2.1. Description of Cells, Ageing Conditions and Initial Visual Inspection of Components
The cells specifications are reported in
Table 1. The cells were aged by cycling with a constant charge current (0.3C or 2C) between 0% and 100% SOC (i.e., between 2.5 V and 3.6 V) at 5 °C inside a ventilated climatic chamber. The discharge C-rate is kept the same (1C). The tests were interrupted regularly to run a performance test at room temperature (RT) to follow up on the evolution of the cell’s characteristics. The cell state of health (SOH) was determined from a 1C discharge constant current/constant voltage (CC-CV) test performed during this reference test.
Acronyms: LFP = LiFePO4; C = conductive carbon; PVDF = Polyvinylidendifluoride; Gr = graphite; CMC = Sodium carboxymethylcellulose; SBR = Styrene Butadiene Rubber; LiPF6 = Lithium hexafluorophosphate EC = Ethylencarbonate; PC = Propylencarbonate; DMC = Dimethylcarbonate; VC = vinylcarbonate; PE/PP = Polyethylene/polypropylene.
The ageing protocol was interrupted when the cell loses at least 20% of its initial capacity. As the SOH is calculated during the performance test, it was not possible to interrupt the ageing at the exact same SOH value. Nevertheless, they have very close SOH values (79.7% and 72%).
Table 2 summarizes the results of the ageing phase. The total capacity throughput indicates the total capacity (charge and discharge) that the cells have cycled during ageing (including the performance test). The equivalent cycle number is obtained by dividing the total capacity throughput by the capacity of one complete cycle (about 32 Ah). Three cells were used for the study: a cell that performed only the formation protocol (named fresh cell) and two cells aged by cycling at 5 °C and 0.3C (named Cell A) or 2C (named Cell B).
After cycle ageing, the cells did not present any clear sign of degradation, such as swelling or electrolyte leakage. Although the cells were completely discharged to 2.5 V, a slightly higher value of OCV was detected for Cell A shortly prior to cell disassembly.
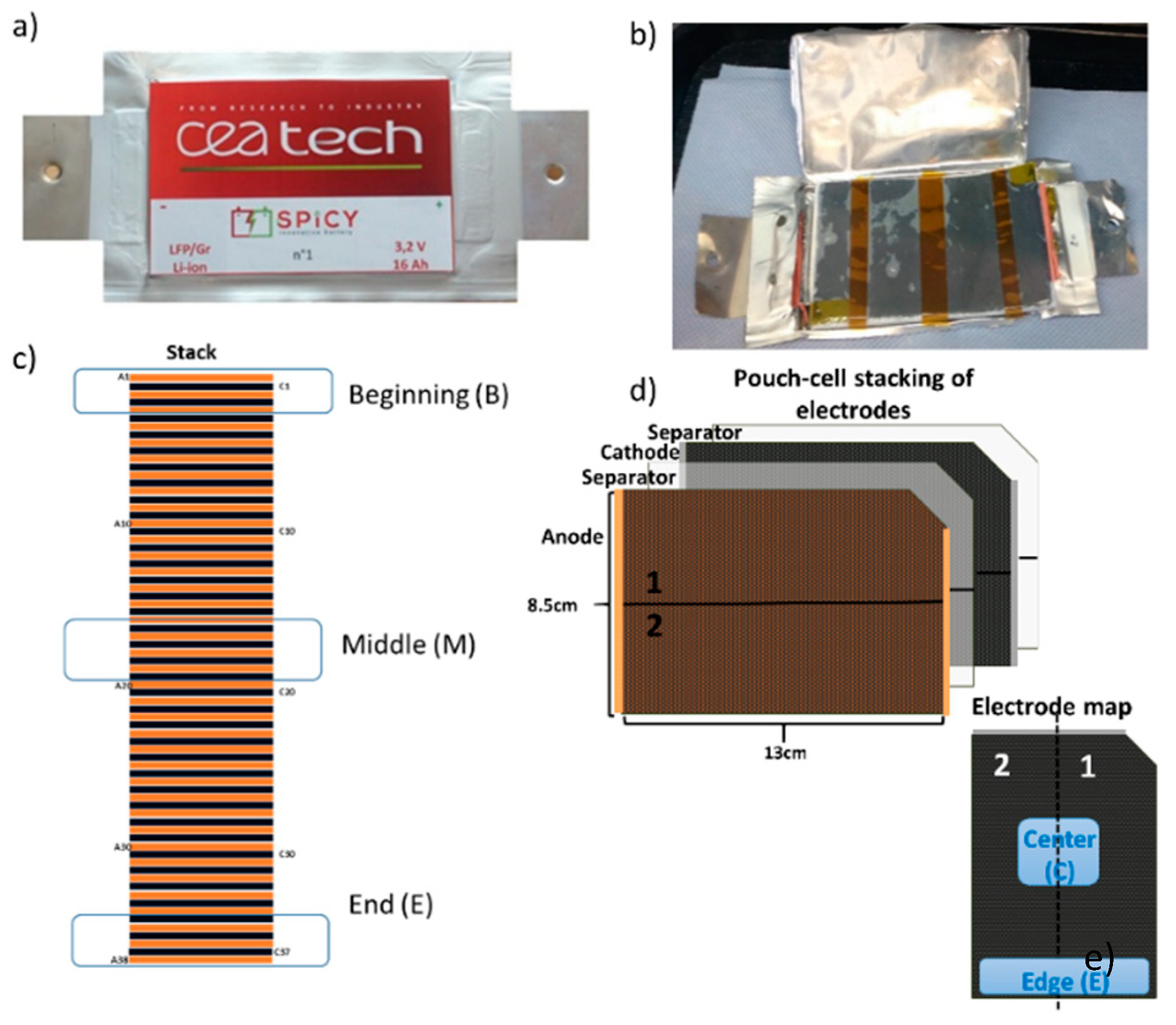
Figure 1a,b shows the pouch cell as made and after opening. Each cell contains a stack (
Figure 1c) of double-side coated electrodes and separators as schematically depicted in
Figure 1d. The electrodes were harvested at the beginning (B), middle (M) and end (E) of the stack. The samples, taken from the edge (e) and the center (c) of the electrode tape (
Figure 1d), were dipped in dimethylcarbonate (DMC) (except some cathode samples from the fresh cell for SEM analysis which were dipped in acetonitrile (ACN) for 30 s and then dried under dynamic vacuum at room temperature for 30 min.
Table S1 in the Supplementary Materials reports a detailed description of the analysis performed on each sample and its position in the stack.
Figure 2 compares the pictures of cells components harvested from the middle of the stack. The separator of Cell A does not show a significant difference respect to the one from fresh cell, while that taken from Cell B shows dark areas, probably due to detachment of active material from anode tape. Compared to the fresh cell, the cathode from cell A and cell B show a grey/brown coloration after cycling at 5 °C. Noteworthy, the cathode tape in different portions of the stack does not show any visual difference (not shown). The anode tape, on the other hand, was rather shiny at the edges with a darker area in the center. The silvery color may be an indication of plated lithium [
5], in both cell A and cell B.
2.3. Morphology of Cells’ Components
The SEM images in
Figure 3 show the appearance of the LiFePO
4 (LFP) electrodes at the beginning of the stack in the center position of the Fresh Cell, Cell A and Cell B. The electrodes are composed of a mixture of spherical particles and small amount of carbon fibers (used as conductive agent) of micrometric length. The larger spherical particles show a diameter comprised between 0.5 and 1 µm while the smaller particles have a diameter lower than 100 nm. Comparing the SEM images, no significant differences are observed. Additionally, no specific damage or deposit is visible.
Also, the cathode morphology did not show any relevant difference when sampled from different position in the stack and in the tape (edge vs. center) (see, respectively,
Figures S2 and S3 in the Supplementary Materials). Furthermore, the energy dispersive X-ray (EDX) analysis (
Figure S4 in the Supplementary Materials) does not reveal differences between Cell A and Cell B, nor the presence of contaminants. The elemental composition (
Figure S4) is also similar to that of the fresh cell (only Fe, P, O and C are detected).
Figure 4 shows a few SEM images of the graphite electrode at the beginning of the stack in the center position, extracted from Fresh Cell (EDX spectra reported in
Figure S5 in the Supplementary Materials), Cell A and Cell B. While no major difference between the electrodes from Fresh Cell and Cell A is observed, the anode of Cell B (cycled at 2C) shows the occurrence of graphite exfoliation (notice that several SEM images were taken using different samples to confirm the observed differences). Noteworthy, such exfoliation is not observed in the edge of the electrode (see
Figure 5). Thus, a major difference between the center and the edge of the anode electrode is confirmed using both visual (see
Figure 2) and SEM observations.
EDX analysis was performed on the samples extracted from both the center and the edge of the anode of Cell B (
Figure S6 in Supplementary Materials). Surprisingly, no significant difference in terms of elemental composition was observed. EDX indicates the presence of O, P and F in both samples, due to electrolyte salt residue and the SEI. However, Fe traces were detected in the center (but not in the edge of the electrode) resulting from the cathode (LFP) decomposition. The Fe traces could be one of the reasons for the graphite exfoliation detected in the center of the electrode, even if Fe is not expected to be intercalated inside graphite.
2.4. Residual Capacity Measurement
All samples used for the residual capacity determination were harvested from the electrode tapes taken from Area 2 in
Figure 1d. The samples are named using the abbreviation given in
Table 4, which describes their position in the stack and the electrode. Due to the poor adhesion of the anodic tape, the half-cell assembly with aged graphite anodes was not always possible therefore the residual capacity is analyzed using only the cathode.
The voltage profile obtained at 0.15 mA∙cm
−2 (corresponding to 0.065C) for a pristine cathode tape is reported in
Figure S8 in the Supplementary Materials. The voltage profiles of the first cycle (charge comes first) of the aged cathodes performed in the same conditions are shown in
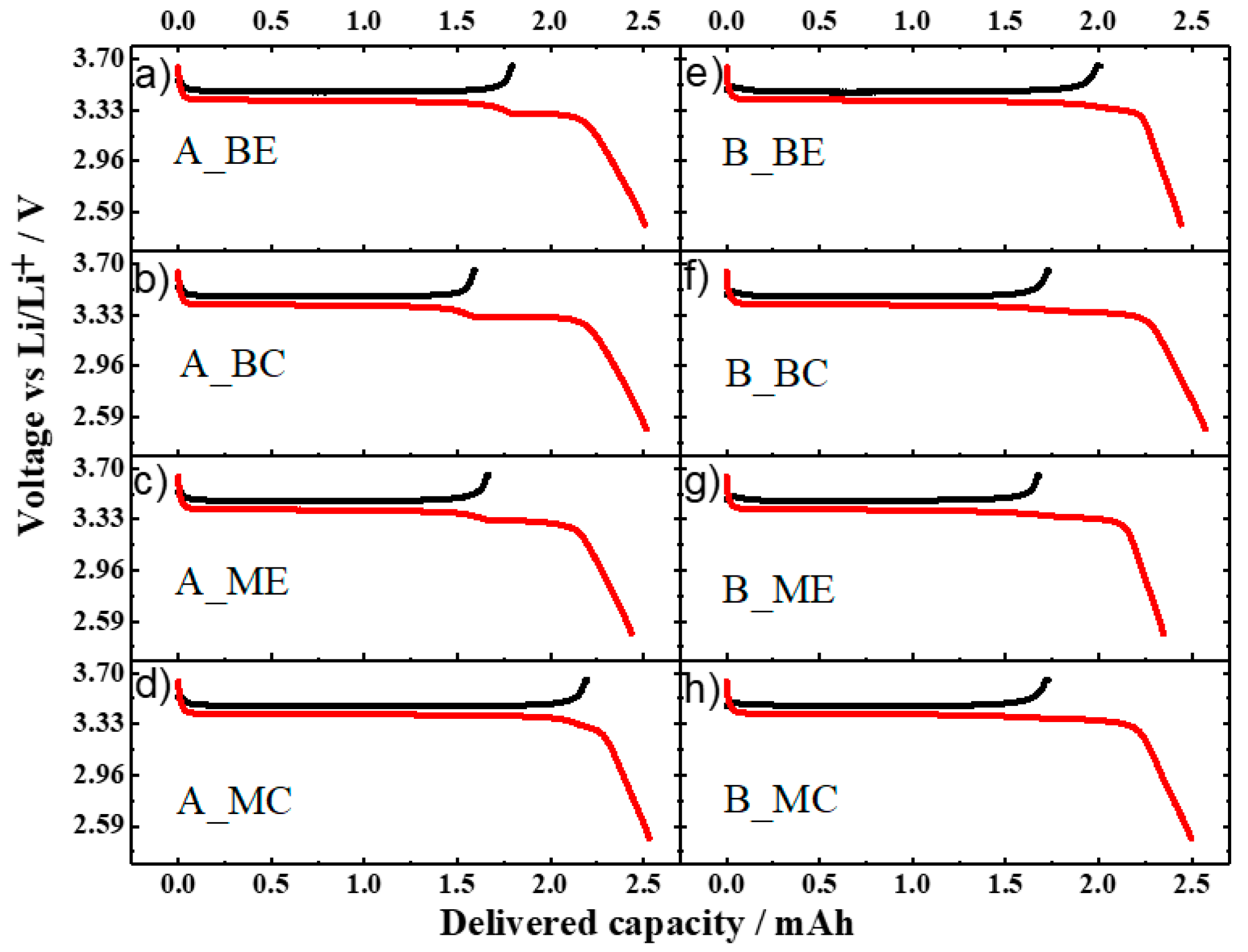
Figure 6. It can be noticed that the residual charge capacity, i.e., the amount of Li
+ extracted from the electrode, is not homogeneous along the stack and across the electrode tape.
For the cell aged at 0.3C, the sample A_MC delivered a high charge capacity of ca. 2.25 mAh, a value close to that of the pristine electrode. On the other hand, for the sample A_ME only 1.66 mAh were obtained. The situation is inverted for the samples taken at the beginning of the stack as the sample from the edge (A_BE) delivered a higher capacity (1.79 mAh) than that from the center (A_BC) (ca. 1.59 mAh). It is worth noting that, upon the subsequent lithiation, a “step” in the voltage profile appears. Its position matches well with the capacity value obtained during the previous de-lithiation. Therefore, we attributed this feature to the insertion of lithium into the LFP cathode, which did not occur during the ageing test. This means that, for all the samples, part of the cathode material was inactive during the ageing test, but not damaged. A similar situation is found for the voltage profiles of the aged cathodes from cell B (see
Figure 6e–h). Here, the sample from the beginning of the stack and edge of the electrode (B _BE) delivered a capacity (ca. 2.0 mAh) higher than all the other samples (ca. 1.7 mAh). It is also noted that the resistance of Cell A cathode is higher than that of Cell B cathode as a marked overvoltage is present at the very beginning of the charge process (
Figure S9 in the Supplementary Materials).
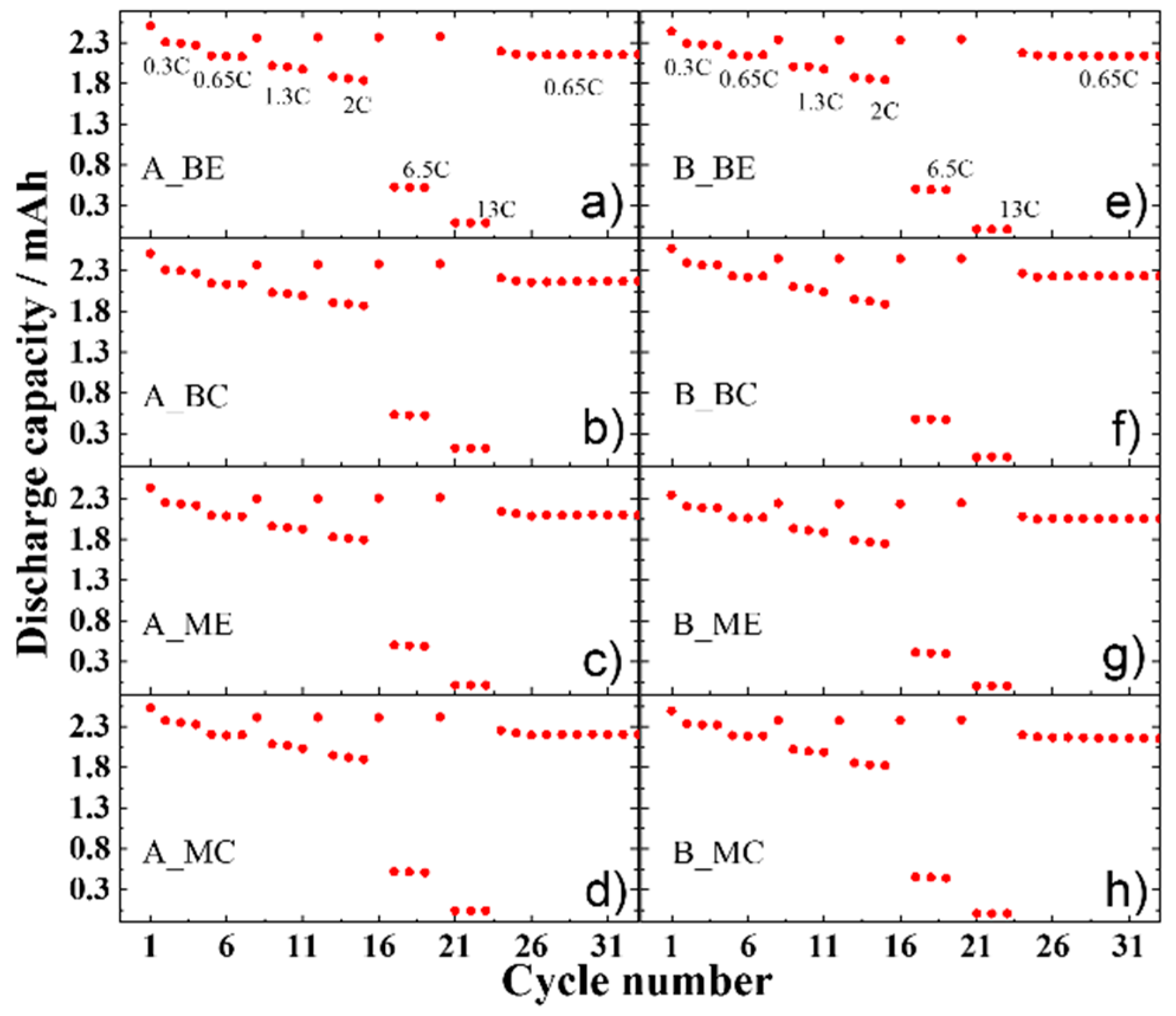
After the first discharge (lithiation of LFP), all the electrodes perform equally in the rate capability test shown in
Figure 7 (cycling protocol described in
Table S2 in the Supplementary Materials), indicating that the cathode material structure did not undergo significant damages independent on the position in the stack and across the tape. Therefore, the loss of active material (LAM) is negligible compared to the loss of lithium inventory (LLI).
Overall, the same trend of the residual capacity is observed for both aged cells, i.e., MC > ME and BE > BC, but the non-uniformity is more marked for Cell A than Cell B. The observed inhomogeneous LLI can be attributed to a different extent of side reactions (e.g., SEI formation at the facing anode) caused by variations of the electronic contact (due to differences in the internal pressure) and/or ionic conduction through the electrode (inhomogeneous electrode wetting).
It can be inferred that, in the middle of the stack and in the center of the electrode (MC), more LFP is reversibly cycled than at the edge, probably due to the better contact induced by the higher internal pressure. This would also result in a better anode SEI, with a lower consumption of the Li+ inventory to repair it upon cycling. On the contrary, at the beginning of the stack, the edge part of the electrode (BE) is more electrochemically active. Recalling the SEM results for Cell B, graphite exfoliation was observed in the center of the electrode, but not at the edges.
2.5. Structural Analysis
The inhomogeneity revealed by the electrochemical tests is confirmed by phase quantification via Rietveld refinement of the XRD patterns of aged cathode electrodes (
Figure S10 in Supplementary Materials). The results are summarized in
Table 5. Considering that the cells were opened in the fully discharged state, the cathode material should mostly consist of LiFePO
4 but FePO
4 could also be present if the discharge process is not complete due to lack of available lithium. For Cell A, the trend that a higher fraction of LiFePO
4 is present in samples BE than BC is confirmed (and double checked with the samples taken at the end of the stack EE). On the other hand, discrepancies are found for electrodes in the middle of the stack for which the XRD investigation detects more LiFePO
4 at the edge than in the center, revealing that the inhomogeneity is more pronounced in this part of the stack.
The diffraction patterns of the anodes have been analysed for the shift of the first graphite peak at 26.8° (
Figure S11). The shift towards lower angle of the
00l reflection is linked with graphite interlayer expansion upon lithium intercalation [
12] The shift is larger in the Fresh Cell, while among the aged samples, Cell B showed the smallest shift, indicative of a lower amount of Li
+ remaining trapped into the graphite layers (the cells are opened in the fully discharged state, i.e., Li
+ ions should be fully removed). This translates into a higher lithiation of the cathode. Indeed, a higher LiFePO
4 content is observed in the cathode sample B_BC than A_BC (75% and 70%, respectively), which matches well with the slightly higher residual capacity (
Figure 6) obtained for the B_BC cathode than the A_BC cathode (1.73 and 1.59 mAh, respectively).
2.6. Surface Analysis
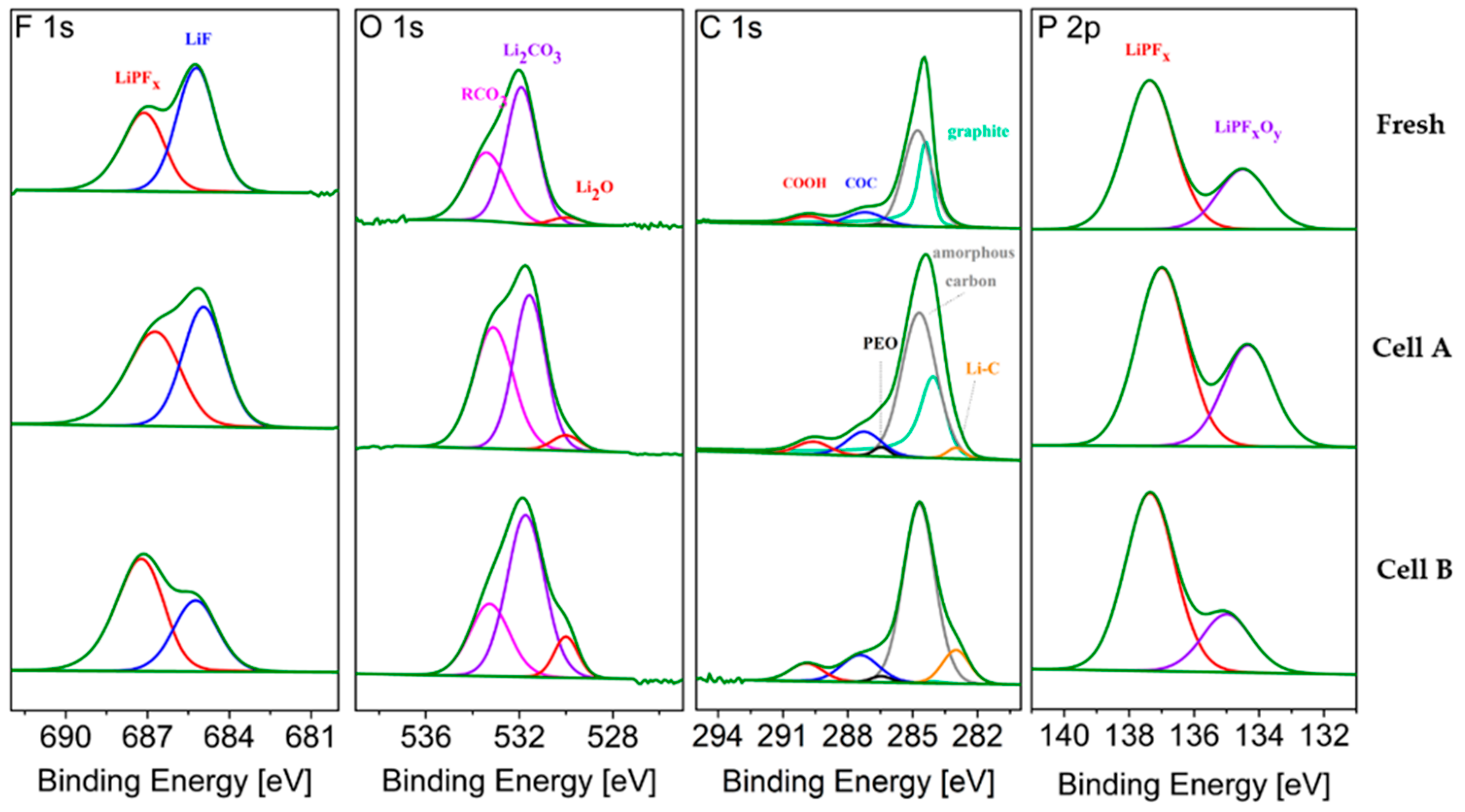
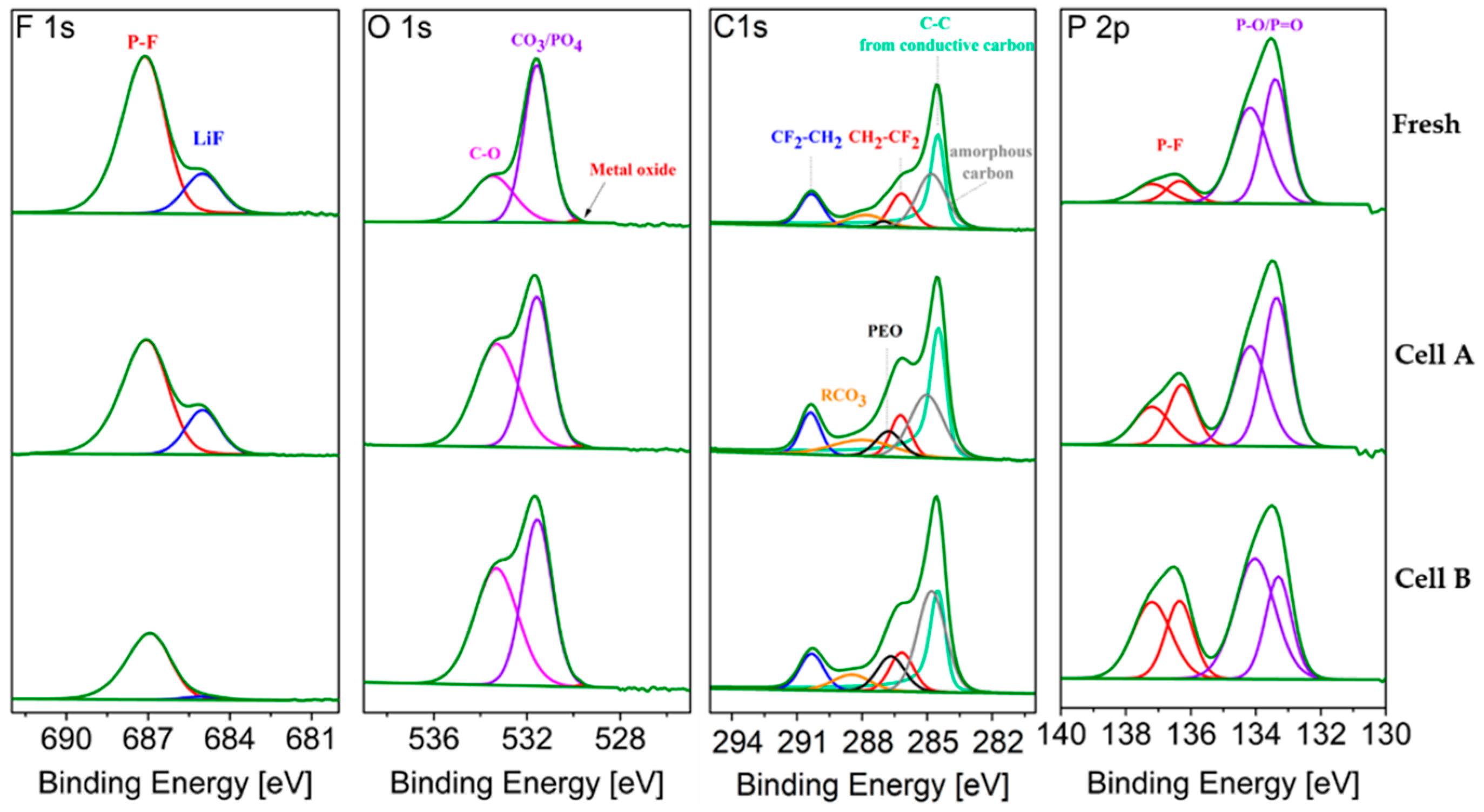
The XPS spectra of graphite samples, harvested from the middle of the stack and at the center of the electrodes (MC), from fresh and aged cells are compared in
Figure 8. The average atomic compositions are reported in
Table 6. Sputter depth profiling was conducted to obtain information on the SEI composition and thickness.
Upon ageing, the SEI on graphite is expected to grow, consuming lithium and thus contributing to the LLI.
Figure 8 shows the typical LiPF
6 (electrolyte salt) decomposition products, i.e., LiF, LiPF
x and LiPO
xF
y, that are detected in the F 1s and P 2p regions [
13]. Their relative ratio changes upon ageing. The sum of the atomic fractions derived from these three peaks after 600 s of sputtering (
Table 6) represents 3.3, 5.0 and 5.6 % at. for the Fresh Cell, Cell A and Cell B, respectively, showing that a slightly higher salt decomposition occurred in Cell B upon cycling, as also highlighted by the higher signal detected for LiPF
x (P 2p region).
The O 1s spectra of the graphite electrodes (
Figure 8) consists of three peaks. The first peak, at the lowest energy, is assigned to lithium oxide, which is a well-known component of the inner part of the SEI [
14], but it also corresponds to the degradation product of Li
2CO
3 induced by the sputtering process itself [
15]. After ageing, the Li
2O feature is more pronounced in the cell cycled at 2C (cell B). The sputtering results in
Table 6 indicate more Li
2O in the inner layer of the SEI of aged graphite electrodes. The second peak, assigned to Li
2CO
3, also grows slightly upon ageing. The peak at the highest binding energy, assigned to organic carbonates, increases more on the surface of the electrode during ageing at 0.3C (cell A), while after 600 s sputtering more organic carbonates are found in Cell B. Before sputtering, the oxygen containing components found in the O 1s region of the graphite from the fresh cell represent 12.11 % at., which increases to 14.98 % at. upon cycling at 0.3 C and 15 % at. 2C.
To eliminate the error caused by the overlapping of graphite and amorphous carbon signals, the sum of their contributions is labeled C 1s in
Table 6. This contribution decreases upon cycling indicating that the SEI becomes thicker, thus hiding the graphite contribution. The Li-C signal at 600 s increases in the cycled electrodes, being more pronounced for Cell A (0.3C). This further confirms that the SEI is thicker in the cell cycled at higher C-rate (Cell B).
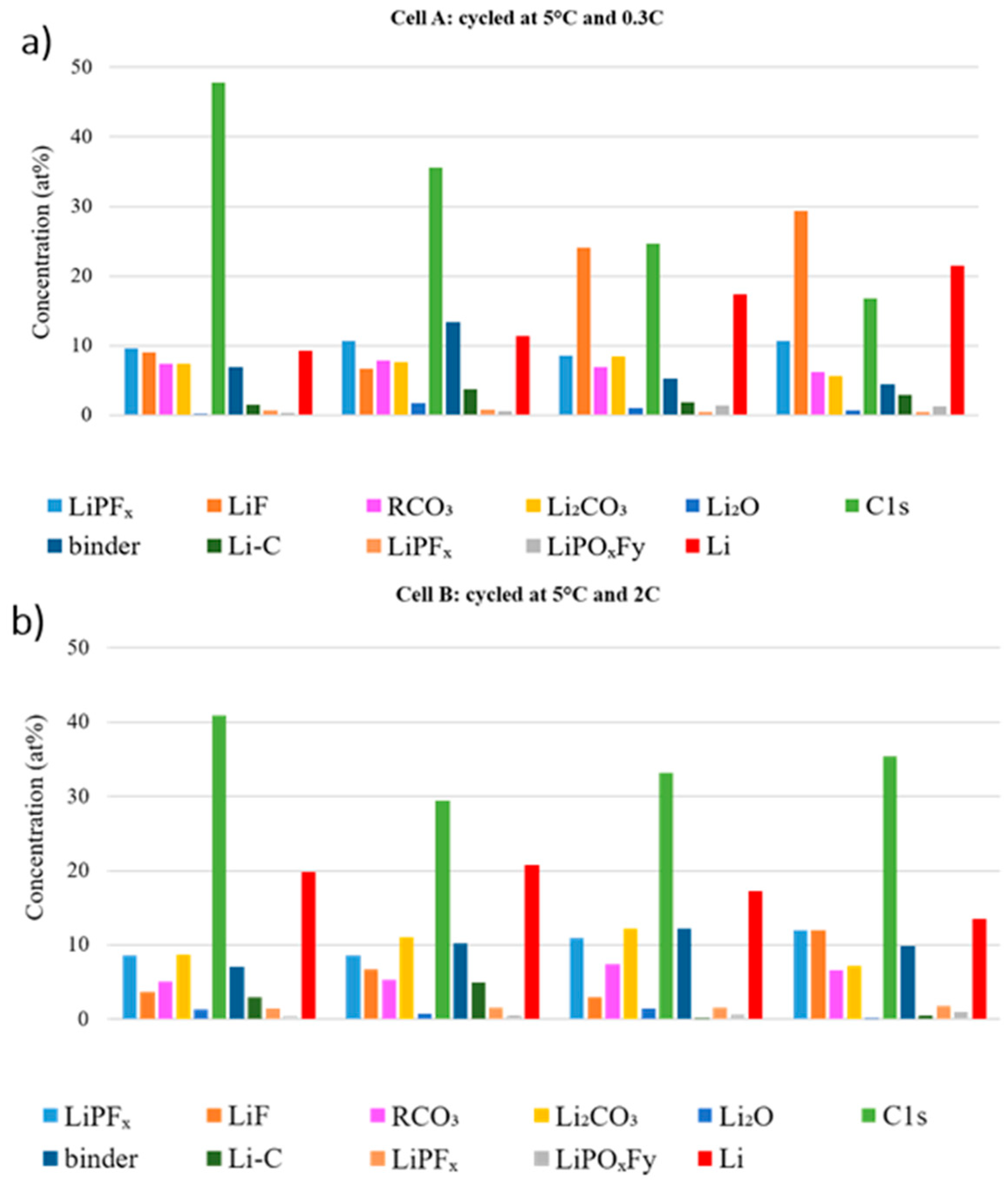
To investigate the effect of electrode position on the SEI build-up,
Figure 9 compares the atomic fraction observed at the center and edge of the electrode tape from the beginning and the middle of the stack for the aged cells. The LiPF
x, LiF and LiPO
xF
y % at. compositions are overall higher at the edge of the graphite tape, which means that LiPF
6 degrades more extensively there. Additionally, more LiPF
6 degradation products are detected in the outer layers of the stack. Overall, the amount of salt decomposition products is higher, whatever the position, in the cell cycled at 0.3C.
In Cell A, the C 1s and binder features decrease dramatically in the beginning of the stack, which indicates that the SEI is thicker than in the middle of the stack. On the other hand, the carbon features in the beginning of the stack in Cell B is higher than Cell A, which is an opposite trend comparing to the middle of the stack. For Li compounds there is no clear trend, which makes it challenging to obtain a reliable relation between different positions.
Generally, for both cycled cells, the differences due to sample location are in the same range as the differences between the two cells. These findings can be correlated to the visual observation of several variations in color and thickness along the electrode stack and across each electrode tape.
The XPS spectra and fitting results for the cathodes extracted from fresh and aged cells are compared in
Figure 10.
Table 7 reports the averaged atomic concentration for the different components. Due to the uncertainties caused by the overlap between the Fe 2p and Fe 3p core level spectra and the fluorine plasmon and, in the Li 1s core spectra, the overlap between Fe and Li contributions, the corresponding spectra were not used for cathode analysis. All samples investigated were taken at the center of electrode at the middle of the stack.
The F 1s spectra show traces of LiF at 685 eV on the fresh and aged LFP electrodes. As found for the anode, LiF represents a higher fraction of the cathode electrolyte interphase (CEI) layer in the cell cycled at 0.3C (Cell A) than at 2C (Cell B).
In the O 1s region, the amount of signal attributed to oxygen atoms in the phosphate group (PO
4)
3− of LFP decreases upon cycling (
Figure 10). This peak is well pronounced for the fresh cathode, indicating the very thin nature of the CEI layer. The peak at higher binding energy (C–O) is attributed to the oxygenated species at the electrode surface and increases in aged cathodes. Upon ageing, the difference between these two peaks decreases, as a result of the growth of the CEI layer. For Cell B the (PO
4)
3− contribution remains dominant (see
Table 7) while the fraction of oxygenated species in Cell A equals that calculated from the LFP feature.
In the C 1s spectrum, the peak at 284.5 eV corresponds to conductive carbon, which conceals the peak of amorphous carbon related to carbon coating [
16]. The two other peaks toward higher energy at 286.3 eV and 290.4 eV are attributed to CF
2CH
2 and CH
2CF
2, corresponding to the PVdF binder [
17]. Moreover, two small peaks are due to polyethylene glycol (PEO) and RCO
3 [
7]. PEO and RCO
3 have been proved to be part of both the SEI and the CEI [
18,
19]. It can be seen that both contributions increase upon cycling in both cases.
In the P 2p region, the peak of PO4 (at 133.3–133.5 eV, labeled P–O/P = O) declines with ageing, which indicates for the CEI growth. The higher amount of phosphate feature in the cell cycled at 2 C is in good agreement with results from the O 1s region, indicating thinner CEI layer. The higher amount of P–F compound (LiPF6 decomposition products) is detected in the cell cycled at higher C-rate, the same result was obtained for graphite electrode.
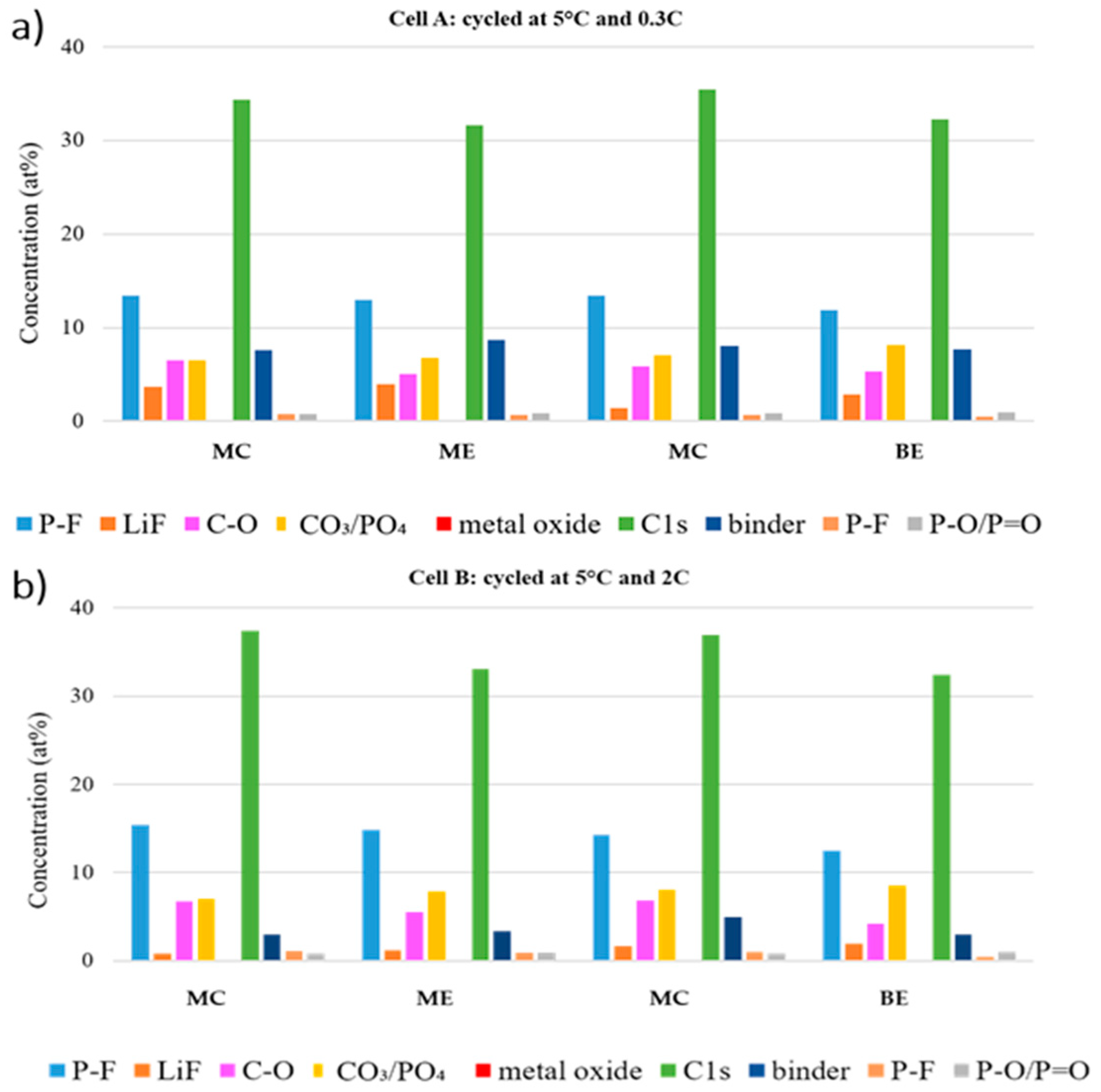
Figure 11 compares the atomic percentages at the center and edge of the electrode tape from different positions of the stack (beginning and middle of the stack) of the aged cathodes. In both cells, a higher LiF amount is detected at the edge of each electrode tape, which means more LiPF
6 degradation. Overall, the amount of LiF is higher at any positions in the cell cycled at 0.3C. However, higher amounts of LiF are found in the middle of the stack, while the opposite is observed for the cell cycled at 2C. Nonetheless, the total amount of fluoride species are found to be higher in the middle of the stack for both cells.
In O 1s and P 2p regions, the amount of oxygenated species (C–O and P–F) is lower at the edge of each stack, while the LFP feature stays more pronounced at all the edges. Thus, we can conclude that the formed CEI is thinner at the electrode edge in both cells. Moreover, the LFP feature (PO4 and P–O/P = O) in O 1s and P 2p region is more pronounced in the beginning of the stack, which implies that the CEI at the middle of the stack is thicker.
Generally, for both cycled cells greater differences were found in electrodes of different stacks. However, the trend of the features in the F 1s, O 1s and P 2p regions suggest that the dissimilarities caused by varying the C rates are as marked as the differences between different locations in the cells.