Genetic and Epigenetic Factors in Risk and Susceptibility for Childhood Asthma
Abstract
1. Introduction
2. Genome-Wide Association Studies (GWAS) on Childhood Asthma
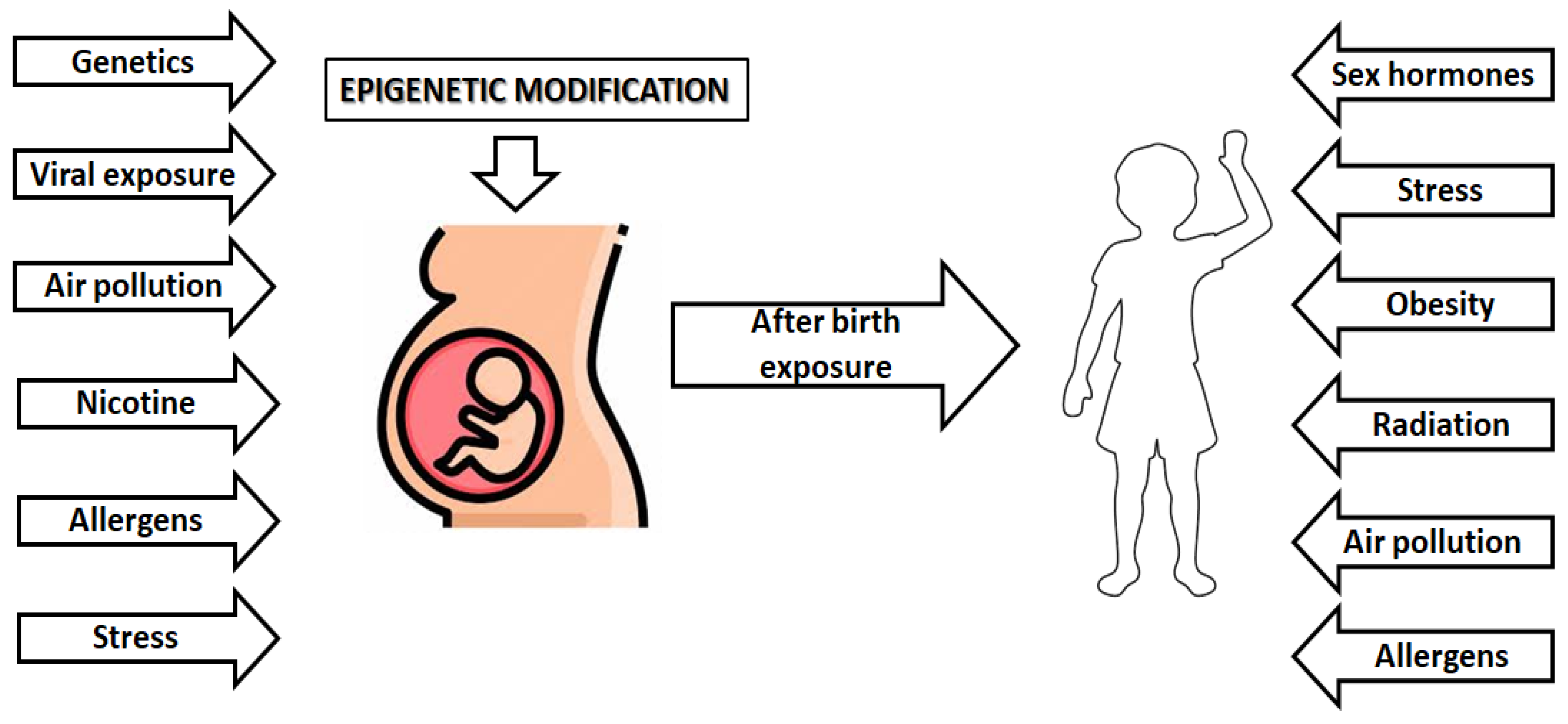
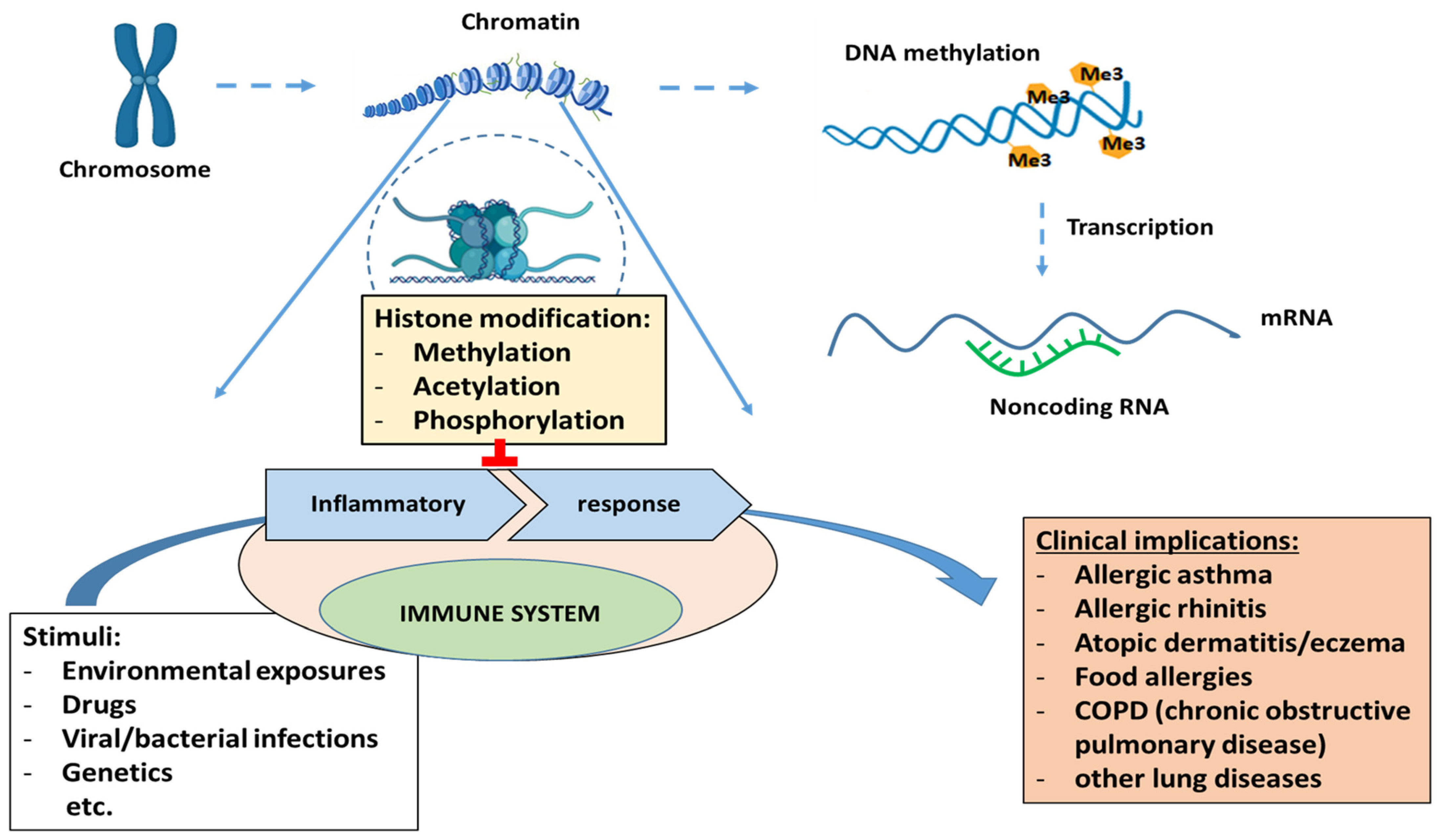
3. Epigenetic and Childhood Asthma Development
3.1. DNA Methylation and Studies of DNA Methylation and Childhood Asthma
| Gene/Candidate-Gene * | Ancestry | Tissue | Ref. |
|---|---|---|---|
| LMAN2; STX3; LPIN1; DICER1; SLC25A25; | European | Whole blood | Xu et al., 2018 [101] |
| ACOT7, EPX, GJA4 and METTL1; | American African | Nasal brush epithelial cells | Yang et al., 2018 [100] |
| ZFPM1; AP2A2; IL5RA; | European | Peripheral blood | Arathimos et al., 2017 [103] |
| CDHR3; CDH26; FBXL7; | Hispanic/Latino | Nasal brushes cells | Forno et al., 2018 [104] |
| EVL; NTRK1; SLC9A3; ACOT7; | Mixed ancestry | Nasal swab cells | Cardenas et al., 2019 [105] |
| CLNS1A, Mir_548; SUB1, LOC100129858; RUNX1; GPATCH2; WDR20; IL5RA; ACOT7; KCNH2; | -13 European cohorts; -3 mixed ancestry cohorts -1 African/American cohort | Whole blood | Reese et al., 2019 [106] |
| ORMDL3; CCL26 | African American and European American | Bronchial airway epithelial cells | Nicodemus-Johnson et al., 2016 [107] |
| SMAD3; DDO/METTL24 | European | Whole blood | Lund et al., 2018 [108] |
| CCL5, IL2RA, TBX21, FCER2, TGFB1, LIF, ADAM17, NHEJ1, AHR, PGDR, PI3K, GNA12 etc. | American cohort | Peripheral blood | Rastogi et al., 2013 [109] |
| ARG1, ARG2, IL6, iNOS | non-Hispanic white and Hispanic white ethnicity | Buccal cells/Nasal epithelium | Breton et al., 2011 [110] |
| KRT5, CRIP1, STAT5A | European | Bronchial epithelium | Stefanowicz et al., 2012 [111] |
3.2. Histone Modification and Studies of Histone Modification and Asthma
3.3. Non-Coding RNAs and Studies of Non-Coding RNA and Asthma
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Global Asthma Report 2018, Global Asthma Network. Available online: http://www.globalasthmanetwork.org (accessed on 20 July 2022).
- Rhodes, L.; Moorman, J.E.; Redd, S.C. Sex Differences in Asthma Prevalence and Other Disease Characteristics in Eight States. J. Asthma 2005, 42, 777–782. [Google Scholar] [CrossRef] [PubMed]
- Morales, E.; Duffy, D. Genetics and Gene-Environment Interactions in Childhood and Adult Onset Asthma. Front. Pediatr. 2019, 7, 499. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Jia, Q.; Jahani, P.S.; Hurrell, B.P.; Pan, C.; Huang, P.; Gukasyan, J.; Woodward, N.C.; Eskin, E.; Gilliland, F.D.; et al. Genome-wide analysis highlights contribution of immune system pathways to the genetic architecture of asthma. Nat. Commun. 2020, 11, 1776. [Google Scholar] [CrossRef]
- Thomsen, S.F.; Duffy, D.L.; Kyvik, K.O.; Backer, V. Genetic influence on the age at onset of asthma: A twin study. J. Allergy Clin. Immunol. 2010, 126, 626–630. [Google Scholar] [CrossRef] [PubMed]
- Polderman, T.J.C.; Benyamin, B.; de Leeuw, C.A.; Sullivan, P.F.; Van Bochoven, A.; Visscher, P.M.; Posthuma, D. Meta-analysis of the heritability of human traits based on fifty years of twin studies. Nat. Genet. 2015, 47, 702–709. [Google Scholar] [CrossRef]
- Ullemar, V.; Magnusson, P.K.E.; Lundholm, C.; Zettergren, A.; Melén, E.; Lichtenstein, P.; Almqvist, C. Heritability and confirmation of genetic association studies for childhood asthma in twins. Allergy 2016, 71, 230–238. [Google Scholar] [CrossRef]
- Almqvist, C.; Worm, M.; Leynaert, B. Working group of GA2LEN WP 2.5 Gender. Impact of gender on asthma in childhood and adolescence: A GA2LEN review. Allergy 2008, 63, 47–57. [Google Scholar] [CrossRef]
- Beasley, R.; Semprini, A.; Mitchell, E.A. Risk factors for asthma: Is prevention possible? Lancet 2015, 386, 1075–1085. [Google Scholar] [CrossRef]
- West, C.E.; Renz, H.; Jenmalm, M.C.; Kozyrskyj, A.L.; Allen, K.J.; Vuillermin, P.; Prescott, S.L.; in-FLAME Microbiome Interest Group. The gut microbiota and inflammatory noncommunicable diseases: Associations and potentials for gut microbiota therapies. J. Allergy Clin. Immunol. 2015, 135, 3–13. [Google Scholar] [CrossRef]
- Bisgaard, H.; Li, N.; Bonnelykke, K.; Chawes, B.L.K.; Skov, T.; Paludan-Müller, G.; Stokholm, J.; Smith, B.; Krogfelt, K.A. Reduced diversity of the intestinal microbiota during infancy is associated with increased risk of allergic disease at school age. J. Allergy Clin. Immunol. 2011, 128, 646–652.e5. [Google Scholar] [CrossRef]
- Hylkema, M.N.; Blacquière, M.J. Intrauterine Effects of Maternal Smoking on Sensitization, Asthma, and Chronic Obstructive Pulmonary Disease. Proc. Am. Thorac. Soc. 2009, 6, 660–662. [Google Scholar] [CrossRef]
- Gehring, U.; Gruzieva, O.; Agius, R.M.; Beelen, R.; Custovic, A.; Cyrys, J.; Eeftens, M.; Flexeder, C.; Fuertes, E.; Heinrich, J.; et al. Air Pollution Exposure and Lung Function in Children: The ESCAPE Project. Environ. Health Perspect. 2013, 121, 1357–1364. [Google Scholar] [CrossRef]
- Bønnelykke, K.; Ober, C. Leveraging gene-environment interactions and endotypes for asthma gene discovery. J. Allergy Clin. Immunol. 2016, 137, 667–679. [Google Scholar] [CrossRef] [PubMed]
- Gasparyan, A.Y.; Ayvazyan, L.; Blackmore, H.; Kitas, G.D. Writing a narrative biomedical review: Considerations for authors, peer reviewers, and editors. Rheumatol. Int. 2011, 31, 1409–1417. [Google Scholar] [CrossRef] [PubMed]
- Banks-Schlegle, S. A genome-wide search for asthma susceptibility loci in ethnically diverse populations. Nat. Genet. 1997, 15, 389–392. [Google Scholar] [CrossRef]
- Moffatt, M.; Kabesch, M.; Liang, L.; Dixon, A.L.; Strachan, D.; Heath, S.; Depner, M.; von Berg, A.; Bufe, A.; Rietschel, E.; et al. Genetic variants regulating ORMDL3 expression contribute to the risk of childhood asthma. Nature 2007, 448, 470–473. [Google Scholar] [CrossRef] [PubMed]
- Sleiman, P.M.; Flory, J.; Imielinski, M.; Bradfield, J.P.; Annaiah, K.; Willis-Owen, S.A.; Wang, K.; Rafaels, N.M.; Michel, S.; Bonnelykke, K.; et al. Variants of DENND1B associated with asthma in children. N. Engl. J. Med. 2010, 362, 36–44. [Google Scholar] [CrossRef]
- Allen, M.; Heinzmann, A.; Noguchi, E.; Abecasis, G.; Broxholme, J.; Ponting, C.; Bhattacharyya, S.; Tinsley, J.; Zhang, Y.; Holt, R.; et al. Positional cloning of a novel gene influencing asthma from Chromosome 2q14. Nat. Genet. 2003, 35, 258–263. [Google Scholar] [CrossRef]
- Mathias, R.A.; Grant, A.V.; Rafaels, N.; Hand, T.; Gao, L.; Vergara, C.; Tsai, Y.J.; Yang, M.; Campbell, M.; Foster, C.; et al. A genome-wide association study on African-ancestry populations for asthma. J. Allergy Clin. Immunol. 2010, 125, 336–346.e4. [Google Scholar] [CrossRef]
- Ober, C.; Tan, Z.; Sun, Y.; Possick, J.D.; Pan, L.; Nicolae, R.; Radford, S.; Parry, R.R.; Heinzmann, A.; Deichmann, K.A.; et al. Effect of variation in CHI3L1 on serum YKL-40 level, risk of asthma, and lung function. N. Engl. J. Med. 2008, 358, 1682–1691. [Google Scholar] [CrossRef]
- Chen, F.; An, Y.; Wang, J. CHI3L1 is correlated with childhood asthma. Int. J. Clin. Exp. Pathol. 2017, 10, 10559–10564. [Google Scholar]
- Demenais, F.; Margaritte-Jeannin, P.; Barnes, K.C.; Cookson, W.O.C.; Altmüller, J.; Ang, W.; Barr, R.G.; Beaty, T.H.; Becker, A.B.; Beilby, J.; et al. Multiancestry association study identifies new asthma risk loci that colocalize with immune-cell enhancer marks. Nat. Genet. 2017, 50, 42–53. [Google Scholar] [CrossRef] [PubMed]
- Lasky-Su, J.; Himes, B.E.; Raby, B.A.; Klanderman, B.J.; Sylvia, J.S.; Lange, C.; Melen, E.; Martinez, F.D.; Israel, E.; Gauderman, J.; et al. HLA-DQ strikes again: Genome-wide association study further confirmsHLA-DQin the diagnosis of asthma among adults. Clin. Exp. Allergy 2012, 42, 1724–1733. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, M.A.; Vonk, J.M.; Baurecht, H.; Marenholz, I.; Tian, C.; Hoffman, J.D.; Helmer, Q.; Tillander, A.; Ullemar, V.; van Dongen, J.; et al. Shared genetic origin of asthma, hay fever and eczema elucidates allergic disease biology. Nat. Genet. 2017, 49, 1752–1757. [Google Scholar] [CrossRef] [PubMed]
- Pividori, M.; Schoettler, N.; Nicolae, D.L.; Ober, C.; Im, H.K. Shared and distinct genetic risk factors for childhood-onset and adult-onset asthma: Genome-wide and transcriptome-wide studies. Lancet Respir. Med. 2019, 7, 509–522. [Google Scholar] [CrossRef]
- Ferreira, M.A.; Mathur, R.; Vonk, J.M.; Szwajda, A.; Brumpton, B.; Granell, R.; Brew, B.K.; Ullemar, V.; Lu, Y.; Jiang, Y.; et al. Genetic Architectures of Childhood- and Adult-Onset Asthma Are Partly Distinct. Am. J. Hum. Genet. 2019, 104, 665–684. [Google Scholar] [CrossRef]
- Hayden, L.P. on behalf of the COPDGene Investigators; Cho, M.H.; Raby, B.A.; Beaty, T.H.; Silverman, E.K.; Hersh, C.P. Childhood asthma is associated with COPD and known asthma variants in COPDGene: A genome-wide association study. Respir. Res. 2018, 19, 209. [Google Scholar] [CrossRef]
- Moffatt, M.F.; Gut, I.G.; Demenais, F.; Strachan, D.P.; Bouzigon, E.; Heath, S.; von Mutius, E.; Farrall, M.; Lathrop, M.; Cookson, W.O.C.M.; et al. A large-scale, consortium-based genome-wide association study of asthma. N. Engl. J. Med. 2010, 363, 1211–1221. [Google Scholar] [CrossRef]
- Li, X.; Howard, T.D.; Zheng, S.L.; Haselkorn, T.; Peters, S.P.; Meyers, D.A.; Bleecker, E.R. Genome-wide association study of asthma identifies RAD50-IL13 and HLA-DR/DQ regions. J. Allergy Clin. Immunol. 2010, 125, 328–335.e11. [Google Scholar] [CrossRef]
- Bønnelykke, K.; Sleiman, P.; Nielsen, K.; Kreiner-Møller, E.; Mercader, J.M.; Belgrave, D.; Dekker, H.T.D.; Husby, A.; Sevelsted, A.; Faura-Tellez, G.; et al. A genome-wide association study identifies CDHR3 as a susceptibility locus for early childhood asthma with severe exacerbations. Nat. Genet. 2013, 46, 51–55. [Google Scholar] [CrossRef]
- Vicente, C.T.; Revez, J.A.; Ferreira, M.A.R. Lessons from ten years of genome-wide association studies of asthma. Clin. Transl. Immunol. 2017, 6, e165. [Google Scholar] [CrossRef]
- Hernandez-Pacheco, N.; Pino-Yanes, M.; Flores, C. Genomic Predictors of Asthma Phenotypes and Treatment Response. Front. Pediatr. 2019, 7, 6. [Google Scholar] [CrossRef] [PubMed]
- Eliasen, A.U.; Pedersen, C.E.T.; Rasmussen, M.A.; Wang, N.; Soverini, M.; Fritz, A.; Stokholm, J.; Chawes, B.L.; Morin, A.; Bork-Jensen, J.; et al. Genome-wide study of early and severe childhood asthma identifies interaction between CDHR3 and GSDMB. J. Allergy Clin. Immunol. 2022, 150, 622–630. [Google Scholar] [CrossRef]
- Thomsen, S.F. Genetics of asthma: An introduction for the clinician. Eur. Clin. Respir. J. 2015, 2, 24643. [Google Scholar] [CrossRef] [PubMed]
- MacArthur, J.; Bowler, E.; Cerezo, M.; Gil, L.; Hall, P.; Hastings, E.; Junkins, H.; McMahon, A.; Milano, A.; Morales, J.; et al. The new NHGRI-EBI Catalog of published genome-wide association studies (GWAS Catalog). Nucleic Acids Res. 2016, 45, D896–D901. [Google Scholar] [CrossRef]
- Schieck, M.; Schouten, J.P.; Michel, S.; Suttner, K.; Toncheva, A.A.; Gaertner, V.D.; Illig, T.; Lipinski, S.; Franke, A.; Klintschar, M.; et al. Doublesex and mab-3 related transcription factor 1 (DMRT1) is a sex-specific genetic determinant of childhood-onset asthma and is expressed in testis and macrophages. J. Allergy Clin. Immunol. 2016, 138, 421–431. [Google Scholar] [CrossRef] [PubMed]
- Gref, A.; Merid, S.K.; Gruzieva, O.; Ballereau, S.; Becker, A.; Bellander, T.; Bergström, A.; Bossé, Y.; Bottai, M.; Chan-Yeung, M.; et al. Genome-Wide Interaction Analysis of Air Pollution Exposure and Childhood Asthma with Functional Follow-up. Am. J. Respir. Crit. Care Med. 2017, 195, 1373–1383. [Google Scholar] [CrossRef]
- Marques, C.R.; Costa, G.N.; da Silva, T.M.; Oliveira, P.; Cruz, A.A.; Alcantara-Neves, N.M.; Fiaccone, R.L.; Horta, B.L.; Hartwig, F.P.; Burchard, E.G.; et al. Suggestive association between variants in IL1RAPL and asthma symptoms in Latin American children. Eur. J. Hum. Genet. 2017, 25, 439–445. [Google Scholar] [CrossRef]
- Spear, M.L.; Hu, D.; Pino-Yanes, M.; Huntsman, S.; Eng, C.; Levin, A.M.; Ortega, V.E.; White, M.J.; McGarry, M.E.; Thakur, N.; et al. A genome-wide association and admixture mapping study of bronchodilator drug response in African Americans with asthma. Pharm. J. 2018, 19, 249–259. [Google Scholar] [CrossRef]
- Yatagai, Y.; Hirota, T.; Sakamoto, T.; Yamada, H.; Masuko, H.; Kaneko, Y.; Iijima, H.; Naito, T.; Noguchi, E.; Tamari, M.; et al. Variants near the HLA complex group 22 gene (HCG22) confer increased susceptibility to late-onset asthma in Japanese populations. J. Allergy Clin. Immunol. 2016, 138, 281–283.e13. [Google Scholar] [CrossRef]
- Almoguera, B.; Vazquez, L.; Mentch, F.; Connolly, J.; Pacheco, J.; Sundaresan, A.S.; Peissig, P.L.; Linneman, J.G.; McCarty, C.; Crosslin, D.; et al. Identification of Four Novel Loci in Asthma in European American and African American Populations. Am. J. Respir. Crit. Care Med. 2017, 195, 456–463. [Google Scholar] [CrossRef]
- Condreay, L.; Chiano, M.; Ortega, H.; Buchan, N.; Harris, E.; Bleecker, E.R.; Thompson, P.J.; Humbert, M.; Gibson, P.; Yancey, S.; et al. No genetic association detected with mepolizumab efficacy in severe asthma. Respir. Med. 2017, 132, 178–180. [Google Scholar] [CrossRef] [PubMed]
- Mosteller, M.; Hosking, L.; Murphy, K.; Shen, J.; Song, K.; Nelson, M.; Ghosh, S. No evidence of large genetic effects on steroid response in asthma patients. J. Allergy Clin. Immunol. 2017, 139, 797–803.e7. [Google Scholar] [CrossRef] [PubMed]
- Vonk, J.M.; Scholtens, S.; Postma, D.S.; Moffatt, M.F.; Jarvis, D.; Ramasamy, A.; Wjst, M.; Omenaas, E.R.; Bouzigon, E.; Demenais, F.; et al. Adult onset asthma and interaction between genes and active tobacco smoking: The GABRIEL consortium. PLoS ONE 2017, 12, e0172716. [Google Scholar] [CrossRef] [PubMed]
- Murk, W.; DeWan, A.T. Genome-wide search identifies a gene-gene interaction between 20p13 and 2q14 in asthma. BMC Genet. 2016, 17, 102. [Google Scholar] [CrossRef]
- Nieuwenhuis, M.A.; Siedlinski, M.; van den Berge, M.; Granell, R.; Li, X.; Niens, M.; van der Vlies, P.; Altmüller, J.; Nürnberg, P.; Kerkhof, M.; et al. Combining genome-wide association study and lung eQTL analysis provides evidence for novel genes associated with asthma. Allergy 2016, 71, 1712–1720. [Google Scholar] [CrossRef]
- Yan, Q.; Brehm, J.; Pino-Yanes, M.; Forno, E.; Lin, J.; Oh, S.S.; Acosta-Perez, E.; Laurie, C.C.; Cloutier, M.M.; Raby, B.A.; et al. A meta-analysis of genome-wide association studies of asthma in Puerto Ricans. Eur. Respir. J. 2017, 49, 1601505. [Google Scholar] [CrossRef]
- Burchard, E.G.; Oh, S.S.; Foreman, M.G.; Celedon, J.C. Moving toward true inclusion of racial/ethnic minorities in federally funded studies. A key step for achieving respiratory health equality in the United States. Am. J. Respir. Crit. Care Med. 2015, 191, 514–521. [Google Scholar] [CrossRef]
- Popejoy, A.B.; Fullerton, S.M. Genomics is failing on diversity. Nature 2016, 538, 161–164. [Google Scholar] [CrossRef]
- Kim, K.W.; Kim, D.Y.; Yoon, D.; Kim, K.; Jang, H.; Schoettler, N.; Kim, E.G.; Na Kim, M.; Hong, J.Y.; Lee, J.; et al. Genome-wide association study identifies TNFSF15 associated with childhood asthma. Allergy 2021, 77, 218–229. [Google Scholar] [CrossRef]
- Israel, E.; Drazen, J.M.; Liggett, S.B.; Boushey, H.A.; Cherniack, R.M.; Chinchilli, V.M.; Cooper, D.M.; Fahy, J.V.; Fish, J.E.; Ford, J.G.; et al. The effect of polymorphisms of the beta(2)-adrenergic receptor on the response to regular use of albuterol in asthma. Am. J. Respir. Crit. Care Med. 2000, 162, 75–80. [Google Scholar] [CrossRef]
- Litonjua, A.; Gong, L.; Duan, Q.L.; Shin, J.; Moore, M.J.; Weiss, S.T.; Johnson, J.A.; Klein, T.E.; Altman, R.B. Very important pharmacogene summary ADRB2. Pharm. Genom. 2010, 20, 64–69. [Google Scholar] [CrossRef]
- Sood, N.; Connolly, J.J.; Mentch, F.D.; Vazquez, L.; Sleiman, P.M.; Hysinger, E.B.; Hakonarson, H. Leveraging electronic health records to assess the role of ADRB2 single nucleotide polymorphisms in predicting exacerbation frequency in asthma patients. Pharm. Genom. 2018, 28, 256–259. [Google Scholar] [CrossRef] [PubMed]
- Scaparrotta, A.; Franzago, M.; Marcovecchio, M.L.; Di Pillo, S.; Chiarelli, F.; Mohn, A.; Stuppia, L. Role of THRB, ARG1, and ADRB2 Genetic Variants on Bronchodilators Response in Asthmatic Children. J. Aerosol Med. Pulm. Drug Deliv. 2019, 32, 164–173. [Google Scholar] [CrossRef]
- Burchard, E.G.; Silverman, E.K.; Rosenwasser, L.J.; Borish, L.; Yandava, C.; Pillari, A.; Weiss, S.T.; Hasday, J.; Lilly, C.M.; Ford, J.G.; et al. Association Between a Sequence Variant in the IL-4 Gene Promoter and FEV1 in Asthma. Am. J. Respir. Crit. Care Med. 1999, 160, 919–922. [Google Scholar] [CrossRef] [PubMed]
- Drazen, J.M.; Yandava, C.N.; Dubé, L.; Szczerback, N.; Hippensteel, R.; Pillari, A.; Israel, E.; Schork, N.; Silverman, E.S.; Katz, D.A.; et al. Pharmacogenetic association between ALOX5 promoter genotype and the response to anti-asthma treatment. Nat. Genet. 1999, 22, 168–170. [Google Scholar] [CrossRef]
- Mougey, E.; Lang, J.E.; Allayee, H.; Teague, W.G.; Dozor, A.J.; Wise, R.A.; Lima, J.J. ALOX5Polymorphism associates with increased leukotriene production and reduced lung function and asthma control in children with poorly controlled asthma. Clin. Exp. Allergy 2012, 43, 512–520. [Google Scholar] [CrossRef] [PubMed]
- Tantisira, K.G.; Lake, S.; Silverman, E.S.; Palmer, L.; Lazarus, R.; Silverman, E.K.; Liggett, S.B.; Gelfand, E.W.; Rosenwasser, L.J.; Richter, B.; et al. Corticosteroid pharmacogenetics: Association of sequence variants in CRHR1 with improved lung function in asthmatics treated with inhaled corticosteroids. Hum. Mol. Genet. 2004, 13, 1353–1359. [Google Scholar] [CrossRef] [PubMed]
- Choudhry, S.; Avila, P.C.; Nazario, S.; Ung, N.; Kho, J.; Rodriguez-Santana, J.R.; Casal, J.; Tsai, H.-J.; Torres, A.; Ziv, E.; et al. CD14 Tobacco Gene–Environment Interaction Modifies Asthma Severity and Immunoglobulin E Levels in Latinos with Asthma. Am. J. Respir. Crit. Care Med. 2005, 172, 173–182. [Google Scholar] [CrossRef]
- Martin, A.C.; Laing, I.A.; Khoo, S.K.; Zhang, G.; Rueter, K.; Teoh, L.; Taheri, S.; Hayden, C.M.; Geelhoed, G.C.; Goldblatt, J.; et al. Faculty Opinions recommendation of Acute asthma in children: Relationships among CD14 and CC16 genotypes, plasma levels, and severity. Am. J. Respir. Crit. Care Med. 2006, 173, 617–622. [Google Scholar] [CrossRef]
- Foley, S.C.; Mogas, A.K.; Olivenstein, R.; Fiset, P.O.; Chakir, J.; Bourbeau, J.; Ernst, P.; Lemière, C.; Martin, J.G.; Hamid, Q. Increased expression of ADAM33 and ADAM8 with disease progression in asthma. J. Allergy Clin. Immunol. 2007, 119, 863–871. [Google Scholar] [CrossRef] [PubMed]
- Tavendale, R.; Macgregor, D.F.; Mukhopadhyay, S.; Palmer, C.N. A polymorphism controlling ORMDL3 expression is associated with asthma that is poorly controlled by current medications. J. Allergy Clin. Immunol. 2008, 121, 860–863. [Google Scholar] [CrossRef] [PubMed]
- Bisgaard, H.; Bønnelykke, K.; Sleiman, P.M.A.; Brasholt, M.; Chawes, B.; Kreiner-Møller, E.; Stage, M.; Kim, C.; Tavendale, R.; Baty, F.; et al. Chromosome 17q21 Gene Variants Are Associated with Asthma and Exacerbations but Not Atopy in Early Childhood. Am. J. Respir. Crit. Care Med. 2009, 179, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Berce, V.; Kozmus, C.; Potočnik, U. Association among ORMDL3 gene expression, 17q21 polymorphism and response to treatment with inhaled corticosteroids in children with asthma. Pharm. J. 2012, 13, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Farzan, N.; Vijverberg, S.J.; Hernandez-Pacheco, N.; Bel, E.H.D.; Berce, V.; Bønnelykke, K.; Bisgaard, H.; Burchard, E.G.; Canino, G.; Celedón, J.C.; et al. 17q21 variant increases the risk of exacerbations in asthmatic children despite inhaled corticosteroids use. Allergy 2018, 73, 2083–2088. [Google Scholar] [CrossRef]
- Tantisira, K.G.; Lasky-Su, J.; Harada, M.; Murphy, A.; Litonjua, A.A.; Himes, B.E.; Lange, C.; Lazarus, R.; Sylvia, J.; Klanderman, B.; et al. Genome-wide association between GLCCI1 and response to glucocorticoid therapy in asthma. N. Engl. J. Med. 2011, 365, 1173–1183. [Google Scholar] [CrossRef]
- Himes, B.E.; Jiang, X.; Hu, R.; Wu, A.C.; Lasky-Su, J.A.; Klanderman, B.J.; Ziniti, J.; Senter-Sylvia, J.; Lima, J.J.; Irvin, C.G.; et al. Genome-Wide Association Analysis in Asthma Subjects Identifies SPATS2L as a Novel Bronchodilator Response Gene. PLOS Genet. 2012, 8, e1002824. [Google Scholar] [CrossRef]
- Israel, E.; Lasky-Su, J.; Markezich, A.; Damask, A.; Szefler, S.J.; Schuemann, B.; Klanderman, B.; Sylvia, J.; Kazani, S.; Wu, R.; et al. Genome-Wide Association Study of Short-Acting β2-Agonists. A Novel Genome-Wide Significant Locus on Chromosome 2 near ASB3. Am. J. Respir. Crit. Care Med. 2015, 191, 530–537. [Google Scholar] [CrossRef]
- Dijk, F.N.; Vijverberg, S.J.; Hernandez-Pacheco, N.; Repnik, K.; Karimi, L.; Mitratza, M.; Farzan, N.; Nawijn, M.C.; Burchard, E.G.; Engelkes, M.; et al. IL1RL1 gene variations are associated with asthma exacerbations in children and adolescents using inhaled corticosteroids. Allergy 2019, 75, 984–989. [Google Scholar] [CrossRef]
- Wan, Z.; Tang, Y.; Song, Q.; Zhang, J.; Xie, W.; He, Y.; Huang, R.; Zheng, X.; Liu, C.; Liu, J. Gene polymorphisms in VEGFA and COL2A1 are associated with response to inhaled corticosteroids in children with asthma. Pharmacogenomics 2019, 20, 947–955. [Google Scholar] [CrossRef]
- Dragicevic, S.; Kosnik, M.; Rankov, A.D.; Rijavec, M.; Milosevic, K.; Korosec, P.; Kavalar, M.S.; Nikolic, A. The Variants in the 3′ Untranslated Region of the Matrix Metalloproteinase 9 Gene as Modulators of Treatment Outcome in Children with Asthma. Lung 2018, 196, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Levin, A.M.; Gui, H.; Hernandez-Pacheco, N.; Yang, M.; Xiao, S.; Yang, J.J.; Hochstadt, S.; Barczak, A.J.; Eckalbar, W.L.; Rynkowski, D.; et al. Integrative approach identifies corticosteroid response variant in diverse populations with asthma. J. Allergy Clin. Immunol. 2018, 143, 1791–1802. [Google Scholar] [CrossRef] [PubMed]
- Himes, B.E.; Jiang, X.; Wagner, P.; Hu, R.; Wang, Q.; Klanderman, B.; Whitaker, R.; Duan, Q.; Lasky-Su, J.; Nikolos, C.; et al. RNA-Seq Transcriptome Profiling Identifies CRISPLD2 as a Glucocorticoid Responsive Gene that Modulates Cytokine Function in Airway Smooth Muscle Cells. PLoS ONE 2014, 9, e99625. [Google Scholar] [CrossRef]
- Ramasamy, A.; Curjuric, I.; Coin, L.J.; Kumar, A.; McArdle, W.L.; Imboden, M.; Leynaert, B.; Kogevinas, M.; Schmid-Grendelmeier, P.; Pekkanen, J.; et al. A genome-wide meta-analysis of genetic variants associated with allergic rhinitis and grass sensitization and their interaction with birth order. J. Allergy Clin. Immunol. 2011, 128, 996–1005. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; McDonald, M.-L.N.; Cho, M.H.; Wan, E.S.; Castaldi, P.J.; Hunninghake, G.M.; Marchetti, N.; Lynch, D.A.; Crapo, J.D.; Lomas, D.A.; et al. DNAH5 is associated with total lung capacity in chronic obstructive pulmonary disease. Respir. Res. 2014, 15, 1–10. [Google Scholar] [CrossRef]
- Mak, A.C.Y.; White, M.J.; Eckalbar, W.L.; Szpiech, Z.A.; Oh, S.S.; Pino-Yanes, M.; Hu, D.; Goddard, P.; Huntsman, S.; Galanter, J.; et al. Whole-Genome Sequencing of Pharmacogenetic Drug Response in Racially Diverse Children with Asthma. Am. J. Respir. Crit. Care Med. 2018, 197, 1552–1564. [Google Scholar] [CrossRef]
- Perez-Garcia, J.; Espuela-Ortiz, A.; Lorenzo-Diaz, F.; Pino-Yanes, M. Pharmacogenetics of Pediatric Asthma: Current Perspectives. Pharm. Pers. Med. 2020, 13, 89–103. [Google Scholar] [CrossRef]
- Lutz, S.M.; Cho, M.H.; Young, K.; Hersh, C.P.; Castaldi, P.J.; McDonald, M.-L.; Regan, E.; Mattheisen, M.; DeMeo, D.L.; Parker, M.; et al. A genome-wide association study identifies risk loci for spirometric measures among smokers of European and African ancestry. BMC Genet. 2015, 16, 138. [Google Scholar] [CrossRef]
- Wain, L.V.; Shrine, N.; Artigas, M.S.; Erzurumluoglu, A.M.; Noyvert, B.; Bossini-Castillo, L.; Obeidat, M.; Henry, A.P.; Portelli, M.A.; Hall, R.J.; et al. Genome-wide association analyses for lung function and chronic obstructive pulmonary disease identify new loci and potential druggable targets. Nat. Genet. 2017, 49, 416–425. [Google Scholar] [CrossRef]
- Karimi, L.; Vijverberg, S.J.; Farzan, N.; Ghanbari, M.; Verhamme, K.M.; der Zee, A.H.M. FCER2 T2206C variant associated with FENO levels in asthmatic children using inhaled corticosteroids: The PACMAN study. Clin. Exp. Allergy 2019, 49, 1429–1436. [Google Scholar] [CrossRef]
- Balantic, M.; Rijavec, M.; Kavalar, M.S.; Suskovic, S.; Silar, M.; Kosnik, M.; Korosec, P. Asthma Treatment Outcome in Children Is Associated with Vascular Endothelial Growth Factor A (VEGFA) Polymorphisms. Mol. Diagn. Ther. 2012, 16, 173–180. [Google Scholar] [CrossRef] [PubMed]
- DeVries, A.; Vercelli, D. Epigenetic Mechanisms in Asthma. Ann. Am. Thorac. Soc. 2016, 13, S48–S50. [Google Scholar] [CrossRef] [PubMed]
- Kelsey, G.; Stegle, O.; Reik, W. Single-cell epigenomics: Recording the past and predicting the future. Science 2017, 358, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Bae, D.-J.; Jun, J.A.; Chang, H.S.; Park, J.S.; Park, C.-S. Epigenetic Changes in Asthma: Role of DNA CpG Methylation. Tuberc. Respir. Dis. 2020, 83, 1–13. [Google Scholar] [CrossRef] [PubMed]
- WHO Global NCD Action Plan 2023-2030 and the United Nations 2030 Agenda for Sustainable Development. Available online: https://www.who.int/teams/noncommunicable-diseases/governance/roadmap (accessed on 4 June 2023).
- Bibi, H.; Shoseyov, D.; Feigenbaum, D.; Genis, M.; Friger, M.; Peled, R.; Sharff, S. The relationship between asthma and obesity in children: Is it real or a case of over diagnosis? J. Asthma 2004, 41, 403–410. [Google Scholar] [CrossRef]
- Lang, J.E.; Feng, H.; Lima, J.J. Body Mass Index-Percentile and Diagnostic Accuracy of Childhood Asthma. J. Asthma 2009, 46, 291–299. [Google Scholar] [CrossRef]
- Di Genova, L.; Penta, L.; Biscarini, A.; Di Cara, G.; Esposito, S. Children with Obesity and Asthma: Which Are the Best Options for Their Management? Nutrients 2018, 10, 1634. [Google Scholar] [CrossRef]
- Belamarich, P.F.; Luder, E.; Kattan, M.; Mitchell, H.; Islam, S.; Lynn, H.; Crain, E.F. Do obese inner-city children with asthma have more symptoms than nonobese children with asthma? Pediatrics 2000, 106, 1436–1441. [Google Scholar] [CrossRef]
- Sithole, F.; Douwes, J.; Burstyn, I.; Veugelers, P. Body Mass Index and Childhood Asthma: A Linear Association? J. Asthma 2008, 45, 473–477. [Google Scholar] [CrossRef]
- Ginde, A.A.; Santillan, A.A.; Clark, S.; Camargo, C.A., Jr. Body mass index and acute asthma severity among children presenting to the emergency department. Pediatr. Allergy Immunol. 2009, 21, 480–488. [Google Scholar] [CrossRef]
- Lovinsky-Desir, S.; Miller, R.L. Epigenetics, Asthma, and Allergic Diseases: A Review of the Latest Advancements. Curr. Allergy Asthma Rep. 2012, 12, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.A. Functions of DNA methylation: Islands, start sites, gene bodies and beyond. Nat. Rev. Genet. 2012, 13, 484–492. [Google Scholar] [CrossRef] [PubMed]
- Thibeault, A.-A.H.; Laprise, C. Cell-Specific DNA Methylation Signatures in Asthma. Genes 2019, 10, 932. [Google Scholar] [CrossRef] [PubMed]
- Gruzieva, O.; Merid, S.K.; Melén, E. An update on epigenetics and childhood respiratory diseases. Paediatr. Respir. Rev. 2014, 15, 348–354. [Google Scholar] [CrossRef]
- Kabesch, M.; Tost, J. Recent findings in the genetics and epigenetics of asthma and allergy. Semin. Immunopathol. 2020, 42, 43–60. [Google Scholar] [CrossRef]
- Brook, P.O.; Perry, M.M.; Adcock, I.M.; Durham, A.L. Epigenome-modifying tools in asthma. Epigenomics 2015, 7, 1017–1032. [Google Scholar] [CrossRef]
- Ntontsi, P.; Photiades, A.; Zervas, E.; Xanthou, G.; Samitas, K. Genetics and Epigenetics in Asthma. Int. J. Mol. Sci. 2021, 22, 2412. [Google Scholar] [CrossRef]
- Yang, I.V.; Pedersen, B.S.; Liu, A.H.; O’Connor, G.T.; Pillai, D.; Kattan, M.; Misiak, R.T.; Gruchalla, R.; Szefler, S.J.; Khurana Hershey, G.; et al. The nasal methylome and childhood atopic asthma. J. Allergy Clin. Immunol. 2017, 139, 1478–1488. [Google Scholar] [CrossRef]
- Xu, C.-J.; Söderhäll, C.; Bustamante, M.; Baïz, N.; Gruzieva, O.; Gehring, U.; Mason, D.; Chatzi, L.; Basterrechea, M.; Llop, S.; et al. DNA methylation in childhood asthma: An epigenome-wide meta-analysis. Lancet Respir. Med. 2018, 6, 379–388. [Google Scholar] [CrossRef]
- Qi, C.; Xu, C.-J.; Koppelman, G.H. The role of epigenetics in the development of childhood asthma. Expert Rev. Clin. Immunol. 2019, 15, 1287–1302. [Google Scholar] [CrossRef]
- Arathimos, R.; Suderman, M.; Sharp, G.C.; Burrows, K.; Granell, R.; Tilling, K.; Gaunt, T.R.; Henderson, J.; Ring, S.; Richmond, R.C.; et al. Epigenome-wide association study of asthma and wheeze in childhood and adolescence. Clin. Epigenetics 2017, 9, 112. [Google Scholar] [CrossRef] [PubMed]
- Forno, E.; Wang, T.; Qi, C.; Yan, Q.; Xu, C.-J.; Boutaoui, N.; Han, Y.-Y.; Weeks, D.E.; Jiang, Y.; Rosser, F.; et al. DNA methylation in nasal epithelium, atopy, and atopic asthma in children: A genome-wide study. Lancet Respir. Med. 2018, 7, 336–346. [Google Scholar] [CrossRef]
- Cardenas, A.; Sordillo, J.E.; Rifas-Shiman, S.L.; Chung, W.; Liang, L.; Coull, B.A.; Hivert, M.-F.; Lai, P.S.; Forno, E.; Celedón, J.C.; et al. The nasal methylome as a biomarker of asthma and airway inflammation in children. Nat. Commun. 2019, 10, 3095. [Google Scholar] [CrossRef] [PubMed]
- Reese, S.E.; Xu, C.-J.; Dekker, H.T.D.; Lee, M.K.; Sikdar, S.; Ruiz-Arenas, C.; Merid, S.K.; Rezwan, F.I.; Page, C.M.; Ullemar, V.; et al. Epigenome-wide meta-analysis of DNA methylation and childhood asthma. J. Allergy Clin. Immunol. 2018, 143, 2062–2074. [Google Scholar] [CrossRef] [PubMed]
- Nicodemus-Johnson, J.; Myers, R.A.; Sakabe, N.J.; Sobreira, D.R.; Hogarth, D.K.; Naureckas, E.T.; Sperling, A.; Solway, J.; White, S.R.; Nobrega, M.A.; et al. DNA methylation in lung cells is associated with asthma endotypes and genetic risk. J. Clin. Investig. 2016, 1, e90151. [Google Scholar] [CrossRef]
- Lund, R.J.; Osmala, M.; Malonzo, M.; Lukkarinen, M.; Leino, A.; Salmi, J.; Vuorikoski, S.; Turunen, R.; Vuorinen, T.; Akdis, C.; et al. Atopic asthma after rhinovirus-induced wheezing is associated with DNA methylation change in the SMAD3 gene promoter. Allergy 2018, 73, 1735–1740. [Google Scholar] [CrossRef]
- Rastogi, D.; Suzuki, M.; Greally, J.M. Differential epigenome-wide DNA methylation patterns in childhood obesity-associated asthma. Sci. Rep. 2013, 3, srep02164. [Google Scholar] [CrossRef]
- Breton, C.V.; Byun, H.-M.; Wang, X.; Salam, M.T.; Siegmund, K.; Gilliland, F.D. DNA Methylation in the Arginase–Nitric Oxide Synthase Pathway Is Associated with Exhaled Nitric Oxide in Children with Asthma. Am. J. Respir. Crit. Care Med. 2011, 184, 191–197. [Google Scholar] [CrossRef]
- Stefanowicz, D.; Hackett, T.-L.; Garmaroudi, F.S.; Günther, O.; Neumann, S.; Sutanto, E.N.; Ling, K.-M.; Kobor, M.; Kicic, A.; Stick, S.; et al. DNA Methylation Profiles of Airway Epithelial Cells and PBMCs from Healthy, Atopic and Asthmatic Children. PLoS ONE 2012, 7, e44213. [Google Scholar] [CrossRef]
- Salam, M.T. Asthma Epigenetics. Heterog. Asthma 2013, 795, 183–199. [Google Scholar] [CrossRef]
- Kidd, C.D.A.; Thompson, P.J.; Barrett, L.; Baltic, S. Histone Modifications and Asthma. The Interface of the Epigenetic and Genetic Landscapes. Am. J. Respir. Cell Mol. Biol. 2016, 54, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Caramori, G.; Lim, S.; Oates, T.; Chung, K.F.; Barnes, P.J.; Adcock, I.M. Expression and Activity of Histone Deacetylases in Human Asthmatic Airways. Am. J. Respir. Crit. Care Med. 2002, 166, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Su, R.-C.; Becker, A.B.; Kozyrskyj, A.L.; HayGlass, K.T. Epigenetic regulation of established human type 1 versus type 2 cytokine responses. J. Allergy Clin. Immunol. 2008, 121, 57–63.e3. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.-H.; Oh, S.-W.; Kang, M.-S.; Kwon, H.; Oh, G.-T.; Kim, D.-Y. Trichostatin A attenuates airway inflammation in mouse asthma model. Clin. Exp. Allergy 2004, 35, 89–96. [Google Scholar] [CrossRef]
- Yang, S.R.; Wright, J.; Bauter, M.; Seweryniak, K.; Kode, A.; Rahman, I. Sirtuin regulates cigarette smoke-induced proinflammatory mediator release via RelA/p65 NF-κB in macrophages in vitro and in rat lungs in vivo: Implications for chronic inflammation and aging. Am. J. Physiol. Lung Cell. Mol. Physiol. 2007, 292, L567–L576. [Google Scholar] [CrossRef]
- Abbring, S.; Wolf, J.; Ayechu-Muruzabal, V.; Diks, M.A.; Alhamdan, F.; Harb, H.; Renz, H.; Garn, H.; Potaczek, D.P.; Van Esch, B.C.; et al. Raw Cow’s Milk Reduces Allergic Symptoms in a Murine Model for Food Allergy—A Potential Role for Epigenetic Modifications. Nutrients 2019, 11, 1721. [Google Scholar] [CrossRef]
- Ren, Y.; Li, M.; Bai, S.; Kong, L.; Su, X. Identification of histone acetylation in a murine model of allergic asthma by proteomic analysis. Exp. Biol. Med. 2020, 246, 929–939. [Google Scholar] [CrossRef]
- Chen, L.-F.; Fischle, W.; Verdin, E.; Greene, W.C. Duration of Nuclear NF-κB Action Regulated by Reversible Acetylation. Science 2001, 293, 1653–1657. [Google Scholar] [CrossRef]
- Grausenburger, R.; Bilic, I.; Boucheron, N.; Zupkovitz, G.; El-Housseiny, L.; Tschismarov, R.; Zhang, Y.; Rembold, M.; Gaisberger, M.; Hartl, A.; et al. Conditional Deletion of Histone Deacetylase 1 in T Cells Leads to Enhanced Airway Inflammation and Increased Th2 Cytokine Production. J. Immunol. 2010, 185, 3489–3497. [Google Scholar] [CrossRef]
- Wang, Y.; Tian, Y.; Morley, M.P.; Lu, M.M.; DeMayo, F.J.; Olson, E.N.; Morrisey, E.E. Development and Regeneration of Sox2+ Endoderm Progenitors Are Regulated by a HDAC1/2-Bmp4/Rb1 Regulatory Pathway. Dev. Cell 2013, 24, 345–358. [Google Scholar] [CrossRef]
- Seumois, G.; Chavez, L.; Gerasimova, A.; Lienhard, M.; Omran, N.; Kalinke, L.; Vedanayagam, M.; Ganesan, A.P.V.; Chawla, A.; Djukanović, R.; et al. Epigenomic analysis of primary human T cells reveals enhancers associated with TH2 memory cell differentiation and asthma susceptibility. Nat. Immunol. 2014, 15, 777–788. [Google Scholar] [CrossRef] [PubMed]
- Harb, H.; Raedler, D.; Ballenberger, N.; Böck, A.; Kesper, D.A.; Renz, H.; Schaub, B. Childhood allergic asthma is associated with increased IL-13 and FOXP3 histone acetylation. J. Allergy Clin. Immunol. 2015, 136, 200–202. [Google Scholar] [CrossRef] [PubMed]
- Stefanowicz, D.; Lee, J.Y.; Lee, K.; Shaheen, F.; Koo, H.-K.; Booth, S.; Knight, D.A.; Hackett, T.-L. Elevated H3K18 acetylation in airway epithelial cells of asthmatic subjects. Respir. Res. 2015, 16, 95. [Google Scholar] [CrossRef]
- Puddicombe, S.M.; Polosa, R.; Richter, A.; Krishna, M.T.; Howarth, P.H.; Holgate, S.T.; Davies, D.E. Involvement of the epidermal growth factor receptor in epithelial repair in asthma. FASEB J. 2000, 14, 1362–1374. [Google Scholar] [CrossRef]
- Boxall, C.; Holgate, S.T.; Davies, D.E. The contribution of transforming growth factor-beta and epidermal growth factor signalling to airway remodelling in chronic asthma. Eur. Respir. J. 2006, 27, 208–229. [Google Scholar] [CrossRef] [PubMed]
- Amishima, M.; Munakata, M.; Nasuhara, Y.; Sato, A.; Takahashi, T.; Homma, Y.; Kawakami, Y. Expression of Epidermal Growth Factor and Epidermal Growth Factor Receptor Immunoreactivity in the Asthmatic Human Airway. Am. J. Respir. Crit. Care Med. 1998, 157, 1907–1912. [Google Scholar] [CrossRef]
- Mullings, R.E.; Wilson, S.J.; Puddicombe, S.M.; Lordan, J.L.; Bucchieri, F.; Djukanović, R.; Howarth, P.H.; Harper, S.; Holgate, S.T.; Davies, D.E. Signal transducer and activator of transcription 6 (STAT-6) expression and function in asthmatic bronchial epithelium. J. Allergy Clin. Immunol. 2001, 108, 832–838. [Google Scholar] [CrossRef]
- Tomita, K.; Caramori, G.; Ito, K.; Sano, H.; Lim, S.; Oates, T.; Cosio, B.; Chung, K.F.; Tohda, Y.; Barnes, P.J.; et al. STAT6 expression in T cells, alveolar macrophages and bronchial biopsies of normal and asthmatic subjects. J. Inflamm. 2012, 9, 5. [Google Scholar] [CrossRef]
- Cheng, Q.; Shang, Y.; Huang, W.; Zhang, Q.; Li, X.; Zhou, Q. p300 mediates the histone acetylation of ORMDL3 to affect airway inflammation and remodeling in asthma. Int. Immunopharmacol. 2019, 76, 105885. [Google Scholar] [CrossRef]
- Wawrzyniak, P.; Wawrzyniak, M.; Wanke, K.; Sokolowska, M.; Bendelja, K.; Rückert, B.; Globinska, A.; Jakiela, B.; Kast, J.I.; Idzko, M.; et al. Regulation of bronchial epithelial barrier integrity by type 2 cytokines and histone deacetylases in asthmatic patients. J. Allergy Clin. Immunol. 2016, 139, 93–103. [Google Scholar] [CrossRef]
- Harb, H.; Alhamwe, B.A.; Garn, H.; Renz, H.; Potaczek, D.P. Recent developments in epigenetics of pediatric asthma. Curr. Opin. Pediatr. 2016, 28, 754–763. [Google Scholar] [CrossRef] [PubMed]
- García-Serna, A.M.; Martín-Orozco, E.; Hernández-Caselles, T.; Morales, E. Prenatal and Perinatal Environmental Influences Shaping the Neonatal Immune System: A Focus on Asthma and Allergy Origins. Int. J. Environ. Res. Public Health 2021, 18, 3962. [Google Scholar] [CrossRef] [PubMed]
- Kabesch, M. Early origins of asthma (and allergy). Mol. Cell. Pediatr. 2016, 3, 31. [Google Scholar] [CrossRef] [PubMed]
- Potaczek, D.P.; Harb, H.; Michel, S.; Alhamwe, B.A.; Renz, H.; Tost, J. Epigenetics and allergy: From basic mechanisms to clinical applications. Epigenomics 2017, 9, 539–571. [Google Scholar] [CrossRef]
- Brand, S.; Teich, R.; Dicke, T.; Harb, H.; Yildirim, A.O.; Tost, J.; Schneider-Stock, R.; Waterland, R.A.; Bauer, U.M.; von Mutius, E. Epigenetic regulation in mouse offspring as a novel mechanism for transmaternal protection against microbial-induced asthma. J. Allergy Clin. Immunol. 2011, 128, 618–625.e7. [Google Scholar] [CrossRef]
- Harb, H.; Alhamwe, B.A.; Acevedo, N.; Frumento, P.; Johansson, C.; Eick, L.; Papadogiannakis, N.; Alm, J.; Renz, H.; Potaczek, D.P.; et al. Epigenetic Modifications in Placenta are Associated with the Child’s Sensitization to Allergens. BioMed Res. Int. 2019, 2019, 1–11. [Google Scholar] [CrossRef]
- Ambros, V. The functions of animal microRNAs. Nature 2004, 431, 350–355. [Google Scholar] [CrossRef]
- Ariel, D.; Upadhyay, D. The role and regulation of microRNAs in asthma. Curr. Opin. Allergy Clin. Immunol. 2012, 12, 49–52. [Google Scholar] [CrossRef]
- Specjalski, K.; Jassem, E. MicroRNAs: Potential biomarkers and targets of therapy in allergic diseases? Arch. Immunol. Ther. Exp. 2019, 67, 213–223. [Google Scholar] [CrossRef]
- Narożna, B.; Langwiński, W.; Szczepankiewicz, A. Non-coding RNAs in pediatric airway diseases. Genes 2017, 8, 348. [Google Scholar] [CrossRef]
- Feketea, G.; Bocsan, C.I.; Popescu, C.; Gaman, M.; Stanciu, L.A.; Zdrenghea, M.T. A Review of Macrophage MicroRNAs’ Role in Human Asthma. Cells 2019, 8, 420. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.B.; Wang, M.Y.; Zhu, H.Y.; Tang, S.Q.; You, Y.D.; Xie, Y.Q. Overexpression of microRNA-21 and microRNA-126 in the patients of bronchial asthma. Int. J. Clin. Exp. Med. 2014, 7, 1307–1312. [Google Scholar] [PubMed]
- Pua, H.H.; Ansel, K.M. MicroRNA regulation of allergic inflammation and asthma. Curr. Opin. Immunol. 2015, 36, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Elbehidy, R.M.; Youssef, D.; El-Shal, A.S.; Shalaby, S.M.; Sherbiny, H.S.; Sherief, L.M.; E Akeel, N. MicroRNA–21 as a novel biomarker in diagnosis and response to therapy in asthmatic children. Mol. Immunol. 2016, 71, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Sawant, D.V.; Yao, W.; Wright, Z.; Sawyers, C.; Tepper, R.S.; Gupta, S.K.; Kaplan, M.H.; Dent, A.L. Serum MicroRNA-21 as a Biomarker for Allergic Inflammatory Disease in Children. MicroRNA 2015, 4, 36–40. [Google Scholar] [CrossRef]
- Hammad Mahmoud Hammad, R.; Hamed, D.H.E.D.; Eldosoky, M.A.E.R.; Ahmad, A.A.E.S.; Osman, H.M.; Abd Elgalil, H.M.; Mahmoud Hassan, M.M. Plasma microRNA-21, microRNA-146a and IL-13 expression in asthmatic children. Innate Immun. 2018, 24, 171–179. [Google Scholar] [CrossRef]
- Karam, R.A.; Elrahman, D.M.A. Differential expression of miR-155 and Let-7a in the plasma of childhood asthma: Potential biomarkers for diagnosis and severity. Clin. Biochem. 2019, 68, 30–36. [Google Scholar] [CrossRef]
- Johansson, K.; Malmhäll, C.; Ramos-Ramírez, P.; Rådinger, M. MicroRNA-155 is a critical regulator of type 2 innate lymphoid cells and IL-33 signaling in experimental models of allergic airway inflammation. J. Allergy Clin. Immunol. 2017, 139, 1007–1016.e9. [Google Scholar] [CrossRef]
- Kumar, M.; Ahmad, T.; Sharma, A.; Mabalirajan, U.; Kulshreshtha, A.; Agrawal, A.; Ghosh, B. Let-7 microRNA-mediated regulation of IL-13 and allergic airway inflammation. J Allergy Clin. Immunol. 2011, 128, 1077–1085.e10. [Google Scholar] [CrossRef]
- Su, X.W.; Yang, Y.; Lv, M.L.; Li, L.J.; Dong, W.; Liao, M.; Gao, L.B.; Luo, H.B.; Liu, Y.; Cong, R.J.; et al. Association between single-nucleotide polymorphisms in pre-miRNAs and the risk of asthma in a Chinese population. DNA Cell Biol. 2011, 30, 919–923. [Google Scholar] [CrossRef]
- Davis, J.S.; Sun, M.; Kho, A.T.; Moore, K.G.; Sylvia, J.M.; Weiss, S.T.; Lu, Q.; Tantisira, K.G. Circulating microRNAs and association with methacholine PC20 in the Childhood Asthma Management Program (CAMP) cohort. PLoS ONE 2017, 12, e0180329. [Google Scholar] [CrossRef] [PubMed]
- Panganiban, R.P.; Wang, Y.; Howrylak, J.; Chinchilli, V.M.; Craig, T.J.; August, A.; Ishmael, F.T. Circulating microRNAs as biomarkers in patients with allergic rhinitis and asthma. J. Allergy Clin. Immunol. 2016, 137, 1423–1432. [Google Scholar] [CrossRef] [PubMed]
- McGeachie, M.J.; Davis, J.S.; Kho, A.T.; Dahlin, A.; Sordillo, J.E.; Sun, M.; Lu, Q.; Weiss, S.T.; Tantisira, K.G. Asthma remission: Predicting future airways responsiveness using an miRNA network. J. Allergy Clin. Immunol. 2017, 140, 598–600.e8. [Google Scholar] [CrossRef]
- Milger, K.; Götschke, J.; Krause, L.; Nathan, P.; Alessandrini, F.; Tufman, A.; Fischer, R.; Bartel, S.; Theis, F.J.; Behr, J.; et al. Identification of a plasma miRNA biomarker signature for allergic asthma: A translational approach. Allergy 2017, 72, 1962–1971. [Google Scholar] [CrossRef]
- Lu, T.X.; Hartner, J.; Lim, E.J.; Fabry, V.; Mingler, M.K.; Cole, E.T.; Orkin, S.H.; Aronow, B.J.; Rothenberg, M.E. Microrna-21 limits in vivo immune response-mediated activation of the il-12/ifn-gamma pathway, th1 polarization, and the severity of delayed-type hypersensitivity. J. Immunol. 2011, 187, 3362–3373. [Google Scholar] [CrossRef]
- Comer, B.; Camoretti-Mercado, B.; Kogut, P.C.; Halayko, A.J.; Solway, J.; Gerthoffer, W.T. MicroRNA-146a and microRNA-146b expression and anti-inflammatory function in human airway smooth muscle. Am. J. Physiol. Cell. Mol. Physiol. 2014, 307, L727–L734. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Li, J.; Gao, P.; Wang, Q.; Zhang, J. miR-155: A Novel Target in Allergic Asthma. Int. J. Mol. Sci. 2016, 17, 1773. [Google Scholar] [CrossRef]
- Simpson, L.J.; Patel, S.; Bhakta, N.R.; Choy, D.; Brightbill, H.D.; Ren, X.; Wang, Y.; Pua, H.H.; Baumjohann, D.; Montoya, M.M.; et al. A microRNA upregulated in asthma airway T cells promotes TH2 cytokine production. Nat. Immunol. 2014, 15, 1162–1170. [Google Scholar] [CrossRef]
- Haj-Salem, I.; Fakhfakh, R.; Bérubé, J.-C.; Jacques, E.; Plante, S.; Simard, M.J.; Bossé, Y.; Chakir, J. MicroRNA-19a enhances proliferation of bronchial epithelial cells by targetingTGFβR2gene in severe asthma. Allergy 2014, 70, 212–219. [Google Scholar] [CrossRef]
- Mattes, J.; Collison, A.; Plank, M.; Phipps, S.; Foster, P.S. Antagonism of microRNA-126 suppresses the effector function of T H 2 cells and the development of allergic airways disease. Proc. Natl. Acad. Sci. USA 2009, 106, 18704–18709. [Google Scholar] [CrossRef]
- Tian, M.; Ji, Y.; Wang, T.; Zhang, W.; Zhou, Y.; Cui, Y. Changes in circulating microRNA-126 levels are associated with immune imbalance in children with acute asthma. Int. J. Immunopathol. Pharmacol. 2018, 32, 2058738418779243. [Google Scholar] [CrossRef] [PubMed]
- Kho, A.T.; Sharma, S.; Davis, J.S.; Spina, J.; Howard, D.; McEnroy, K.; Moore, K.; Sylvia, J.; Qiu, W.; Weiss, S.T.; et al. Circulating MicroRNAs: Association with Lung Function in Asthma. PLoS ONE 2016, 11, e0157998. [Google Scholar] [CrossRef] [PubMed]
- Naydenova, K.; Dimitrov, V.; Velikova, T. Immunological and microRNA Features of Allergic Rhinitis in the Context of United Airway Disease. Sinusitis 2021, 5, 45–52. [Google Scholar] [CrossRef]
| Genome-Wide Association Studies | ||||
|---|---|---|---|---|
| Gene | Locus/Chr. Region * | Function/Biomarker | Population | Ref. |
| DENND1B | 1q31 | Involved in T cell receptor signaling pathway and positive regulation of T-helper 2 cell cytokine production | North American children of European ancestry | Sleiman et al., 2010 [18] |
| DPP10 | 2q14 | Involved in positive regulation of protein localization to plasma membrane and proteolysis; IL-1 gene cluster | English and German; American Blacks, African Caribbean | Allen et al., 2003 [19] |
| ORMDL3 GSDMB | 17q21 | Acts upstream or within negative regulation of B cell apoptotic process; | Europeans, Asians, Hispanics, | Moffatt et al., 2007 [17] Moffatt et al., 2010 [29] Thomsen et al., 2015 [35] Demenais et al., 2019 [23] |
| CHI3LI | 1q32.1 | YKL-40 plays a role in the pathogenesis of autoimmune diseases; | Europeans, Hutterites, American, German | Ober et al., 2008 [21] Chen & Wang, 2017 [22] |
| HLA-DQ | 6p21 | MHC class II loci; involved in immunoglobulin production and immunoglobulin-mediated immune response | American Whites, Europeans | Li et al., 2010 [30] Thomsen et al., 2015 [35] Demenais et al., 2019 [23] |
| RAD50 | 5q31 | Involved in DNA double-strand break processing; involved in mitotic G2/M transition checkpoint; | Northeastern Han Chinese, | Li et al., 2010 [30] Thomsen, 2015 [35] |
| IL13 | 5q31 | Inflammation; involved in the positive regulation of macrophage activation | Northeastern Han Chinese, Europeans | Li et al., 2010 [30] Thomsen, 2015 [35] |
| CDHR3 | 7q22 | Pronounced severe and early onset of disease; related to increased IL-17A response to viral infections; | Europeans | Bonnelykke et al., 2014 [31] Eliasen et al., 2022 [34] |
| IL1RL1 IL18R1 | 2q11 2q12 | Encodes a protein in the Toll-like receptor superfamily/encodes the IL-18 receptor; | Icelanders, Europeans, East Asians | Moffatt et al., 2010 [29] Hayden et al., 2018 [28] Ferreira et al., 2017 [25] |
| IL33 | 9p24 | Encodes IL-33, belongs to the IL-1 superfamily of proteins; | Europeans | Moffat et al., 2007 [17] |
| IRF4 NOD2 IL4R IL2RA | 6p25 16q12 16p12 10p15 | Involved in the regulation of T-helper cell differentiation and innate immune response; cytokine signaling in the immune system; | Europeans | Ferreira et al., 2017 [25] |
| KIAA1958 IL1RL1 IL13 LINC01149 | 9q32 2q12 5q31 6p21 | Encodes a protein in the Toll-like receptor superfamily/encodes the IL-18 receptor; involved in positive regulation of macrophage activation; | Europeans | Li et al., 2010 [30] Hayden et al., 2018 [25] |
| DMRT1 | 9p24 | Influence sex-specific patterns of childhood asthma; potential involvement in hormone or immune cell regulation; | Europeans | Schieck et al., 2016 [37] |
| TNFSF15 | 9q32 | Not expressed in B and T cells; the expression of the gene product is inducible by TNF and IL-1 alpha; | Koreans, Asia | Kim et al., 2022 [51] |
| Gene | Locus/Chr. Region * | Association Effect (Pathogenesis, Function, Severity, Treatment) | Ref. |
|---|---|---|---|
| ADBR2 | 5q32 | Arg16-variant is associated with improved response to Beta-2 agonism; asthma severity; | Israel et al., 2000 [52] Litonjua et al., 2010 [53] Sood et al., 2018 [54] Scaparrotta et al., 2019 [55] |
| IL4 | 5q31 | Encodes interleukin-4, associated with reduced forced expiratory volume in 1 s; associated with asthma severity; | Burchard et al., 1999 [56] |
| ALOX5 | 10q11 | 5-lipoxygenase, associated with improved response to ALOX5 inhibitor; worse lung function and asthma control; | Drazen et al., 1999 [57] Mougey et al., 2012 [58] |
| CRHR1 | 17q21 | Intronic region of the corticotrophin-releasing hormone receptor 1 gene, response to inhaled corticosteroids; | Tantisira et al., 2004 [59] |
| CD14 | 5q31 | Associated with asthma severity with modification from secondhand smoke; associated with asthma severity; | Choudhry et al., 2005 [60] Martin et al., 2006 [61] |
| ADAM33 | 20p13 | Increased in severe asthma vs. mild asthma or no asthma; moderate and severe asthma; | Foley et al., 2007 [62] |
| ORMDL3 cluster | 17q21 | Association with exacerbations and poor asthma control on controller medication; response to treatment with inhaled corticosteroids; proinflammatory airway epithelial response; | Tavendale et al., 2008 [63] Bisgaard et al., 2009 [64] Berce et al., 2013 [65] Farzan et al., 2018 [66] |
| GLCCI1 | 7p21 | Response to inhaled corticosteroids; | Tantisira et al., 2011 [67] |
| SPATS2L | 2q33 | Related to bronchodilator responsiveness; related to different ranges of asthma severity, age, and baseline; | Himes et al., 2012 [68] |
| ASB3 | 2p16 | Modulates pulmonary function; bronchodilator response to inhaledβ2-agonists; | Israel et al., 2015 [69] |
| IL1RL1 | 2q12 | Response to inhaled corticosteroids; related to asthma susceptibility and severity; | Dijk et al., 2019 [70] |
| COL2A1 | 12q13 | Response to inhaled corticosteroids; related to mild-to-moderate asthma; | Wan et al., 2019 [71] |
| MMP9 | 20q13 | MMP-9 is released by inflammatory cells in response to allergen provocation in asthma, declining lung function; | Dragicevic et al., 2018 [72] |
| EDDM3B | 14q11 | Inhaled corticosteroids in pediatric asthma; | Levin et al., 2019 [73] |
| CRISPLD2 | 16q24 | Associated with an adaptive response to asthma; a glucocorticoid responsive gene in airway smooth muscle cells; | Himes et al., 2014 [74] Almoguera et al., 2017 [42] |
| DNAH5 | 5p15 | Associated with lung function or the presence/absence of COPD; moderate-to-severe persistent asthma; | Ramasamy et al., 2011 [75] Lee et al., 2014 [76] Mak et al., 2018 [77] Perez-Garcia et al., 2020 [78] |
| FCER2 | 19p13 | Associated with response to inhaled corticosteroids; associated with asthma severity; | Lutz et al., 2015 [79] Wain et al., 2017 [80] Karimi et al., 2019 [81] |
| VEGFA | 6p21 | Associated with response to inhaled corticosteroids therapy severity of bronchial asthma; | Balantic et al., 2012 [82] Wan et al., 2019 [71] |
| Genes/Candidate-Genes * | Histone Modifications | Tissue | Ref. |
|---|---|---|---|
| IL-13, FOXP3 | histone H3 acetylation | Peripheral blood | Harb et al., 2015 [124] |
| IL-4, IL-13 | higher expression of HDACs 1 and 9 | Bronchial epithelium | Wawrzyniak et al., 2016 [132] |
| STAT6, EGFR and ΔNp63 | histone H3 acetylation | Bronchial airway epithelial cells | Stefanowics et al., 2015 [125] |
| IFNG, SH2B3 HDAC4 | histone H3 acetylation histone H4 acetylation | Blood samples Placenta samples | Harb et al., 2019 [138] |
| ORMDL3 | histone H3 acetylation | Bronchial epithelium | Cheng et al., 2019 [131] |
| miRNAs | Level | Function/Role in Asthma | Tissue | Ref. |
|---|---|---|---|---|
| miR-21 | increased | - eosinophilic accumulation - correlation with IL-13 - Th2 response - suppression of IL-12p35 | bronchial epithelial cells | Lu et al., 2011 [157] Wu et al., 2014 [144] Pua et al., 2015 [145] Elbehidy R., 2016 [146] |
| miR-146 | increased | - increase the eosinophilic level - anti-inflammatory - negative regulation of IL1β and COX2 | blood samples, human ASM cells | Su et al., 2011 [152] Comer et al., 2014 [158] Hammad et al., 2018 [148] |
| miR-155 | increased | - decreased level of Let-7a - regulation of IL-13 - in Th2 immune responses | blood samples | Zhou et al., 2016 [159] Karam et al., 2019 [149] |
| miR-19 | increased | -mediate the production of IL-13 -targets mRNA of the TGFβR2 gene -regulation in responses of the Th2 cell | bronchial epithelial cells, airway T cells | Simpson et al., 2014 [160] Haj-Salem et al., 20015 [161] |
| miR-126 | Increased | - increase AHR and immune migration - Th2 response - increase the eosinophilic level | bronchial epithelial cells, peripheral blood | Mattes et al., 2009 [162] Wu et al., 2014 [144] Tian et al., 2018 [163] |
| miR-15b, 126, -139, -142, -186 etc. | - lung function parameters in children | Blood samples | Kho et al., 2016 [164] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miteva, D.; Lazova, S.; Velikova, T. Genetic and Epigenetic Factors in Risk and Susceptibility for Childhood Asthma. Allergies 2023, 3, 115-133. https://doi.org/10.3390/allergies3020009
Miteva D, Lazova S, Velikova T. Genetic and Epigenetic Factors in Risk and Susceptibility for Childhood Asthma. Allergies. 2023; 3(2):115-133. https://doi.org/10.3390/allergies3020009
Chicago/Turabian StyleMiteva, Dimitrina, Snezhina Lazova, and Tsvetelina Velikova. 2023. "Genetic and Epigenetic Factors in Risk and Susceptibility for Childhood Asthma" Allergies 3, no. 2: 115-133. https://doi.org/10.3390/allergies3020009
APA StyleMiteva, D., Lazova, S., & Velikova, T. (2023). Genetic and Epigenetic Factors in Risk and Susceptibility for Childhood Asthma. Allergies, 3(2), 115-133. https://doi.org/10.3390/allergies3020009









