Cyanobacteria: A Natural Source for Controlling Agricultural Plant Diseases Caused by Fungi and Oomycetes and Improving Plant Growth
Abstract
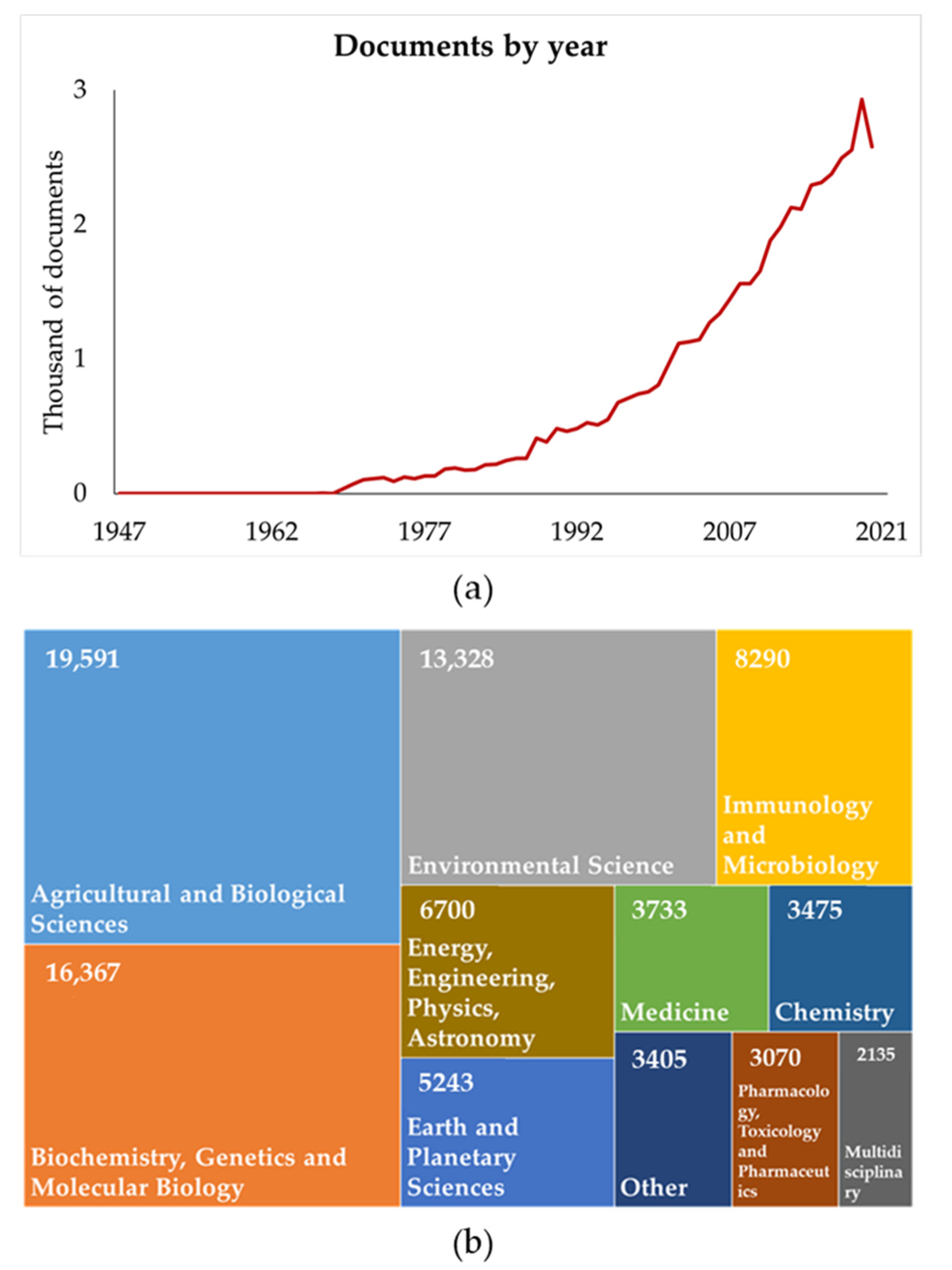
1. Introduction
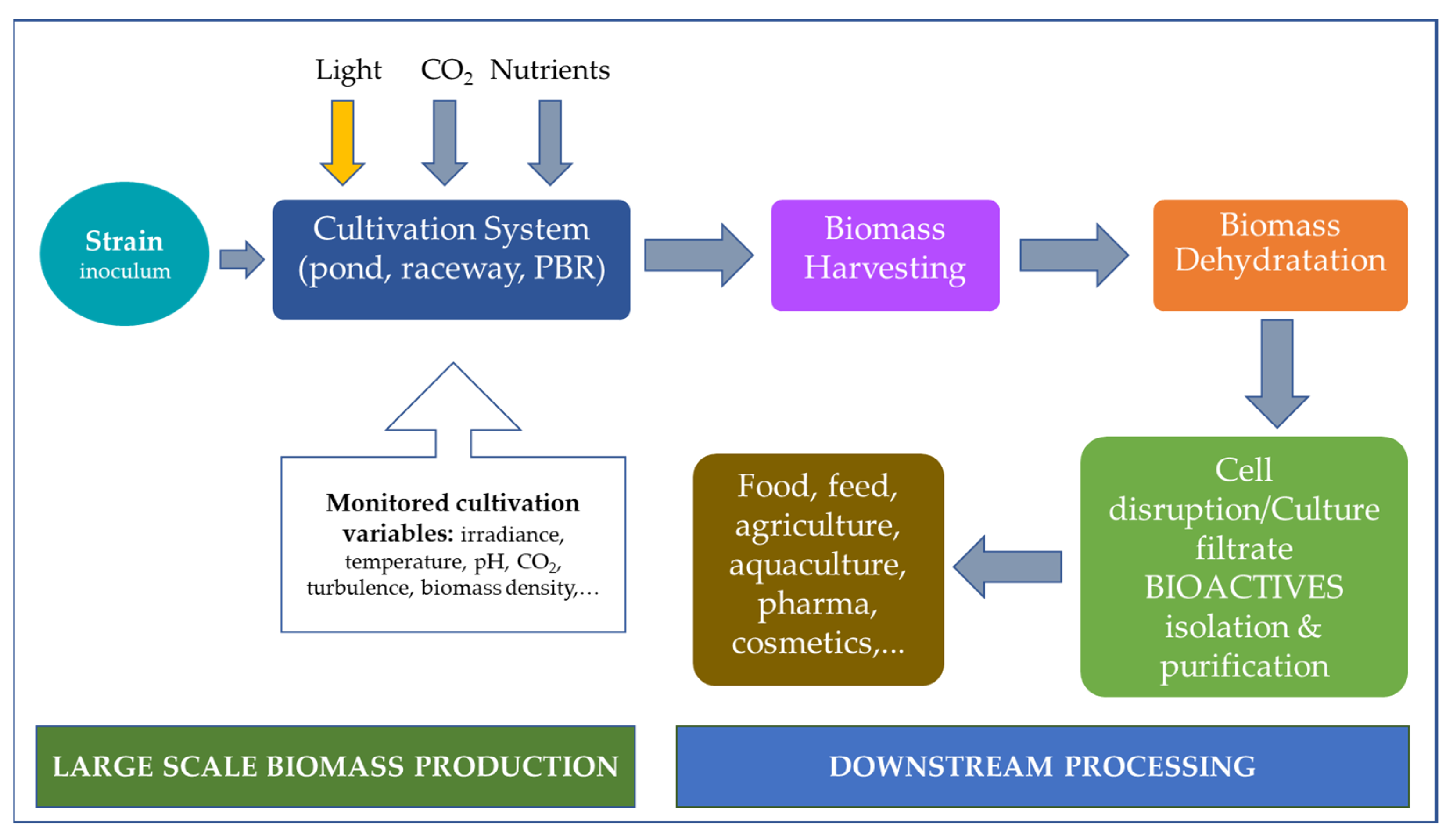
2. Cultivation, Growth, and Extraction Methods
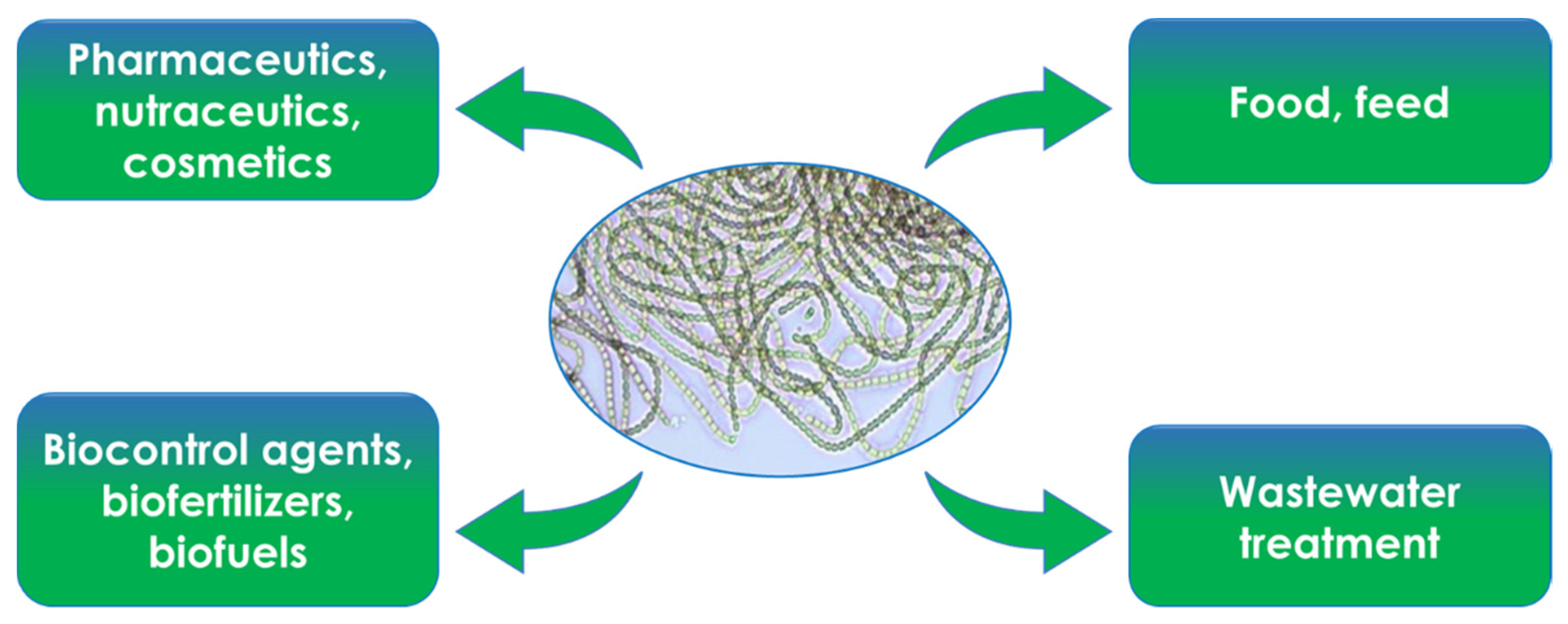
3. General Uses
4. Toxins
5. Plant Pathogen Control
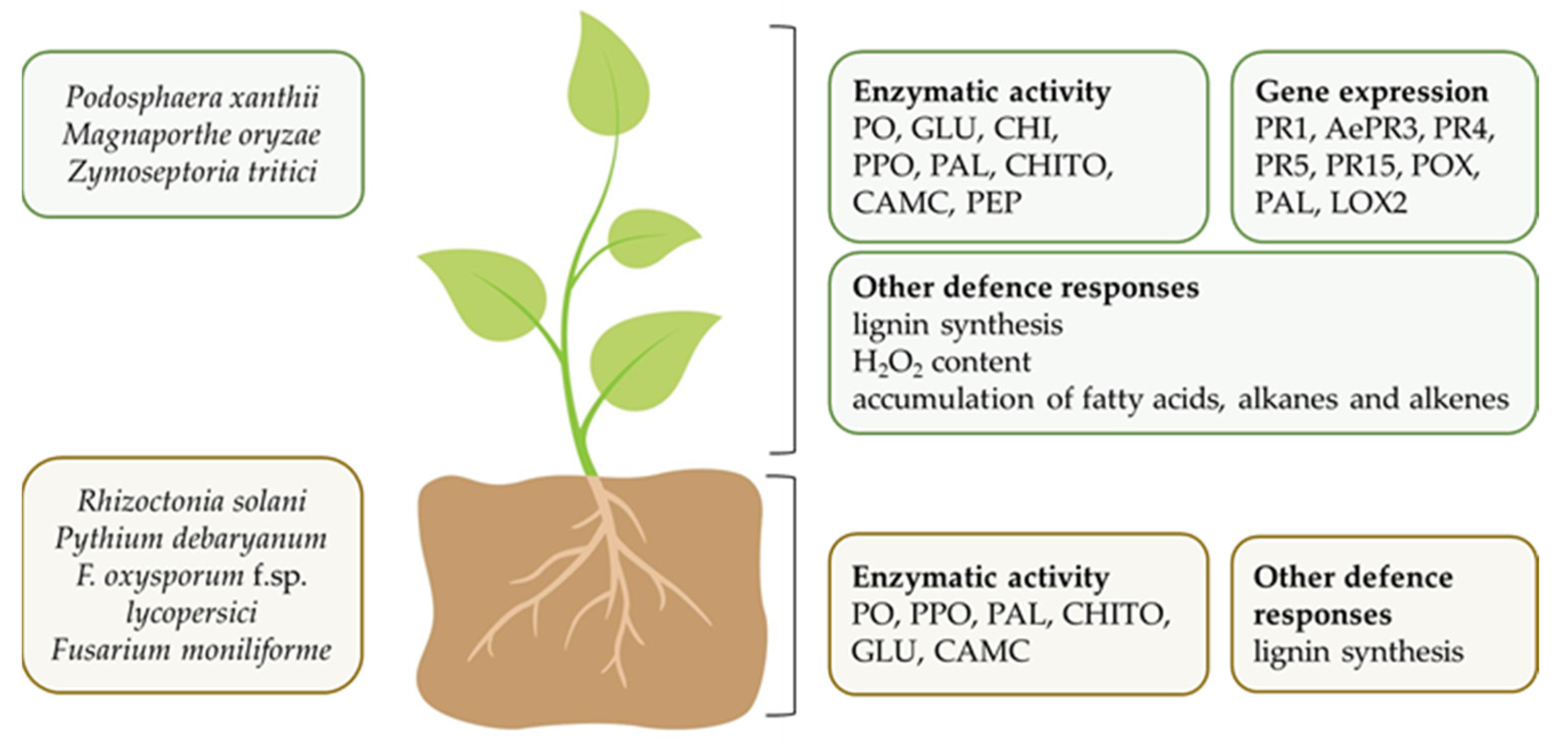
5.1. Plant Pathogens
5.2. Cyanobacteria as Biocontrol Agents
5.2.1. In Vitro Studies and Mechanisms of Fungal and Oomycete Growth Inhibition
| Cyanobacterium | Extract/Culture Filtrate 1 | Plant Pathogen | Reference |
|---|---|---|---|
| Chroococcales | |||
| Microcystis aeruginosa | ME | Aspergillus carbonarius, A. niger | [97] |
| ETH | A. flavus, A. niger, A. parasiticus | ||
| AC | A. flavus, A. niger, Fusarium proliferatum | ||
| MC | A. flavus, A. parasiticus, F. proliferatum | ||
| DE | A. carbonarius, A. flavus, A. niger, A. ochraceus, A. westerdijkiae, F. proliferatum, F. verticillioides, Penicillium verrucosum | ||
| EA | A. carbonarius, A. flavus, A. niger, A. westerdijkiae, F. verticillioides | ||
| Nostocales | |||
| Anabaena spp. | CFILT | Alternaria solani, Drechslera oryzae, Fusarium moniliforme, F. solani, Macrophomina phaseolina, Pythium aphanidermatum | [17] |
| Anabaena sp. | PE | Alternaria alternata | [101] |
| Anabaena sp. | ME | Aspergillus flavus | [98] |
| A. cylindrica | ME | A. flavus | [98] |
| A. oscillarioides | CFILT, L | F. moniliforme, F. oxysporum f. sp. lycopersici, Pythium debaryanum, Rhizoctonia solani | [21] |
| A. solitaria | ME | Alternaria alternata | [101] |
| A. variabilis | CFILT, L | F. moniliforme, F. oxysporum f. sp. lycopersici, P. debaryanum, R. solani | [21] |
| A. variabilis | CFILT | F. oxysporum f. sp. lycopersici | [22] |
| A. laxa clones | CFILT | Pythium aphanidermatum | [105] |
| Calothrix brevissima | PE | Alternaria alternata | [101] |
| ME | A. alternata, Botrytis cinerea, F. oxysporum | [101] | |
| Fischerella sp. | ME | Aspergillus flavus | [98] |
| Nodularia sp. | ME | F. oxysporum | [101] |
| Nostoc sp. | ME | A. flavus | [98] |
| Nostoc strain ATCC 53789 | ME | Armillaria sp., Fusarium solani, F. oxysporum f. sp. melonis, Penicillium expansum, Phytophthora cambivora, P. cinnamomi, Rhizoctonia solani, Rosellinia sp., Sclerotinia sclerotiorum, Verticillium albo-atrum | [102] |
| Nostoc strain UTEX 2493 | ME | Rosellinia sp. | [102] |
| N. calcicula | ME | Aspergillus flavus | [98] |
| N. commune | ME | F. oxysporum f. sp. lycopersici | [104] |
| N. commune | PE | Phytophthora capsici, Pythium ultimum | [101] |
| ME | F. oxysporum, P. capsici | [101] | |
| N.commune | PE | Alternaria alternata | [101] |
| N.commune | ME | A. niger | [100] |
| PE | A. flavus, A. niger | [100] | |
| N.entophytum | AC, CHL, ME | R. solani | [106] |
| N. linckia | ME | F. oxysporum f. sp. lycopersici | [103] |
| N.muscorum | ME | A. alternata, B. cinerea, Colletotrichum gleosporioides | [101] |
| N.muscorum | CFILT | Aspergillus flavus, A. niger, Fusarium microsporium, Penicillium sp. | [99] |
| N.muscorum | AC, CHL, ME | R. solani | [106] |
| Scytonema sp., S. hofmanni | ME | A. flavus | [98] |
| Oscillatoriales | |||
| Arthrospira platensis | PBPs | B. cinerea | [107] |
| Lyngbya lutea | W | A. niger | [100] |
| ME | A. niger, Colletotrichum musae, F. oxysporum | ||
| nPROP | A. flavus, F. oxysporum | ||
| PEE | A. niger, C. musae, F. oxysporum | ||
| Oscillatoria amphibia | W | A. flavus, C. musae | [100] |
| ME | F. oxysporum | ||
| nPROP | A. flavus, C. musae, F. oxysporum | ||
| O. angustissima | PE | C. gleosporioides, F. oxysporum | [101] |
| O. limosa | W | A. niger, C. musae | [100] |
| ME | A. flavus, A. niger, C. musae, F. oxysporum | ||
| nPROP | A. flavus, A. niger, C. musae, F. oxysporum | ||
| O. ornata | ME | A. flavus, A. niger, C. musae, F. oxysporum | |
| nPROP | C. musae, F. oxysporum | ||
| PEE | A. niger | ||
| O. tenuis | PE | A. alternata, P. capsici | [101] |
| ME | P. capsici | ||
| Phormidium autumnale | ME | F. oxysporum f. sp. lycopersici | [103] |
| P. tenue | W | A. niger, C. musae, F. oxysporum | [100] |
| ME | A. niger, F. oxysporum | ||
| nPROP | C. musae, F. oxysporum | ||
| PEE | A. niger, P. lilacimus | ||
| Trichodesmium hildebrantii | W | C. musae | [100] |
| ME | A. niger, C. musae, F. oxysporum | ||
| nPROP | A. flavus, A. niger, C. musae, P. lilacimus | ||
| PEE | C. musae, F. oxysporum | ||
| Synechococcales | |||
| Synechococcus elongates | W | C. musae | [100] |
| ME | A. niger, C. musae, F. oxysporum | ||
| nPROP | A. flavus, A. niger, P. lilacimus | ||
| PEE | C. musae, F. oxysporum | ||
| Synechocystis sp. | W | A. flavus, A. niger, C. musae, P. lilacimus | [100] |
| ME, nPROP | A. flavus, A. niger, C. musae, P. lilacimus, F. oxysporum | ||
| PEE | C. musae |
5.2.2. In Vivo Studies and Mechanisms of Biocontrol Ability
| Cyanobacterium | Extract/ Biomass/ Compound 1 | Plant/ Treatment 3 | Pathogen Control 4/ Plant Defense Responses 5 | Reference |
|---|---|---|---|---|
| Nostocales | ||||
| Anabaena laxa | Biomass culture | Coriander GS | Shoot and root: PO activity; shoot: GLU activity | [111] |
| Cumin GS | Shoot and root: PO activity | |||
| Fennel GS | Shoot: PO activity | |||
| A. minutissima | W | Zucchini L | Podosphaera xanthii (25%) CHI, GLU, PO activity, isoforms of CHI, GLU, PO | [110] |
| W | Cucumber L | P. xanthii (31%) PR1, AePR3 genes | [24] | |
| W | Tomato S | Rhizoctonia solani Seedling: CHI activity, lignin content | [20] | |
| PBPs | Tomato F | Botrytis cinerea cutin and pectin preservation | [23] | |
| POL | Strawberry F | B. cinerea | [109] | |
| A. variabilis | Biomass 2 | Tomato GS | Pythium debaryanum, R. solani, Fusarium moniliforme, F. oxysporum f. sp. lycopersici | [21] |
| A. variabilis | Biomass 2 | Tomato seedling | F. oxysporum f. sp. lycopersici (100%) GLU, PPO, PAL activity | [22] |
| Calothrix elenkinii | Biomass culture | Coriander GS | Shoot and root: PO activity | [111] |
| Cumin GS | Shoot and root: PO activity | |||
| Fennel GS | Shoot: PO, GLU activity | |||
| C. elenkinii | Biomass | Rice GS | Shoot and root: PPO, PAL, PO, CHITO, GLU, CAMC activity | [115] |
| C. elenkinii | Biomass | Rice GS | Magnaporthe oryzae (50%) Leaf: PO, PPO, PAL, PEP activity | [114] |
| Nostoc linkia | Biomass | Tomato GS | F. oxyporum f. sp. lycopersici | [103] |
| N. punctiforme | Medium culture | Arabidopsis thaliana | WRKY | [112] |
| Nostoc-Anabaena consortium | Biomass | Rice GS | Magnaporthe oryzae (69%) PPO activity | [114] |
| Oscillatoriales | ||||
| Arthrospira platensis | DB | Wheat L | Zymoseptoria tritici (~70%) PR4, PR5, PR15, PO, PAL, LOX genes | [116] |
| A. platensis | PBPs | Tomato F | B. cinerea | [107] |
| A. platensis | POL | Tomato L | PAL, CHI, GLU, PO activity; H2O2 content; accumulation of fatty acids, azelaic acid, alkanes, alkenes, other metabolites | [117] |
6. Biostimulant Effects
7. Current Market and Regulations
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Schopf, J.W. The fossil record of cyanobacteria. In Ecology of Cyanobacteria II; Whitton, B., Ed.; Springer: Dordrecht, The Netherlands, 2012; pp. 15–36. [Google Scholar]
- Blankenship, R.E. Origin and early evolution of photosynthesis. Photosynth. Res. 1992, 33, 91–111. [Google Scholar] [CrossRef]
- Buick, R. The antiquity of oxygenic photosynthesis: Evidence from stromatolites in sulphate-deficient archaean lakes. Science 1992, 255, 74–77. [Google Scholar] [CrossRef]
- Barsanti, L.; Gualtieri, P. General Overview. In Algae: Anatomy, Biochemistry, and Biotechnology; Barsanti, L., Gualtieri, P., Eds.; CRC Press Taylor & Francis: Boca Raton, FL, USA, 2005; pp. 1–6. [Google Scholar]
- Dvořák, P.; Casamatta, D.A.; Hašler, P.; Jahodářová, E.; Norwich, A.R.; Poulíčková, A. Diversity of the Cyanobacteria. In Modern Topics in the Phototrophic Prokaryotes; Hallenbeck, P., Ed.; Springer: Cham, Switzerland, 2017; pp. 3–17. [Google Scholar]
- Demoulin, C.F.; Lara, Y.J.; Cornet, L.; François, C.; Baurain, D.; Wilmotte, A.; Javaux, E.J. Cyanobacteria evolution: Insight from the fossil record. Free Radic. Biol. Med. 2019, 140, 206–223. [Google Scholar] [CrossRef]
- Stal, L.J. Cyanobacteria: Diversity and versatility, clues to life in extreme environments. In Algae and Cyanobacteria in Extreme Environments; Seckbach, J., Ed.; Springer: Dordrecht, The Netherlands, 2007; Volume 11, pp. 661–682. [Google Scholar]
- Whitton, B.A.; Potts, M. Ecology of Cyanobacteria II: Their Diversity in Space and Time; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2012; pp. 1–13. [Google Scholar]
- Kurmayer, R.; Deng, L.; Entfellner, E. Role of toxic and bioactive secondary metabolites in colonization and bloom formation by filamentous cyanobacteria Planktothrix. Harmful Algae 2016, 54, 69–86. [Google Scholar] [CrossRef] [PubMed]
- Mazard, S.; Penesyan, A.; Ostrowski, M.; Paulsen, I.T.; Egan, S. Tiny microbes with a big impact: The role of cyanobacteria and their metabolites in shaping our future. Mar. Drugs 2016, 14, 97. [Google Scholar] [CrossRef] [PubMed]
- Mantzouki, E.; Visser, P.M.; Bormans, M.; Ibelings, B.W. Understanding the key ecological traits of cyanobacteria as a basis for their management and control in changing lakes. Aquat. Ecol. 2016, 50, 333–350. [Google Scholar] [CrossRef]
- Herrero, A.; Flores, E. The Cyanobacteria: Molecular Biology, Genomics and Evolution; Caister Academic Press: Sevilla, Spain, 2008; p. 484. [Google Scholar]
- Boden, J.S.; Konhauser, K.O.; Robbins, L.J.; Sánchez-Baracaldo, P. Timing the evolution of antioxidant enzymes in cyanobacteria. Nat. Commun. 2021, 12, 4742. [Google Scholar] [CrossRef]
- Demay, J.; Bernard, C.; Reinhardt, A.; Marie, B. Natural products from cyanobacteria: Focus on beneficial activities. Mar. Drugs 2019, 17, 320. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.A.A.; Akhter, N.; Auckloo, B.N.; Khan, I.; Lu, Y.; Wang, K.; Wu, B.; Guo, Y.-W. Structural diversity, biological properties and applications of natural products from cyanobacteria. A review. Mar. Drugs 2017, 15, 354. [Google Scholar] [CrossRef]
- Carpine, R.; Sieber, S. Antibacterial and antiviral metabolites from cyanobacteria: Their application and their impact on human health. Curr. Res. Biotechnol. 2021, 3, 65–81. [Google Scholar] [CrossRef]
- Prasanna, R.; Nain, L.; Tripathi, R.; Gupta, V.; Chaudhary, V.; Middha, S.; Joshi, M.; Ancha, R.; Kaushik, B.D. Evaluation of fungicidal activity of extracellular filtrates of cyanobacteria—Possible role of hydrolytic enzymes. J. Basic Microbiol. 2008, 48, 186–194. [Google Scholar] [CrossRef]
- Santini, G.; Biondi, N.; Rodolfi, L.; Tredici, M.R. Plant biostimulants from cyanobacteria: An emerging strategy to improve yields and sustainability in agriculture. Plants 2021, 10, 643. [Google Scholar] [CrossRef]
- Berthon, J.-Y.; Michel, T.; Wauquier, A.; Joly, P.; Gerbore, J.; Filaire, E. Seaweed and microalgae as major actors of blue biotechnology to achieve plant stimulation and pest and pathogen—A review of the latest advances and future prospects. J. Agric. Sci. 2021, 1, 12. [Google Scholar] [CrossRef]
- Righini, H.; Francioso, O.; Di Foggia, M.; Prodi, A.; Martel Quintana, A.; Roberti, R. Tomato seed biopriming with water extracts from Anabaena minutissima, Ecklonia maxima and Jania adhaerens as a new agro-ecological option against Rhizoctonia solani. Sci. Hortic. 2021, 281, 109921. [Google Scholar] [CrossRef]
- Chaudhary, V.; Prasanna, R.; Nain, L.; Dubey, S.C.; Gupta, V.; Singh, R.; Jaggi, S.; Bhatnagar, A.K. Bioefficacy of novel cyanobacteria-amended formulations in suppressing damping off disease in tomato seedlings. World J. Microbiol. Biotechnol. 2012, 28, 3301–3310. [Google Scholar] [CrossRef] [PubMed]
- Prasanna, R.; Chaudhary, V.; Gupta, V.; Babu, S.; Kumar, A.; Singh, R.; Shivay, Y.S.; Nain, L. Cyanobacteria mediated plant growth promotion and bioprotection against Fusarium wilt in tomato. Eur. J. Plant Pathol. 2013, 136, 337–353. [Google Scholar] [CrossRef]
- Righini, H.; Francioso, O.; Di Foggia, M.; Martel Quintana, A.; Roberti, R. Assessing the potential of the terrestrial cyanobacterium Anabaena minutissima for controlling Botrytis cinerea on tomato fruits. Horticulturae 2021, 7, 210. [Google Scholar] [CrossRef]
- Righini, H.; Somma, A.; Cetrullo, S.; D’Adamo, S.; Flamigni, F.; Martel Quintana, A.; Roberti, R. Inhibitory activity of aqueous extracts from Anabaena minutissima, Ecklonia maxima and Jania adhaerens on the cucumber powdery mildew pathogen in vitro and in vivo. J. Appl. Phycol. 2020, 32, 3363–3375. [Google Scholar] [CrossRef]
- Koksharova, O.A.; Wolk, C.P. Genetic tools for cyanobacteria. Appl. Microbiol. Biotechnol. 2002, 58, 123–137. [Google Scholar]
- Wijffels, R.H.; Kruse, O.; Hellingwerf, K.J. Potential of industrial biotechnology with cyanobacteria and eukaryotic microalgae. Curr. Opin. Biotech. 2013, 24, 405–413. [Google Scholar] [CrossRef]
- Balasubramaniam, V.; Gunasegavan, R.D.N.; Mustar, S.; Lee, J.C.; Mohd Noh, M.F. Isolation of industrial important bioactive compounds from microalgae. Molecules 2021, 26, 943. [Google Scholar] [CrossRef]
- Masojídek, J.; Torzillo, G. Mass Cultivation of Freshwater Microalgae. In Encyclopedia of Ecology, 1st ed.; Jørgensen, S.E., Fath, B.D., Eds.; Elsevier B.V.: Amsterdam, The Netherlands, 2008; pp. 2226–2235. [Google Scholar]
- Acién, F.G.; Molina, E.; Reis, A.; Torzillo, G.; Zittelli, G.C.; Sepúlveda, C.; Masojídek, J. Photobioreactors for the production of microalgae. In Microalgae-Based Biofuels and Bioproducts; Gonzales-Fernandez, C., Munoz, R., Eds.; Woodhead Publishing; Elsevier: Kidlington, UK, 2017; pp. 1–44. [Google Scholar]
- Masojídek, J.; Ranglová, K.; Lakatos, G.E.; Silva Benavides, A.M.; Torzillo, G. Variables governing photosynthesis and growth in microalgae mass cultures. Processes 2021, 9, 820. [Google Scholar] [CrossRef]
- Darvehei, P.; Bahri, P.A.; Moheimani, N.R. Model development for the growth of microalgae: A review. Renew. Sust. Energ. Rev. 2018, 97, 233–258. [Google Scholar] [CrossRef]
- Show, K.Y.; Lee, D.J.; Chang, J.S. Algal biomass dehydration. Bioresour. Technol. 2013, 135, 720–729. [Google Scholar] [CrossRef]
- Mohn, F.H. Improved technologies for the harvesting and processing of microalgae and their impact on production costs. In Microalgae for Food and Feed/Ergebn. Limnol; Soeder, C.J., Binsack, R., Eds.; Schweizerbart Science Publisher: Stuttgart, Germany, 1978; Volume II, pp. 228–253. [Google Scholar]
- Ventura, S.P.M.; Nobre, B.P.; Ertekin, F.; Hayes, M.; Garciá-Vaquero, M.; Vieira, F.; Koc, M.; Gouveia, L.; Aires-Barros, M.R.; Palavra, A.M.F. Extraction of value-added compounds from microalgae. In Microalgae-Based Biofuels and Bioproducts; Gonzalez-Fernandez, C., Munoz, R., Eds.; Woodhead Publishing; Elsevier: Kidlington, UK, 2017; pp. 461–483. [Google Scholar]
- Sanz, N.; García-Blanco, A.; Gavalás-Olea, A.; Loures, P.; Garrido, J.L. Phytoplankton pigment biomarkers: HPLC separation using a pentafluorophenyloctadecyl silica column. Methods Ecol. Evol. 2015, 6, 1199–1209. [Google Scholar] [CrossRef]
- Desai, R.K.; Streefland, M.; Wijffels, R.H.; Eppink, M.H.M. Novel astaxanthin extraction from Haematococcus pluvialis using cell permeabilising ionic liquids. Green Chem. 2016, 18, 1261–1267. [Google Scholar] [CrossRef]
- Benedetti, M.; Vecchi, V.; Barera, S.; Dall’Osto, L. Biomass from microalgae: The potential of domestication towards sustainable biofactories. Microb. Cell Fact. 2018, 17, 173. [Google Scholar] [CrossRef] [PubMed]
- Favas, R.; Morone, J.; Martins, R.; Vasconcelos, V.; Lopes, G. Cyanobacteria and microalgae bioactive compounds in skin-ageing: Potential to restore extracellular matrix filling and overcome hyperpigmentation. J. Enzym. Inhib. Med. Chem. 2021, 36, 1829–1838. [Google Scholar] [CrossRef]
- Fernandes, T.; Cordeiro, N. Microalgae as sustainable biofactories to produce high-value lipids: Biodiversity, exploitation, and biotechnological applications. Mar. Drugs 2021, 19, 573. [Google Scholar] [CrossRef]
- Pattnaik, S.; Singh, L. Cyanobacteria bioactive compound, their production and extraction with pharmaceutical applications—A review. Int. J. Curr. Microb. Appl. Sci. 2020, 9, 3394–3405. [Google Scholar] [CrossRef]
- Kultschar, B.; Llewellyn, C. Secondary Metabolites in Cyanobacteria. In Secondary Metabolites—Sources and Applications; Vijayakumar, R., Ed.; IntechOpen: London, UK, 2018. [Google Scholar] [CrossRef]
- Gademann, K.; Portmann, C. Secondary metabolites from cyanobacteria: Complex structures and powerful bioactivities. Curr. Org. Chem. 2008, 12, 326–341. [Google Scholar] [CrossRef]
- Luesch, H.; Harrigan, G.G.; Goetz, G.; Horgen, F.D. The cyanobacterial origin of potent anticancer agents originally isolated from sea hares. Curr. Med. Chem. 2002, 9, 1791–1806. [Google Scholar] [CrossRef] [PubMed]
- Burja, A.M.; Banaigs, B.; Abou-Mansour, E.; Burgess, J.G.; Wright, P.C. Marine cyanobacteria—A prolific source of natural products. Tetrahedron 2002, 57, 9347–9377. [Google Scholar] [CrossRef]
- Jones, M.R.; Pinto, E.; Torres, M.A.; Dörr, F.; Mazur-Marzec, H.; Szubert, K.; Tartaglione, L.; Dell’Aversano, C.; Miles, C.O.; Beach, D.G.; et al. CyanoMetDB, a comprehensive public database of secondary metabolites from cyanobacteria. Water Res. 2021, 196, 117017. [Google Scholar] [CrossRef]
- Nunnery, J.K.; Mevers, E.; Gerwick, W.H. Biologically active secondary metabolites from marine cyanobacteria. Curr. Opin. Biotechnol. 2010, 21, 787–793. [Google Scholar] [CrossRef] [PubMed]
- Cabanillas, A.H.; Tena Pérez, V.; Maderuelo Corral, S.; Rosero Valencia, D.F.; Martel Quintana, A.; Ortega Doménech, M.; Rumbero Sánchez, Á. Cybastacines A and B: Antibiotic sesterterpenes from a Nostoc sp. cyanobacterium. J. Nat. Prod. 2018, 81, 410–413. [Google Scholar] [CrossRef] [PubMed]
- Marahiel, M.A.; Essen, L.O. Nonribosomal peptide synthetases mechanistic and structural aspects of essential domains. Method Enzymol. 2009, 458, 337–351. [Google Scholar]
- Llewellyn, C.A.; Kapoore, R.V.; Lovitt, R.W.; Greig, C.; Fuentes-Grünewald, C.; Kultschar, B. Deriving economic value from metabolites in cyanobacteria. In Grand Challenges in Algae Biotechnology; Hallmann, A., Rampelotto, P., Eds.; Springer: Cham, Switzerland, 2019; pp. 535–576. [Google Scholar]
- Kapoore, R.V.; Wood, E.E.; Llewellyn, C.A. Algae biostimulants: A critical look at microalgal biostimulants for sustainable agricultural practices. Biotechnol. Adv. 2021, 49, 107754. [Google Scholar] [CrossRef]
- Parwani, L.; Bhatt, M.; Singh, J. Potential biotechnological applications of cyanobacterial exopolysaccharides. Braz. Arch. Biol. Technol. 2021, 64, e21200401. [Google Scholar] [CrossRef]
- Rosic, N.N. Recent advances in the discovery of novel marine natural products and mycosporine-like amino acid UV-absorbing compounds. Appl. Microbiol. Biotechnol. 2021, 105, 7053–7067. [Google Scholar] [CrossRef]
- Morone, J.; Alfeus, A.; Vasconcelos, V.; Martins, R. Revealing the potential of cyanobacteria in cosmetics and cosmeceuticals—A new bioactive approach. Algal Res. 2019, 41, 101541. [Google Scholar] [CrossRef]
- Vega, J.; Bonomi-Barufi, J.; Gómez-Pinchetti, J.L.; Figueroa, F.L. Cyanobacteria and red macroalgae as potential sources of antioxidants and UV radiation-absorbing compounds for cosmeceutical applications. Mar. Drugs 2020, 18, 659. [Google Scholar] [CrossRef] [PubMed]
- Mitra, S.; Siddiqui, W.A.; Khandelwal, S. C-Phycocyanin protects against acute tributyltin chloride neurotoxicity by modulating glial cell activity along with its anti-oxidant and anti-inflammatory property: A comparative efficacy evaluation with N-acetyl cysteine in adult rat brain. Chem. Biol. Interact. 2015, 238, 138–150. [Google Scholar] [CrossRef]
- Vijayakumar, S.; Menakha, M. Pharmaceutical applications of cyanobacteria—A review. J. Acute Med. 2015, 5, 15–23. [Google Scholar] [CrossRef]
- Tan, L.T.; Phyo, M.Y. Marine cyanobacteria: A source of lead compounds and their clinically-relevant molecular targets. Molecules 2020, 25, 2197. [Google Scholar] [CrossRef] [PubMed]
- Castaneda, A.; Ferraz, R.; Vieira, M.; Cardoso, I.; Vasconcelos, V.; Martins, R. Bridging cyanobacteria to neurodegenerative diseases: A new potential source of bioactive compounds against Alzheimer’s disease. Mar. Drugs 2021, 19, 343. [Google Scholar] [CrossRef]
- Saad, M.H.; El-Fakharany, E.M.; Salem, M.S.; Sidkey, N.M. The use of cyanobacterial metabolites as natural medical and biotechnological tools: Review article. J. Biomol. Struct. Dyn. 2020, 8, 1–23. [Google Scholar] [CrossRef]
- Van Apeldoorn, M.E.; van Egmond, H.P.; Speijers, G.J.A.; Bakker, G.J.I. Toxins of cyanobacteria. Mol. Nutr. Food Res. 2007, 51, 7–60. [Google Scholar] [CrossRef]
- Carmichael, W.W. The toxins of cyanobacteria. Sci. Am. 1994, 270, 78–86. [Google Scholar] [CrossRef]
- Kubickova, B.; Babica, P.; Hilscherová, K.; Šindlerová, L. Effects of cyanobacterial toxins on the human gastrointestinal tract and the mucosal innate immune system. Environ. Sci. Eur. 2019, 31, 31. [Google Scholar] [CrossRef]
- Carmichael, W.W. Health effects of toxin-producing cyano-bacteria, “The CyanoHabs”. Hum. Ecol. Risk Assess. 2001, 7, 1393–1407. [Google Scholar] [CrossRef]
- Bláha, L.; Babica, P.; Maršálek, B. Toxins produced in cyanobacterial water blooms—Toxicity and risks. Interdisc. Toxicol. 2009, 2, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Corbel, S.; Mougin, C.; Bouaïcha, N. Cyanobacterial toxins: Modes of actions, fate in aquatic and soil ecosystems, phytotoxicity and bioaccumulation in agricultural crops. Chemosphere 2014, 96, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, J.S.; Tischbein, M.; Cox, P.A.; Stommel, E.W. Cyanotoxins and the nervous system. Toxins 2021, 13, 660. [Google Scholar] [CrossRef] [PubMed]
- Diez-Quijada, L.; Benítez-González, M.d.M.; Puerto, M.; Jos, A.; Cameán, A.M. Immunotoxic effects induced by microcystins and cylindrospermopsin: A review. Toxins 2021, 13, 711. [Google Scholar] [CrossRef]
- Machado, J.; Campos, A.; Vasconcelos, V.; Freitas, M. Effects of microcystin-LR and cylindrospermopsin on plant-soil systems: A review of their relevance for agricultural plant quality and public health. Environ. Res. 2017, 153, 191–204. [Google Scholar] [CrossRef]
- Máthé, C.; M-Hamvas, M.; Vasas, G.; Garda, T.; Freytag, C. Subcellular alterations induced by cyanotoxins in vascular plants—A review. Plants 2021, 10, 984. [Google Scholar] [CrossRef]
- M-Hamvas, M.; Vasas, G.; Beyer, D.; Nagylaki, E.; Máthé, C. Microcystin-LR, a cyanobacterial toxin, induces DNA strand breaks correlated with changes in specific nuclease and protease activities in white mustard (Sinapis alba) seedlings. Plants 2021, 10, 2045. [Google Scholar] [CrossRef]
- Campos, A.; Redouane, E.M.; Freitas, M.; Amaral, S.; Azevedo, T.; Loss, L.; Máthé, C.; Mohamed, Z.A.; Oudra, B.; Vasconcelos, V. Impacts of microcystins on morphological and physiological parameters of agricultural plants: A review. Plants 2021, 10, 639. [Google Scholar] [CrossRef]
- Zhang, Y.; Whalen, J.K.; Sauvé, S. Phytotoxicity and bioconcentration of microcystins in agricultural plants: Meta-analysis and risk assessment. Environ. Pollut. 2021, 272, 115966. [Google Scholar] [CrossRef]
- Liang, C.; Wang, W. Response and recovery of rice (Oryza sativa) seedlings to irrigation with microcystin-contaminated water. Environ. Earth Sci. 2015, 73, 4573–4580. [Google Scholar] [CrossRef]
- Abe, T.; Lawson, T.; Weyers, J.D.; Codd, G.A. Microcystin-LR inhibits photosynthesis of Phaseolus vulgaris primary leaves: Implications for current spray irrigation practice. New Phytol. 1996, 133, 651–658. [Google Scholar] [CrossRef]
- Westphal, S.; Soll, J.; Vothknecht, U.C. A vesicle transport system inside chloroplasts. FEBS Lett. 2001, 506, 257–261. [Google Scholar] [CrossRef]
- Máthé, C.; M-Hamvas, M.; Vasas, G. Microcystin-LR and cylindrospermopsin induced alterations in chromatin organization of plant cells. Mar. Drugs 2013, 11, 3689–3717. [Google Scholar] [CrossRef] [PubMed]
- McElhiney, J.; Lawton, L.A.; Leifert, C. Investigations into the inhibitory effects of microcystins on plant growth, and the toxicity of plant tissues following exposure. Toxicon 2001, 39, 1411–1420. [Google Scholar] [CrossRef]
- Babica, P.; Bláha, L.; Maršálek, B. Exploring the natural role of microcystins—A review of effects on photoautotrophic organisms. J. Phycol. 2006, 42, 9–20. [Google Scholar] [CrossRef]
- Knogge, W. Fungal infection of plants. Plant Cell 1996, 8, 1711–1722. [Google Scholar] [CrossRef] [PubMed]
- Dean, R.; Van Kan, J.A.L.; Pretorius, Z.A.; Hammond-Kosack, K.E.; Di Pietro, A.; Spanu, P.D.; Rudd, J.J.; Dickman, M.; Kahmann, R.; Ellis, J.; et al. The Top 10 fungal pathogens in molecular plant pathology. Mol. Plant Pathol. 2012, 13, 414–430. [Google Scholar] [CrossRef] [PubMed]
- Gil-Serna, J.; Vázquez, C.; Patiño, B. Genetic regulation of aflatoxin, ochratoxin A, trichothecene, and fumonisin biosynthesis: A review. Int. Microbiol. 2020, 23, 89–96. [Google Scholar] [CrossRef]
- Morales, H.; Marín, S.; Rovira, A.; Ramos, A.J.; Sanchis, V. Patulin accumulation in apples by Penicillium expansum during postharvest stages. Lett. Appl. Microbiol. 2007, 44, 30–35. [Google Scholar] [CrossRef]
- Bertero, A.; Moretti, A.; Spicer, L.J.; Caloni, F. Fusarium molds and mycotoxins: Potential species-specific effects. Toxins 2018, 10, 244. [Google Scholar] [CrossRef] [PubMed]
- Carris, L.M.; Little, C.R.; Stiles, C.M. Introduction to Fungi. 2012. Available online: https://www.apsnet.org/edcenter/disandpath/fungalasco/intro/Pages/IntroFungi.aspx (accessed on 15 October 2021).
- Barberis, C.L.; Dalcero, A.M.; Magnoli, C.E. Evaluation of aflatoxin B1 and ochratoxin A in interacting mixed cultures of Aspergillus sections Flavi and Nigri on peanut grains. Mycotoxin Res. 2012, 28, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Gnat, S.; Łagowski, D.; Nowakiewicz, A.; Dyląg, M. A global view on fungal infections in humans and animals: Opportunistic infections and microsporidioses. J. Appl. Microbiol. 2021, 131, 2095–2113. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, S.; Taylor, A.; Jackson, A.C.; Armitage, A.D.; Bates, H.J.; Mead, A.; Harrison, R.J.; Clarkson, J.P. Identification and expression of secreted in xylem pathogenicity genes in Fusarium oxysporum f. sp. pisi. Front. Microbiol. 2021, 12, 593140. [Google Scholar] [CrossRef]
- Michielse, C.B.; Rep, M. Pathogen profile update: Fusarium oxysporum. Mol. Plant Pathol. 2009, 10, 311–324. [Google Scholar] [CrossRef] [PubMed]
- Agrios, G. Plant Pathology, 5th ed.; Elsevier Academic Press: San Diego, CA, USA, 2005; p. 952. [Google Scholar]
- Batista, B.G.; de Chaves, M.A.; Reginatto, P.; Saraiva, O.J.; Fuentefria, A.M. Human fusariosis: An emerging infection that is difficult to treat. Rev. Soc. Bras. Med. Trop. 2020, 53, e20200013. [Google Scholar] [CrossRef]
- Lamour, K.H.; Stam, R.; Jupe, J.; Huitema, E. The oomycete broad-host-range pathogen Phytophthora capsici. Mol. Plant. Pathol. 2012, 13, 329–337. [Google Scholar] [CrossRef]
- Baker, K.F. Types of Rhizoctonia diseases and their occurrence. In Rhizoctonia solani: Biology and Pathology; Parmeter, J.R., Jr., Ed.; University of California Press: Berkeley, CA, USA, 1970; pp. 125–148. [Google Scholar]
- Fillinger, S.; Elad, Y. Botrytis—The Fungus, the Pathogen and Its Management in Agricultural Systems; Springer International Publishing: Cham, Switzerland, 2016; p. 486. [Google Scholar]
- Pérez-García, A.; Romero, D.; Fernández-Ortuño, D.; López-Ruiz, F.; De Vicente, A.; Tores, J.A. The powdery mildew fungus Podosphaera fusca (synonym Podosphaera xanthii), a constant threat to cucurbits. Mol. Plant Pathol. 2009, 10, 153–160. [Google Scholar] [CrossRef]
- Keinath, A.P.; DuBose, V.B. Evaluation of fungicides for prevention and management of powdery mildew on watermelon. Crop Prot. 2004, 23, 35–42. [Google Scholar] [CrossRef]
- Boddy, L. Pathogens of autotrophs. In The Fungi, 3rd ed.; Watkinson, S.C., Boddy, L., Money, N.P., Eds.; Academic Press: London, UK, 2016; pp. 245–292. [Google Scholar]
- Marrez, D.A.; Sultan, Y.Y. Antifungal activity of the cyanobacterium Microcystis aeruginosa against mycotoxigenic fungi. J. Appl. Pharm. Sci. 2016, 6, 191–198. [Google Scholar] [CrossRef]
- Shishido, T.K.; Humisto, A.; Jokela, J.; Liu, L.; Wahlsten, M.; Tamrakar, A.; Fewer, D.P.; Permi, P.; Andreote, A.P.D.; Fiore, M.F.; et al. Antifungal compounds from cyanobacteria. Mar. Drugs 2015, 13, 2124–2140. [Google Scholar] [CrossRef]
- El-Sheekh, M.M.; Osman, M.E.H.; Dyab, M.A.; Amer, M.S. Production and characterization of antimicrobial active substance from the cyanobacterium Nostoc muscorum. Environ. Toxicol. Phar. 2006, 21, 42–50. [Google Scholar] [CrossRef]
- Pawar, S.T.; Puranik, P.R. Screening of terrestrial and freshwater halotolerant cyanobacteria for antifungal activities. World J. Microbiol. Biotechnol. 2008, 24, 1019–1025. [Google Scholar] [CrossRef]
- Kim, J.D. Screening of cyanobacteria (blue-green algae) from rice paddy soil for antifungal activity against plant pathogenic fungi. Microbiology 2006, 34, 138–142. [Google Scholar] [CrossRef]
- Biondi, N.; Piccardi, R.; Margheri, M.C.; Rodolfi, L.; Smith, G.D.; Tredici, M.R. Evaluation of Nostoc strain ATCC 53789 as a potential source of natural pesticides. Appl. Environ. Microbiol. 2004, 70, 3313–3320. [Google Scholar] [CrossRef]
- Alwathnani, H.A.; Perveen, K. Biological control of Fusarium wilt of tomato by antagonist fungi and cyanobacteria. Afr. J. Biotechnol. 2012, 11, 1100–1105. [Google Scholar] [CrossRef]
- Kim, J.; Kim, J.-D. Inhibitory effect of algal extracts on mycelial growth of the tomato-wilt pathogen, Fusarium oxysporum f. sp. lycopersici. Mycobiology 2008, 36, 242–248. [Google Scholar] [CrossRef]
- Gupta, V.; Prasanna, R.; Natarajan, C.; Srivastava, A.K.; Sharma, J. Identification, characterization, and regulation of a novel antifungal chitosanase gene (cho) in Anabaena spp. Appl. Environ. Microbiol. 2010, 76, 2769–2777. [Google Scholar] [CrossRef]
- Osman, M.E.A.H.; El-Sheekh, M.M.; Metwally, M.A.; Ismail, A.E.W.A.; Ismail, M.M. Antagonistic activity of some fungi and cyanobacteria species against Rhizoctonia solani. Int. J. Plant Pathol. 2011, 2, 101–114. [Google Scholar] [CrossRef][Green Version]
- Righini, H.; Francioso, O.; Di Foggia, M.; Martel Quintana, A.; Roberti, R. Preliminary study on the activity of phycobiliproteins against Botrytis cinerea. Mar. Drugs 2020, 18, 600. [Google Scholar] [CrossRef]
- Moreno, A.B.; Martínez del Pozo, Á.; Borja, M.; San Segundo, B. Activity of the antifungal protein from Aspergillus giganteus against Botrytis cinerea. Phytopathology 2003, 93, 1344–1353. [Google Scholar] [CrossRef]
- Righini, H.; Baraldi, E.; García Fernández, Y.; Martel Quintana, A.; Roberti, R. Different Antifungal Activity of Anabaena sp., Ecklonia sp., and Jania sp. against Botrytis cinerea. Mar. Drugs 2019, 17, 299. [Google Scholar] [CrossRef]
- Roberti, R.; Galletti, S.; Burzi, P.L.; Righini, H.; Cetrullo, S.; Perez, C. Induction of defence responses in zucchini (Cucurbita pepo) by Anabaena sp. water extract. Biol. Control 2015, 82, 61–68. [Google Scholar] [CrossRef]
- Kumar, M.; Prasanna, R.; Bidyarani, N.; Babu, S.; Mishra, B.K.; Kumar, A.; Adak, A.; Jauhari, S.; Yadav, K.; Singh, R.; et al. Evaluating the plant growth promoting ability of thermotolerant bacteria and cyanobacteria and their interactions with seed spice crops. Sci. Hortic. 2013, 164, 94–101. [Google Scholar] [CrossRef]
- Belton, S.; McCabe, P.F.; Ng, C.K.Y. The cyanobacterium, Nostoc punctiforme can protect against programmed cell death and induce defence genes in Arabidopsis thaliana. J. Plant Interact. 2021, 16, 64–74. [Google Scholar] [CrossRef]
- Zheng, J.; Zhang, Z.; Tong, T.; Fang, Y.; Zhang, X.; Niu, C.; Li, J.; Wu, Y.; Xue, D.; Zhang, X. Genome-wide identification of wrky gene family and expression analysis under abiotic stress in barley. Agronomy 2021, 11, 521. [Google Scholar] [CrossRef]
- Thapa, S.; Prasanna, R.; Ramakrishnan, B.; Mahawar, H.; Bharti, A.; Kumar, A.; Velmourougane, K.; Shivay, Y.S.; Kumar, A. Microbial inoculation elicited changes in phyllosphere microbial communities and host immunity suppress Magnaporthe oryzae in a susceptible rice cultivar. Physiol. Mol. Plant Pathol. 2021, 114, 101625. [Google Scholar] [CrossRef]
- Priya, H.; Prasanna, R.; Ramakrishnan, B.; Bidyarani, N.; Babu, S.; Thapa, S.; Renuka, N. Influence of cyanobacterial inoculation on the culturable microbiome and growth of rice. Microbiol. Res. 2015, 171, 78–89. [Google Scholar] [CrossRef]
- Le Mire, G.; Siah, A.; Marolleau, B.; Gaucher, M.; Maumené, C.; Brostaux, Y.; Massart, S.; Brisset, M.N.; Jijakli, M.H. Evaluation of λ-carrageenan, CpG-ODN, glycine betaine, Spirulina platensis, and ergosterol as elicitors for control of Zymoseptoria tritici in wheat. Phytopathology 2019, 109, 409–417. [Google Scholar] [CrossRef]
- Rachidi, F.; Benhima, R.; Kasmi, Y.; Sbabou, L.; El Arroussi, H. Evaluation of microalgae polysaccharides as biostimulants of tomato plant defense using metabolomics and biochemical approaches. Sci. Rep. 2021, 11, 930. [Google Scholar] [CrossRef]
- Chiaiese, P.; Corrado, G.; Colla, G.; Kyriacou, M.C.; Rouphael, Y. Renewable sources of plant biostimulation: Microalgae as a sustainable means to improve crop performance. Front. Plant Sci. 2018, 9, 1782. [Google Scholar] [CrossRef]
- Sharma, S.B.; Sayyed, R.Z.; Trivedi, M.H.; Gobi, T.A. Phosphate solubilizing microbes: Sustainable approach for managing phosphorus deficiency in agricultural soils. Springerplus 2013, 2, 587. [Google Scholar] [CrossRef]
- Li, J.; Li, C.; Smith, S.M. Hormone Metabolism and Signaling in Plants; Academic Press Elsevier: London, UK, 2017; p. 597. [Google Scholar]
- Karthikeyan, N.; Prasanna, R.; Sood, A.; Jaiswal, P.; Nayak, S.; Kaushik, B.D. Physiological characterization and electron microscopic investigation of cyanobacteria associated with wheat rhizosphere. Folia Microbiol. 2009, 54, 43–51. [Google Scholar] [CrossRef]
- Maqubela, M.P.; Mnkeni, P.N.S.; Malam Issa, O.; Pardo, M.T.; D’Acqui, L.P. Nostoc cyanobacterial inoculation in South African agricultural soils enhances soil structure, fertility, and maize growth. Plant Soil 2009, 315, 79–92. [Google Scholar] [CrossRef]
- Řezanka, T.; Palyzová, A.; Sigler, K. Isolation and identification of siderophores produced by cyanobacteria. Folia Microbiol. 2018, 63, 569–579. [Google Scholar] [CrossRef]
- Shukia, S.P.; Singh, J.S.; Kashyap, S.; Giri, D.D.; Kashyap, A.K. Antarctic cyanobacteria as a source of phycocyanin: An assessment. Indian J. Mar. Sci. 2008, 37, 446–449. [Google Scholar]
- Godlewska, K.; Michalak, I.; Pacyga, P.; Baśladyńska, S.; Chojnacka, K. Potential applications of cyanobacteria: Spirulina platensis filtrates and homogenates in agriculture. World J. Microbiol. Biotechnol. 2019, 35, 80. [Google Scholar] [CrossRef]
- Markets and Markets. Available online: https://www.marketsandmarkets.com/Market-Reports/biostimulant-market-1081.html (accessed on 9 October 2021).
- Arnau, L. Techno-Economic Feasibility Study for the Production of Microalgae Based Plant Biostimulant. Master’s Thesis, KTH, Royal Institute of Technology School of Chemical Science and Engineering, Stockholm, Sweden, 2016. [Google Scholar]
- El Boukhari, M.E.M.E.; Barakate, M.; Bouhia, Y.; Lyamlouli, K. Trends in seaweed extract based biostimulants: Manufacturing process and beneficial effect on soil-plant systems. Plants 2020, 9, 359. [Google Scholar] [CrossRef]
- Siddiki, S.Y.A.; Mofijur, M.; Kumar, S.P.; Ahmed, S.F.; Inayat, A.; Kusumo, F.; Badruddin, I.A.; Yunus Khan, T.M.; Nghiem, L.D.; Ong, H.C.; et al. Microalgae biomass as a sustainable source for biofuel, biochemical and biobased value-added products: An integrated biorefinery concept. Fuel 2022, 307, 121782. [Google Scholar] [CrossRef]
- Romero Villegas, G.I.; Fiamengo, M.; Acién Fernández, F.G.; Molina Grima, E. Outdoor production of microalgae biomass at pilot-scale in seawater using centrate as the nutrient source. Algal Res. 2017, 25, 538–548. [Google Scholar] [CrossRef]
- Aghofack-Nguemezi, J.; Schinzoumka, P.A.; Tatchago, V. Effects of extracts or powder of Jatropha curcas and Spirulina platensis on the growth and development of tomato plant. J. Appl. Biosci. 2015, 90, 8413–8420. [Google Scholar] [CrossRef][Green Version]
- Renuka, N.; Guldhe, A.; Prasanna, R.; Singh, P.; Bux, F. Microalgae as multi-functional options in modern agriculture: Current trends, prospects and challenges. Biotechnol. Adv. 2018, 36, 1255–1273. [Google Scholar] [CrossRef]
- Mógor, Á.F.; Ördög, V.; Pereira Lima, G.P.; Molnár, Z.; Mógor, G. Biostimulant properties of cyanobacterial hydrolysate related to polyamines. J. Appl. Phycol. 2018, 30, 453–460. [Google Scholar] [CrossRef]
- Supraja, K.V.; Behera, B.; Balasubramanian, P. Efficacy of microalgal extracts as biostimulants through seed treatment and foliar spray for tomato cultivation. Ind. Crops Prod. 2020, 151, 112453. [Google Scholar]
- Toribio, A.J.; Suárez-Estrella, F.; Jurado, M.M.; López, M.J.; López-González, J.A.; Moreno, J. Prospection of cyanobacteria producing bioactive substances and their application as potential phytostimulating agents. Biotechnol. Rep. 2020, 26, e00449. [Google Scholar] [CrossRef]
- Singh, S. A review on possible elicitor molecules of cyanobacteria: Their role in improving plant growth and providing tolerance against biotic or abiotic stress. J. Appl. Microbiol. 2014, 117, 1221–1244. [Google Scholar] [CrossRef]
- Žižková, E.; Kubeš, M.; Dobrev, P.I.; Přibyl, P.; Šimura, J.; Zahajská, L.; Drábková, L.Z.; Novák, O.; Motyka, V. Control of cytokinin and auxin homeostasis in cyanobacteria and algae. Ann. Bot. 2017, 119, 151–166. [Google Scholar] [CrossRef]
- Bareke, T. Biology of seed development and germination physiology. Adv. Plants Agric. Res. 2018, 8, 336–346. [Google Scholar] [CrossRef]
- Barone, V.; Baglieri, A.; Stevanato, P.; Broccanello, C.; Bertoldo, G.; Bertaggia, M.; Cagnin, M.; Pizzeghello, D.; Moliterni, V.M.C.; Mandolino, G.; et al. Root morphological and molecular responses induced by microalgae extracts in sugar beet (Beta vulgaris L.). J. Appl. Phycol. 2017, 30, 1061–1071. [Google Scholar] [CrossRef]
- Araújo, R.; Vázquez Calderón, F.; Sánchez López, J.; Azevedo, I.C.; Bruhn, A.; Fluch, S.; Garcia Tasende, M.; Ghaderiardakani, F.; Ilmjärv, T.; Laurans, M.; et al. Current status of the algae production industry in Europe: An emerging sector of the blue bioeconomy. Front. Mar. Sci. 2021, 7, 626389. [Google Scholar] [CrossRef]
- Rumin, J.; Gonçalves de Oliveira Junior, R.; Bérard, J.-B.; Picot, L. Improving microalgae research and marketing in the European Atlantic area: Analysis of major gaps and barriers limiting sector development. Mar. Drugs 2021, 19, 319. [Google Scholar] [CrossRef]
- Lee, S.-M.; Ryu, C.-M. Algae as new kids in the beneficial plant microbiome. Front. Plant Sci. 2021, 12, 599742. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Righini, H.; Francioso, O.; Martel Quintana, A.; Roberti, R. Cyanobacteria: A Natural Source for Controlling Agricultural Plant Diseases Caused by Fungi and Oomycetes and Improving Plant Growth. Horticulturae 2022, 8, 58. https://doi.org/10.3390/horticulturae8010058
Righini H, Francioso O, Martel Quintana A, Roberti R. Cyanobacteria: A Natural Source for Controlling Agricultural Plant Diseases Caused by Fungi and Oomycetes and Improving Plant Growth. Horticulturae. 2022; 8(1):58. https://doi.org/10.3390/horticulturae8010058
Chicago/Turabian StyleRighini, Hillary, Ornella Francioso, Antera Martel Quintana, and Roberta Roberti. 2022. "Cyanobacteria: A Natural Source for Controlling Agricultural Plant Diseases Caused by Fungi and Oomycetes and Improving Plant Growth" Horticulturae 8, no. 1: 58. https://doi.org/10.3390/horticulturae8010058
APA StyleRighini, H., Francioso, O., Martel Quintana, A., & Roberti, R. (2022). Cyanobacteria: A Natural Source for Controlling Agricultural Plant Diseases Caused by Fungi and Oomycetes and Improving Plant Growth. Horticulturae, 8(1), 58. https://doi.org/10.3390/horticulturae8010058







