Cerebral Hemodynamic Influences in Task-Related Functional Magnetic Resonance Imaging and Near-Infrared Spectroscopy in Acute Sport-Related Concussion: A Review
Abstract
1. Introduction
2. Hemodynamic Impairments in Acute Concussion
3. Task-Related Functional Magnetic Resonance Imaging in Acute Concussion
3.1. Hyperactivation
3.2. Hypoactivation
4. Limits of the Task-Related Functional Magnetic Resonance Imaging in Acutely Concussed Patients
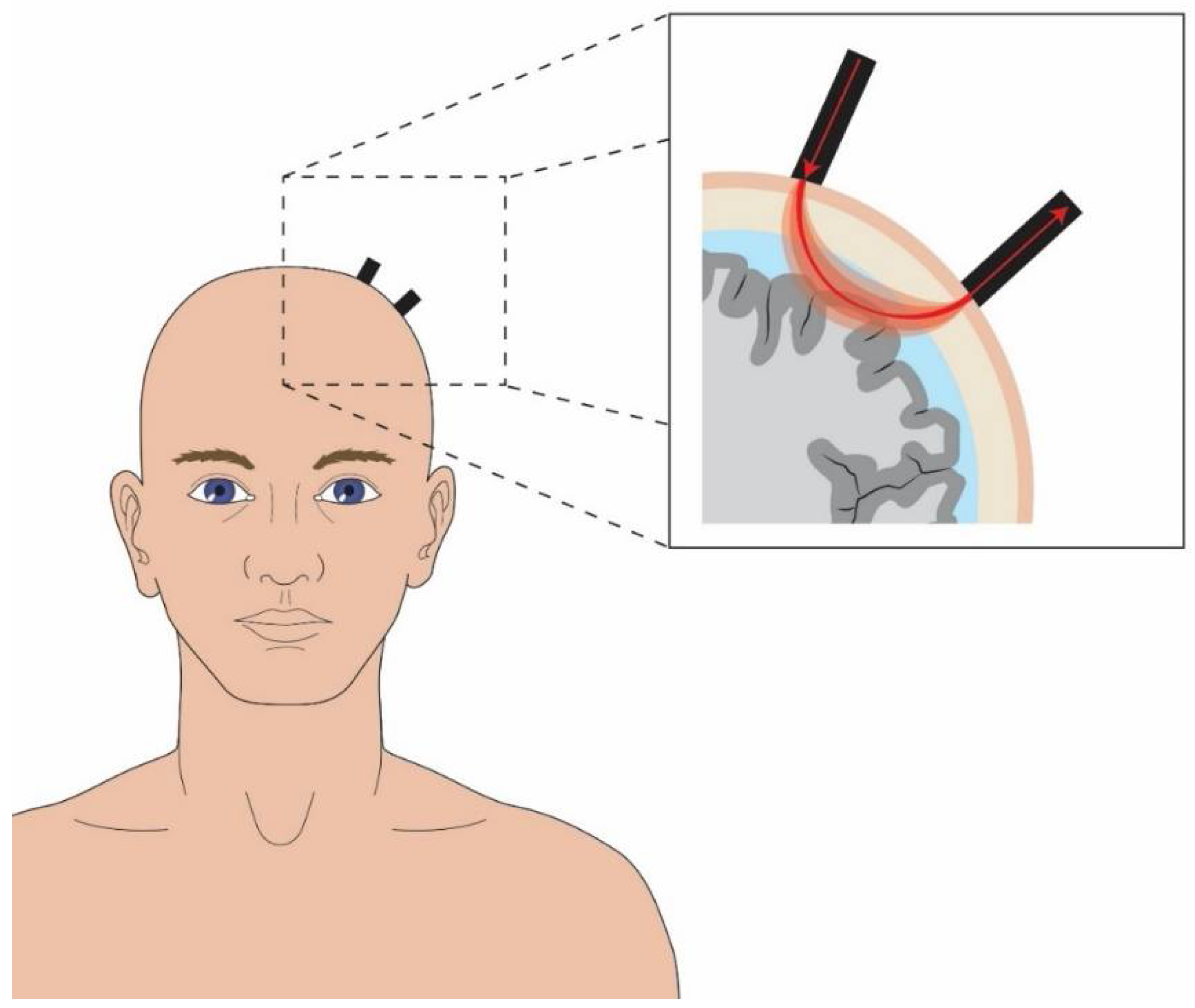
5. Near-Infrared Spectroscopy in Acutely Concussed Patients
6. Conclusions
Author Contributions
Conflicts of Interest
References
- McCrory, P.; Meeuwisse, W.; Dvorak, J.; Aubry, M.; Bailes, J.; Broglio, S.; Cantu, R.C.; Cassidy, D.; Echemendia, R.J.; Castellani, R.J.; et al. Consensus statement on concussion in sport-the 5th international conference on concussion in sport held in Berlin, October 2016. Br. J. Sports Med. 2017. [Google Scholar] [CrossRef] [PubMed]
- Daneshvar, D.H.; Nowinski, C.J.; McKee, A.C.; Cantu, R.C. The epidemiology of sport-related concussion. Clin. Sports Med. 2011, 30, 1–17. [Google Scholar] [CrossRef] [PubMed]
- McCrea, M.; Hammeke, T.; Olsen, G.; Leo, P.; Guskiewicz, K. Unreported concussion in high school football players: Implications for prevention. Clin. J. Sport Med. 2004, 14, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Lovell, M.R.; Collins, M.W.; Maroon, J.C.; Cantu, R.; Hawn, M.A.; Burke, C.J.; Fu, F. Inaccuracy of symptom reporting following concussion in athletes. Med. Sci. Sports Exerc. 2002, 34, S298. [Google Scholar] [CrossRef]
- Giza, C.C.; Hovda, D.A. The new neurometabolic cascade of concussion. Neurosurgery 2014, 75 (Suppl. 4), S24–S33. [Google Scholar] [CrossRef] [PubMed]
- Vagnozzi, R.; Tavazzi, B.; Signoretti, S.; Amorini, A.M.; Belli, A.; Cimatti, M.; Delfini, R.; Di Pietro, V.; Finocchiaro, A.; Lazzarino, G. Temporal window of metabolic brain vulnerability to concussions: Mitochondrial-related impairment—Part I. Neurosurgery 2007, 61, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Tavazzi, B.; Vagnozzi, R.; Signoretti, S.; Amorini, A.M.; Belli, A.; Cimatti, M.; Delfini, R.; Di Pietro, V.; Finocchiaro, A.; Lazzarino, G. Temporal window of metabolic brain vulnerability to concussions: Oxidative and nitrosative stresses—Part II. Neurosurgery 2007, 61, 390–396. [Google Scholar] [CrossRef] [PubMed]
- Echemendia, R.J.; Giza, C.C.; Kutcher, J.S. Developing guidelines for return to play: Consensus and evidence-based approaches. Brain Inj. 2015, 29, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Echemendia, R.J.; Putukian, M.; Mackin, R.S.; Julian, L.; Shoss, N. Neuropsychological test performance prior to and following sports-related mild traumatic brain injury. Clin. J. Sport Med. 2001, 11, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Lovell, M.R.; Collins, M.W.; Iverson, G.L.; Field, M.; Maroon, J.C.; Cantu, R.; Podell, K.; Powell, J.W.; Belza, M.; Fu, F.H. Recovery from mild concussion in high school athletes. J. Neurosurg. 2003, 98, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Broglio, S.P.; Macciocchi, S.N.; Ferrara, M.S. Neurocognitive performance of concussed athletes when symptom free. J. Athletic Train. 2007, 42, 504–508. [Google Scholar]
- Van Kampen, D.A.; Lovell, M.R.; Pardini, J.E.; Collins, M.W.; Fu, F.H. The “value added” of neurocognitive testing after sports-related concussion. Am. J. Sports Med. 2006, 34, 1630–1635. [Google Scholar] [CrossRef] [PubMed]
- Slobounov, S.; Tutwiler, R.; Sebastianelli, W.; Slobounov, E. Alteration of postural responses to visual field motion in mild traumatic brain injury. Neurosurgery 2006, 59, 134–193. [Google Scholar] [CrossRef] [PubMed]
- Slobounov, S.; Slobounov, E.; Sebastianelli, W.; Cao, C.; Newell, K. Differential rate of recovery in athletes after first and second concussion episodes. Neurosurgery 2007, 61, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Vagnozzi, R.; Signoretti, S.; Cristofori, L.; Alessandrini, F.; Floris, R.; Isgro, E.; Ria, A.; Marziali, S.; Zoccatelli, G.; Tavazzi, B.; et al. Assessment of metabolic brain damage and recovery following mild traumatic brain injury: A multicentre, proton magnetic resonance spectroscopic study in concussed patients. Brain 2010, 133, 3232–3242. [Google Scholar] [CrossRef] [PubMed]
- McCrea, M.; Guskiewicz, K.; Randolph, C.; Barr, W.B.; Hammeke, T.A.; Marshall, S.W.; Kelly, J.P. Effects of a symptom-free waiting period on clinical outcome and risk of reinjury after sport-related concussion. Neurosurgery 2009, 65, 876–883. [Google Scholar] [CrossRef] [PubMed]
- Eierud, C.; Craddock, R.C.; Fletcher, S.; Aulakh, M.; King-Casas, B.; Kuehl, D.; LaConte, S.M. Neuroimaging after mild traumatic brain injury: Review and meta-analysis. Neuroimage Clin. 2014, 4, 283–294. [Google Scholar] [CrossRef] [PubMed]
- Yuh, E.L.; Hawryluk, G.W.; Manley, G.T. Imaging concussion: A review. Neurosurgery 2014, 75 (Suppl. 4), S50–S63. [Google Scholar] [CrossRef] [PubMed]
- Jantzen, K.J. Functional magnetic resonance imaging of mild traumatic brain injury. J. Head Trauma Rehabil. 2010, 25, 256–266. [Google Scholar] [CrossRef] [PubMed]
- La Fountaine, M.F.; Toda, M.; Testa, A.; Bauman, W.A. Cardioautonomic instability following a sports-related concussion in a 20-year-old male. Int. J. Cardiol. 2014, 172, e511–e512. [Google Scholar] [CrossRef] [PubMed]
- La Fountaine, M.F.; Gossett, J.D.; De Meersman, R.E.; Bauman, W.A. Increased QT interval variability in 3 recently concussed athletes: An exploratory observation. J. Athletic Train. 2011, 46, 230–233. [Google Scholar] [CrossRef]
- Gall, B.; Parkhouse, W.; Goodman, D. Heart rate variability of recently concussed athletes at rest and exercise. Med. Sci. Sports Exerc. 2004, 36, 1269–1274. [Google Scholar] [CrossRef] [PubMed]
- La Fountaine, M.F.; Heffernan, K.S.; Gossett, J.D.; Bauman, W.A.; De Meersman, R.E. Transient suppression of heart rate complexity in concussed athletes. Auton. Neurosci. Basic Clin. 2009, 148, 101–103. [Google Scholar] [CrossRef] [PubMed]
- La Fountaine, M.F.; Toda, M.; Testa, A.J.; Hill-Lombardi, V. Autonomic nervous system responses to concussion: Arterial pulse contour analysis. Front. Neurol. 2016, 7, 13. [Google Scholar] [CrossRef] [PubMed]
- Dobson, J.L.; Yarbrough, M.B.; Perez, J.; Evans, K.; Buckley, T. Sport-related concussion induces transient cardiovascular autonomic dysfunction. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2017, 312, R575–R584. [Google Scholar] [CrossRef] [PubMed]
- Middleton, K.; Krabak, B.J.; Coppel, D.B. The influence of pediatric autonomic dysfunction on recovery after concussion. Clin. J. Sport Med. 2010, 20, 491–492. [Google Scholar] [CrossRef] [PubMed]
- Len, T.K.; Neary, J.P.; Asmundson, G.J.; Goodman, D.G.; Bjornson, B.; Bhambhani, Y.N. Cerebrovascular reactivity impairment after sport-induced concussion. Med. Sci. Sports Exerc. 2011, 43, 2241–2248. [Google Scholar] [CrossRef] [PubMed]
- Slobounov, S.M.; Gay, M.; Zhang, K.; Johnson, B.; Pennell, D.; Sebastianelli, W.; Horovitz, S.; Hallett, M. Alteration of brain functional network at rest and in response to YMCA physical stress test in concussed athletes: RsFMRI study. NeuroImage 2011, 55, 1716–1727. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, A.; Put, E.; Ingels, M.; Bossuyt, A. Prospective evaluation of technetium-99m-HMPAO SPECT in mild and moderate traumatic brain injury. J. Nucl. Med. 1994, 35, 942–947. [Google Scholar] [PubMed]
- Gowda, N.K.; Agrawal, D.; Bal, C.; Chandrashekar, N.; Tripati, M.; Bandopadhyaya, G.P.; Malhotra, A.; Mahapatra, A.K. Technetium Tc-99m ethyl cysteinate dimer brain single-photon emission CT in mild traumatic brain injury: A prospective study. Am. J. Neuroradiol. 2006, 27, 447–451. [Google Scholar] [PubMed]
- Lorberboym, M.; Lampl, Y.; Gerzon, I.; Sadeh, M. Brain SPECT evaluation of amnestic ED patients after mild head trauma. Am. J. Emerg. Med. 2002, 20, 310–313. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, D.; Gowda, N.K.; Bal, C.S.; Pant, M.; Mahapatra, A.K. Is medial temporal injury responsible for pediatric postconcussion syndrome? A prospective controlled study with single-photon emission computerized tomography. J. Neurosurg. 2005, 102, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Maugans, T.A.; Farley, C.; Altaye, M.; Leach, J.; Cecil, K.M. Pediatric sports-related concussion produces cerebral blood flow alterations. Pediatrics 2012, 129, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Metting, Z.; Rodiger, L.A.; Stewart, R.E.; Oudkerk, M.; De Keyser, J.; van der Naalt, J. Perfusion computed tomography in the acute phase of mild head injury: Regional dysfunction and prognostic value. Ann. Neurol. 2009, 66, 809–816. [Google Scholar] [CrossRef] [PubMed]
- Meier, T.B.; Bellgowan, P.S.; Singh, R.; Kuplicki, R.; Polanski, D.W.; Mayer, A.R. Recovery of cerebral blood flow following sports-related concussion. JAMA Neurol. 2015, 72, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Churchill, N.W.; Hutchison, M.G.; Richards, D.; Leung, G.; Graham, S.J.; Schweizer, T.A. The first week after concussion: Blood flow, brain function and white matter microstructure. Neuroimage Clin. 2017, 14, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Mark, C.I.; Mazerolle, E.L.; Chen, J.J. Metabolic and vascular origins of the bold effect: Implications for imaging pathology and resting-state brain function. J. Magn. Reson. Imaging 2015, 42, 231–246. [Google Scholar] [CrossRef] [PubMed]
- Heeger, D.J.; Ress, D. What does fMRI tell us about neuronal activity? Nat. Rev. Neurosci. 2002, 3, 142–151. [Google Scholar] [CrossRef] [PubMed]
- McAllister, T.W.; Saykin, A.J.; Flashman, L.A.; Sparling, M.B.; Johnson, S.C.; Guerin, S.J.; Mamourian, A.C.; Weaver, J.B.; Yanofsky, N. Brain activation during working memory 1 month after mild traumatic brain injury: A functional MRI study. Neurology 1999, 53, 1300–1308. [Google Scholar] [CrossRef] [PubMed]
- McAllister, T.W.; Sparling, M.B.; Flashman, L.A.; Guerin, S.J.; Mamourian, A.C.; Saykin, A.J. Differential working memory load effects after mild traumatic brain injury. NeuroImage 2001, 14, 1004–1012. [Google Scholar] [CrossRef] [PubMed]
- Krivitzky, L.S.; Roebuck-Spencer, T.M.; Roth, R.M.; Blackstone, K.; Johnson, C.P.; Gioia, G. Functional magnetic resonance imaging of working memory and response inhibition in children with mild traumatic brain injury. J. Int. Neuropsychol. Soc. 2011, 17, 1143–1152. [Google Scholar] [CrossRef] [PubMed]
- Talavage, T.M.; Nauman, E.A.; Breedlove, E.L.; Yoruk, U.; Dye, A.E.; Morigaki, K.E.; Feuer, H.; Leverenz, L.J. Functionally-detected cognitive impairment in high school football players without clinically-diagnosed concussion. J. Neurotrauma 2014, 31, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Jantzen, K.J.; Anderson, B.; Steinberg, F.L.; Kelso, J.A. A prospective functional MR imaging study of mild traumatic brain injury in college football players. Am. J. Neuroradiol. 2004, 25, 738–745. [Google Scholar] [PubMed]
- Lovell, M.R.; Pardini, J.E.; Welling, J.; Collins, M.W.; Bakal, J.; Lazar, N.; Roush, R.; Eddy, W.F.; Becker, J.T. Functional brain abnormalities are related to clinical recovery and time to return-to-play in athletes. Neurosurgery 2007, 61, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Smits, M.; Dippel, D.W.; Houston, G.C.; Wielopolski, P.A.; Koudstaal, P.J.; Hunink, M.G.; van der Lugt, A. Postconcussion syndrome after minor head injury: Brain activation of working memory and attention. Hum. Brain Mapp. 2009, 30, 2789–2803. [Google Scholar] [CrossRef] [PubMed]
- Pardini, J.E.; Pardini, D.A.; Becker, J.T.; Dunfee, K.L.; Eddy, W.F.; Lovell, M.R.; Welling, J.S. Postconcussive symptoms are associated with compensatory cortical recruitment during a working memory task. Neurosurgery 2010, 67, 1020–1028. [Google Scholar] [CrossRef] [PubMed]
- Slobounov, S.M.; Zhang, K.; Pennell, D.; Ray, W.; Johnson, B.; Sebastianelli, W. Functional abnormalities in normally appearing athletes following mild traumatic brain injury: A functional MRI study. Exp. Brain Res. 2010, 202, 341–354. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Johnson, B.; Pennell, D.; Ray, W.; Sebastianelli, W.; Slobounov, S. Are functional deficits in concussed individuals consistent with white matter structural alterations: Combined fMRI & DTI study. Exp. Brain Res. 2010, 204, 57–70. [Google Scholar] [PubMed]
- Maruishi, M.; Miyatani, M.; Nakao, T.; Muranaka, H. Compensatory cortical activation during performance of an attention task by patients with diffuse axonal injury: A functional magnetic resonance imaging study. J. Neurol. Neurosurg. Psychiatry 2007, 78, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Turner, G.R.; Levine, B. Augmented neural activity during executive control processing following diffuse axonal injury. Neurology 2008, 71, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Hammeke, T.A.; McCrea, M.; Coats, S.M.; Verber, M.D.; Durgerian, S.; Flora, K.; Olsen, G.S.; Leo, P.D.; Gennarelli, T.A.; Rao, S.M. Acute and subacute changes in neural activation during the recovery from sport-related concussion. J. Int. Neuropsychol. Soc. 2013, 19, 863–872. [Google Scholar] [CrossRef] [PubMed]
- Mayer, A.R.; Mannell, M.V.; Ling, J.; Elgie, R.; Gasparovic, C.; Phillips, J.P.; Doezema, D.; Yeo, R.A. Auditory orienting and inhibition of return in mild traumatic brain injury: A fMRI study. Hum. Brain Mapp. 2009, 30, 4152–4166. [Google Scholar] [CrossRef] [PubMed]
- Stulemeijer, M.; Vos, P.E.; van der Werf, S.; van Dijk, G.; Rijpkema, M.; Fernandez, G. How mild traumatic brain injury may affect declarative memory performance in the post-acute stage. J. Neurotrauma 2010, 27, 1585–1595. [Google Scholar] [CrossRef] [PubMed]
- Witt, S.T.; Lovejoy, D.W.; Pearlson, G.D.; Stevens, M.C. Decreased prefrontal cortex activity in mild traumatic brain injury during performance of an auditory oddball task. Brain Imaging Behav. 2010, 4, 232–247. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Yeo, R.A.; Pena, A.; Ling, J.M.; Klimaj, S.; Campbell, R.; Doezema, D.; Mayer, A.R. An fMRI study of auditory orienting and inhibition of return in pediatric mild traumatic brain injury. J. Neurotrauma 2012, 29, 2124–2136. [Google Scholar] [CrossRef] [PubMed]
- Keightley, M.L.; Saluja, R.S.; Chen, J.K.; Gagnon, I.; Leonard, G.; Petrides, M.; Ptito, A. A functional magnetic resonance imaging study of working memory in youth after sports-related concussion: Is it still working? J. Neurotrauma 2014, 31, 437–451. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, C.J.; Madjar, C.; Desjardins-Crepeau, L.; Bellec, P.; Bherer, L.; Hoge, R.D. Age dependence of hemodynamic response characteristics in human functional magnetic resonance imaging. Neurobiol. Aging 2013, 34, 1469–1485. [Google Scholar] [CrossRef] [PubMed]
- Davies, D.J.; Su, Z.; Clancy, M.T.; Lucas, S.J.; Dehghani, H.; Logan, A.; Belli, A. Near-infrared spectroscopy in the monitoring of adult traumatic brain injury: A review. J. Neurotrauma 2015, 32, 933–941. [Google Scholar] [CrossRef] [PubMed]
- Davie, S.N.; Grocott, H.P. Impact of extracranial contamination on regional cerebral oxygen saturation: A comparison of three cerebral oximetry technologies. Anesthesiology 2012, 116, 834–840. [Google Scholar] [CrossRef] [PubMed]
- Strangman, G.E.; Li, Z.; Zhang, Q. Depth sensitivity and source-detector separations for near infrared spectroscopy based on the Colin27 brain template. PLoS ONE 2013, 8, e66319. [Google Scholar] [CrossRef] [PubMed]
- Strangman, G.; Boas, D.A.; Sutton, J.P. Non-invasive neuroimaging using near-infrared light. Biol. Psychiatry 2002, 52, 679–693. [Google Scholar] [CrossRef]
- Fantini, S.; Franceschini, M.-A.; Maier, J.S.; Walker, S.A.; Barbieri, B.B.; Gratton, E. Frequency-Domain Multichannel Optical Detector for Noninvasive Tissue Spectroscopy and Oximetry. Opt. Eng. 1995, 34, 32–43. [Google Scholar] [CrossRef]
- Fantini, S.; Franceschini, M.A.; Gratton, E. Semi-infinite-geometry boundary problem for light migration in highly scattering media: A frequency-domain study in the diffusion approximation. J. Opt. Soc. Am. B 1994, 11, 2128–2138. [Google Scholar] [CrossRef]
- Matcher, S.J.; Cooper, C.E. Absolute quantification of deoxyhaemoglobin concentration in tissue near infrared spectroscopy. Phys. Med. Biol. 1994, 39, 1295–1312. [Google Scholar] [CrossRef] [PubMed]
- Clancy, M.; Belli, A.; Davies, D.; Lucas, S.J.E.; Su, Z.J.; Dehghani, H. Comparison of neurological NIRS signals during standing valsalva maneuvers, pre and post vasopressor injection. In Diffuse Optical Imaging V; Dehghani, H., Taroni, P., Eds.; SPIE-International Society for Optical Engineering: Bellingham, WA, USA, 2015; Volume 9538. [Google Scholar]
- Clancy, M.; Belli, A.; Davies, D.; Lucas, S.J.E.; Su, Z.J.; Dehghani, H. Monitoring the injured brain—Registered, patient specific atlas models to improve accuracy of recovered brain saturation values. In Diffuse Optical Imaging V; Dehghani, H., Taroni, P., Eds.; SPIE-International Society for Optical Engineering: Bellingham, WA, USA, 2015; Volume 9538. [Google Scholar]
- Boas, D.A.; Gaudette, T.; Strangman, G.; Cheng, X.; Marota, J.J.; Mandeville, J.B. The accuracy of near infrared spectroscopy and imaging during focal changes in cerebral hemodynamics. NeuroImage 2001, 13, 76–90. [Google Scholar] [CrossRef] [PubMed]
- Chiarelli, A.M.; Maclin, E.L.; Low, K.A.; Fabiani, M.; Gratton, G. Comparison of procedures for co-registering scalp-recording locations to anatomical magnetic resonance images. J. Biomed. Opt. 2015, 20, 016009. [Google Scholar] [CrossRef] [PubMed]
- Amyot, F.; Zimmermann, T.; Riley, J.; Kainerstorfer, J.M.; Chernomordik, V.; Mooshagian, E.; Najafizadeh, L.; Krueger, F.; Gandjbakhche, A.H.; Wassermann, E.M. Normative database of judgment of complexity task with functional near infrared spectroscopy—Application for TBI. NeuroImage 2012, 60, 879–883. [Google Scholar] [CrossRef] [PubMed]
- Fantini, S.; Sassaroli, A.; Tgavalekos, K.T.; Kornbluth, J. Cerebral blood flow and autoregulation: Current measurement techniques and prospects for noninvasive optical methods. Neurophotonics 2016, 3, 031411. [Google Scholar] [CrossRef] [PubMed]
- Tachtsidis, I.; Scholkmann, F. False positives and false negatives in functional near-infrared spectroscopy: Issues, challenges, and the way forward. Neurophotonics 2016, 3, 031405. [Google Scholar] [CrossRef] [PubMed]
- Kontos, A.P.; Huppert, T.J.; Beluk, N.H.; Elbin, R.J.; Henry, L.C.; French, J.; Dakan, S.M.; Collins, M.W. Brain activation during neurocognitive testing using functional near-infrared spectroscopy in patients following concussion compared to healthy controls. Brain Imaging Behav. 2014, 8, 621–634. [Google Scholar] [CrossRef] [PubMed]
- Fabiani, M.; Low, K.A.; Tan, C.H.; Zimmerman, B.; Fletcher, M.A.; Schneider-Garces, N.; Maclin, E.L.; Chiarelli, A.M.; Sutton, B.P.; Gratton, G. Taking the pulse of aging: Mapping pulse pressure and elasticity in cerebral arteries with optical methods. Psychophysiology 2014, 51, 1072–1088. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.H.; Low, K.A.; Kong, T.; Fletcher, M.A.; Zimmerman, B.; Maclin, E.L.; Chiarelli, A.M.; Gratton, G.; Fabiani, M. Mapping cerebral pulse pressure and arterial compliance over the adult lifespan with optical imaging. PLoS ONE 2017, 12, e0171305. [Google Scholar] [CrossRef] [PubMed]
| Author | Year | Method | Time of Assessment | N mTBI | Sport-Related Concussion | Main Findings |
|---|---|---|---|---|---|---|
| Gall et al. | 2004 | ECG | 24 h | 14 | Yes | Increase of HR |
| La Fountaine et al. | 2009 | ECG | 48 h; 2 weeks | 3 | Yes | Increase of HR complexity |
| Middleton et al. | 2010 | Photoplethysmography | 3 weeks | 1 | Yes | Abnormal response to physical tasks |
| Len et al. | 2011 | Transcranial Doppler Ultrasonography | 1 week | 10 | Yes | Abnormal cerebrovascular reactivity |
| Slobounov et al. | 2011 | Photoplethysmography | 12 days | 17 | Yes | No abnormalities |
| La Fountaine et al. | 2016 | ECG; Photoplethysmography | 48 h; 1 week | 10 | Yes | Reduction of stroke volume; increase of HR |
| Dobson et al. | 2017 | Photoplethysmography | 48 h; 72 h; 1 week; 2 weeks | 12 | Yes | Abnormal response to physical tasks |
| Author | Year | Method | Time of Assessment | N mTBI | Sport-Related Concussion | Main Findings |
|---|---|---|---|---|---|---|
| Jacobs et al. | 1994 | SPECT | 4 weeks | 25 | No | Reduced rCRF |
| Lorberboym et al. | 2001 | SPECT | 6 h | 16 | No | Reduced rCBF |
| Agrawal et al. | 2005 | SPECT | 10 days | 30 | No | Reduced rCBF |
| Gowda et al. | 2006 | SPECT | 72 h | 92 | No | Reduced rCBF |
| Metting et al. | 2009 | CT with contrast dye | Mean time 3.9 h | 76 | No | Reduced rCBF |
| Maugans et al. | 2012 | MRI with contrast dye | 72 h; 2 weeks; 4 weeks or more | 12 | Yes | Reduced CBF |
| Meier et al. | 2015 | MRI ASL | 1 day; 1 week; 4 weeks | 17 | Yes | Reduced rCBF |
| Churchill et al. | 2017 | MRI ASL | 1–3 days; 5–7 days | 26 | Yes | Elevated rCBF and subsequently reduced rCBF |
| Author | Year | Task | Time of Assessment | N mTBI | Sport-Related Concussion | Main Findings |
|---|---|---|---|---|---|---|
| McAllister et al. | 1999 | N-back | 4 weeks | 12 | Yes | Hyperactivation |
| McAllister et al. | 2000 | N-back | 4 weeks | 18 | Yes | No abnormalities |
| Jantzen et al. | 2004 | Finger sequences; Calculation; Digit span | 1 week | 4 | Yes | Hyperactivation |
| Lovell et al. | 2007 | N-back | 1 week | 28 | Yes | Hyperactivation |
| Smit et al. | 2009 | N-back; Stroop; Finger sequence | 4 weeks and more | 21 | No | Hyperactivation |
| Mayer et al. | 2009 | Auditory orienting | 3 weeks | 16 | No | Hypoactivation |
| Pardini et al. | 2010 | N-back | 2 weeks | 16 | Yes | Hyperactivation |
| Slobounov et al. | 2010 | Virtual reality | 4 weeks | 15 | Yes | Hyperactivation |
| Stulemeijer et al. | 2010 | N-back | 6 weeks | 43 | No | Hypoactivation |
| Witt et al. | 2010 | Auditory oddball | 13–200 days | 31 | No | Hypoactivation |
| Yang et al. | 2012 | Auditory orienting | 3 weeks or more | 14 | No | Hypoactivation |
| Hammeke et al. | 2013 | Sternberg | 48 h | 12 | Yes | Hypoactivation |
| Keightley et al. | 2014 | Visual memory | 90 days | 15 | Yes | Hypoactivation |
| Talavage et al. | 2014 | N-back | 72 h | 4 | Yes | Hyperactivation |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Forcione, M.; Colonnese, C.; Belli, A. Cerebral Hemodynamic Influences in Task-Related Functional Magnetic Resonance Imaging and Near-Infrared Spectroscopy in Acute Sport-Related Concussion: A Review. J. Imaging 2018, 4, 59. https://doi.org/10.3390/jimaging4040059
Forcione M, Colonnese C, Belli A. Cerebral Hemodynamic Influences in Task-Related Functional Magnetic Resonance Imaging and Near-Infrared Spectroscopy in Acute Sport-Related Concussion: A Review. Journal of Imaging. 2018; 4(4):59. https://doi.org/10.3390/jimaging4040059
Chicago/Turabian StyleForcione, Mario, Claudio Colonnese, and Antonio Belli. 2018. "Cerebral Hemodynamic Influences in Task-Related Functional Magnetic Resonance Imaging and Near-Infrared Spectroscopy in Acute Sport-Related Concussion: A Review" Journal of Imaging 4, no. 4: 59. https://doi.org/10.3390/jimaging4040059
APA StyleForcione, M., Colonnese, C., & Belli, A. (2018). Cerebral Hemodynamic Influences in Task-Related Functional Magnetic Resonance Imaging and Near-Infrared Spectroscopy in Acute Sport-Related Concussion: A Review. Journal of Imaging, 4(4), 59. https://doi.org/10.3390/jimaging4040059





