Prospects of Red King Crab Hepatopancreas Processing: Fundamental and Applied Biochemistry
Abstract
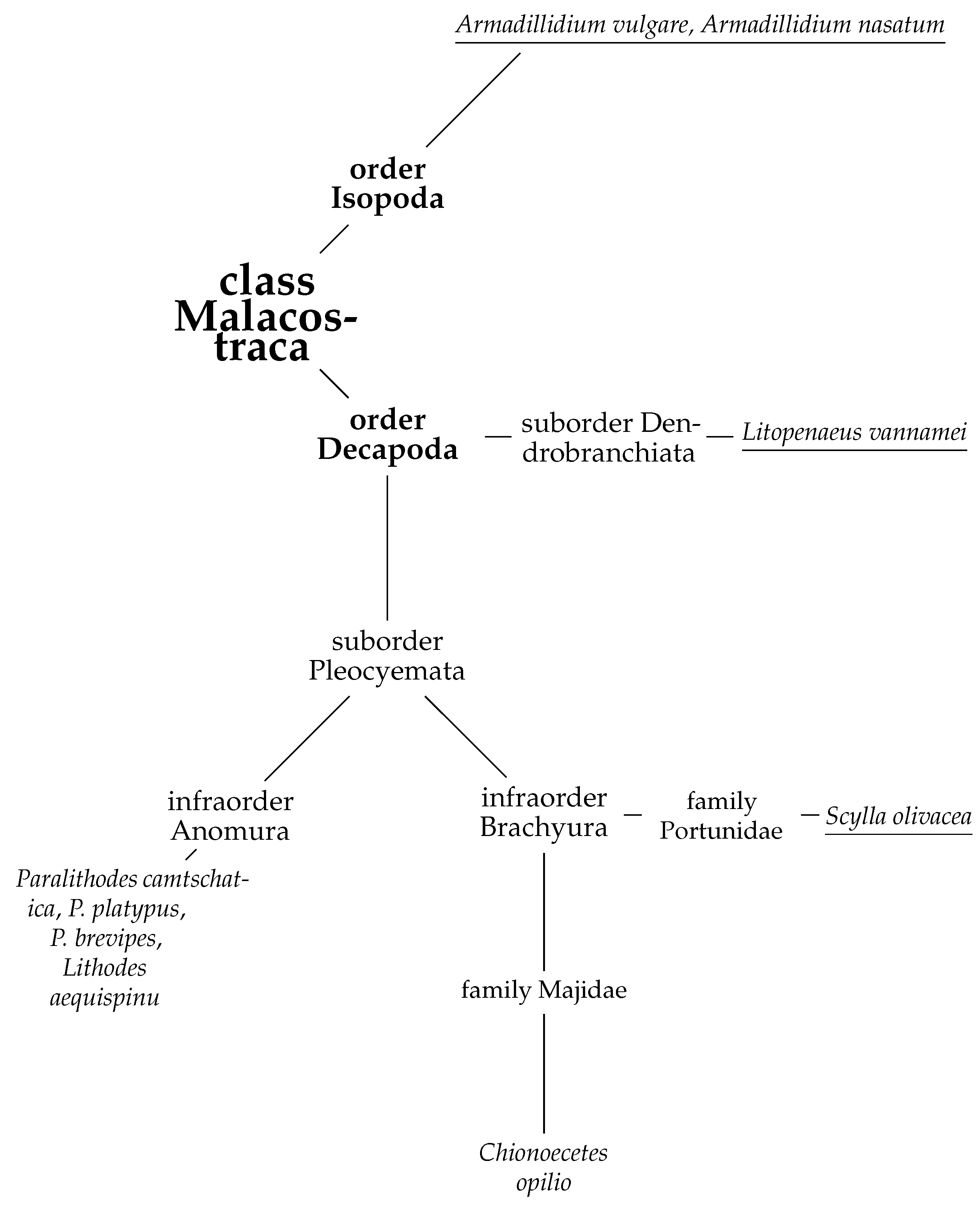
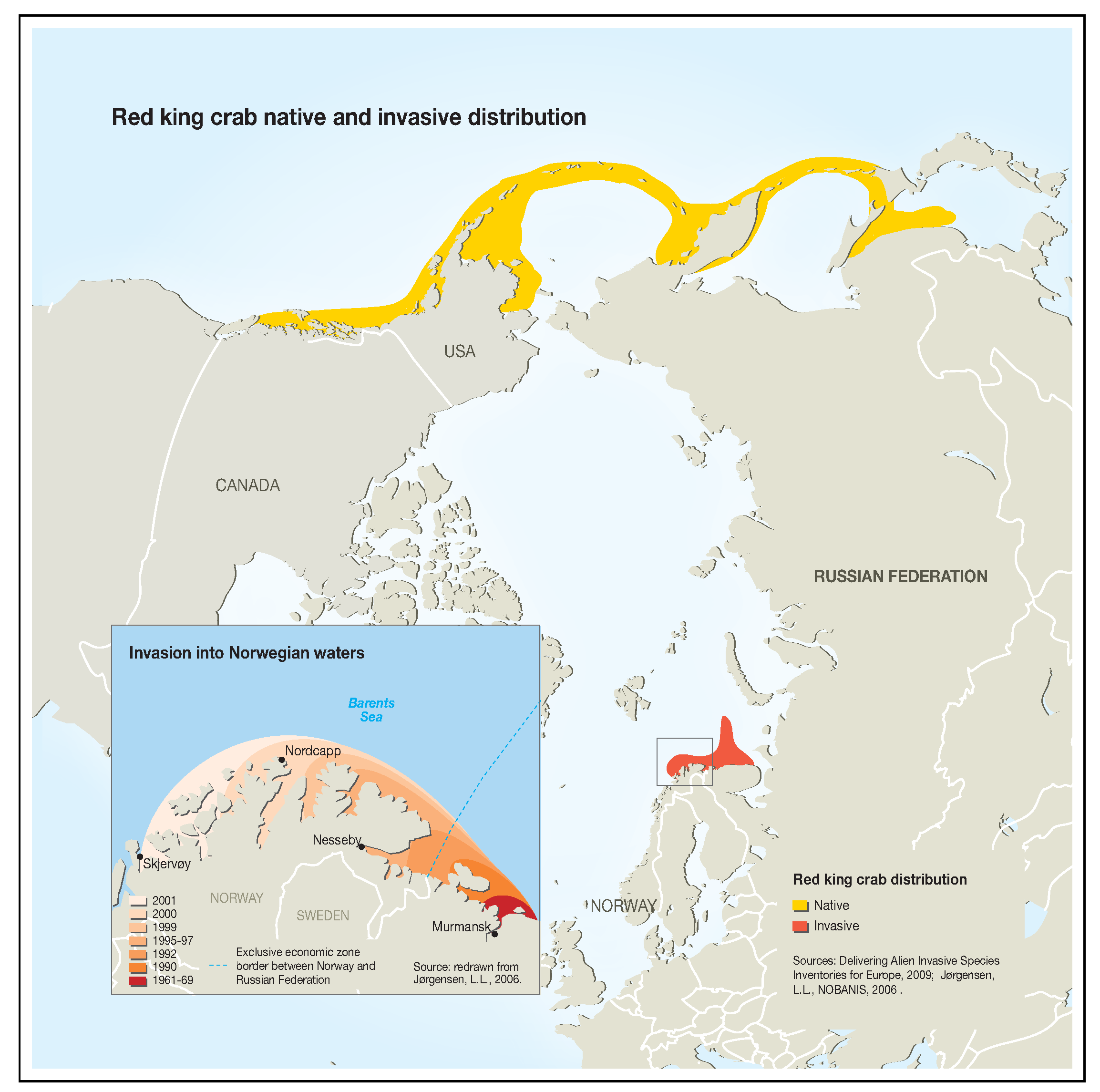
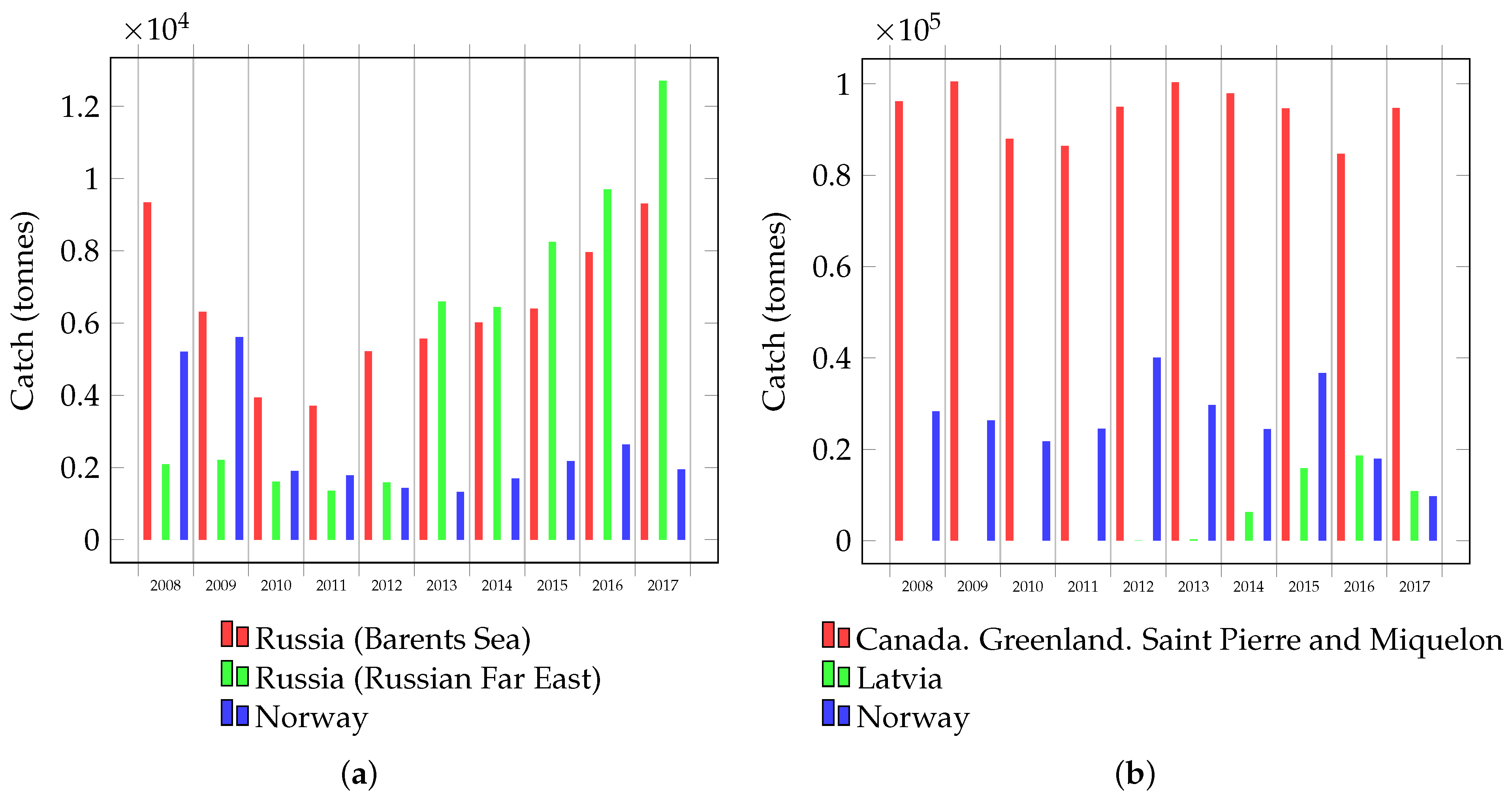
1. Introduction
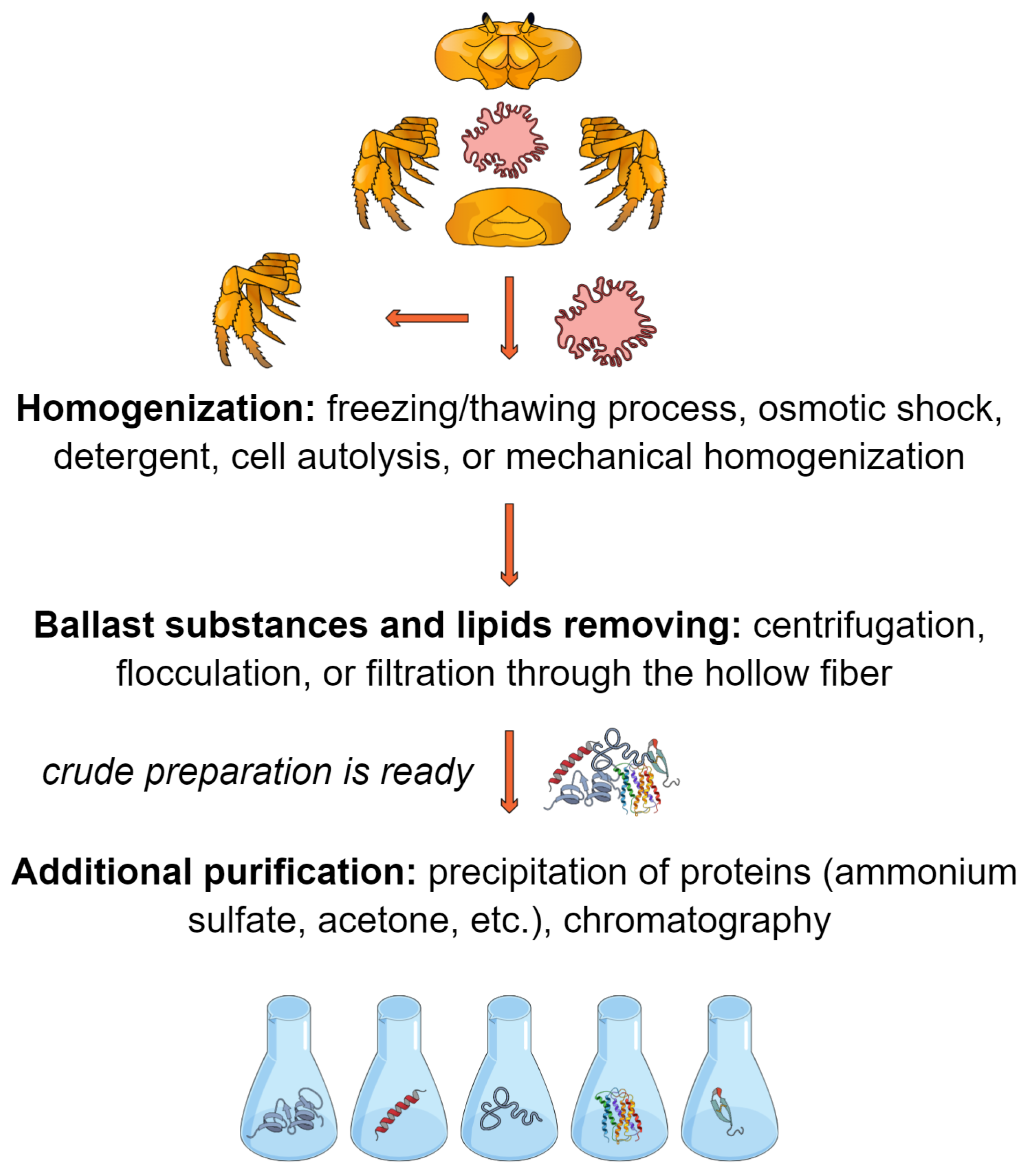
2. Materials and Methods
3. Results
3.1. Crab Hepatopancreas
3.2. Use of Red King Crab Hepatopancreatic Enzymes
3.3. Hepatopancreas Recycling Technologies
3.4. Enzymes of the Red King Crab Hepatopancreas
3.4.1. Proteolytic Enzymes
3.4.2. Nucleases and Other Enzymes of Hepatopancreas
3.4.3. Hyaluronidase Activity of Hepatopancreas Homogenate
3.5. Other Valuable Non-Protein Components of the Red King Crab Hepatopancreas
4. Discussion
4.1. Prospects of Processing Waste from Other Commercial Crab Species
4.2. Development Strategy for Waste Processing
5. Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Slizkin, A.; Safronov, S. Commercial crabs of Kamchatka waters. Petropavlovsk Kamchatskii Sev. Patsifika 2000, 180, 12. [Google Scholar]
- Taxonomy Browser (Root)-NCBI-NIH. Available online: https://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?mode=Undef&id=6681&lvl=3&lin=f&keep=1&srchmode=1&unlock (accessed on 16 December 2020).
- Tavares, M. True crabs. In FAO Species Identification Sheets for Fishery Purposes: Western Central Atlantic (Fishing Area 31); FAO: Rome, Italy, 2003; pp. 327–352. [Google Scholar]
- Garth, J.S.; Abbott, D.P. Brachyura: The True Crabs. In Intertidal Invertebrates of California; Stanford University Press: Stanford, CA, USA, 1980; pp. 594–630. [Google Scholar]
- Steneck, R.S. Functional Groups. In Encyclopedia of Biodiversity, 2nd ed.; Levin, S.A., Ed.; Academic Press: Waltham, MA, USA, 2001. [Google Scholar] [CrossRef]
- Maksimova, S.; Poleschuk, D.; Surovtseva, E.; Vereshchagina, K.; Milovanov, A. King crab wastes potential as the technological valuable raw materials. Food Ind. 2019, 4. [Google Scholar] [CrossRef]
- Dvoretsky, A.G.; Dvoretsky, V.G. Red king crab (Paralithodes camtschaticus) fisheries in Russian waters: Historical review and present status. Rev. Fish Biol. Fish. 2018, 28, 331–353. [Google Scholar] [CrossRef]
- Matishov, G. Marine Ecosystems and Communities in the Conditions of Current Climate Changes; MMBI KSC RAS: St. Petersburg, Russia, 2014. [Google Scholar]
- Podkorytova, A.; Strokova, N.; Semikova, N. Complex processing of Kamchatka crab in the production of food products and biologically active substances. Tr. VNIRO 2018, 172, 198–212. [Google Scholar] [CrossRef]
- Nemtsev, S. Comprehensive Technology for Obtaining Chitin and Chitosan from Crustacean Shell; VNIRO: Moscow, Russia, 2006. [Google Scholar]
- Šimat, V.; Elabed, N.; Kulawik, P.; Ceylan, Z.; Jamroz, E.; Yazgan, H.; Čagalj, M.; Regenstein, J.M.; Özogul, F. Recent Advances in Marine-Based Nutraceuticals and Their Health Benefits. Mar. Drugs 2020, 18, 627. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.B.; Wang, S.L. Reclamation of marine chitinous materials for the production of α-glucosidase inhibitors via microbial conversion. Mar. Drugs 2017, 15, 350. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.B.; Nguyen, D.N.; Nguyen, A.D.; Ngo, V.A.; Ton, T.Q.; Doan, C.T.; Pham, T.P.; Tran, T.P.H.; Wang, S.L. Utilization of Crab Waste for Cost-Effective Bioproduction of Prodigiosin. Mar. Drugs 2020, 18, 523. [Google Scholar] [CrossRef]
- Casadidio, C.; Peregrina, D.V.; Gigliobianco, M.R.; Deng, S.; Censi, R.; Di Martino, P. Chitin and chitosans: Characteristics, eco-friendly processes, and applications in cosmetic science. Mar. Drugs 2019, 17, 369. [Google Scholar] [CrossRef]
- Hulme, P.E. DAISIE (Delivering Alien Invasive Species Inventories for Europe). Handbook of Alien Species in Europe; Springer: Dordrecht, The Netherlands, 2009. [Google Scholar]
- Jørgensen, L.L. NOBANIS—Invasive Alien Species Fact Sheet—Paralithodes camtschaticus.—From: Online Database of the European Network on Invasive Alien Species—NOBANIS www.nobanis.org. 2013. Available online: https://www.nobanis.org/globalassets/speciesinfo/p/paralithodes-camtschatica/paralithodes_camtschaticus.pdf (accessed on 19 October 2020).
- Gibson, R. The decapod hepatopancreas. Oceanogr. Mar. Biol. Ann. Rev. 1979, 17, 285–346. [Google Scholar]
- Kas’janov, S.; Kuklev, D.; Kucheravenko, K.; Blinov, J.; Akulin, V. Method of Extracting Fat from Crab Liver. RU 2162648 C2, Filed 7 April 1999, and Issued 10 February 2001. Patent Holder Pacific Branch of VNIRO (TINRO).
- Muranova, T.; Zinchenko, D.; Melanyina, L.; Miroshnikov, A. Hydrolysis of soybean proteins with Red king crab hepatopancreas enzyme complex. Appl. Biochem. Microbiol. 2018, 54, 74–81. [Google Scholar] [CrossRef]
- Sova, V. Method for Preparing Caviar from Salmon Species Fish. RU 2111681 C1, Filed 12 March 2003, and Issued 10 April 2004. Patent Holder Sova, V.
- Lyzhov, I.; Rysakova, K.; Mukhin, V.; Novikov, V.; Shironina, A. Applying of snow crab hepatopancreas for obtaining of protein hydrolysates from cod filleting wastes. In Proceedings of the Biological Resources of the White Sea and Inland Waters of the European North, Materials of the XXVIII International Conference, Petrozavodsk, Russia, 5–8 October 2009; pp. 337–340. [Google Scholar]
- Mukhin, V.; Novikov, V. Enzymatic hydrolysis of proteins from crustaceans of the Barents Sea. Appl. Biochem. Microbiol. 2001, 37, 538–542. [Google Scholar] [CrossRef]
- Novikov, V.; Portsel, M. Method of Producing Chondroitin Sulphate From Sea Hydrobiont Tissue. RU 2458134 C1, Filed 27 December 2010, and Issued 10 August 2012. Patent Holder POLAR Branch of VNIRO (PINRO).
- Glyantsev, S.; Vishnevsky, A.; Adamyan, A.; Vishnevsky, A.; Sakharov, I. Crab collagenase in wound debridement. J. Wound Care 1997, 6, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Sakharov, I.; Glyanzev, S.; Litvin, F.; Savvina, T. Potent debriding ability of collagenolytic protease isolated from the hepatopancreas of the king crab Paralithodes camtschatica. Arch. Dermatol. Res. 1993, 285, 32–35. [Google Scholar] [CrossRef] [PubMed]
- Rudenskaya, G. Brachyurins—Serine collagenolytic enzymes from crabs. Bioorg. Khim. 2003, 29, 117–128. [Google Scholar]
- Belov, A.; Filatov, V.; Belova, E.; Filatov, N. Medical Bandage Containing Proteolytic Enzyme Complex Including Collagenolytic Proteases from Crab Hepatopancreas. RU 2268751 C2, Filed 25 April 2003, and Issued 27 January 2006. Patent Holders Belov, A.; Filatov, V.; Belova, E.; Filatov, N.
- Belov, A.; Filatov, V.; Belova, E. A Medical Dressing Containing a Complex of Enzymes from the Hepatopancreas of a Crab, and a Method for Its Production. RU 2323748 C2, Filed 21 February 2006, and Issued 10 May 2008. Patent Holders Belov, A.; Filatov, V.; Belova, E.
- Sova, V.; Strongyn, A.; Klimovava, O.; Stadnikov, V. A Collagenolytic Activity Exhibiting Enzyme Complex and the Method for Isolation and Purification Thereof. EP 0402321 A1, Filed 8 June 1990, and Issued 12 December 1990, Patent Holder SEATEC.
- Artjukov, A.; Sakharov, I. Method for Production of Collagenase. RU 2039819 C1, Filed 29 March 1990, and Issued 20 July 1995. Patent holders Artjukov, A.; Sakharov, I.
- Majorov, A.; Albulov, A.; Samujlenko, A. Method of Collagenase Preparing. RU 2112036 C1, Filed 9 October 1992, and Issued 27 May 1998. Patent Holders Majorov, A.; Albulov, A.; Samujlenko, A.
- Pivnenko, T.; Zhdanjuk, V.; Ehpshtejn, L. Method of Preparing of Proteolytic Complex. RU 2034028 C1, Filed 7 February 1992, and Issued 30 April 1995. Patent Holder “Tikhookeanskij nauchno-issledovatel’skij institut rybnogo khozjajstva i okeanografii”.
- Artjukov, A.; Menzorova, N.; Kozlovskaja, E.; Kofanova, N.; Kozlovskij, A.; Rasskazov, V. Enzyme Preparation from Hepatopancreas of Commercial Crab Species and Method for Production of the Same. RU 2280076 C1, Filed 6 December 2004, and Issued 20 July 2006. Patent Holder “Tikhookeanskij institut bioorganicheskoj khimii Dal’nevostochnogo otdelenija Rossijskoj Akademii nauk”.
- Kireev, V.; Titov, O.; Sheveleva, O. Method for Preparing Enzyme Preparation from Crab Hepatopancreas. RU 2285041 C1, Filed 14 April 2005, and Issued 10 October 2006. Patent Holder “FGUP Poljarnyj nauchno-issledovatel’skij institut morskogo rybnogo khozjajstva i okeanografii im. N.M. Knipovicha (FGUP PINRO)”.
- Stadnikov, V.; Erkhov, S. Method of Preparing Enzyme Preparation Showing Collagenolytic Activity. RU 2096456 C1, Filed 10 November 1996, and Issued 20 November 1997. Patent Holders Stadnikov, V.; Erkhov, S.
- Erkhov, S. Biologically Active Substance, Raw Material and Method of Its Preparing. RU 2132876 C1, Filed 13 October 1998, and Issued 10 July 1999. Patent Holder Erkhov, S.
- Sakharov, I.; Artyukov, A.; Berezin, V. Method of Purification of Collagenase. SU 1699350 A3, filed 29 March 1990, and issued 15 December 1991.
- Isaev, V.; Rudenskaja, G.; Kupenko, O.; Stepanov, V.; Popova, I.; Didenko, J. Method of Collagenase Preparation Producing. RU 2008353 C1, Filed 5 August 1991 and Issued 28 February 1994. Patent Holder “NAUCHNO-PROIZVODSTVENNOE PREDPRIJATIE “TRINITA””.
- Sandakhchiev, L.; Danilov, A.; Malygin, E.; Zinov’ev, V.; Ovechkina, L.; Zakabunin, A.; Mistjurin, J. Method of Collase Purification. RU 2121503 C1, Filed 5 Spetember 1996, and Issued 10 November 1998. Patent Holder “Gosudarstvennyj nauchnyj tsentr virusologii i biotekhnologii “Vektor””.
- Isaev, V.; Rudenskaja, G.; Shmojlov, A. Method for Preparing Collagenase Preparation. RU 2225441 C1, Filed 5 March 2003, and Issued 10 March 2004. Patent Holder “Zakrytoe aktsionernoe obshchestvo Nauchno-proizvodstvennoe predprijatie “Trinita””.
- Sakharov, I.; Dzhunkovskaya, A.; Artyukov, A.; Sova, V.; Sakandelidze, O.; Kozlovskaya, E. Method of Producing Collagenase. SU 1343591 A1, Filed 13 December 1985, and Issued 30 April 1992.
- Klimova, O.; Chebotarev, V. Collagenolytic complex of proteases from the hepatopancreas of the Kamchatka crab: Enzymologic activity of the individual components. Biulleten’eksperimental’noi Biologii I Meditsiny 1999, 128, 391–396. [Google Scholar] [CrossRef]
- Štancl, J.; Skočilas, J.; Landfeld, A.; Žitnỳ, R.; Houška, M. Electrical and thermodynamic properties of a collagen solution. Acta Polytech. 2017, 57, 229–234. [Google Scholar] [CrossRef]
- Rudenskaia, G.; Isaev, V.; Stepanov, V.; Dunaevskii, I.; Baratova, L.; Kalebina, T.; Nurminskaia, M. Isolation and properties of serine PC proteinase from the Kamchatka crab, Paralithodes camtschatica—A proteolytic enzyme with broad specificity. Biochem. Mosc. 1996, 61, 1119. [Google Scholar]
- Rudenskaya, G.N.; Kislitsin, Y.A.; Rebrikov, D.V. Collagenolytic serine protease PC and trypsin PC from king crab Paralithodes camtschaticus: CDNA cloning and primary structure of the enzymes. BMC Struct. Biol. 2004, 4, 2. [Google Scholar] [CrossRef]
- Papisova, A.; Semenova, S.; Kislitsyn, Y.; Rudenskaya, G. Peculiarities of substrate hydrolysis by endopeptidases from hepatopancreas of king crab. Russ. J. Bioorg. Chem. 2008, 34, 428–434. [Google Scholar] [CrossRef]
- Semenova, S.; Rudenskaya, G.; Lyutova, L.; Nikitina, O. Isolation and properties of collagenolytic serine proteinase isoenzyme from king crab Paralithodes camtschatica. Biochem. Mos. 2008, 73, 1125–1133. [Google Scholar] [CrossRef] [PubMed]
- Sakharov, I.; Litvin, F.; Artyukov, A.; Kofanova, N. Purification and characterization of collagenolytic protease from the Paralithodes camtschatica hepatopancreas. Biochem. Mos. 1988, 53, 1844–1849. [Google Scholar]
- Sakharov, I.Y.; Litvin, F.E.; Artyukov, A.A. Purification and characterization of two serine collagenolytic proteases from crab Paralithodes camtschatica. Comp. Biochem. Physiol. Part B Comp. Biochem. 1994, 108, 561–568. [Google Scholar] [CrossRef]
- Sakharov, I.Y.; Litvin, F.E.; Artyukov, A.A. Some physico-chemical properties of collagenolytic protease C of the crab (Paralithodes camtschatica). Biochem. Mosc. 1992, 57, 40–45. [Google Scholar]
- Rudenskaya, G.; Shmoilov, A.; Isaev, V.; Ksenofontov, A.; Shvets, S. Aminopeptidase PC from the hepatopancreas of the Kamchatka crab Paralithodes camtschatica. Biochem. Mos. 2000, 65, 164–170. [Google Scholar]
- Rudenskaia, G.; Kupenko, O.; Isaev, V.; Stepanov, V.; Dunaevskii, I. Isolation and properties of carboxypeptidase from the Kamchatka crab Paralithodes camtshatica. Russ. J. Bioorg. Chem. 1995, 21, 249–255. [Google Scholar]
- Rudenskaia, G.; Isaev, V.; Kalebina, T.; Stepanov, V.; Maltsev, K.; Shvets, S.; Lukianova, N.; Kislitsin, I.; Miroshnikov, A. Isolation of trypsin PC from the Kamchatka crab Paralithodes camtschatica and its properties. Russ. J. Bioorg. Chem. 1998, 24, 112–118. [Google Scholar]
- Kislitsyn, I.; Rebrikov, D.; Dunaevskii, I.; Rudenskaia, G. Isolation and primary structure of trypsin from the red king crab Paralithodes camtschaticus. Russ. J. Bioorg. Chem. 2003, 29, 269–276. [Google Scholar] [CrossRef]
- Dzhunkovskaia, A.; Zakharova, N. Pancreatic elastase is a new serine protease isolated from the hepatopancreas of the king crab. In Proceedings of the Abstracts of the All-Union Meeting Biologically Active Substances of Aquatic Organisms—New Medicinal, Therapeutic and Prophylactic and Technical Preparations, Vladivostok, Russia, 23–27 September 1991; p. 8. [Google Scholar]
- Sakharov, I.; Dzhunkovkaia, A. Elastase from the hepatopancreas of the king crab. Biochem. Mos. 1993, 58, 1445–1453. [Google Scholar]
- Sakharov, I.Y.; Dzunkovskaya, A.V.; Artyukov, A.A.; Zakharova, N.N. Purification and some properties of elastase from hepatopancreas of king crab Paralithodes camtschatica. Comp. Biochem. Physiol. Part B Comp. Biochem. 1993, 106, 681–684. [Google Scholar] [CrossRef]
- Isaev, V.; Balashova MV, S.D.; Shagin, D.; Shagina, I.; Eremeev, N.; Rudenskaya, G. New psychrophilic cathepsin L from the hepatopancreas of the red king crab (Paralithodes camtschaticus). Sci. Rev. Biol. Sci. 2016, 6, 81–89. [Google Scholar]
- Semenova, S.A.; Rudenskaya, G.N.; Rebrikov, D.V.; Isaev, V.A. cDNA cloning, purification and properties of Paralithodes camtschatica metalloprotease. Protein Pept. Lett. 2006, 13, 571–575. [Google Scholar] [CrossRef] [PubMed]
- Menzorova, N.; Markova, A.; Rasskazov, V. Highly stable Ca2+, Mg2+-dependent DNAase from crab hepatopancreas. Biochem. Mos. 1994, 59, 449–456. [Google Scholar]
- Vakorina, T.; Menzorova, N.; Rasskazov, V. Study of conformational stability of DNAse from hepatopancreas of the crab Paralithodes camtschatica. Biochem. Mos. 1997, 62, 1642–1647. [Google Scholar]
- Anisimova, V.E.; Rebrikov, D.V.; Shagin, D.A.; Kozhemyako, V.B.; Menzorova, N.I.; Staroverov, D.B.; Ziganshin, R.; Vagner, L.L.; Rasskazov, V.A.; Lukyanov, S.A.; et al. Isolation, characterization and molecular cloning of duplex-specific nuclease from the hepatopancreas of the Kamchatka crab. BMC Biochem. 2008, 9, 14. [Google Scholar] [CrossRef]
- Shagin, D.A.; Rebrikov, D.V.; Kozhemyako, V.B.; Altshuler, I.M.; Shcheglov, A.S.; Zhulidov, P.A.; Bogdanova, E.A.; Staroverov, D.B.; Rasskazov, V.A.; Lukyanov, S. A novel method for SNP detection using a new duplex-specific nuclease from crab hepatopancreas. Genome Res. 2002, 12, 1935–1942. [Google Scholar] [CrossRef]
- Zhulidov, P.A.; Bogdanova, E.A.; Shcheglov, A.S.; Vagner, L.L.; Khaspekov, G.L.; Kozhemyako, V.B.; Matz, M.V.; Meleshkevitch, E.; Moroz, L.L.; Lukyanov, S.A. Simple cDNA normalization using kamchatka crab duplex-specific nuclease. Nucleic Acids Res. 2004, 32, e37. [Google Scholar] [CrossRef]
- Lukyanov, S.A.; Rebrikov, D.V.; Shagin, D.A. Methods and Compositions for Selectively Cleaving Dna Containing Duplex Nucleic Acids in a Complex Nucleic Acid Mixture, and Nuclease Compositions for Use in Practicing the Same. US 7435794 B2, Filed 5 December 2004, and Issued 14 October 2008.
- Shagina, I.; Bogdanova, E.; Al’tshuler, I.; Luk’ianov, S.; Shagin, D. Application of the duplex-specific nuclease for fast analysis of single nucleotide polymorphisms and detection of target DNA in complex PCR products. Bioorg. Khim. 2011, 37, 522–529. [Google Scholar]
- Menzorova, N.I.; Sibirtsev, J.T.; Rasskazov, V.A. Ribonuclease from the Hepatopancreas of the Red King Crab Paralithodes camtschatica. Appl. Biochem. Microbiol. 2009, 45, 369–373. [Google Scholar] [CrossRef]
- Menzorova, N.; Ivleva, A.; Sibirtsev, Y.T.; Rasskazov, V. Phosphatases and phosphodiesterases isolated from the red king crab (Paralithodes camtschatica) hepatopancreas. Appl. Biochem. Microbiol. 2008, 44, 93–97. [Google Scholar] [CrossRef]
- Novikov, V.; Mukhin, V. Chitosan Depolymerization by Enzymes from the Hepatopancreas of the Crab Paralithodes camtschaticus. Appl. Biochem. Microbiol. 2003, 39, 464–468. [Google Scholar] [CrossRef]
- Novikov, V.; Mukhin, V.; Rysakova, K. Properties of chitinolytic enzymes from the hepatopancreas of the red king crab (Paralithodes camtschaticus). Appl. Biochem. Microbiol. 2007, 43, 159–163. [Google Scholar] [CrossRef]
- Rysakova, K.; Novikov, V.; Mukhin, V.; Serafimchik, E. Glycolytic activity of enzyme preparation from the red king crab (Paralithodes camtschaticus) hepatopancreas. Appl. Biochem. Microbiol. 2008, 44, 281–286. [Google Scholar] [CrossRef]
- Turkovski, I.; Paramonov, B.; Antonov, S.; Kozlov, D.; Klimova, O.; Pomorski, K. Comparative evaluation of the depth of collagen and hyaluronic acid hydrolysis in vitro by collagenase and hyaluronidase preparations. Bull. Exp. Biol. Med. 2008, 146, 89–90. [Google Scholar] [CrossRef]
- Ponomareva, T.; Sliadovskii, D.; Timchenko, M.; Molchanov, M.; Timchenko, A.; Sogorin, E. The effect of hepatopancreas homogenate of the Red king crab on HA-based filler. PeerJ 2020, 8, e8579. [Google Scholar] [CrossRef]
- Balashova, M.; Liutova, L.; Rudenskaia, I.; Isaev, V.; Andina, S.; Kozlov, L.; Rudenskaia, G. Anticoagulative and anticomplementary activity of endogenous inhibitor preparation from hepatopancreas of red king crab (Paralithosed camtschaticus) towards human blood. Biomed. Khim. 2012, 58, 176–188. [Google Scholar] [CrossRef][Green Version]
- Isaev, V.; Rudenskaya, G.; Rudenskaya, Y.; Lyutova, L.; Balashova, M. Method for Producing a Collagenase Inhibitor with Anticoagulative Action from the King Crab Hepatopancreas. RU 2403284 C1, Filed 25 January 2009, and Issued 10 November 2010. Patent Holder “Zakrytoe Aktsionernoe Obshchestvo Nauchno-proizvodstvennoe predprijatie “TRINITA””.
- Isaev, V.; Rudenskaja, G.; Kostin, N.; Sergeeva, E.; Statsenko, I.; Kolesnikova, T. Method of Production of Recombinant Serine Protease Inhibitor of King Crab. RU 2560264 C1, Filed 17 April 2014, and Issued 20 August 2015. Patent Holder “Obshchestvo s ogranichennoj otvetstvennost’ju Nauchno-proizvodstvennoe predprijatie “TRINITA” (OOO NPP “TRINITA”)”.
- Zikeeva, B. Processing of Water Non-Fish Raw Materials; Pishchepromizda: Moscow, Russia, 1950; p. 277. [Google Scholar]
- Boeva, N.; Petrova, M.; Makarova, A. Production Method of Crab Fat. RU 2390274 C1, Filed 18 December 2008, and Issued 27 May 2010. Patent Holder “FGUP “Vserossijskij nauchno-issledovatel’skij institut rybnogo khozjajstva i okeanografii” (VNIRO)”.
- Klimova, O.; Borukhov, S.; Solovyeva, N.; Balaevskaya, T.; Strongin, A. The isolation and properties of collagenolytic proteases from crab hepatopancreas. Biochem. Biophys. Res. Commun. 1990, 166, 1411–1420. [Google Scholar] [CrossRef]
- Klimova, O.; Vedishcheva, I.; Strongin, A. Isolation and characteristics of collagenolytic enzymes from the hepatopancreas of the crab Chionoecetes opilio. Dokl. Akad. Nauk SSSR 1991, 317, 482. [Google Scholar]
- Shkuratova, E.; Shokina, Y.; Mukhin, V. Development of technology for gourmet smoked products from cod species using enzyme preparation from hepatopancreas of snow crab Chionoecetes opilio. Proc. Voronezh State Univ. Eng. Technol. 2017, 79, 126–137. [Google Scholar] [CrossRef]
- The Food and Agriculture Organization (FAO). Fishery and Aquaculture Statistics: B-44. Available online: http://www.fao.org/fishery/static/Yearbook/YB2017_USBcard/root/capture/b44.pdf (accessed on 5 May 2020).
- National Oceanic and Atmospheric Administration (NOAA). Fisheries Catch and Landings Reports: BSAI Crab. Available online: https://www.fisheries.noaa.gov/alaska/commercial-fishing/fisheries-catch-and-landings-reports (accessed on 5 May 2020).
- The Food and Agriculture Organization (FAO). Fishery and Aquaculture Statistics: B-42. Available online: http://www.fao.org/fishery/static/Yearbook/YB2017_USBcard/root/capture/b42.pdf (accessed on 5 May 2020).
| Proteinase | kDa (Based on Electrophoresis) | Opt. pH | Opt. °C | pI | Substrate Specificity and Other |
|---|---|---|---|---|---|
| Collagenolytic serine proteinase PC [44] | 29 23.5 1 [45] | 7.5 | 47–55 | 3 | Preferably hydrolyzes peptide bonds, the carbonyl formed by Arg, Lys, and hydrophobic amino acids. Hydrolyzes native collagen type I even at 4 °C [46]. The mRNA nucleotide sequence was determined [45], European Molecular Biology Laboratory (EMBL) Nucleotide Sequence Database: AF461035. |
| Collagenolytic serine proteinase PC 2 [47] | 25 | 8.5 | 38–40 | ? | Preferably hydrolyzes peptide bonds, carbonyl formed by positively charged amino acids. Hydrolyzes native collagen types I–III. |
| Trypsin-like proteinase A [48] | 27 30 [49] | 7.9 | 55 [49] | 2.5 | Preferably hydrolyzes peptide bonds, the carbonyl formed by Arg and Lys. Proteolytic activity is not inhibited by ethylenediaminetetraacetic acid (EDTA), and partially inhibited by soybean trypsin inhibitor. |
| Chymotrypsin-like proteinase C [50] | 24 | 9 | 55 [49] | 2.9 [49] | Preferably hydrolyzes peptide bonds, carbonyl formed by hydrophobic amino acids (Phe, Val, and Leu). Not inhibited by Tos-Phe-CH2Cl (chymotrypsin inhibitor). |
| Aminopeptidase PC [51] | 110 | 6 | 36–40 | 4.1 | Effectively cleaves N-terminal amino acids: Arg, Lys, Leu, Phe, and Met. Most likely it is a homodimeric, Zn-containing enzyme. |
| Proteinase | kDa (Based on Electrophoresis) | Opt. pH | Opt. °C | pI | Substrate Specificity and Other |
| Carboxypeptidase PC [52] | 34 | 6.5 | 55 | 3.1 | Effectively cleaves C-terminal amino acids: Arg, Lys, Phe, Tyr, Leu, and Ile. The enzyme is inhibited by 0.5 mM Ag, Zn, Cd and 1 mM EDTA, whereas it is activated by Co and Ca. |
| Trypsin PC [53] | 29 23 [54] 24.8 1 [45] 24.8 2 [54] | 7.5–8 | 55 | <2.5 | Preferably hydrolyzes peptide bonds, carbonyl formed by Arg and Lys. The mRNA nucleotide sequence was determined [45], EMBL: AF461036. |
| Elastase [55,56] | 28.5 | 8–8.5 | 50 [57] | 4.5 | Hydrolyzes native elastin (inhibited by elastinal). NaCl, MnCl2, CdCl2 at a concentration of 1–100 mM stimulate elastase activity, whereas it is inhibited by HgCl2 (100 mM). |
| Cathepsin L [58] | 29 24 1 | 8 | 25 | ? | Enzyme has cathepsin activity, hydrolyzes Z-Phe-Arg-pNA substrate. Hydrolyzes collagen types X and VI. HgCl2, E64, and leupeptin inhibit cathepsin activity; soybean trypsin inhibitor practically does not suppress activity. The mRNA nucleotide sequence was determined, EMBL: HQ437281 |
| Metalloproteinase [59] | 22.2 1 | 8–8.5 | 45 | 4.43 | Destroys peptide bonds formed by both acidic and hydrophobic amino acids. Hydrolyzes azocollagen. Proteolytic activity is maintained at 1–3 M NaCl and is inhibited by isopropanol, o-phenanthroline, and EDTA. Zn-containing enzyme. The mRNA nucleotide sequence was determined, EMBL: AF492483 |
| Enzymes | kDa (Based on Gel-Chromatography) | Opt. pH | Known Properties |
|---|---|---|---|
| Ca- and Mg-dependent DNase [60] | 53 47 1 42 2 41.5 3 [62] | 7–8 6.6 [62] | The primary structure was determined. There are two sequences in UniProtKB, Q8I9M9 (2003) and B6ZLK3 (2009), differing by two amino acids. |
| Alkaline RNase (AlkR) [67] | 19 | 7.2–7.5 | Broad specificity. Poorly hydrolyzes poly(AUC). MgCl2 at a concentration of 10–50 mM stimulates the activity of enzyme. 0.1 M NaCl inhibits the enzyme activity by 50%. |
| Acid RNase (AcR and AcR’) [67] | 33 and 70 | 5.5 | Does not hydrolyze poly(C) and poly(AUC). MgCl2 inhibits its activity, 0.25 M NaCl inhibits its activity by 50%. Most probably, these are monomeric and dimeric forms of the same protein. |
| Two acidic phosphomonoesterases [68] | 80 and 82 | 5.5 | Do not hydrolyze (3′,5′)cAMP (cyclic adenosine monophosphate); 1.5 M NaCl inhibits the enzyme activity by 20% (protein 80 kDa); 1.1M NaCl inhibits the enzyme activity by 50% (protein 82 kDa) |
| Alkaline phosphomonoesterase [68] | 80 | 7.2–7.5 | Does not hydrolyze (3′,5′)cAMP. 0.4 M NaCl inhibits the enzyme activity by 50%. |
| Acidic phosphodiesterase [68] | 57 | 4.8–5 | 50% inhibition at 1.4 M NaCl. |
| Alkaline phosphodiesterase [68] | 51 | 7.2–7.5 | No inhibition up to 1.4 M NaCl is observed. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ponomareva, T.; Timchenko, M.; Filippov, M.; Lapaev, S.; Sogorin, E. Prospects of Red King Crab Hepatopancreas Processing: Fundamental and Applied Biochemistry. Recycling 2021, 6, 3. https://doi.org/10.3390/recycling6010003
Ponomareva T, Timchenko M, Filippov M, Lapaev S, Sogorin E. Prospects of Red King Crab Hepatopancreas Processing: Fundamental and Applied Biochemistry. Recycling. 2021; 6(1):3. https://doi.org/10.3390/recycling6010003
Chicago/Turabian StylePonomareva, Tatyana, Maria Timchenko, Michael Filippov, Sergey Lapaev, and Evgeny Sogorin. 2021. "Prospects of Red King Crab Hepatopancreas Processing: Fundamental and Applied Biochemistry" Recycling 6, no. 1: 3. https://doi.org/10.3390/recycling6010003
APA StylePonomareva, T., Timchenko, M., Filippov, M., Lapaev, S., & Sogorin, E. (2021). Prospects of Red King Crab Hepatopancreas Processing: Fundamental and Applied Biochemistry. Recycling, 6(1), 3. https://doi.org/10.3390/recycling6010003







