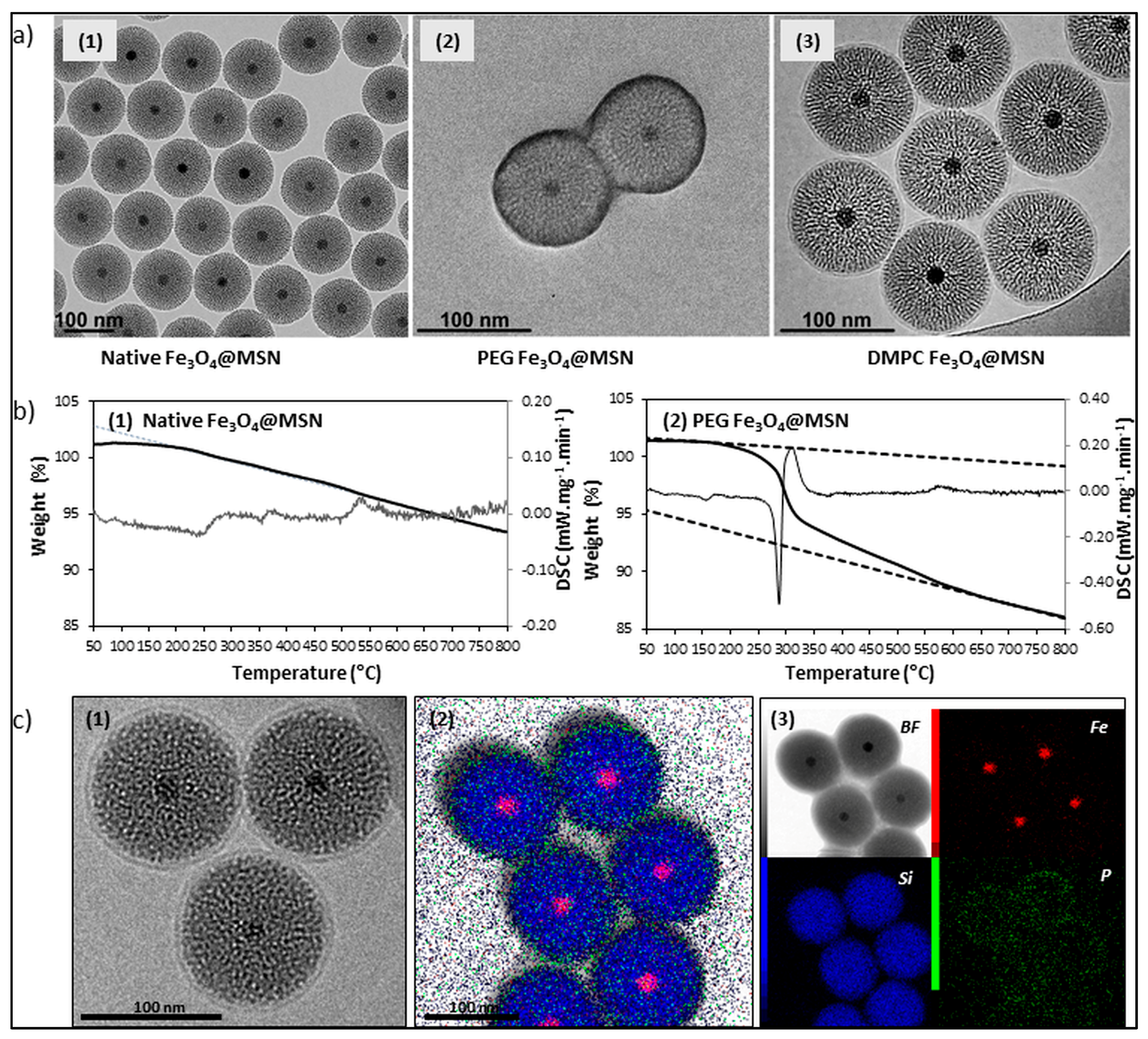
2. Preparation of Magnetic Mesoporous Silica Nanoparticles
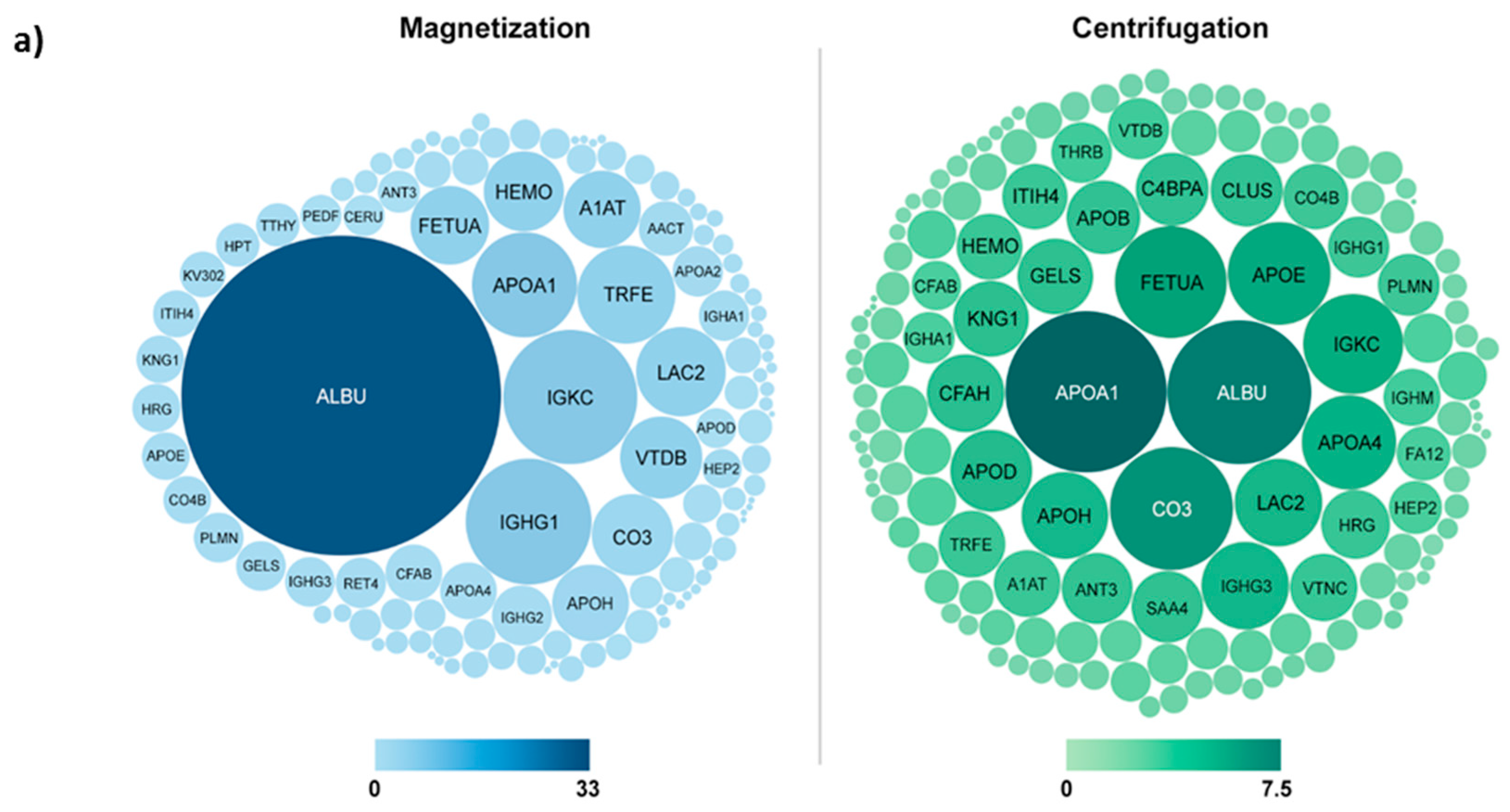
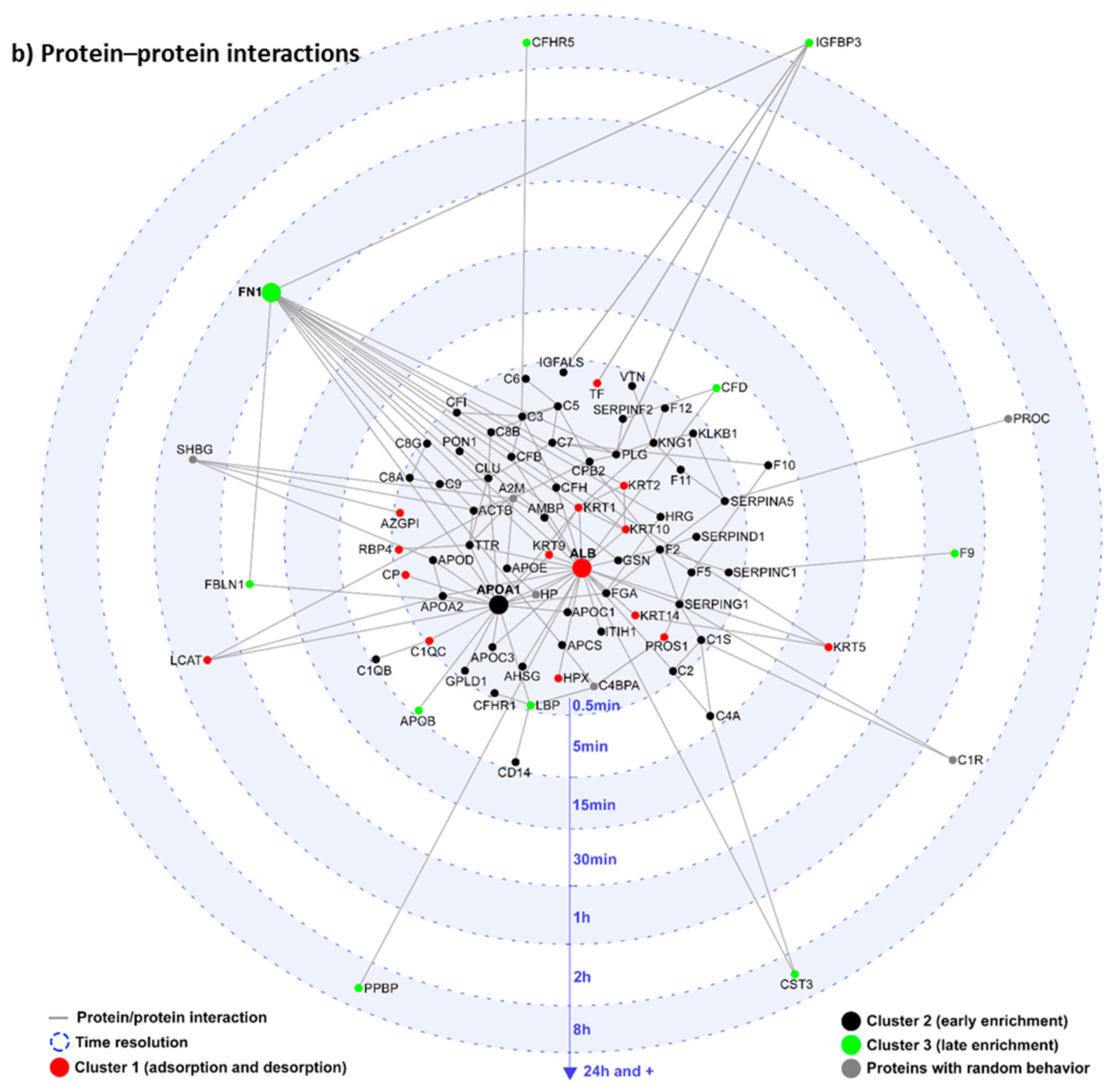
3. Magnetic Separation for Corona Characterization
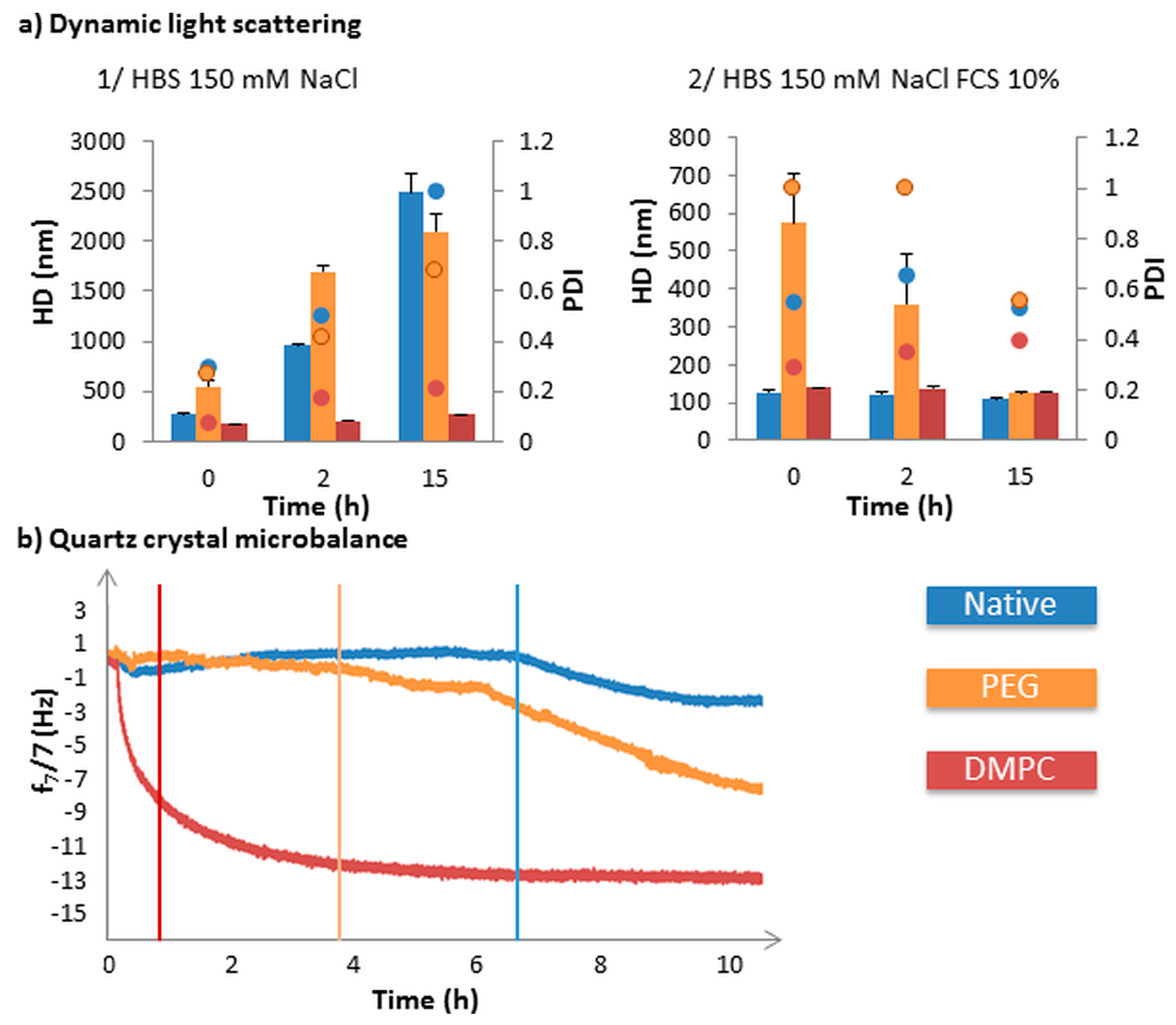
4. Influence of Magnetic Mesoporous Silica Nanoparticles’ Coverage on Their Interaction with Proteins and Cell Membranes
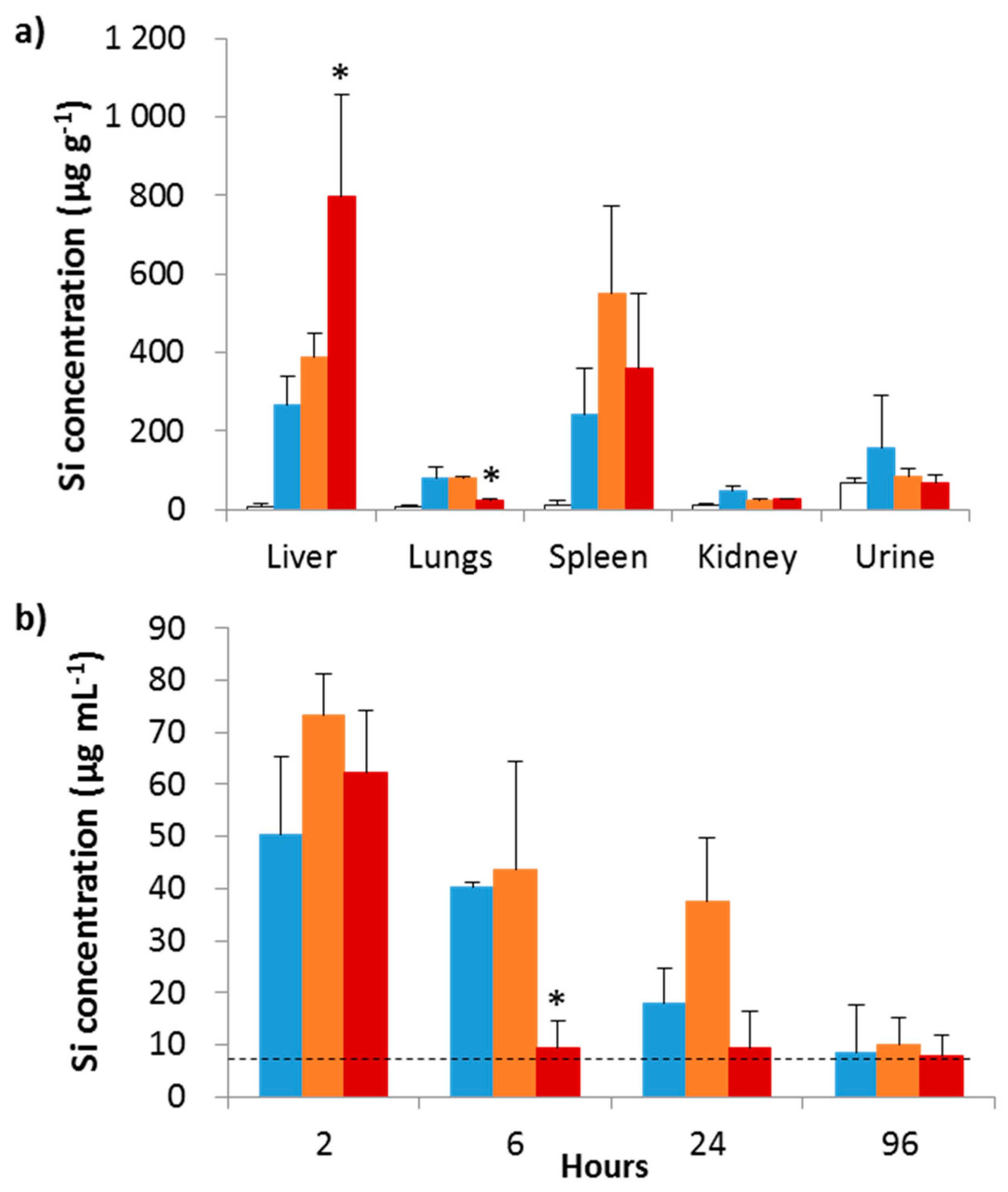
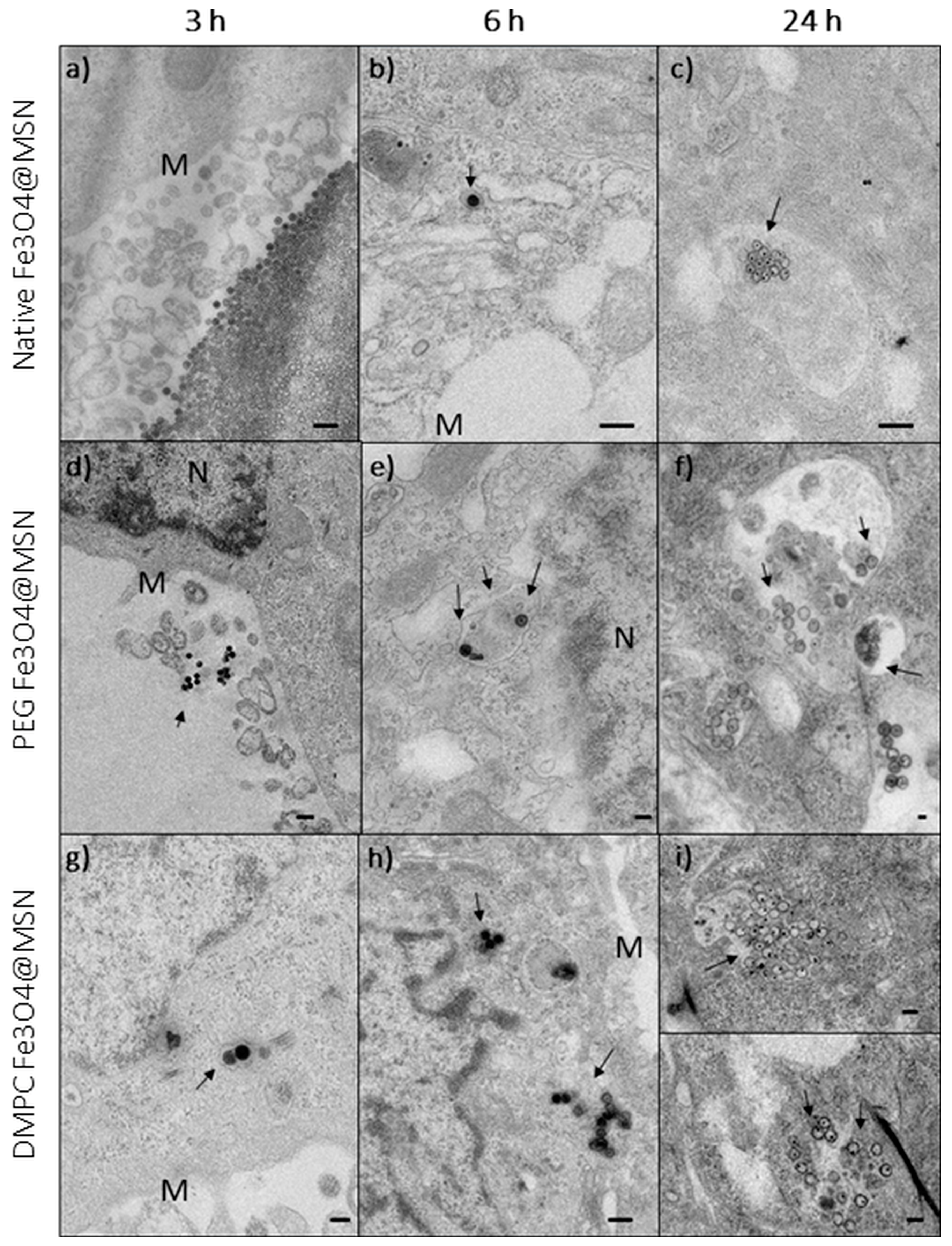
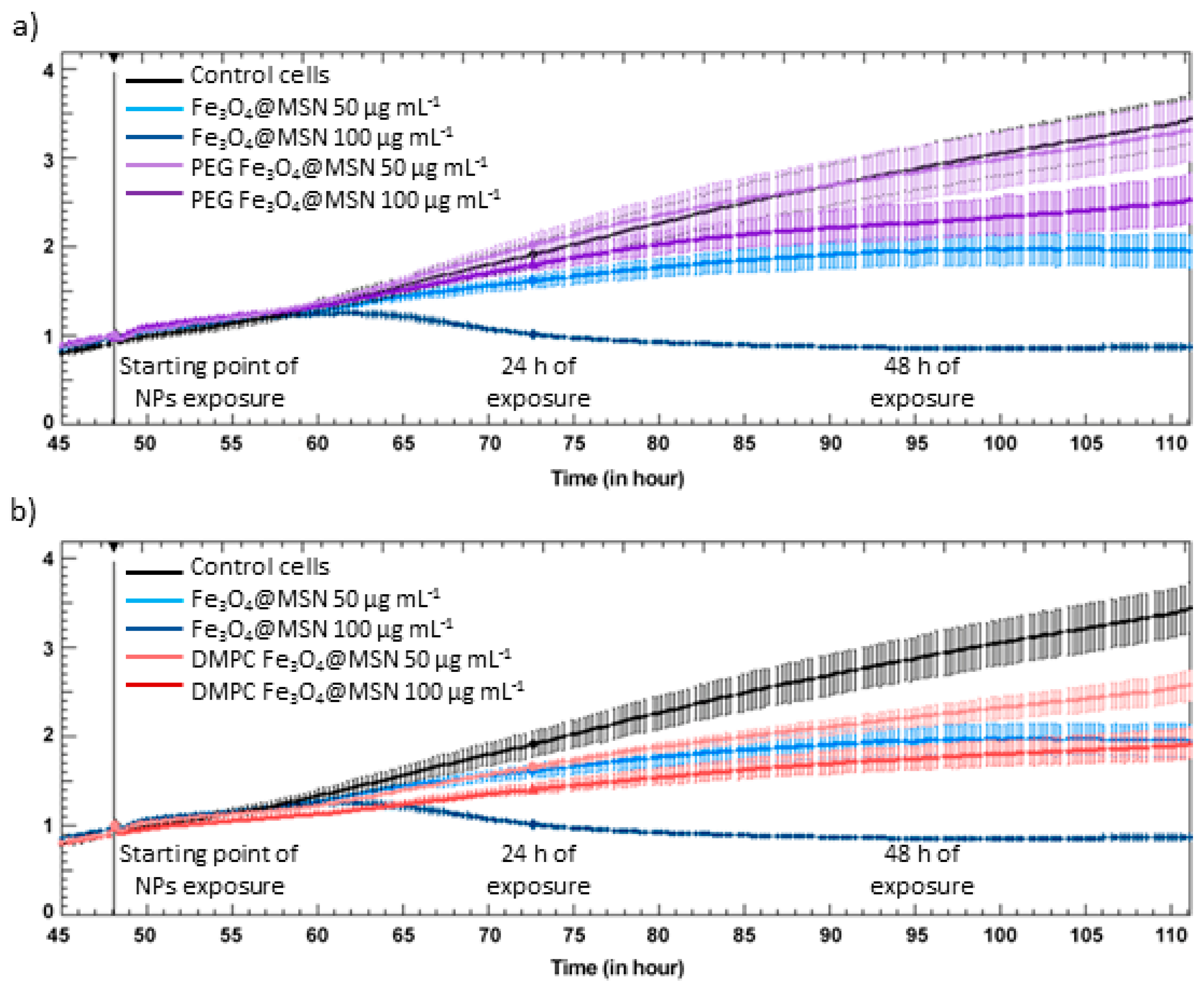
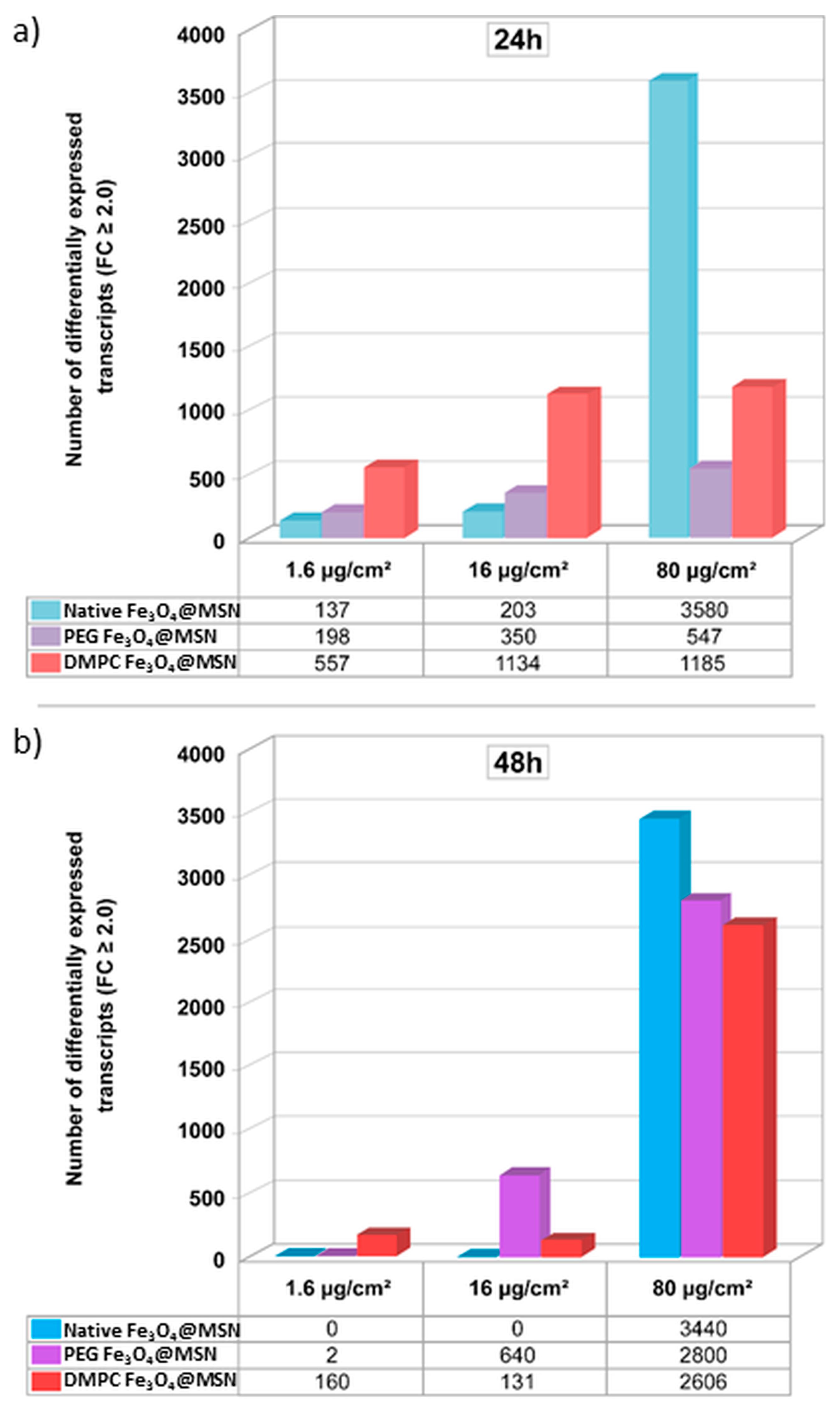
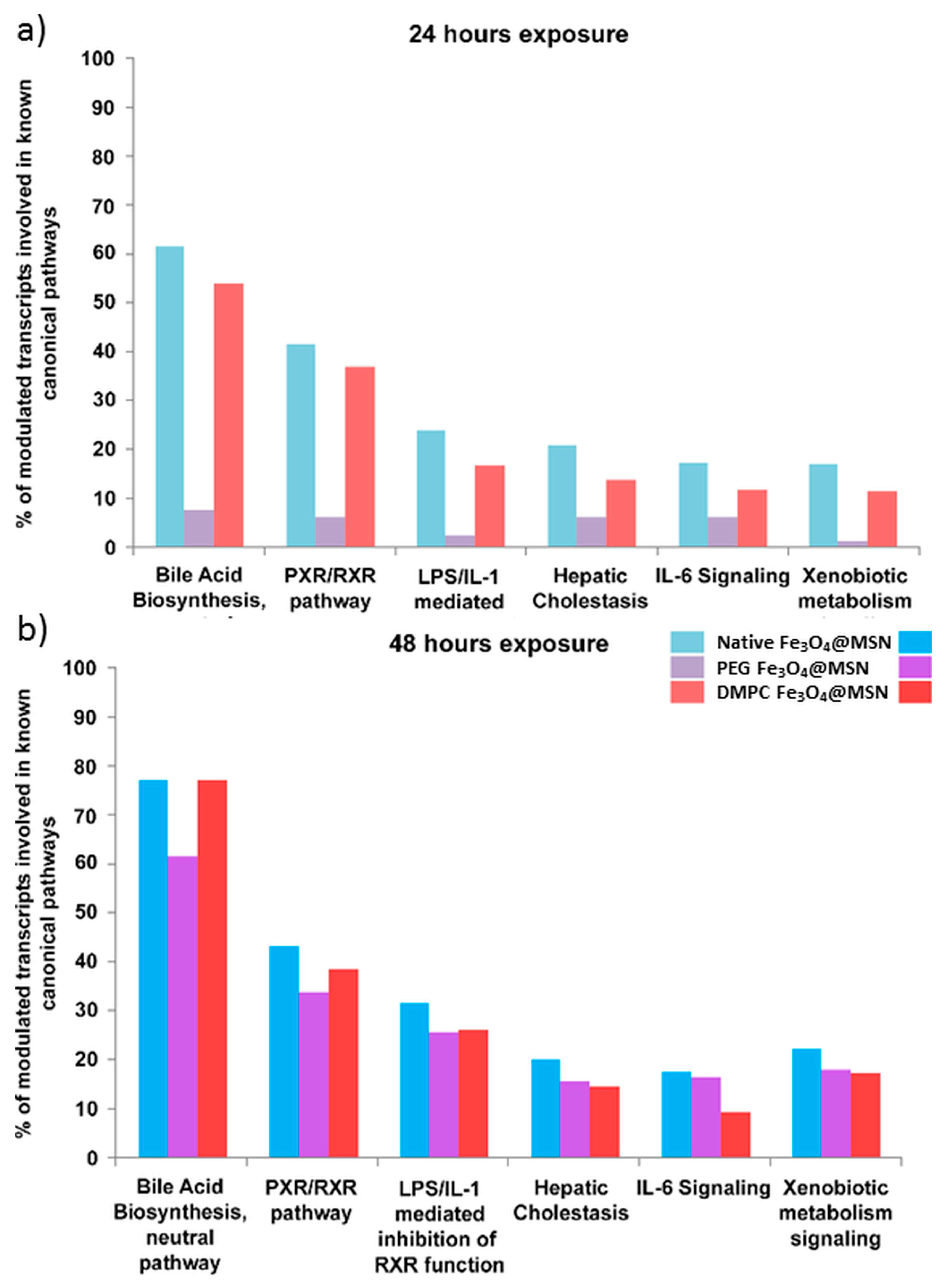
5. Magnetic Mesoporous Silica Nanoparticles’ In Vivo Toxicity and In Vitro Mechanisms
6. Conclusions
Funding
Conflicts of Interest
References
- Chang, B.; Guo, J.; Liu, C.; Qian, J.; Yang, W. Surface functionalization of magnetic mesoporous silica nanoparticles for controlled drug release. J. Mater. Chem. 2010, 20, 9941. [Google Scholar] [CrossRef]
- Perrier, M.; Gary-Bobo, M.; Lartigue, L.; Brevet, D.; Morère, A.; Garcia, M.; Maillard, P.; Raehm, L.; Guari, Y.; Larionova, J.; et al. Mannose-functionalized porous silica-coated magnetic nanoparticles for two-photon imaging or PDT of cancer cells. J. Nanoparticle Res. 2013, 15, 1602. [Google Scholar] [CrossRef]
- Nyalosaso, J.L.; Rascol, E.; Pisani, C.; Dorandeu, C.; Dumail, X.; Maynadier, M.; Gary-Bobo, M.; Kee Him, J.L.; Bron, P.; Garcia, M.; et al. Synthesis, decoration, and cellular effects of magnetic mesoporous silica nanoparticles. RSC Adv. 2016, 6, 57275–57283. [Google Scholar] [CrossRef]
- Rascol, E.; Daurat, M.; Da Silva, A.; Maynadier, M.; Dorandeu, C.; Charnay, C.; Garcia, M.; Lai-Kee-Him, J.; Bron, P.; Auffan, M.; et al. Biological fate of Fe3O4 core-shell mesoporous silica nanoparticles depending on particle surface chemistry. Nanomaterials 2017, 7, 162. [Google Scholar] [CrossRef] [PubMed]
- Pisani, C.; Gaillard, J.-C.; Odorico, M.; Nyalosaso, J.L.; Charnay, C.; Guari, Y.; Chopineau, J.; Devoisselle, J.-M.; Armengaud, J.; Prat, O. The timeline of corona formation around silica nanocarriers highlights the role of the protein interactome. Nanoscale 2017, 9, 1840–1851. [Google Scholar] [CrossRef] [PubMed]
- Pisani, C.; Rascol, E.; Dorandeu, C.; Charnay, C.; Guari, Y.; Chopineau, J.; Devoisselle, J.-M.; Prat, O. Biocompatibility assessment of functionalized magnetic mesoporous silica nanoparticles in human HepaRG cells. Nanotoxicology 2017, 11, 871–890. [Google Scholar] [CrossRef] [PubMed]
- Roggers, R.; Kanvinde, S.; Boonsith, S.; Oupický, D. The practicality of mesoporous silica nanoparticles as drug delivery devices and progress toward this goal. AAPS PharmSciTech 2014, 15, 1163–1171. [Google Scholar] [CrossRef] [PubMed]
- Fontecave, T.; Sanchez, C.; Azaïs, T.; Boissiére, C. Chemical modification as a versatile tool for tuning stability of silica based mesoporous carriers in biologically relevant conditions. Chem. Mater. 2012, 24, 4326–4336. [Google Scholar] [CrossRef]
- Bass, J.D.; Grosso, D.; Boissiere, C.; Belamie, E.; Coradin, T.; Sanchez, C. Stability of mesoporous oxide and mixed metal oxide materials under biologically relevant conditions. Chem. Mater. 2007, 19, 4349–4356. [Google Scholar] [CrossRef]
- He, Q.; Shi, J.; Zhu, M.; Chen, Y.; Chen, F. The three-stage in vitro degradation behavior of mesoporous silica in simulated body fluid. Microporous Mesoporous Mater. 2010, 131, 314–320. [Google Scholar] [CrossRef]
- Cauda, V.; Argyo, C.; Bein, T. Impact of different PEGylation patterns on the long-term bio-stability of colloidal mesoporous silica nanoparticles. J. Mater. Chem. 2010, 20, 8693–8699. [Google Scholar] [CrossRef]
- Braun, K.; Pochert, A.; Beck, M.; Fiedler, R.; Gruber, J.; Lindén, M. Dissolution kinetics of mesoporous silica nanoparticles in different simulated body fluids. J. Sol-Gel Sci. Technol. 2016, 79, 319–327. [Google Scholar] [CrossRef]
- Rimola, A.; Costa, D.; Sodupe, M.; Lambert, J.F.; Ugliengo, P. Silica surface features and their role in the adsorption of biomolecules: Computational modeling and experiments. Chem. Rev. 2013, 113, 4216–4313. [Google Scholar] [CrossRef] [PubMed]
- Tarn, D.; Ashley, C.E.; Xue, M.; Carnes, E.C.; Zink, J.I.; Brinker, C.J. Masoporous Silica Nanocarriers: Biofunctionality and Biocompatibility. Acc. Chem. Res. 2013, 46, 792–801. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Qiao, S.Z.; Hu, Q.H.; Max Lu, G.Q. Magnetic Nanocomposites with Mesoporous Structures: Synthesis and Applications. Small 2011, 7, 425–443. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Detrembleur, C.; De Pauw-Gillet, M.C.; Mornet, S.; Elst, L.V.; Laurent, S.; Jérôme, C.; Duguet, E. Heat-triggered drug release systems based on mesoporous silica nanoparticles filled with a maghemite core and phase-change molecules as gatekeepers. J. Mater. Chem. B 2014, 2, 59–70. [Google Scholar] [CrossRef]
- Pisani, C.; Gaillard, J.C.; Dorandeu, C.; Charnay, C.; Guari, Y.; Chopineau, J.; Devoisselle, J.M.; Armengaud, J.; Prat, O. Experimental separation steps influence the protein content of corona around mesoporous silica nanoparticles. Nanoscale 2017, 9, 5769–5772. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Gill, E.E.; Hancock, R.E.W. NetworkAnalyst for statistical, visual and network-based meta-analysis of gene expression data. Nat. Protoc. 2015, 10, 823–844. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Benner, M.J.; Hancock, R.E.W. NetworkAnalyst—integrative approaches for protein–protein interaction network analysis and visual exploration. Nucleic Acids Res. 2014, 42, W167–W174. [Google Scholar] [CrossRef] [PubMed]
- Innate DB. Available online: www.innatedb.com (accessed on 1 May 2016).
- He, Q.; Zhang, Z.; Gao, F.; Li, Y.; Shi, J. In vivo Biodistribution and Urinary Excretion of Mesoporous Silica Nanoparticles: Effects of Particle Size and PEGylation. Small 2011, 7, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Ashley, C.E.; Carnes, E.C.; Phillips, G.K.; Padilla, D.; Durfee, P.N.; Brown, P.A.; Hanna, T.N.; Liu, J.; Phillips, B.; Carter, M.B.; et al. The targeted delivery of multicomponent cargos to cancer cells by nanoporous particle-supported lipid bilayers. Nat. Mater. 2011, 10, 389–397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Butler, K.S.; Durfee, P.N.; Theron, C.; Ashley, C.E.; Carnes, E.C.; Brinker, J. Protocells: Modular Mesoporous Silica Nanoparticle- Supported Lipid Bilayers for Drug Delivery. Small 2016, 12, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Durfee, P.N.; Lin, Y.S.; Dunphy, D.R.; Muñiz, A.J.; Butler, K.S.; Humphrey, K.R.; Lokke, A.J.; Agola, J.O.; Chou, S.S.; Chen, I.M.; et al. Mesoporous Silica Nanoparticle-Supported Lipid Bilayers (Protocells) for Active Targeting and Delivery to Individual Leukemia Cells. ACS Nano 2016, 10, 8325–8345. [Google Scholar] [CrossRef] [PubMed]
- Tada, D.B.; Suraniti, E.; Rossi, L.M.; Leite, C.A.P.; Oliveira, C.S.; Tumolo, T.C.; Calemczuk, R.; Livache, T.; Baptista, M.S. Effect of lipid coating on the interaction between silica nanoparticles and membranes. J. Biomed. Nanotechnol. 2014, 10, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Savarala, S.; Ahmed, S.; Ilies, M.A.; Wunder, S.L. Formation and colloidal stability of dmpc supported lipid bilayers on SiO 2 nanobeads. Langmuir 2010, 26, 12081–12088. [Google Scholar] [CrossRef] [PubMed]
- Mornet, S.; Lambert, O.; Duguet, E.; Brisson, A. The formation of supported lipid bilayers on silica nanoparticles revealed by cryoelectron microscopy. Nano Lett. 2005, 5, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Weingart, J.; Vabbilisetty, P.; Sun, X.L. Membrane mimetic surface functionalization of nanoparticles: Methods and applications. Adv. Colloid Interface Sci. 2013, 197–198, 68–84. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, R.T.; Braga, V.H.A.; Carmona-Ribeiro, A.M. Biomimetic Cationic Nanoparticles Based on Silica: Optimizing Bilayer Deposition from Lipid Films. Biomimetics 2017, 2, 20. [Google Scholar] [CrossRef]
- Moura, S.P.; Carmona-Ribeiro, A.M. Biomimetic particles: Optimization of phospholipid bilayer coverage on silica and colloid stabilization. Langmuir 2005, 21, 10160–10164. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Savarala, S.; Chen, Y.; Bothun, G.; Wunder, S.L. Formation of lipid sheaths around nanoparticle-supported lipid bilayers. Small 2012, 8, 1740–1751. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Drazenovic, J.; Luo, Z.; Zhang, J.; Zhou, H.; Wunder, S.L. Mechanism of supported bilayer formation of zwitterionic lipids on SiO2nanoparticles and structure of the stable colloids. RSC Adv. 2012, 2, 11336–11348. [Google Scholar] [CrossRef]
- Lin, Y.S.; Haynes, C.L. Synthesis and characterization of biocompatible and size-tunable multifunctional porous silica nanoparticles. Chem. Mater. 2009, 21, 3979–3986. [Google Scholar] [CrossRef]
- Zhang, Q.; Ye, Z.; Wang, S.T.; Yin, J. Facile one-pot synthesis of PEGylated monodisperse mesoporous silica nanoparticles with controllable particle sizes. Chinese Chem. Lett. 2014, 25, 257–260. [Google Scholar] [CrossRef]
- Maurice, V.; Georgelin, T.; Siaugue, J.M.; Cabuil, V. Synthesis and characterization of functionalized core-shell γFe2O3-SiO2 nanoparticles. J. Magn. Magn. Mater. 2009, 321, 1408–1413. [Google Scholar] [CrossRef]
- Graf, C.; Gao, Q.; Schütz, I.; Noufele, C.N.; Ruan, W.; Posselt, U.; Korotianskiy, E.; Nordmeyer, D.; Rancan, F.; Hadam, S.; et al. Surface functionalization of silica nanoparticles supports colloidal stability in physiological media and facilitates internalization in cells. Langmuir 2012, 28, 7598–7613. [Google Scholar] [CrossRef] [PubMed]
- Van Schooneveld, M.M.; Vucic, E.; Koole, R.; Zhou, Y.; Stocks, J.; Cormode, D.P.; Tang, C.Y.; Gordon, R.E.; Nicolay, K.; Meijerink, A.; et al. Improved biocompatibility and pharmacokinetics of silica nanoparticles by means of a lipid coating: A multimodality investigation. Nano Lett. 2008, 8, 2517–2525. [Google Scholar] [CrossRef] [PubMed]
- Ross, E.E.; Mok, S.W.; Bugni, S.R. Assembly of lipid bilayers on silica and modified silica colloids by reconstitution of dried lipid films. Langmuir 2011, 27, 8634–8644. [Google Scholar] [CrossRef] [PubMed]
- Anthérieu, S.; Chesné, C.; Li, R.; Guguen-Guillouzo, C.; Guillouzo, A. Optimization of the HepaRG cell model for drug metabolism and toxicity studies. Toxicol. Vitr. 2012, 26, 1278–1285. [Google Scholar] [CrossRef] [PubMed]
- Guillouzo, A. Liver cell models in in vitro toxicology. Environ. Health Perspect. 1998, 106 (Suppl. 2), 511–532. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rascol, E.; Pisani, C.; Dorandeu, C.; Nyalosaso, J.L.; Charnay, C.; Daurat, M.; Da Silva, A.; Devoisselle, J.-M.; Gaillard, J.-C.; Armengaud, J.; et al. Biosafety of Mesoporous Silica Nanoparticles. Biomimetics 2018, 3, 22. https://doi.org/10.3390/biomimetics3030022
Rascol E, Pisani C, Dorandeu C, Nyalosaso JL, Charnay C, Daurat M, Da Silva A, Devoisselle J-M, Gaillard J-C, Armengaud J, et al. Biosafety of Mesoporous Silica Nanoparticles. Biomimetics. 2018; 3(3):22. https://doi.org/10.3390/biomimetics3030022
Chicago/Turabian StyleRascol, Estelle, Cédric Pisani, Christophe Dorandeu, Jeff L. Nyalosaso, Clarence Charnay, Morgane Daurat, Afitz Da Silva, Jean-Marie Devoisselle, Jean-Charles Gaillard, Jean Armengaud, and et al. 2018. "Biosafety of Mesoporous Silica Nanoparticles" Biomimetics 3, no. 3: 22. https://doi.org/10.3390/biomimetics3030022
APA StyleRascol, E., Pisani, C., Dorandeu, C., Nyalosaso, J. L., Charnay, C., Daurat, M., Da Silva, A., Devoisselle, J.-M., Gaillard, J.-C., Armengaud, J., Prat, O., Maynadier, M., Gary-Bobo, M., Garcia, M., Chopineau, J., & Guari, Y. (2018). Biosafety of Mesoporous Silica Nanoparticles. Biomimetics, 3(3), 22. https://doi.org/10.3390/biomimetics3030022






