Biological Control of Pear Valsa Canker Caused by Valsa pyri Using Penicillium citrinum
Abstract
:1. Introduction
2. Materials and Methods
2.1. Pathogenic Fungal Isolates
2.2. Isolation of Endophytic Fungi from the Branches of Pear
2.3. Dual Culture Test for Screening Potential Biocontrol Fungi
2.4. Assessment of the Impact of Antagonistic Strains on V. pyri Hyphal Morphology
2.5. Assessment of Antagonistic Strain-Derived Culture Filtrates on V. pyri Mycelial Growth
2.6. Assessment of Antagonistic Strain-Derived Culture Filtrates on V. pyti Conidial Germination
2.7. Disease Suppression Assay
2.8. Antagonistic Strain Identification
3. Results
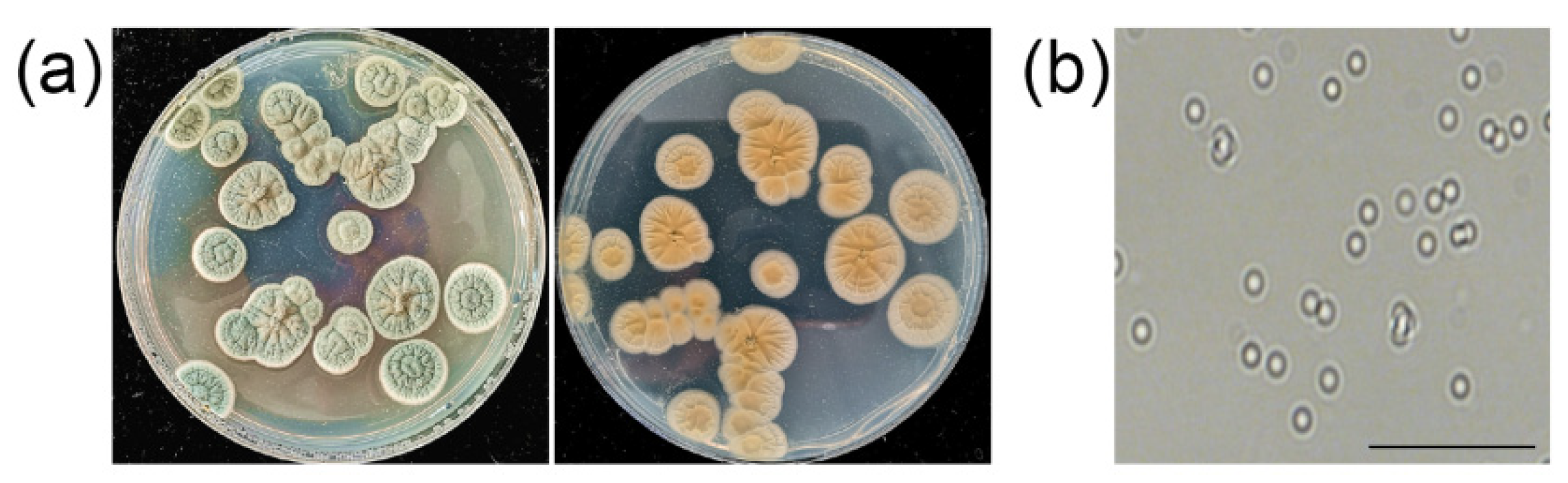
3.1. Isolation and Assessment of Antifungal Activity of ZZ1 against Phytopathogenic Fungi
3.2. Assessment of the Antagonistic Effects of Isolate ZZ1 on V. pyri Hyphal Morphology
3.3. Analysis of the Ability of ZZ1 Conidial Suspensions to Control Pear Valsa Canker Disease
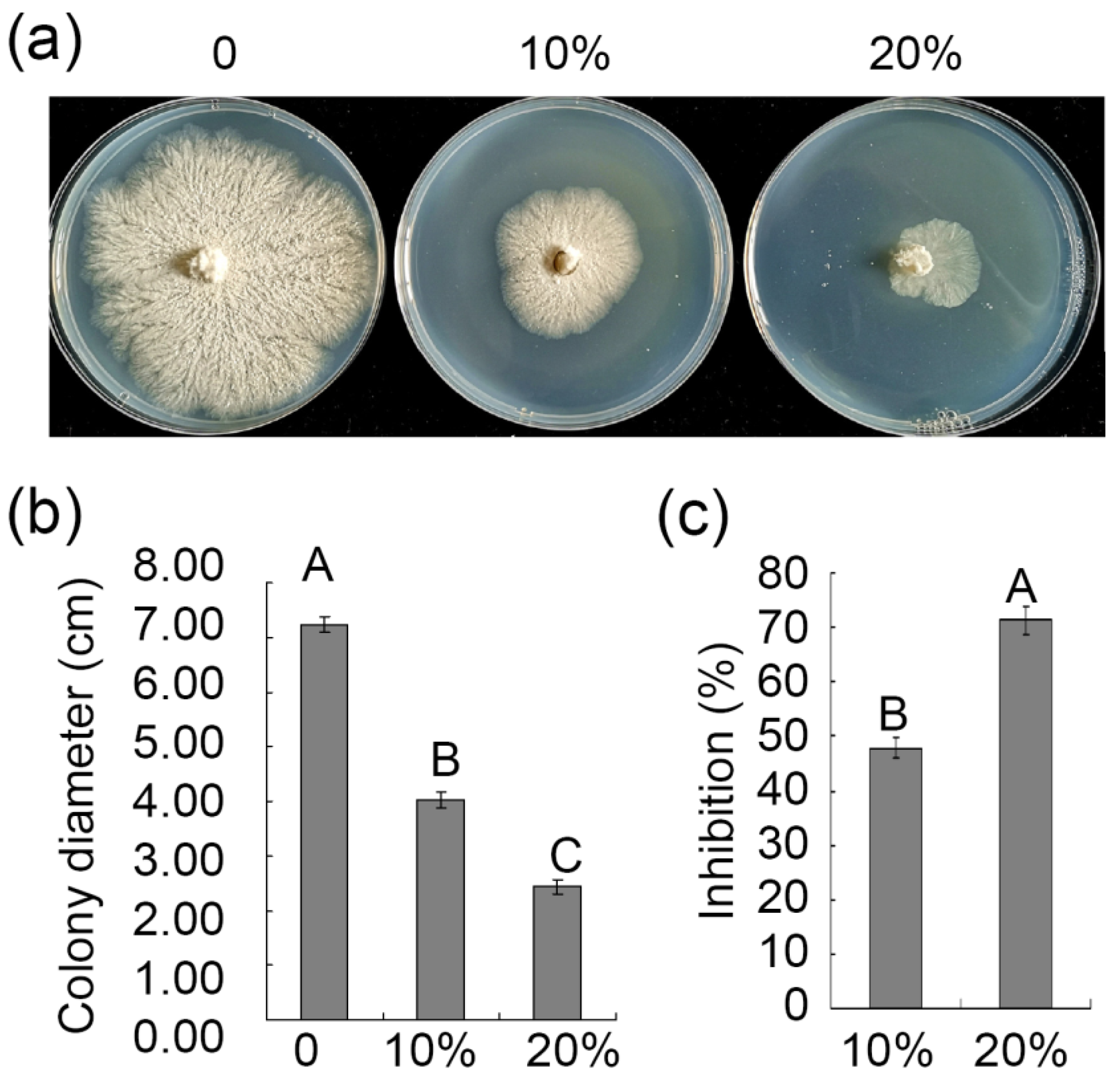
3.4. Assessment of the Ability of ZZ1 Culture Filtrates to Suppress V. pyri Mycelial Growth and Spore Germination
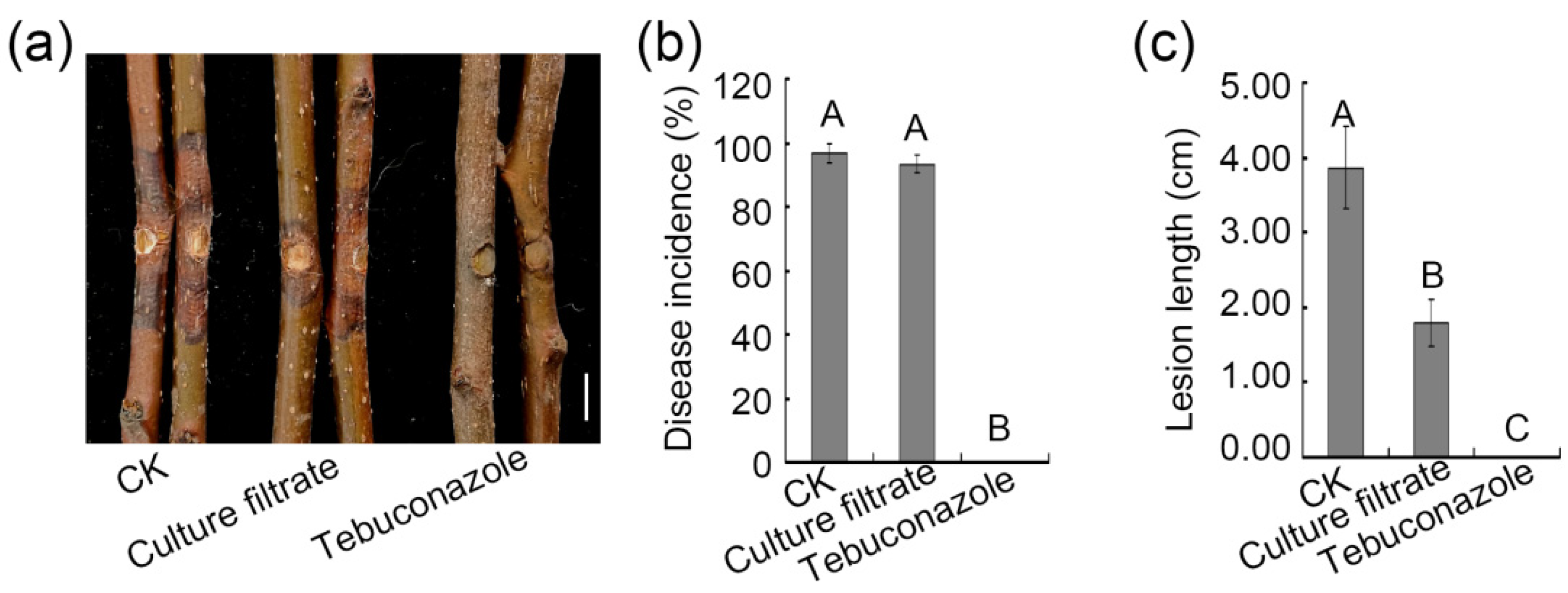
3.5. Assessment of the Ability of ZZ1 Culture Filtrates to Control Pear Valsa Canker Disease
3.6. Identification of Strain ZZ1
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kange, A.M.; Xia, A.; Si, J.; Li, B.; Zhang, X.; Ai, G.; He, F.; Dou, D. The fungal-specific transcription factor VpFSTF1 is required for virulence in Valsa pyri. Front. Microbiol. 2020, 10, 2945. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.; Liu, H.; Li, Z.; Ke, X.; Dou, D.; Gao, X.; Song, N.; Dai, Q.; Wu, Y.; Xu, J.-R.; et al. Genome sequence of Valsa canker pathogens uncovers a potential adaptation of colonization of woody bark. New Phytol. 2015, 208, 1202–1216. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zang, R.; Yin, Z.; Kang, Z.; Huang, L. Delimiting cryptic pathogen species causing apple Valsa canker with multilocus data. Ecol. Evol. 2014, 4, 1369–1380. [Google Scholar] [CrossRef]
- Dong, R.; Wei, S.; Ran, K.; Wang, H.; Dong, X.; Wang, S. The identification and the pathogenicity of analysis for the pathogen of pear Valsa canker. J. Fruit Sci. 2018, 35, 135–138. [Google Scholar]
- Latz, M.A.C.; Jensen, B.; Collinge, D.B.; Jørgensen, H.J.L. Endophytic fungi as biocontrol agents: Elucidating mechanisms in disease suppression. Plant Ecol. Divers. 2018, 11, 555–567. [Google Scholar] [CrossRef] [Green Version]
- Zheng, R.; Li, S.; Zhang, X.; Zhao, C. Biological activities of some new secondary metabolites isolated from endophytic fungi: A review study. Int. J. Mol. Sci. 2021, 22, 959. [Google Scholar] [CrossRef]
- Rodriguez, R.J.; White, J.F., Jr.; Arnold, A.E.; Redman, R.S. Fungal endophytes: Diversity and functional roles. New Phytol. 2009, 182, 314–330. [Google Scholar] [CrossRef]
- Liu, Y.; Zhou, S.; Zhang, Y.; Wang, Z.; Liu, G.; Dao, L.; Kang, X.; Li, S. Field control effects of 5 fungicides on diseases of silage maize. Plant Prot. 2019, 45, 280–284. [Google Scholar]
- Rusita, I.; Sasongko, H. Effectivity of Trichoderma harzianum as bio-fungicide against moler disease and bio-stimulator of shallot growth. J. Agri-Food Sci. Technol. 2020, 1, 12–17. [Google Scholar] [CrossRef]
- Song, X.; Han, M.; He, F.; Wang, S.; Li, C.; Wu, G.; Huang, Z.; Liu, D.; Liu, F.; Laborda, P.; et al. Antifungal mechanism of dipicolinic acid and its efficacy for the biocontrol of pear Valsa canker. Front. Microbiol. 2020, 11, 958. [Google Scholar] [CrossRef]
- Gulizige, M.; Xu, L.; Li, Y.; Han, J.; Yu, F.; Luo, M. Antagonistic bacteria against Korla fragrant pear canker: Screening and control effects. Chin. Agric. Sci. Bull. 2020, 36, 127–134. [Google Scholar]
- Savita, S.A. Fungi as biological control agents. In Biofertilizers for Sustainable Agriculture and Environment; Giri, B., Prasad, R., Wu, Q.-S., Varma, A., Eds.; Springer International Publishing: Cham, Switzerland, 2019; Volume 55, pp. 395–411. [Google Scholar]
- Zhao, X.; Liu, X.; Zhao, H.; Ni, Y.; Lian, Q.; Qian, H.; He, B.; Liu, H.; Ma, Q. Biological control of Fusarium wilt of sesame by Penicillium bilaiae 47M-1. Biol. Control 2021, 158, 104601. [Google Scholar] [CrossRef]
- Chaibub, A.A.; de Sousa, T.P.; de Araújo, L.G.; de Filippi, M.C.C. Cladosporium cladosporioides C24G modulates gene expression and enzymatic activity during leaf blast suppression in rice plants. J. Plant Growth Regul. 2019, 39, 1140–1152. [Google Scholar] [CrossRef]
- Yuan, H.; Hou, H.; Zhou, Z.; Wang, L.; Tu, H. Isolation and identification of endophytic fungi JK2 antagonistic against pear Valsa canker caused by Valsa pyri. J. Fruit Sci. 2021. [Google Scholar] [CrossRef]
- Hou, H.; Zhang, H.; Zhou, Z.; Wang, L.; Yan, Z.; Wang, S. Evaluation of resistance to apple ring rot in Malus Germplasms. Acta Hortic. Sin. 2017, 44, 1559–1568. [Google Scholar]
- Xu, T.; Hu, T.; Wang, Y.; Wang, S.; Cao, K. Isolation of endophytic fungi from apple bark and their potential for biological control of Valsa ceratosperma. Acta Phytophylacica Sinica 2012, 39, 327–333. [Google Scholar]
- Wang, Y.; Li, Y.; Chang, L.; Yu, H.; Zhou, Z.; Qu, L. Isolation and identification of endophytic fungi resistant to three apple pathogens from the branches of Malus sieversii. J. Fruit Sci. 2020, 37, 390–396. [Google Scholar]
- Dai, P.; Zong, Z.; Ma, Q.; Wang, Y. Isolation, evaluation and identification of rhizosphere actinomycetes with potential application for biocontrol of Valsa mali. Eur. J. Plant Pathol. 2018, 153, 119–130. [Google Scholar] [CrossRef]
- White, T.; Bruns, T.; Lee, S.; Taylor, F.; White, T.; Lee, S.H.; Taylor, L.; Shawetaylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols; Elsevier; Academic Press: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Lousie, G.N.; Donaldson, G.C. Development of primer sets designed for use with the PCR to amplify conserved genes from filamentous ascomycetes. Appl. Environ. Microbiol. 1995, 61, 1320–1330. [Google Scholar]
- Peterson, S.W. Multilocus DNA sequence analysis shows that Penicillium biourgeianum is a distinct species closely related to P. brevicompactum and P. olsonii. Mycol. Res. 2004, 108, 434–440. [Google Scholar] [CrossRef]
- Haggag, W.M. The role of entophytic microorganisms in biocontrol of plant diseases. Life Sci. J. 2010, 7, 57–62. [Google Scholar]
- Toghueo, R.M.K.; Boyom, F.F. Endophytic Penicillium species and their agricultural, biotechnological, and pharmaceutical applications. 3 Biotech 2020, 10, 1–35. [Google Scholar] [CrossRef] [PubMed]
- Sreevidya, M.; Gopalakrishnan, S.; Melø, T.M.; Simic, N.; Bruheim, P.; Sharma, M.; Srinivas, V.; Alekhya, G. Biological control of Botrytis cinerea and plant growth promotion potential by Penicillium citrinum in chickpea (Cicer arietinum L.). Biocontrol. Sci. Technol. 2015, 25, 739–755. [Google Scholar] [CrossRef]
- Ma, Y.; Chang, Z.Z.; Zhao, J.T.; Zhou, M.G. Antifungal activity of Penicillium striatisporum Pst10 and its biocontrol effect on Phytophthora root rot of chilli pepper. Biol. Control 2008, 44, 24–31. [Google Scholar] [CrossRef]
- Ab De L-Motaal, F.F.; Nassar, M.S.M.; El-Zayat, S.A.; El-Sayed, M.A.; Ito, S.I. Antifungal activity of endophytic fungi isolated from Egyptian henbane (Hyoscyamus muticus L.). Pak. J. Bot. 2010, 42, 2883–2894. [Google Scholar]
- Ting, A.; Mah, S.W.; Tee, C.S. Evaluating the feasibility of induced host resistance by endophytic isolate Penicillium citrinum BTF08 as a control mechanism for Fusarium wilt in banana plantlets. Biol. Control 2012, 61, 155–159. [Google Scholar] [CrossRef]
- González-Estrada, R.R.; de Jesus Ascencio-Valle, F.; Ragazzo-Sánchez, J.A.; Santoyo, M.C. Use of a marine yeast as a biocontrol agent of the novel pathogen Penicillium citrinum on Persian Lime. Emir. J. Food Agric. 2017, 29, 114–122. [Google Scholar] [CrossRef] [Green Version]
- Mohammadiani, E.; Aliakbarlu, J.; Ownagh, A.; Kaboudari, A. Antifungal interactions of Persian shallot (Allium hirtifolium) extracts and potassium sorbate against Aspergillus flavus and Penicillium citrinum. Flavour Fragr. J. 2021, 36, 332–338. [Google Scholar] [CrossRef]
- Thamara, C.C.; Mariana, C.F.; Luiz, H.R.; Ana, M.; Enio, N. Penicillium citrinum and Penicillium mallochii: New phytopathogens of orange fruit and their control using chitosan. Carbohydr. Polym. 2020, 234, 115918. [Google Scholar]
- Luo, H.; Zhen, Q.; Deng, Y.; Deng, Z.; Lin, W. Two polyketides produced by endophytic Penicillium citrinum DBR-9 from medicinal plant Stephania kwangsiensis and their antifungal activity against plant pathogenic fungi. Nat. Prod. Commun. 2019, 14. [Google Scholar] [CrossRef] [Green Version]
- Vinale, F.; Nicoletti, R.; Lacatena, F.; Marra, R.; Sacco, A.; Lombardi, N.; d’Errico, G.; Digilio, M.C.; Lorito, M.; Woo, S.L. Secondary metabolites from the endophytic fungus Talaromyces pinophilus. Nat. Prod. Res. 2017, 31, 1778–1785. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ali, T.; Pham, T.M.; Ju, K.S.; Rakotondraibe, H.L. Ent-homocyclopiamine B, a prenylated indole alkaloid of biogenetic interest from the endophytic fungus Penicillium concentricum. Molecules 2019, 24, 218. [Google Scholar] [CrossRef] [PubMed] [Green Version]





| Endophytic Strains | Inhibition (%) |
|---|---|
| ZZ1 | 95.2 ± 2.1 |
| ZZ2 | 7.4 ± 0.8 |
| ZZ3 | 3.9 ± 0.7 |
| ZZ4 | 5.7 ± 1.1 |
| ZZ5 | 2.7 ± 0.8 |
| ZZ6 | 3.5 ± 1.2 |
| ZZ7 | 12.3 ± 1.4 |
| ZZ8 | 32.5 ± 3.1 |
| ZZ9 | 13.5 ± 2.3 |
| ZZ10 | 5.3 ± 1.6 |
| ZZ11 | 3.5 ± 0.5 |
| ZZ12 | 55.0 ± 2.6 |
| ZZ13 | 22.3 ± 3.1 |
| ZZ14 | 15.4 ± 2.4 |
| ZZ15 | 5.9 ± 1.5 |
| ZZ16 | 14.6 ± 2.4 |
| ZZ17 | 58.7 ± 3.2 |
| ZZ18 | 8.6 ± 0.9 |
| ZZ19 | 5.4 ± 0.4 |
| ZZ20 | 23.5 ± 3.5 |
| ZZ21 | 16.5 ± 2.6 |
| ZZ22 | 7.4 ± 1.8 |
| ZZ23 | 8.9 ± 1.1 |
| ZZ24 | 3.3 ± 0.8 |
| ZZ25 | 5.4 ± 1.3 |
| ZZ26 | 8.4 ± 2.1 |
| ZZ27 | 4.9 ± 0.5 |
| ZZ28 | 9.4 ± 1.7 |
| ZZ29 | 10.5 ± 2.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuan, H.; Shi, B.; Huang, T.; Zhou, Z.; Wang, L.; Hou, H.; Tu, H. Biological Control of Pear Valsa Canker Caused by Valsa pyri Using Penicillium citrinum. Horticulturae 2021, 7, 198. https://doi.org/10.3390/horticulturae7070198
Yuan H, Shi B, Huang T, Zhou Z, Wang L, Hou H, Tu H. Biological Control of Pear Valsa Canker Caused by Valsa pyri Using Penicillium citrinum. Horticulturae. 2021; 7(7):198. https://doi.org/10.3390/horticulturae7070198
Chicago/Turabian StyleYuan, Hongbo, Bingke Shi, Tianxiang Huang, Zengqiang Zhou, Li Wang, Hui Hou, and Hongtao Tu. 2021. "Biological Control of Pear Valsa Canker Caused by Valsa pyri Using Penicillium citrinum" Horticulturae 7, no. 7: 198. https://doi.org/10.3390/horticulturae7070198
APA StyleYuan, H., Shi, B., Huang, T., Zhou, Z., Wang, L., Hou, H., & Tu, H. (2021). Biological Control of Pear Valsa Canker Caused by Valsa pyri Using Penicillium citrinum. Horticulturae, 7(7), 198. https://doi.org/10.3390/horticulturae7070198






