Variation of Used Vegetable Oils’ Composition upon Treatment with Algerian Clays
Abstract
:1. Introduction
2. Results and Discussion
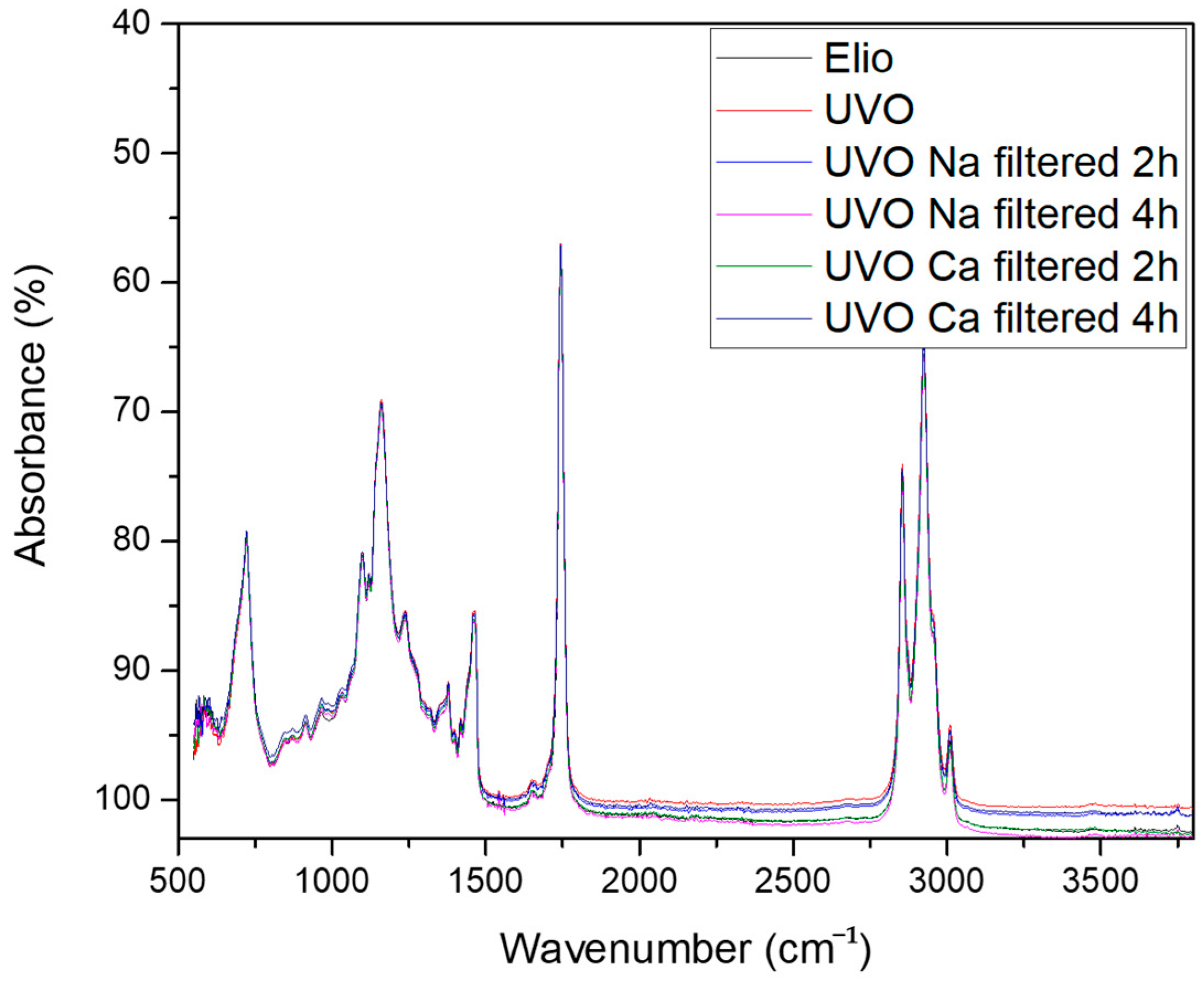
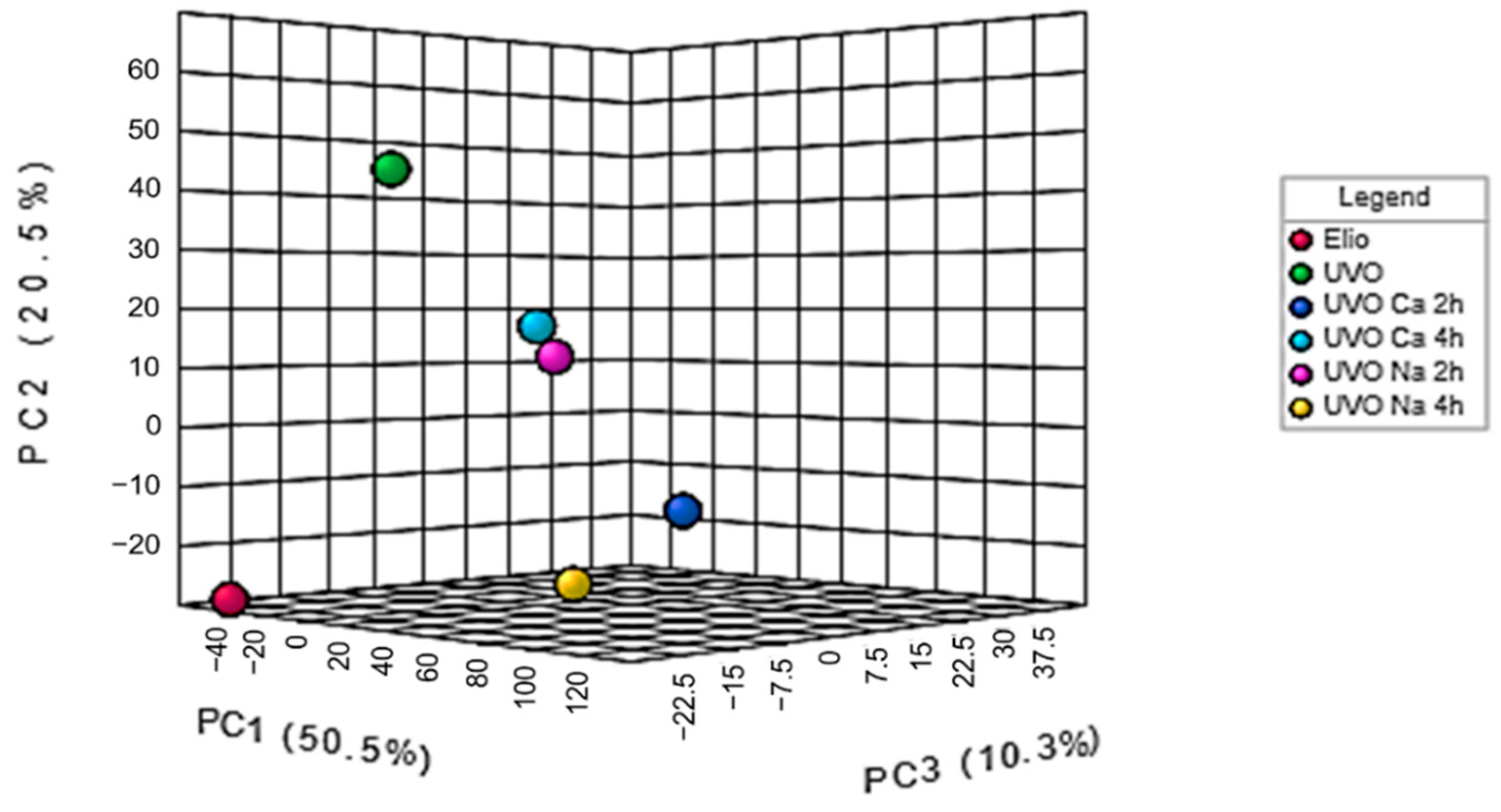
2.1. FT-IR and Principal Component Analysis
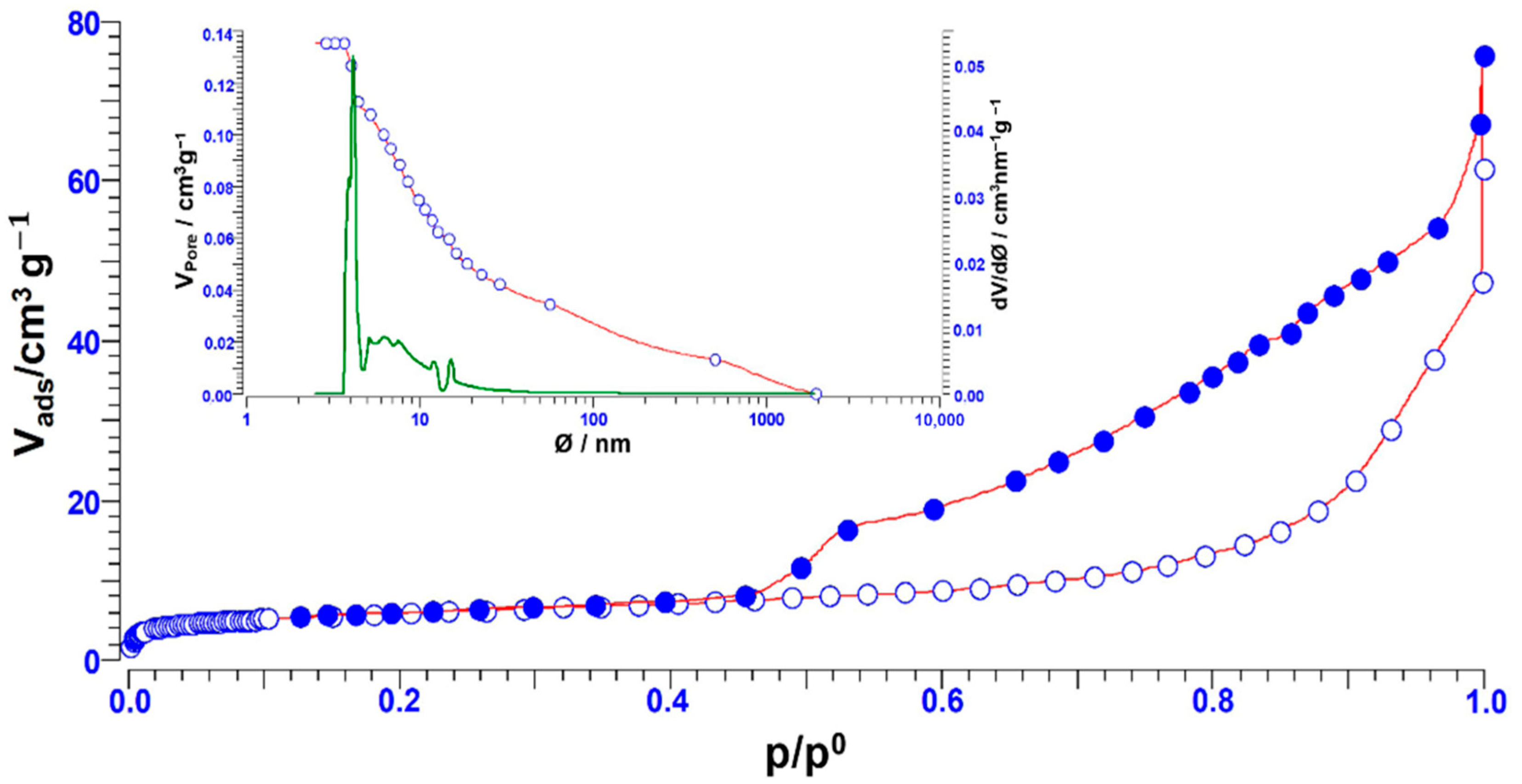
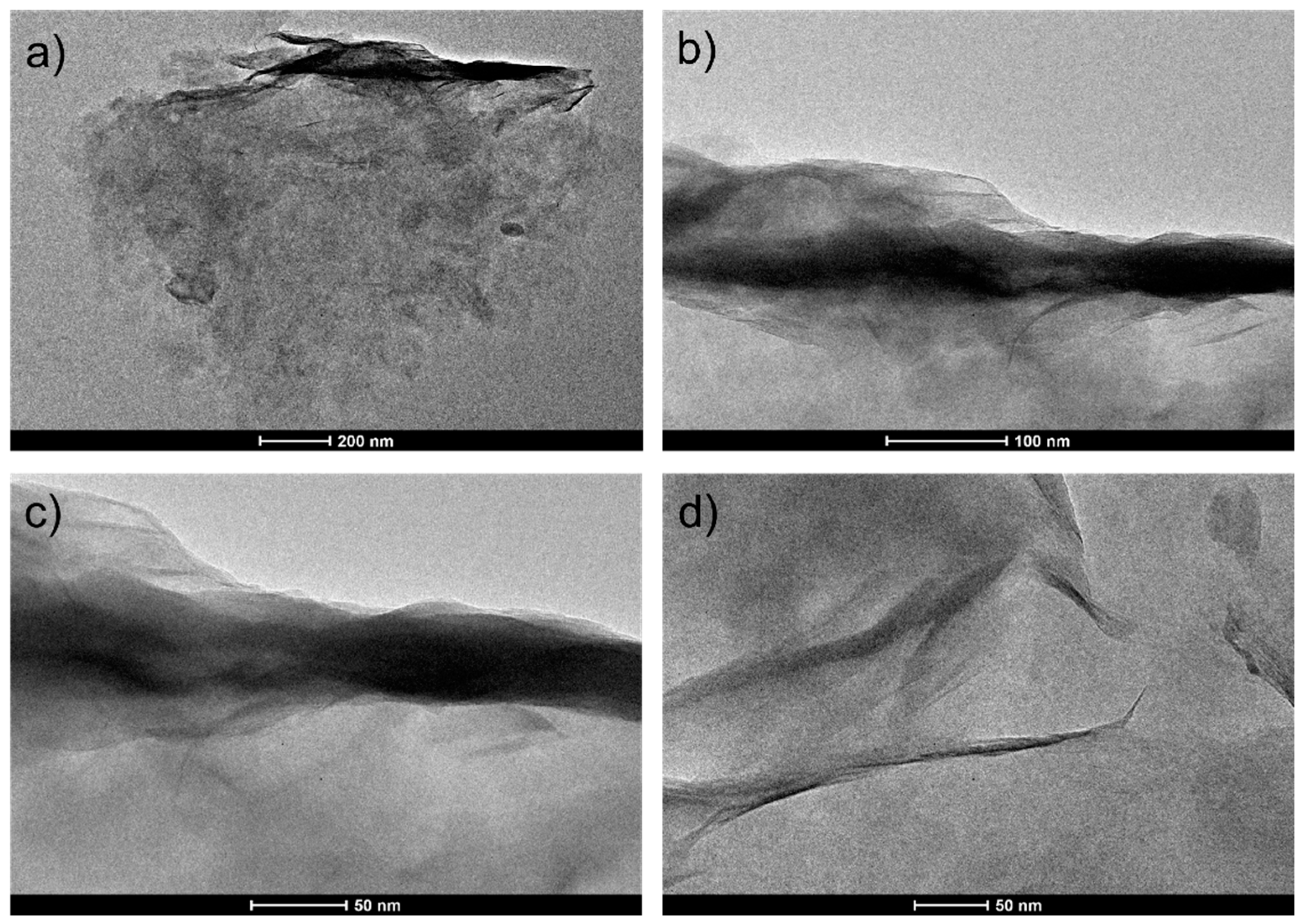
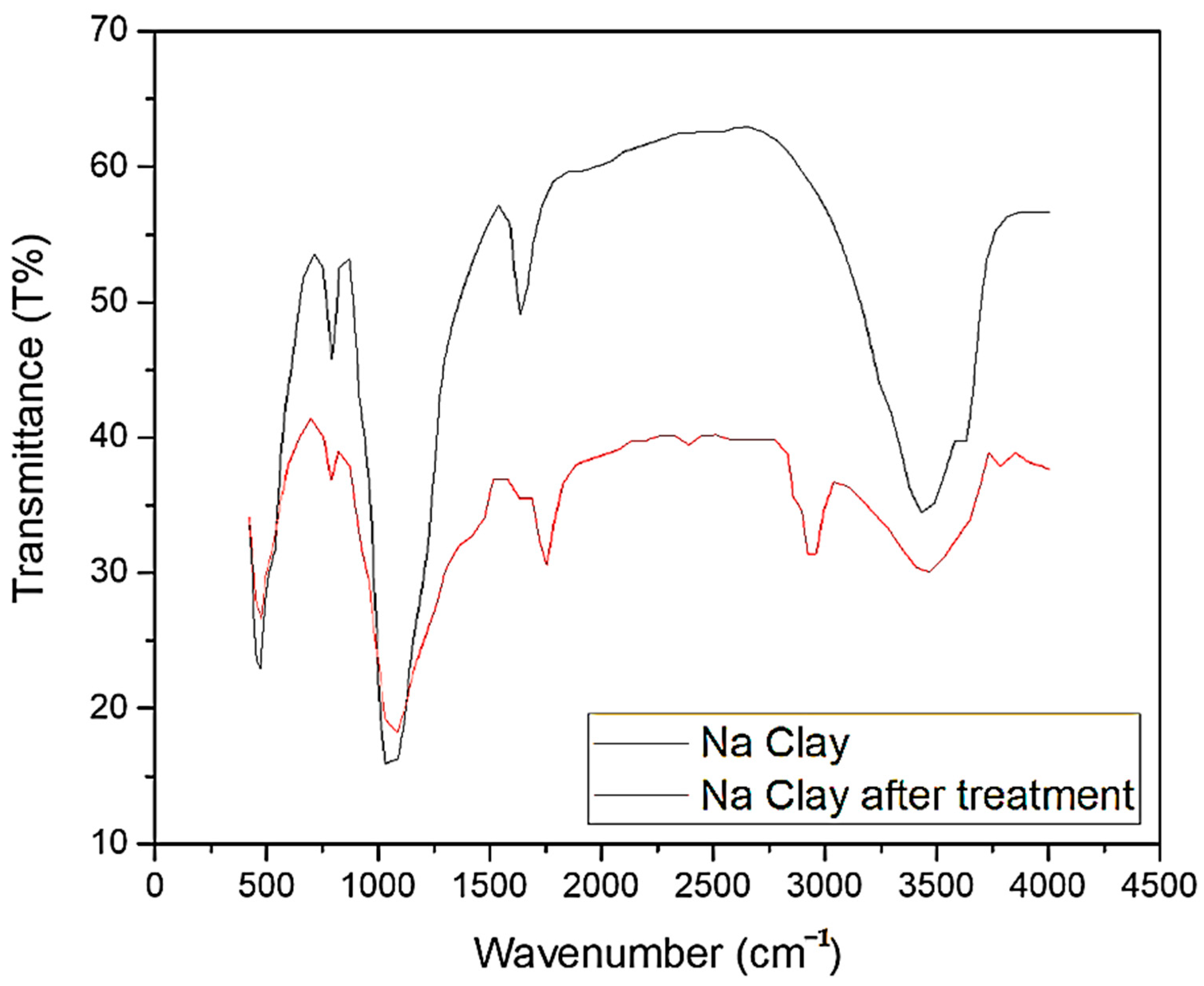
2.2. Characterization of Na-Based Clay
3. Materials and Methods
3.1. Raw Materials
3.2. Physico Chemical Analyses
3.3. Experimental Protocol
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Corona, B.; Shen, L.; Reike, D.; Rosales Carreón, J.; Worrell, E. Towards sustainable development through the circular economy—A review and critical assessment on current circularity metrics. Resour. Conserv. Recycl. 2019, 151, 104498. [Google Scholar] [CrossRef]
- Rodriguez-Anton, J.M.; Rubio-Andrada, L.; Celemín-Pedroche, M.S.; Alonso-Almeida, M.D.M. Analysis of the relations between circular economy and sustainable development goals. Int. J. Sustain. Dev. World Ecol. 2019, 26, 708–720. [Google Scholar] [CrossRef]
- Zeller, V.; Towaa, E.; Degrez, M.; Achten, W.M.J. Urban waste flows and their potential for a circular economy model at city-region level. Waste Manag. 2019, 83, 83–94. [Google Scholar] [CrossRef] [PubMed]
- Awasthi, A.K.; Li, J.; Koh, L.; Ogunseitan, O.A. Circular economy and electronic waste. Nat. Electron. 2019, 2, 86–89. [Google Scholar] [CrossRef] [Green Version]
- Pires, A.; Martinho, G. Waste hierarchy index for circular economy in waste management. Waste Manag. 2019, 95, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Ibanez, J.; Martel Martín, S.; Baldino, S.; Prandi, C.; Mannu, A. European Union Legislation Overview about Used Vegetable Oils Recycling: The Spanish and Italian Case Studies. Processes 2020, 8, 798. [Google Scholar] [CrossRef]
- Mannu, A.; Garroni, S.; Ibanez Porras, J.; Mele, A. Available Technologies and Materials for Waste Cooking Oil Recycling. Processes 2020, 8, 366. [Google Scholar] [CrossRef] [Green Version]
- Mannu, A.; Ferro, M.; Di Pietro, M.E.; Mele, A. Innovative applications of waste cooking oil as raw material. Sci. Prog. 2019, 102, 153–160. [Google Scholar] [CrossRef] [PubMed]
- No, S.Y. Inedible vegetable oils and their derivatives for alternative diesel fuels in CI engines: A review. Renew. Sust. Energy Rev. 2011, 15, 131–149. [Google Scholar] [CrossRef]
- Panadare, D.C.; Rathod, V.K. Applications of Waste Cooking Oil Other Than Biodiesel. Iran. J. Chem. Eng. 2015, 12, 55. [Google Scholar]
- Biermann, U.; Meier, M.A.R.; Metzger, J.O.; Schafer, H.J. Oils and fats as renewable raw materials in chemistry. Angew. Chem. Int. Ed. 2011, 50, 3854. [Google Scholar] [CrossRef] [PubMed]
- Asli, H.; Ahmadinia, E.; Zargar, M.; Karim, M.R. Investigation on physical properties of waste cooking oil. Constr. Build. Mater. 2012, 37, 398. [Google Scholar] [CrossRef]
- Choe, E.; Min, D.B. Chemistry of Deep-Fat Frying Oils. J. Food Sci. 2007, 72, R77–R86. [Google Scholar] [CrossRef] [PubMed]
- Implementation of the Waste Framework Directive. Available online: http://ec.europa.eu/environment/waste/framework/list.htm (accessed on 22 July 2021).
- Di Pietro, M.E.; Mannu, A.; Mele, A. NMR determination of free fatty acids in vegetable oils. Processes 2020, 8, 410. [Google Scholar] [CrossRef] [Green Version]
- Andrikopoulos, N.K.; Boskou, G.; Dedoussis, G.V.Z.; Chiou, A.; Tzamtzis, V.A.; Papathanasiou, A. Quality assessment of frying oils and fats from 63 restaurants in Athens, Greece. Food Serv. Technol. 2003, 3, 49–59. [Google Scholar] [CrossRef]
- Ziaiifar, A.M.; Achir, N.; Courtois, F.; Trezzani, I.; Trystram, G. Review of mechanisms, conditions, and factors involved in the oil uptake phenomenon during the deep-fat frying process. Int. J. Food Sci. Technol. 2008, 43, 1410–1423. [Google Scholar] [CrossRef]
- Lin, C.S.K.; Pfaltzgraff, L.A.; Herrero-Davila, L.; Mubofu, E.B.; Abderrahim, S.; Clark, J.H.; Koutinas, A.A.; Kopsahelis, N.; Stamatelatou, K.; Dickson, F.; et al. Food waste as a valuable resource for the production of chemicals, materials and fuels. Current situation and global perspective. Energy Environ. Sci. 2013, 6, 426–464. [Google Scholar] [CrossRef]
- Hymore, F.K. Effects of some additives on the performance of acid-activated clays in the bleaching of palm oil. Appl. Clay Sci. 1996, 10, 379–385. [Google Scholar] [CrossRef]
- Komadel, P. Acid activated clays: Materials in continuous demand. Appl. Clay Sci. 2016, 131, 84–99. [Google Scholar] [CrossRef]
- Murray, H.H. Overview—Clay mineral applications. Appl. Clay Sci. 1991, 5, 379–395. [Google Scholar] [CrossRef]
- Luna, F.M.T.; Cecilia, J.A.; Saboya, R.M.A.; Barrera, D.; Sapag, K.; Rodríguez-Castellón, E.; Cavalcante, C.L., Jr. Natural and Modified Montmorillonite Clays as Catalysts for Synthesis of Biolubricants. Materials 2018, 1, 1764. [Google Scholar] [CrossRef] [Green Version]
- Richardson, L.L. Use of bleaching, clays, in processing edible oils. J. Am. Oil Chem. Soc. 1978, 55, 777–780. [Google Scholar] [CrossRef]
- Mannu, A.; Vlahopoulou, G.; Urgeghe, P.; Ferro, M.; Del Caro, A.; Taras, A.; Garroni, S.; Rourke, J.P.; Cabizza, R.; Petretto, G.L. Variation of the Chemical Composition of Waste Cooking Oils upon Bentonite Filtration. Resources 2019, 8, 108. [Google Scholar] [CrossRef] [Green Version]
- Taleb, Z.; Ramdani, A.; Berenguer, R.; Ramdani, N.; Adjir, M.; Taleb, S.; Morallón, E.; Nemmich, S.; Tilmatine, A. Combined ozonation process and adsorption onto bentonite natural adsorbent for the o-cresol elimination. Int. J. Environ. Anal. Chem. 2021, 1–18. [Google Scholar] [CrossRef]
- Feddal, I.; Taleb, Z.; Ramdani, A.; Herbache, H.; Taleb, S. Discoloration of contaminated water by an industrial dye: Methylene Blue, by two Algerian bentonites, thermally activated. Alger. J. Environ. Sci. Technol. 2019, 5, 1141. [Google Scholar]
- Alexa, E.; Dragomirescu, A.; Pop, G.; Jianu, C.; Dragoş, D. The use of FT-IR spectroscopy in the identification of vegetable oils adulteration. J. Food Agric. Environ. 2009, 7, 20–24. [Google Scholar]
- Mannu, A.; Ferro, M.; Colombo Dugoni, G.; Panzeri, W.; Petretto, G.L.; Urgeghe, P.; Mele, A. Improving the recycling technology of waste cooking oils: Chemical fingerprint as tool for non-biodiesel application. Waste Manag. 2019, 96, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ferro, M.; Mannu, A.; Panzeri, W.; Theeuwen, C.H.J.; Mele, A. An integrated approach to optimizing cellulose mercerization. Polymers 2020, 12, 1559. [Google Scholar] [CrossRef]
- Mannu, A.; Karabagias, I.K.; Di Pietro, M.E.; Baldino, S.; Karabagias, V.K.; Badeka, A.V. 13C NMR-based chemical fingerprint for the varietal and geographical discrimination of wines. Foods 2020, 9, 1040. [Google Scholar] [CrossRef]
- Sahasrabudhe, S.N.; Rodriguez-Martinez, V.; O’Meara, M.; Farkas, B.E. Density, viscosity, and surface tension of five vegetable oils at elevated temperatures: Measurement and modeling. Int. J. Food Prop. 2017, 20, 1965–1981. [Google Scholar] [CrossRef] [Green Version]
- Varma, R.S. Clay and clay-supported reagents in inorganic synthesis. Tetrahedron 2002, 58, 1235–1255. [Google Scholar] [CrossRef]
- Makhoukhi, B.; Didi, M.A.; Villemin, D.; Azzouz, A. Acid activation of Bentonite for use as a vegetable oil bleaching agent. Grasas Y Aceites 2009, 60, 343–349. [Google Scholar] [CrossRef] [Green Version]
- Taylor, D.R.; Jenkins, D.B.; Ungermann, C.B. Bleaching with alternative layered minerals: A comparison with acid-activated montmorillonite for bleaching soybean oil. J. Am. Oil Chem. Soc. 1989, 66, 334–341. [Google Scholar] [CrossRef]
- Mannu, A.; Vlahopoulou, G.; Sireus, V.; Petretto, G.L.; Mulas, G.; Garroni, S. Bentonite as a Refining Agent in Waste Cooking Oils Recycling: Flash Point, Density and Color Evaluation. Nat. Prod. Commun. 2018, 13, 613–616. [Google Scholar] [CrossRef] [Green Version]
- Jalil, M.E.R.; Vieira, R.S.; Azevedo, D.; Baschini, M.; Sapag, K. Improvement in the adsorption of thiabendazole by using aluminum pillared clays. Appl. Clay Sci. 2013, 71, 55–63. [Google Scholar] [CrossRef]
- Song, K.; Sandi, G. Characterization of Montmorillonite Surfaces After Modification by Organosilane. Clays Clay Miner. 2001, 49, 119. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. The Solubility of Oil-Soluble Dyes in Aqueous Solutions of Stable Protecting Colloids as Examples of True Reversible Equilibrium. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Lomić, G.A.; Kiš, E.E.; Dimić, E.B.; Romanić, R. Investigation of activated Al pillared clay efficiency in vegetable oil purification. Acta Period. Technol. 2014, 35, 31–36. [Google Scholar]
| EO | UVO | Ca Treated | Na Treated | |||
|---|---|---|---|---|---|---|
| 2 h | 4 h | 2 h | 4 h | |||
| pH | 5.507 | 5.104 | 7.05 | 6.75 | 6.68 | 6.39 |
| 0.1 | 0.1 | 1.23 | 0.8 | 1.7 | 0.9 | |
| TDS | 0 | 0 | 1 | 1 | 2 | 1 |
| salinity 1 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| turbidity (UTN) | 0 | 69 | 26 | 57 | 37 | 14 |
| density | 0.938 | 0.9048 | 0.9221 | 0.9212 | 0.9215 | 0.9224 |
| Elements | Na Clays Weight % |
|---|---|
| C | 23.84 |
| O | 42.94 |
| Na | 2.72 |
| Mg | 2.85 |
| Al | 7.19 |
| Si | 19.43 |
| K | 0.16 |
| Fe | 0.83 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serouri, A.; Taleb, Z.; Mannu, A.; Garroni, S.; Senes, N.; Taleb, S.; Brini, S.; Abdoun, S.K. Variation of Used Vegetable Oils’ Composition upon Treatment with Algerian Clays. Recycling 2021, 6, 68. https://doi.org/10.3390/recycling6040068
Serouri A, Taleb Z, Mannu A, Garroni S, Senes N, Taleb S, Brini S, Abdoun SK. Variation of Used Vegetable Oils’ Composition upon Treatment with Algerian Clays. Recycling. 2021; 6(4):68. https://doi.org/10.3390/recycling6040068
Chicago/Turabian StyleSerouri, Abdelhak, Zoubida Taleb, Alberto Mannu, Sebastiano Garroni, Nina Senes, Safia Taleb, Sara Brini, and Sabrine Kawther Abdoun. 2021. "Variation of Used Vegetable Oils’ Composition upon Treatment with Algerian Clays" Recycling 6, no. 4: 68. https://doi.org/10.3390/recycling6040068
APA StyleSerouri, A., Taleb, Z., Mannu, A., Garroni, S., Senes, N., Taleb, S., Brini, S., & Abdoun, S. K. (2021). Variation of Used Vegetable Oils’ Composition upon Treatment with Algerian Clays. Recycling, 6(4), 68. https://doi.org/10.3390/recycling6040068








