Xylella fastidiosa, Possible New Threat to Chestnut (Castanea sativa Mill.) in Italy
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material, Symptom Observation and Diagnostic Analysis
2.2. MLST and Phylogenetic Analysis
2.3. FISH-CLSM
2.4. Possible Vectors of X. fastidiosa for This Species
3. Results
3.1. Symptom Observation and Diagnostic Analysis
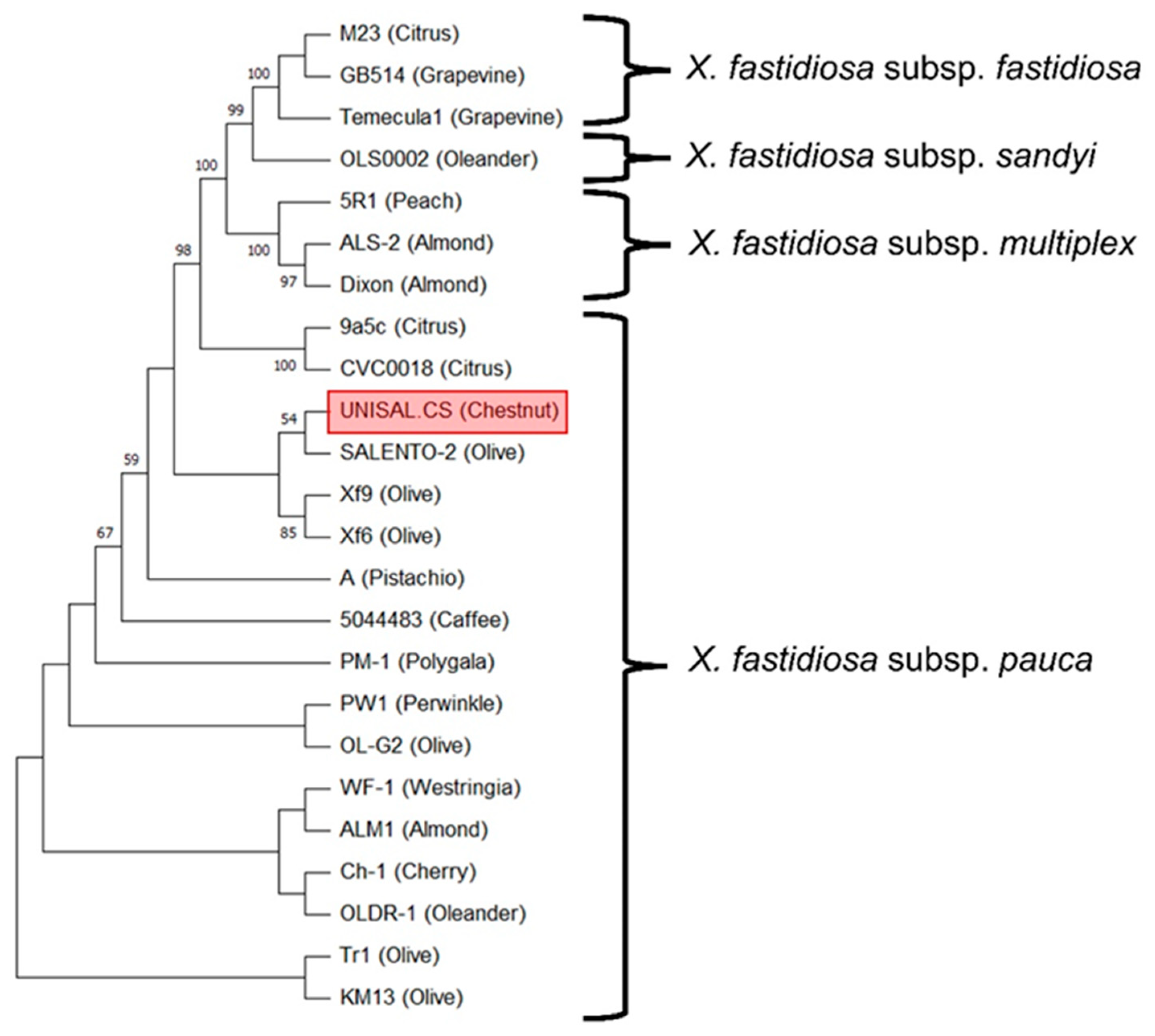
3.2. MLST and Phylogenetic Analysis
3.3. Xylella fastidiosa Distribution in C. sativa Xylem Vessels
3.4. Possible Vectors of X. fastidiosa for This Species
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Saponari, M.; Giampetruzzi, A.; Loconsole, G.; Boscia, D.; Saldarelli, P. Xylella fastidiosa in Olive in Apulia: Where We Stand. Phytopathology 2019, 109, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Picciotti, U.; Araujo Dalbon, V.; Ciancio, A.; Colagiero, M.; Cozzi, G.; De Bellis, L.; Finetti-Sialer, M.M.; Greco, D.; Ippolito, A.; Lahbib, N.; et al. “Ectomosphere”: Insects and Microorganism Interactions. Microorganisms 2023, 9, 440. [Google Scholar] [CrossRef] [PubMed]
- Loconsole, G.; Zicca, S.; Manco, L.; El Hatib, O.; Altamura, G.; Potere, O.; Elicio, V.; Valentini, F.; Boscia, D.; Saponari, M. Diagnostic Procedures to Detect Xylella fastidiosa in Nursery Stocks and Consignments of Plants for Planting. Agriculture 2021, 11, 922. [Google Scholar] [CrossRef]
- Saponari, M.; Boscia, D.; Nigro, F.; Martelli, G.P. Identification of DNA sequences related to Xylella fastidiosa in oleander, almond and olive trees exhibiting leaf scorch symptoms in Apulia (Southern Italy). J. Plant Pathol. 2013, 95, 3. [Google Scholar] [CrossRef]
- Martelli, G.P.; Boscia, D.; Porcelli, F.; Saponari, M. The olive quick decline syndrome in South-east Italy: A threatening phytosanitary emergency. Eur. J. Plant Pathol. 2016, 144, 235–243. [Google Scholar] [CrossRef]
- Greco, D.; Aprile, A.; De Bellis, L.; Luvisi, A. Diseases Caused by Xylella fastidiosa in Prunus Genus: An Overview of the Research on an Increasingly Widespread Pathogen. Front. Plant Sci. 2021, 13, 712452. [Google Scholar] [CrossRef]
- EFSA (European Food Safety Authority); Gibin, D.; Pasinato, L.; Delbianco, A. Scientific Report on the update of the Xylella spp. host plant database—Systematic literature search up to 31 December 2022. EFSA J. 2023, 21, 8061. [Google Scholar] [CrossRef]
- EFSA Panel on Plant Health (PLH); Bragard, C.; Dehnen-Schmutz, K.; Di Serio, F.; Gonthier, P.; Jacques, M.A.; Jaques Miret, J.A.; Justesen, A.F.; MacLeod, A.; Magnusson, C.S.; et al. Update of the Scientific Opinion on the risks to plant health posed by Xylella fastidiosa in the EU territory. EFSA J. 2019, 17, 5665. [Google Scholar] [CrossRef]
- EFSA (European Food Safety Authority). Statement of EFSA on host plants, entry and spread pathways and risk reduction options for Xylella fastidiosa Wells et al. EFSA J. 2013, 11, 3468. [Google Scholar] [CrossRef]
- EFSA (European Food Safety Authority). Scientific report on the update of the Xylella spp. host plant database. EFSA J. 2018, 16, 5408. [Google Scholar] [CrossRef]
- Hopkins, D.L.; Purcell, A.H. Xylella fastidiosa: Cause of Pierce’s Disease of Grapevine and Other Emergent Diseases. Plant Dis. 2002, 86, 1056–1066. [Google Scholar] [CrossRef] [PubMed]
- Regione Puglia, Piante Specificate Sensibili Alla Xylella fastidiosa Sottospecie pauca Riscontrate in Puglia. 2023. Available online: www.emergenzaxylella.it (accessed on 21 October 2023).
- Saponari, M.; Boscia, D.; Altamura, G.; Loconsole, G.; Zicca, S.; D’Attoma, G.; Morelli, M.; Palmisano, M.; Saponari, A.; Tavano, D.; et al. Isolation and pathogenicity of Xylella fastidiosa associated to the olive quick decline syndrome in Southern Italy. Sci. Rep. 2017, 7, 17723. [Google Scholar] [CrossRef] [PubMed]
- Vergine, M.; Pavan, S.; Negro, C.; Nicolì, F.; Greco, D.; Sabella, E.; Aprile, A.; Ricciardi, L.; De Bellis, L.; Luvisi, A. Phenolic characterization of olive genotypes potentially resistant to Xylella. J. Plant Interact. 2022, 17, 462–474. [Google Scholar] [CrossRef]
- White, S.M.; Bullock, J.M.; Hooftman, D.A.P.; Chapman, D.S. Modelling the spread and control of Xylella fastidiosa in the early stages of invasion in Apulia, Italy. Biol. Invasions 2017, 19, 1825–1837. [Google Scholar] [CrossRef] [PubMed]
- Conedera, M.; Tinner, W.; Krebs, P.; de Rigo, D.; Caudullo, G. Castanea sativa in Europe: Distribution, habitat, usage and threats. In European Atlas of Forest Tree Species; San-Miguel-Ayanz, J., de Rigo, D., Caudullo, G., Houston Durrant, T., Mauri, A., Eds.; European Commission: Luxembourg, 2016; p. e0125e0+. [Google Scholar]
- Beccaro, G.L.; Donno, D.; Lione, G.G.; De Biaggi, M.; Gamba, G.; Rapalino, S.; Riondato, I.; Gonthier, P.; Mellano, M.G. Castanea spp. Agrobiodiversity Conservation: Genotype Influence on Chemical and Sensorial Traits of Cultivars Grown on the Same Clonal Rootstock. Foods 2020, 9, 1062. [Google Scholar] [CrossRef] [PubMed]
- Alessandri, S.; Krznar, M.; Ajolfi, D.; Ramos Cabrer, A.M.; Pereira-Lorenzo, S.; Dondini, L. Genetic Diversity of Castanea sativa Mill. Accessions from the Tuscan-Emilian Apennines and Emilia Romagna Region (Italy). Agronomy 2020, 10, 1319. [Google Scholar] [CrossRef]
- Costantini, E.A.C.; Dazzi, C. The Soils of Italy, World Soils Book Series. Geol. Geomorphol. 2013, 3, 39–56. [Google Scholar] [CrossRef]
- Del Coco, L.; Migoni, D.; Girelli, C.R.; Angilè, F.; Scortichini, M.; Fanizzi, F.P. Soil and Leaf Ionome Heterogeneity in Xylella fastidiosa subsp. pauca-Infected, Non-Infected and Treated Olive Groves in Apulia, Italy. Plants 2020, 9, 760. [Google Scholar] [CrossRef]
- Official Bulletin of the Apulia Region (BURP) n. 15 of 29-01-2015. Available online: https://burp.regione.puglia.it/documents/20135/899904/DELIBERAZIONE+DEL+CONSIGLIO+REGIONALE+20+gennaio+2015%2C+n.293+%28id+4729422%29.pdf/aff953bd-db88-ffcc-c79c-c2fc9fa69f86?t=1622798897598 (accessed on 7 July 2023).
- European Commission Directorate-General for Health and Food Safety—Safety of the Food Chain Plant health. In Guidelines for the Survey of Xylella fastidiosa (Wells et al.) in the Union Territory; European Commission: Brussels, Belgium, 2015.
- Luvisi, A.; Aprile, A.; Sabella, E.; Vergine, M.; Nutricati, E.; Miceli, A.; Negro, C.; De Bellis, L. Xylella fastidiosa subsp. pauca (CoDiRO strain) infection in four olive (Olea europaea L.) cultivars: Profile of phenolic compounds in leaves and progression of leaf scorch symptoms. Phytopathol. Mediterr. 2017, 56, 259–273. [Google Scholar] [CrossRef]
- Loconsole, G.; Potere, O.; Boscia, D.; Altamura, G.; Djelouah, K.; Elbeaino, T.; Frascheri, D.; Lorusso, D.; Palmisano, F.; Pollastro, P.; et al. Detection of Xylella fastidiosa in olive trees by molecular and serological methods. J. Plant Pathol. 2014, 96, 7–14. [Google Scholar] [CrossRef]
- Harper, S.J.; Ward, L.I.; Clover, G.R.G. Development of LAMP and Real-Time PCR Methods for the Rapid Detection of Xylella fastidiosa for Quarantine and Field Applications. Phytopathology 2010, 100, 1282–1288. [Google Scholar] [CrossRef] [PubMed]
- D’Attoma, G.; Morelli, M.; Saldarelli, P.; Saponari, M.; Giampetruzzi, A.; Boscia, D.; Savino, V.N.; De La Fuente, L.; Cobine, P.A. Ionomic Differences between Susceptible and Resistant Olive Cultivars Infected by Xylella fastidiosa in the Outbreak Area of Salento, Italy. Pathogens 2019, 8, 272. [Google Scholar] [CrossRef]
- Yuan, X.; Morano, L.; Bromley, R.; Spring-Pearson, S.; Stouthamer, R.; Nunney, L. Multilocus sequence typing of Xylella fastidiosa causing Pierce’s disease and oleander leaf scorch in the United States. Phytopathology 2010, 100, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Higgins, D.G.; Gibson, T.J. CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar] [CrossRef] [PubMed]
- Elbeaino, T.; Valentini, F.; Abou Kubaa, R.; Moubarak, P.; Yaseen, T.; Digiaro, M. Multilocus sequence typing of Xylella fastidiosa isolated from olive affected by ‘olive quick decline syndrome’ in Italy. Phytopathol. Mediterr. 2014, 53, 533–542. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular Evolutionary Genetics Analysis version 11. Mol. Biol. Evol. 2021, 38, 3022–3027. [Google Scholar] [CrossRef] [PubMed]
- Cardinale, M.; Luvisi, A.; Meyer, J.B.; Sabella, E.; De Bellis, L.; Cruz, A.C.; Ampatzidis, Y.; Cherubini, P. Specific fluorescence in situ hybridization (FISH) test to highlight colonization of xylem vessels by Xylella fastidiosa in naturally infected olive trees (Olea europaea L.). Front. Plant Sci. 2018, 9, 431. [Google Scholar] [CrossRef]
- Sabella, E.; Luvisi, A.; Aprile, A.; Negro, C.; Vergine, M.; Nicolì, F.; Miceli, A.; De Bellis, L. Xylella fastidiosa induces differential expression of lignification related-genes and lignin accumulation in tolerant olive trees cv. Leccino. J. Plant. Phys. 2018, 220, 60–68. [Google Scholar] [CrossRef]
- Drosopoulos, S. New data on the nature and origin of colour polymorphism in the spittlebug genus Philaenus (Hemiptera: Aphorophoridae). Ann. Soc. Entomol. 2003, 39, 31–42. [Google Scholar] [CrossRef]
- Kaplan, C.; Turanlı, T.; Çeliker, N.M. Pest species in chestnut growing areas of Izmir and Manisa provinces and their economic importance. In Proceedings of the VIII International Scientific Agriculture Symposium, Jahorina, Bosnia and Herzegovina, 5–8 October 2017; pp. 756–762. [Google Scholar]
- Elbeaino, T.; Yaseen, T.; Valentini, F.; Ben Moussa, I.E.; Mazzoni, V.; D’onghia, A.M. Identification of three potential insect vectors of Xylella fastidiosa in Southern Italy. Phytopathol. Mediterr. 2014, 53, 328–332. [Google Scholar] [CrossRef]
- Lahbib, N.; Picciotti, U.; Bouhachem, S.; Garganese, F.; Porcelli, F. Morphs of Philaenus species, candidate Xylella fastidiosa vectors. Bull. Insectology 2022, 75, 197–209. [Google Scholar]
- Mazzoni, V. Contribution to the knowledge of the Auchenorrhyncha (Hemiptera Fulgoromorpha and Cicadomorpha) of Tuscany (Italy). Redia 2005, 88, 85–102. [Google Scholar]
- EFSA PLH Panel (EFSA Panel on Plant Health). Scientific Opinion on the risks to plant health posed by Xylella fastidiosa in the EU territory, with the identification and evaluation of risk reduction options. EFSA J. 2015, 13, 3989. [Google Scholar] [CrossRef]
- Demichelis, S.; Bosco, D. Host-plant relationship and life history of some Alebra species in Italy (Auchenorrhyncha: Cicadellide). Eur. J. Entomol. 1995, 92, 683–690. [Google Scholar]
- EFSA (European Food Safety Authority); Delbianco, A.; Gibin, D.; Pasinato, L.; Boscia, D.; Morelli, M. Update of the Xylella spp. host plant database—systematic literature search up to 30 June 2022. EFSA J. 2023, 21, 7726. [Google Scholar] [CrossRef]
- Gould, A.B.; Lashomb, J.H. Bacterial Leaf Scorch (BLS) of Shade Trees. Plant Health Instr. 2007. [Google Scholar] [CrossRef]
- Tkaczyk, M. Worldwide review of bacterial diseases of oaks (Quercus sp.) and their potential threat to trees in Central Europe. For. Int. J. For. Res. 2022, 96, 425–433. [Google Scholar] [CrossRef]
- Huang, Q.; Wenbin, L.; Hartung, J.S. Association of Xylella fastidiosa with leaf scorch in Japanese beech bonsai. Can. J. Plant Pathol. 2003, 25, 401–405. [Google Scholar] [CrossRef]
- EFSA Panel on Plant Health (PLH); Jeger, M.; Bragard, C.; Caffier, D.; Chatzivassiliou, E.; Dehnen-Schmutz, K.; Gilioli, G.; Grégoire, J.-C.; Jaques Miret, J.A.; MacLeod, A.; et al. Statement on susceptibility of Citrus spp., Quercus ilex and Vitis spp. to Xylella fastidiosa strain CoDiRO. EFSA J. 2016, 14, 4601. [Google Scholar] [CrossRef]
- Denancé, N.; Legendre, B.; Briand, M.; Olivier, V.; de Boisseson, C.; Poliakoff, F.; Jacques, M.A. Several subspecies and sequence types are associated with the emergence of Xylella fastidiosa in natural settings in France. Plant Pathol. 2017, 66, 1054–1064. [Google Scholar] [CrossRef]
- Nunney, L.; Yuan, X.; Bromley, R.E.; Stouthamer, R. Detecting genetic introgression: High levels of intersubspecific recombination found in Xylella fastidiosa in Brazil. Appl. Environ. Microbiol. 2012, 78, 4702–4714. [Google Scholar] [CrossRef] [PubMed]
- Nunney, L.; Ortiz, B.; Russell, S.A.; Ruiz Sànchez, R.; Stouthamer, R. The Complex Biogeography of the Plant Pathogen Xylella fastidiosa: Genetic Evidence of Introductions and Subspecific Introgression in Central America. PLoS ONE 2014, 9, e112463. [Google Scholar] [CrossRef] [PubMed]
- Krivanek, A.F.; Walker, M.A. Vitis resistance to Pierce’s disease is characterized by differential Xylella fastidiosa populations in stems and leaves. Phytopathology 2005, 95, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Nicolì, F.; Negro, C.; Nutricati, E.; Vergine, M.; Aprile, A.; Sabella, E.; Damiano, G.; De Bellis, L.; Luvisi, A. Accumulation of Azelaic Acid in Xylella fastidiosa-Infected Olive Trees: A Mobile Metabolite for Health Screening. Phytopathology 2019, 109, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Janse, J.; Obradovic, A. Xylella fastidiosa: Its biology, diagnosis, control and risks. J. Plant Pathol. 2010, 92, 35–148. [Google Scholar]
- Nunne, L.; Azad, H.; Stouthamer, R. An Experimental test of the Host-Plant Range of Nonrecombinant Strains of North American Xylella fastidiosa subsp. multiplex. Phytopathology 2019, 109, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Groves, R.L.; Chen, J.; Civerolo, E.L.; Freeman, M.W.; Viveros, M.A. Spatial Analysis of Almond Leaf Scorch Disease in the San Joaquin Valley of California: Factors Affecting Pathogen Distribution and Spread. Plant Dis. 2005, 89, 581–589. [Google Scholar] [CrossRef]
- Rapicavoli, J.N.; Blanco-Ulate, B.; Muszyński, A.; Figueroa-Balderas, R.; Morales-Cruz, A.; Azadi, P.; Dobruchowska, J.M.; Castro, C.; Cantu, D.; Roper, M.C. Lipopolysaccharide O-antigen delays plant innate immune recognition of Xylella fastidiosa. Nat. Commun. 2018, 9, 390. [Google Scholar] [CrossRef]
- Chatterjee, S.; Almeida, R.P.P.; Lindow, S. Living in two Worlds: The Plant and Insect Lifestyles of Xylella fastidiosa. Annu. Rev. Phytopathol. 2008, 46, 243–271. [Google Scholar] [CrossRef]
- Sun, Q.; Greve, L.C.; Labavitch, J.M. Polysaccharide compositions of intervessel pit membranes contribute to Pierce’s disease resistance of grapevines. Plant Physiol. 2011, 155, 1976–1987. [Google Scholar] [CrossRef][Green Version]
- Cornara, D.; Cavalieri, V.; Dongiovanni, C.; Altamura, G.; Palmisano, F.; Bosco, D.; Porcelli, F.; Almeida, R.P.P.; Saponari, M. Transmission of Xylella fastidiosa by naturally infected Philaenus spumarius (Hemiptera, Aphrophoridae) to different host plants. J. Appl. Entomol. 2017, 141, 80–87. [Google Scholar] [CrossRef]
- Gonella, E.; Picciau, L.; Pippinato, L.; Cavagna, B.; Alma, A. Host plant identification in the generalist xylem feeder Philaenus spumarius through gut content analysis. Entomol. Exp. Appl. 2020, 168, 890–899. [Google Scholar] [CrossRef]
- Martelli, G.P. The current status of the quick decline syndrome of olive in Southern Italy. Phytoparasitica 2016, 44, 1–10. [Google Scholar] [CrossRef]
- Cavalieri, V.; Altamura, G.; Fumarola, G.; di Carolo, M.; Saponari, M.; Cornara, D.; Bosco, D.; Dongiovanni, C. Transmission of Xylella fastidiosa subspecies pauca Sequence Type 53 by Different Insect Species. Insects 2019, 10, 324. [Google Scholar] [CrossRef] [PubMed]



| April | June | ||||
|---|---|---|---|---|---|
| Site of Analysis | Sampled Trees | Infection Incidence | cfu mL−1 | Infection Incidence | cfu mL−1 |
| Martano | 2 | 50% | 3.01 × 103 | 50% | 1.38 × 103 |
| Corigliano d’Otranto | 5 | 40% | 3.08 × 104 | 40% | 1.74 × 103 |
| Taviano | 8 | 25% | 4.61 × 102 | 0 | 0 |
| Average | 15 | 38% | 1.14 × 104 | 30% | 1.56 × 103 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Greco, D.; Sabella, E.; Carluccio, G.; Delle Donne, A.G.; De Bellis, L.; Luvisi, A. Xylella fastidiosa, Possible New Threat to Chestnut (Castanea sativa Mill.) in Italy. Horticulturae 2023, 9, 1315. https://doi.org/10.3390/horticulturae9121315
Greco D, Sabella E, Carluccio G, Delle Donne AG, De Bellis L, Luvisi A. Xylella fastidiosa, Possible New Threat to Chestnut (Castanea sativa Mill.) in Italy. Horticulturae. 2023; 9(12):1315. https://doi.org/10.3390/horticulturae9121315
Chicago/Turabian StyleGreco, Davide, Erika Sabella, Giambattista Carluccio, Angelo Giovanni Delle Donne, Luigi De Bellis, and Andrea Luvisi. 2023. "Xylella fastidiosa, Possible New Threat to Chestnut (Castanea sativa Mill.) in Italy" Horticulturae 9, no. 12: 1315. https://doi.org/10.3390/horticulturae9121315
APA StyleGreco, D., Sabella, E., Carluccio, G., Delle Donne, A. G., De Bellis, L., & Luvisi, A. (2023). Xylella fastidiosa, Possible New Threat to Chestnut (Castanea sativa Mill.) in Italy. Horticulturae, 9(12), 1315. https://doi.org/10.3390/horticulturae9121315









