Zero Liquid Discharge System for the Tannery Industry—An Overview of Sustainable Approaches
Abstract
:1. Introduction
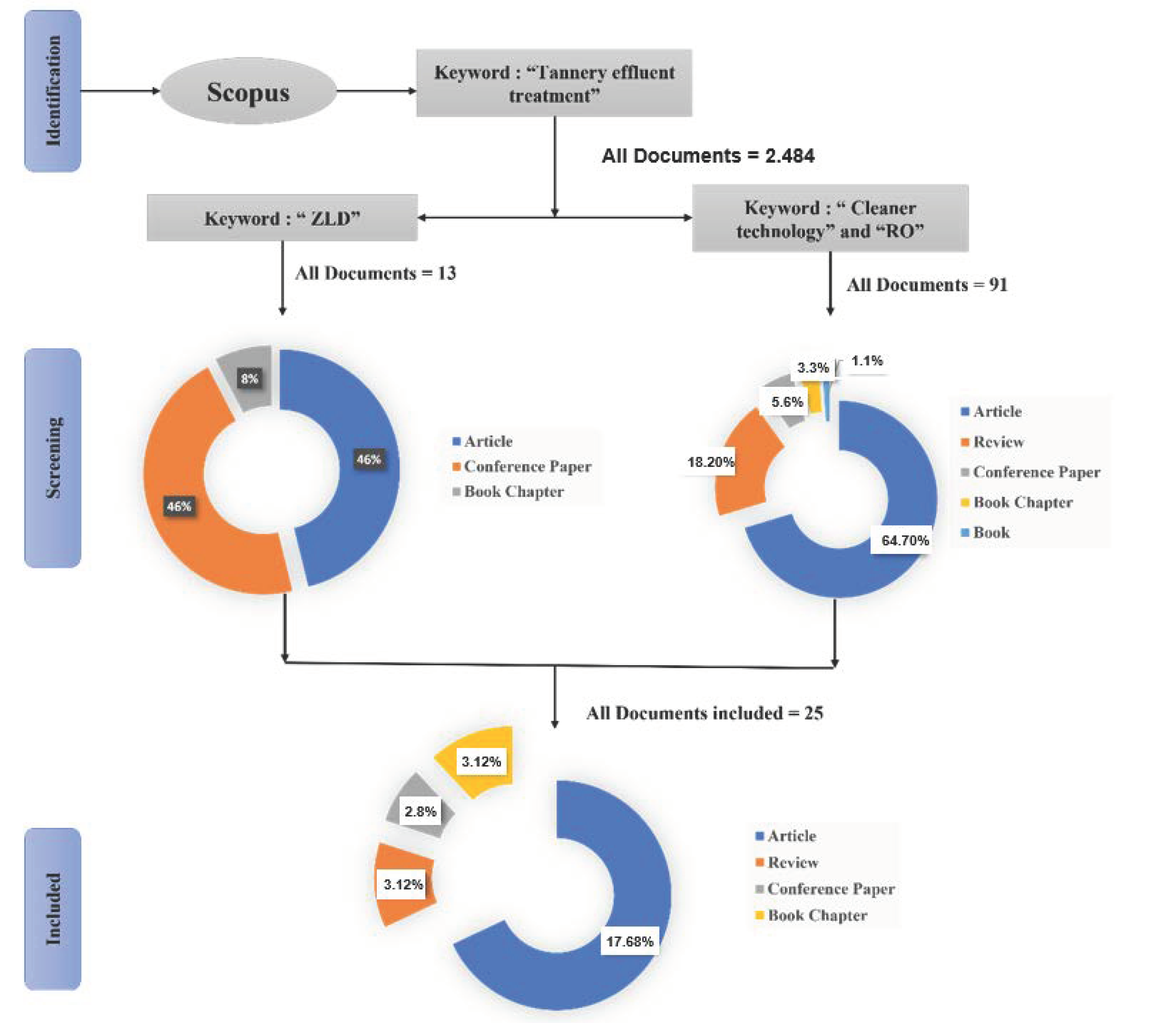
2. Methodology
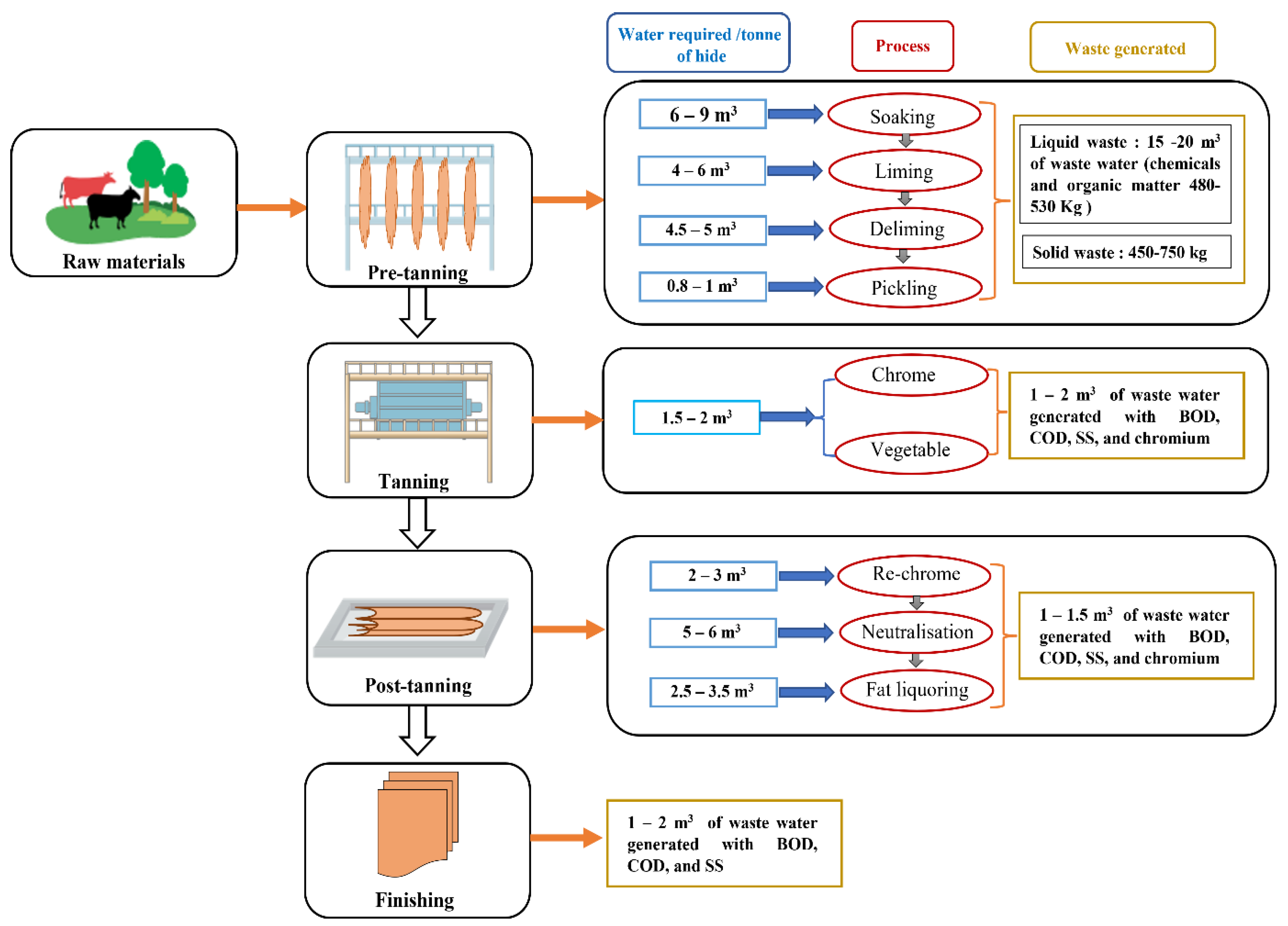
3. Tanning Process and Related Environmental Issues
4. Clean Technology Approach in Tanneries
- Use of enzymes in the dehairing bath: enzymes are added in the soaking phase to improve the water uptake and to degrade the proteins and fats present in the skin [34]. This approach reduces the processing time.
- Precipitation of chrome: to recover and reuse chrome in spent liquor by raising the pH to minimize the solubility of chromium in the liquor [35].
- Recycling the dehairing bath: instead of discharging it to the treatment plant after a single use, it can be reused after a simple filtration [36].
- Recycling the chrome tanning bath (can reduce chrome use by 20%): reusing the contents in the tanning bath after a simple filtration process [37].
| Process | Waste Generated | Category of Approach | Cleaner Technology | Reference |
|---|---|---|---|---|
| Mechanical shaking | Salts | Reuse of collected salts | [38] | |
| Fleshing | 100 kg of solid waste/ton of processed hide | Recycling of dehairing bath | Recycling the solid waste to poultry feed or soap industry | [39] |
| Soaking and washing | Wastewater with brine solution (contaminated with fats, soap, and dirt) | Addition of sodium carbonate and sodium hydroxide to increase the pH for better results | [39,40] | |
| Liming and unhairing | Wastewater contaminated with lime, hair, and sulfides | Using enzymes in the dehairing bath | Enzyme-assisted unhairing to reduce COD and sulfide content in the wastewater and recycling of spent liquor | [40,41] |
| Deliming | Wastewater contaminated with lime, hair, and sulfides | Using organic acid and carbon dioxide-based deliming and reusing the deliming liquor | [40,41,42] | |
| Pickling | No wastewater is released | Decreasing the pH to 2.4 to increase the tanning conditions and using a salt-free organic acid pickling system | [40,43] | |
| Tanning | Wastewater with chromium | High chrome exhaustion and precipitation of chrome | Adding soda-ash to increase pH from 2.4 to 4.1 to improve chromium utilization | [38] |
| Re-tanning | Wastewater contaminated with dyes and oils | Recycling of chrome tanning bath | Non-spraying dying methods are preferred with the use of liquid and low-dust dyes | [38] |
5. Need for Water Recycling in Tanneries
6. Zero Liquid Discharge system
6.1. Thermal-Based ZLD Systems
6.1.1. Drawbacks of Thermal-Based ZLD Systems
6.1.2. Approaches for Overcoming These Drawbacks
6.2. RO-Based ZLD Systems
6.2.1. Limitations of RO-Based ZLD Systems
6.2.2. Systems for Overcoming These Limitations
7. Critical Assessment of ZLD Economics
8. Application of ZLD in the Tannery Industry
8.1. India
8.2. United States
8.3. China
8.4. Other Countries
9. Discussion
- Reduction in freshwater consumption;
- Increase in water recovery;
- Reduction in the cost of operation; and
- Reduction/disposal of generated solid waste/brine liquor.
- In India, novel marine disposal of saline streams is explored as the desalination plant is integrated into a leather complex. It draws 30,000 m3/day and discharges 20,000 m3/day of the saline stream back into the Bay of Bengal under the overall control of environmental protection authorities with a special bio-control and dispersion system to safeguard the aquatic life [111].
- The volume of RO rejection can be reduced using an alternative technology known as the high-pressure RO system, where 30–40% of the water can be recovered from the rejected water of the RO plant. This reduces the volume of rejected water by 10%, thus reducing the cost of thermal evaporation by 35–40%. The overall operation cost can be reduced by 10–15% [112,113].
- RO-based ZLD systems can be improved by upgrading the system with a new technology named high-efficiency RO, with a recovery rate higher than 90% and a low tendency for scaling and fouling. It overcomes the restriction of salinity limit in RO and treats the brine concentrate efficiently and economically as RO [114].
- Membrane distillation incorporated in ZLD systems is a developing technology that has the potential to concentrate saline wastewater by using waste heat. This system is more economical than the brine concentrator as it can recover 60–90% of water and it works by the waste heat. Therefore, MD-ZLD has the potential to replace brine concentrators. However, at the moment, it has not yet been applied to large-scale treatment plants [115].
- Implementation of forward osmosis in ZLD is an alternative approach to the RO system. It is an energy-efficient separation technique to recover the water from the brine solution. Permeation of water molecules occurs under the influence of osmotic pressure without the requirement of hydraulic pressure. Less energy consumption and reversible membrane fouling are the advantages of this technology. It offers a cleaner alternative way to achieve higher water flux, thereby reducing the generation of brine solution [116,117].
- Electrodialysis can be used to preconcentrate the brine before the crystallizer as the ion exchange membrane eliminates the ions in the brine solution. As a result, a salt-depleted brine solution is obtained at the end of the process. Scaling and fouling can be easily removed by reversing the polarity of the electrodialysis. Pilot-scale studies have shown that this technology is capable of concentrating feed water to relatively high conductivity (above 120 mS/cm) compared to RO capability (80–90 mS/cm) [118].
- Solar evaporation technologies are suitable in arid regions and require low maintenance and low operating costs. Wind aid-intensified evaporation technology uses wind energy to evaporate the brine and can be 10-fold over the natural evaporation at the rate of 0.55–1.7 m3/h with 500 m2 of a wetted surface for 300–1000 m3/d [119].
- The use of chlorine-resistant membranes, such as sulfonated polysulfone composite membranes, can reduce membrane degradation, thus restricting the need for de-chlorination of the RO feed [120].
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sivaram, N.M.; Barik, D. Toxic waste from leather industries. In Energy from Toxic Organic Waste for Heat and Power Generation; Woodhead Publishing: Cambridge, UK, 2018; pp. 55–67. ISBN 9780081025284. [Google Scholar]
- Saran, S.; Chib, S.; Saxena, R.K. Biotechnology of leather: An alternative to conventional leather processing. A Handb. High Value Ferment. Prod. Hum. Welf. 2019, 2, 23–47. [Google Scholar]
- Saranya, D.; Shanthakumar, S. Opportunities for phycoremediation approach in tannery effluent: A treatment perspective. Environ. Prog. Sustain. Energy 2019, 38, e13078. [Google Scholar] [CrossRef]
- Omoloso, O.; Mortimer, K.; Wise, W.R.; Jraisat, L. Sustainability research in the leather industry: A critical review of progress and opportunities for future research. J. Clean. Prod. 2021, 285, 125441. [Google Scholar] [CrossRef]
- Singh, A. Livestock Production Statistics of India—2019. Available online: https://www.vetextension.com/livestock-animal-production-statistics-of-india-2019/ (accessed on 22 May 2022).
- Nisa, S. A Study of Export Potential of Indian Leather Industry and Strategies for Growth. SSRN’s Elibrary 2007. [Google Scholar] [CrossRef]
- Suman, H.; Sangal, V.K.; Vashishtha, M. Treatment of tannery industry effluent by electrochemical methods: A review. Mater. Today Proc. 2021, 47, 1438–1444. [Google Scholar] [CrossRef]
- Rajamani, S. Sustainable Environmental Technologies Including Water Recovery for Reuse from Tannery and Industrial Wastewater–Indian and Asian Scenario. Ann. Univ. Oradea Fascicle Text. Oradea Rom. 2017, 173–179. [Google Scholar]
- Zhao, C.; Chen, W. A review for tannery wastewater treatment: Some thoughts under stricter discharge requirements. Environ. Sci. Pollut. Res. 2019, 26, 26102–26111. [Google Scholar] [CrossRef]
- Doble, M.; Kruthiventi, A.K. Industrial Examples. In Green Chemistry and Engineering; Academic Press: Burlington, ON, Canada, 2007; pp. 245–296. ISBN 978-0-12-372532-5. [Google Scholar]
- Lofrano, G.; Meriç, S.; Zengin, G.E.; Orhon, D. Chemical and biological treatment technologies for leather tannery chemicals and wastewaters: A review. Sci. Total Environ. 2013, 461–462, 265–281. [Google Scholar] [CrossRef]
- Moktadir, M.A.; Ahmadi, H.B.; Sultana, R.; Zohra, F.T.; Liou, J.J.H.; Rezaei, J. Circular economy practices in the leather industry: A practical step towards sustainable development. J. Clean. Prod. 2020, 251, 119737. [Google Scholar] [CrossRef]
- Famielec, S. Chromium concentrate recovery from solid tannery waste in a thermal process. Materials 2020, 13, 1533. [Google Scholar] [CrossRef]
- Sundar, V.J.; Ramesh, R.; Rao, P.S.; Saravanan, P.; Sridharnath, B.; Muralidharan, C. Water management in leather industry. J. Sci. Ind. Res. 2001, 60, 443–450. [Google Scholar]
- Sivakumar, V.; Swaminathan, G.; Rao, P.G.; Ramasami, T. Influence of ultrasound on diffusion through skin/leather matrix. Chem. Eng. Process. Process Intensif. 2008, 47, 2076–2083. [Google Scholar] [CrossRef]
- Thazeem, B.; Umesh, M.; Mani, V.M.; Beryl, G.P.; Preethi, K. Biotransformation of bovine tannery fleshing into utilizable product with multifunctionalities. Biocatal. Biotransformation 2021, 39, 81–99. [Google Scholar] [CrossRef]
- Saravanabhavan, S.; Thanikaivelan, P.; Rao, J.R.; Nair, B.U.; Ramasami, T. Reversing the conventional leather processing sequence for cleaner leather production. Environ. Sci. Technol. 2006, 40, 1069–1075. [Google Scholar] [CrossRef]
- Tariq, S.R.; Shah, M.H.; Shaheen, N. Comparative statistical analysis of chrome and vegetable tanning effluents and their effects on related soil. J. Hazard. Mater. 2009, 169, 285–290. [Google Scholar] [CrossRef]
- Covington, A.D. Modern tanning chemistry. Chem. Soc. Rev. 1997, 26, 111. [Google Scholar] [CrossRef]
- Hansen, É.; Monteiro de Aquim, P.; Hansen, A.W.; Cardoso, J.K.; Ziulkoski, A.L.; Gutterres, M. Impact of post-tanning chemicals on the pollution load of tannery wastewater. J. Environ. Manag. 2020, 269, 110787. [Google Scholar] [CrossRef]
- Saxena, G.; Chandra, R.; Bharagava, R.N. Environmental pollution, toxicity profile and treatment approaches for tannery wastewater and its chemical pollutants. In Reviews of Environmental Contamination and Toxicology; de Voogt, P., Ed.; Springer: Cham, Switzerland, 2017; Volume 240, pp. 31–69. ISBN 978-3-319-42300-5. [Google Scholar]
- Bhattacharya, P.; Swarnakar, S.; Mukhopadhyay, A.; Ghosh, S. Exposure of composite tannery effluent on snail, Pila globosa: A comparative assessment of toxic impacts of the untreated and membrane treated effluents. Ecotoxicol. Environ. Saf. 2016, 126, 45–55. [Google Scholar] [CrossRef]
- Saxena, G.; Purchase, D.; Bharagava, R.N. Environmental Hazards and Toxicity Profile of Organic and Inorganic Pollutants of Tannery Wastewater and Bioremediation Approaches. In Bioremediation of Industrial Waste for Environmental Safety; Saxena, G., Bharagava, R.N., Eds.; Springer: Singapore, 2020; pp. 381–398. ISBN 978-981-13-1891-7. [Google Scholar]
- Costa, M.; Klein, C.B. Toxicity and carcinogenicity of chromium compounds in humans. Crit. Rev. Toxicol. 2006, 36, 155–163. [Google Scholar] [CrossRef]
- Bauchinger, M.; Schmid, E.; Dresp, J.; Kolin-Gerresheim, J.; Hauf, R.; Suhr, E. Chromosome changes in lymphocytes after occupational exposure to toluene. Mutat. Res. Toxicol. 1982, 102, 439–445. [Google Scholar] [CrossRef]
- Sharma, A.; Thakur, I.S.; Dureja, P. Enrichment, isolation and characterization of pentachlorophenol degrading bacterium Acinetobacter sp. ISTPCP-3 from effluent discharge site. Biodegradation 2009, 20, 643–650. [Google Scholar] [CrossRef]
- Shekhar Thakur, I.; Verma, P.K.; Upadhaya, K.C. Involvement of plasmid in degradation of pentachlorophenol by Pseudomonas sp. from a chemostat. Biochem. Biophys. Res. Commun. 2001, 286, 109–113. [Google Scholar] [CrossRef]
- Verma, T.; Tiwari, S.; Tripathi, M.; Ramteke, P.W. Treatment and Recycling of Wastewater from Tannery. In Advances in Biological Treatment of Industrial Waste Water and Their Recycling for a Sustainable Future; Singh, R.L., Singh, R.P., Eds.; Springer: Singapore, 2019; pp. 51–90. ISBN 978-981-13-1468-1. [Google Scholar]
- Kavitha, P.R.; Ganapathy, G.P. Tannery process and its environmental impacts a case study: Vellore District, Tamil Nadu, India. J. Chem. Pharm. Sci. 2015, 8, 759–764. [Google Scholar]
- Buljan, J.; Emmanuel, K.V.; Viswanathan, M.; Bosnić, M.; Král, I. Assessment of Performance of Zero Liquid Discharge (ZLD) Operations in Some Tannery Clusters Vellore Districts, Tamil Nadu, India; United Nations Industrial Development Organization: Vienna, Austria, 2017. [Google Scholar]
- Karuppiah, K.; Sankaranarayanan, B.; Ali, S.M.; Jabbour, C.J.C.; Bhalaji, R.K.A. Inhibitors to circular economy practices in the leather industry using an integrated approach: Implications for sustainable development goals in emerging economies. Sustain. Prod. Consum. 2021, 27, 1554–1568. [Google Scholar] [CrossRef]
- Morera, J.M.; Bacardit, A.; Ollé, L.; Bartolí, E.; Borràs, M.D. Minimization of the environmental impact of chrome tanning: A new process with high chrome exhaustion. Chemosphere 2007, 69, 1728–1733. [Google Scholar] [CrossRef]
- Bacardit, A.; Morera, J.M.; Ollé, L.; Bartolí, E.; Dolors Borràs, M. High chrome exhaustion in a non-float tanning process using a sulphonic aromatic acid. Chemosphere 2008, 73, 820–824. [Google Scholar] [CrossRef]
- Singhania, R.R.; Patel, A.K.; Thomas, L.; Goswami, M.; Giri, B.S.; Pandey, A. Industrial Enzymes. In Industrial Biorefineries and White Biotechnology; Pandey, A., Höfer, R., Taherzadeh, M., Nampoothiri, K.M., Larroche, C., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 473–497. ISBN 9780444634535. [Google Scholar]
- Kanagaraj, J.; Chandra Babu, N.K.; Mandal, A.B. Recovery and reuse of chromium from chrome tanning waste water aiming towards zero discharge of pollution. J. Clean. Prod. 2008, 16, 1807–1813. [Google Scholar] [CrossRef]
- Collivignarelli, C.; Barducci, G. Waste recovery from the tanning industry. Waste Manag. Res. 1984, 2, 265–278. [Google Scholar] [CrossRef]
- Blackman, A. Adoption of Clean Leather-Tanning Technologies in Mexico. Discussion Papers 10881, Resources for the Future; Washington, DC, USA, 2005; DP 05-38; Available online: https://ageconsearch.umn.edu/record/10881 (accessed on 22 May 2022). [CrossRef]
- Al-Jabari, M.; Sawalha, H.; Pugazhendhi, A.; Rene, E.R. Cleaner production and resource recovery opportunities in leather tanneries: Technological applications and perspectives. Bioresour. Technol. Rep. 2021, 16, 100815. [Google Scholar] [CrossRef]
- Varaidzo S Dandira, I.M. Design of a Cleaner Production Framework to Enhance Productivity: Case Study of Leather Company. Int. J. Sci. Res. 2013, 2, 18–37. [Google Scholar]
- Raghava Rao, J.; Chandrababu, N.K.; Muralidharan, C.; Nair, B.U.; Rao, P.G.; Ramasami, T. Recouping the wastewater: A way forward for cleaner leather processing. J. Clean. Prod. 2003, 11, 591–599. [Google Scholar] [CrossRef]
- Galiana-Aleixandre, M.V.; Mendoza-Roca, J.A.; Bes-Piá, A. Reducing sulfates concentration in the tannery effluent by applying pollution prevention techniques and nanofiltration. J. Clean. Prod. 2011, 19, 91–98. [Google Scholar] [CrossRef]
- Deng, W.; Chen, D.; Huang, M.; Hu, J.; Chen, L. Carbon dioxide deliming in leather production: A literature review. J. Clean. Prod. 2015, 87, 26–38. [Google Scholar] [CrossRef]
- Hu, J.; Xiao, Z.; Zhou, R.; Deng, W.; Wang, M.; Ma, S. Ecological utilization of leather tannery waste with circular economy model. J. Clean. Prod. 2011, 19, 221–228. [Google Scholar] [CrossRef]
- Bień, J.; Celary, P.; Wystalska, K. The problems in achieving sustainable development in the tannery industry in regard to sewage sludge management. J. Ecol. Eng. 2017, 18, 13–20. [Google Scholar] [CrossRef]
- Hutton, M.; Shafahi, M. Water pollution caused by leather industry: A review. In Proceedings of the ASME 2019 13th International Conference on Energy Sustainability, ES 2019, Collocated with the ASME 2019 Heat Transfer Summer Conference, American Society of Mechanical Engineers. Bellevue, WA, USA, 14–17 July 2019; Volume 59094, p. V001T10A002. [Google Scholar]
- Sreeram, K.J.; Ramasami, T. Sustaining tanning process through conservation, recovery and better utilization of chromium. Resour. Conserv. Recycl. 2003, 38, 185–212. [Google Scholar] [CrossRef]
- Mutlu, M.M.; Crudu, M.; Maier, S.S.; Deselnicu, D.C.; Albu, L.; Gulumser, G.; Bitlisli, B.O.; Basaran, B.; Tosun, C.C.; Adiguzel Zengin, A.C. Eko-leather: Properties of chromium-free leathers produced with titanium tanning materials obtained from the wastes of the metal industry. Ekoloji 2014, 23, 83–90. [Google Scholar] [CrossRef]
- China, C.R.; Maguta, M.M.; Nyandoro, S.S.; Hilonga, A.; Kanth, S.V.; Njau, K.N. Alternative tanning technologies and their suitability in curbing environmental pollution from the leather industry: A comprehensive review. Chemosphere 2020, 254, 126804. [Google Scholar] [CrossRef]
- Zhang, C.; Lin, J.; Jia, X.; Peng, B. A salt-free and chromium discharge minimizing tanning technology: The novel cleaner integrated chrome tanning process. J. Clean. Prod. 2016, 112, 1055–1063. [Google Scholar] [CrossRef]
- Yao, Q.; Wang, Y.; Chen, H.; Huang, H.; Liu, B. Mechanism of High Chrome Uptake of Tanning Pickled Pelt by Carboxyl-Terminated Hyper-Branched Polymer Combination Chrome Tanning. ChemistrySelect 2019, 4, 670–680. [Google Scholar] [CrossRef]
- Kanagaraj, J.; Chandra Babu, N.K.; Sadulla, S.; Suseela Rajkumar, G.; Visalakshi, V.; Chandra Kumar, N. Cleaner techniques for the preservation of raw goat skins. J. Clean. Prod. 2001, 9, 261–268. [Google Scholar] [CrossRef]
- Thanikaivelan, P.; Rao, J.R.; Nair, B.U.; Ramasami, T. Recent trends in leather making: Processes, problems, and pathways. Crit. Rev. Environ. Sci. Technol. 2005, 35, 37–79. [Google Scholar] [CrossRef]
- Molden, D.; Amarasinghe, U.; Hussain, I. Water for Rural Development—Background Paper on Water for Rural Development; International Water Management Institute: Anand, India, 2001; Volume 32, ISBN 9290904593. [Google Scholar]
- Ahamed, M.I.N.; Kashif, P.M. Safety Disposal of Tannery Effluent Sludge: Challenges to Researchers- a Review. Int. J. Pharm. Sci. Res. 2014, 5, 733–736. [Google Scholar]
- Ramteke, P.W.; Awasthi, S.; Srinath, T.; Joseph, B. Efficiency assessment of Common Effluent Treatment Plant (CETP) treating tannery effluents. Environ. Monit. Assess. 2010, 169, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Mendoza-Roca, J.A.; Galiana-Aleixandre, M.V.; Lora-García, J.; Bes-Piá, A. Purification of tannery effluents by ultrafiltration in view of permeate reuse. Sep. Purif. Technol. 2010, 70, 296–301. [Google Scholar] [CrossRef]
- Zakmout, A.; Sadi, F.; Portugal, C.A.M.; Crespo, J.G.; Velizarov, S. Tannery effluent treatment by nanofiltration, reverse osmosis and chitosan modified membranes. Membranes 2020, 10, 378. [Google Scholar] [CrossRef]
- Suthanthararajan, R.; Ravindranath, E.; Chitra, K.; Umamaheswari, B.; Ramesh, T.; Rajamani, S. Membrane application for recovery and reuse of water from treated tannery wastewater. Desalination 2004, 164, 151–156. [Google Scholar] [CrossRef]
- Kiril Mert, B.; Kestioglu, K. Application of nanofiltration and reverse osmosis for tanning wastewater. Int. J. Environ. Res. 2014, 8, 789–798. [Google Scholar]
- Fababuj-Roger, M.; Mendoza-Roca, J.A.; Galiana-Aleixandre, M.V.; Bes-Piá, A.; Cuartas-Uribe, B.; Iborra-Clar, A. Reuse of tannery wastewaters by combination of ultrafiltration and reverse osmosis after a conventional physical-chemical treatment. Desalination 2007, 204, 219–226. [Google Scholar] [CrossRef]
- Shenvi, S.S.; Isloor, A.M.; Ismail, A.F. A review on RO membrane technology: Developments and challenges. Desalination 2015, 368, 10–26. [Google Scholar] [CrossRef]
- Sinha, S.; Singh, S.; Mallick, S. Comparative growth response of two varieties of Vigna radiata L. (var. PDM 54 and var. NM 1) grown on different tannery sludge applications: Efects of treated wastewater and ground water used for irrigation. Environ. Geochem. Health 2008, 30, 407–422. [Google Scholar] [CrossRef] [PubMed]
- Byers, B. Zero discharge: A systematic approach to water reuse. Chem. Eng. 1995, 102, 96. [Google Scholar]
- Yang, F.; Huang, Z.; Huang, J.; Wu, C.; Zhou, R.; Jin, Y. Tanning wastewater treatment by ultrafiltration: Process efficiency and fouling behavior. Membranes 2021, 11, 461. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Lin, X.; Kong, X.; Duan, Q.; Wang, P.; Mei, X.; Ma, J. Making waves: Zero liquid discharge for sustainable industrial effluent management. Water 2021, 13, 2852. [Google Scholar] [CrossRef]
- Tong, T.; Elimelech, M. The Global Rise of Zero Liquid Discharge for Wastewater Management: Drivers, Technologies, and Future Directions. Environ. Sci. Technol. 2016, 50, 6846–6855. [Google Scholar] [CrossRef]
- Yaqub, M.; Lee, W. Zero-liquid discharge (ZLD) technology for resource recovery from wastewater: A review. Sci. Total Environ. 2019, 681, 551–563. [Google Scholar] [CrossRef]
- Qiblawey, H.M.; Banat, F. Solar thermal desalination technologies. Desalination 2008, 220, 633–644. [Google Scholar] [CrossRef]
- Bostjancic, J.; Ludlum, R. Getting to Zero Discharge: How to Recycle That Last Bit of Really Bad Wastewater A Brief History of Evaporation. In Proceedings of the International Water Conference, Engineers Society of Western Pennsylvania, West Chester, PA, USA, 21–24 April 1996; Volume 57, pp. 290–295. [Google Scholar]
- Giwa, A.; Dufour, V.; Al Marzooqi, F.; Al Kaabi, M.; Hasan, S.W. Brine management methods: Recent innovations and current status. Desalination 2017, 407, 1–23. [Google Scholar] [CrossRef]
- Boopathy, R.; Karthikeyan, S.; Mandal, A.B.; Sekaran, G. Characterisation and recovery of sodium chloride from salt-laden solid waste generated from leather industry. Clean Technol. Environ. Policy 2013, 15, 117–124. [Google Scholar] [CrossRef]
- Alasfour, F.N.; Abdulrahim, H.K. The effect of stage temperature drop on MVC thermal performance. Desalination 2011, 265, 213–221. [Google Scholar] [CrossRef]
- Griffin, S.J.; Schooley, K.E.; Solomon, R.L. The Advantage of Mixed Salt Crystallizers in Zero Liquid Discharge (ZLD) Wastewater Treatment Systems. GE Water Process Technol. 2021, 12, 998. [Google Scholar]
- Faes, W.; Van Bael, J.; Lecompte, S.; Verbeken, K. Optimization of heat exchanger design taking corrosion into account. Therm. Sci. Eng. Prog. 2022, 30, 101277. [Google Scholar] [CrossRef]
- Wang, C.P.; Wang, H.Z.; Ruan, G.L.; Wang, S.H.; Xiao, Y.X.; Jiang, L.D. Applications and prospects of titanium and its alloys in seawater desalination industry. In Proceedings of the IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2019; Volume 688, p. 33036. [Google Scholar]
- Srithar, K.; Mani, A. Studies on solar flat plate collector evaporation systems for tannery effluent (soak liquor). J. Zhejiang Univ. Sci. 2006, 7, 1870–1877. [Google Scholar] [CrossRef]
- Srithar, K.; Mani, A. Open fibre reinforced plastic (FRP) flat plate collector (FPC) and spray network systems for augmenting the evaporation rate of tannery effluent (soak liquor). Sol. Energy 2007, 81, 1492–1500. [Google Scholar] [CrossRef]
- Al-Karaghouli, A.; Kazmerski, L.L. Energy consumption and water production cost of conventional and renewable-energy-powered desalination processes. Renew. Sustain. Energy Rev. 2013, 24, 343–356. [Google Scholar] [CrossRef]
- Elimelech, M.; Phillip, W.A. The future of seawater desalination: Energy, technology, and the environment. Science 2011, 333, 712–717. [Google Scholar] [CrossRef] [PubMed]
- Shaffer, D.L.; Arias Chavez, L.H.; Ben-Sasson, M.; Romero-Vargas Castrillón, S.; Yip, N.Y.; Elimelech, M. Desalination and reuse of high-salinity shale gas produced water: Drivers, technologies, and future directions. Environ. Sci. Technol. 2013, 47, 9569–9583. [Google Scholar] [CrossRef]
- Prihasto, N.; Liu, Q.F.; Kim, S.H. Pre-treatment strategies for seawater desalination by reverse osmosis system. Desalination 2009, 249, 308–316. [Google Scholar] [CrossRef]
- Loganathan, K.; Chelme-Ayala, P.; Gamal El-Din, M. Pilot-scale study on the treatment of basal aquifer water using ultrafiltration, reverse osmosis and evaporation/crystallization to achieve zero-liquid discharge. J. Environ. Manag. 2016, 165, 213–223. [Google Scholar] [CrossRef]
- Pérez-González, A.; Urtiaga, A.M.; Ibáñez, R.; Ortiz, I. State of the art and review on the treatment technologies of water reverse osmosis concentrates. Water Res. 2012, 46, 267–283. [Google Scholar] [CrossRef]
- Almulla, A.; Eid, M.; Côté, P.; Coburn, J. Developments in high recovery brackish water desalination plants as part of the solution yp water quantity problems. Desalination 2003, 153, 237–243. [Google Scholar] [CrossRef]
- Li, W.; Krantz, W.B.; Cornelissen, E.R.; Post, J.W.; Verliefde, A.R.D.; Tang, C.Y. A novel hybrid process of reverse electrodialysis and reverse osmosis for low energy seawater desalination and brine management. Appl. Energy 2013, 104, 592–602. [Google Scholar] [CrossRef]
- Robertson, A.; Nghiem, L.D. Treatment of High TDS Liquid Waste: Is Zero Liquid Discharge Feasible? J. Water Sustain. 2011, 1, 1–11. [Google Scholar]
- Younos, T. The Economics of Desalination. J. Contemp. Water Res. Educ. 2009, 132, 39–45. [Google Scholar] [CrossRef]
- Mickley, M.C. Membrane Concentrate Disposal: Practices and Regulation; US Department of the Interior, Bureau of Reclamation, Technical Service: Denver, CO, USA, 2006; Volume 123, ISBN 1856173895.
- Pancharatnam, S. Transient Behavior of a Solar Pond and Prediction of Evaporation Rates. Ind. Eng. Chem. Process Des. Dev. 1972, 11, 287–292. [Google Scholar] [CrossRef]
- Ahmed, M.; Shayya, W.H.; Hoey, D.; Mahendran, A.; Morris, R.; Al-Handaly, J. Use of evaporation ponds for brine disposal in desalination plants. Desalination 2000, 130, 155–168. [Google Scholar] [CrossRef]
- Rajkumar, R.; Sathish, S.; Senthilkumar, P. Studies on enhancing the efficiency of ZLD plant for tannery effluent by implementing Low-Cost ambient air evaporator system. Rasayan J. Chem. 2018, 11, 13–17. [Google Scholar] [CrossRef]
- Martínez, J.; León, E.; Baena-Moreno, F.M.; Rodríguez-Galán, M.; Arroyo-Torralvo, F.; Vilches, L.F. Techno-economic analysis of a membrane-hybrid process as a novel low-energy alternative for zero liquid discharge systems. Energy Convers. Manag. 2020, 211, 112783. [Google Scholar] [CrossRef]
- Buljan, J.; Emmanuel, K.V.; Viswanathan, M.; Bosnić, M.; Král, I. Analysis of flow and energy aspects of Zero Liquid Discharge (ZLD) technology in treatment of tannery effluents in Tamil Nadu, India. In Proceedings of the 34th IULTCS Congress: Science and Technology for Sustainability of Leather, Chennai, India, 6 February 2017; pp. 244–259. [Google Scholar]
- Patel, T.; Bagrecha, D. Zero Liquid Discharge. Zero Waste 2019, 2, 13–23. [Google Scholar] [CrossRef]
- CPCB Guidelines on Techno—Economic Feasibility of Implementation of Zero Liquid Discharge (Zld) For Water Polluting Industries. Available online: https://ueppcb.uk.gov.in/downloads/view/28 (accessed on 22 May 2022).
- Greenlee, L.F.; Lawler, D.F.; Freeman, B.D.; Marrot, B.; Moulin, P. Reverse osmosis desalination: Water sources, technology, and today’s challenges. Water Res. 2009, 43, 2317–2348. [Google Scholar] [CrossRef]
- Afonso, M.D.; Jaber, J.O.; Mohsen, M.S. Brackish groundwater treatment by reverse osmosis in Jordan. Desalination 2004, 164, 157–171. [Google Scholar] [CrossRef]
- Ahmed, M.; Shayya, W.H.; Hoey, D.; Al-Handaly, J. Brine disposal from reverse osmosis desalination plants in Oman and the United Arab Emirates. Desalination 2001, 133, 135–147. [Google Scholar] [CrossRef]
- Vigneswaran, V.S.; Ganesh Kumar, P.; Jeyachandran, J.; Britto Sahayaraj, S.; Kumaresan, G. Usage of solar greenhouse evaporator to enhance dehydration and potable water extraction from tannery effluent. Process Saf. Environ. Prot. 2021, 147, 912–923. [Google Scholar] [CrossRef]
- Namasivayam, C. Reutilization of Industrial Solid Wastes for Industrial Effluent Treatments. Freshw. Manag. 2005, 2, 124. [Google Scholar]
- Govindasamy, P.; Madhavan, S.D.; Revathi, S.; Shanmugam, P. Performance evaluation of common effluent treatment plant for tanneries at Pallavaram CETP. J. Environ. Sci. Eng. 2006, 48, 213–220. [Google Scholar]
- TNPCB Details of Common Effluent Treatment Plants pertaining to the Cluster of Tannery Industries in Tamil Nadu. Available online: https://tnpcb.gov.in/cetp.php (accessed on 22 May 2022).
- Mickley, M. Survey of High-Recovery and Zero Liquid Discharge Technologies for Water Utilities; WateReuse Foundation: Alexandria, VA, USA, 2008. Available online: https://www.waterboards.ca.gov/water_issues/programs/grants_loans/water_recycling/research/02_006a_01.pdf (accessed on 22 May 2022).
- United States Environmental Protection Agency Effluent Limitations Guidelines and Standards for the Steam Electric Power Generating Point Source Category; Final Rule, 40 CFR Part 423. Fed. Regist. 2015, 80, 67838–67903.
- Kimberly-clark Economic zero liquid discharge at US chemical facility. Filtr. Sep. 2018, 55, 12. [CrossRef]
- Rajamani, S. Novel industrial wastewater treatment integrated with recovery of water and salt under a zero liquid discharge concept. Rev. Environ. Health 2016, 31, 63–66. [Google Scholar] [CrossRef]
- Panagopoulos, A.; Haralambous, K.J. Minimal Liquid Discharge (MLD) and Zero Liquid Discharge (ZLD) strategies for wastewater management and resource recovery-Analysis, challenges and prospects. J. Environ. Chem. Eng. 2020, 8, 104418. [Google Scholar] [CrossRef]
- Suwaileh, W.; Pathak, N.; Shon, H.; Hilal, N. Forward osmosis membranes and processes: A comprehensive review of research trends and future outlook. Desalination 2020, 485, 114455. [Google Scholar] [CrossRef]
- Fu, J.; Hu, N.; Yang, Z.; Wang, L. Experimental study on zero liquid discharge (ZLD) of FGD wastewater from a coal-fired power plant by flue gas exhausted heat. J. Water Process Eng. 2018, 26, 100–107. [Google Scholar] [CrossRef]
- Nair, M.; Kumar, D. Water desalination and challenges: The Middle East perspective: A review. Desalin. Water Treat. 2013, 51, 2030–2040. [Google Scholar] [CrossRef]
- Rajamani, S.G. Innovative environmental technologies including water recovery for reuse from tannery and industrial wastewater—Indian and Asian scenario. In Proceedings of the International Conference on Advanced Materials and Systems, The National Research & Development Institute for Textiles and Leather-INCDTP, Bucharest, Romania, 20–22 October 2016; pp. 513–518. [Google Scholar]
- Günther, R.; Perschall, B.; Reese, D.; Hapke, J. Engineering for high pressure reverse osmosis. J. Memb. Sci. 1996, 121, 95–107. [Google Scholar] [CrossRef]
- Davenport, D.M.; Deshmukh, A.; Werber, J.R.; Elimelech, M. High-Pressure Reverse Osmosis for Energy-Efficient Hypersaline Brine Desalination: Current Status, Design Considerations, and Research Needs. Environ. Sci. Technol. Lett. 2018, 5, 467–475. [Google Scholar] [CrossRef]
- Subramani, A.; Jacangelo, J.G. Treatment technologies for reverse osmosis concentrate volume minimization: A review. Sep. Purif. Technol. 2014, 122, 472–489. [Google Scholar] [CrossRef]
- Schwantes, R.; Chavan, K.; Winter, D.; Felsmann, C.; Pfafferott, J. Techno-economic comparison of membrane distillation and MVC in a zero liquid discharge application. Desalination 2018, 428, 50–68. [Google Scholar] [CrossRef] [Green Version]
- Zhao, S.; Zou, L.; Tang, C.Y.; Mulcahy, D. Recent developments in forward osmosis: Opportunities and challenges. J. Memb. Sci. 2012, 396, 1–21. [Google Scholar] [CrossRef]
- Zhou, X.; Gingerich, D.B.; Mauter, M.S. Water Treatment Capacity of Forward-Osmosis Systems Utilizing Power-Plant Waste Heat. Ind. Eng. Chem. Res. 2015, 54, 6378–6389. [Google Scholar] [CrossRef]
- Zhu, Y.; Yan, H.; Lu, F.; Su, Y.; Li, W.; An, J.; Wang, Y.; Xu, T. Electrodialytic concentration of landfill leachate effluent: Lab- and pilot-scale test, and assessment. Sep. Purif. Technol. 2021, 276, 119311. [Google Scholar] [CrossRef]
- Morillo, J.; Usero, J.; Rosado, D.; El Bakouri, H.; Riaza, A.; Bernaola, F.J. Comparative study of brine management technologies for desalination plants. Desalination 2014, 336, 32–49. [Google Scholar] [CrossRef]
- Park, H.B.; Freeman, B.D.; Zhang, Z.B.; Sankir, M.; McGrath, J.E. Highly chlorine-tolerant polymers for desalination. Angew. Chemie—Int. Ed. 2008, 47, 6019–6024. [Google Scholar] [CrossRef] [PubMed]

| Parameter | Raw Effluent | Raw Effluent after Primary and Secondary Treatment | RO Feed | RO Permeate | RO Reject |
|---|---|---|---|---|---|
| pH | 5–7.6 | 7.3–8.2 | 6.6–7.3 | 6.5–7.1 | 7.2–7.5 |
| Total suspended solids (mg/L) | 1540–2010 | 55–90 | 2.5 | nil | 2.2–15 |
| Chemical oxygen demand (COD) (mg/L) | 4340–5342 | 260–647 | 190 | 20–BDL | 510–3280 |
| Biochemical oxygen demand (BOD) (mg/L) | 640–1570 | 20–69 | 10 | 1–BDL | 5.2–90 |
| Total dissolved solids (TDS) (mg/L) | 11,800–20,090 | 17,980–9986 | 17830 | 860–388 | 39,210–36,100 |
| Chlorides (mg/L) | 1945–10,880 | 1945–9280 | 8450 | 400–226 | 24,760–9725 |
| Sulfates (mg/L) | 3590–3870 | 2290–3766 | 1710 | 110–9 | 17225 |
| Sulfide (mg/L) | 26–180 | 1.8–2 | 0.1 | BDL | 0.1 |
| Total chromium (mg/L) | 22–120 | 0.2–0.4 | 0.05 | BDL | 0.1 |
| Oil and grease (mg/L) | 12–27 | 2.1–2.4 | 0.5 | BDL | 1.7 |
| Ammonium (mg/L) | 22 | 16.1 | 14 | 3.2 | 31–450 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ricky, R.; Shanthakumar, S.; Ganapathy, G.P.; Chiampo, F. Zero Liquid Discharge System for the Tannery Industry—An Overview of Sustainable Approaches. Recycling 2022, 7, 31. https://doi.org/10.3390/recycling7030031
Ricky R, Shanthakumar S, Ganapathy GP, Chiampo F. Zero Liquid Discharge System for the Tannery Industry—An Overview of Sustainable Approaches. Recycling. 2022; 7(3):31. https://doi.org/10.3390/recycling7030031
Chicago/Turabian StyleRicky, Rajamanickam, Subramanian Shanthakumar, Ganapathy Pattukandan Ganapathy, and Fulvia Chiampo. 2022. "Zero Liquid Discharge System for the Tannery Industry—An Overview of Sustainable Approaches" Recycling 7, no. 3: 31. https://doi.org/10.3390/recycling7030031
APA StyleRicky, R., Shanthakumar, S., Ganapathy, G. P., & Chiampo, F. (2022). Zero Liquid Discharge System for the Tannery Industry—An Overview of Sustainable Approaches. Recycling, 7(3), 31. https://doi.org/10.3390/recycling7030031








