Titan Yellow and Congo Red Removal with Superparamagnetic Iron-Oxide-Based Nanoparticles Doped with Zinc
Abstract
:1. Introduction
2. Experimental Section
2.1. Chemicals
2.2. Synthesis of Nanoparticles
2.3. Characterization
2.4. Adsorption Study
3. Results
3.1. Morphology Studies
3.2. BET Analysis
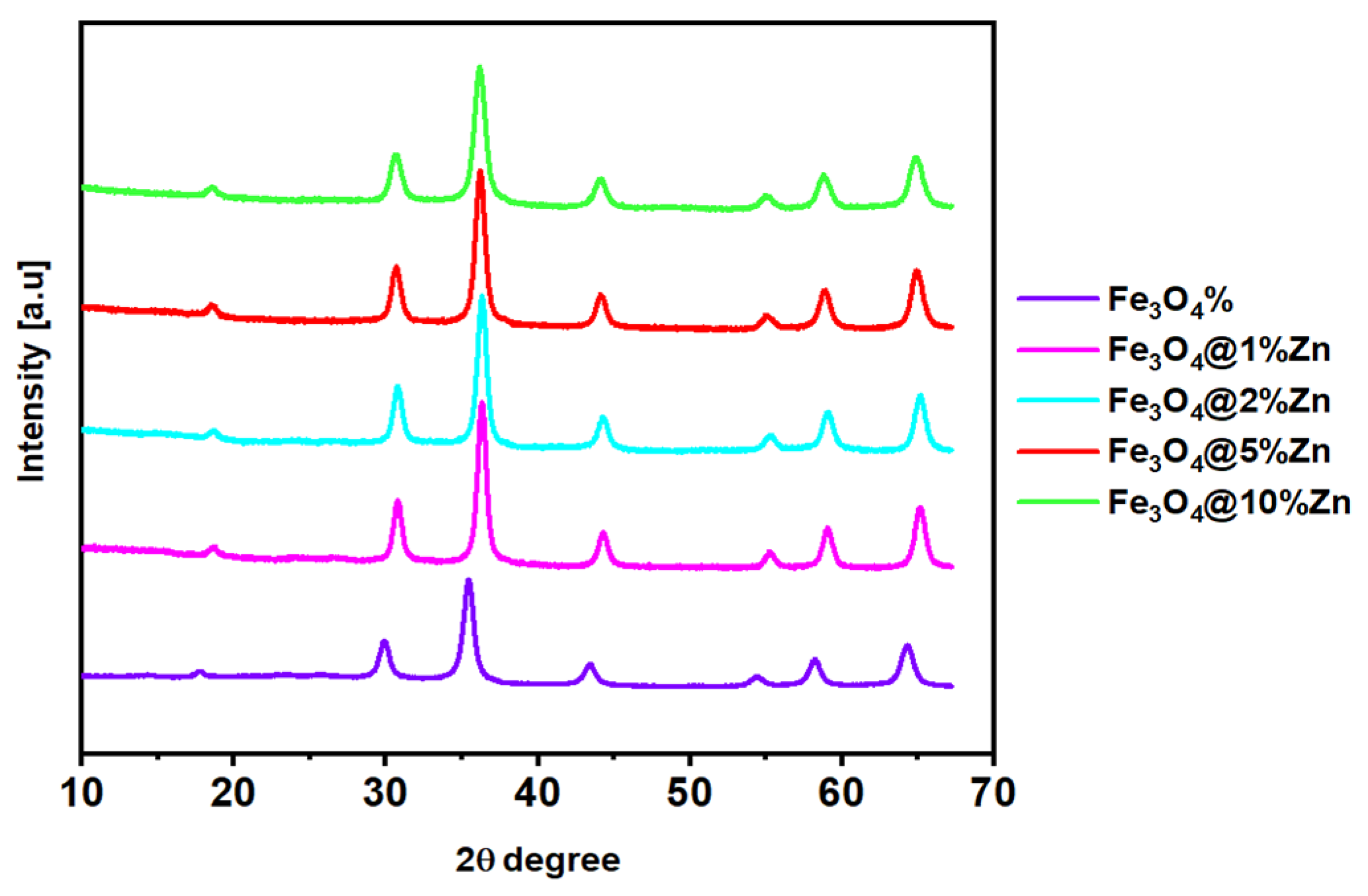
3.3. Crystallinity
3.4. Magnetic Properties
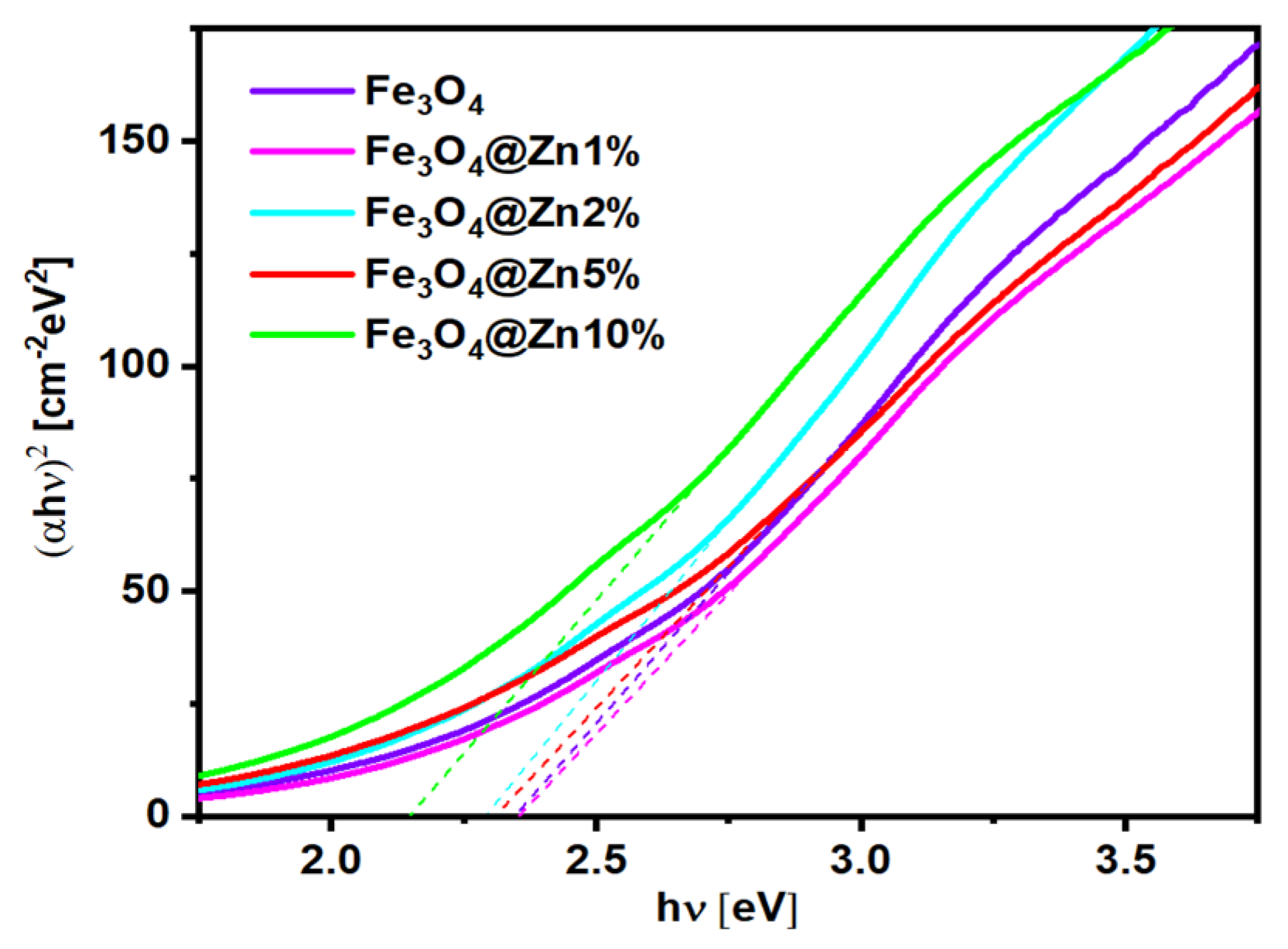
3.5. Optical Absorption Properties
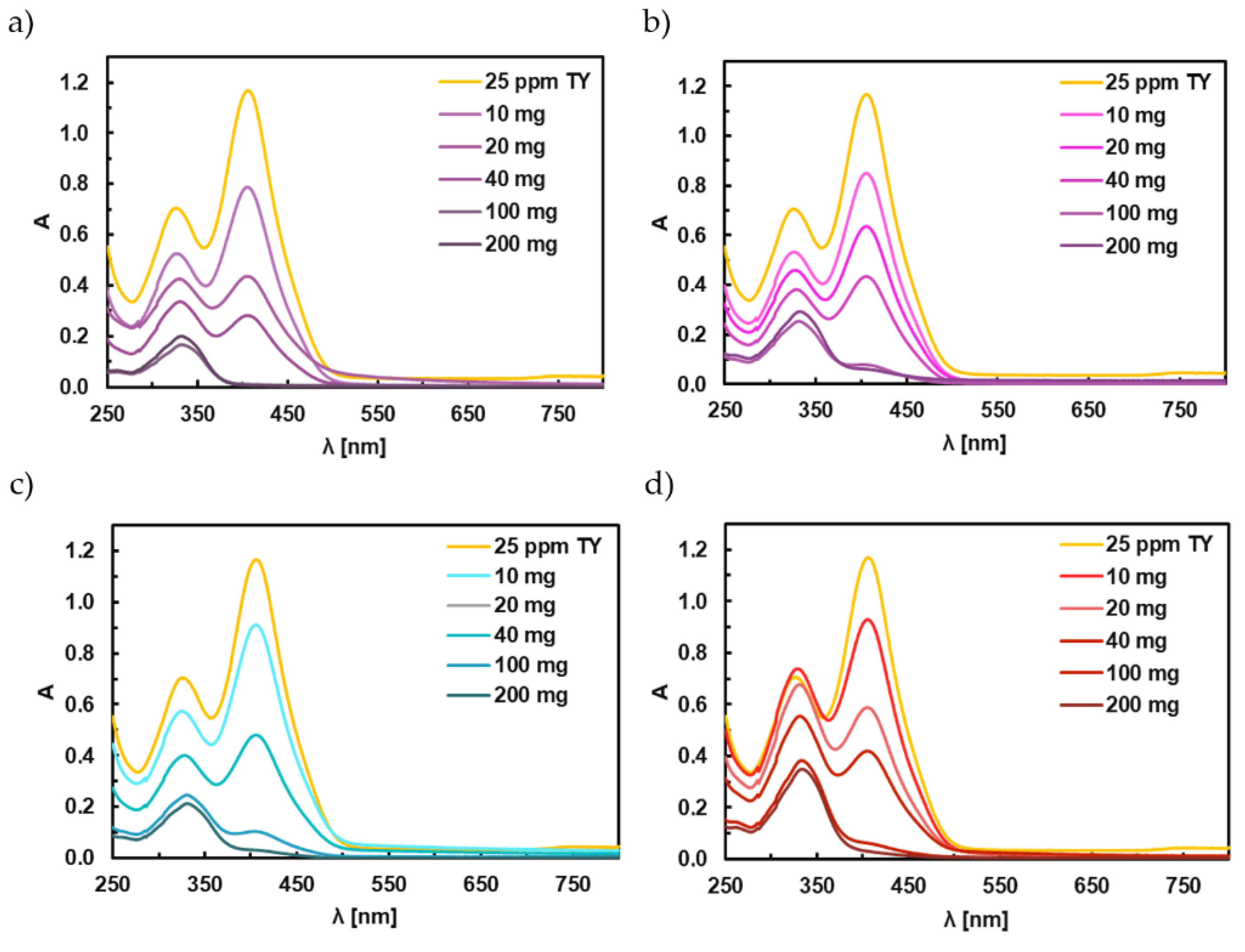
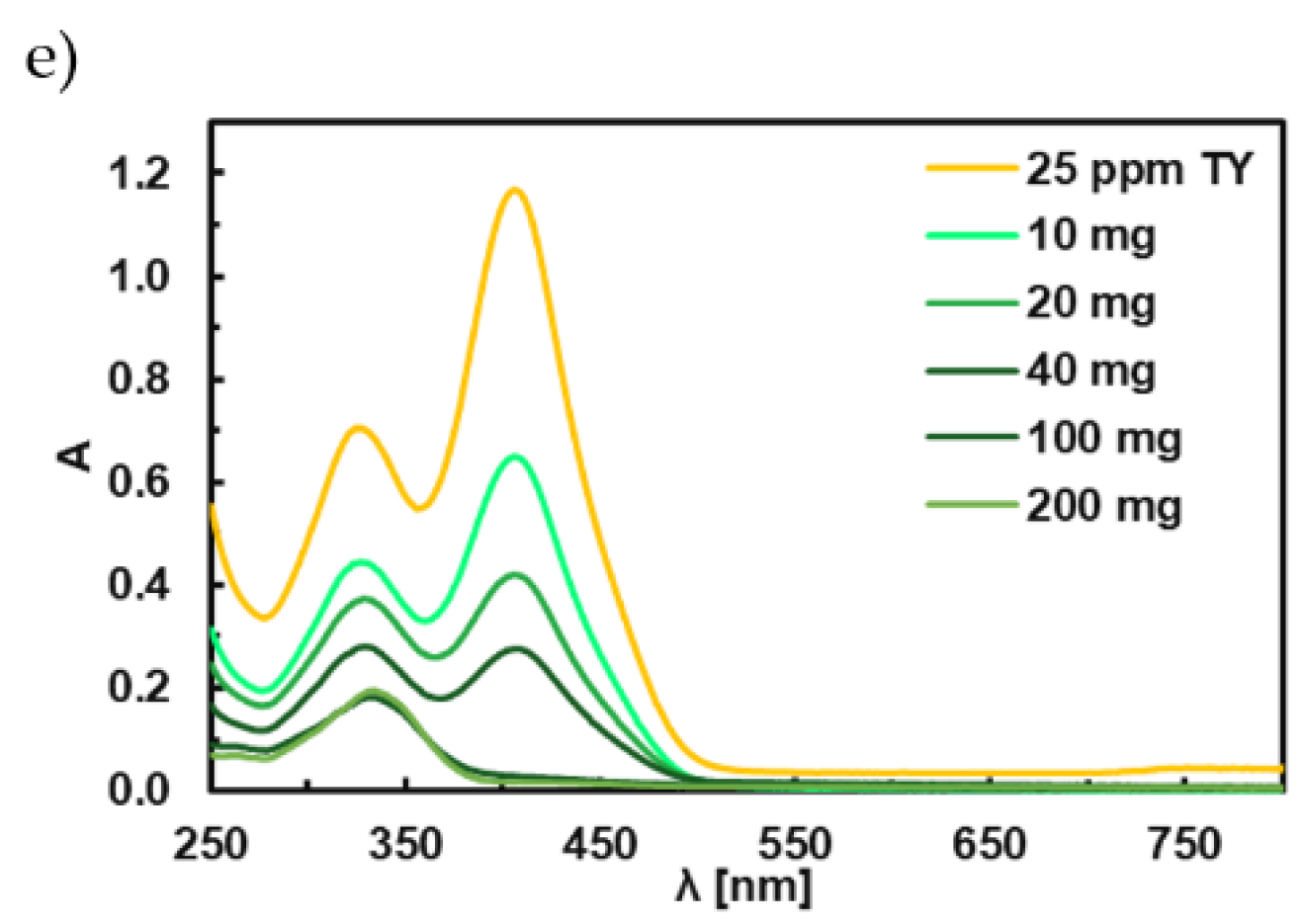
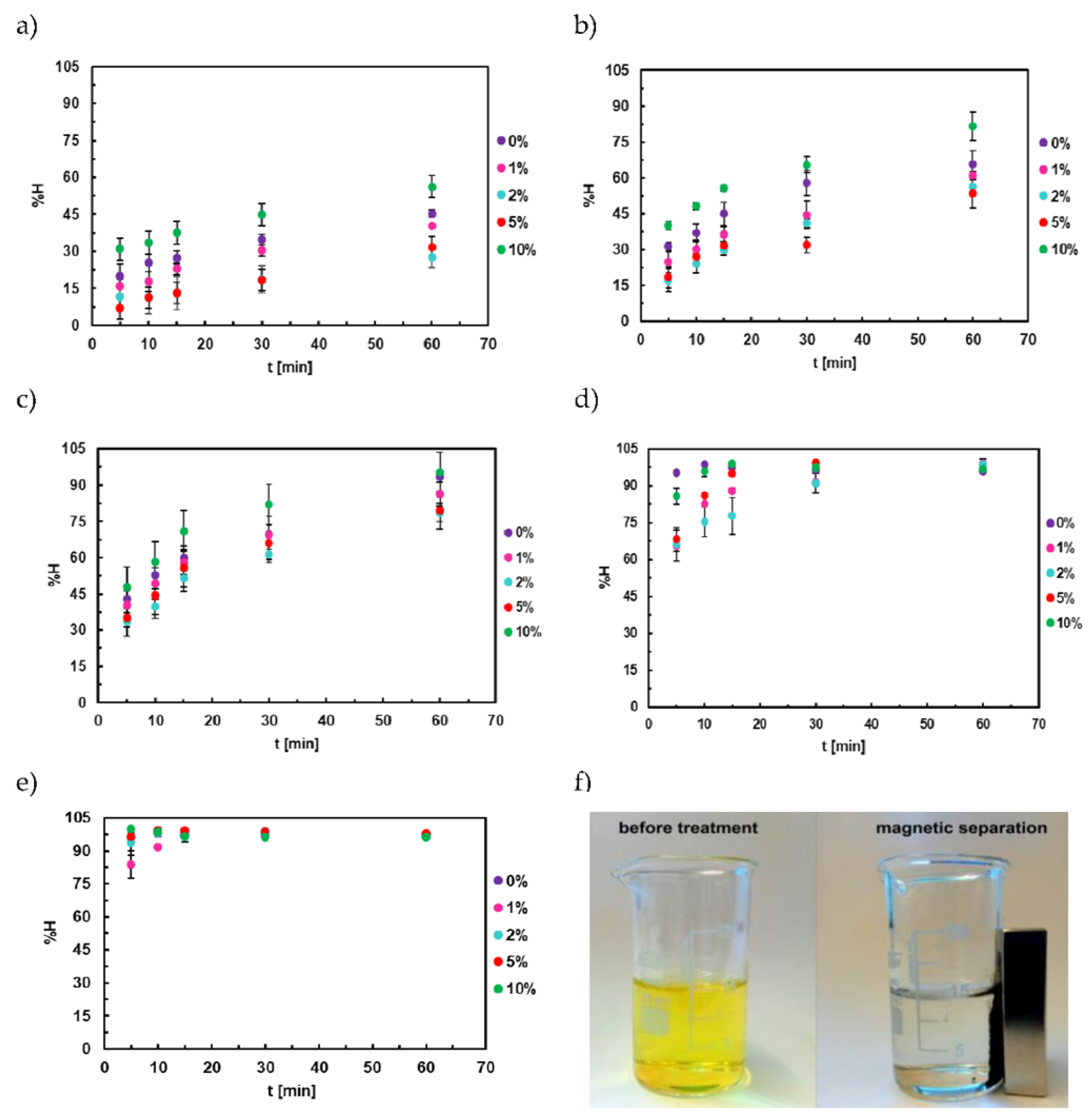
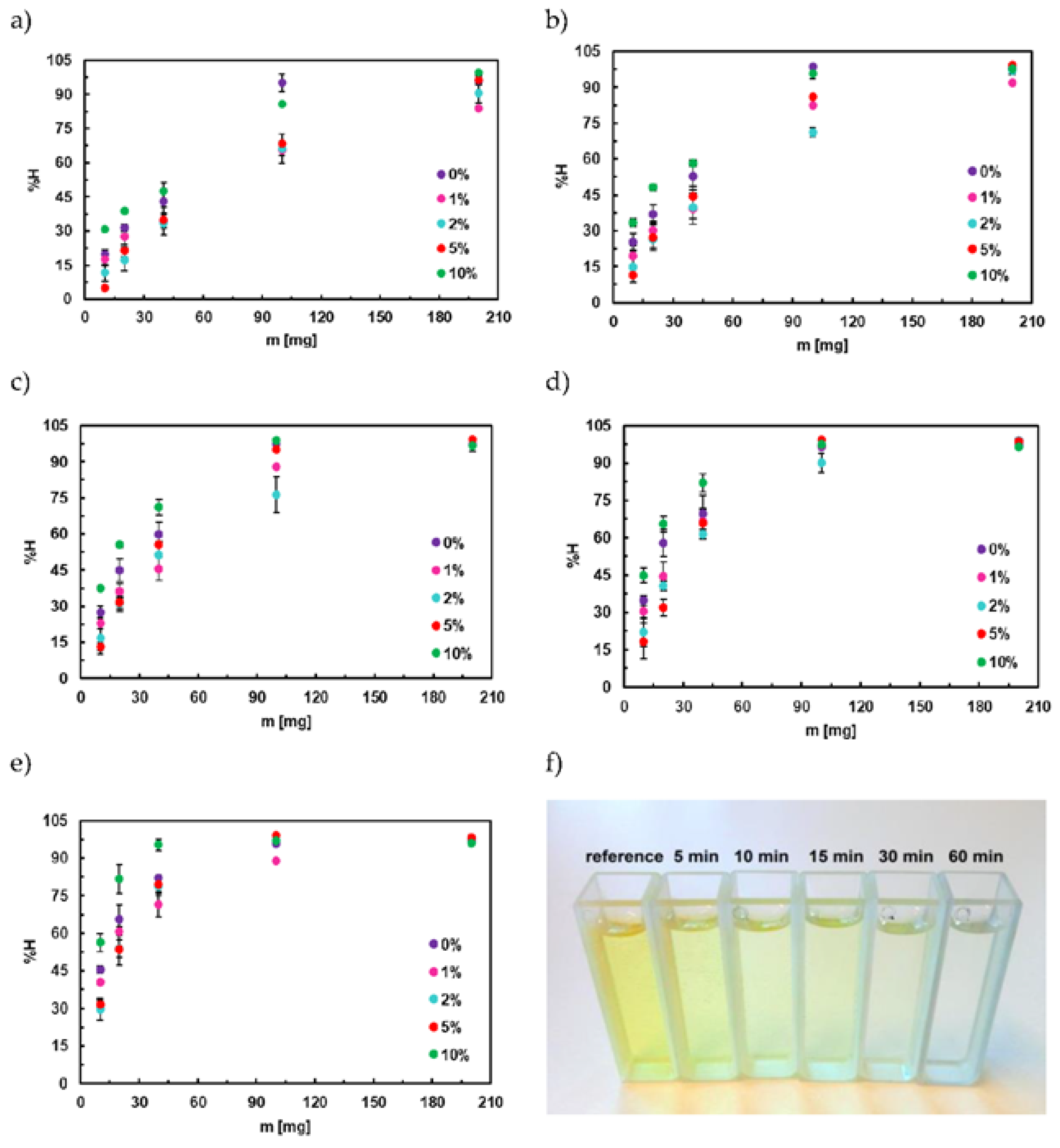
3.6. Adsorption Studies of Titan Yellow
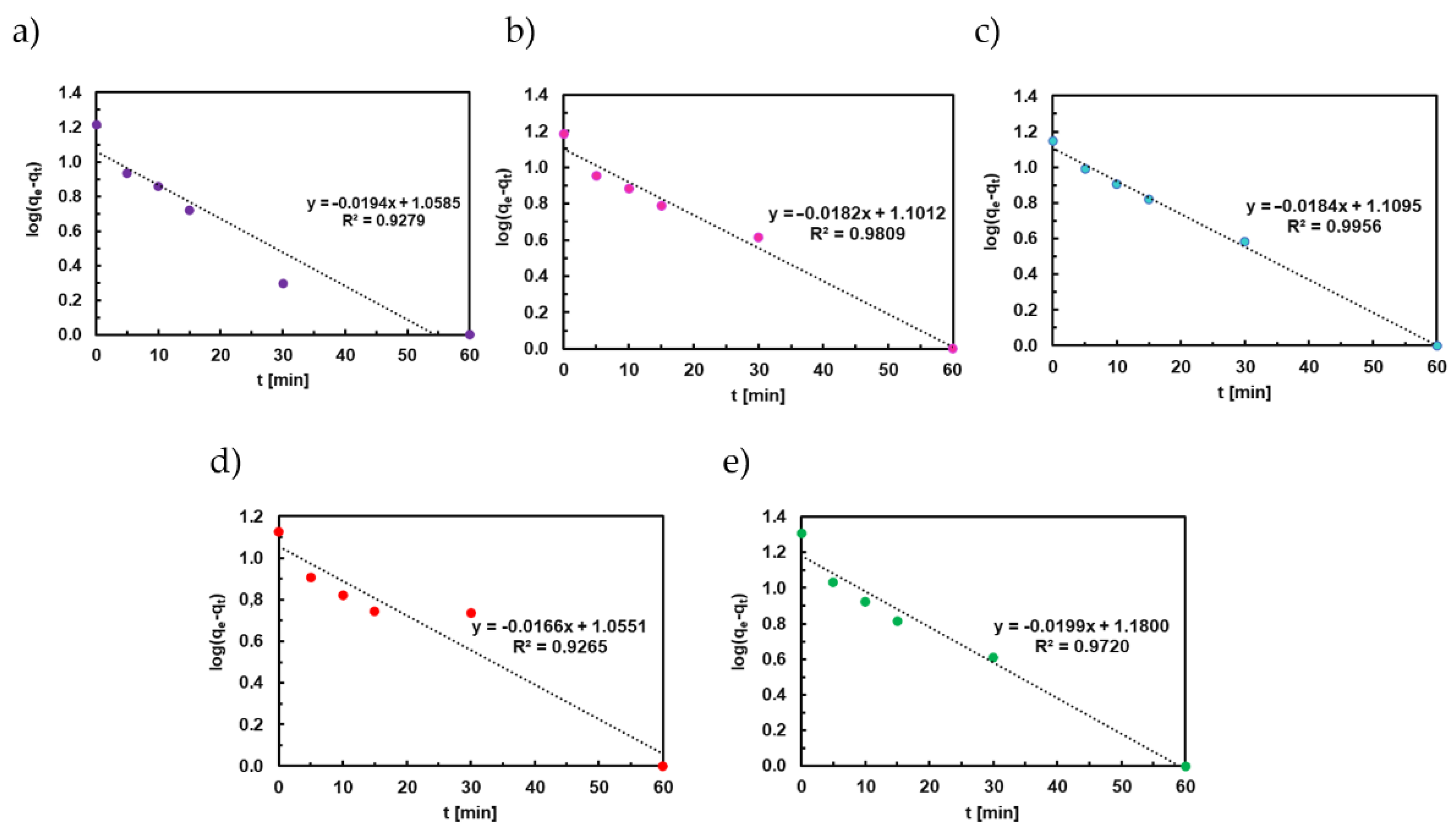
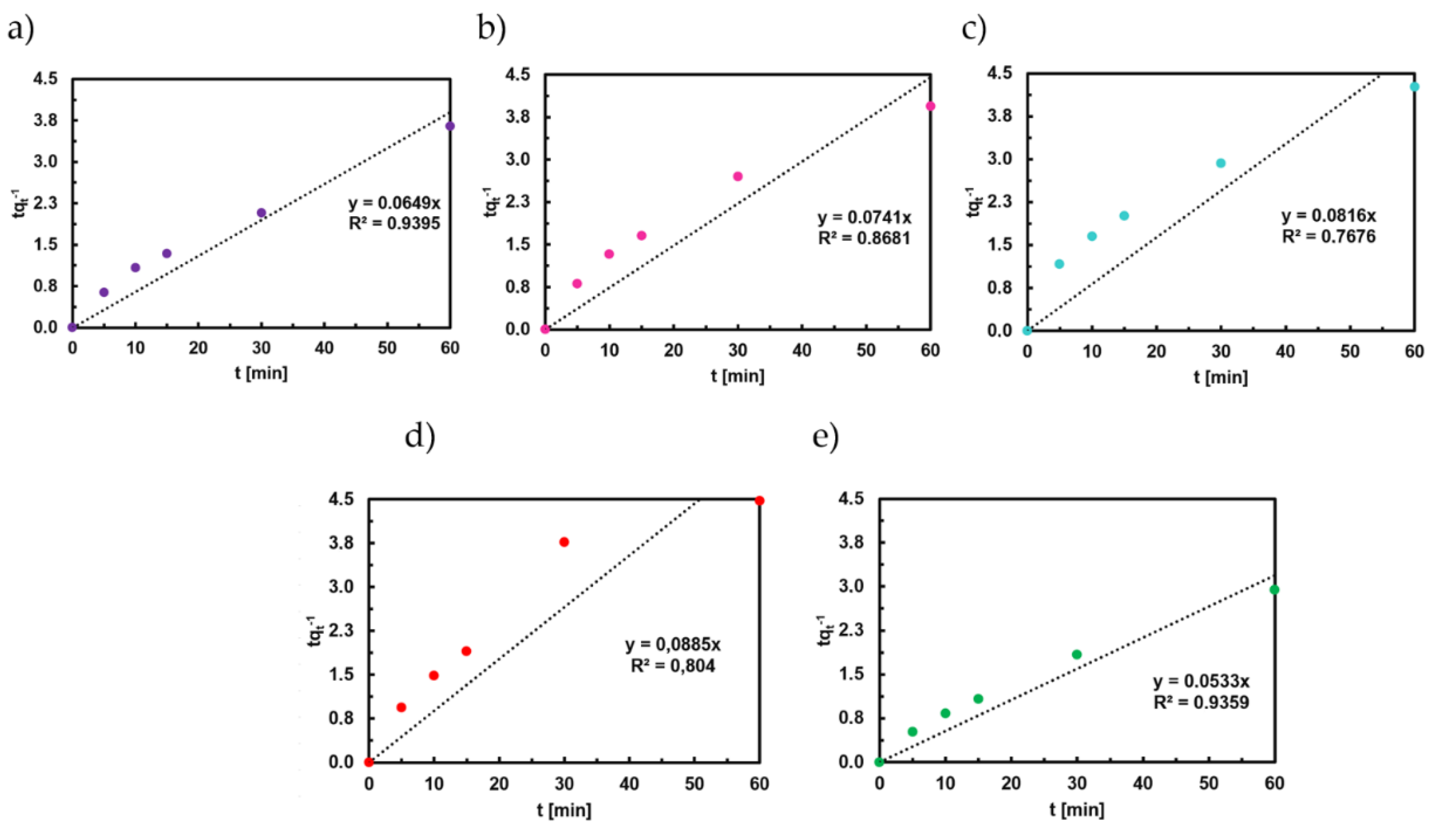
3.7. Adsorption Kinetics of Titan Yellow
3.8. Adsorption Isotherm of Titan Yellow
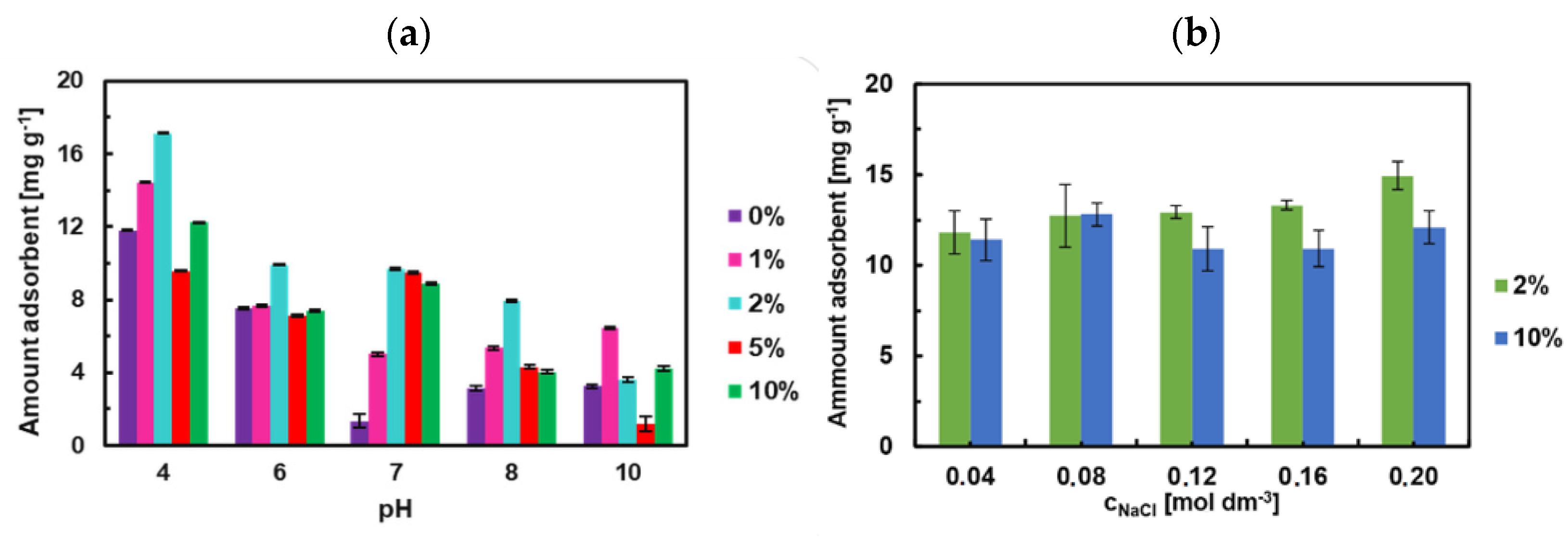
3.9. Effect of pH and Ionic Strength on the Adsorption of TY
4. Photocatalytic Studies
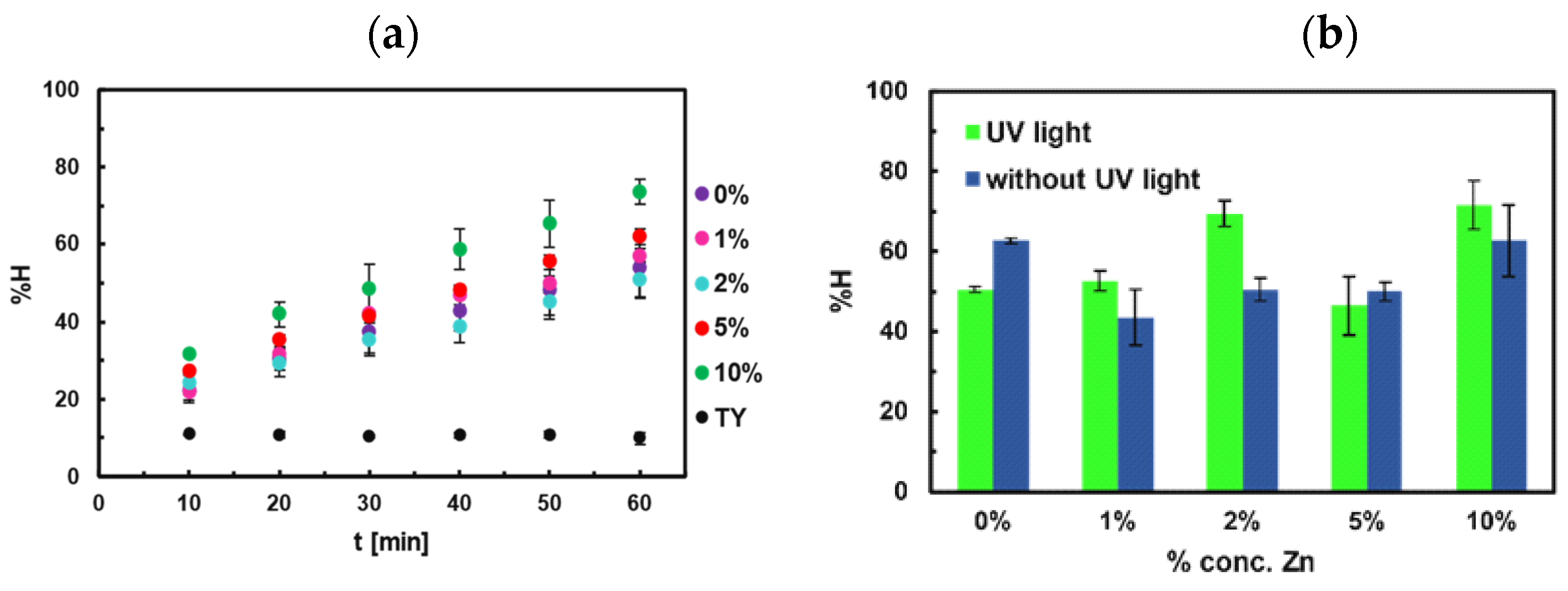
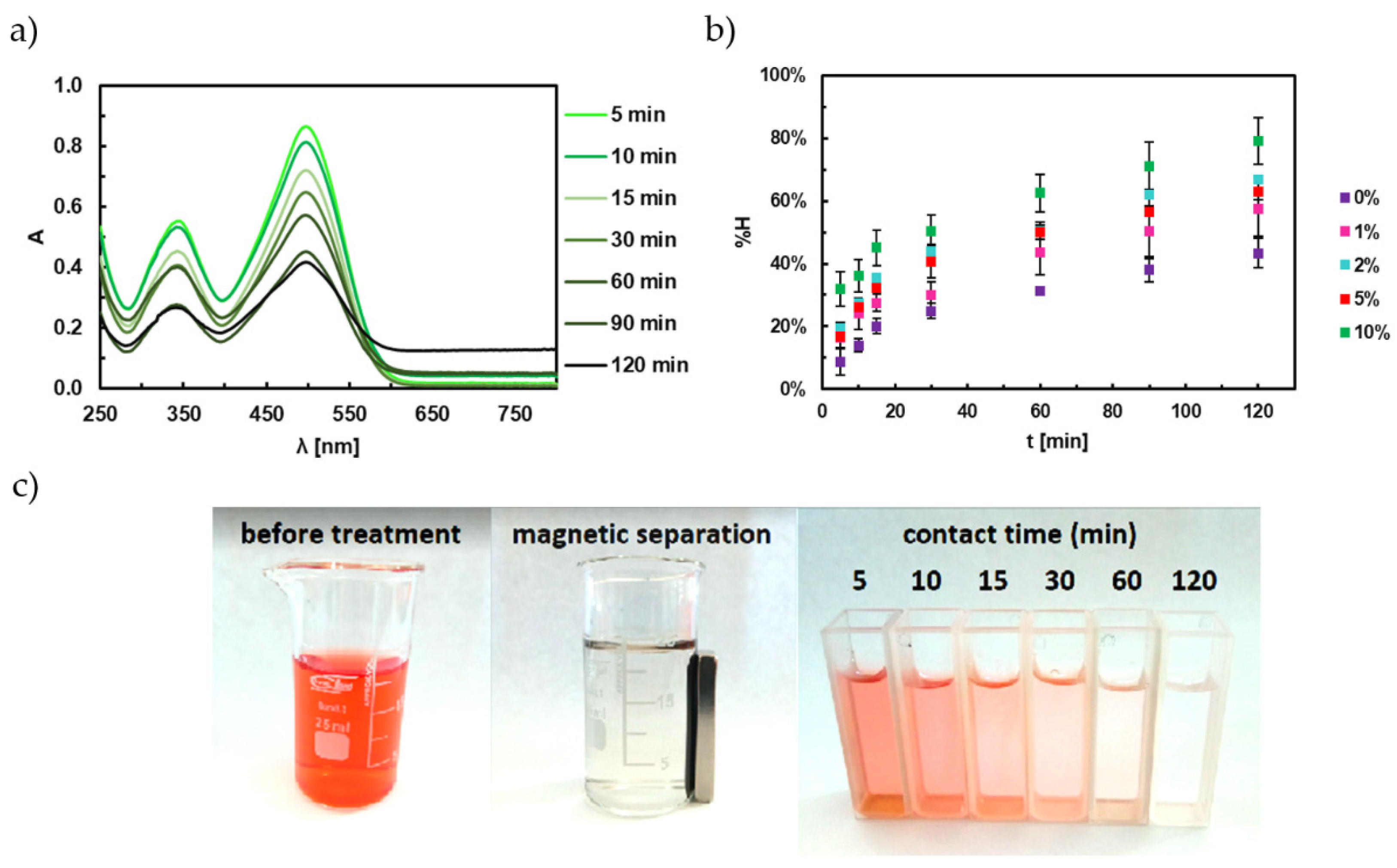
4.1. Adsorption Studies of Congo Red
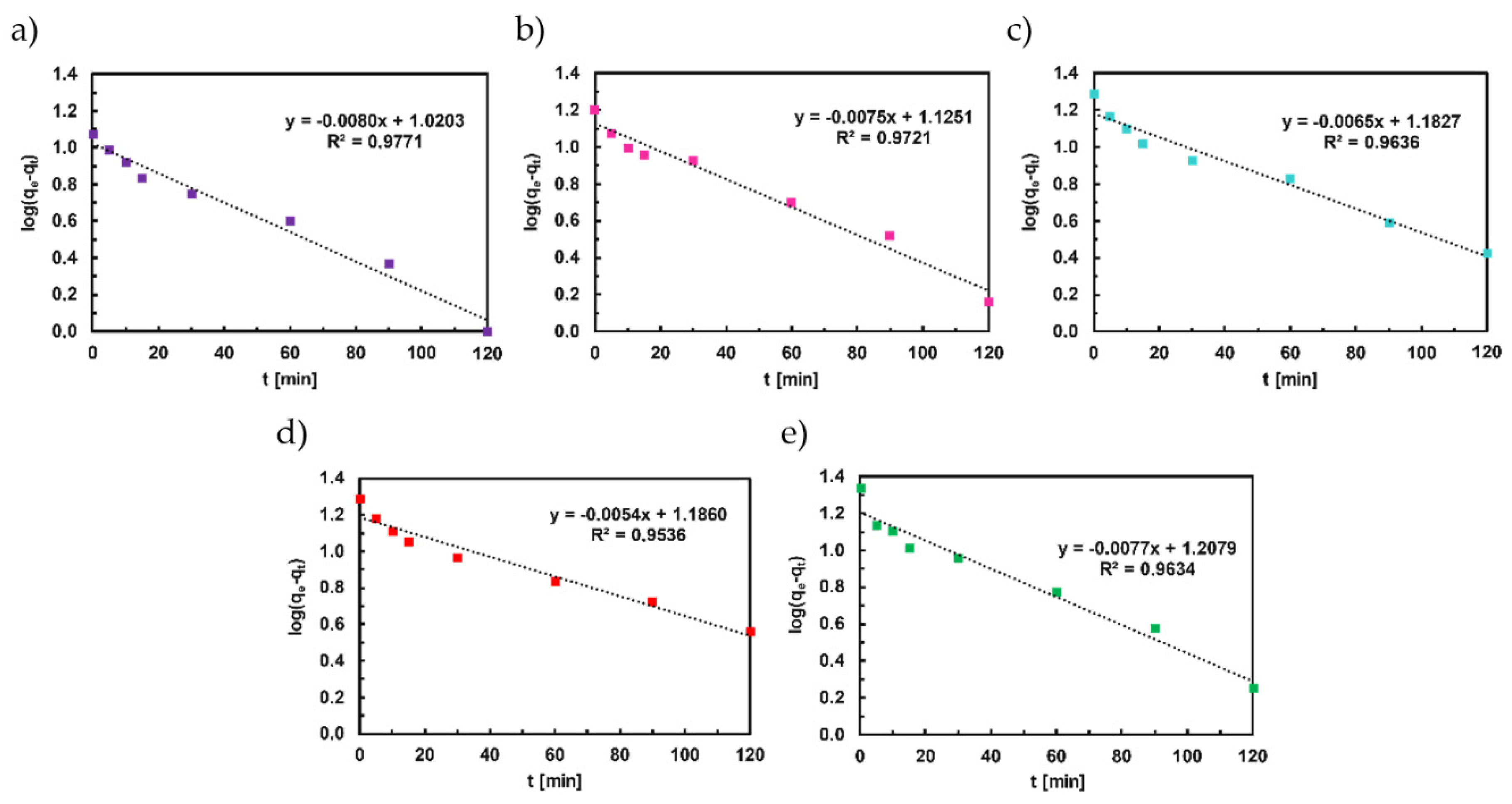
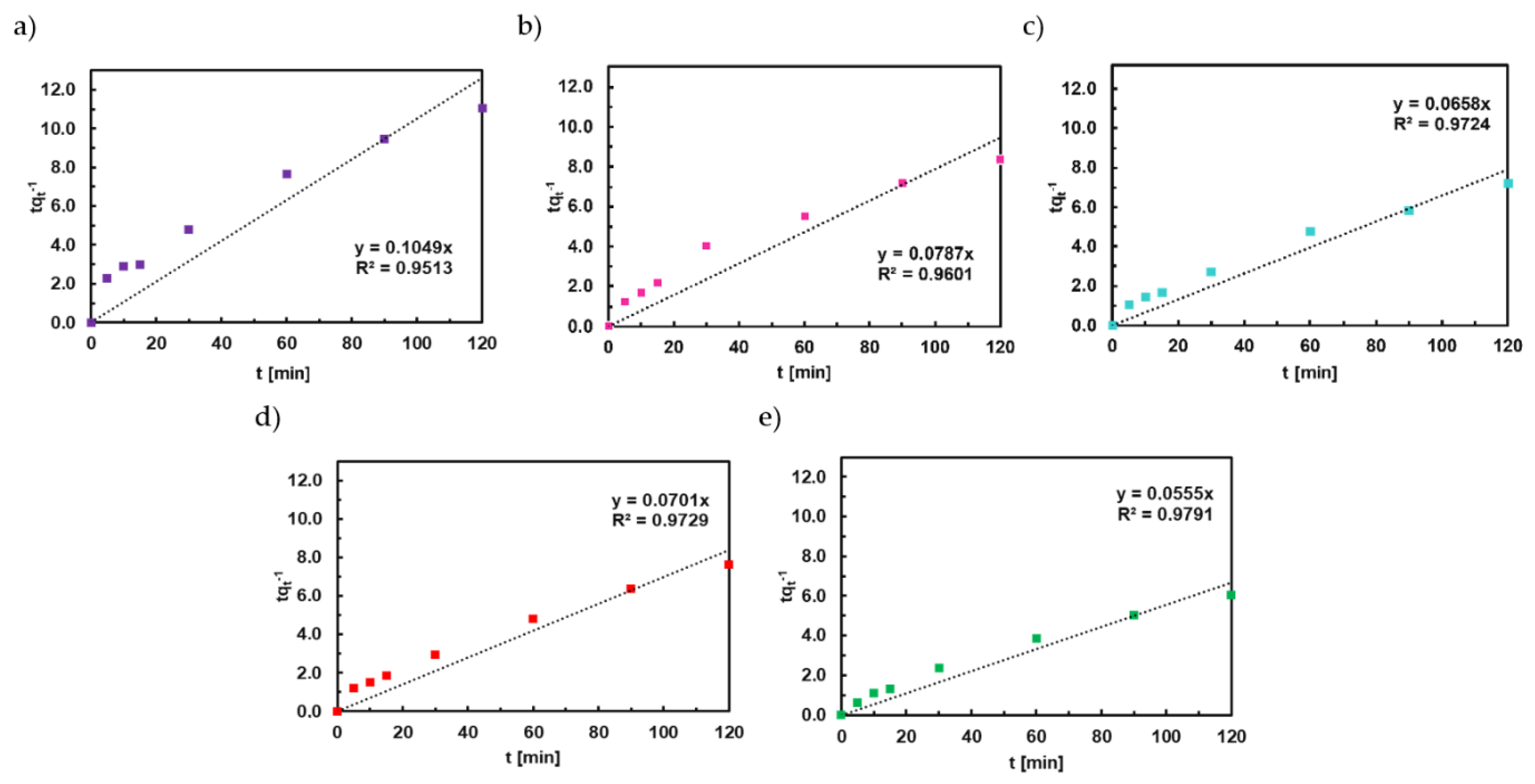
4.2. Adsorption Kinetics of Congo Red
4.3. Effect of pH and Ionic Strength on the Adsorption of CR
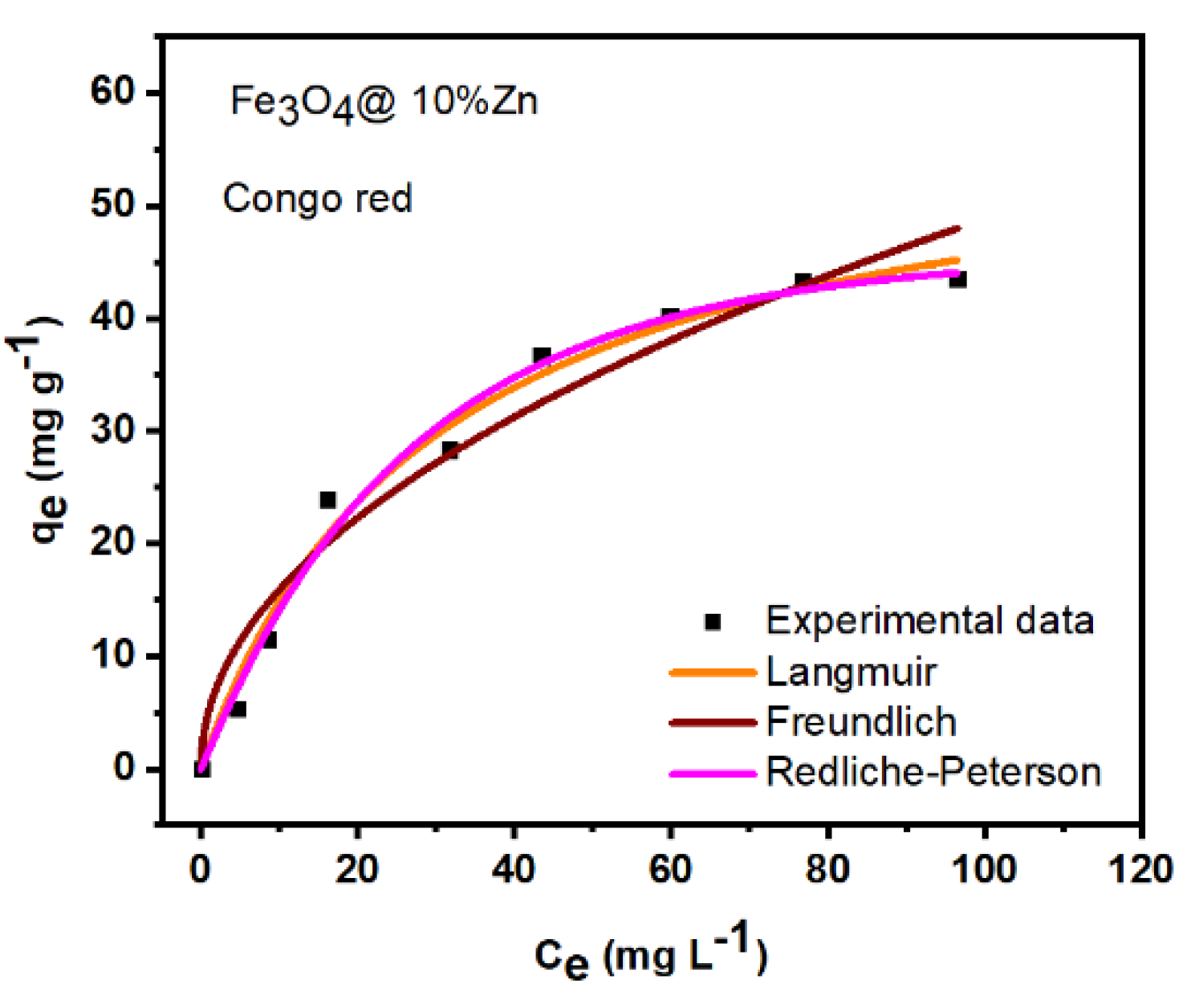
4.4. Adsorption Isotherm for Congo Red
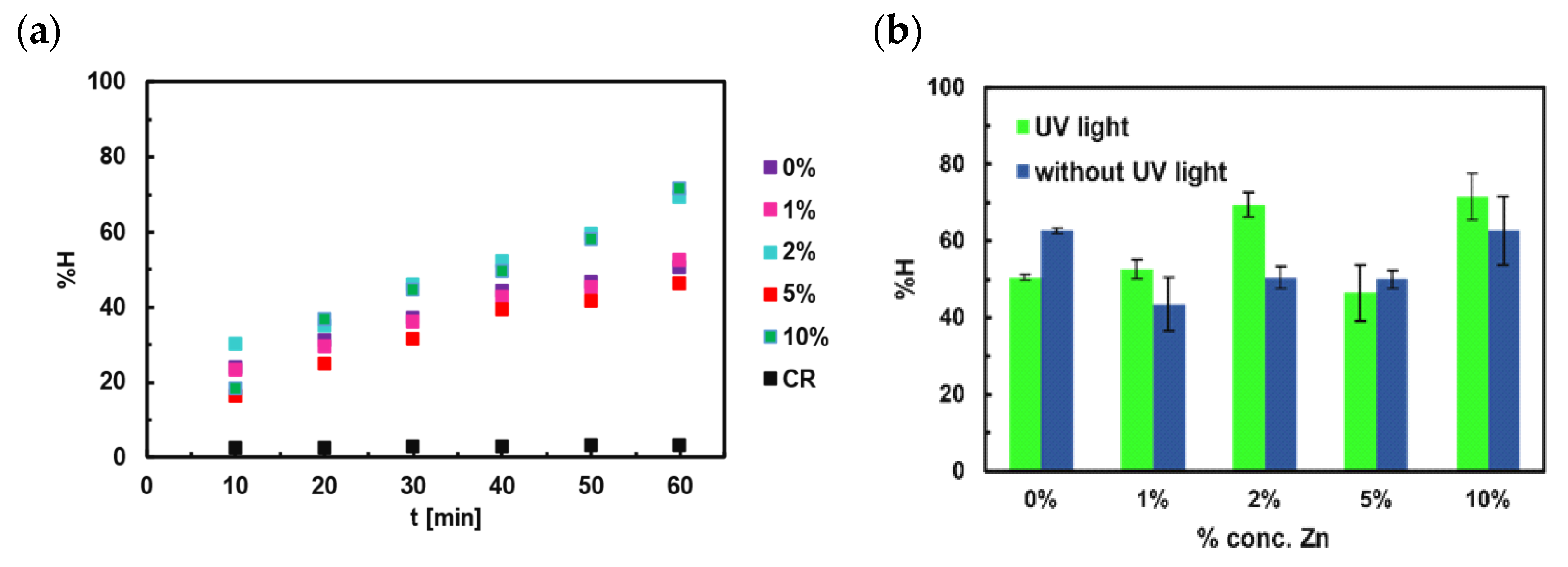
4.5. Photocatalytic Studies for Congo Red
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maheshwari, K.; Agrawal, M.; Gupta, A.B. Dye Pollution in Water and Wastewater. In Novel Materials for Dye-Containing Wastewater Treatment. Sustainable Textiles: Production, Processing, Manufacturing & Chemistry; Muthu, S.S., Khadir, A., Eds.; Springer: Singapore, 2021; pp. 1–25. [Google Scholar] [CrossRef]
- Lorenc-Grabowska, E.; Gryglewicz, G. Adsorption characteristics of Congo Red on coal-based mesoporous activated carbon. Dye. Pigment. 2007, 74, 34–40. [Google Scholar] [CrossRef]
- Le, P.T.; Bui, H.T.; Le, D.N.; Nguyen, T.H.; Pham, L.A.; Nguyen, H.N.; Nguyen, Q.S.; Nguyen, T.P.; Bich, N.T.; Duong, T.T.; et al. Preparation and Characterization of Biochar Derived from Agricultural By-Products for Dye Removal. Adsorpt. Sci. Technol. 2021, 2021, 14. [Google Scholar] [CrossRef]
- Maru, K.; Kalla, S.; Jangir, R. Dye contaminated wastewater treatment through metal–organic framework (MOF) based materials. New J. Chem. 2022, 46, 3054–3072. [Google Scholar] [CrossRef]
- Armağan, B.; Turan, M.; Çelik, M.S. Equilibrium studies on the adsorption of reactive azo dyes into zeolite. Desalination 2004, 170, 33–39. [Google Scholar] [CrossRef]
- Yang, X.; Wang, L.; Shao, X.; Tong, J.; Chen, R.; Yang, Q.; Yang, X.; Li, G.; Zimmerman, A.R.; Gao, B. Preparation of biosorbent for the removal of organic dyes from aqueous solution via one-step alkaline ball milling of hickory wood. Bioresour. Technol. 2022, 348, 126831. [Google Scholar] [CrossRef] [PubMed]
- Ayati, A.; Shahrak, M.N.; Tanhaei, B.; Sillanpää, M. Emerging adsorptive removal of azo dye by metal–organic frameworks. Chemosphere 2016, 160, 30–44. [Google Scholar] [CrossRef] [PubMed]
- Elwakeel, K.Z.; Shahat, A.; Khan, Z.A.; Alshitari, W.; Guibal, E. Magnetic metal oxide-organic framework material for ultrasonic-assisted sorption of titan yellow and rose bengal from aqueous solutions. Chem. Eng. J. 2020, 392, 123635. [Google Scholar] [CrossRef]
- Mishra, A.K.; Arockiadoss, T.; Ramaprabhu, S. Study of removal of azo dye by functionalized multi walled carbon nanotubes. Chem. Eng. J. 2010, 162, 1026–1034. [Google Scholar] [CrossRef]
- Hou, H.; Zhou, R.; Wu, P.; Wu, L. Removal of Congo red dye from aqueous solution with hydroxyapatite/chitosan composite. Chem. Eng. J. 2012, 211–212, 336–342. [Google Scholar] [CrossRef]
- Jiang, Y.; Gong, J.L.; Zeng, G.M.; Ou, X.M.; Chang, Y.N.; Deng, C.H.; Zhang, J.; Liu, H.Y.; Huang, S.Y. Magnetic chitosan-graphene oxide composite for anti-microbial and dye removal applications. Int. J. Biol. Macromol. 2016, 82, 702–710. [Google Scholar] [CrossRef] [PubMed]
- Nwaji, N.; Akinoglu, E.M.; Giersig, M. Gold Nanoparticle-Decorated Bi2S3 Nanorods and Nanoflowers for Photocatalytic Wastewater Treatment. Catalysts 2021, 11, 355. [Google Scholar] [CrossRef]
- Ghaemi, M.; Absalan, G.; Sheikhian, L. Adsorption characteristics of Titan yellow and Congo red on CoFe2O4 magnetic nanoparticles. J. Iran. Chem. Soc. 2014, 11, 1759–1766. [Google Scholar] [CrossRef]
- Hou, X.; Feng, J.; Liu, X.; Ren, Y.; Fan, Z.; Wei, T.; Meng, J.; Zhang, M. Synthesis of 3D porous ferromagnetic NiFe2O4 and using as novel adsorbent to treat wastewater. J Colloid Interface Sci. 2011, 362, 477–485. [Google Scholar] [CrossRef]
- Tabaraki, R.; Sadeghinejad, N. Comparison of magnetic Fe3O4/chitosan and arginine-modified magnetic Fe3O4/chitosan nanoparticles in simultaneous multidye removal: Experimental design and multicomponent analysis. Int. J. Biol. Macromol. 2018, 120, 2313–2323. [Google Scholar] [CrossRef]
- Hu, L.; Guang, C.; Liu, Y.; Su, Z.; Gong, S.; Yao, Y.; Wang, Y. Adsorption behavior of dyes from an aqueous solution onto composite magnetic lignin adsorbent. Chemosphere 2020, 246, 125757. [Google Scholar] [CrossRef] [PubMed]
- Salem, M.A.; Salem, I.A.; Hanfy, M.G.; Zaki, A.B. Removal of titan yellow dye from aqueous solution by polyaniline/Fe3O4 nanocomposite. Eur. Chem. Bull 2016, 5, 113–118. [Google Scholar]
- Ahmed, K.A.M. Formation Fe2O3 Nanowalls through Solvent-Assisted Hydrothermal Process and Their Application for Titan Yellow GR Dye Degradation. J. Korean Chem. Soc. 2014, 58, 205–209. [Google Scholar] [CrossRef]
- Akrami, A.; Niazi, A. Synthesis of maghemite nanoparticles and its application for removal of Titan yellow from aqueous solutions using full factorial design. Desalin. Water Treat. 2016, 57, 22618–22631. [Google Scholar] [CrossRef]
- Brown, M.A.; De Vito, S.C. Predicting Azo Dye Toxicity. Crit. Rev. Environ. Sci. Technol. 1993, 23, 249–324. [Google Scholar] [CrossRef]
- Golka, K.; Kopps, S.; Myslak, Z.W. Carcinogenicity of azo colorants: Influence of solubility and bioavailability. Toxicol. Lett. 2004, 151, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Zamora, M.; Martínez-Jerónimo, F. Congo red dye diversely affects organisms of different trophic levels: A comparative study with microalgae, cladocerans, and zebrafish embryos. Environ. Sci. Pollut. Res. Int. 2019, 26, 11743–11755. [Google Scholar] [CrossRef]
- Sharmila, M.; Mani, R.J.; Kader, S.M.A. Novel synthesis of undoped and zinc doped iron oxide nanoparticles using Apis Mellifera. Mater. Today Proc. 2022, 50, 2647–2649. [Google Scholar] [CrossRef]
- Kiełbik, P.; Jończy, A.; Kaszewski, J.; Gralak, M.; Rosowska, J.; Sapierzyński, R.; Witkowski, B.; Wachnicki, Ł.; Lawniczak-Jablonska, K.; Kuzmiuk, P.; et al. Biodegradable Zinc Oxide Nanoparticles Doped with Iron as Carriers of Exogenous Iron in the Living Organism. Pharmaceuticals 2021, 14, 859. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Bu, W.; Lu, C.; Chen, C.; Chen, M.; Shen, X.; Liu, R.; Shi, J. Hydrothermal synthesis of a highly sensitive T2-weigthed MRI contrast agent: Zinc-doped superparamagnetic iron oxide nanocrystals. J. Nanosci. Nanotechnol. 2011, 11, 10438–10443. [Google Scholar] [CrossRef]
- Moise, S.; Céspedes, E.; Soukup, D.; Byrne, J.M.; El Haj, A.J.; Telling, N.D. The cellular magnetic response and biocompatibility of biogenic zinc- and cobalt-doped magnetite nanoparticles. Sci. Rep. 2017, 7, 39922. [Google Scholar] [CrossRef]
- Paganini, M.C.; Giorgini, A.; Gonçalves, N.P.F.; Gionco, C.; Prevot, A.B.; Calza, P. New insight into zinc oxide doped with iron and its exploitation to pollutants abatement. Catal. Today 2019, 328, 230–234. [Google Scholar] [CrossRef]
- Joseph, J.A.; Nair, S.B.; John, S.S.; Remillard, S.K.; Shaji, S.; Philip, R.R. Zinc-doped iron oxide nanostructures for enhanced photocatalytic and antimicrobial applications. J. Appl. Electrochem. 2021, 51, 521–538. [Google Scholar] [CrossRef]
- Olusegun, S.J.; Mohallem, N.D.S. Comparative adsorption mechanism of doxycycline and Congo red using synthesized kaolinite supported CoFe2O4 nanoparticles. Environ. Pollut. 2020, 260, 114019. [Google Scholar] [CrossRef]
- Wang, J.; Yang, J.; Li, X.; Wang, D.; Wei, B.; Song, H.; Li, X.; Fu, S. Preparation and photocatalytic properties of magnetically reusable Fe3O4@ZnO core/shell nanoparticles. Phys. E Low-Dimens. Syst. Nanostruct. 2016, 75, 66–71. [Google Scholar] [CrossRef]
- Liu, J.; Zeng, M.; Yu, R. Surfactant-free synthesis of octahedral ZnO/ZnFe2O4 heterostructure with ultrahigh and selective adsorption capacity of malachite green. Sci. Rep. 2016, 6, 25074. [Google Scholar] [CrossRef]
- Yang, C.; Florent, M.; de Falco, G.; Fan, H.; Bandosz, T.J. ZnFe2O4/activated carbon as a regenerable adsorbent for catalytic removal of H2S from air at room temperature. Chem. Eng. J. 2020, 394, 124906. [Google Scholar] [CrossRef]
- Yavuz, E.; Tokalıoğlu, Ş.; Patat, Ş. Magnetic dispersive solid phase extraction with graphene/ZnFe2O4 nanocomposite adsorbent for the sensitive determination of mercury in water and fish samples by cold vapor atomic absorption spectrometry. Microchem. J. 2018, 142, 85–93. [Google Scholar] [CrossRef]
- Wang, W.D.; Cui, Y.X.; Zhang, L.K.; Li, Y.M.; Sun, P.; Han, J.H. Synthesis of a novel ZnFe2O4/porous biochar magnetic composite for Th(IV) adsorption in aqueous solutions. Int. J. Environ. Sci. Technol. 2021, 18, 2733–2746. [Google Scholar] [CrossRef]
- Olusegun, S.J.; Larrea, G.; Osial, M.; Jackowska, K.; Krysinski, P. Photocatalytic Degradation of Antibiotics by Superparamagnetic Iron Oxide Nanoparticles. Tetracycline Case. Catalysts 2021, 11, 1243. [Google Scholar] [CrossRef]
- Osial, M.; Rybicka, P.; Pękała, M.; Cichowicz, G.; Cyrański, M.K.; Krysiński, P. Easy Synthesis and Characterization of Holmium-Doped SPIONs. Nanomaterials 2018, 8, 430. [Google Scholar] [CrossRef] [PubMed]
- Nieciecka, D.; Celej, J.; Żuk, M.; Majkowska-Pilip, A.; Żelechowska-Matysiak, K.; Lis, A.; Osial, M. Hybrid System for Local Drug Delivery and Magnetic Hyperthermia Based on SPIONs Loaded with Doxorubicin and Epirubicin. Pharmaceutics 2021, 13, 480. [Google Scholar] [CrossRef]
- Strobel, R.; Pratsinis, S.E. Direct synthesis of maghemite, magnetite and wustite nanoparticles by flame spray pyrolysis. Adv. Powder Technol. 2009, 20, 190–194. [Google Scholar] [CrossRef]
- Jozwiak, W.K.; Kaczmarek, E.; Maniecki, T.P.; Ignaczak, W.; Maniukiewicz, W. Reduction behavior of iron oxides in hydrogen and carbon monoxide atmospheres. Appl. Catal. A Gen. 2007, 326, 17–27. [Google Scholar] [CrossRef]
- Amos-Tautua, B.M.; Fakayode, O.J.; Songca, S.P.; Oluwafemi, O.S. Effect of synthetic conditions on the crystallinity, porosity and magnetic properties of gluconic acid capped iron oxide nanoparticles. Nano-Struct. Nano-Objects 2020, 23, 100480. [Google Scholar] [CrossRef]
- Demortière, A.; Panissod, P.; Pichon, B.P.; Pourroy, G.; Guillon, D.; Donnio, B.; Bégin-Colin, S. Size-dependent properties of magnetic iron oxidenanocrystals. Nanoscale 2011, 3, 225–232. [Google Scholar] [CrossRef]
- Saritha, A.; Raju, B.; Rao, D.N.; Roychowdhury, A.; Das, D.; Hussain, K.A. Facile green synthesis of iron oxide nanoparticles via solid-state thermolysis of a chiral, 3D anhydrous potassium tris(oxalato)ferrate(III) precursor. Adv. Powder Technol. 2015, 26, 349–354. [Google Scholar] [CrossRef]
- Anjana, P.M.; Bindhu, M.R.; Umadevi, M.; Rakhi, R.B. Antimicrobial, electrochemical and photo catalytic activities of Zn doped Fe3O4 nanoparticles. J. Mater. Sci. Mater. Electron. 2018, 29, 6040–6050. [Google Scholar] [CrossRef]
- Nguyen, X.S.; Zhang, G.; Yang, X. Mesocrystalline Zn-Doped Fe3O4 Hollow Submicrospheres: Formation Mechanism and Enhanced Photo-Fenton Catalytic Performance. ACS Appl. Mater. Interfaces 2017, 9, 8900–8909. [Google Scholar] [CrossRef] [PubMed]
- Aghazadeh, M. Zn-doped magnetite nanoparticles: Development of novel preparation method and evaluation of magnetic and electrochemical capacitance performances. J. Mater. Sci. Mater. Electron. 2017, 28, 18755–18764. [Google Scholar] [CrossRef]
- Paz-Díaz, B.; Vázquez-Olmos, A.R.; Almaguer-Flores, A.; García-Pérez, V.I.; Sato-Berrú, R.Y.; Almanza-Arjona, Y.C.; Garibay-Febles, V. ZnFe2O4 and CuFe2O4 Nanocrystals: Synthesis, Characterization, and Bactericidal Application. J. Clust. Sci. 2021, 32, 1–9. [Google Scholar] [CrossRef]
- Shanmugavania, A.; Selvan, R.K. Synthesis of ZnFe2O4 nanoparticles and their asymmetric configuration with Ni(OH)2 for a pseudocapacitor. RSC Adv. 2014, 4, 27022–27029. [Google Scholar] [CrossRef]
- Sun, S.; Yang, X.; Zhang, X.; Zhang, F.; Ding, J.; Bao, J.; Gao, C. Enhanced photocatalytic activity of sponge-like ZnFe2O4 synthesized by solution combustion method. Prog. Nat. Sci. Mater. Int. 2012, 22, 639–643. [Google Scholar] [CrossRef]
- Yao, J.; Yan, J.; Huang, Y.; Li, Y.; Xiao, S.; Xiao, J. Preparation of ZnFe2O4/α-Fe2O3 Nanocomposites from Sulfuric Acid Leaching Liquor of Jarosite Residue and Their Application in Lithium-Ion Batteries. Front. Chem. 2018, 6, 422. [Google Scholar] [CrossRef]
- Briche, S.; Belaiche, M. Photocatalytic study of nanoferrites elaborated by sol-gel process for environmental applications. In Proceedings of the 2016 International Renewable and Sustainable Energy Conference (IRSEC), Marrakech, Morocco, 14–17 November 2016; pp. 789–792. [Google Scholar] [CrossRef]
- Fuentes-García, J.A.; Diaz-Cano, A.I.; Guillen-Cervantes, A.; Santoyo-Salazar, J. Magnetic domain interactions of Fe3O4 nanoparticles embedded in a SiO2 matrix. Sci. Rep. 2018, 8, 5096. [Google Scholar] [CrossRef]
- Sunil, K.C.; Utsav, S.; Nairy, R.K.; Chethan, G.; Shenoy, S.P.; Mustak, M.S.; Yerol, N. Synthesis and characterization of Zn0.4Co0.6Fe2O4 superparamagnetic nanoparticles as a promising agent against proliferation of colorectal cancer cells. Ceram. Int. 2021, 47, 19026–19035. [Google Scholar] [CrossRef]
- Correa-Duarte, M.A.; Grzelczak, M.; Salgueiriño-Maceira, V.; Giersig, M.; Liz-Marzán, L.M.; Farle, M.; Sierazdki, K.; Diaz, R. Alignment of Carbon Nanotubes under Low Magnetic Fields through Attachment of Magnetic Nanoparticles. J. Phys. Chem. B 2005, 109, 19060–19063. [Google Scholar] [CrossRef] [PubMed]
- Le, D.T.; Le, T.P.T.; Do, H.T.; Vo, H.T.; Pham, N.T.; Nguyen, T.T.; Cao, H.T.; Nguyen, P.T.; Dinh, T.M.T.; Le, H.V.; et al. Fabrication of Porous Hydroxyapatite Granules as an Effective Adsorbent for the Removal of Aqueous Pb(II) Ions. J. Chem. 2019, 2019, 8620181. [Google Scholar] [CrossRef]
- Tran, H.N.; You, S.J.; Hosseini-Bandegharaei, A.; Chao, H.P. Mistakes and inconsistencies regarding adsorption of contaminants from aqueous solutions: A critical review. Water Res. 2017, 120, 88–116. [Google Scholar] [CrossRef] [PubMed]
- Lima, É.C.; Adebayo, M.A.; Fernando, M.M. Chapter 3—Kinetic and Equilibrium Models of Adsorption. In Carbon Nanomaterials as Adsorbents for Environmental and Biological Applications; Bergmann, C.P., Machado, F.M., Eds.; Springer: São Paulo, Brazil, 2015; pp. 33–71. [Google Scholar] [CrossRef]
- Bameri, I.; Saffari, J.; Baniyaghoob, S.; Ekrami-Kakhki, M.S. Synthesis of magnetic nano-NiFe2O4 with the assistance of ultrasound and its application for photocatalytic degradation of Titan Yellow: Kinetic and isotherm studies. Colloids Interface Sci. Commun. 2020, 48, 100610. [Google Scholar] [CrossRef]
- Shi, Q.X.; Li, Y.; Wang, L.; Wang, J.; Cao, Y.L. Preparation of supported chitosan adsorbent with high adsorption capacity for Titan Yellow removal. Int. J. Biol. Macromol. 2020, 152, 449–455. [Google Scholar] [CrossRef]
- Lakshmi, A.N.; Dhamodaran, M.; Samu Solomon, J. Thermodynamics and kinetics of adsorption of azo dye titan yellow from aqueous solutions on natural plant material Saccharum spontaneum. Pharma Chem. 2015, 7, 36–45. [Google Scholar]
- Ibrahim, H.K.; Albo, M.A.; Allah, H.; Al-da, M.A. Adsorption of Titan Yellow Using Walnut Husks: Thermodynamics, Kinetics and Isotherm Studies. Ann. Rom. Soc. Cell Biol. 2021, 25, 12576–12587. [Google Scholar]
- Okoronkwo, A.E.; Olusegun, S.J. Biosorption of nickel using unmodified and modified lignin extracted from agricultural waste. Desalin. Water Treat. 2013, 51, 1989–1997. [Google Scholar] [CrossRef]
- Vidya, C.; Manjunatha, C.; Sudeep, M.; Ashoka, S.; Lourdu Antony Raj, M.A. Photo-assisted mineralisation of titan yellow dye using ZnO nanorods synthesised via environmental benign route. SN Appl. Sci. 2020, 2, 743. [Google Scholar] [CrossRef]
- Kooh, M.R.R.; Dahri, M.K.; Lim, L.B.L. The removal of rhodamine B dye from aqueous solution using Casuarina equisetifolia needles as adsorbent. Cogent Environ. Sci. 2016, 2, 1140553. [Google Scholar] [CrossRef]
- Olusegun, S.J.; Mohallem, N.D.S. Insight into the adsorption of doxycycline hydrochloride on di ff erent thermally treated hierarchical CoFe2O4/bio-silica nanocomposite. J. Environ. Chem. Eng. 2019, 7, 103442. [Google Scholar] [CrossRef]
- Samiey, B.; Ashoori, F. Adsorptive removal of methylene blue by agar: Effects of NaCl and ethanol. Chem. Cent. J. 2012, 6, 14. [Google Scholar] [CrossRef] [PubMed]
- Olusegun, S.J.; Freitas, E.T.F.; Lara, L.R.S.; Mohallem, N.D.S. Synergistic effect of a spinel ferrite on the adsorption capacity of nano bio-silica for the removal of methylene blue. Environ. Technol. 2021, 42, 2163–2176. [Google Scholar] [CrossRef]
- Azazy, M.; Dimassi, S.N.; El-shafie, A.S.; Issa, A.A. Bio-Waste Aloe vera Leaves as an Efficient Adsorbent for Titan Yellow from Wastewater: Structuring of a Novel Adsorbent Using Plackett-Burman Factorial Design. Appl. Sci. 2019, 9, 4856. [Google Scholar] [CrossRef]
- Raval, N.P.; Shah, P.U.; Shah, N.K. Adsorptive amputation of hazardous azo dye Congo red from wastewater: A critical review. Environ. Sci. Pollut. Res. 2016, 23, 14810–14853. [Google Scholar] [CrossRef] [PubMed]
- Farrokhi, M.; Hosseini, S.C.; Yang, J.K.; Shirzad-Siboni, M. Application of ZnO–Fe3O4 Nanocomposite on the Removal of Azo Dye from Aqueous Solutions: Kinetics and Equilibrium Studies. Water Air Soil Pollut. 2014, 225, 2113. [Google Scholar] [CrossRef]
- Chakraborty, S.; Farida, J.J.; Simon, R.; Kasthuri, S.; Mary, N.L. Averrhoe carrambola fruit extract assisted green synthesis of zno nanoparticles for the photodegradation of congo red dye. Surf. Interfaces 2020, 19, 100488. [Google Scholar] [CrossRef]
- Karamipour, A.; Rasouli, N.; Movahedi, M.; Salavati, H. A Kinetic Study on Adsorption of Congo Red from Aqueous Solution by ZnOZnFe2O4-polypyrrole Magnetic Nanocomposite. Phys. Chem. Res. 2016, 4, 291–301. [Google Scholar] [CrossRef]
- Rath, P.P.; Behera, S.S.; Priyadarshini, B.; Panda, S.R.; Mandal, D.; Sahoo, T.; Mishra, S.; Sahoo, T.R.; Parhi, P.K. Influence of Mg doping on ZnO NPs for enhanced adsorption activity of Congo Red dye. Appl. Surf. Sci. 2019, 491, 256–266. [Google Scholar] [CrossRef]
- Xu, Y.; Jin, J.; Li, X.; Han, Y.; Meng, H.; Wang, T.; Zhang, X. Fabrication of hybrid magnetic HKUST-1 and its highly efficient adsorption performance for Congo red dye. RSC Adv. 2015, 25, 19199–19202. [Google Scholar] [CrossRef]
- Li, L.; Li, X.; Duan, H.; Wang, X.; Luo, C. Removal of Congo Red by magnetic mesoporous titanium dioxide–graphene oxide core–shell microspheres for water purification. Dalton Trans. 2014, 43, 8431–8438. [Google Scholar] [CrossRef] [PubMed]
- Mou, Y.; Yang, H.; Xu, Z. Morphology, Surface Layer Evolution, and Structure–Dye Adsorption Relationship of Porous Fe3O4 MNPs Prepared by Solvothermal/Gas Generation Process. ACS Sustain. Chem. Eng. 2017, 5, 2339–2349. [Google Scholar] [CrossRef]
- Maiti, D.; Mukhopadhyay, S.; Devi, P.S. Evaluation of Mechanism on Selective, Rapid, and Superior Adsorption of Congo Red by Reusable Mesoporous α-Fe2O3 Nanorods. ACS Sustain. Chem. Eng. 2017, 5, 11255–11267. [Google Scholar] [CrossRef]
- Said, M.; Rizki, W.T.; Asri, W.R.; Desnelli, D.; Rachmat, A.; Hariani, P.L. SnO2–Fe3O4 nanocomposites for the photodegradation of the Congo red dye. Heliyon 2022, 8, E09204. [Google Scholar] [CrossRef] [PubMed]






| Source of Ions | FeCl3·6H2O | FeCl2·4H2O | ZnCl2·4H2O |
| Fe3O4 | 1.35 g | 497.0 mg | - |
| Fe3O4@1%Zn | 493.6 mg | 3.40 mg | |
| Fe3O4@2%Zn | 490.2 mg | 6.80 mg | |
| Fe3O4@5%Zn | 472.1 mg | 24.85 mg | |
| Fe3O4@10%Zn | 447.3 mg | 49.70 mg |
| Sample | STotal (m2 g−1) | SMicro (cm2 g−1) | SMeso (cm2 g−1) | VMeso (cm3 g−1) | dMean pore (nm) |
|---|---|---|---|---|---|
| Fe3O4 | 97.9 | 0 | 100.4 | 0.17 | 6.78 |
| Fe3O4@1%Zn | 85.7 | 2.3 | 54.5 | 0.03 | 2.41 |
| Fe3O4@2%Zn | 93.1 | 0 | 70.0 | 0.04 | 2.29 |
| Fe3O4@5%Zn | 91.4 | 1.6 | 66.9 | 0.04 | 2.30 |
| Fe3O4@10%Zn | 101.5 | 5.2 | 63.3 | 0.04 | 2.40 |
| Nanoparticles | Band-Gap Energy [eV] |
|---|---|
| Fe3O4 | 2.35 ± 0.08 |
| Fe3O4@1%Zn | 2.35 ± 0.10 |
| Fe3O4@2%Zn | 2.34 ± 0.29 |
| Fe3O4@5%Zn | 2.33 ± 0.30 |
| Fe3O4@10%Zn | 2.24 ± 0.08 |
| Parameters | Fe3O4 | Fe3O4@10%Zn |
|---|---|---|
| Langmuir | ||
| qmax (mg g−1) | 30.0 | 43.0 |
| KL (L mg−1) | 0.041 | 0.094 |
| 0.966 | 0.985 | |
| SD (mg g−1) | 1.72 | 1.77 |
| Redlich-Peterson | ||
| KRP (L g−1) | 0.843 | 4.28 |
| aRP (mg L−1)-g | 0.004 | 0.107 |
| g | 1.39 | 0.981 |
| 0.985 | 0.983 | |
| SD (mg g−1) | 1.13 | 1.86 |
| Freundlich | ||
| KF (mg g−1) (mg L−1)−1/nF | 4.66 | 10.36 |
| nF | 2.79 | 3.40 |
| 0.889 | 0.937 | |
| SD (mg g−1) | 3.14 | 3.61 |
| Adsorbents | qmax (mg g−1) | References |
|---|---|---|
| NiFe2O4 | 19.193 | [56] |
| Chitosan | 58.76 | [57] |
| Saccharum spontaneum | 3.984 | [58] |
| Walnut Husks | 7.6982 | [59] |
| Bio-Waste Aloe vera Leaves | 55.25 | [60] |
| Fe3O4 | 30.0 | This work |
| Fe3O4@10%Zn | 43.0 | This work |
| Fe3O4@10%Zn | |
|---|---|
| Langmuir | |
| qmax (mg g−1) | 59 |
| KL (L mg−1) | 0.033 |
| 0.984 | |
| SD (mg g−1) | 2.07 |
| Redlich-Peterson | |
| KRP (L g−1) | 1.64 |
| aRP (mg L−1)-g | 0.009 |
| g | 1.2 |
| 0.985 | |
| SD (mg g−1) | 2.04 |
| Freundlich | |
| KF (mg g−1) (mg L−1)−1/nF | 5.183 |
| nF | 2.06 |
| 0.945 | |
| SD (mg g−1) | 3.75 |
| Adsorbents | qmax (mg g−1) | References |
|---|---|---|
| ZnO-polypyrrole magnetic nanocomposite | 38.0 | [71] |
| ZnO | 25.0 | [72] |
| magnetic HKUST-1 | 49.5 | [73] |
| mesoporous-TiO2/Fe3O4 | 50.0 | [74] |
| Fe3O4 | 28.46 | [75] |
| α-Fe3O4 | 57.2 | [76] |
| SnO2 | 24.27 | [77] |
| Fe3O4@10%Zn | 59.0 | This work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pietrzyk, P.; Phuong, N.T.; Olusegun, S.J.; Hong Nam, N.; Thanh, D.T.M.; Giersig, M.; Krysiński, P.; Osial, M. Titan Yellow and Congo Red Removal with Superparamagnetic Iron-Oxide-Based Nanoparticles Doped with Zinc. Magnetochemistry 2022, 8, 91. https://doi.org/10.3390/magnetochemistry8080091
Pietrzyk P, Phuong NT, Olusegun SJ, Hong Nam N, Thanh DTM, Giersig M, Krysiński P, Osial M. Titan Yellow and Congo Red Removal with Superparamagnetic Iron-Oxide-Based Nanoparticles Doped with Zinc. Magnetochemistry. 2022; 8(8):91. https://doi.org/10.3390/magnetochemistry8080091
Chicago/Turabian StylePietrzyk, Paulina, Nguyen Thu Phuong, Sunday Joseph Olusegun, Nguyen Hong Nam, Dinh Thi Mai Thanh, Michael Giersig, Paweł Krysiński, and Magdalena Osial. 2022. "Titan Yellow and Congo Red Removal with Superparamagnetic Iron-Oxide-Based Nanoparticles Doped with Zinc" Magnetochemistry 8, no. 8: 91. https://doi.org/10.3390/magnetochemistry8080091
APA StylePietrzyk, P., Phuong, N. T., Olusegun, S. J., Hong Nam, N., Thanh, D. T. M., Giersig, M., Krysiński, P., & Osial, M. (2022). Titan Yellow and Congo Red Removal with Superparamagnetic Iron-Oxide-Based Nanoparticles Doped with Zinc. Magnetochemistry, 8(8), 91. https://doi.org/10.3390/magnetochemistry8080091








