Inferring the Potential Geographic Distribution and Reasons for the Endangered Status of the Tree Fern, Sphaeropteris lepifera, in Lingnan, China Using a Small Sample Size
Abstract
1. Introduction
2. Study Area and Methods
2.1. Field Investigation
2.2. Reintroduction Experiments
2.3. Gametophytic Development
2.4. Ecological Niche Modeling
3. Results
3.1. Germination and Fertilization Observations and Experiments
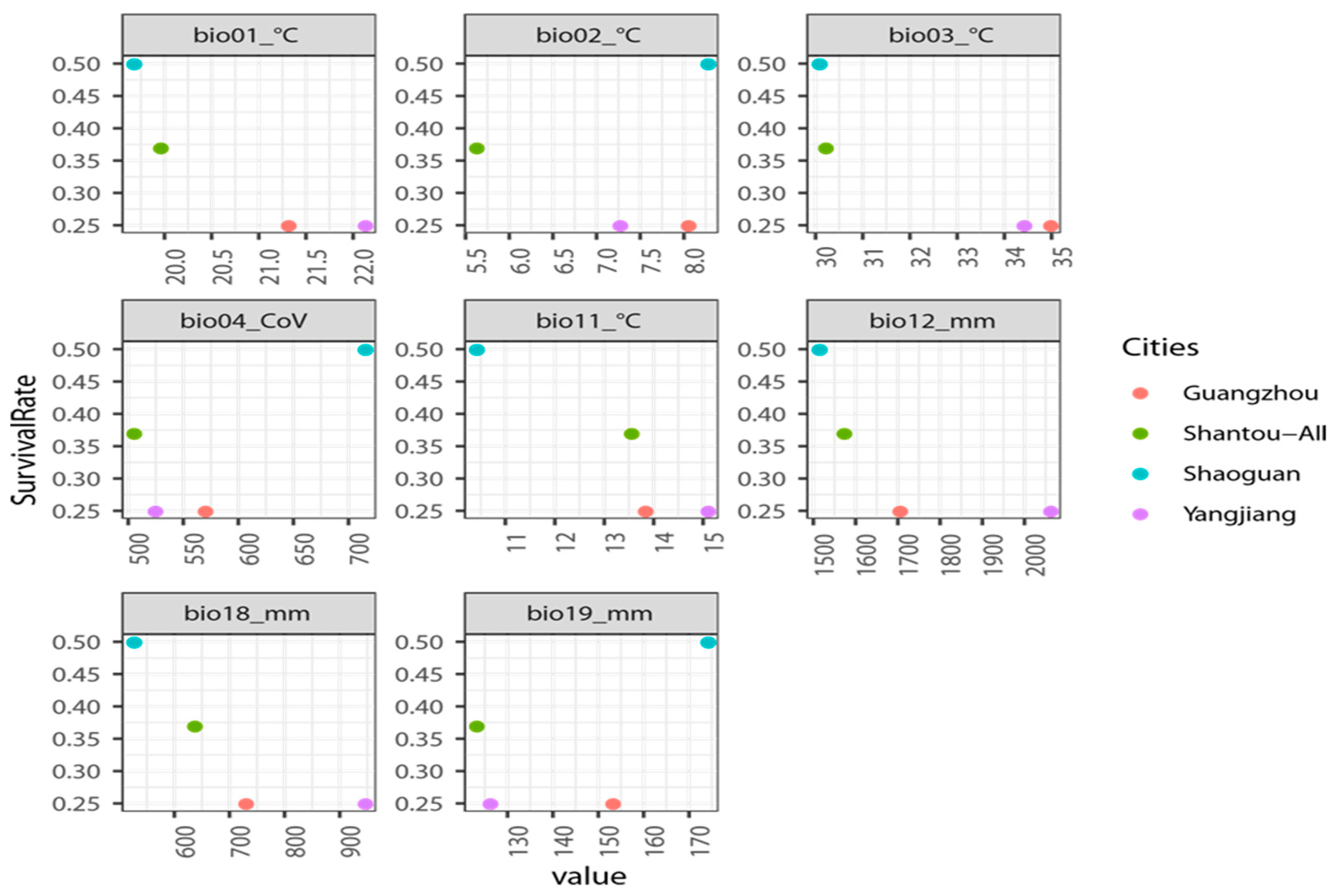
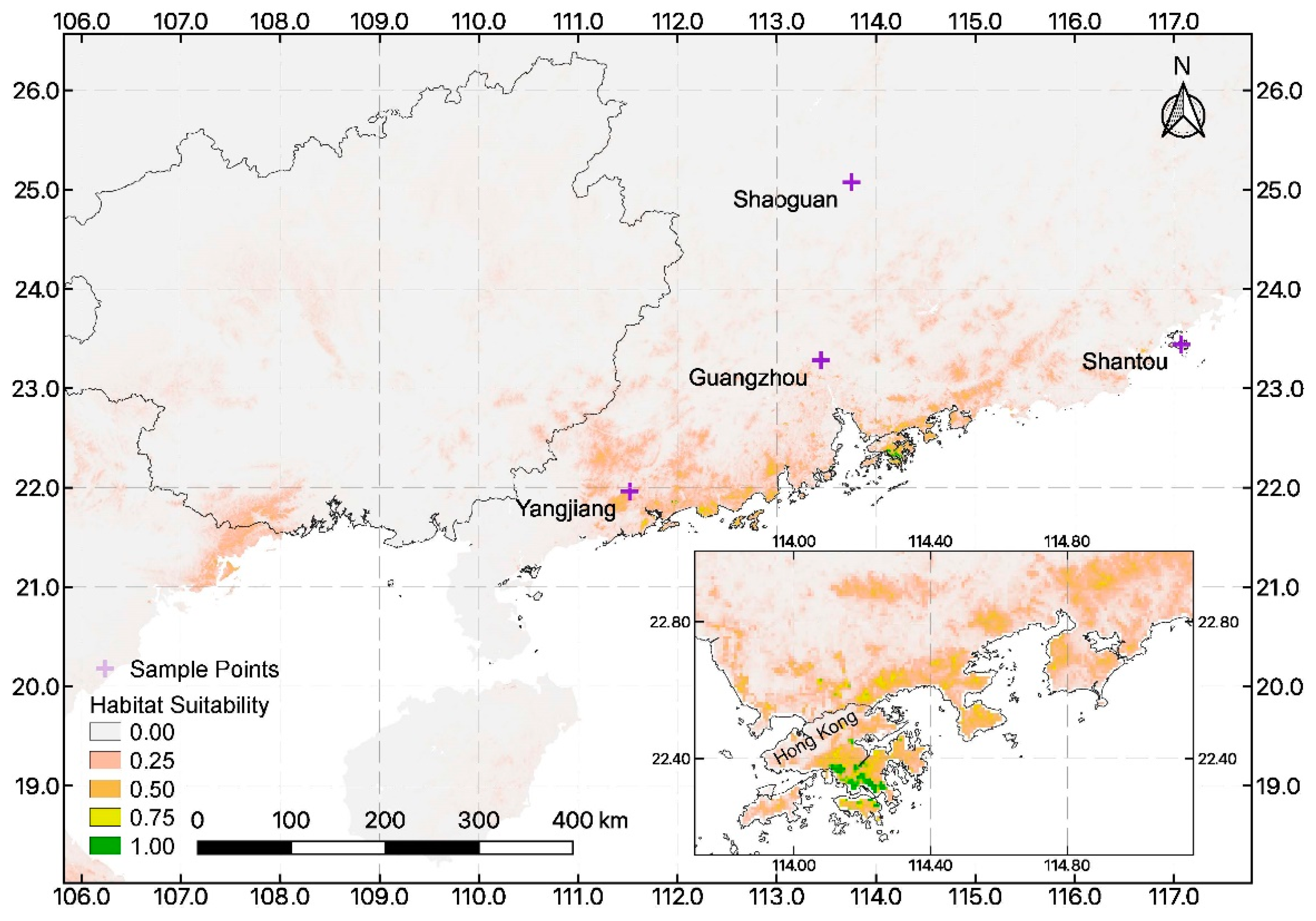
3.2. Ecological Niche Modeling and Importance of Environmental Variables
3.3. Community Composition
3.4. Preliminary Reintroduction Experiment
4. Discussion
4.1. Factors and Reasons Affecting the Distribution of Sphaeropteris lepifera during Life History
4.2. Factors and Causes of Community Composition Affecting the Distribution of Sphaeropteris lepifera
4.3. Suitable Habitat for Reintroduction within the Chinese Mainland and Near-Shore Islands
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ehlers, E.; Krafft, T. The “Anthropocene”. In Earth System Science in the Anthropocene; Springer: Berlin/Heidelberg, Germany, 2006; pp. 13–18. [Google Scholar]
- Harris, A.; Stefanie, I.B.; Aarón, R. Long distance dispersal in the assembly of floras: A review of progress and prospects in north America. J. Syst. Evol. 2018, 56, 430–448. [Google Scholar] [CrossRef]
- Xu, W.B.; Svenning, J.C.; Chen, G.K.; Zhang, M.G.; Huang, J.H.; Chen, B.; Ordonez, A.; Ma, K.P. Human Activities Have Opposing Effects on Distributions of Narrow—Ranged and Widespread Plant Species in China. Proc. Natl. Acad. Sci. USA 2019, 116, 266–274. [Google Scholar] [CrossRef]
- Peterson, A.T. Uses and requirements of ecological niche models and related distributional models. Biodivers. Inform. 2006, 3, 59–72. [Google Scholar] [CrossRef]
- Blonder, B. Hypervolume concepts in niche- and trait-based ecology. Ecography 2018, 41, 1441–1455. [Google Scholar] [CrossRef]
- Peterson, A.T.; Pape, M.; Soberón, J. Mechanistic and correlative models of ecological niches. Eur. J. Ecol. 2015, 1, 28–38. [Google Scholar] [CrossRef]
- Phillips, S.J.; Anderson, R.P.; Schapire, R.E. Maximum Entropy Modeling of Species Geographic Distributions. Ecol. Model. 2006, 190, 231–259. [Google Scholar] [CrossRef]
- Carsten, F.D.; Stanislaus, J.S.; Juliano, C.; Isabelle, C.; Catherine, G.; Florian, H.; Michael, K.; Xavier, M.; Römermann, C.; Schröder, B.; et al. Correlation and process in species distribution models: Bridging a dichotomy. J. Biogeogr. 2012, 39, 2119–2131. [Google Scholar]
- GBIF.org. GBIF Occurrence Download. Available online: https://doi.org/10.15468/dl.j8abq5 (accessed on 5 July 2021). [CrossRef]
- Zhang, X.C.; Nishida, H. Cyatheaceae. In Flora of China; Wu, Z.Y., Raven, P.H., Hong, D.Y., Eds.; Science Press: Beijing, China, 2013; Volume 2, pp. 134–138. [Google Scholar]
- Gu, Y.F.; Jiang, R.H.; Liu, B.D.; Yan, Y.H. Sphaeropteris guangxiensis Y. F. Gu & Y. H. Yan (Cyatheaceae), a new species of tree fern from Southern China. Phytotaxa 2021, 518, 69–74. [Google Scholar]
- National Forestry and Grassland Administration, Ministry of Agriculture and Rural Affairs. List of National Key Protected Wild Plants in China. 2021-9-7. Available online: http://www.gov.cn/zhengce/zhengceku/2021-09/09/content_5636409.htm (accessed on 25 September 2021).
- Ye, H.G. Chinese Medicinal Plants; Chemical Industry Press: Beijing, China, 2017; Volume 25, pp. 356–357. [Google Scholar]
- Shi, L. Ornamental Ferns of China; China Forestry Publishing House: Beijing, China, 2002; p. 87. [Google Scholar]
- Fujian Forestry Department. Sphaeropteris lepifera (J. Sm. ex Hook.). For. China 2003, 11, 29. [Google Scholar]
- Morigengaowa, H.S.; Liu, B.; Kang, M.; Yan, Y. One or More Species? GBS Sequencing and Mmorphological Traits Evidence Reveal Species Diversification of Sphaeropteris brunoniana in China. Biodivers. Sci. 2019, 27, 36–44. [Google Scholar] [CrossRef]
- Huang, C.H.; Qi, X.P.; Chen, D.Y.; Qi, J.; Ma, H. Recurrent genome duplication events likely contributed to both the ancient and recent rise of ferns. J. Integr. Plant Biol. 2020, 62, 433–455. [Google Scholar] [CrossRef]
- Chen, X.X.; Pan, T.Z. The Report of a New Record Genera (Sphaeropteris) and a New Record Species (Cyathea lepifera) from Zhejiang, China. J. Wenzhou Univ. 2016, 37, 34–37. [Google Scholar]
- Yin, H. Rare and Endangered Plants in China; China Forestry Publishing House: Beijing, China, 2013; pp. 13–26. [Google Scholar]
- Zhao, X.Q. The Rare Wild Tree Fern Found in Xiamen: Sphaeropteris lepifera (J. Sm. ex Hook.) (Cycinaceae). Wuyi Sci. J. 1983, 3, 181. [Google Scholar]
- Ebihara, A.; Yamaoka, A.; Mizukami, N.; Sakoda, A.; Nitta, J.H.; Imaichi, R. A survey of the fern gametophyte flora of japan: Frequent independent occurrences of noncordiform gametophytes. Am. J. Bot. 2013, 100, 735–743. [Google Scholar] [CrossRef] [PubMed]
- Pelser, P.B.; Barcelona, J.F.; Nickrent, D.L. (Eds.) 2011 Onwards. Co’s Digital Flora of the Philippines. Available online: www.philippineplants.org (accessed on 20 September 2021).
- Barcelona, J.F. The Taxonomy and Ecology of the Pteridophytes of Mt. Iraya and Vicinity, Batan Island, Batanes Province, Northern Philippines. In Pteridology in the New Millennium; Springer: Dordrecht, The Netherlands, 2003. [Google Scholar] [CrossRef]
- Holttum, R.E. Tree-Ferns of the Genus Cyathea Sm. in Asia (Excluding Malaysia). Kew Bull. 1965, 19, 463–487. [Google Scholar] [CrossRef]
- Wang, B.S.; Yu, S.X.; Peng, S.L.; Li, M.G. Handbook of Plant Community Experiments; Guangdong Higher Education Press: Guangzhou, China, 1996. [Google Scholar]
- Ji, S.B.; Xiang, J.Y.; Liu, B.D.; Ji, H.L. Behavior and Morphology of Sphaeropteris lepifera (J. Sm. ex Hook.) (Cyatheaceae) Swimming Sperm. Acta Bot. Boreali-Occident. Sin. 2018, 38, 92–97. [Google Scholar]
- Zhang, X.; Xiang, J.J.; Xiao, X.; Ji, S.B.; Liu, B.D. Influence of Lead and Cadmium Stress on Sperm’s Behavior and Morphology of Sphaeropteris lepifera (J. Sm. ex Hook.) (Cyatheaceae). Acta Bot. Boreali-Occident. Sin. 2019, 5, 776–783. [Google Scholar]
- Cheng, M.C.; You, C.F. Sources of major ions and heavy metals in rainwater associated with typhoon events in southwestern taiwan. J. Geochem. Explor. 2010, 105, 106–116. [Google Scholar] [CrossRef]
- Jiao, Y.; Wang, H.; Zhang, S.Z. Fern Atlas; China Forestry Publishing House: Beijing, China, 2014. [Google Scholar]
- Wang, J.J.; Zhang, X.C.; Liu, B.D.; Cheng, X. Gametophyte Development of Three Species in Cyatheaceae. J. Trop. Subtrop. Bot. 2007, 15, 115–120. [Google Scholar]
- Wani, I.A.; Verma, S.; Mustahq, S.; Alsahli, A.A.; Alyemeni, M.N.; Tariq, M.; Pant, S. Ecological analysis and environmental niche modelling of D. hatagirea (D. Don) Soo: A conservation approach for critically endangered medicinal orchid. Saudi J. Biol. Sci. 2021, 28, 2109–2122. [Google Scholar] [CrossRef]
- Papeş, M.; Gaubert, P. Modelling ecological niches from low numbers of occurrences: Assessment of the conservation status of poorly known viverrids (Mammalia, Carnivora) across two continents. Divers. Distrib. 2010, 13, 890–902. [Google Scholar] [CrossRef]
- Banks, S.C.; Finlayson, G.R.; Lawson, S.J.; Lindenmayer, D.B.; Paetkau, D.; Ward, S.J.; Taylor, A.C. The effects of habitat fragmentation due to forestry plantation establishment on the demography and genetic variation of a marsupial carnivore, Antechinus agilis. Biol. Conserv. 2005, 122, 581–597. [Google Scholar] [CrossRef]
- Rhodes, J.R.; Wiegand, T.; McAlpine, C.A.; Callaghan, J.; Lunney, D.; Bowen, M.; Possingham, H.P. Modeling species’ dis-tributions to improve conservation in semiurban landscapes: Koala case study. Conserv. Biol. 2006, 20, 449–459. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Fu, L.; Wang, F.G.; Xing, F.W. Michelia guang¬dongensis (Magnoliaceae), an Endangered Plant Species with Ex-tremely Small Population, should be Evaluated as CR C2a (i); D. Biodivers. Sci. 2017, 25, 91–93. [Google Scholar] [CrossRef][Green Version]
- Ren, Z.; Chen, L.; Peng, H. Orophea yunnanensis should be Listed as a Plant Species with Extremely Small Population. Biodivers. Sci. 2016, 24, 358–359. (In Chinese) [Google Scholar] [CrossRef]
- Ptsch, R.; Jaková, A.; Chytr, M.; Kucherov, I.B.; Janssen, J. Making them visible and usable—vegetation lot observations from fennoscandia based on historical species quantity scales. Appl. Veg. Sci. 2019, 22, 465–473. [Google Scholar] [CrossRef]
- Ma, H.N.; Li, Y.; Tan, L.Y.; Liu, B.D. Spore Propagation and Rejuvenation of Sphaeropteris lepifera (J. Sm. ex Hook.). Acta Hortic. Sin. 2010, 37, 1679–1684. [Google Scholar]
- Robert, J.H.; Phillips, S.; Leathwick, J.; Elith, J. Dismo: Species Distribution Modeling. R Package Version 1.3-3. Available online: https://CRAN.R-project.org/package=dismo (accessed on 10 September 2020).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria; Available online: https://www.R-project.org/ (accessed on 12 June 2021).
- Tennekes, M. tmap: Thematic Maps in R. J. Stat. Softw. 2018, 84, 1–39. [Google Scholar] [CrossRef]
- Fick, S.E.; Hijmans, R.J. Worldclim 2: New 1-km spatial resolution climate surfaces for global land areas. Int. J. Climatol. 2017, 37, 4302–4315. [Google Scholar] [CrossRef]
- Booth, T.H.; Nix, H.A.; Busby, J.R.; Hutchinson, M.F. Bioclim: The first species distribution modelling package, its early applications and relevance to most current MaxEnt studies. Divers. Distrib. 2014, 20, 1–9. [Google Scholar]
- Zuo, H.; Hutchinson, M.F.; Mcmahon, J.P.; Nix, H.A. Developing a mean monthly climatic database for China and Southeast Asias. In Proceedings of the International Workshop Held in Bangkok, Bangkok, Thailand, 27–30 March 1995.
- Buchhorn, M.; Smets, B.; Bertels, L.; Lesiv, M.; Tsendbazar, N.-E.; Masiliunas, D.; Linlin, L.; Herold, M.; Fritz, S. Copernicus Global Land Service: Land Cover 100 m: Collection 3: Epoch 2019: Globe (Version V3.0.1) [Data set]. Zenodo. 2020. Available online: https://land.copernicus.eu/global/products/lc (accessed on 10 July 2021).
- WorldPop (www.worldpop.org-School of Geography and Environmental Science, University of Southampton; Department of Geography and Geosciences, University of Louisville; Departement de Geographie, Universite de Namur) and Center for International Earth Science Information Network (CIESIN), Columbia University. Global High Resolution Population Denominators Project-Funded by the Bill and Melinda Gates Foundation (OPP1134076). 2018. Available online: https://www.worldpop.org/geodata/summary?id=94 (accessed on 10 September 2020).
- Chitta, R.D.; Jamir, N.S.; Zubenthung, P.K. Distribution Prediction Model of a Rare Orchid Species (Vanda bicolor Griff.) Using Small Sample Size. Am. J. Plant Sci. 2017, 8, 1388–1398. [Google Scholar]
- Su, Z.R.; Zhang, H.D. The Relationship between the Flora of Guangxi and the Flora of Neighboring Areas. J. South China Agric. Univ. 1994, 15, 38–43. [Google Scholar]
- Riano, K.; Briones, O. Sensitivity of three tree ferns during their first phase of life to the variation of solar radiation and water availability in a mexican cloud forest. Am. J. Bot. 2015, 102, 1472–1481. [Google Scholar] [CrossRef] [PubMed]
- Brock, J.M.R.; Burns, B.R.; Perry, G.; Lee, W.G. Gametophyte niche differences among sympatric tree ferns. Biol. Lett. 2019, 15, 20180659. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.B. Carbon cost of plant nitrogen acquisition: A mechanistic, globally applicable model of plant nitrogen uptake, retranslocation, and fixation. Glob. Biogeochem. Cycles 2010, 24. [Google Scholar] [CrossRef]
- Brock, J.; Perry, G.; Lee, W.G.; Burns, B.R. Tree fern ecology in New Zealand: A model for southern temperate rainforests. For. Ecol. Manag. 2016, 375, 112–126. [Google Scholar] [CrossRef]
- Bernabe, N.; Williams-Linera, G.; Palacios-Rios, M. Tree ferns in the interior and at the edge of a mexican cloud forest remnant: Spore germination and sporophyte survival and establishment1. Biotropica 1999, 31, 83–88. [Google Scholar]
- Rotach, P. In situ conservation methods. In Conservation and Management of Forest Genetic Resources in Europe; Geburek, T., Turok, J., Eds.; Arbora Publishers: Zvolen, Slovakia, 2005; pp. 535–565. [Google Scholar]
- Wei, K.H.; Liang, Y.; Yang, C.; Li, L.X.; Hu, Y.; Liao, J.H.; Gao, W.Y. Conservation Research of Bletilla striata. Mod. Chin. Med. 2019, 21, 689–693. [Google Scholar]



| Source | Province | City | Longitude | Latitude |
|---|---|---|---|---|
| field site | Guangdong | Shantou | 117.072 | 23.444 |
| ex situ | Guangdong | Shaoguan | 113.751 | 25.076 |
| ex situ | Guangdong | Yangjiang | 111.520 | 21.963 |
| ex situ | Guangdong | Shantou | 117.072 | 23.444 |
| ex situ | Guangdong | Guangzhou | 113.444 | 23.283 |
| ex situ | Guangdong | Shantou | 117.072 | 23.444 |
| Code | Environmental Factor | Percent Contribution | Permutation Importance |
|---|---|---|---|
| Bio01 | Annual Mean Temperature | 0.4 | 0 |
| Bio02 | Mean Diurnal Range (Mean of monthly (max temp-min temp)) | 29.3 | 4.9 |
| Bio03 | Isothermality (Bio2/Bio7) (× 100) | 3.7 | 7.4 |
| Bio04 | Temperature Seasonality (standard deviation × 100) | 24.7 | 0.6 |
| Bio05 | Max Temperature of Warmest Month | 1 | 1.2 |
| Bio06 | Min Temperature of Coldest Month | 0.1 | 0.1 |
| Bio07 | Temperature Annual Range (Bio5-Bio6) | 4.1 | 0.4 |
| Bio08 | Mean Temperature of Wettest Quarter | 0.6 | 1.1 |
| Bio09 | Mean Temperature of Driest Quarter | 0.1 | 0 |
| Bio10 | Mean Temperature of Warmest Quarter | 0.5 | 0.2 |
| Bio11 | Mean Temperature of Coldest Quarter | 2.1 | 5.7 |
| Bio12 | Annual Precipitation | 11.7 | 3.7 |
| Bio13 | Precipitation of Wettest Month | 0.1 | 0.3 |
| Bio14 | Precipitation of Driest Month | 0.9 | 0.8 |
| Bio15 | Precipitation Seasonality (Coefficient of Variation) | 0.2 | 0.5 |
| Bio16 | Precipitation of Wettest Quarter | 0 | 0 |
| Bio17 | Precipitation of Driest Quarter | 0.3 | 0.3 |
| Bio18 | Precipitation of Warmest Quarter | 4.7 | 3.7 |
| Bio19 | Precipitation of Coldest Quarter | 13.5 | 66.6 |
| Slope | Slope | 0.3 | 0.1 |
| Aspect | Aspect | 0.3 | 0.3 |
| Landcover | Landcover | 0.1 | 0.1 |
| Human
population | Human population | 1.4 | 1.9 |
| Life Form | Frequency | Species | Drude Abundance | Relative Cover (%) |
|---|---|---|---|---|
| Tree | Dominant species | Machilus chinensis (Benth.) Hemsl. | Soc | 211 |
| Schefflera heptaphylla (L.) Frodin | Soc | 136.5 | ||
| Acacia confusa Merr. | Soc | 90 | ||
| Cunninghamia lanceolata (Lamb.) Hook. | Soc | 89.1 | ||
| Companion species | Ilex viridis Champ. ex Benth. | Cop3 | 52.8 | |
| Litsea glutinosa (Lour.) C.B. Rob. | Cop2 | 36 | ||
| Aporosa dioica (Roxb.) Müll. Arg. | Sp | 9 | ||
| Rare species | Archidendron lucidum (Benth.) I.C. Nielsen | Uni | 1.28 | |
| Shrub | Dominant species | Psychotria rubra (Lour.) Poir. | Sp | 9.6 |
| Companion species | Camellia sinensis (L.) Kuntze | Uni | 2.92 | |
| Diplospora dubia (Lindl.) Masam. | Uni | 1.44 | ||
| Herb | Dominant species | Blechnum orientale L. | Sol | 4.5 |
| Boehmeria nivea (L.) Gaudich. | Sol | 3.5 | ||
| Companion species | Eleutherococcus trifoliatus (L.) S.Y. Hu | Sol | 3 | |
| Cyclosorus parasiticus (L.) Farw. | Uni | 2.2 | ||
| Alocasia odora (Roxb.) K. Koch | Uni | 2 | ||
| Dryopteris fuscipes C. Chr. | Uni | 1.2 | ||
| Pteris fauriei Hieron. | Uni | 0.6 | ||
| Rare species | Deparia lancea (Thunb.) Fraser-Jenk. | Uni | 0.5 | |
| Adiantum flabellulatum L. | Uni | 0.4 | ||
| Lygodium japonicum (Thunb.) Sw. | Uni | 0.3 | ||
| Mussaenda pubescens W.T. Aiton | Uni | 0.3 | ||
| Alpinia hainanensis K. Schum. | Uni | 0.3 | ||
| Solanum Americanum Mill. | Uni | 0.3 | ||
| Liriope spicata (Thunb.) Lour. | Uni | 0.2 |
| Nanao Island | Maofeng Mountain | Renhua County | Bajia Town | |
|---|---|---|---|---|
| City | Shantou | Guangzhou | Shaoguan | Yangchun |
| Longitude | 117.07190393 | 113.444498 | 113.751431 | 111.520311 |
| Latitude | 23.44393082 | 23.283165 | 25.07638 | 21.963179 |
| Mean annual temperature | 21.5 °C | 21.9 °C | 19.6 °C | 22.3 °C |
| Average annual precipitation | 1372.5 mm | 1623.6 mm | 1665 mm | 2392.3 mm |
| Frost season | no | no | 57 d | no |
| Annual minimum temperature | 8 °C | 3 °C | −3 °C | 2 °C |
| Coldest month | January | January | January | January |
| Mean temperature of the coldest month | 14 °C | 15 °C | 11 °C | 16 °C |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, X.; Harris, A.; Cui, Y.; Dai, Y.; Hu, H.; Yu, X.; Jiang, R.; Wang, F. Inferring the Potential Geographic Distribution and Reasons for the Endangered Status of the Tree Fern, Sphaeropteris lepifera, in Lingnan, China Using a Small Sample Size. Horticulturae 2021, 7, 496. https://doi.org/10.3390/horticulturae7110496
Wei X, Harris A, Cui Y, Dai Y, Hu H, Yu X, Jiang R, Wang F. Inferring the Potential Geographic Distribution and Reasons for the Endangered Status of the Tree Fern, Sphaeropteris lepifera, in Lingnan, China Using a Small Sample Size. Horticulturae. 2021; 7(11):496. https://doi.org/10.3390/horticulturae7110496
Chicago/Turabian StyleWei, Xueying, AJ Harris, Yuwen Cui, Yangwu Dai, Hanjia Hu, Xiaoling Yu, Rihong Jiang, and Faguo Wang. 2021. "Inferring the Potential Geographic Distribution and Reasons for the Endangered Status of the Tree Fern, Sphaeropteris lepifera, in Lingnan, China Using a Small Sample Size" Horticulturae 7, no. 11: 496. https://doi.org/10.3390/horticulturae7110496
APA StyleWei, X., Harris, A., Cui, Y., Dai, Y., Hu, H., Yu, X., Jiang, R., & Wang, F. (2021). Inferring the Potential Geographic Distribution and Reasons for the Endangered Status of the Tree Fern, Sphaeropteris lepifera, in Lingnan, China Using a Small Sample Size. Horticulturae, 7(11), 496. https://doi.org/10.3390/horticulturae7110496






