Multivariate Analysis of the Phenological Stages, Yield, Bioactive Components, and Antioxidant Capacity Effects in Two Mulberry Cultivars under Different Cultivation Modes
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials and Experimental Design
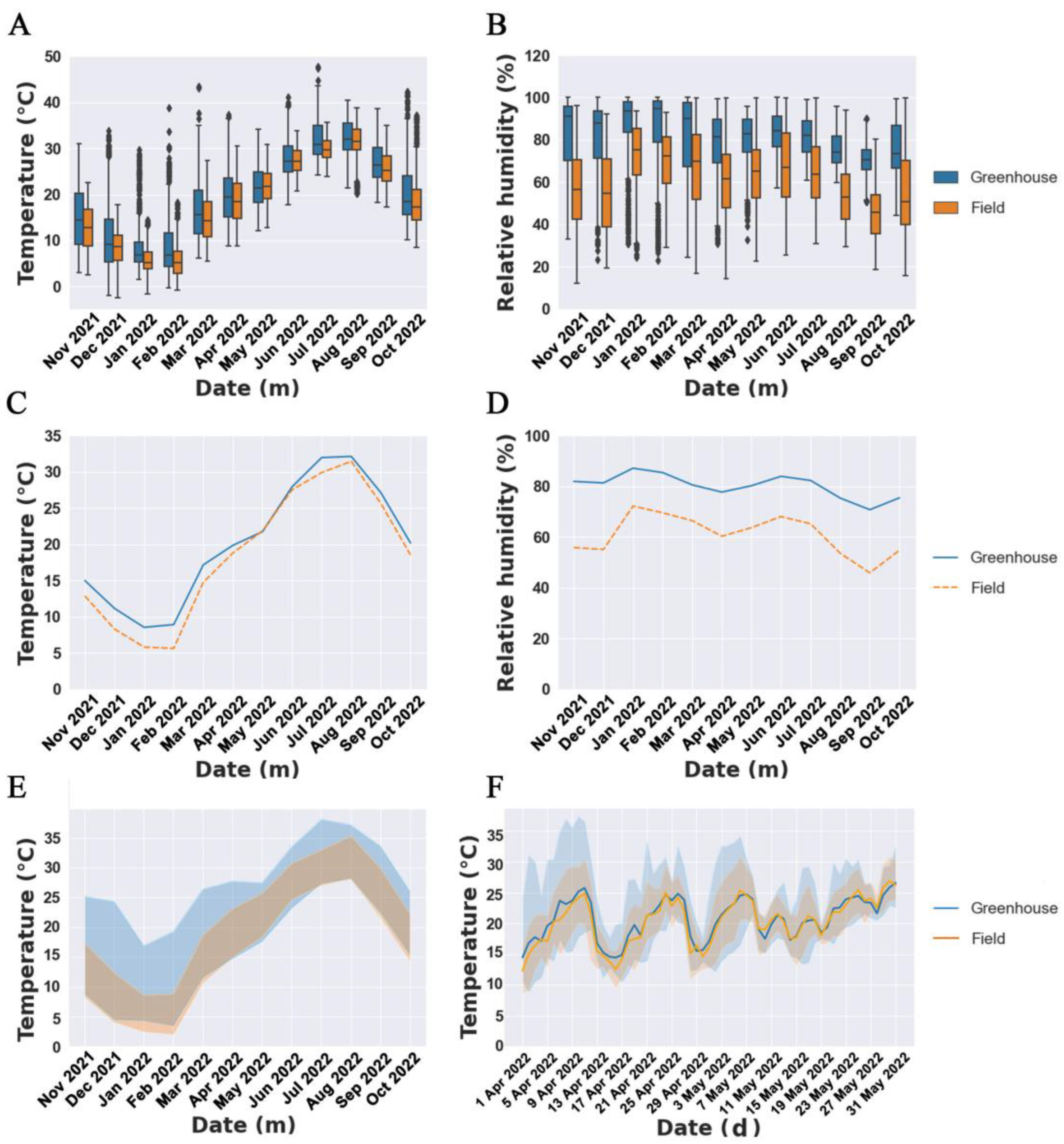
2.2. Climate Factors
2.3. Phenological Observation
2.4. Fruit Firmness and Yield
2.5. Fruit Quality
2.5.1. Sugar and Acid Determination
2.5.2. Athocyanin and Vc Determination
2.5.3. Preparation of Mulberry Fruit Extract
2.5.4. Total Polyphenols and Flavonoids Determination
2.5.5. Antioxidant Activity Analysis
2.6. Statistical Analysis
3. Results
3.1. Climate Factor Variation and Phenological Stages in Various Cultivation Modes
3.2. Fruit Firmness and Yield in Different Cultivation Modes
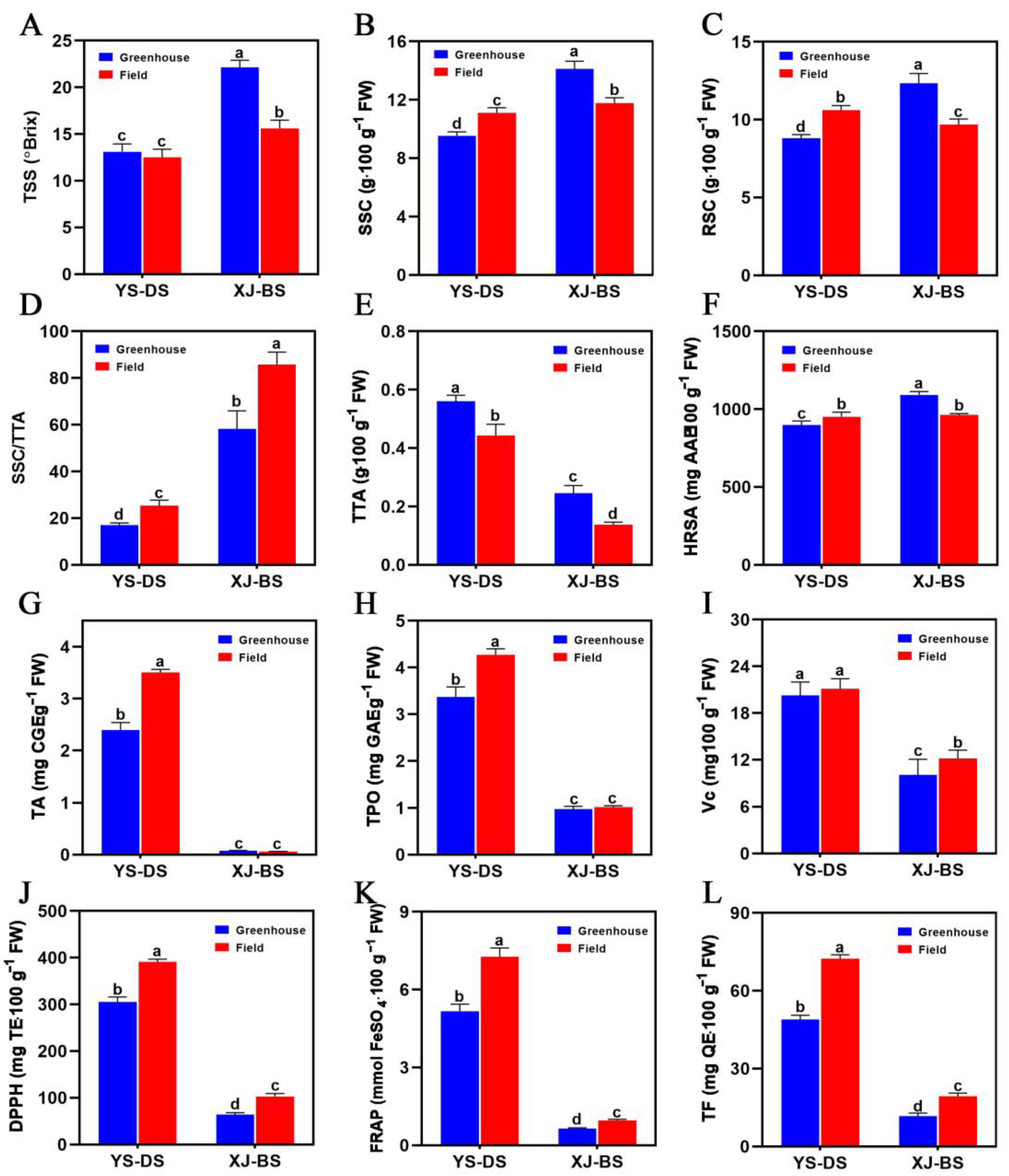
3.3. Fruit Quality across Different Cultivation Modes
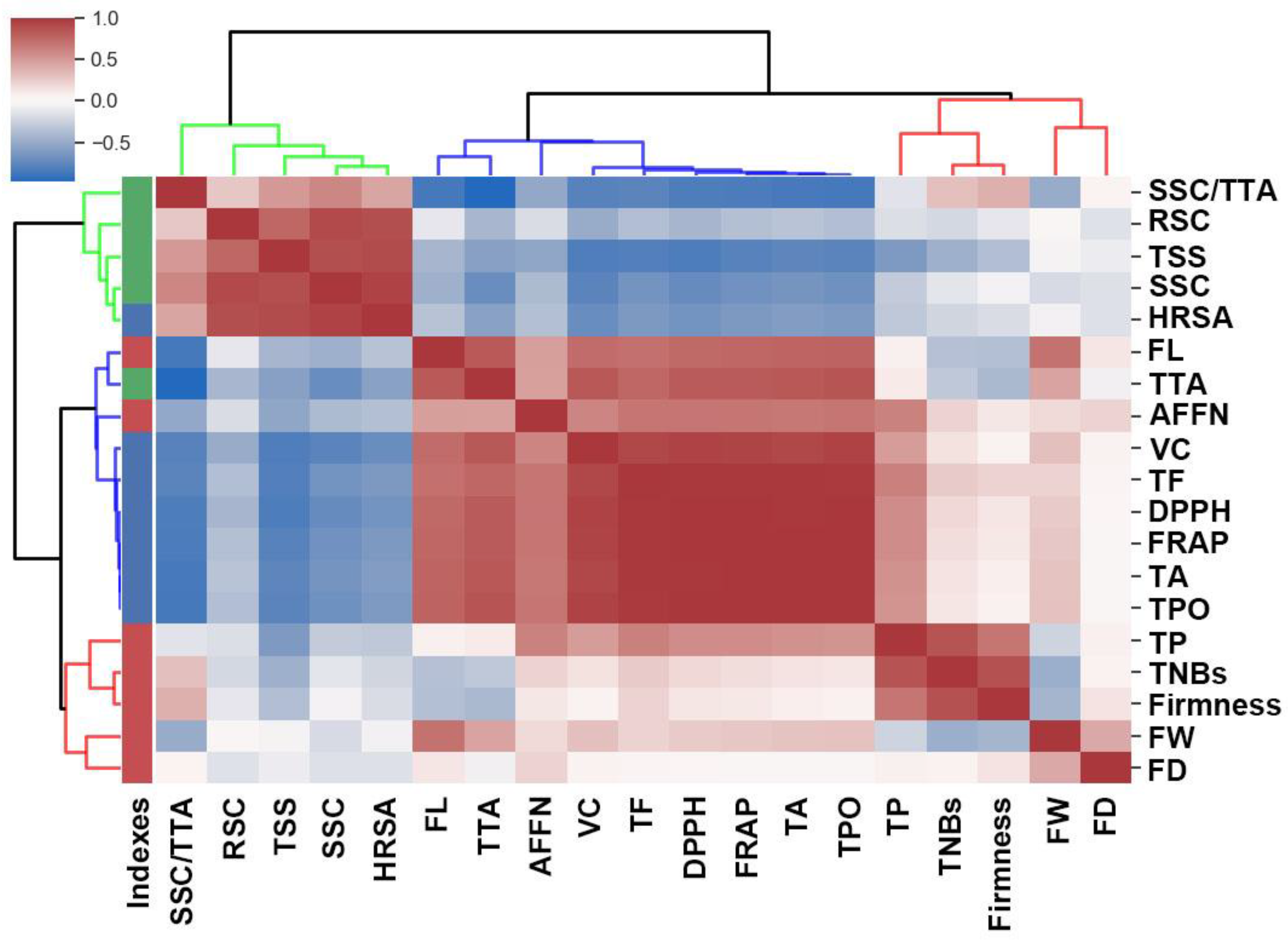
3.4. Correlation Analysis of Fruit Yield and Quality Traits
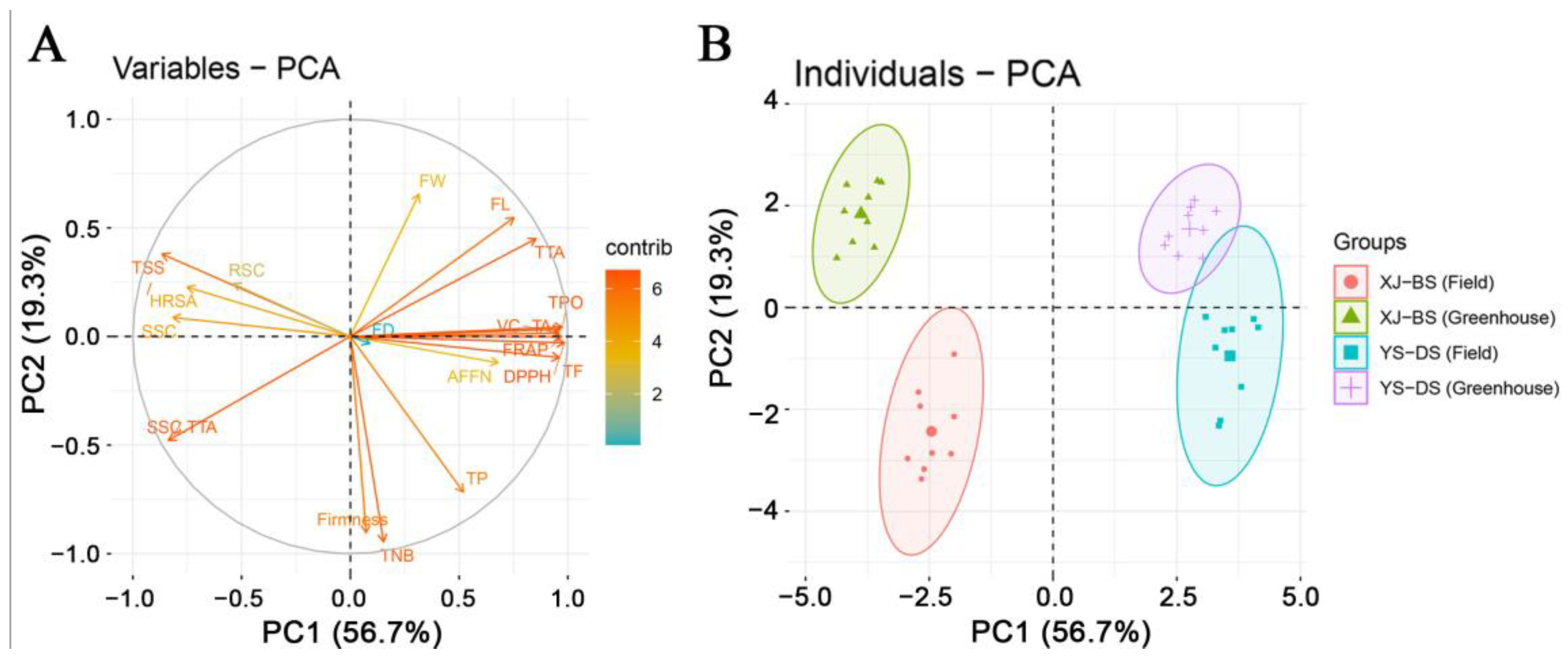
3.5. Principal Component Analysis (PCA) of Fruit Yield and Quality Traits
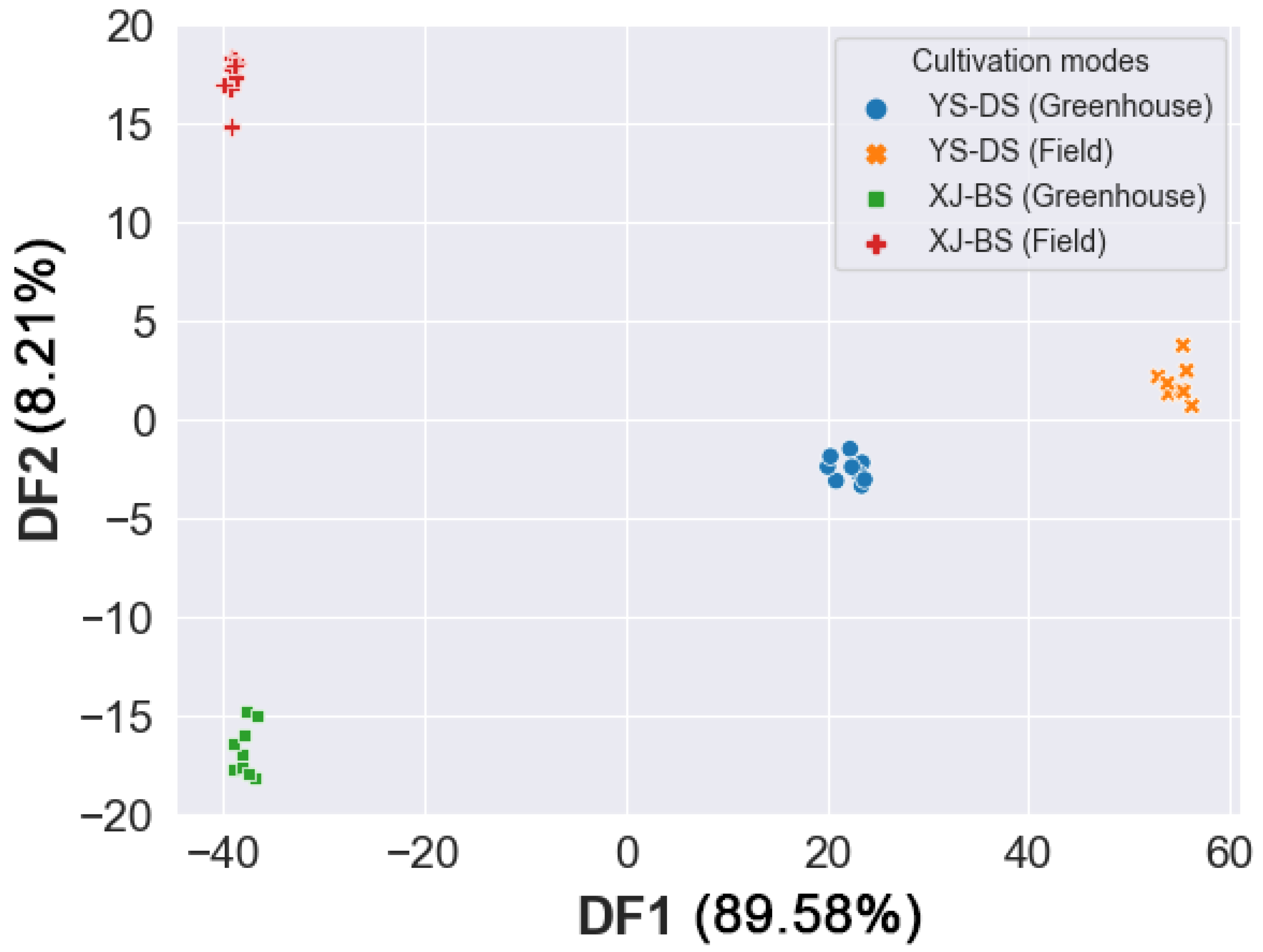
3.6. Linear Discriminant Analysis (LDA)
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Cui, J.; Li, R.H.; Zhang, L.Y.; Jing, Y. Spatially illustrating leisure agriculture: Empirical evidence from picking orchards in China. Land 2021, 10, 631. [Google Scholar] [CrossRef]
- Tao, C. Development Efficiency of Leisure Agriculture Based on DEA Model in the Background of Rural Revitalization. Rev. Cercet. Interv. Soc. 2019, 67, 169–187. [Google Scholar] [CrossRef]
- Liang, A.R.D.; Hsiao, T.Y.; Chen, D.J.; Lin, J.H. Agritourism: Experience design, activities, and revisit intention. Tour. Rev. 2021, 76, 1181–1196. [Google Scholar] [CrossRef]
- Gungor, N.; Sengul, M. Antioxidant activity, total phenolic content and selected physicochemical properties of white mulberry (Morus alba L.) fruits. Int. J. Food Prop. 2008, 11, 44–52. [Google Scholar] [CrossRef]
- Natić, M.M.; Dabić, D.Č.; Papetti, A.; Akšić, M.M.F.; Ognjanov, V.; Ljubojević, M.; Tešić, Ž.L. Analysis and characterisation of phytochemicals in mulberry (Morus alba L.) fruits grown in Vojvodina, North Serbia. Food Chem. 2015, 171, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Li, K.F.; Ni, L.; Yang, P.; Jia, H.J.; Lu, X.M. Preliminary study on trellis cultivation technology of mulberry. China Sericult. 2019, 40, 14–16. [Google Scholar]
- Xu, W.; Wang, F.; Wang, R.; Sui, Y.; Zhou, Z.; Xie, J.; Xiang, Z. Seasonal Characterization of the endophytic fungal microbiome of mulberry (Morus spp.) cultivars resistant and susceptible to sclerotiniosis. Microorganisms 2021, 9, 2052. [Google Scholar] [CrossRef]
- Shamshiri, R.R.; Jones, J.W.; Thorp, K.R.; Ahmad, D.; Man, H.C.; Taheri, S. Review of optimum temperature, humidity, and vapour pressure deficit for microclimate evaluation and control in greenhouse cultivation of tomato: A review. Int. Agrophys. 2018, 32, 287–302. [Google Scholar] [CrossRef]
- Liu, P.G.; Zhu, Y.; Ye, J.J.; Lin, T.B.; Lv, Z.Q.; Xu, Z.L.; Xu, L.S.; Chen, L.Y.; Wei, J. Biological characteristics, bioactive compounds, and antioxidant activities of off-season mulberry fruit. Front. Plant Sci. 2022, 13, 1034013. [Google Scholar] [CrossRef]
- Kandel, D.R.; Marconi, T.G.; Badillo-Vargas, I.E.; Enciso, J.; Zapata, S.D.; Lazcano, C.A.; Crosby, K.; Acila, C.A. Yield and fruit quality of high-tunnel tomato cultivars produced during the off-season in south Texas. Sci. Hortic. 2020, 272, 109582. [Google Scholar] [CrossRef]
- Ahn, M.G.; Kim, D.S.; Ahn, S.R.; Sim, H.S.; Kim, S. Characteristics and trends of strawberry cultivars throughout the cultivation season in a greenhouse. Horticulturae 2021, 7, 30. [Google Scholar] [CrossRef]
- Kim, H.C.; Kwon, T.O.; Bae, J.H.; Kim, T.C. Shoot growth characteristics and climatic factors in greenhouse cultivation of mulberry. J. Bio-Environ. Control 2012, 21, 74–78. [Google Scholar]
- Kim, H.Y.; Lee, J.Y.; Hwang, I.G.; Han, H.M.; Park, B.R.; Han, G.J.; Park, J.T. Analysis of Functional constituents of mulberries (Morus alba L.) cultivated in a greenhouse and open field during maturation. J. Korean Soc. Food Sci. Nutr. 2015, 44, 1588–1593. [Google Scholar] [CrossRef]
- Lee, J.Y.; Hwang, I.G.; Kim, H.Y.; Yoo, S.M.; Park, J.T. Quality characteristics of mulberry cultivated under greenhouse and open field conditions. J. Korean Soc. Food Sci. Nutr. 2014, 43, 1964–1968. [Google Scholar] [CrossRef][Green Version]
- Lee, J.Y.; Hwang, I.G.; Park, B.R.; Han, H.M.; Yoo, S.M.; Han, G.J.; Park, J.T.; Kim, H.Y. Physicochemical characteristics and antioxidant activities of mulberries by greenhouse and open field cultivation in maturation degrees. J. Korean Soc. Food Sci. Nutr. 2015, 44, 1476–1483. [Google Scholar] [CrossRef]
- Mo, R.L.; Zhang, N.; Hu, D.; Jin, Q.; Li, J.X.; Dong, Z.X.; Zhu, Z.X.; Li, Y.; Zhang, C.; Yu, C. Identification of phenological growth stages of four Morus species based on the extended BBCH-Scale and its application in fruit development with morphological profiles and color characteristics. Horticulturae 2022, 8, 1140. [Google Scholar] [CrossRef]
- Mo, R.L.; Zhang, N.; Zhou, Y.; Dong, Z.X.; Zhu, Z.X.; Li, Y.; Zhang, C.; Jin, Q.; Yu, C. Influence of eight rootstocks on fruit quality of Morus multicaulis cv.‘Zing’and the comprehensive evaluation of fruit quality traits. Not. Bot. Horti Agrobot. 2022, 50, 12598. [Google Scholar] [CrossRef]
- Morris, D.L. Quantitative determination of carbohydrates with Dreywood’s anthrone reagent. Science 1948, 107, 254–255. [Google Scholar] [CrossRef]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Giusti, M.M.; Wrolstad, R.E. Characterization and measurement of anthocyanins by UV–visible spectroscopy. Curr. Protoc. Food Anal. Chem. 2001, 1, F1.2.1–F1.2.12. [Google Scholar] [CrossRef]
- Zhang, W.H. Determination of ascorbic acid (vitamin C) content. In Experimental Principles and Techniques of Plant Physiology and Biochemistry; Li, H.S., Ed.; Higher Education Press: Beijing, China, 2000; pp. 246–249. [Google Scholar]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrate and antioxidants by means of Folin-Ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Lin, C.Y.; Lay, H.L. Characteristics of fruit growth, component analysis and antioxidant activity of mulberry (Morus spp.). Sci. Hortic. 2013, 162, 285–292. [Google Scholar] [CrossRef]
- Chen, H.; Chen, J.; Yang, H.; Chen, W.; Gao, H.; Lu, W. Variation in total anthocyanin, phenolic contents, antioxidant enzyme and antioxidant capacity among different mulberry (Morus sp.) cultivars in China. Sci. Hortic. 2016, 213, 186–192. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Waskom, M.L. Seaborn: Statistical data visualization. J. Open Source Softw. 2021, 6, 3021. [Google Scholar] [CrossRef]
- Sanders, A.R.; Bhongir, N.; vonHoldt, B.; Pellegrini, M. Association of DNA methylation with energy and fear-related behaviors in canines. Front. Psychol. 2022, 13, 1025494. [Google Scholar] [CrossRef] [PubMed]
- Baccichet, I.; Chiozzotto, R.; Spinardi, A.; Gardana, C.; Bassi, D.; Cirilli, M. Evaluation of a large apricot germplasm collection for fruit skin and flesh acidity and organic acids composition. Sci. Hortic. 2022, 294, 110780. [Google Scholar] [CrossRef]
- Yahiro, M. Dormancy and sprouting of winter buds of a mulberry grown in a greenhouse. Jap. J. Trop. Agric. 1975, 18, 189–193. [Google Scholar]
- Faust, M.; Erez, A.; Rowland, L.J.; Wang, S.Y.; Norman, H.A. Bud dormancy in perennial fruit trees: Physiological basis for dormancy induction, maintenance, and release. Hortscience 1997, 32, 623–629. [Google Scholar] [CrossRef]
- Horvath, D.P.; Anderson, J.V.; Chao, W.S.; Foley, M.E. Knowing when to grow: Signals regulating bud dormancy. Trends Plant Sci. 2003, 8, 534–540. [Google Scholar] [CrossRef]
- Lang, G.A. Dormancy–a new universal terminology. Hortscience 1987, 22, 817–820. [Google Scholar] [CrossRef]
- Alburquerque, N.; García-Montiel, F.; Carrillo, A.; Burgos, L. Chilling and heat requirements of sweet cherry cultivars and the relationship between altitude and the probability of satisfying the chill requirements. Environ. Exp. Bot. 2008, 64, 162–170. [Google Scholar] [CrossRef]
- Figàs, M.R.; Prohens, J.; Raigón, M.D.; Pereira-Dias, L.; Casanova, C.; García-Martínez, M.D.; Rosa, E.; Soler, E.; Plazas, M.; Soler, S. Insights into the adaptation to greenhouse cultivation of the traditional Mediterranean long shelf-life tomato carrying the alc mutation: A multi-trait comparison of landraces, selections, and hybrids in open field and greenhouse. Front. Plant Sci. 2018, 9, 1774. [Google Scholar] [CrossRef]
- Ozkan, B.; Fert, C.; Karadeniz, C.F. Energy and cost analysis for greenhouse and open-field grape production. Energy 2007, 32, 1500–1504. [Google Scholar] [CrossRef]
- Birgi, J.; Peri, P.L.; Gargaglione, V. Raspberries and gooseberries in south Patagonia: Production, fruit quality, morphology and phenology in two environmental conditions. Sci. Hortic. 2019, 258, 108574. [Google Scholar] [CrossRef]
- Sønsteby, A.; Myrheim, U.; Heiberg, N.; Heide, O.M. Production of high yielding red raspberry long canes in a Northern climate. Sci. Hortic. 2009, 121, 289–297. [Google Scholar] [CrossRef]
- Wada, T.; Ikeda, H.; Matsushita, K.; Kambara, A.; Hirai, H.; Abe, K. Effects of shading in summer on yield and quality of tomatoes grown on a single-truss system. J. Jpn. Soc. Hort. Sci. 2006, 75, 51–58. [Google Scholar] [CrossRef]
- Zha, Q.; Yin, X.; Yin, X.J.; Jiang, A.L. Colored shade nets can relieve abnormal fruit softening and premature leaf senescence of “Jumeigui” grapes during ripening under greenhouse conditions. Plants 2022, 11, 1227. [Google Scholar] [CrossRef]
- Woolf, A.B.; Ferguson, I.B.; Requejo-Tapia, L.C.; Boyd, L.; Laing, W.A.; White, A. Impact of sun exposure on harvest quality of ‘Hass’ avocado fruit. Rev. Chapingo Ser. Hortic. 1999, 5, 352–358. [Google Scholar]
- Qiao, L.; Cao, M.; Zheng, J.; Zhao, Y.; Zheng, Z.L. Gene coexpression network analysis of fruit transcriptomes uncovers a possible mechanistically distinct class of sugar/acid ratio-associated genes in sweet orange. BMC Plant Biol. 2017, 17, 186. [Google Scholar] [CrossRef]
- Marsh, K.B.; Richardson, A.C.; Macrae, E.A. Early- and mid-season temperature effects on the growth and composition of satsuma mandarins. J. Hortic. Sci. Biotechnol. 1999, 74, 443–451. [Google Scholar] [CrossRef]
- Gautier, Y.; Diakou-Verdin, V.; Bénard, C.; Reich, M.; Buret, M.; Bourgaud, F.; Reich, R.; Buret, M.; Bourgaud, F.; Poëssel, J.; et al. How does tomato quality (sugar, acid, and nutritional quality) vary with ripening stage, temperature, and irradiance? J. Agric. Food Chem. 2008, 56, 1241–1250. [Google Scholar] [CrossRef]
- Agüero, J.J.; Salazar, S.M.; Kirschbaum, D.S.; Jerez, E.F. Factors affecting fruit quality in strawberries grown in a subtropical environment. Int. J. Fruit Sci. 2015, 15, 223–234. [Google Scholar] [CrossRef]
- Wang, S.Y.; Camp, M.J. Temperatures after bloom affect plant growth and fruit quality of strawberry. Sci. Hortic. 2000, 85, 183–199. [Google Scholar] [CrossRef]
- Kliewer, W.M. Berry composition of Vitis vinifera cultivars as influenced by photo-and nycto-temperatures during maturation. J. Am. Soc. Hortic. Sci. 1973, 98, 153–159. [Google Scholar] [CrossRef]
- Huet, R.; Tisseau, M.A. Observation sur l’évolution de l’ananas après lacoupe. Fruits 1959, 14, 271–275. [Google Scholar]
- Zhao, X.X.; Gao, H.P.; Wang, Y.F.; Zheng, Z.L.; Bao, L.J.; Su, C.; Zhang, M.J.; Qian, Y.H. Changes in content of endogenous hormones during mulberry fruit development and its relationship with maturation. Acta Sericologica Sin. 2019, 45, 643–650. [Google Scholar]
- Dorey, E.; Fournier, P.; Léchaudel, M.; Tixier, P. A statistical model to predict titratable acidity of pineapple during fruit developing period responding to climatic variables. Sci. Hortic. 2016, 210, 19–24. [Google Scholar] [CrossRef]
- Zhang, H.X.; Ma, Z.F.; Luo, X.Q.; Li, X.L. Effects of mulberry fruit (Morus alba L.) consumption on health outcomes: A mini-review. Antioxidants 2018, 7, 69. [Google Scholar] [CrossRef]
- Ferreyra, M.L.F.; Serra, P.; Casati, P. Recent advances on the roles of flavonoids as plant protective molecules after UV and high light exposure. Physiol. Plant. 2021, 173, 736–749. [Google Scholar] [CrossRef] [PubMed]
- Grisebach, H. Biosynthesis of anthocyanins. In Anthocyanins as Food Colors; Markakis, P., Ed.; Academic Press: New York, NY, USA, 1982; pp. 47–67. [Google Scholar]
- Zha, Q.; Xi, X.J.; He, Y.N.; Jiang, A.L. Bagging affecting sugar and anthocyanin metabolism in the ripening period of grape berries. Not. Bot. Horti Agrobot. 2019, 47, 1194–1205. [Google Scholar] [CrossRef]
- Wang, S.Y.; Chen, C.T.; Wang, C.Y. The influence of light and maturity on fruit quality and flavonoid content of red raspberries. Food Chem. 2009, 112, 676–684. [Google Scholar] [CrossRef]
- Wang, F.; Wang, X.J.; Zhao, S.N.; Min, J.R.; Bo, X.; Zhang, Y.; Liu, Y.F.; Xu, T.; Qi, M.F.; Qi, H.Y.; et al. Light regulation of anthocyanin biosynthesis in horticultural crops. Sci. Agric. Sin. 2020, 53, 4904–4917. [Google Scholar]
- Gundogdu, M.; Muradoglu, F.; Sensoy, R.I.G.; Yilmaz, H. Determination of fruit chemical properties of Morus nigra L., Morus alba L. and Morus rubra L. by HPLC. Sci. Hortic. 2011, 132, 37–41. [Google Scholar] [CrossRef]
- Zhang, W.; Han, F.; He, J.; Duan, C. HPLC-DAD-ESI-MS/MS analysis and antioxidant activities of nonanthocyanin phenolics in mulberry (Morus alba L.). J. Food Sci. 2008, 73, C512–C518. [Google Scholar] [CrossRef] [PubMed]
- Hegedűs, A.; Balogh, E.; Engel, R.; Sipos, B.Z.; Papp, J.; Blázovics, A.; Stefanovits-Bányai, E. Comparative nutrient element and antioxidant characterization of berry fruit species and cultivars grown in Hungary. HortScience 2008, 43, 1711–1715. [Google Scholar] [CrossRef]
- Wang, R.S.; Dong, P.H.; Shuai, X.X.; Chen, M.S. Evaluation of different black mulberry fruits (Morus nigra L.) based on phenolic compounds and antioxidant activity. Foods 2022, 11, 1252. [Google Scholar] [CrossRef]
- Patanè, C.; Malvuccio, A.; Saita, A.; Rizzarelli, P.; Siracusa, L.; Rizzo, V.; Muratore, G. Nutritional changes during storage in fresh-cut long storage tomato as affected by biocompostable polylactide and cellulose based packaging. LWT–Food Sci. Technol. 2019, 101, 618–624. [Google Scholar] [CrossRef]
- Valko, M.; Rhodes, C.J.B.; Moncol, J.; Izakovic, M.M.; Mazur, M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem.-Biol. Interact. 2006, 160, 1–40. [Google Scholar] [CrossRef]
- Wang, W.; Li, X.W.; Bao, X.W.; Gao, L.; Tao, Y.X. Extraction of polysaccharides from black mulberry fruit and their effect on enhancing antioxidant activity. Int. J. Biol. Macromol. 2018, 120, 1420–1429. [Google Scholar] [CrossRef]
- Guo, Z.Y.; Xing, R.G.; Liu, S.; Yu, H.H.; Wang, P.B.; Li, C.P.; Li, P.C. The synthesis and antioxidant activity of the Schiff bases of chitosan and carboxymethyl chitosan. Bioorg. Med. Chem. Lett. 2005, 15, 4600–4603. [Google Scholar] [CrossRef]

| Cultivars | Cultivation Modes | Buds Elongating and Bursting | Full Flowering | Fruit Ripe for Picking |
|---|---|---|---|---|
| YS-DS | Greenhouse | 17 January 2022 | 10 March 2022 | 15 April 2022 |
| Field | 24 January 2022 | 15 March 2022 | 24 April 2022 | |
| XJ-BS | Greenhouse | 28 February 2022 | 14 March 2022 | 1 May 2022 |
| Field | 5 March 2022 | 27 March 2022 | 7 May 2022 |
| Cultivars | Cultivation Modes | FW (Grams per Fruit) | FL (Millimeters per Fruit) | FD (Millimeters per Fruit) | Firmness (Newtons) | TNBs (Number per Plant) | AFFN (Number per Bud) | TP (Kilograms per Plant) |
|---|---|---|---|---|---|---|---|---|
| YS-DS | Greenhouse | 3.83 ± 0.53 a | 33.16 ± 1.48 a | 14.37 ± 0.87 a | 0.53 ± 0.01 b | 276.44 ± 17.52 b | 5.98 ± 0.45 a | 6.32 ± 0.65 c |
| Field | 3.53 ± 0.49 a | 33.14 ± 1.44 a | 14.50 ± 0.66 a | 0.64 ± 0.02 a | 486.56 ± 82.49 a | 6.39 ± 0.54 a | 10.99 ± 2.14 a | |
| XJ-BS | Greenhouse | 3.59 ± 0.71 a | 28.98 ± 1.21 b | 14.12 ± 0.61 a | 0.54 ± 0.01 b | 250.11 ± 56.56 b | 5.48 ± 0.26 c | 4.92 ± 1.06 d |
| Field | 2.85 ± 0.59 b | 24.93 ± 3.01 c | 14.76 ± 1.91 a | 0.65 ± 0.03 a | 515.22 ± 69.97 a | 5.55 ± 0.34 c | 8.19 ± 1.42 b |
| Principal Component | Eigenvalues | Proportion of Variance | Cumulative Proportion |
|---|---|---|---|
| 1 | 3.28 | 56.74 | 56.74 |
| 2 | 1.92 | 19.35 | 76.09 |
| 3 | 1.37 | 9.93 | 86.02 |
| 4 | 1.16 | 7.11 | 93.13 |
| Indices | Principal Component One | Principal Component Two |
|---|---|---|
| TNBs | 1.17 | 15.74 |
| AFFN | 5.21 | 2.02 |
| TP | 3.98 | 11.93 |
| FW | 2.42 | 10.90 |
| FL | 5.76 | 9.12 |
| FD | 0.68 | 0.54 |
| Firmness | 0.56 | 15.05 |
| TSS | 6.63 | 6.32 |
| SSC | 6.25 | 1.45 |
| RSC | 4.12 | 4.03 |
| SSC/TTA | 6.41 | 7.97 |
| TTA | 6.54 | 7.49 |
| HRSA | 5.75 | 3.78 |
| TA | 7.39 | 0.55 |
| TPO | 7.46 | 0.73 |
| VC | 7.34 | 0.26 |
| DPPH | 7.54 | 0.49 |
| FRAP | 7.44 | 0.02 |
| TF | 7.36 | 1.62 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, N.; Li, J.; Qiu, C.; Wei, W.; Huang, S.; Li, Y.; Deng, W.; Mo, R.; Lin, Q. Multivariate Analysis of the Phenological Stages, Yield, Bioactive Components, and Antioxidant Capacity Effects in Two Mulberry Cultivars under Different Cultivation Modes. Horticulturae 2023, 9, 1334. https://doi.org/10.3390/horticulturae9121334
Zhang N, Li J, Qiu C, Wei W, Huang S, Li Y, Deng W, Mo R, Lin Q. Multivariate Analysis of the Phenological Stages, Yield, Bioactive Components, and Antioxidant Capacity Effects in Two Mulberry Cultivars under Different Cultivation Modes. Horticulturae. 2023; 9(12):1334. https://doi.org/10.3390/horticulturae9121334
Chicago/Turabian StyleZhang, Na, Jinxin Li, Changyu Qiu, Wei Wei, Sheng Huang, Yong Li, Wen Deng, Rongli Mo, and Qiang Lin. 2023. "Multivariate Analysis of the Phenological Stages, Yield, Bioactive Components, and Antioxidant Capacity Effects in Two Mulberry Cultivars under Different Cultivation Modes" Horticulturae 9, no. 12: 1334. https://doi.org/10.3390/horticulturae9121334
APA StyleZhang, N., Li, J., Qiu, C., Wei, W., Huang, S., Li, Y., Deng, W., Mo, R., & Lin, Q. (2023). Multivariate Analysis of the Phenological Stages, Yield, Bioactive Components, and Antioxidant Capacity Effects in Two Mulberry Cultivars under Different Cultivation Modes. Horticulturae, 9(12), 1334. https://doi.org/10.3390/horticulturae9121334





