Potato Aeroponics: Effects of Cultivar and Plant Origin on Minituber Production
Abstract
1. Introduction
2. Materials and Methods
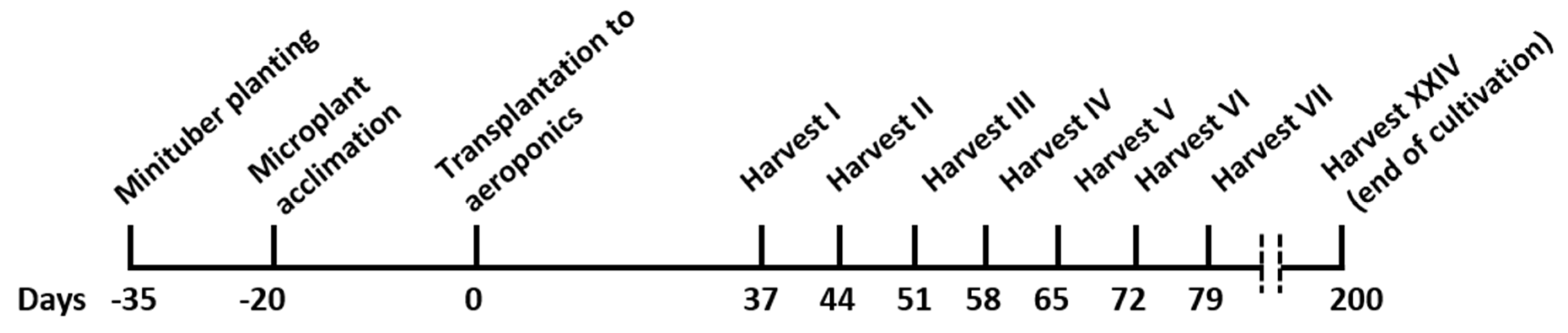
2.1. Plant Material and Establishment of In Vitro Culture
2.2. Preparation of Plants for Aeroponic Cultivation
2.3. Minituber Production in Aeroponic System
2.4. Statistical Analysis
3. Results
3.1. Potato Cultivation in an Aeroponic System
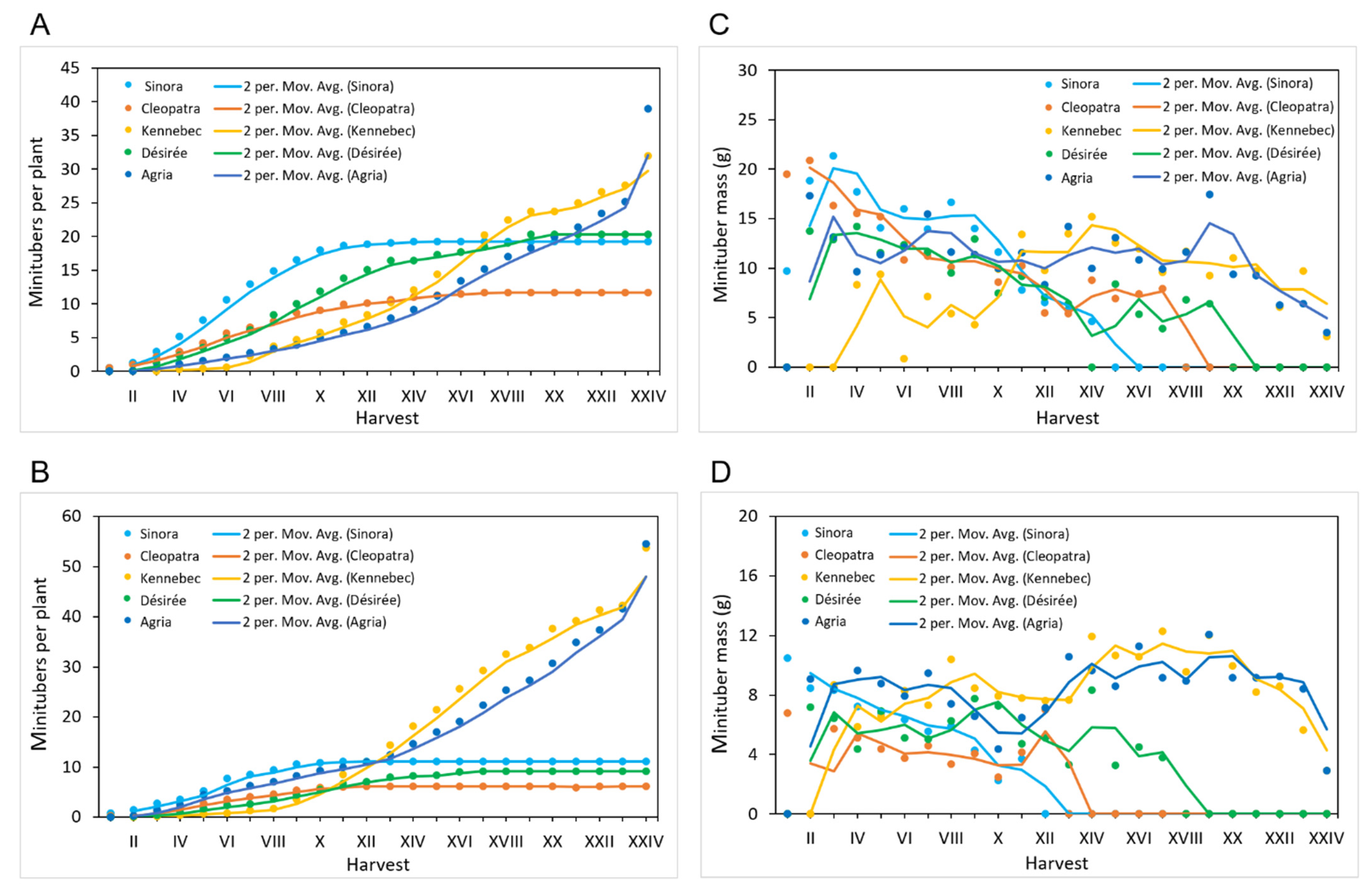
3.2. Dynamics of Minituber Formation
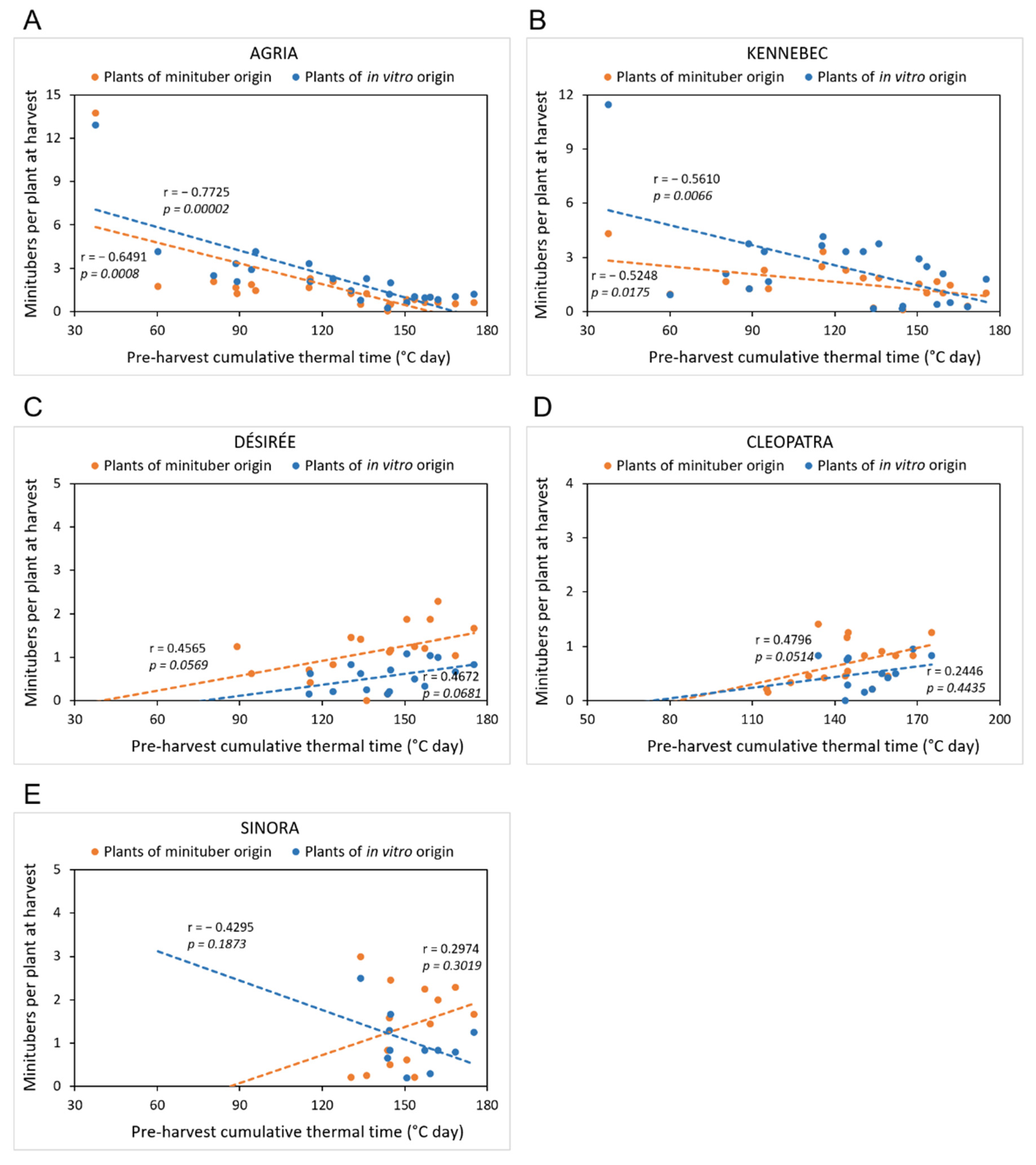
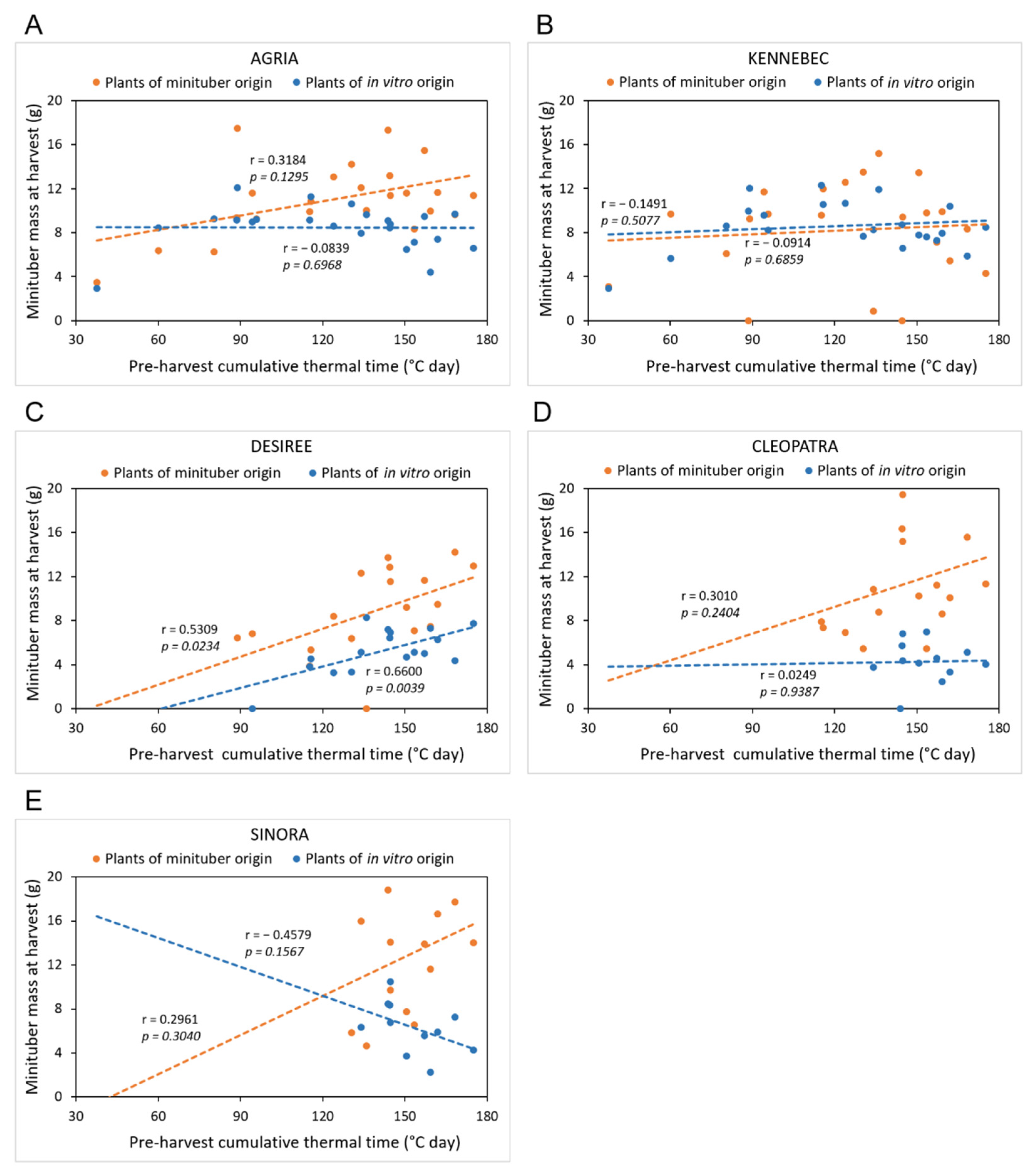
3.3. Effects of Cultivar and Plant Origin on Minituber Production
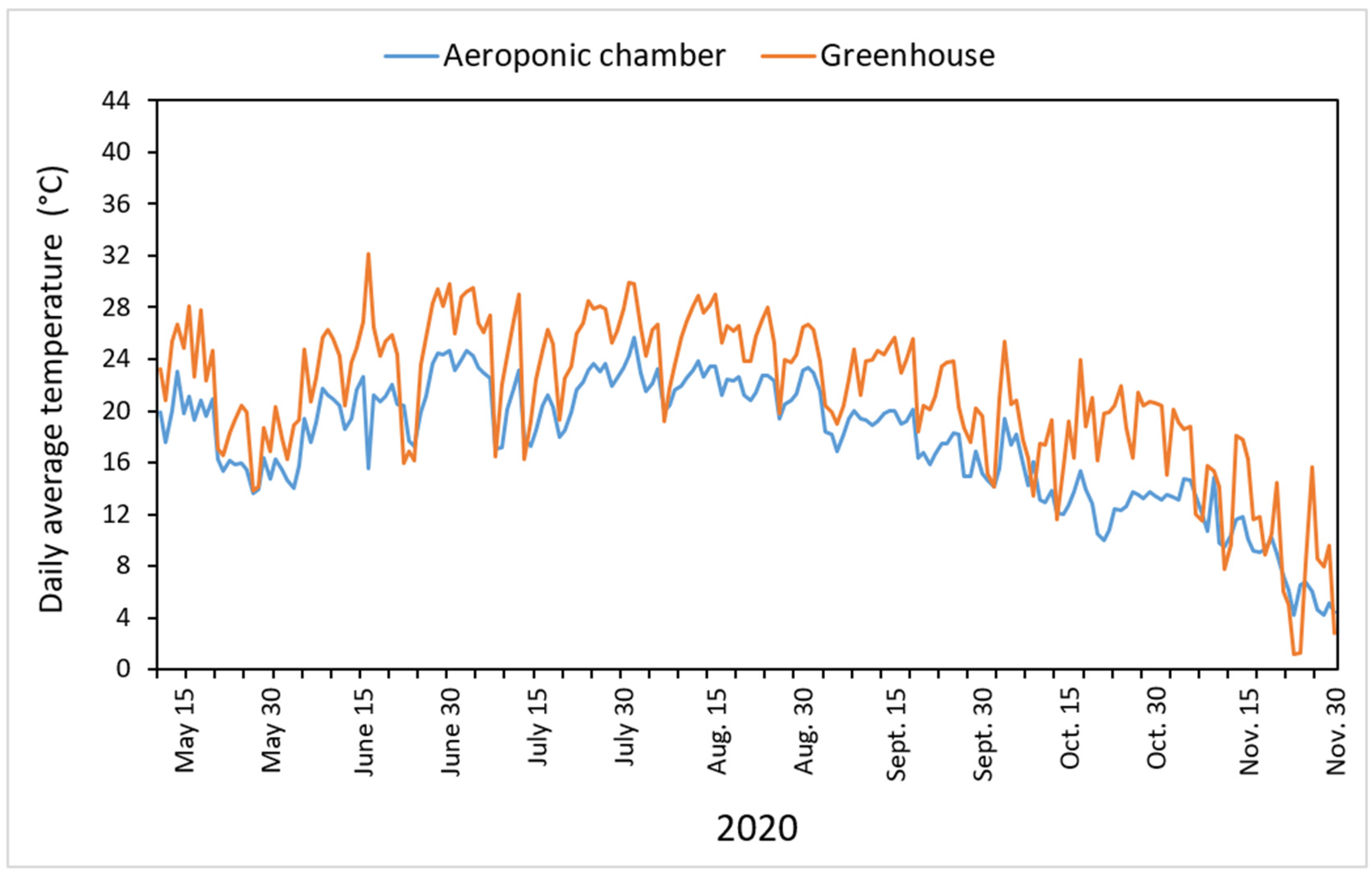
3.4. Effects of Temperature in the Aeroponic Module on Tuber Formation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Food and Agriculture Organization of the United Nations—FAOSTAT. Available online: http://www.fao.org/faostat/en/#data (accessed on 7 September 2022).
- Devaux, A.; Goffart, J.P.; Kromann, P.; Andrade-Piedra, J.; Polar, V.; Hareau, G. The potato of the future: Opportunities and challenges in sustainable agri-food systems. Potato Res. 2021, 64, 681–720. [Google Scholar] [CrossRef] [PubMed]
- Thomas-Sharma, S.; Abdurahman, A.; Ali, S.; Andrade-Piedra, J.L.; Bao, S.; Charkowski, A.O.; Crook, D.; Kadian, M.; Kromann, P.; Struik, P.C.; et al. Seed degeneration in potato: The need for an integrated seed health strategy to mitigate the problem in developing countries. Plant Pathol. 2016, 65, 3–16. [Google Scholar] [CrossRef]
- Broćić, Z.; Momčilović, I.; Poštić, D.; Oljača, J.; Veljković, B. Production of High-Quality Seed Potato by Aeroponics. In The Potato Crop: Management, Production, and Food Security; Villa, P.M., Ed.; Nova Science Publishers: New York, NY, USA, 2021; pp. 25–59. [Google Scholar] [CrossRef]
- Buckseth, T.; Sharma, A.K.; Pandey, K.K.; Singh, B.P.; Muthuraj, R. Methods of pre-basic seed potato production with special reference to aeroponics—A review. Sci. Hortic. 2016, 204, 79–87. [Google Scholar] [CrossRef]
- Struik, P.C. The canon of potato science: 25. Minitubers. Potato Res. 2007, 50, 305–308. [Google Scholar] [CrossRef]
- Broćić, Z.; Milinković, M.; Momčilović, I.; Oljača, J.; Veljković, B.; Milošević, D.; Poštić, D. Comparison of aeroponics and conventional production system of virus-free potato mini tubers in Serbia. Agro-Knowl. J. 2019, 20, 95–105. [Google Scholar] [CrossRef]
- Mateus-Rodriguez, J.R.; de Haan, S.; Andrade-Piedra, J.L.; Maldonado, L.; Hareau, G.; Barker, I.; Chuquillanqui, C.; Otazú, V.; Frisancho, R.; Bastos, C.; et al. Technical and economic analysis of aeroponics and other systems for potato mini-tuber production in Latin America. Am. J. Potato Res. 2013, 90, 357–368. [Google Scholar] [CrossRef]
- Otazú, V. Manual on Quality Seed Potato Production Using Aeroponics; International Potato Center (CIP): Lima, Peru, 2010; pp. 1–44. [Google Scholar]
- da Silva Filho, J.B.; Fontes, P.C.R.; Cecon, P.R.; McGiffen, M.E. Evaluation of “UFV aeroponic system” to produce basic potato seed minitubers. Am. J. Potato Res. 2018, 95, 443–450. [Google Scholar] [CrossRef]
- Gopinath, P.; Vethamoni, P.I.; Gomathi, M. Aeroponics soilless cultivation system for vegetable crops. Chem. Sci. Rev. Lett 2017, 6, 838–849. [Google Scholar]
- Rykaczewska, K. The potato minituber production from microtubers in aeroponic culture. Plant Soil Environ. 2016, 62, 210–214. [Google Scholar] [CrossRef]
- Muthoni, J.; Mbiyu, M.; Lung’aho, C.; Otieno, S.; Pwaipwai, P. Performance of two potato cultivars derived from in-vitro plantlets, mini-tubers and stem cuttings using aeroponics technique. Int. J. Hortic. 2017, 7, 246–249. [Google Scholar] [CrossRef]
- Broćić, Z.; Milinković, M.; Momčilović, I.; Poštić, D.; Oljača, J.; Veljković, B.; Milošević, D. Effect of the variety and origin of plants on the production of virus-free potato minitubers in the aeroponic growing system. J. Proc. Ener. Agri. 2019, 23, 147–149. [Google Scholar] [CrossRef]
- da Silva Filho, J.B.; Fontes, P.C.R.; Cecon, P.R.; Ferreira, J.F.; McGiffen, M.E.; Montgomery, J.F. Yield of potato minitubers under aeroponics, optimized for nozzle type and spray direction. HortScience 2020, 55, 14–22. [Google Scholar] [CrossRef]
- Farran, I.; Mingo-Castel, A.M. Potato minituber production using aeroponics: Effect of plant density and harvesting intervals. Am. J. Potato Res. 2006, 83, 47–53. [Google Scholar] [CrossRef]
- Çalışkan, M.E.; Yavuz, C.; Yağız, A.K.; Demirel, U.; Çalışkan, S. Comparison of aeroponics and conventional potato mini tuber production systems at different plant densities. Potato Res. 2020, 64, 41–52. [Google Scholar] [CrossRef]
- Chang, D.C.; Park, C.S.; Kim, S.Y.; Kim, S.J.; Lee, Y.B. Physiological growth responses by nutrient interruption in aeroponically grown potatoes. Am. J. Potato Res. 2008, 85, 315–323. [Google Scholar] [CrossRef]
- Mbiyu, M.W.; Muthoni, J.; Kabira, J.; Elmar, G.; Muchira, C.; Pwaipwai, P.; Onditi, J. Use of aeroponics technique for potato (Solanum tuberosum) minitubers production in Kenya. J. Hortic. For. 2012, 4, 172–177. [Google Scholar] [CrossRef]
- Momčilović, I.; Pantelić, D.; Hfidan, M.; Savić, J.; Vinterhalter, D. Improved procedure for detection of superoxide dismutase isoforms in potato, Solanum tuberosum L. Acta Physiol. Plant. 2014, 36, 2059–2066. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Linsmaier, E.M.; Skoog, F. Organic growth factor requirements of tobacco tissue cultures. Physiol. Plant. 1965, 18, 100–127. [Google Scholar] [CrossRef]
- Broćić, Z. Tehničko rešenje aeroponik sistema za proizvodnju bezvisusnih mini krtola krompira u Srbiji. J. Proc. Ener. Agri. 2019, 23, D2–D5. [Google Scholar]
- Broćić, Z.; Milinković, M.; Momčilović, I.; Poštić, D.; Oljača, J.; Veljković, B.; Milošević, D. Production of potato mini-tubers in the aeroponic growing system. J. Proc. Ener. Agri. 2018, 22, 49–52. [Google Scholar] [CrossRef]
- Broćić, Z.; Stefanović, R. Potato Production, and Market Economics; Faculty of Agriculture: Belgrade, Serbia, 2012; pp. 1–408. [Google Scholar]
- Mateus-Rodriguez, J.F.; De Haan, S.; Rodríguez-Delfín, A. Genotype by environment effects on potato mini-tuber seed production in an aeroponics system. Agronomy 2014, 4, 514–528. [Google Scholar] [CrossRef]
- Tierno, R.; Carrasco, A.; Ritter, E.; de Galarreta, J.I.R. Differential growth response and minituber production of three potato cultivars under aeroponics and greenhouse bed culture. Am. J. Potato Res. 2014, 91, 346–353. [Google Scholar] [CrossRef]
- Lommen, W.J.M.; Struik, P.C. Production of potato minitubers by repeated harvesting: Effects of crop husbandry on yield parameters. Potato Res. 1992, 35, 419–432. [Google Scholar] [CrossRef]
- Hancock, R.D.; Morris, W.L.; Ducreux, L.J.; Morris, J.A.; Usman, M.; Verrall, S.R.; Fuller, J.; Simpson, C.G.; Zhang, R.; Hedley, P.E.; et al. Physiological, biochemical and molecular responses of the potato (Solanum tuberosum L.) plant to moderately elevated temperature. Plant Cell Environ. 2014, 37, 439–450. [Google Scholar] [CrossRef] [PubMed]
- Pantelić, D.; Dragićević, I.Č.; Rudić, J.; Fu, J.; Momčilović, I. Effects of high temperature on in vitro tuberization and accumulation of stress-responsive proteins in potato. Hortic. Environ. Biotechnol. 2018, 59, 315–324. [Google Scholar] [CrossRef]
- Struik, P.C. Responses of the potato plant to temperature. In Potato Biology and Biotechnology, 1st ed.; Vreugdenhil, D., Bradshaw, J., Gebhardt, C., Govers, F., Taylor, M., MacKerron, D., Ross, H., Eds.; Elsevier, B.V.: Amsterdam, The Netherlands, 2007; pp. 367–393. [Google Scholar] [CrossRef]

| Parameter | Factor | df | SS | MS | F | p | Sig. |
|---|---|---|---|---|---|---|---|
| Number of minitubers per plant | Cultivar | 4 | 7151.313 | 1787.828 | 109.663 | 2.63 × 10−13 | *** |
| Plant origin | 1 | 74.419 | 74.419 | 4.565 | 4.52 × 10−2 | * | |
| Cultivar × Plant origin | 4 | 1569.557 | 392.389 | 24.069 | 2.10 × 10−7 | *** | |
| Minituber mass | Cultivar | 4 | 68.338 | 17.084 | 8.505 | 3.53 × 10−4 | *** |
| Plant origin | 1 | 145.332 | 145.332 | 72.350 | 4.46 × 10−8 | *** | |
| Cultivar × Plant origin | 4 | 58.251 | 14.563 | 7.250 | 8.88 × 10−4 | *** | |
| Yield per m2 | Cultivar | 4 | 244.702 | 61.176 | 60.483 | 6.90 × 10−11 | *** |
| Plant origin | 1 | 9.905 | 9.905 | 9.792 | 5.28 × 10−3 | ** | |
| Cultivar × Plant origin | 4 | 78.390 | 19.598 | 19.376 | 1.18 × 10−6 | *** |
| Parameter | Levene’s Statistic | df1 | df2 | p | Sig. |
|---|---|---|---|---|---|
| Number of minitubers per plant | 2.475 | 9 | 20 | 0.068 | ns |
| Minituber mass | 0.833 | 9 | 20 | 0.595 | ns |
| Yield per m2 | 1.845 | 9 | 20 | 0.147 | ns |
| Parameter | Factor | df | SS | MS | F | p | Sig. |
|---|---|---|---|---|---|---|---|
| Number of minitubers per plant | Between groups | 9 | 8795.288 | 977.254 | 59.943 | 0.000 | *** |
| Within groups | 20 | 326.060 | 16.303 | ||||
| Total | 29 | 9121.348 | |||||
| Minituber mass | Between groups | 9 | 271.920 | 30.213 | 15.041 | 0.000 | *** |
| Within groups | 20 | 40.175 | 2.009 | ||||
| Total | 29 | 312.095 | |||||
| Yield per m2 | Between groups | 9 | 332.941 | 36.993 | 36.568 | 0.000 | *** |
| Within groups | 20 | 20.233 | 1.012 | ||||
| Total | 29 | 353.173 |
| Cultivar | Plant Origin | Minitubers per Plant | Minituber Mass (g) | Yield per m2 (kg) |
|---|---|---|---|---|
| Sinora | Minituber | 19.33 bc | 15.48 a | 7.17 cd |
| In vitro | 11.16 ab | 8.07 cde | 2.17 ab | |
| Cleopatra | Minituber | 11.75 ab | 12.28 ab | 3.43 ab |
| In vitro | 6.25 a | 4.54 e | 0.69 a | |
| Désirée | Minituber | 20.37 bc | 9.51 bcd | 4.61 bc |
| In vitro | 9.16 ab | 5.69 de | 1.23 a | |
| Kennebec | Minituber | 28.58 cd | 10.57 bc | 7.32 cd |
| In vitro | 53.79 e | 8.13 cde | 10.50 e | |
| Agria | Minituber | 39.08 d | 8.17 cde | 7.56 d |
| In vitro | 54.50 e | 7.57 cde | 9.76 de |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Broćić, Z.; Oljača, J.; Pantelić, D.; Rudić, J.; Momčilović, I. Potato Aeroponics: Effects of Cultivar and Plant Origin on Minituber Production. Horticulturae 2022, 8, 915. https://doi.org/10.3390/horticulturae8100915
Broćić Z, Oljača J, Pantelić D, Rudić J, Momčilović I. Potato Aeroponics: Effects of Cultivar and Plant Origin on Minituber Production. Horticulturae. 2022; 8(10):915. https://doi.org/10.3390/horticulturae8100915
Chicago/Turabian StyleBroćić, Zoran, Jasmina Oljača, Danijel Pantelić, Jelena Rudić, and Ivana Momčilović. 2022. "Potato Aeroponics: Effects of Cultivar and Plant Origin on Minituber Production" Horticulturae 8, no. 10: 915. https://doi.org/10.3390/horticulturae8100915
APA StyleBroćić, Z., Oljača, J., Pantelić, D., Rudić, J., & Momčilović, I. (2022). Potato Aeroponics: Effects of Cultivar and Plant Origin on Minituber Production. Horticulturae, 8(10), 915. https://doi.org/10.3390/horticulturae8100915








