Abstract
Lilies are important crops that are commonly used as cut flowers (Lilium spp.) and edible bulb crops (Lilium davidii var. unicolor). However, virus infections can significantly impact the quantity and quality of lily production. Various methods have been developed to eliminate viruses in lilies, including in vitro culture and virus detection techniques. Meristem culture is the most effective method, which can be combined with other techniques such as thermotherapy and chemotherapy. Nonetheless, virus elimination is affected by several factors, including cultivar, explants used, virus type, and duration of treatments. Efficient diagnostic methods, such as serological and molecular techniques, have been developed to detect viral infections in lilies, including enzyme-linked immunosorbent assay (ELISA) and real-time reverse transcriptase polymerase chain reaction (real-time RT-PCR). However, cross-contamination and multiple-virus contamination can lead to unreliable results, and more sophisticated protocols and systems have been developed to address these issues. The objective of this review is to provide a comprehensive overview of the development of lily virus eradication, detection strategies, challenges, and solutions associated with these procedures, and how more sophisticated approaches such as multiplex RT-PCR, indirect ELISA (ID-ELISA), immunocapture RT-PCR (IC-RT-PCR), and immunochromatographic test strips (ICSs) can alleviate some of these setbacks.
1. Introduction
Lily is one of the world’s most important agro-industrial crops that is used as a cut flower (Lilium spp.) [1], food as edible bulbs (Lilium davidii var. unicolor) [2], and as a traditional medicinal plant [3]. The Lilium species known as ‘Baihe’ is grown in Northwestern China [1,2,4,5,6,7]. Lanzhou lily (Lilium davidii var. unicolor), a mutation of Lilium davidii ‘Duchartre ex Elwes’, has been used as an edible plant for over 150 years. The profitability of lily production has been enhanced by the effects of globalization of trade and it is ranked among the top ten most exported flowers on the global market [2,3]. It is also used in medicine as an antibacterial and anti-inflammatory product for bronchitis and pneumonia therapy [8]. Originating from the cold and temperate regions of the northern hemisphere, lily has a taxonomy that consists of about 110 accepted species [9,10]. Genetic studies have been conducted to explore the potential for interbreeding among various species within each taxonomic group [3,10]. Easter lily (Lilium longiflorum) and Asiatic and Oriental hybrids were found to be the three most commercially important types of lily [10,11,12,13]. Lilies can be infected by several viruses, including the lily symptomless virus (LSV), cucumber mosaic virus (CMV), and lily mottle virus (LMoV) (Figure 1). LSV is the most widespread virus in lilies, and it can cause vein clearing, light green stripes, reduced growth, smaller flowers, and shorter vase life of cut flowers. CMV has a wide host range and can cause vein clearing, chlorotic spots, and color-breaking patterns in flowers. LMoV is associated with light yellow mottling or mosaic and twisted leaves. These viruses can be detected through ELISA or PCR, and testing of bulbs after storage is more reliable than testing of leaves during flowering. The most sensitive detection method for these viruses is the tissue blot immunoassay (TBIA) on nitrocellulose membranes [14].
Vegetative propagation using bulblet scales is the conventional method used by farmers for lily plant propagation [15]. However, this method is associated with the susceptibility to viral, bacterial, and fungal diseases from one generation to another [2,16]. In addition, it has been found that aphids can serve as vectors for the transmission of viruses within and between crops [2]. It has been reported that lily-producing regions around the world are highly threatened by the aforementioned viral infections, causing serious loss of quality and yield [3,17].
Different cultivars have shown a reduction of 0–20% in bulb and flower yield due to the lily symptomless virus (LSV) [18,19]. In Lanzhou lily, virus infection has been reported to cause a decrease in yield production from 23,000 to 11,000 kg per hectare, accounting for over 50% of the reduction [19,20].
More than 20 species have been identified, as reported in previous research [17,20]. Research is actively underway to address the spread of lily viruses by developing a standardized protocol for producing virus-free seeds and seedlings. The aim is to establish an economically viable approach for lily plant production [5]. While it is important to acknowledge the significant progress made in reducing the incidence of major viruses in planting stock through advanced virus detection techniques such as immunoassay and molecular methods, it is essential to recognize that detection alone is insufficient in minimizing the presence of viruses [21]. To achieve effective virus control, a comprehensive approach encompassing both detection and elimination strategies is required. Therefore, in addition to the advancements in detection methods, efforts are being directed towards the development of reliable and efficient tools for virus elimination in lily plants. By combining these two aspects, researchers aim to establish a holistic management approach that ensures the production of healthy and virus-free lily plants for sustainable cultivation. The objective of this review is to compile and present knowledge on advancements in lily virus management techniques, including elimination and detection methods. The aim is to provide a comprehensive overview of the challenges faced in lily virus management and discuss potential solutions. By highlighting the limitations of current techniques and reviewing the emerging new sophisticated methods, the review seeks to address gaps in scientific knowledge and contribute to the improvement of lily virus management practices.

Figure 1.
Images of lily viruses: (I) each leaf has been infected by the virus and shows symptoms: (A) CMV, (B) LMoV, (C) LSV, (D) LMoV and LSV, (E) CMV, LMoV, and LSV, images acquired from Kwon, Ryu, and Choi, 2013 [22]. (II) Lilium martagon ‘Pink Taurade’ infected by lily symptomless virus, image acquired from Fisher, 2013 [23]. (III) Symptoms caused by lily-infecting viruses. Leaves are infected by (A) LSV, (B) CMV, and (C) LMoV, images acquired from Jo and Cho, 2018 [17]. (IV) Lily plant with mosaic symptoms on leaves, image acquired from Aravintharaj et al., 2017 [24].
Figure 1.
Images of lily viruses: (I) each leaf has been infected by the virus and shows symptoms: (A) CMV, (B) LMoV, (C) LSV, (D) LMoV and LSV, (E) CMV, LMoV, and LSV, images acquired from Kwon, Ryu, and Choi, 2013 [22]. (II) Lilium martagon ‘Pink Taurade’ infected by lily symptomless virus, image acquired from Fisher, 2013 [23]. (III) Symptoms caused by lily-infecting viruses. Leaves are infected by (A) LSV, (B) CMV, and (C) LMoV, images acquired from Jo and Cho, 2018 [17]. (IV) Lily plant with mosaic symptoms on leaves, image acquired from Aravintharaj et al., 2017 [24].

2. Methods for Lily Virus Elimination
2.1. Production of Virus-Free Lilies
Propagation through seed production and vegetative methods are the two commonly used methods for lily plant reproduction [3]. However, reliable in vitro techniques have been widely used in combination with other complementary techniques, including advanced virus detection methods and genetic engineering, in order to expedite lily multiplication, ensure disease-free crops, and shorten breeding programs [1,25,26,27,28,29]. In particular, the detection of viruses during the in vitro culture process holds significant importance, as it plays a vital role in generating virus-free lilies [3].
2.2. Methods to Eliminate Viruses from Lily Tissues
To control plant viral diseases and produce healthy plants free of viruses, the indexing process in lilies focuses on both axillary and apical meristems of the parent plant, the resulting plant, as well as the plant coming from the seed [30,31]. Viruses can be transmitted through the parent plant or the seed itself to the new plant, making indexing essential. By indexing meristems, newly grown plants, or seeds, experts can evaluate their virus-free status and ensure their health [32,33]. This is particularly crucial for viruses that can be vertically transmitted from the parent plant to the seed. The specific indexing practices may vary based on the type of virus, the plant species, and the desired level of assurance for producing healthy plants free of viruses [30,31,32]. Additionally, producing healthy seeds that have been indexed and shown to be free of known viruses is a common method for controlling plant viral diseases. Indexing methods involve selecting a representative subset of seeds from the batch for testing, typically based on statistical principles to ensure accuracy. Additionally, indexing includes the use of resistant indicator plants or acquiring seed, cuttings, or plants from certified virus-free sources [30,31,32,33]. Furthermore, ongoing investigations are exploring the elimination of lily virus through tissue culture-based methods (Table 1).
Thermotherapy, meristem tissue culture, and chemotherapy have been developed for lily virus elimination [5,34,35]. Studies show that the efficiency of the therapy depends upon the type of virus, the explants used for culture, and also on the hybrid infected [34,36,37]. In the case of eradicating lily symptomless virus (LSV) from Asiatic hybrids, in vitro culture of regenerated shoots from infected bulbs’ internal scales was employed successfully. However, this method was found to be ineffective in eliminating LSV from L. longiflorum Asiatic hybrids, such as ‘Ace’ and ‘Nellie White’ [36]. The above techniques can be used alone or as modified complex therapies [38,39,40]. Moreover, thermotherapy, meristem tissue culture, and chemotherapy techniques can be applied in in vitro culture systems or ex vitro environments [36]. Figure 2 illustrates a systematical approach encompassing sequential steps for the successful eradication of lily viruses, including LSV, LS, and LMoV, utilizing meristematic tips derived from bulblets generated ex vitro through scaling.
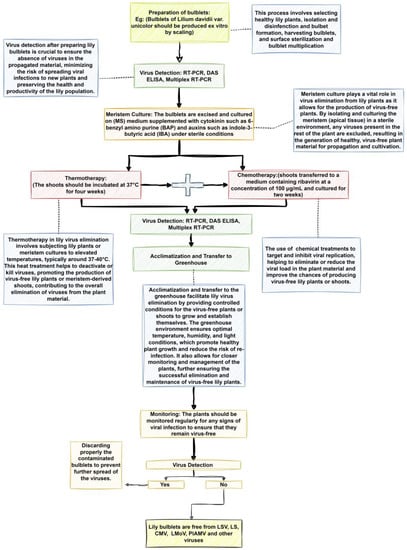
Figure 2.
Figure showing systematical steps for successful elimination of lily viruses (LSV, LS, and LMoV) using meristematic tips from bulblets produced ex vitro by scaling. It showcases the step-by-step process, including the collection of bulblets, production of virus-free meristematic tips, and their cultivation in a controlled environment. The figure emphasizes the importance of sterilization, nutrient media, and advanced diagnostic techniques to ensure the virus-free status of the meristematic tips. Ultimately, the figure highlights the potential for producing healthy lily plants that are free from targeted viruses (LSV, LS, and LMoV) by utilizing these systematic steps.
2.3. Meristem Tissue Culture for Lily Virus Infection Treatment
The use of meristem tissue culture is an important in vitro technique that consists of culturing on a nutrient medium a small (0.1–0.5 mm) piece of tissue removed from apical buds or nodal segments with an axillary bud [35,39]. Research has shown that the probability of obtaining plants without virus infection is inversely proportional to the size of the excised meristem [38]. Virus elimination by meristem tissue culture is challenged by the difficulty of tissue excision, the small size of the excised meristem, and the risk of contamination. These challenges can be overcome through careful handling of the plant material using a sharp, sterile scalpel or razor blade to excise the meristem. This requires skill and experience, as the meristem is a delicate tissue that can be easily damaged. Another solution is to use ex vitro scaling from tissue culture of bulb scales to obtain meristem tips. This method involves growing bulbs in tissue culture, then transferring the bulb scales to a new medium to induce the formation of meristem tips. This can be a more reliable and efficient method than direct meristem excision, as it allows for the production of multiple meristem tips from a single bulb.
In addition, it is highly recommended to use appropriate culture conditions to reduce the risk of contamination throughout the tissue culture process. This includes sterilizing all equipment and culture media and working in a laminar flow hood or other sterile environments [41]. Moreover, meristem tissue culture can be used alone or in combination with other techniques such as chemotherapy, thermotherapy, and cryotherapy to further reduce the risk of virus infection [36,42]. Studies have examined the elimination of lily mottle virus (LMoV) and lily symptomless virus (LSV) from lily plants through meristem tip culture. The results indicate that this technique is effective, cost-effective, and carries a minimal risk of contamination when utilizing meristem tips obtained from ex vitro scaling of bulb scales in tissue culture [36,39].
2.4. Thermotherapy for In Vitro Lily Virus Eradication
Studies have shown the relationship between high temperature and viral RNA silencing, and high temperatures are frequently associated with low virus content [6]. Therefore, thermotherapy is one of the conventional in vitro methods used for virus elimination in different plants [30]. For example, a recent study has explored virus-induced gene silencing (VIGS) and its potential for manipulating gene expression without transgenesis. While VIGS has been widely used in various plant species, its efficiency can be influenced by developmental, physiological, and environmental factors. The researchers investigated the impact of temperature on two forms of gene silencing, ViPTGS and ViTGS, using GFP as a reporter gene in N. benthamiana 16c plants infected with recombinant tobacco rattle viruses (TRVs). They found that high temperatures impair ViTGS due to a lack of secondary siRNA production, hindering the transmission of gene silencing to subsequent generations. Understanding the environmental influence on gene silencing mechanisms could aid in optimizing ViTGS for transgene-free crop improvement. Additionally, this research highlights the potential application of thermotherapy in eliminating viruses, as higher temperatures negatively impact ViTGS efficiency [43]. Thermotherapy can be used alone or in combination with other techniques such as meristem tissue culture or chemotherapy to eliminate lily viruses [38]. However, one major challenge that must be addressed is that the extended duration of thermotherapy at a high temperature leads to significant impacts on plant regeneration, growth, and overall survival, thus, an appropriate period must be further researched to establish the plant regeneration time during temperature-based therapy [37,38]. Elimination by meristem tip culture of LSV from Asiatic hybrids has been enhanced by the application of thermotherapy using bulbs obtained ex vitro [36,44]. However, a high-temperature treatment during ex vitro scaling has a negative effect on plant regeneration (Table 1). Studies have shown that the effectiveness of thermotherapy varies with plant genotype [8,39], which means that different lily varieties may require different temperatures or durations of treatment for effective virus elimination. Therefore, it is important to determine the appropriate conditions for each plant genotype to ensure successful virus eradication.
2.5. Chemotherapy for Eradicating Lily Viruses
Chemotherapy is an important in vitro culture method with a defined concentration of antiviral drugs used for the production of virus-free plants. It is used alone or in combination with other therapies [36]. Other chemical control methods involve the use of pesticides to manage the vectors or the virus itself. This can be carried out by using insecticides to regulate the vector insects or by using antiviral compounds to reduce the viral load in infected plants [45]. Although successful in some instances, chemotherapy possesses some challenges and limitations. For example, one of the major challenges in chemotherapy is finding the appropriate concentration of antiviral drugs that can effectively eliminate viruses without adversely affecting plant growth and development. Furthermore, some viruses may develop resistance to antiviral drugs, making chemotherapy ineffective in certain cases [46]. Another challenge is the phytotoxicity of the antiviral drugs used, which can have negative effects on plant regeneration and survival. These effects may include stunted growth, reduced photosynthesis, cellular damage, and even plant death [47]. For example, the application of 2,4-dioxohexahydro-1,3,5-triazine (DHT) or virazole was used to eliminate CMV and LSV infection in L. longiflorum, and the eradication effect of DHT on CMV and LSV is higher than that of virazole [35,39].
To overcome these challenges, researchers have experimented with different antiviral drugs and concentrations to determine the most effective and least toxic ones. They have also investigated the utilization of combinations of antiviral drugs to enhance the effectiveness of treatment and minimize the risk of viral resistance. Furthermore, integrating chemotherapy with other approaches like meristem tissue culture or thermotherapy can enhance virus elimination and shorten the duration of treatment [48].
Previous studies have shown that a combination of callus culture with chemotherapy was used to eliminate CMV from L. longiflorum [34]. When chemotherapy was combined with meristem tip culture, a noticeable decrease in the presence of lily symptomless virus (LSV) and/or tulip breaking virus (TBV) was observed in L. longiflorum ‘Arai’. However, the same technique was ineffective to eliminate the above viruses from the Asiatic hybrid ‘Enchantment’ (Table 1). According to reports, chemotherapy demonstrated higher efficacy when applied to bulbs obtained through in vitro scaling compared to ex vitro scaling [36].
2.6. Development of Combined In Vitro Methods for Lily Virus Elimination
The development of combined in vitro methods for lily virus elimination has shown promising results in effectively eradicating these viruses. However, this development is accompanied by certain challenges that need to be overcome. One of the main impediments is the optimization of each individual method to ensure their synergistic action rather than conflicting with one another in the elimination of lily viruses [36,49].
Researchers such as Masuda et al. (2011) have reported on the application of a combination of chemotherapy methods with callus culture for eliminating cucumber mosaic virus (CMV) in L. longiflorum [39]. Various techniques, including callus culture, meristem culture, and their combinations with thermotherapy or chemotherapy have been developed [34,35]. Notably, the combination of thermotherapy with meristem culture has been studied and found to be more efficient than using these methods individually [35,44]. This involves the excision and propagation of meristems, followed by a second round of meristematic tissue culture and subsequent application of thermotherapy to produce virus-free mother lily plants. In the case of lily symptomless virus (LSV) and lily mottle virus (LMoV) elimination from infected lily bulblets, the establishment of a successful complex therapy involved the combination of meristem tip culture with chemotherapy [39]. This approach effectively eliminated these viruses from the infected lily bulblets.
The development of combined in vitro methods for lily virus elimination requires the careful optimization of each individual method to ensure their synergistic action in targeting and eliminating specific viruses or virus combinations. This optimization process fine-tunes the parameters and conditions of each technique, maximizing their efficacy and minimizing potential drawbacks. Strict contamination control measures and a comprehensive understanding of the biology and behavior of each virus are also crucial aspects to consider. By harmoniously integrating different methods, researchers can achieve maximum success rates in producing virus-free lily plants, contributing to the overall health and resilience of lilies.

Table 1.
Different elimination techniques, specifically tailored to target various types of viruses and different plant genotypes, while also highlighting their respective treatment effects.
Table 1.
Different elimination techniques, specifically tailored to target various types of viruses and different plant genotypes, while also highlighting their respective treatment effects.
| Treatment Techniques 1 | Virus 2 | Hybrids 3 | Type of Treatment | Treatment Effects 4 | Elimination Rate (%) 5 | Reference |
|---|---|---|---|---|---|---|
| Meristem tip culture | LSV | Asiatic hybrid ‘Enchantment’ | -Meristem tip culture with 0 to 40 µM of ribavirin | Effective elimination of LSV | 74 56 | [38,41,46] |
| LSV | L. longiflorum ‘Fire King’ L. longiflorum ‘Ace’ L. longiflorum ‘Nellie White’ | Meristem tip culture with 0 to 40 µM of ribavirin | 16.7 25 | |||
| -LSV and/or TBV | L. longiflorum ‘Arai’ | -Meristem tip culture with 0 to 40 µM of ribavirin | Partial elimination of LSV and/or TBV | 35.46–61.4 | ||
| Thermotherapy | LSV | Oriental hybrid ‘Casa Blanca’ | 25 °C | Significant reduction in LSV with bulblet differentiation and multiplication | 80 | [41,46] |
| Oriental hybrid ‘Casa Blanca’ | 35 °C | Moderate reduction in LSV with limited bulblet differentiation | 60 | |||
| -L. longiflorum ‘Georgia’ | 25 °C | 78% bulblet differentiation and 2.3 bulblets per scale | 88 | |||
| 35 °C | 0% bulblet differentiation and 0 bulblets per scale | - | ||||
| Chemotherapy | LSV | L. longiflorum ‘Royal Respect’ | Ribavirin at 25 °C under ex vitro | Complete elimination of LSV with no phytotoxic effects | 100 | [44] |
| L. longiflorum ‘Georgia’ | Ribavirin at 35 °C | Phytotoxic effect Low number of bulblets | Low elimination rate | |||
| L. longiflorum ‘Arai’ | Ribavirin at 0.5 to 50 µM 0 to 40 µM 400 µM | Phytotoxic effects observed with a low number of bulblets No effect on the number of microbulbs, reduction in the number of bulblets | Varied elimination rate | |||
| L. longiflorum ‘Arai’ | Ribavirin between 100 and 300 µM at 25 °C | No phytotoxic effects observed | - |
1 Treatment techniques: the proposed protocols used for treating diseases. 2 Virus: intracellular pathogenic particles that infect other living organisms [39]. 3 Hybrids: the offspring of two or more individuals of different species or varieties [50]. 4 Treatment effects: the measures applied to evaluate the vegetative growth response to the treatment. 5 Elimination rate: the ratio at which the virus is removed from the lily plant.
2.7. Alternative Approaches for Managing Lily Viruses
To manage lily viruses, various approaches can be employed, including producing virus-free bulbs, eliminating infected plants and weeds, utilizing meristem culture, chemotherapy, and thermotherapy. Additionally, strategies such as genetic transformation, quarantine measures, cultural control methods, and biological control methods can be implemented to mitigate the impact of viruses. These approaches offer additional avenues for effectively managing lily viruses, complementing traditional control methods, and contributing to sustainable and integrated disease management strategies in lily cultivation.
2.7.1. Genetic Transformation
Genetic transformation is a promising tool that can be used to enhance the resistance of lily plants to viral infections. By introducing genes that encode resistance factors, it is possible to confer durable resistance to viruses. However, the genetic transformation of lilies is challenging due to their recalcitrance to regeneration, low transformation efficiency, and the lack of a comprehensive genome sequence. Despite these challenges, progress has been made, with successful transformation reported in several species. For example, Gladiolus plants transformed with a defective replicase and protein subgroup II gene were found to be resistant to cucumber mosaic virus (CMV). In another study Azadi et al. (2011) reported the successful transformation of Lilium cv Acapulco with a defective cucumber mosaic virus (CMV) replicase gene (CMV2-GDD) constructed using Agrobacterium tumefaciens that confers resistance to CMV. The results suggested that the two lines have increased resistance to CMV and that the CMV-GDD replicase gene is an effective construct that provides protection against CMV in Lilium [51]. In other studies, Agrobacterium-mediated transformation was used to generate improved lily virus resistance [52,53]. These studies demonstrate the potential of genetic transformation as a tool for improving the resistance of lily plants to viral infections, and further research is needed to optimize transformation protocols and identify new resistance genes.
2.7.2. Quarantine Measures
Quarantine measures involve isolating infected plants and preventing them from coming into contact with healthy plants. This can be carried out by physically separating infected plants, using physical barriers, or restricting the movement of plants and plant material from infected areas [54]. Other strategies include crop rotation, controlling weeds, destroying old crops, avoiding planting new crops on diseased plantings [55], and controlling vectors [56]. All these strategies help in eliminating and controlling lily viruses by reducing viral reservoirs and preventing transmission by carriers such as aphids.
2.7.3. Cultural Control Methods
Cultural control methods involve modifying the growing environment to make it less favorable for the virus or its vectors. This can be carried out by selecting disease-resistant varieties, planting at the appropriate time, crop rotation, improving soil fertility and drainage, and using appropriate irrigation and fertilization practices. More than any other form of control, cultural methods emphasize that the objective of horticulture is to produce fruitful, high-yielding crops rather than simply to control plant pathogens. These controls act largely in a preventive manner and are applied in advance of invasion [57]. Moreover, cultural control practices such as sanitation, proper plant hygiene, and the removal of infected plants (roguing) help prevent the buildup of virus reservoirs and decrease the chances of viral spread through vectors or mechanical means [58]. In the case of lily viruses, the use of cultural control methods has been limited due to the lack of known resistant varieties [36]. However, recent studies have shown that certain cultivars of lilies may exhibit some level of resistance to specific viruses [59,60]. Therefore, the selection of resistant varieties can be a potential solution for the cultural control of lily viruses.
Another challenge in the cultural control of lily viruses is the difficulty in early identification of infected plants. This is due to the lack of visible symptoms in some cultivars, which can result in the spread of the virus to other plants before detection [61]. To overcome this challenge, it is recommended to carry out routine virus testing, using methods such as ELISA and reverse transcription loop-mediated isothermal amplification (RT-LAMP) [29]. Improving soil fertility and drainage can also be a solution for cultural control of lily viruses. This can be achieved by ensuring proper crop rotation, use of organic fertilizers, and avoiding overwatering. By improving soil conditions, the plants can be healthier and less susceptible to viral infections [58]. While improving soil fertility alone may not directly eliminate lily viruses, it can contribute to a more robust plant defense system, reducing the severity and impact of viral infections [62,63].
2.7.4. Biological Control Methods
Biological control methods involve the use of natural enemies to control the virus or its vectors. This can be carried out by introducing predators or parasites that feed on the vectors or by using beneficial microorganisms that compete with the virus for resources or produce antiviral compounds [64]. The use of virus-free suckers, entomopathogens, predators, and parasitoids has been applied to control viruses [65,66]. In addition, predators, parasitoids, entomopathogenic fungi, and hyper-parasitoids are also used as good natural enemies to control aphids [67]. Biological control has been used to suppress virus infection in various plants. For example, the use of entomopathogenic fungi has been shown to be effective in controlling viruses from the genera Orthotospovirus, Ilarvirus, Alphacarmovirus, and Machlomovirus that affect a Western flower thrip, Frankliniella occidentalis (Pergande) [68]. These agents can be categorized into two groups: macrobials, which include predators and parasitoids, and microbials, such as fungal pathogens and entomopathogenic nematodes [69]. Notably, macrobials like anthocorid bugs (Orius spp.), green lacewing species, predatory phytoseiid mites, and predaceous laelapid mites have shown significant efficacy in combating thrips infestation at different life stages [70,71,72,73,74,75]. These biological control agents specifically target first instar thrips on foliage and thrips pupae in soil, offering promising potential for managing plant viruses and their vectors [76].
3. Development of Lily Virus Detection Methods
For surveillance of lily viruses, reproducible, scalable, reliable, precise, and rapid detection methods were developed (Table 2). Simplicity, turnaround time, specificity, sensitivity, and cost-efficiency are also key criteria for choosing the best method for plant virus detection [19]. RT-PCR, real-time RT-PCR, and ELISA have been developed to detect lily viruses in infected leaves and bulbs [19,77,78].
3.1. Serological-Based Methods
The lily bulb is the most common explant used when manipulating plants both for detection and tissue culture [34]. Virus detection using serological methods has been widely reported and embraced by researchers for their practicality and reliability [79]. Techniques such as enzyme-linked immunosorbent assay (ELISA) and immunoblotting (Western blot) are commonly employed for serological detection of lily viruses. Even though serological techniques are not specific and sensitive compared to other molecular biology techniques [77], they compensate for it in several key ways. Firstly, they are not expensive and do not require qualified trained experts for molecular detection and can also be used during routine activities of laboratory analysis, especially in laboratories with limited resources [80]. Many researchers have reported the use of monoclonal and polyclonal antibodies for lily virus detection using immunoserological tests [81,82]. Immunoassays offer robustness and reduced costs.
3.1.1. Enzyme-Linked Immunosorbent Assay (ELISA)
The development of simple, economical, and routine methods has been a prerequisite to managing the infection of lilies by viruses. ELISA was found to be the most valuable and rapid method that does not need qualified experts to carry out testing [77,80,82]. ELISA is used in agriculture to detect plant viruses such as PVX, PVY, and PLRV. The method uses protein A in two applications to sandwich antibody–antigen–antibody layers. The specificity of the test can preclude detection of closely related strains of the same virus [83]. ELISA is the preferred technique for indexing and controlling plant viruses due to its ability to efficiently test a large number of samples. In order to attain a high sensitivity, it is crucial to develop and test a qualitative antibody of high quality [28]. To optimize the specificity of ELISA for lily virus detection, researchers produced two recombinant proteins that were specific to LMoV and LSV. These recombinant proteins were designed to target and bind specifically to the viral coat protein (CP) of each virus. By using these recombinant proteins in the ELISA, researchers were able to enhance the specificity of the technique, enabling accurate and reliable detection of LMoV and LSV in lily plants [28,84]. Ha et al. (2012) reported that, using ID-ELISA, lily virus can be detected in the field without contamination. However, it is used to detect a single infection, which is a limitation as lily plants are often infected by more than one virus [2]. One of the major challenges of using ELISA is the need for a high concentration of virus, which indicates its low sensitivity [28]. To overcome this limitation, the optimization of the technique through the utilization of high-quality antibodies and virus-specific recombinant proteins can significantly enhance the sensitivity and specificity of the ELISA. Consequently, ELISA might emerge as a valuable method in comparison to other molecular techniques. [82]. ELISA can be used for routine virus detection in lilies and provide accurate and sensitive detection of viruses in lilies.
3.1.2. Immunochromatographic Tests
LSV and LMoV are the most prevalent viruses in lily plants [28]. Immunochromatographic tests (ICTs) have exhibited substantial potential for field diagnosis of lily virus infections. This method is favored due to its independence from sophisticated equipment and specialized expertise, making it an accessible approach for virus detection [77,85,86]. An immunochromatographic strip (ICS) test has been developed as a diagnostic tool for the outbreak of viral diseases for common plant viruses, such as lily viruses, including LSV and LMoV (Table 2). The method exhibits strong agreement with the reference test of quadruplex RT-PCR when detecting LMoV and LSV at low concentrations. Additionally, compared to other techniques, it demonstrates the highest sensitivity and specificity [19]. One challenge is the cross-reactivity of the antibodies used in the test, which may lead to false positive or false negative results [19]. To overcome this problem, it is important to use antibodies that are highly specific for the target molecule. This can be achieved by using monoclonal antibodies that are produced by a single clone of cells and are highly specific for the target molecule. Another way to overcome this problem is to use multiple antibodies that recognize different epitopes on the target molecule. This can increase the specificity of the test and reduce the risk of cross-reactivity [87] The turnaround time for sample analysis of 10 min has defined ICSs as rapid in comparison to other techniques and they have been adopted to be used for the detection of LMoV and LSV under field conditions [19]. IC-RT-PCR methods have been applied to detect many other plant viruses [28]. For example, in a recent study, an IC-RT-PCR assay using newly prepared polyclonal antibodies was developed. When co-infected leaf sap (PV-0155) was tested with IC-RT-PCR, it was observed that a fragment of the expected size (904 bp) was successfully amplified. To assess the sensitivity of IC-RT-PCR for CMV, researchers performed IC-RT-PCR on 10-fold serial dilutions of CMV-infected lily extracts (PV-0188). They obtained the expected amplicon size (904 bp) from test samples diluted up to 10−6, but no amplification product beyond a 10−6 dilution of CMV-infected leaf samples or from the healthy control was observed [82]. The results suggest that the IC-RT-PCR technique exhibits greater sensitivity than ELISA, even when the lily virus is present in low concentrations.
Another challenge is the sensitivity of the test. While the sensitivity of ICTs is generally high, it may not be sufficient to detect low levels of viral infections. For example, the sensitivity of the immunochromatographic test for lily viruses was evaluated by testing a series of 10-fold dilutions of purified LSV solution (at 6.0 × 10−1 mg/mL), which was analyzed as virus antigen utilizing Western blotting using the IgG2 protein at a concentration of 2.0 × 10−2 mg/mL. When serial dilutions of purified LSV were tested, the LSV detection limit of the ICS test was 6.0 × 10−8 mg/mL, which was the same as the quadruplex RT-PCR assay. Relative to quadruplex RT-PCR, the specificity and sensitivity of the ICS was 98.6% and 100%, respectively, for field leaf samples. To improve the sensitivity, the use of signal amplification strategies such as gold nanoparticles or enzyme-conjugated antibodies may be considered [19]. Furthermore, the stability and storage of the test strips can also affect the accuracy and reliability of the results [88]. The optimal storage conditions for ICSs may vary depending on the manufacturer and type of test strip. However, it is generally recommended to store the test strips in a cool and dry place away from direct sunlight. The recommended ambient storage conditions are usually below 25 °C and below 60% RH [87].
3.2. Molecular Techniques
Molecular techniques have revolutionized the detection of lily viruses, surpassing the sensitivity and specificity of immunoserological methods [32,78]. In particular, the RT-PCR technique has emerged as a reliable tool, surpassing the limitations of ELISA when applied at various stages of plant growth and development [22]. By studying the genomes of lily viruses, researchers have gained valuable insights and developed molecular biology techniques that greatly enhance the detection of these viruses [84,89]. With knowledge of the virus genome, one can use RT-PCR to detect viruses with great sensitivity and specificity, contributing to improved monitoring and management of lily virus infections [84,89].
3.2.1. Reverse Transcription Polymerase Chain Reaction-Based System for the Simultaneous Detection of Multiple Lily-Infecting Viruses
The co-infection of lily plants by more than one virus resulted in severe expressions of viral symptoms [81,90]. In many lily plantations, CMV, LMoV, and LSV have been detected as mixed infections [2,22]. Many techniques, including both immunoserological and molecular biology-based methods, have been developed to detect single viruses such as LSV and LMoV and it has been found that the results vary from one hybrid to another [22]. A multiplex RT-PCR assay to detect lily plant viruses including CMV, LMoV, and LSV was developed [22,91]. However, the use of agarose gel for target detection in this method has been identified as a limitation in advanced research. It is crucial to carefully design detection primer pairs for simultaneous amplification of multiple targets, ensuring distinctive discrimination of multiple bands [78,92]. Chun et al. (2007) reported a modified PCR method that utilizes a dual priming oligonucleotide (DPO) system, which exhibits structural and functional differences compared to the conventional primer system to manage a mixed infection of lily plants by a virus. This modified method incorporates a poly(I) linker between two segments of primer sequences [22]. The simultaneous detection of three viral genes, of CMV, LMoV, and LSV, and the 18 S ribosomal RNA (rRNA) internal control in one reaction was found to be the most effective and valuable compared to other conventional methods.
3.2.2. Detection Using Luminex-Based Liquid Bead Array
A microsphere-based flow cytometric system was developed by Luminex and validated for the efficacy of detecting multiple viral genes simultaneously in lily plant tissues [93]. It is a bead-based array detection comparable to the methods developed for ELISA that can allow the detection of a large number of viruses in a single test [78]. It is among the recently developed techniques and found to be economic, sensitive, and specific compared to multiplex real-time RT-PCR.
3.2.3. Triplex IC-RT-PCR for the Simultaneous Detection of Three Lily Viruses and Hybrid Diagnostic Techniques
Multiple-virus infections in lilies have been observed in various regions and their prevalence is steadily rising, as indicated by numerous studies [2,91,94]. Consequently, reliable and precise detection systems that can simultaneously detect a mixed infection are needed. In addition, the sensitivity of virus detection by the PCR technique is limited by polysaccharose, polyphenol, and other plant inhibitors from RNA extraction [81,95]. As a result, a qualitative, semi-automated PCR-based method, immunocapture RT-PCR (IC-RT-PCR), was developed to simultaneously detect several targets in the same sample [78,96]. The method is characterized by its rapidity, sensitivity, and specificity, enabling simultaneous detection of LSV, CMV, and LMoV [97]. In a recent study that focused on the development of a triplex immunocapture reverse transcription polymerase chain reaction (IC-RT-PCR) assay for the simultaneous detection of these three viruses, the triplex IC-RT-PCR assay demonstrated high specificity (100%) and sensitivity (ranging from 81.4% to 99.4%) for detecting LSV, CMV, and LMoV in field samples, respectively. The agreement between the triplex assay and a quadruplex RT-PCR assay was high, with κ values of 0.844 to 0.984. These results obtained from detecting these three highly prevalent viruses affirm the reliability and accuracy of the technique [81].
3.2.4. Reverse Transcription Loop-Mediated Isothermal Amplification
LSV and its co-infections with CMV or LMoV have been reported in various endemic regions worldwide. Some of the regions where these viruses have been identified include: the Netherlands, China, Japan, and the United States [21,27,98]. Mixed infection with these viruses leads to severe symptoms like smaller flowers, lower bulb yield, and growth reduction. This highlights the need for a sophisticated detection technique to effectively identify and monitor these infections.
Furthermore, the challenge of early-stage virus identification has posed difficulties for scientists. This difficulty, combined with the failure to remove infected plants promptly, has resulted in significant outbreaks of infection [28]. Therefore, the development of a rapid, low-cost, and simple alternative diagnostic method has been a prerequisite to controlling, surveying, and preventing this type of infection [61]. The need for a rapid, accurate, field-based detection technology has been solved by reverse transcription loop-mediated isothermal amplification (RT-LAMP). The same technique has also been applied to other crops such as potato to detect potato viruses [99]. He et al. (2016) found that RT-LAMP detects not only the virus in a symptomatic plant but also in symptomless infected plants. RT-LAMP is a preferred method for LSV detection during field activities due to its simplicity, specificity, rapidity, and the advantage of a lower detection limit. Although it has a detection limit that is 10 times higher compared to RT-PCR, the technique still offers a significant benefit with a turnaround reaction time of less than 30 min. This makes it highly practical and efficient for on-site detection, ensuring timely identification of the presence of LSV even at lower concentrations [29,61]. The technique is conducted in a single tube at a constant temperature of 60–70 °C, utilizing two primer pairs targeting the conserved sequence of LSV coat protein [61]. To detect amplified amplicons, SYBR Green I, ethidium bromide, or calcein can be added to the amplified reaction mixture [78].
3.2.5. Real-Time RT-PCR
Real-time PCR techniques have been developed specifically for detecting plant viruses. These techniques serve a dual purpose: identifying viral infections and monitoring virus quantities in collected samples [25,100,101]. Their high sensitivity and specificity have made them valuable in the diagnosis of lily viruses [89]. Two common types of real-time PCR used in plant virus detection are multiplex RT-PCR and TaqMan real-time RT-PCR [32,90]. Studies have demonstrated that these techniques are effective in detecting CMV, LSV, LMoV, TVX, and TVR in lily samples, surpassing the capabilities of conventional RT-PCR and ELISA in terms of sensitivity and specificity [22,48].
Despite its benefits, real-time RT-PCR for lily virus detection presents several challenges. Firstly, it requires highly skilled personnel and costly equipment for the detection process [91]. Secondly, the technique is susceptible to interference from inhibitors found in plant tissue [94]. Finally, achieving accurate detection necessitates the development of highly specific and sensitive primers and probes, a task that can be time-consuming and expensive [48].
To overcome the challenges of real-time RT-PCR in lily virus detection, several solutions have been proposed. Simplified protocols for RNA extraction and purification have been developed, facilitating a more accessible and efficient process. Some examples of simplified RNA extraction methods include the use of silica-based spin columns, magnetic bead-based extraction kits, and rapid lysis buffer-based protocols. These methods often involve simplified sample preparation steps, such as direct cell lysis or quick homogenization, to streamline the extraction process. Additionally, commercial RNA extraction kits with pre-packaged reagents and optimized protocols are available, allowing for convenient and efficient RNA isolation [91]. Internal controls are utilized to monitor and ensure the accuracy of results by detecting the presence of inhibitors in the sample [32]. Additionally, the use of multiplex RT-PCR and TaqMan real-time RT-PCR enhances the technique’s efficiency by enabling the simultaneous detection of multiple viruses in a single reaction [94,102]. These solutions simplify the detection process, reducing the reliance on specialized personnel and expensive equipment, while also minimizing the time and cost associated with primer and probe development.
3.3. Generation and Purification of Recombinant Coat Proteins
The choice of a high-quality antibody was found to be very important for detecting lily virus using immunoserological methods such as ELISA [82]. The virus-specific DNA recombinant technology was used for the production of purified coated proteins that capture viral antigens from infected samples; the method is based on a hybrid diagnostic technique [96]. A purified coat protein can capture a virus antigen through specific binding interactions. When the recombinant coat proteins are produced using virus-specific DNA recombinant technology, they possess the ability to bind to viral antigens present in infected samples. This binding occurs due to the specific recognition between the coat protein and the viral antigen, allowing for the capture and detection of the virus. However, it should be noted that the purification process of coat proteins from lily symptomless virus (LSV) and lily mottle virus (LMoV) for the generation of antiserum is a challenging task [28]. In capture (indirect coating) ELISA, antigen-specific antibody is adsorbed onto the plastic, which in turn binds and immobilizes the antigen upon incubation with the antigen sample [103]. To increase efficiency, the expression of recombinant proteins in prokaryotes has been utilized to produce polyclonal antibodies (PAbs) as an immunogen. This is due to the stability, high productivity, and easy protein purification [28,82]. Consequently, the ID-ELISA method and IC-RT-PCR assays have been developed for efficient lily virus detection [104]. These methods were recommended for further control, surveys, and also to prevent lily infection by most of the viruses that occur in Lanzhou/Gansu Province and NingXia, the most important lily production areas in China [21,98].

Table 2.
Overview of different techniques used for the detection of lily viruses.
Table 2.
Overview of different techniques used for the detection of lily viruses.
| Techniques | Principle | Detected Virus | Features | Samples | Reference |
|---|---|---|---|---|---|
| ELISA | Used to detect single virus infection | LSV and LMoV |
| Large-scale samples | [28] |
| ID-ELISA | Separately detect LSV and LMoV | LSV, LMoV, CMV, and PlAMV |
| Large-scale bulbs Field activities | [23,84] |
| ICS | Double-antibody Sandwich format and employs two distinct anti-viral Polyclonal antibodies | LMoV and LSV -Triplex IC-RT-PCR for simultaneous detection of LMoV, CMV, and LSV |
| Lily leaf samples | [28,81] |
| Real-time PCR | Determine the number of viruses within tested samples | CMV, LSV, and other virus | 1000 times more sensitive than conventional RT-PCR, need for expert personnel and expensive equipment, no risk of contamination | More efficient in leaves than in bulbs | [90] |
| IC-RT-PCR | RNA extraction carried out in a single tube | LMoV Simultaneously detecting LSV, CMV, and LMoV |
| Leaf samples | [28] |
| Multiplex RT-PCR | The simultaneous detection of multiple viruses in a single reaction, amplifying multiple target sequences using multiple primer pairs in a single reaction tube | LSV LMoV CMV PlAMV |
| Fresh leaves | [94] |
| Luminex-based liquid bead array | Specific oligonucleotide probes labeled with an amino group and coupled to fluorescence-encoded microspheres | LSV LMoV CMV |
|
| [82] |
| Reverse transcriptionloop-mediated isothermal amplification | Within a closed tube at a constant temperature with specifically designed loop primers and results observed using SYBR Green I | LSV, LMoV |
| Lily bulbs Field detection | [61,105] |
| Triplex IC-RT-PCR | LSV-CP, CMV-CP, and LMoV-CPI200 use proteins against viral antigens | LSV, CMV, and LMoV |
| Leaves | [81] |
3.4. Emerging Assays for Lily Virus Detection: Expanding the Toolbox for Portable, Low-Cost, and Rapid On-Site Testing
Several other new, portable, low-cost, rapid, and on-site assays could be developed for lily viruses besides the ones mentioned previously. These are:
- CRISPR-based assays: These assays use the CRISPR/Cas system to detect and amplify viral RNA/DNA in a sample. They are highly specific and can provide results in less than an hour [106].
- DNA-based biosensors: These are devices that detect specific molecules (such as viral proteins or nucleic acids) and convert them into a measurable signal. They can be designed to be portable, rapid, and low-cost, making them ideal for on-site detection [107].
- Digital PCR: Digital PCR is a highly sensitive nucleic acid quantification technique that partitions a sample into thousands of individual reactions. It can detect low levels of viral RNA/DNA in a sample and is highly precise. This method has already shown a positive outcome. According to [99], an experiment was conducted using leaf samples from lily plants, specifically the oriental hybrid ‘Siberia’ cultivar, which had low viral loads. The results showed that the reverse transcription droplet digital polymerase chain reaction (RT-ddPCR) outperformed RT-qPCR in diagnosing PlAMV infection in lily plants. Based on these findings, it was recommended to utilize RT-ddPCR, especially in plant quarantine inspection and PlAMV-free certification programs, for the accurate detection of PlAMV infection in lily plants.
- Next-generation sequencing: This is a high-throughput sequencing technique that can detect and identify viral sequences in a sample. It can be used to identify new and emerging viral strains and to monitor viral diversity within a population [108].
4. Conclusions and Future Perspectives
Viral diseases are affecting both the qualitative and quantitative aspects of lily production. These diseases continue to spread in lily-producing countries worldwide. Genomic characterization and sequencing of different types of lily viruses have been carried out, especially for the most prevalent viruses: LSV, CMV, PlAMV, and LMoV. The current objective in many studies is to find a reliable and efficient method for producing certified lily seeds from in vitro culture that is delivered to lily growers for commercial production. On one hand, designing a genetic engineering method that can produce lily viral resistance might be a sustainable solution. However, on the other hand, the use of genetically modified organisms is still debatable according to international bioethics organizations.
Lily virus eradication using in vitro culture methods is currently the common method used to increase lily production, as lily cultivars are vegetatively propagated. Plant tissue culture is the most common technique for the production of quality planting material, especially using meristem culture, which is a conventional and well-known method for the production of virus-free plants. However, the extraction and culture of a small amount of meristem tissue is problematic and can result in non-desirable soma clonal variations. The use of other techniques such as chemotherapy, thermotherapy, and their improved combined methods, particularly with meristem culture, is much more effective for the enhancement of lily virus elimination.
Predicting future technologies that are specifically suitable for each lily virus and plant species is challenging. However, advancements in molecular biology techniques are expected to lead to the development of new assays in the future. These assays are anticipated to offer higher levels of sensitivity, specificity, and speed in detecting and studying lily viruses and their interactions with different plant species. These technologies may be tailored to detect specific viral strains, allowing for more targeted and efficient detection and control strategies. In addition, these methods can be complemented with more serological techniques that have been developed to enhance lily virus control. To detect eradication efficiency and in terms of reliability and accuracy, ELISA was found to be useful in terms of high-volume samples; however, due to its low sensitivity and risk of contamination, ICSs and IC-RT-PCR were developed as rapid, cost-effective, sensitive, and specific techniques for detection of the most prevalent viruses. Various efforts have been made in the field of virus detection. However, it is important to note that the development of the recombinase polymerase amplification (RPA) assay for lily virus detection is still pending, despite its numerous advantages such as high convenience in sample preparation, portability, and applicability at temperatures ranging from 37 to 42 °C. The authors recommend placing further emphasis on the development of the RPA assay as a promising approach for the detection of lily viruses [109]. In addition, several methods for virus detection discussed in this review can be applied to detect more than one virus at a time which raises a question of specificity and demonstrates the urgency to address this problem by developing a more suitable lily virus genotype-specific protocol which works well for a single virus such as reverse transcription loop-mediated isothermal amplification and IC-RT-PCR which can only detect LSV and LMoV, respectively [61,80]. The authors acknowledge that while the recommended advanced techniques for virus detection and elimination may primarily be adopted in modern laboratories, even non-modern labs should strive to find alternative methods and optimize the currently used protocols. This will facilitate a wide accurate diagnosis of these viruses by increasing simplicity, speed, specificity, sensitivity, robustness, and cost-efficiency, which are the major criteria that should be focused on while selecting the pre-eminent detection methods. By adopting the suggested approaches in this review, the accurate detection and control of lily viruses can be achieved, contributing to the improvement of lily production worldwide.
Author Contributions
H.G.: Conceptualization, resources, supervision, and revision of the manuscript, L.D., P.L. and C.I.: Writing—original draft preparation, editing, preparation of graphs. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Foundation of China (Grant Nos. 31860397 and 31360296).
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Fatihah, H.N.N.; Moñino López, D.; van Arkel, G.; Schaart, J.G.; Visser, R.G.F.; Krens, F.A. The ROSEA1 and DELILA Transcription Factors Control Anthocyanin Biosynthesis in Nicotiana benthamiana and Lilium Flowers. Sci. Hortic. 2019, 243, 327–337. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Y.; Xie, Z.; Yang, G.; Guo, Z.; Wang, L. The Occurrence and Distribution of Viruses Infecting Lanzhou Lily in Northwest, China. Crop Prot. 2018, 110, 73–76. [Google Scholar] [CrossRef]
- Bakhshaie, M.; Khosravi, S.; Azadi, P.; Bagheri, H.; van Tuyl, J.M. Biotechnological Advances in Lilium. Plant Cell Rep. 2016, 35, 1799–1826. [Google Scholar] [CrossRef] [PubMed]
- LingFei, X.; FengWang, M.; Dong, L. Plant Regeneration from in Vitro Cultured Leaves of Lanzhou Lily (Lilium davidii var. unicolor). Sci. Hortic. 2009, 119, 458–461. [Google Scholar] [CrossRef]
- Prudente, D.d.O.; Paiva, R.; de Souza, L.B.; Paiva, P.D.d.O. Cryotherapy as a Technique for Virus Elimination in Ornamental Species. Plant Cell Cult. Microprogation 2018, 13, 29–33. [Google Scholar]
- Wang, F.; Wang, W.; Niu, X.; Huang, Y.; Zhang, J. Isolation and Structural Characterization of a Second Polysaccharide from Bulbs of Lanzhou Lily. Appl. Biochem. Biotechnol. 2018, 186, 535–546. [Google Scholar] [CrossRef]
- Wu, Y.; Ma, Y.; Li, Y.; Zhang, L.; Xia, Y. Plantlet Regeneration from Primary Callus Cultures of Lilium brownii F.E.Br. Ex Miellez var. giganteum G. Y. Li & Z. H. Chen, a Rare Bulbous Germplasm. Vitr. Cell. Dev. Biol.—Plant 2019, 55, 44–59. [Google Scholar] [CrossRef]
- Wang, M.-R.; Cui, Z.-H.; Li, J.-W.; Hao, X.-Y.; Zhao, L.; Wang, Q.-C. In Vitro Thermotherapy-Based Methods for Plant Virus Eradication. Plant Methods 2018, 14, 87. [Google Scholar] [CrossRef]
- Bos, F. Lilium bulbiferum subsp. croceum in the Netherlands and Nothern Germany. Bentham-Moxon Trust 1993, 10, 190–197. [Google Scholar]
- Grassotti, A.; Nesi, B.; Lazzereschi, S.; Cacini, S.; Pacifici, S. Breeding Asiatic Hybrid Lilies: An Italian Experience. Acta Hort Hort 2011, 900, 237–242. [Google Scholar] [CrossRef]
- Van Tuyl, J.M.; Van Holsteijn, H.C.M. Lily Breeding Research in the Netherlands. Acta Hortic. 1996, 414, 35–45. [Google Scholar] [CrossRef]
- Roh, M.S.; Plants, N. Controlled Flowering in the Genus Lilium—Review of the Past Achievements and the Future Direction of Research. Acta Hort 2011, 900, 189–204. [Google Scholar] [CrossRef]
- Suh, J.K.; Roh, M.S. Scientia Horticulturae New Technique for Cut Flower Production from Bulbils of the Asiatic Hybrid Lily (Lilium × Elegans Thunb.). Sci. Hortic. 2014, 165, 374–383. [Google Scholar] [CrossRef]
- Lawson, R.H. Detection, Diagnosis and Control of Lily Diseases. Acta Hortic. 2011, 900, 313–324. [Google Scholar] [CrossRef]
- Bi, W.-L.; Chen, L.; Guo, L.; Pan, C.; Yin, Z.-F.; Wang, Q.-C. Plant Regeneration via Embryo-like Structures: Histological Observations and Genetic Stability in Regenerants of Lilium spp. J. Hortic. Sci. Biotechnol. 2015, 90, 626–634. [Google Scholar] [CrossRef]
- Shang, Q.H.; Zhao, X.; Li, Y.Y.; Xie, Z.K.; Wang, R.Y. First Report of Fusarium tricinctum Causing Stem and Root Rot on Lanzhou Lily (Lilium davidii var. unicolor) in China. Plant Dis. 2014, 98, 999. [Google Scholar] [CrossRef]
- Jo, Y.; Cho, W.K. RNA Viromes of the Oriental Hybrid Lily Cultivar “Sorbonne”. BMC Genom. 2018, 19, 748. [Google Scholar] [CrossRef]
- Asjes, C.J. Control of Aphid-Borne Lily Symptomless Tirus and Lily Mottle Tirus in Lilium in the Netherlands. Virus Res. 2000, 71, 23–32. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Y.; Meng, J.; Xie, Z.; Wang, R.; Kutcher, H.R.; Guo, Z. Development of an Immunochromatographic Strip Test for Rapid Detection of Lily Symptomless Virus. J. Virol. Methods 2015, 220, 13–17. [Google Scholar] [CrossRef]
- Fan, X.; Du, Y.; Cai, Y.; Zhang, Y.; Zhao, X.; Liang, J.; Yang, D.; Zhang, Q.; Zhang, X.; Zhang, W.; et al. Rapid and Sensitive Detection of Cucumber Mosaic Virus by Reverse Transcription Loop-Mediated Isothermal Amplification. Acta Biochim. Biophys. Sin. 2018, 51, 223–226. [Google Scholar] [CrossRef]
- Cardoso, E.; Bodi, D.A.; Harakava, R. Occurrence and Molecular Analysis of Quarantine Virus in Lily Cultivation Areas in Brazil. Pesqui. Agropecuária Bras. 2016, 51, 615–622. [Google Scholar] [CrossRef]
- Kwon, J.Y.; Ryu, K.H.; Choi, S.H. Reverse Transcription Polymerase Chain Reaction-Based System for Simultaneous Detection of Multiple Lily-Infecting Viruses. Plant Pathol. J. 2013, 29, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J. Lily Symptomless Virus (Carlavirus LSV) on Martagon Lily (Lilium martagon). Available online: https://www.forestryimages.org/browse/detail.cfm?imgnum=5503429 (accessed on 27 March 2020).
- Aravintharaj, R.; Balaji, C.G.; Nagendran, K.; Priyanka, R.; Karthikeyan, G. First Report of Lily Mottle Virus on Lily (Lilium sp.) in Southern India. Virus Dis. 2017, 28, 222–223. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Cheng, J.; Zhang, J.; Teixeira da Silva, J.A.; Wang, C.; Sun, H. Validation of Reference Genes for Accurate Normalization of Gene Expression in Lilium davidii var. unicolor for Real Time Quantitative PCR. PLoS ONE 2015, 10, e0141323. [Google Scholar] [CrossRef]
- Chen, M.; Zhang, J.; Zhou, Y.; Li, S.; Fan, X.; Yang, L.; Guan, Y.; Zhang, Y. Transcriptome Analysis of Lilium Oriental × Trumpet Hybrid Roots Reveals Auxin-Related Genes and Stress-Related Genes Involved in Picloram-Induced Somatic Embryogenesis Induction. J. Hortic. Sci. Biotechnol. 2019, 94, 317–330. [Google Scholar] [CrossRef]
- Sharma, A.; Mahinghara, B.K.K.; Singh, A.K.K.; Kulshrestha, S.; Raikhy, G.; Singh, L.; Verma, N.; Hallan, V.; Ram, R.; Zaidi, A.A.A. Identification, Detection and Frequency of Lily Viruses in Northern India. Sci. Hortic. 2005, 106, 213–227. [Google Scholar] [CrossRef]
- Yoo, H.N.; Jung, Y.T. Expression of Lily Mottle Virus Coat Protein and Preparation of Igy Antibody against the Recombinant Coat Protein. Korean J. Hortic. Sci. Technol. 2014, 32, 544–549. [Google Scholar] [CrossRef]
- Zhao, X.; Du, Y.; Zhang, Y.; Liang, J.; Cai, Y.; Xu, Y.; Fan, X.; Wu, W.; Zhang, Q.; Zhang, X.; et al. Effective Detection of Lily Symptomless Virus Using the Reverse Transcription Loop–Mediated Isothermal Amplification Method. Australas. Plant Pathol. 2019, 48, 373–374. [Google Scholar] [CrossRef]
- Panattoni, A.; Luvisi, A.; Triolo, E. Review. Elimination of Viruses in Plants: Twenty Years of Progress. Span. J. Agric. Res. 2013, 11, 173–188. [Google Scholar] [CrossRef]
- Montero-astúa, M. Detection of Plantago Asiatica Mosaic Virus in Lily Hybrid Plants (Lilium spp.) in Costa Rica Grown from Imported Bulbs. Australas. Plant Dis. Notes 2017, 12, 57. [Google Scholar] [CrossRef]
- Gong, H.; Igiraneza, C.; Dusengemungu, L. Major In Vitro Techniques for Potato Virus Elimination and Post Eradication Detection Methods. A Review. Am. J. Potato Res. 2019, 96, 379–389. [Google Scholar] [CrossRef]
- Bhojwani, S.S.; Dantu, P.K. Production of Virus-Free Plants. In Plant Tissue Culture: An Introductory Text; Bhojwani, S.S., Dantu, P.K., Eds.; Springer: New Delhi, India, 2013; pp. 227–243. ISBN 978-81-322-1026-9. [Google Scholar]
- Dapküniene, S.; Indrißiünaite, G.; Juodkaite, R.; Navalinskiene, M.; Samuitiene, M. Tissue Culture for Elimination of Lily Viruses Depending on Explant Type. Acta Univ. Latv. Biol. 2004, 676, 163–166. [Google Scholar]
- Milosevic, S.; Cingel, A.; Jevremovic, S.; Stankovic, I.; Bulajic, A.; Krstic, B.; Subotic, A.; Milošević, S.; Cingel, A.; Jevremović, S.; et al. Virus Elimination from Ornamental Plants Using in Vitro Culture Techniques. Pestic. Fitomedicina 2012, 27, 203–211. [Google Scholar] [CrossRef]
- Chinestra, S.C.; Curvetto, N.R.; Marinangeli, P.A. Production of Virus-Free Plants of Lilium spp. from Bulbs Obtained in Vitro and Ex Vitro. Sci. Hortic. 2015, 194, 304–312. [Google Scholar] [CrossRef]
- Moses, W.; Rogers, K.; Mildred, O.-S. Effect of Thermotherapy Duration, Virus Type and Cultivar Interactions on Elimination of Potato Viruses X and S in Infected Seed Stocks. Afr. J. Plant Sci. 2017, 11, 61–70. [Google Scholar] [CrossRef]
- Nesi, B.; Trinchello, D.; Lazzereschi, S.; Grassotti, A.; Ruffoni, B. Production of Lily Symptomless Virus-Free Plants by Shoot Meristem Tip Culture and in Vitro Thermotherapy. HortScience 2009, 44, 217–219. [Google Scholar] [CrossRef]
- Masuda, J.I.; Thien, N.Q.; Thi, N.; Hai, L.; Hiramatsu, M.; Takeshita, M.; Kim, J.-H.H.; Nakamura, M.; Iwai, H.; Okubo, H.; et al. Production of Virus-Free Bulblets by Meristematic Tip Culture with Antiviral Chemical in Lilium brownii var. colchesteri. J. Jpn. Soc. Hortic. Sci. 2011, 80, 469–474. [Google Scholar] [CrossRef]
- Antonova, O.Y.; Apalikova, O.V.; Ukhatova, Y.V.; Krylova, E.A.; Shuvalov, O.Y.; Shuvalova, A.R.; Gavrilenko, T.A. Eradication of viruses in microplants of three cultivated potato species (Solanum tuberosum L., S. phureja Juz. & Buk., S. stenotomum Juz. & Buk.) using combined thermo-chemotherapy method. Sel’skokhozyaistvennaya Biol. 2017, 52, 95–104. [Google Scholar] [CrossRef]
- Retheesh, S.T.; Bhat, A.I. Simultaneous Elimination of Cucumber Mosaic Virus and Cymbidium Mosaic Virus Infecting Vanilla Planifolia through Meristem Culture. Crop Prot. 2010, 29, 1214–1217. [Google Scholar] [CrossRef]
- Kaur, C.; Raj, R.; Kumar, S.; Lalit, D.K.P.; Puneet, A. Elimination of Bean Yellow Mosaic Virus from Infected Cormels of Three Cultivars of Gladiolus Using Thermo-, Electro- and Chemotherapy. 3 Biotech 2019, 9, 154. [Google Scholar] [CrossRef]
- Fei, Y.; Pyott, D.E.; Molnar, A. Temperature Modulates Virus-Induced Transcriptional Gene Silencing via Secondary Small RNAs. New Phytol. 2021, 232, 356–371. [Google Scholar] [CrossRef] [PubMed]
- Nesi, B.; Lazzereschi, S.; Pecchioli, S.; Grassotti, A.; Ruffoni, B. Virus Detection and Propagation of Virus-Free Bulbs from Selected Progenies of Pollenless Lilies. In Proceedings of the Acta Horticulturae; International Society for Horticultural Science (ISHS), Leuven, Belgium, 1 July 2011; Volume 8, pp. 325–331. [Google Scholar]
- Conijna, C.G.M. Developments in the Control of Lily Diseases. Acta Hortic. 2014, 1027, 213–230. [Google Scholar] [CrossRef]
- Bachar, S.C.; Mazumder, K.; Bachar, R.; Aktar, A.; Al Mahtab, M. A Review of Medicinal Plants with Antiviral Activity Available in Bangladesh and Mechanistic Insight Into Their Bioactive Metabolites on SARS-CoV-2, HIV and HBV. Front. Pharmacol. 2021, 12, 732891. [Google Scholar] [CrossRef]
- Kräusslich, H.; Müller, B. Antiviral Drugs. In Encyclopedia of Molecular Pharmacology; Offermanns, S., Rosenthal, W., Eds.; Springer: Berlin/Heidelberg, Germany, 2008; pp. 196–201. ISBN 978-3-540-38918-7. [Google Scholar]
- Niimi, Y.; Han, D.-S.S.; Mori, S.; Kobayashi, H. Detection of Cucumber Mosaic Virus, Lily Symptomless Virus and Lily Mottle Virus in Lilium Species by RT-PCR Technique. Sci. Hortic. 2003, 97, 57–63. [Google Scholar] [CrossRef]
- Gong, H.L.; Dusengemungu, L.; Igiraneza, C.; Rukundo, P. Molecular Regulation of Potato Tuber Dormancy and Sprouting: A Mini-Review. Plant Biotechnol. Rep. 2021, 15, 417–434. [Google Scholar] [CrossRef]
- Rhymer, J.M.; Simberloff, D. Extinction by Hybridization and Introgression. Annu. Rev. Ecol. Syst. 1996, 27, 83–109. [Google Scholar] [CrossRef]
- Azadi, P.; Otang, N.V.; Supaporn, H.; Khan, R.S.; Chin, D.P.; Nakamura, I.; Mii, M. Increased Resistance to Cucumber Mosaic Virus (CMV) in Lilium Transformed with a Defective CMV Replicase Gene. Biotechnol. Lett. 2011, 33, 1249–1255. [Google Scholar] [CrossRef]
- Nehra, N.S.; Kartha, K.K. Meristem and Shoot Tip Culture: Requirements and Applications. In Plant Cell and Tissue Culture; Springer: Dordrecht, The Netherlands, 1994; pp. 37–70. [Google Scholar]
- Chen, Y.; Hou, X.; Zheng, Y.; Lyu, Y. The Establishment of a Genetic Transformation System and the Acquisition of Transgenic Plants of Oriental Hybrid Lily (Lilium L.). Int. J. Mol. Sci. 2023, 24, 782. [Google Scholar] [CrossRef]
- Rubio, L.; Galipienso, L.; Ferriol, I. Detection of Plant Viruses and Disease Management: Relevance of Genetic Diversity and Evolution. Front. Plant Sci. 2020, 11, 1092. [Google Scholar] [CrossRef]
- Munir, M. Management of Plant Virus Diseases; Farmer’s Knowledge and Our Suggestions. Hosts Viruses 2017, 4, 28–33. [Google Scholar] [CrossRef]
- Roudine, S.; Le Lann, C.; Bouvaine, S.; Le Ralec, A.; van Baaren, J. Can Biological Control Be a Strategy to Control Vector-Borne Plant Viruses? J. Pest Sci. 2023, 96, 451–470. [Google Scholar] [CrossRef]
- Dixon, G.R. Control: Cultural and Biological Methods. In Plant Pathogens and their Control in Horticulture; Dixon, G.R., Ed.; Macmillan Education: London, UK, 1984; pp. 202–226. ISBN 978-1-349-06923-1. [Google Scholar]
- Zalom, F.G. Pesticide Use Practices in Integrated Pest Management. In Handbook of Pesticide Toxicology; Elsevier: London, UK, 2001; pp. 275–283. [Google Scholar]
- Chastagner, G.A.; van Tuyl, J.M.; Verbeek, M.; Miller, W.B.; Westerdahl, B.B. Diseases of Lily. In Disease of Lily; McGovern, R.J., Elmer, W.H., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 1229–1288. ISBN 978-3-319-39670-5. [Google Scholar]
- Dhiman, M.R.; Sharma, P.; Bhargava, B. Lilium: Conservation, Characterization, and Evaluation. In Floriculture and Ornamental Plants; Datta, S.K., Gupta, Y.C., Eds.; Springer: Singapore, 2021; pp. 1–36. ISBN 978-981-15-1554-5. [Google Scholar]
- He, X.; Xue, F.; Xu, S.; Wang, W. Rapid and Sensitive Detection of Lily Symptomless Virus by Reverse Transcription Loop-Mediated Isothermal Amplification. J. Virol. Methods 2016, 238, 38–41. [Google Scholar] [CrossRef]
- Nega, A. Review on Concepts in Biological Control of Plant Pathogens. J. Biol. Agric. Healthc. 2014, 4, 33–54. [Google Scholar]
- Trivedi, P.; Leach, J.E.; Tringe, S.G.; Sa, T.; Singh, B.K. Plant–Microbiome Interactions: From Community Assembly to Plant Health. Nat. Rev. Microbiol. 2020, 18, 607–621. [Google Scholar] [CrossRef]
- Wagemans, J.; Holtappels, D.; Vainio, E.; Rabiey, M.; Marzachì, C.; Herrero, S.; Ravanbakhsh, M.; Tebbe, C.C.; Ogliastro, M.; Ayllón, M.A.; et al. Going Viral: Virus-Based Biological Control Agents for Plant Protection. Annu. Rev. Phytopathol. 2022, 60, 21–42. [Google Scholar] [CrossRef]
- Tricahyati, T.; Suparman; Irsan, C. Natural Enemies of Pentalonia Nigronervosa, Vector of Banana Bunchy Top Virus. Biodiversitas 2022, 23, 3675–3684. [Google Scholar] [CrossRef]
- Kakati, N.; Nath, P. First Report on Development of Sustainable Management Strategy against Pentalonia Nigronervosa Coq. Vector of Banana Bunchy Top Virus Disease, Its Seasonal Variation and Effect on Yield of Banana in Jorhat District of Assam—A North Eastern State of India. J. Entomol. Zool. Stud. 2019, 7, 158–167. [Google Scholar] [CrossRef]
- de Oliveira, C.F.; Long, E.Y.; Finke, D.L. A Negative Effect of a Pathogen on Its Vector? A Plant Pathogen Increases the Vulnerability of Its Vector to Attack by Natural Enemies. Oecologia 2014, 174, 1169–1177. [Google Scholar] [CrossRef]
- He, Z.; Guo, J.-F.; Reitz, S.R.; Lei, Z.-R.; Wu, S.-Y. A Global Invasion by the Thrip, Frankliniella Occidentalis: Current Virus Vector Status and Its Management. Insect Sci. 2020, 27, 626–645. [Google Scholar] [CrossRef]
- Mouden, S.; Sarmiento, K.F.; Klinkhamer, P.G.L.; Leiss, K.A. Integrated Pest Management in Western Flower Thrips: Past, Present and Future. Pest Manag. Sci. 2017, 73, 813–822. [Google Scholar] [CrossRef]
- Wu, S.; Zhang, Z.; Gao, Y.; Xu, X.; Lei, Z. Interactions between Foliage- and Soil-Dwelling Predatory Mites and Consequences for Biological Control of Frankliniella Occidentalis. BioControl 2016, 61, 717–727. [Google Scholar] [CrossRef]
- Mo, L.F.; Zhi, J.R.; Tian, T. Biological Control Efficiency of Orius Similis Zheng (Hemiptera: Anthocoridae) on Frankliniella Occidentalis (Pergande) under Different Spatial and Caged Conditions. Acta Ecol. Sin. 2013, 33, 7132–7139. [Google Scholar]
- Aragón-Sánchez, M.; Román-Fernández, L.R.; Martínez-García, H.; Aragón-García, A.; Pérez-Moreno, I.; Marco-Mancebón, V.S. Rate of Consumption, Biological Parameters, and Population Growth Capacity of Orius Laevigatus Fed on Spodoptera Exigua. BioControl 2018, 63, 785–794. [Google Scholar] [CrossRef]
- Sarkar, S.C.; Wang, E.; Zhang, Z.; Wu, S.; Lei, Z. Laboratory and Glasshouse Evaluation of the Green Lacewing, Chrysopa Pallens (Neuroptera: Chrysopidae) against the Western Flower Thrips, Frankliniella Occidentalis (Thysanoptera: Thripidae). Appl. Entomol. Zool. 2019, 54, 115–121. [Google Scholar] [CrossRef]
- Messelink, G.J.; Van Steenpaal, S.E.F.; Ramakers, P.M.J. Evaluation of Phytoseiid Predators for Control of Western Flower Thrips on Greenhouse Cucumber. BioControl 2006, 51, 753–768. [Google Scholar] [CrossRef]
- Ahmed, N.; Lou, M. Efficacy of Two Predatory Phytoseiid Mites in Controlling the Western Flower Thrips, Frankliniella Occidentalis (Pergande) (Thysanoptera: Thripidae) on Cherry Tomato Grown in a Hydroponic System. Egypt. J. Biol. Pest Control 2018, 28, 15. [Google Scholar] [CrossRef]
- Pandit, M.A.; Kumar, J.; Gulati, S.; Bhandari, N.; Mehta, P.; Katyal, R.; Rawat, C.D.; Mishra, V.; Kaur, J. Major Biological Control Strategies for Plant Pathogens. Pathogens 2022, 11, 273. [Google Scholar] [CrossRef]
- Sato, H.; Hagiwara, K.; Nakamura, S.; Morikawa, T.; Honda, Y.; Omura, T. A Comparison of Sensitive and Specific Methods for the Detection of Lily Mottle Virus in Lily Plants. J. Phytopathol. 2002, 150, 20–24. [Google Scholar] [CrossRef]
- Boonham, N.; Kreuze, J.; Winter, S.; Van Der Vlugt, R.; Bergervoet, J.; Tomlinson, J.; Mumford, R.; van der Vlugt, R.; Bergervoet, J.; Tomlinson, J.; et al. Methods in Virus Diagnostics: From ELISA to next Generation Sequencing. Virus Res. 2014, 186, 20–31. [Google Scholar] [CrossRef]
- Cassedy, A.; Parle-McDermott, A.; O’Kennedy, R. Virus Detection: A Review of the Current and Emerging Molecular and Immunological Methods. Front. Mol. Biosci. 2021, 8, 637559. [Google Scholar] [CrossRef]
- Kim, J.H. Development of an Indirect ELISA and Immunocapture RT-PCR for Lily Virus Detection. J. Microbiol. Biotechnol. 2012, 22, 1776–1781. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Y.; Xie, Z.; Yang, G.; Guo, Z.; Wang, L. Simultaneous Detection of Three Lily Viruses Using Triplex IC-RT-PCR. J. Virol. Methods 2017, 249, 69–75. [Google Scholar] [CrossRef]
- Kim, H.S.; Kim, D.W.; Jung, Y.T. Development of Serological Procedures for Sensitive, Rapid Detection of Cucumber Mosaic Virus in Lilium. Hortic. Environ. Biotechnol. 2016, 57, 633–639. [Google Scholar] [CrossRef]
- Edwards, M.L.; Cooper, J.I. Plant Virus Detection Using a New Form of Indirect ELISA. J. Virol. Methods 1985, 11, 309–319. [Google Scholar] [CrossRef]
- Wang, R.; Wang, G.; Zhao, Q.; Zhang, Y.; An, L.; Wang, Y. Expression, Purification and Characterization of the Lily Symptomless Virus Coat Protein from Lanzhou Isolate. Virol. J. 2010, 7, 34. [Google Scholar] [CrossRef]
- Sun, N.; Deng, C.; Zhao, X.; Zhou, Q.; Ge, G.; Liu, Y.; Yan, W.; Xia, Q. Extraction of Total Nucleic Acid Based on Silica-Coated Magnetic Particles for RT-QPCR Detection of Plant RNA Virus/Viroid. J. Virol. Methods 2014, 196, 204–211. [Google Scholar] [CrossRef]
- Jeong, J.; Cho, S.; Lee, W.-H.; Lee, K.; Ju, H. Development of a Rapid Detection Method for Potato Virus X by Reverse Transcription Loop-Mediated Isothermal Amplification. Plant Pathol. J. 2015, 31, 219–225. [Google Scholar] [CrossRef]
- Guedez-López, G.V.; Alguacil-Guillén, M.; González-Donapetry, P.; Bloise, I.; Tornero-Marin, C.; González-García, J.; Mingorance, J.; García-Rodríguez, J.; Montero-Vega, M.D.; Romero, M.P.; et al. Evaluation of Three Immunochromatographic Tests for Rapid Detection of Antibodies against SARS-CoV-2. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 39, 2289–2297. [Google Scholar] [CrossRef]
- Jeong, J.-J.; Ju, H.-J.; Noh, J. A Review of Detection Methods for the Plant Viruses. Res. Plant Dis. 2014, 20, 173–181. [Google Scholar] [CrossRef]
- Flores, F.; Garrido, P.; Guevara, F.; Granda, R. Complete Genome Sequence of Lily Symptomless Virus, Isolated from Alstroemeria Plants in Ecuador. Microbiol. Resour. Announc. 2019, 8, 4–6. [Google Scholar] [CrossRef]
- Li, J.-W.; Zhang, X.-C.; Wang, M.-R.; Bi, W.-L.; Faisal, M.; da Silva, J.A.T.; Volk, G.M.; Wang, Q.-C. Development, Progress and Future Prospects in Cryobiotechnology of Lilium spp. Plant Methods 2019, 15, 125. [Google Scholar] [CrossRef] [PubMed]
- Lim, M.S.; Min, D.J.; Hong, J.S.; Choi, S.H. Simultaneous Detection of Four Lily-Infecting Viruses by a Multiplex RT-PCR Assay. J. Gen. Plant Pathol. 2021, 87, 219–224. [Google Scholar] [CrossRef]
- Van Brunschot, S.L.; Bergervoet, J.H.W.; Pagendam, D.E.; De Weerdt, M.; Geering, A.D.W.; Drenth, A.; Vlugt, R.A.A. Van Der A Bead-Based Suspension Array for the Multiplexed Detection of Begomoviruses and Their Whitefly Vectors. J. Virol. Methods 2014, 198, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Lim, M.S.; Kim, S.M.; Choi, S.H. Simultaneous Detection of Three Lily-Infecting Viruses Using a Multiplex Luminex Bead Array. J. Virol. Methods 2016, 231, 34–37. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Ming, J. Development of a Multiplex RT-PCR Assay for Simultaneous Detection of Lily Symptomless Virus, Lily Mottle Virus, Cucumber Mosaic Virus, and Plantago Asiatica Mosaic Virus in Lilies. Virol. J. 2022, 19, 4–9. [Google Scholar] [CrossRef]
- Gambley, C.F.; Geering, A.D.W.; Thomas, J.E. Development of an Immunomagnetic Capture-Reverse Transcriptase-PCR Assay for Three Pineapple Ampeloviruses. J. Virol. Methods 2009, 155, 187–192. [Google Scholar] [CrossRef]
- Huber, I.; Block, A.; Sebah, D.; Debode, F.; Morisset, D.; Grohmann, L.; Berben, G.; Štebih, D.; Milavec, M.; Žel, J.; et al. Development and Validation of Duplex, Triplex, and Pentaplex Real-Time PCR Screening Assays for the Detection of Genetically Modified Organisms in Food and Feed. J. Agric. Food Chem. 2013, 61, 10293–10301. [Google Scholar] [CrossRef]
- Rigotti, S.; Gugerli, P. Rapid Identification of Potato Virus Y Strains by One-Step Triplex RT-PCR. J. Virol. Methods 2007, 140, 90–94. [Google Scholar] [CrossRef]
- Zhang, Y.B.; Wang, Y.J.; Xie, Z.K.; Wang, R.Y.; Yang, G.; Guo, Z.H.; Qiu, Y. A Study of Viral Coat Protein Accumulation in Lily Chloroplasts from Mixed Virus Infections of Lily Mottle Virus and Cucumber Mosaic Virus. Plant Pathol. 2019, 68, 261–268. [Google Scholar] [CrossRef]
- Lee, H.-J.; Choi, S.; Cho, I.-S.; Yoon, J.-Y.; Jeong, R.-D. Development and Application of a Reverse Transcription Droplet Digital PCR Assay for Detection and Quantification of Plantago Asiatica Mosaic Virus. Crop Prot. 2023, 169, 106255. [Google Scholar] [CrossRef]
- Munro, S.B.; Kuypers, J.; Jerome, K.R. Comparison of a Multiplex Real-Time PCR Assay with a Multiplex Luminex Assay for Influenza Virus Detection. J. Clin. Microbiol. 2013, 51, 1124–1129. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Xu, H.; Cao, Y.; Yang, P.; Feng, Y.; Tang, Y.; Yuan, S.; Ming, J. Validation of Reference Genes for Quantitative Real-Time PCR during Bicolor Tepal Development in Asiatic Hybrid Lilies (Lilium spp.). Front. Plant Sci. 2017, 8, 669. [Google Scholar] [CrossRef]
- Sun, B.; Bosch, A.; Myrmel, M. Extended Direct Lysis Method for Virus Detection on Berries Including Droplet Digital RT-PCR or Real Time RT-PCR with Reduced Influence from Inhibitors. J. Virol. Methods 2019, 271, 113638. [Google Scholar] [CrossRef]
- Ryabchevskaya, E.M.; Evtushenko, E.A.; Arkhipenko, M.V.; Donchenko, E.K.; Nikitin, N.A.; Atabeko, J.G.; Karpova, O. V A Recombinant Rotavirus Antigen Based on the Coat Protein of Alternanthera Mosaic Virus. Mol. Biol. 2020, 54, 278–284. [Google Scholar] [CrossRef]
- Kim, M.H.; Park, I.S.; Il Park, K.; Oh, W.; Kim, K.W. Elimination of Lily Symptomless Virus by in Vitro Scaling and Reinfection Rates under Various Culture Conditions in Korean Native Lilies. Korean J. Hortic. Sci. Technol. 2015, 33, 891–899. [Google Scholar] [CrossRef]
- Zhao, B.; Yang, D.; Zhang, Y.; Xu, Y.; Zhao, X.; Liang, J.; Fan, X.; Du, Y.; Zhu, Z.; Shi, B.; et al. Rapid Visual Detection of Lily Mottle Virus Using a Loop-Mediated Isothermal Amplification Method. Arch. Virol. 2018, 163, 545–548. [Google Scholar] [CrossRef]
- Yin, L.; Man, S.; Ye, S.; Liu, G.; Ma, L. CRISPR-Cas Based Virus Detection: Recent Advances and Perspectives. Biosens. Bioelectron. 2021, 193, 113541. [Google Scholar] [CrossRef]
- Khater, M.; de la Escosura-Muñiz, A.; Merkoçi, A. Biosensors for Plant Pathogen Detection. Biosens. Bioelectron. 2017, 93, 72–86. [Google Scholar] [CrossRef]
- Quer, J.; Colomer-Castell, S.; Campos, C.; Andrés, C.; Piñana, M.; Cortese, M.F.; González-Sánchez, A.; Garcia-Cehic, D.; Ibáñez, M.; Pumarola, T.; et al. Next-Generation Sequencing for Confronting Virus Pandemics. Viruses 2022, 14, 600. [Google Scholar] [CrossRef]
- Babu, B.; Ochoa-Corona, F.M.; Paret, M.L. Recombinase Polymerase Amplification Applied to Plant Virus Detection and Potential Implications. Anal. Biochem. 2018, 546, 72–77. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).