Synergistic Effect of a Plant-Derived Protein Hydrolysate and Arbuscular Mycorrhizal Fungi on Eggplant Grown in Open Fields: A Two-Year Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Site and Plant Material
2.2. Plant-Derived Protein Hydrolysate Application and Arbuscular Mycorrhizal Fungi Inoculation
2.3. Statistics and Design
2.4. Plant Growth and Yield
2.5. Fruit Composition
2.6. Mineral Profile and Nitrogen Indices
3. Results
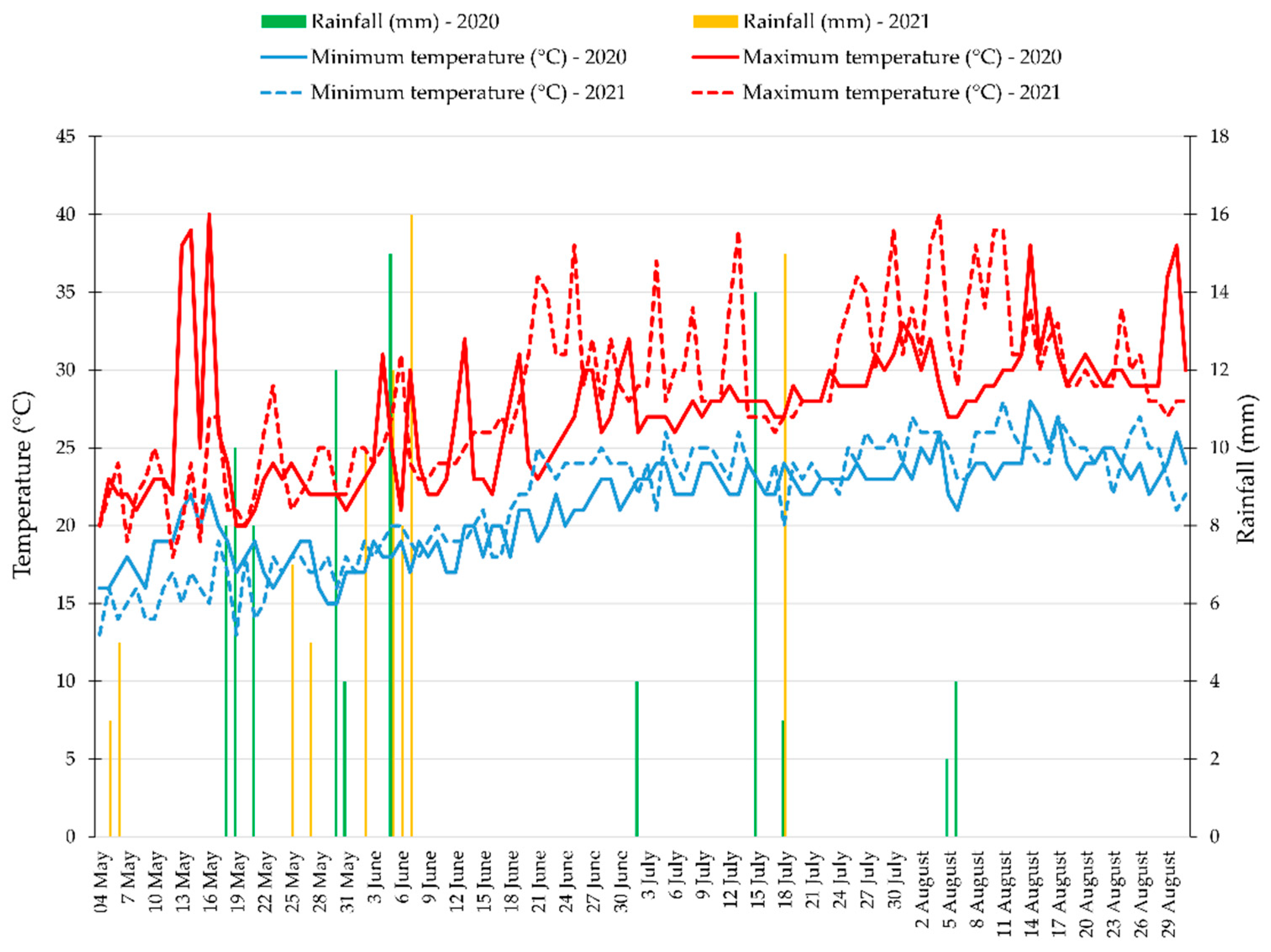
3.1. Weather Data and Mycorrhizal Inoculation
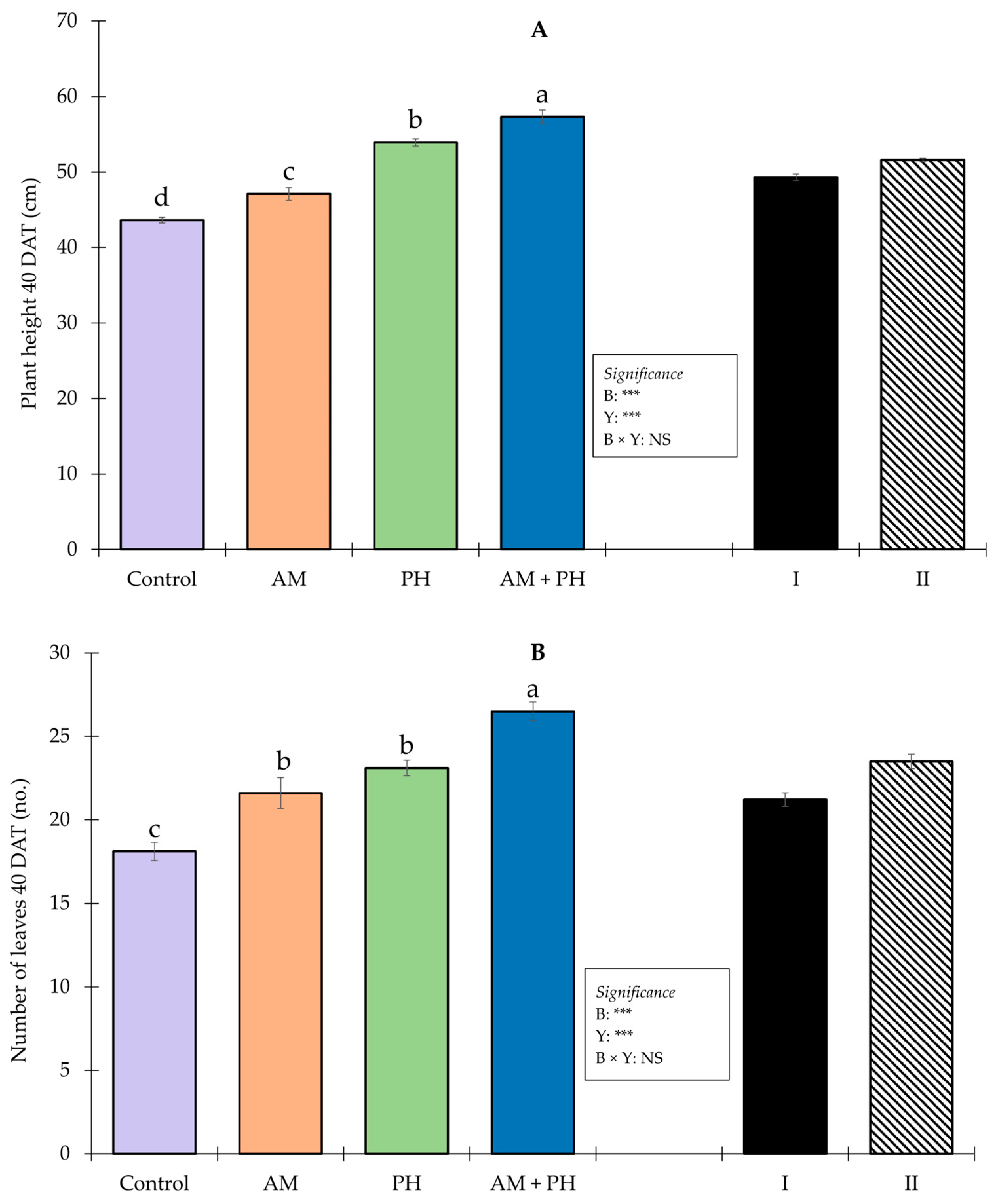
3.2. Plant Growth and Yield
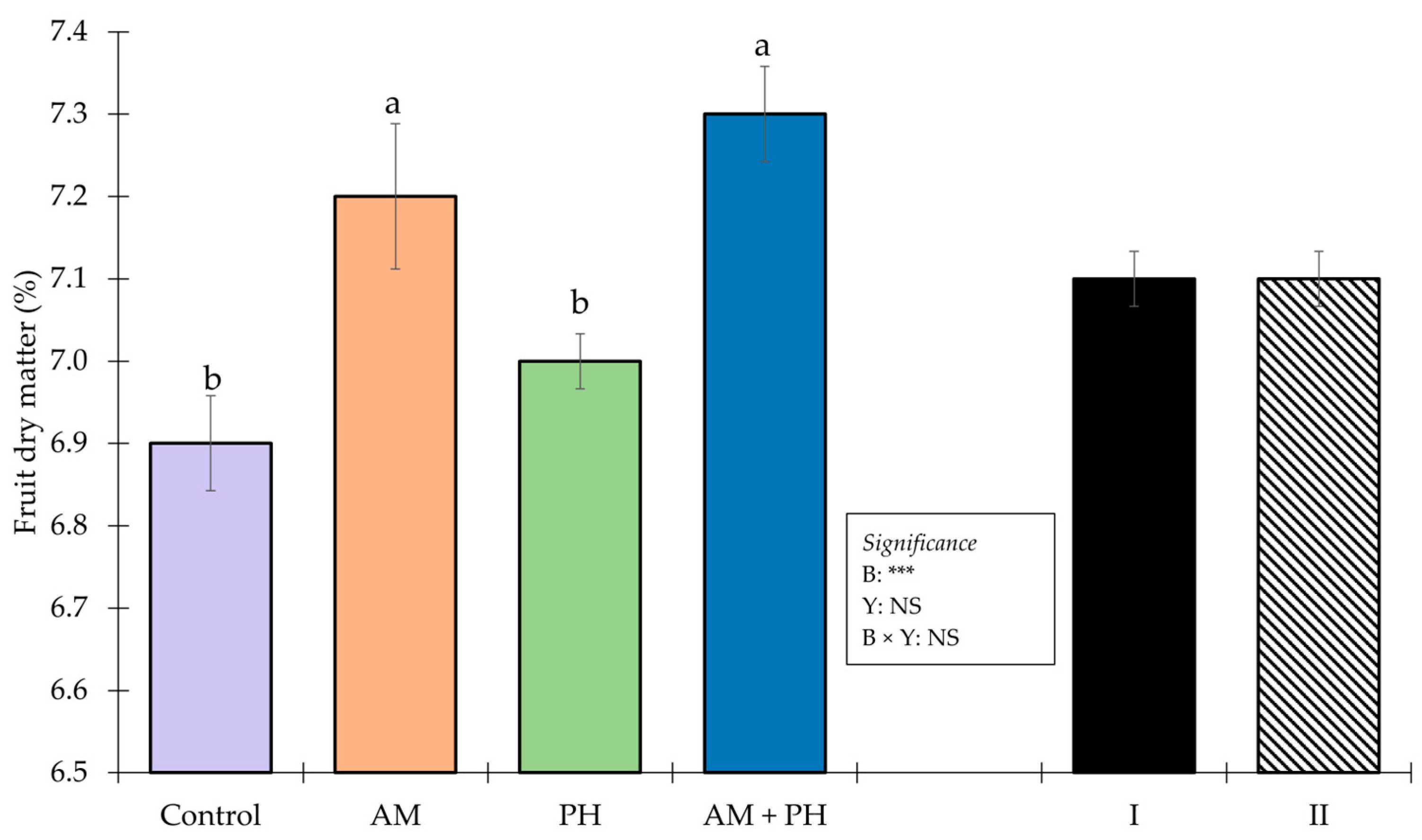
3.3. Fruit Quality and Browning
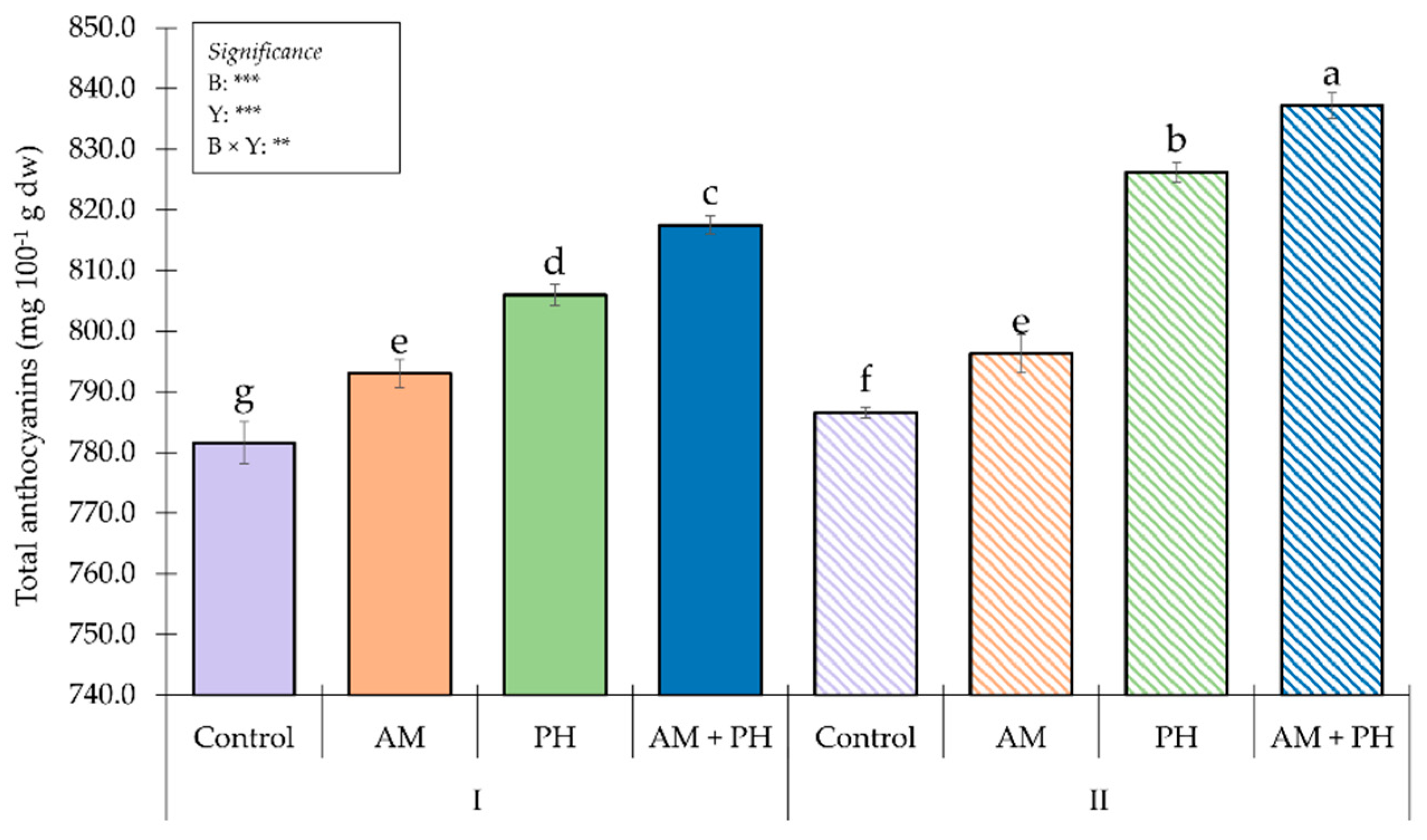
3.4. Fruit Proteins and Mineral Profile
3.5. Nitrogen Indices
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Zenia, M.; Halina, B. Content of microelements in eggplant fruits depending on nitrogen fertilization and plant training method. J. Elementol. 2008, 13, 269–274. [Google Scholar]
- Faostat. Available online: https://www.fao.org/faostat/en/ (accessed on 20 March 2023).
- Agristat. Available online: http://dati.istat.it/Index.aspx (accessed on 20 March 2023).
- Tesi, R. Orticoltura Mediterranea Sostenibile; Pàtron Editore: Bologna, Italy, 2010. [Google Scholar]
- Colla, G.; Rouphael, Y. Biostimulants in horticulture. Sci. Hortic. 2015, 196, 1–134. [Google Scholar] [CrossRef]
- Di Mola, I.; Conti, S.; Cozzolino, E.; Melchionna, G.; Ottaiano, L.; Testa, A.; Sabatino, L.; Rouphael, Y.; Mori, M. Plant-based protein hydrolysate improves salinity tolerance in hemp: Agronomical and physiological aspects. Agronomy 2021, 11, 342. [Google Scholar] [CrossRef]
- D’Anna, F.; Sabatino, L. Morphological and agronomical characterization of eggplant genetic resources from the Sicily area. J. Food Agric. Environ. 2013, 11, 401–404. [Google Scholar]
- Consentino, B.B.; Aprile, S.; Rouphael, Y.; Ntatsi, G.; De Pasquale, C.; Iapichino, G.; Alibrandi, P.; Sabatino, L. Application of PGPB combined with variable n doses affects growth, yield-related traits, n-fertilizer efficiency and nutritional status of lettuce grown under controlled condition. Agronomy 2022, 12, 236. [Google Scholar] [CrossRef]
- La Bella, S.; Consentino, B.B.; Rouphael, Y.; Ntatsi, G.; De Pasquale, C.; Iapichino, G.; Sabatino, L. Impact of Ecklonia maxima seaweed extract and Mo foliar treatments on biofortification, spinach yield, quality and NUE. Plants 2021, 10, 1139. [Google Scholar] [CrossRef]
- Sabatino, L.; La Bella, S.; Ntatsi, G.; Iapichino, G.; D’Anna, F.; De Pasquale, C.; Consentino, B.B.; Rouphael, Y. Selenium biofortification and grafting modulate plant performance and functional features of cherry tomato grown in a soilless system. Sci. Hortic. 2021, 285, 110095. [Google Scholar] [CrossRef]
- Consentino, B.B.; Ciriello, M.; Sabatino, L.; Vultaggio, L.; Baldassano, S.; Vasto, S.; Rouphael, Y.; La Bella, S.; De Pascale, S. Current acquaintance on agronomic biofortification to modulate the yield and functional value of vegetable crops: A review. Horticulturae 2023, 9, 219. [Google Scholar] [CrossRef]
- Consentino, B.B.; Vultaggio, L.; Iacuzzi, N.; La Bella, S.; De Pasquale, C.; Rouphael, Y.; Ntatsi, G.; Virga, G.; Sabatino, L. Iodine biofortification and seaweed extract-based biostimulant supply interactively drive the yield, quality, and functional traits in strawberry fruits. Plants 2023, 12, 245. [Google Scholar] [CrossRef]
- Sabatino, L.; Consentino, B.B.; Rouphael, Y.; Baldassano, S.; De Pasquale, C.; Ntatsi, G. Ecklonia maxima-derivate seaweed extract supply as mitigation strategy to alleviate drought stress in chicory plants. Sci. Hortic. 2023, 312, 111856. [Google Scholar] [CrossRef]
- Consentino, B.B.; Sabatino, L.; Mauro, R.P.; Nicoletto, C.; De Pasquale, C.; Iapichino, G.; La Bella, S. Seaweed extract improves Lagenaria siceraria young shoot production, mineral profile and functional quality. Horticulturae 2021, 7, 549. [Google Scholar] [CrossRef]
- Consentino, B.B.; Sabatino, L.; Vultaggio, L.; Rotino, G.L.; La Placa, G.G.; D’Anna, F.; Leto, C.; Iacuzzi, N.; De Pasquale, C. Grafting eggplant onto underutilized solanum species and biostimulatory action of Azospirillum brasilense modulate growth, yield, nue and nutritional and functional traits. Horticulturae 2022, 8, 722. [Google Scholar] [CrossRef]
- Giovannetti, M.; Fortuna, P.; Citernesi, A.S.; Morini, S.; Nuti, M.P. The occurrence of anastomosis formation and nuclear exchange in intact arbuscular mycorrhizal networks. New Phytol. 2001, 151, 717–724. [Google Scholar] [CrossRef] [PubMed]
- Avio, L.; Pellegrino, E.; Bonari, E.; Giovannetti, M. Functional diversity of arbuscular mycorrhizal fungal isolates in relation to extraradical mycelial networks. New Phytol. 2006, 172, 347–357. [Google Scholar] [CrossRef] [PubMed]
- Sbrana, C.; Turrini, A.; Giovannetti, M. The crosstalk between plants and their arbuscular mycorrhizal symbionts: A mycocentric view. In Biocommunication: Sign-Mediated Interactions Between Cells and Organisms; World Scientific: Singapore, 2017; pp. 285–308. [Google Scholar]
- Wu, Q.S.; Srivastava, A.K.; Zou, Y.N. AMF-induced tolerance to drought stress in citrus: A review. Sci. Hortic. 2013, 164, 77–87. [Google Scholar] [CrossRef]
- Smith, S.E.; Smith, F.A. Roles of arbuscular mycorrhizas in plant nutrition and growth: New paradigms from cellular to ecosystem scales. Annu. Rev. Plant Biol. 2011, 63, 227–250. [Google Scholar] [CrossRef]
- Rouphael, Y.; Franken, P.; Schneider, C.; Schwarz, D.; Giovannetti, M.; Agnolucci, M. Arbuscular mycorrhizal fungi act as bio-stimulants in horticultural crops. Sci. Hortic. 2015, 196, 91–108. [Google Scholar] [CrossRef]
- Sbrana, C.; Avio, L.; Giovannetti, M. Beneficial mycorrhizal symbionts affecting the production of health-promoting phytochemicals. Electrophoresis 2014, 35, 1535–1546. [Google Scholar] [CrossRef]
- Strack, D.; Fester, T. Isoprenoid metabolism and plastid reorganization in arbuscular mycorrhizal roots. New Phytol. 2006, 172, 22–34. [Google Scholar] [CrossRef]
- du Jardin, P. Plant biostimulants: Definition, concept, main categories and regulation. Sci. Hortic. 2015, 196, 3–14. [Google Scholar] [CrossRef]
- Colla, G.; Nardi, S.; Cardarelli, M.; Ertani, A.; Lucini, L.; Canaguier, R.; Rouphael, Y. Protein hydrolysates as biostimulants in horticulture. Sci. Hortic. 2015, 196, 28–38. [Google Scholar] [CrossRef]
- Colla, G.; Rouphael, Y. Biostimolanti per un’Agricoltura Sostenibile: Cosa Sono, Come Agiscono e Modalità di Utilizzo; Edizioni L’Informatore Agrario: Verona, Italy, 2019; p. 189. [Google Scholar]
- Sabatino, L.; Consentino, B.B.; Rouphael, Y.; De Pasquale, C.; Iapichino, G.; D’Anna, F.; La Bella, S. Protein hydrolysates and mo-biofortification interactively modulate plant performance and quality of ‘canasta’ lettuce grown in a protected environment. Agronomy 2021, 11, 1023. [Google Scholar] [CrossRef]
- Rouphael, Y.; Colla, G. Synergistic biostimulatory action: Designing the next generation of plant biostimulants for sustainable agriculture. Front. Plant Sci. 2018, 9, 1655. [Google Scholar] [CrossRef] [PubMed]
- Sabatino, L.; Consentino, B.B.; Ntatsi, G.; La Bella, S.; Baldassano, S.; Rouphael, Y. Stand-alone or combinatorial effects of grafting and microbial and non-microbial derived compounds on vigour, yield and nutritive and functional quality of greenhouse eggplant. Plants 2022, 11, 1175. [Google Scholar] [CrossRef]
- Baixauli, C. Berenjena. In La Horticultura Espãnola; Nuez, F., Llácer, G., Eds.; Ediciones de Horticultura: Reus, Spain, 2001; pp. 104–108. [Google Scholar]
- Phillips, J.; Hayman, D. Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans. Br. Mycol. Soc. 1970, 55, 158–168. [Google Scholar] [CrossRef]
- Torta, L.; Mondello, V.; Burruano, S. Valutazione delle caratteristiche morfo-anatomiche di alcune simbiosi micorriziche mediante tecniche colorimetriche usuali e innovative. Micol. Ital. 2003, 2, 53–59. [Google Scholar]
- Larrigaudiere, C.; Lentheric, I.; Vendrell, M. Relationship between enzymatic browning and internal disorders in controlled atmosphere stored pears. J. Sci. Food Agric. 1998, 78, 232–236. [Google Scholar] [CrossRef]
- Concellón, A.; Añón, M.C.; Chaves, A.R. Effect of low temperature storage on physical and physiological characteristics of eggplant fruit (Solanum melongena L.). LWT Food Sci. Technol. 2007, 40, 389–396. [Google Scholar] [CrossRef]
- Stommel, J.R.; Whitaker, B.D. Phenolic acid content and composition of eggplant fruit in a germplasm core subset. J. Am. Soc. Hortic. Sci. 2003, 128, 704–710. [Google Scholar] [CrossRef]
- Mennella, G.; Lo Scalzo, R.; Fibiani, M.; D’Alessandro, A.; Francese, G.; Toppino, L.; Acciarri, N.; De Almeida, A.E.; Rotino, G.L. Chemical and bioactive quality traits during fruit ripening in eggplant (S. melongena L.) and allied species. J. Agric. Food Chem. 2012, 60, 11821–11831. [Google Scholar] [CrossRef]
- Birner, J. A method for the determination of total steroid bases. J. Pharm. Sci. 1969, 58, 258–259. [Google Scholar] [CrossRef]
- Kuronen, P.; Väänänen, T.; Pehu, E. Reversed-phase liquid chromatographic separation and simultaneous profiling of steroidal glycoalkaloids and their aglycones. J. Chromatogr. A 1999, 863, 25–35. [Google Scholar] [CrossRef]
- Fogg, D.N.; Wilkinson, A.N. The colorimetric determination of phosphorus. Analyst 1958, 83, 406–414. [Google Scholar] [CrossRef]
- Morand, P.; Gullo, J.L. Mineralisation des tissus vegetaux en vue du dosage de P., Ca, Mg, Na, K. Ann. Agron. 1970, 21, 229–236. [Google Scholar]
- Sabatino, L.; Iapichino, G.; Consentino, B.B.; D’Anna, F.; Rouphael, Y. Rootstock and arbuscular mycorrhiza combinatorial effects on eggplant crop performance and fruit quality under greenhouse conditions. Agronomy 2020, 10, 693. [Google Scholar] [CrossRef]
- Oztekin, G.B.; Tuzel, Y.; Tuzel, I.H. Does mycorrhiza improve salinity tolerance in grafted plants? Sci. Hortic. 2013, 149, 55–60. [Google Scholar] [CrossRef]
- De Pascale, S.; Rouphael, Y.; Colla, G. Plant biostimulants: Innovative tool for enhancing plant nutrition in organic farming. Eur. J. Hortic. Sci. 2018, 82, 277–285. [Google Scholar] [CrossRef]
- Rouphael, Y.; Colla, G.; Hoagland, L.; Giordano, M.; El-Nakhel, C.; Cardarelli, M. Vegetal-protein hydrolysates based microgranule enhances growth, mineral content, and quality traits of vegetable transplants. Sci. Hortic. 2021, 290, 110554. [Google Scholar] [CrossRef]
- Consentino, B.B.; Virga, G.; La Placa, G.G.; Sabatino, L.; Rouphael, Y.; Ntatsi, G.; Iapichino, G.; La Bella, S.; Mauro, R.P.; D’Anna, F.; et al. Celery (Apium graveolens L.) performances as subjected to different sources of protein hydrolysates. Plants 2020, 9, 1633. [Google Scholar] [CrossRef]
- Colla, G.; Rouphael, Y.; Canaguier, R.; Svecova, E.; Cardarelli, M. Biostimulant action of a plant-derived protein hydrolysate produced through enzymatic hydrolysis. Front. Plant Sci. 2014, 5, 448. [Google Scholar] [CrossRef]
- Sharma, M.; Delta, A.K.; Dhanda, P.S.; Kaushik, P.; Mohanta, Y.K.; Saravanan, M.; Mohanta, T.K. AMF and PSB applications modulated the biochemical and mineral content of the eggplants. J. Basic Microbiol. 2022, 62, 1371–1378. [Google Scholar] [CrossRef]
- Lucini, L.; Rouphael, Y.; Cardarelli, M.; Canaguier, R.; Kumar, P.; Colla, G. The effect of a plant-derived biostimulant on metabolic profiling and crop performance of lettuce grown under saline conditions. Sci. Hortic. 2015, 182, 124–133. [Google Scholar] [CrossRef]
- Miceli, A.; Romano, C.; Moncada, A.; Piazza, G.; Torta, L.; D’Anna, F.; Vetrano, F. Yield and quality of mini-watermelon as affected by grafting and mycorrhizal inoculum. J. Agric. Sci. Technol. 2016, 18, 505–516. [Google Scholar]
- Maboko, M.M.; Bertling, I.; du Plooy, C.P. Arbuscular mycorrhiza has limited effects on yield and quality of tomatoes grown under soilless cultivation. Acta Agric. Scand. Sect. B Soil Plant. Sci. 2013, 63, 261–270. [Google Scholar] [CrossRef]
- Cozzolino, E.; Di Mola, I.; Ottaiano, L.; El-Nakhel, C.; Rouphael, Y.; Mori, M. Foliar application of plant-based biostimulants improve yield and upgrade qualitative characteristics of processing tomato. Ital. J. Agron. 2021, 16, 1825. [Google Scholar] [CrossRef]
- Soteriou, G.A.; Rouphael, Y.; Emmanouilidou, M.G.; Antoniou, C.; Kyratzis, A.C.; Kyriacou, M.C. Biostimulatory action of vegetal protein hydrolysate and the configuration of fruit physicochemical characteristics in grafted watermelon. Horticulturae 2021, 7, 313. [Google Scholar] [CrossRef]
- Ordookhani, K.; Zare, M. Effect of pseudomonas, azotobacter and arbuscular mycorrhiza fungi on lycopene, antioxidant activity and total soluble solid in tomato (Lycopersicon esculentum F1 Hybrid, Delba). Adv. Environ. Biol. 2011, 5, 1290–1294. [Google Scholar]
- Rouphael, Y.; Colla, G.; Giordano, M.; El-Nakhel, C.; Kyriacou, M.C.; De Pascale, S. Foliar applications of a legume-derived protein hydrolysate elicit dose-dependent increases of growth, leaf mineral composition, yield and fruit quality in two greenhouse tomato cultivars. Sci. Hortic. 2017, 226, 353–360. [Google Scholar] [CrossRef]
- Ertani, A.; Pizzeghello, D.; Francioso, O.; Sambo, P.; Sanchez-Cortes, S.; Nardi, S. Capsicum chinensis L. growth and nutraceutical properties are enhanced by biostimulants in a long-term period: Chemical and metabolomic approaches. Front. Plant Sci. 2014, 5, 375. [Google Scholar] [CrossRef]
- Helyes, L.; Nagy, Z.; Daood, H.; Pék, Z.; Lugasi, A. The simultaneous effect of heat stress and water supply on total polyphenol content of eggplant. Appl. Ecol. Environ. Res. 2015, 13, 583–595. [Google Scholar]
- Papathanasiou, F.; Mitchell, S.H.; Watson, S.; Harvey, B.M.R. Effect of environmental stress during tuber development on accumulation of glycoalkaloids in potato (Solanum tuberosum L.). J. Sci. Food Agric. 1999, 79, 1183–1189. [Google Scholar] [CrossRef]
- Van Oosten, M.J.; Pepe, O.; De Pascale, S.; Silletti, S.; Maggio, A. The role of biostimulants and bioeffectors as alleviators of abiotic stress in crop plants. Chem. Biol. Technol. Agric. 2017, 4, 5. [Google Scholar] [CrossRef]
- Amr, A.; Hadidi, N. Effect of cultivar and harvest data on nitrate (NO3) and nitrite (NO2) content of selected vegetables gown under open field and greenhouse conditions in Jordan. J. Food Comp. Anal. 2001, 14, 59–67. [Google Scholar] [CrossRef]
- Calvo, P.; Nelson, L.; Kloepper, J.W. Agricultural uses of plant biostimulants. Plant Soil 2014, 383, 3–41. [Google Scholar] [CrossRef]
- Ma, K.K.; Greis, M.; Lu, J.; Nolden, A.A.; McClements, D.J.; Kinchla, A.J. Functional Performance of Plant Proteins. Foods 2022, 11, 594. [Google Scholar] [CrossRef]
- Badr, M.A.; El-Tohamy, W.A.; Abou-Hussein, S.D.; Gruda, N.S. Deficit irrigation and arbuscular mycorrhiza as a water-saving strategy for eggplant production. Horticulturae 2020, 6, 45. [Google Scholar] [CrossRef]
- Zhu, X.; Song, F.; Liu, S.; Liu, F. Arbuscular mycorrhiza improve growth, nitrogen uptake, and nitrogen use efficiency in wheat grown under elevated CO2. Mycorrhiza 2015, 26, 133–140. [Google Scholar] [CrossRef]
- Di Mola, I.; Cozzolino, E.; Ottaiano, L.; Nocerino, S.; Rouphael, Y.; Colla, G.; El-Nakhel, C.; Mori, M. Nitrogen Use and Uptake Efficiency and Crop Performance of Baby Spinach (Spinacia oleracea L.) and Lamb’s Lettuce (Valerianella locusta L.) Grown under Variable Sub-Optimal N Regimes Combined with Plant-Based Biostimulant Application. Agronomy 2020, 10, 278. [Google Scholar] [CrossRef]
- Copetta, A.; Lingua, G.; Berta, G. Effects of three AM fungi on growth, distribution of glandular hairs, and essential oil production in Ocimum basilicum L. var. Genovese. Mycorrhiza 2006, 16, 485–494. [Google Scholar] [CrossRef]
- Toussaint, J.P.; Smith, F.A.; Smith, S.E. Arbuscular mycorrhizal fungi caninduce the production of phytochemicals in sweet basil irrespective of phosphorus nutrition. Mycorrhiza 2007, 17, 291–297. [Google Scholar] [CrossRef]
- Rasouli-Sadaghiani, M.; Hassani, A.; Barin, M.; Rezaee Danesh, Y.; Sefidkon, F. Effects of arbuscular mycorrhizal (AM) fungi on growth, essential oil production and nutrients uptake in basil. J. Med. Plant Res. 2010, 4, 2222–2228. [Google Scholar]
- Baslam, M.; Garmendia, I.; Goicoechea, N. Arbuscular mycorrhizal fungi (AMF) improved growth and nutritional quality of greenhouse-grown lettuce. J. Agric. Food Chem. 2011, 59, 5504–5515. [Google Scholar] [CrossRef] [PubMed]
- Castellanos-Morales, V.; Villegas, J.; Wendelin, S.; Vierheilig, H.; Eder, R.; Cardenas-Navarro, R. Root colonization by the arbuscular mycorrhizal fungus Glomus intraradices alters the quality of strawberry fruit (Fragaria × ananassa Duch.) at different nitrogen levels. J. Sci. Food Agric. 2010, 90, 1774–1782. [Google Scholar]
- Shao, L.; Shu, Z.; Sun, S.; Peng, C.; Wang, X.; Lin, Z. Antioxidation of anthocyanins in photosynthesis under high temperature stress. J. Integr. Plant Biol. 2007, 49, 1341–1351. [Google Scholar] [CrossRef]
- Mishra, B.B.; Gautam, S.; Sharma, A. Free phenolics and polyphenol oxidase (PPO): The factors affecting post-cut browning in eggplant (Solanum melongena). Food Chem. 2013, 139, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Gisbert, C.; Prohens, J.; Raig’on, M.D.; Stommel, J.R.; Nuez, F. Eggplant relatives as sources of variation for developing new rootstocks: Effects of grafting on eggplant yield and fruit apparent quality and composition. Sci. Hortic. 2011, 128, 14–22. [Google Scholar] [CrossRef]

| Treatments | Total Yield (kg Plant−1) | Marketable Yield (kg Plant−1) | Average Marketable Fruit Weight (g) | No. of Marketable Fruits (no.) | Discarded Production (%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Biostimulant (B) | ||||||||||
| Control | 3.8 | d | 3.3 | d | 373.3 | d | 9.0 | b | 11.0 | a |
| AM | 4.0 | c | 3.7 | c | 396.2 | c | 9.5 | b | 5.8 | c |
| PH | 4.4 | b | 4.0 | b | 430.0 | b | 9.4 | b | 8.3 | b |
| AM + PH | 4.7 | a | 4.5 | a | 444.2 | a | 10.0 | a | 6.3 | c |
| Year (Y) | ||||||||||
| I | 4.3 | 4.0 | 417.0 | 9.6 | 7.3 | |||||
| II | 4.2 | 3.8 | 404.8 | 9.4 | 8.3 | |||||
| Significance | ||||||||||
| B | *** | *** | *** | *** | *** | |||||
| Y | *** | *** | ** | * | NS | |||||
| B × Y | NS | NS | NS | NS | NS | |||||
| Treatments | Firmness (N) | SSC (°Brix) | Chlorogenic Acid (mg 100 g−1 of dw) | Glycoalkaloids (mg 100 g−1 of dw) | ||||
|---|---|---|---|---|---|---|---|---|
| Biostimulant (B) | ||||||||
| Control | −40.9 | b | 4.4 | c | 763.4 | c | 90.3 | a |
| AM | −44.1 | c | 4.8 | b | 778.5 | b | 78.1 | b |
| PH | −38.2 | a | 4.7 | b | 776.2 | b | 72.2 | c |
| AM + PH | −44.2 | c | 5.1 | a | 798.7 | a | 72.4 | c |
| Year (Y) | ||||||||
| I | −41.9 | 4.7 | 772.0 | 79.0 | ||||
| II | −41.8 | 4.7 | 786.2 | 77.5 | ||||
| Significance | ||||||||
| B | *** | *** | *** | *** | ||||
| Y | NS | NS | *** | NS | ||||
| B × Y | NS | NS | NS | NS | ||||
| Treatments | Proteins (g 100 g−1 dw) | P (mg 100 g−1 dw) | K (mg 100 g−1 dw) | Ca (mg 100 g−1 dw) | Mg (mg 100 g−1 dw) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Biostimulant (B) | ||||||||||
| Control | 9.66 | a | 558.0 | a | 346.3 | c | 112.6 | a | 18.51 | c |
| AM | 9.66 | a | 559.1 | a | 346.4 | c | 112.8 | a | 20.71 | b |
| PH | 8.68 | b | 558.9 | a | 368.9 | b | 112.6 | a | 18.50 | c |
| AM + PH | 8.68 | b | 558.6 | a | 376.0 | a | 112.7 | a | 22.93 | a |
| Year (Y) | ||||||||||
| I | 9.10 | 558.4 | 335.6 | 112.6 | 19.92 | |||||
| II | 9.25 | 558.9 | 363.2 | 112.7 | 20.40 | |||||
| Significance | ||||||||||
| B | *** | NS | *** | NS | *** | |||||
| Y | *** | NS | *** | NS | * | |||||
| B × Y | NS | NS | NS | NS | NS | |||||
| Treatments | NUE (t/kg) | UE (kg/kg) | ||
|---|---|---|---|---|
| Biostimulant (B) | ||||
| Control | 0.30 | d | 0.32 | c |
| AM | 0.32 | c | 0.36 | b |
| PH | 0.35 | b | 0.34 | b |
| PH + AM | 0.38 | a | 0.38 | a |
| Year (Y) | ||||
| I | 0.34 | 0.34 | ||
| II | 0.33 | 0.36 | ||
| Significance | ||||
| B | *** | *** | ||
| Y | *** | *** | ||
| B × Y | NS | NS | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Miceli, G.; Vultaggio, L.; Sabatino, L.; De Pasquale, C.; La Bella, S.; Consentino, B.B. Synergistic Effect of a Plant-Derived Protein Hydrolysate and Arbuscular Mycorrhizal Fungi on Eggplant Grown in Open Fields: A Two-Year Study. Horticulturae 2023, 9, 592. https://doi.org/10.3390/horticulturae9050592
Di Miceli G, Vultaggio L, Sabatino L, De Pasquale C, La Bella S, Consentino BB. Synergistic Effect of a Plant-Derived Protein Hydrolysate and Arbuscular Mycorrhizal Fungi on Eggplant Grown in Open Fields: A Two-Year Study. Horticulturae. 2023; 9(5):592. https://doi.org/10.3390/horticulturae9050592
Chicago/Turabian StyleDi Miceli, Giuseppe, Lorena Vultaggio, Leo Sabatino, Claudio De Pasquale, Salvatore La Bella, and Beppe Benedetto Consentino. 2023. "Synergistic Effect of a Plant-Derived Protein Hydrolysate and Arbuscular Mycorrhizal Fungi on Eggplant Grown in Open Fields: A Two-Year Study" Horticulturae 9, no. 5: 592. https://doi.org/10.3390/horticulturae9050592
APA StyleDi Miceli, G., Vultaggio, L., Sabatino, L., De Pasquale, C., La Bella, S., & Consentino, B. B. (2023). Synergistic Effect of a Plant-Derived Protein Hydrolysate and Arbuscular Mycorrhizal Fungi on Eggplant Grown in Open Fields: A Two-Year Study. Horticulturae, 9(5), 592. https://doi.org/10.3390/horticulturae9050592










