Moringa oleifera Lam.: A Phytochemical and Pharmacological Overview
Abstract
:1. Introduction
2. Plant Description
3. Phytochemistry
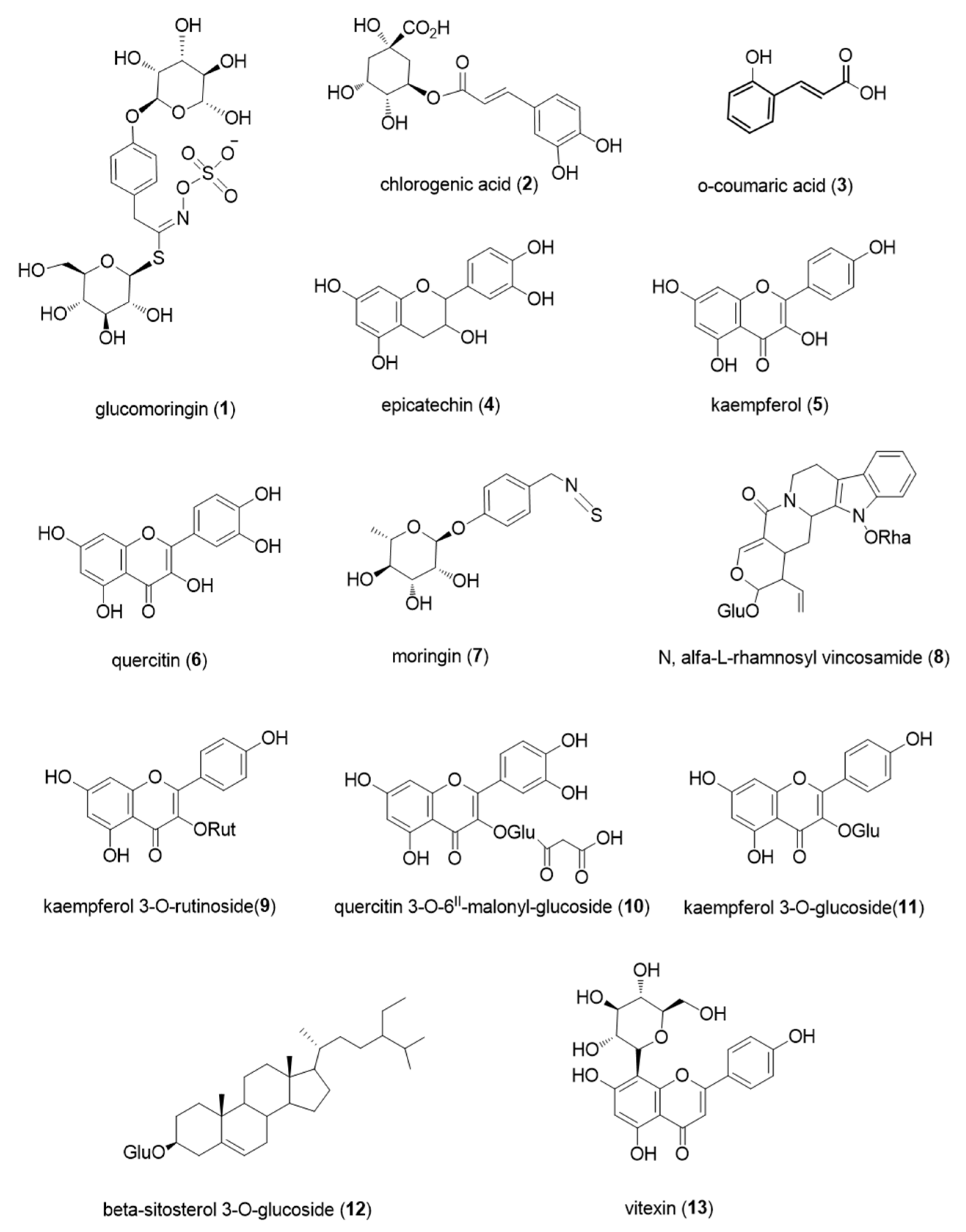
3.1. Non-Polar Compounds
3.2. Polar Compounds
4. Pharmacology
4.1. Leaves: Cardiovascular Activity
4.2. Leaves: Anti-Inflammatory Activity
4.3. Leaves: Antihypertensive Activity
4.4. Leaves: Radical Scavenging and Antioxidant Activity
4.5. Leaves: Anticancer Activity
4.6. Leaves: Hepatoprotective and Nutraceutical Activity
4.7. Leaves: Antimicrobial Activity
4.8. Leaves, Stems, Pods: Anti-Allergic Activity
4.9. Seeds: Anti-Inflammatory Activity
4.10. Seeds: Antiviral Activity
4.11. Seeds: Anticancer Activity
4.12. Seeds: Antioxidant Activity
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Olson, M.E.; Fahey, J.W. Moringa oleifera: Un árbol multiusos para las zonas tropicales secas. Rev. Mex. Biodivers 2011, 82, 1071–1082. [Google Scholar] [CrossRef] [Green Version]
- Olson, M.E. Moringaceae. In Flora of North America North of Mexico. Flora N. Am. Assoc. 2010, 7, 167–169. [Google Scholar]
- Yadava, U.L. Exotic horticultural plants with commercial potential in the UnitedStates market: Introduction to the Workshop. HortScience 1996, 31, 764–765. [Google Scholar] [CrossRef] [Green Version]
- Zheng, Y.; Zhang, Y.; Wu, J. Yield and quality of Moringa oleifera under different planting densities and cutting heights in southwest China. Ind. Crops Prod. 2016, 91, 88–96. [Google Scholar] [CrossRef]
- Alli, E.; And, R.; Arumugam, T. Moringa oleifera (Lam)-A nutritional powerhouse. J. Crop Weed 2017, 13, 238–246. [Google Scholar]
- Fahey, J.W. Moringa oleifera: A review of the medicinal potential. Acta Hortic. 2017, 1158, 209–224. [Google Scholar] [CrossRef]
- Renityas, N.N. The Effectiveness of Moringa Leaves Extract and Cancunpoint Massage Towards Breast Milk Volume on Breastfeeding Mothers. J. Ners Kebidanan (J. Ners Midwifery) 2018, 5, 150–153. [Google Scholar] [CrossRef] [Green Version]
- Fahey, J.R. Microbiological monitoring of laboratory mice. Genet. Eng. Mice Handb. 2016, 157–164. [Google Scholar] [CrossRef]
- Lee, J.H.; Kim, Y.G.; Park, J.G.; Lee, J. Supercritical fluid extracts of Moringa oleifera and their unsaturated fatty acid components inhibit biofilm formation by Staphylococcus aureus. Food Control 2017, 80, 74–82. [Google Scholar] [CrossRef]
- Chelliah, R.; Ramakrishnan, S.; Antony, U. Nutritional quality of Moringa oleifera for its bioactivity and antibacterial properties. Int. Food Res. J. 2017, 24, 825–833. [Google Scholar]
- Chuang, P.H.; Lee, C.W.; Chou, J.Y.; Murugan, M.; Shieh, B.J.; Chen, H.M. Anti-fungal activity of crude extracts and essential oil of Moringa oleifera Lam. Bioresour. Technol. 2007, 98, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Zhang, D. Supercritical fluid extraction and characterisation of Moringa oleifera leaves oil. Sep. Purif. Technol. 2013, 118, 497–502. [Google Scholar] [CrossRef]
- Bhalla, N.; Ingle, N.; Patri, S.V.; Haranath, D. Phytochemical analysis of Moringa oleifera leaves extracts by GC-MS and free radical scavenging potency for industrial applications. Saudi J. Biol. Sci. 2021. [Google Scholar] [CrossRef]
- Lalas, S.; Athanasiadis, V.; Karageorgou, I.; Batra, G.; Nanos, G.D.; Makris, D.P. Nutritional Characterization of Leaves and Herbal Tea of Moringa oleifera Cultivated in Greece. J. Herbs Spices Med. Plants 2017, 23, 320–333. [Google Scholar] [CrossRef]
- Lopez-Rodriguez, N.A.; Gaytán-Martínez, M.; de la Luz Reyes-Vega, M.; Loarca-Piña, G. Glucosinolates and Isothiocyanates from Moringa oleifera: Chemical and Biological Approaches. Plant Foods Hum. Nutr. 2020, 75, 447–457. [Google Scholar] [CrossRef]
- Leone, A.; Spada, A.; Battezzati, A.; Schiraldi, A.; Aristil, J.; Bertoli, S. Cultivation, genetic, ethnopharmacology, phytochemistry and pharmacology of Moringa oleifera leaves: An overview. Int. J. Mol. Sci. 2015, 16, 12791–12835. [Google Scholar] [CrossRef]
- Balamurugan, R.; Duraipandiyan, V.; Ignacimuthu, S. Antidiabetic activity of γ-sitosterol isolated from Lippia nodiflora L. in streptozotocin induced diabetic rats. Eur. J. Pharmacol. 2011, 667, 410–418. [Google Scholar] [CrossRef]
- Balamurugan, R.; Stalin, A.; Ignacimuthu, S. Molecular docking of γ-sitosterol with some targets related to diabetes. Eur. J. Med. Chem. 2012, 47, 38–43. [Google Scholar] [CrossRef]
- Dhifi, W.; Bellili, S.; Jazi, S.; Bahloul, N.; Mnif, W. Essential Oils’ Chemical Characterization and Investigation of Some Biological Activities: A Critical Review. Medicines 2016, 3, 25. [Google Scholar] [CrossRef] [Green Version]
- Salvamani, S.; Gunasekaran, B.; Shaharuddin, N.A.; Ahmad, S.A.; Shukor, M.Y. Antiartherosclerotic effects of plant flavonoids. BioMed Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [Green Version]
- Montone, A.M.I.; Papaianni, M.; Malvano, F.; Capuano, F.; Capparelli, R.; Albanese, D. Lactoferrin, Quercetin, and Hydroxyapatite Act Synergistically against Pseudomonas fluorescens. Int. J. Mol. Sci. 2021, 22, 9247. [Google Scholar] [CrossRef] [PubMed]
- Nocerino, N.; Fulgione, A.; Iannaccone, M.; Tomasetta, L.; Ianniello, F.; Martora, F.; Lelli, M.; Roveri, N.; Capuano, F.; Capparelli, R. Biological activity of lactoferrin-functionalized biomimetic hydroxyapatite nanocrystals. Int. J. Nanomed. 2014, 9, 1175–1184. [Google Scholar] [CrossRef] [Green Version]
- Cuomo, P.; Papaianni, M.; Fulgione, A.; Guerra, F.; Capparelli, R.; Medaglia, C. An innovative approach to control H. Pylori-induced persistent inflammation and colonization. Microorganisms 2020, 8, 1214. [Google Scholar] [CrossRef] [PubMed]
- Fulgione, A.; Nocerino, N.; Iannaccone, M.; Roperto, S.; Capuano, F.; Roveri, N.; Lelli, M.; Crasto, A.; Calogero, A.; Pilloni, A.P.; et al. Lactoferrin adsorbed onto biomimetic hydroxyapatite nanocrystals controlling—In vivo—The Helicobacter pylori infection. PLoS ONE 2016, 11, e0158646. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vergara-Jimenez, M.; Almatrafi, M.M.; Fernandez, M.L. Bioactive components in Moringa oleifera leaves protect against chronic disease. Antioxidants 2017, 6, 91. [Google Scholar] [CrossRef] [Green Version]
- Oluduro, A.O. Evaluation of Antimicrobial properties and nutritional potentials of Moringa oleifera Lam.leaf in South-Western Nigeria. Malays. J. Microbiol. 2012, 8, 59–67. [Google Scholar] [CrossRef]
- Siddhuraju, P.; Becker, K. Antioxidant properties of various solvent extracts of total phenolic constituents from three different agroclimatic origins of drumstick tree (Moringa oleifera Lam.) leaves. J. Agric. Food Chem. 2003, 51, 2144–2155. [Google Scholar] [CrossRef]
- Xu, Y.; Chen, G.; Guo, M. Correlations between phytochemical fingerprints of Moringa oleifera leaf extracts and their antioxidant activities revealed by chemometric analysis. Phytochem. Anal. 2021, 32, 698–709. [Google Scholar] [CrossRef]
- Hossain, M.A.; Disha, N.K.; Shourove, J.H.; Dey, P. Determination of Antioxidant Activity and Total Tannin from Drumstick (Moringa oleifera Lam.) Leaves Using Different Solvent Extraction Methods. Turk. J. Agric.—Food Sci. Technol. 2020, 8, 2749–2755. [Google Scholar] [CrossRef]
- Kooltheat, N.; Pankla Sranujit, R.; Chumark, P.; Potup, P.; Laytragoon-Lewin, N.; Usuwanthim, K. An ethyl acetate fraction of Moringa oleifera Lam. inhibits human macrophage cytokine production induced by cigarette smoke. Nutrients 2014, 6, 697–710. [Google Scholar] [CrossRef] [Green Version]
- Khalafalla, M.M.; Abdellatef, E.; Dafalla, H.M.; Nassrallah, A.A.; Aboul-Enein, K.M.; Lightfoot, D.A.; El-Deeb, F.E.; El-Shemy, H.A. Active principle from Moringa oleifera Lam leaves effective against two leukemias and a hepatocarcinoma. Afr. J. Biotechnol. 2010, 9, 8467–8471. [Google Scholar] [CrossRef]
- Das, N.; Sikder, K.; Ghosh, S.; Fromenty, B.; Dey, S. Moringa oleifera lam. leaf extract prevents early liver injury and restores antioxidant status in mice fed with high-fat diet. Indian J. Exp. Biol. 2012, 50, 404–412. [Google Scholar]
- Almatrafi, M.M.; Vergara-Jimenez, M.; Smyth, J.A.; Medina-Vera, I.; Fernandez, M.L. Moringa oleifera leaves do not alter adi-pose tissue colesterol accumulation or inflammation in guinea pigs fed a hypercholesterolemic diet. EC Nutr. 2017, 18, 1330. [Google Scholar]
- Halaby, M.S.; Elmetwaly, E.M.; Omar, A.A.A. Effect of Moringa oleifera on serum lipids and kidney function of hyperlipidemic rats. J. Appl. Sci. Res. 2013, 9, 5189–5198. [Google Scholar]
- Almatrafi, M.M.; Vergara-Jimenez, M.; Murillo, A.G.; Norris, G.H.; Blesso, C.N.; Fernandez, M.L. Moringa leaves prevent hepatic lipid accumulation and inflammation in guinea pigs by reducing the expression of genes involved in lipid metabolism. Int. J. Mol. Sci. 2017, 18, 1330. [Google Scholar] [CrossRef] [PubMed]
- Richter, N.; Siddhuraju, P.; Becker, K. Evaluation of nutritional quality of moringa (Moringa oleifera Lam.) leaves as an alternative protein source for Nile tilapia (Oreochromis niloticus L.). Aquaculture 2003, 217, 599–611. [Google Scholar] [CrossRef]
- Bais, S.; Singh, G.S.; Sharma, R. Antiobesity and Hypolipidemic Activity of Moringa oleifera Leaves against High Fat Diet-Induced Obesity in Rats. Adv. Biol. 2014, 2014, 9. [Google Scholar] [CrossRef] [Green Version]
- Panda, S.; Kar, A.; Sharma, P.; Sharma, A. Cardioprotective potential of N,α-l-rhamnopyranosyl vincosamide, an indole alkaloid, isolated from the leaves of Moringa oleifera in isoproterenol induced cardiotoxic rats: In Vivo and in vitro studies. Bioorg. Med. Chem. Lett. 2013, 23, 959–962. [Google Scholar] [CrossRef] [PubMed]
- Dangi, S.Y.; Jolly, C.I.; Narayanan, S. Antihypertensive activity of the total alkaloids from the leaves of Moringa oleifera. Pharm. Biol. 2002, 40, 144–148. [Google Scholar] [CrossRef]
- Tiloke, C.; Anand, K.; Gengan, R.M.; Chuturgoon, A.A. Moringa oleifera and their phytonanoparticles: Potential antiproliferative agents against cancer. Biomed. Pharmacother. 2018, 108, 457–466. [Google Scholar] [CrossRef]
- Wahyuni, R.; Wignyanto, W.; Wijana, S.; Sucipto, S. Optimization of protein and tannin extraction in Moringa oleifera leaf as antioxidant source. Food Res. 2020, 4, 2224–2232. [Google Scholar] [CrossRef]
- Adisakwattana, S.; Chanathong, B. A-Glucosidase Inhibitory Activity and Lipid-Lowering. Eur. Rev. Med. Pharmacol. Sci. 2011, 15, 803–808. [Google Scholar]
- Araújo, L.C.C.; Aguiar, J.S.; Napoleão, T.H.; Mota, F.V.B.; Barros, A.L.S.; Moura, M.C.; Coriolano, M.C.; Coelho, L.C.B.B.; Silva, T.G.; Paiva, P.M.G. Evaluation of cytotoxic and anti-inflammatory activities of extracts and lectins from Moringa oleifera seeds. PLoS ONE 2013, 8, e81973. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiong, Y.; Riaz Rajoka, M.S.; Zhang, M.X.; He, Z. Isolation and identification of two new compounds from the seeds of Moringa oleifera and their antiviral and anti-inflammatory activities. Nat. Prod. Res. 2020, 1–9. [Google Scholar] [CrossRef]
- Govardhan Singh, R.S.; Negi, P.S.; Radha, C. Phenolic composition, antioxidant and antimicrobial activities of free and bound phenolic extracts of Moringa oleifera seed flour. J. Funct. Foods 2013, 5, 1883–1891. [Google Scholar] [CrossRef]
- Liao, P.C.; Lai, M.H.; Hsu, K.P.; Kuo, Y.H.; Chen, J.; Tsai, M.C.; Li, C.X.; Yin, X.J.; Jeyashoke, N.; Chao, L.K.P. Identification of β-Sitosterol as in Vitro Anti-Inflammatory Constituent in Moringa oleifera. J. Agric. Food Chem. 2018, 66, 10748–10759. [Google Scholar] [CrossRef] [PubMed]
- Abd Rani, N.Z.; Kumolosasi, E.; Jasamai, M.; Jamal, J.A.; Lam, K.W.; Husain, K. In Vitro anti-allergic activity of Moringa oleifera Lam. extracts and their isolated compounds. BMC Complement. Altern. Med. 2019, 19, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Wolff, K.; Jaja-Chimedza, A.; Kim, Y.; Waterman, C.; Poulev, A.; Raskin, I.; Ribnicky, D. Moringa isothiocyanate-1 is bioaccessible and bioavailable as a stable unmodified compound. Phytochem. Lett. 2020, 38, 33–38. [Google Scholar] [CrossRef]
- Famurewa, A.C.; Asogwa, N.T.; Aja, P.M.; Akunna, G.G.; Awoke, J.N.; Ekeleme-Egedigwe, C.A.; Maduagwuna, E.K.; Folawiyo, A.M.; Besong, E.E.; Ekpono, E.U.; et al. Moringa oleifera seed oil modulates redox imbalance and iNOS/NF-ĸB/caspase-3 signaling pathway to exert antioxidant, anti-inflammatory and antiapoptotic mechanisms against anticancer drug 5-fluorouracil-induced nephrotoxicity in rats. S. Afr. J. Bot. 2019, 127, 96–103. [Google Scholar] [CrossRef]

| Compounds | Molecular Formula | Quantity | Method | Reference | |
|---|---|---|---|---|---|
| Leaves | Seeds | ||||
| Caproic acid | C6H12O2 | 0.1% | - | GC-MS | [9] |
| Capric acid | C10H20O2 | 0.1% | - | GC-MS | [9] |
| Lauric acid | C12H24O2 | 0.72% | 0.03% | GC-MS | [9] |
| Myristic acid | C14H28O2 | 1.13% | 0.16% | GC-MS | [9] |
| Palmitic acid | C16H32O2 | 23.65% | 7.92% | GC-MS | [9] |
| - | Madurai: 6.17% a | GC-MS | [10] | ||
| 1.08% a | - | GC-MS | [11] | ||
| 0.15% | - | GC-MS | [12] | ||
| 2.5703% a | - | GC-MS | [13] | ||
| Palmitoleic acid | C16H30O2 | - | 1.49% | GC-MS | [9] |
| Heptadecanoic acid | C17H34O2 | 0.46% | 0.32% | GC-MS | [9] |
| Stearic acid | C18H36O2 | 1.2051% a | - | GC-MS | [13] |
| 3.93% | 7.10% | GC-MS | [9] | ||
| Oleic acid | C18H34O2 | 5.92% | 74.50% | GC-MS | [9] |
| - | Madurai: 5.12% a | GC-MS | [10] | ||
| Linoleic acid | C18H32O2 | 6.84% | 0.62% | GC-MS | [9] |
| Arachidic acid | C20H40O2 | 0.72% | 4.28% | GC-MS | [9] |
| Cis-11-Eicosenoic acid | C20H38O2 | 54.44% | - | GC-MS | [9] |
| Linolenic acid | C18H30O2 | - | 2.50% | GC-MS | [9] |
| Cis-11, 14-Eicosadienoic acid | C20H36O2 | - | 0.48% | GC-MS | [9] |
| Cis-5, 8, 11, 14, 17-Eicosapentaenoic acid. | C21H32O2 | 1.99% | 0.60% | GC-MS | [9] |
| Lysine | C6H14N2O2 | Madurai: 4.10 g/16 gN Chennai: 3.30 g/16 gN | Madurai: 4.20 g/16 gN Chennai: 3.70 g/16 gN | HPLC | [10] |
| 129.4 mg/100g dw | - | HPLC | [14] | ||
| Histidine | C6H9N3O2 | Madurai: 2.50 g/16 gN Chennai: 3.12 g/16 gN | Madurai: 6.30 g/16 gN Chennai: 5.70 g/16 gN | HPLC | [10] |
| 73.9 mg/100g dw | - | HPLC | [14] | ||
| Valine | C5H11NO2 | Madurai: 4.55 g/16 gN Chennai: 4.75 g/16 gN | Madurai: 3.36 g/16 gN Chennai: 2.37 g/16 gN | HPLC | [10] |
| 111.5 mg/100g dw | - | HPLC | [14] | ||
| Leucine | C6H13NO2 | Madurai: 4.80 g/16 gN Chennai: 4.40 g/16 gN | Madurai: 4.80 g/16 gN Chennai: 4.70 g/16 gN | HPLC | [10] |
| 211.5 mg/100g dw | - | HPLC | [14] | ||
| Isoleucine | C6H13NO2 | Madurai: 5.90 g/16 gN Chennai: 9.10 g/16 gN | Madurai: 8.30 g/16 gN Chennai: 7.70 g/16 gN | HPLC | [10] |
| 81.3 mg/100g dw | - | HPLC | [14] | ||
| Threonine | C4H9NO3 | Madurai: 4.10 g/16 gN Chennai: 4.50 g/16 gN | Madurai: 3.20 g/16 gN Chennai: 3.80 g/16 gN | HPLC | [10] |
| 91.8 mg/100g dw | - | HPLC | [14] | ||
| Alanine | C3H7NO2 | Madurai: 3.23 g/16 gN Chennai: 3.90 g/16 gN | Madurai: 4.29 g/16 gN Chennai: 3.55 g/16 gN | HPLC | [10] |
| 121.2 mg/100 g dw | - | HPLC | [14] | ||
| Aspartic acid | C4H7NO4 | Madurai: 6.44 g/16 gN Chennai: 6.86 g/16 gN | Madurai: 6.02 g/16 gN Chennai: 6.37 g/16 gN | HPLC | [10] |
| 195.8 mg/100 g dw | - | HPLC | [14] | ||
| Serine | C3H7NO3 | Madurai: 4.25 g/16 gN Chennai: 4.66 g/16 gN | Madurai: 4.22 g/16 gN Chennai: 4.11 g/16 gN | HPLC | [10] |
| 90.2 mg/100 g dw | - | HPLC | [14] | ||
| Proline | C5H9NO2 | 128.9 mg/100 g dw | - | HPLC | [14] |
| Glutamic acid | C5H9NO4 | Madurai: 15.86 g/16 gN Chennai: 15.33 g/16 gN | Madurai: 14.23 g/16 gN Chennai: 14.74 g/16 gN | HPLC | [10] |
| 268.7 mg/100 g dw | - | HPLC | [14] | ||
| Glycine | C2H5NO2 | Madurai: 5.13 g/16 gN Chennai: 5.11 g/16 gN | Madurai: 5.00 g/16 gN Chennai: 4.70 g/16 gN | HPLC | [10] |
| 102.4 mg/100 g dw | - | HPLC | [14] | ||
| Arginine | C6H14N4O2 | Madurai: 4.30 g/16 gN Chennai: 3.70 g/16 gN | Madurai: 9.44 g/16 gN Chennai: 11.00 g/16 gN | HPLC | [10] |
| 125.4 mg/100 g dw | - | HPLC | [14] | ||
| Cysteine | C3H7NO2S | 29.6 mg/100 g dw | - | HPLC | [14] |
| Tyrosine | C9H11NO3 | 79.3 mg/100 g dw | - | HPLC | [14] |
| Methionine | C5H11NO2S | Madurai: 1.90 g/16 gN Chennai: 3.40 g/16 gN | Madurai: 3.50 g/16 gN Chennai: 3.90 g/16 gN | HPLC | [10] |
| 221.4 mg/100 g dw | - | HPLC | [14] | ||
| Phenylalanine | C9H11NO2 | Madurai: 4.20 g/16 gN Chennai: 4.60 g/16 gN | Madurai: 3.50 g/16 gN Chennai: 3.90 g/16 gN | HPLC | [10] |
| 117.2 mg/100 g | - | HPLC | [14] | ||
| 1,30-Triacontanediol | C30H62O2 | 3.06% | - | GC-MS | [12] |
| Madurai: 14.98% a | - | GC-MS | [10] | ||
| Octacosane | C28H58 | Madurai: 8.57% a | - | GC-MS | [10] |
| Z-14-nonacosane | C29H58 | Madurai: 8.3% a | - | GC-MS | [10] |
| 2,2-dimethyl-1-oxa-2-silacyclotrid ecanone-13 | C6H14OSi | Madurai: 8.28% a | - | GC-MS | [10] |
| Nonacosane | C29H60 | Chennai: 15.55% a | - | GC-MS | [10] |
| 18,65% | - | GC-MS | [12] | ||
| γ-Sitosterol | C29H50O | 0,86% | - | GC-MS | [12] |
| Chennai: 9.56% a | - | GC-MS | [10] | ||
| Campesterol | C28H48O | Chennai: 5.00% a | Chennai: 1.21% a | GC-MS | [10] |
| 13-docosenamide, (Z)- | C22H43NO | - | Chennai: 13.62% a | GC-MS | [10] |
| 6-octadecenoic acid | C18H34O2 | - | Madurai: 52.24% a | GC-MS | [10] |
| Propionamide | C3H7NO | 0.7335% a | - | GC-MS | [13] |
| - | Chennai: 4.48% a | GC-MS | [10] | ||
| Ethyl oleate | C20H38O2 | - | Chennai: 4.33% a | GC-MS | [10] |
| Glucomoringin | C21H31NO14S2 | 16.14–17.3 μmol/g dry weight | - | UPLC | [15] |
|
Domesticated: 75.29 μmol/g dry weight Wild: 18.00 μmol/g dry weight | - | HPLC | [15] | ||
| 110–112 μmol/g dry weight | - | HPLC | [15] | ||
| 21.84–22.56 mg/g dry weight | 32.88 mg/g dry weight | HPLC | [15] | ||
| 0.77 mg/g dry weight | 86.19 mg/g dry weight | HPLC-ESI-MS/MS | [15] | ||
| 33.9–59.4 mg/g dry weight | 202 mg/g dry weight | [15] | |||
| Glucomoringin Isomer I | C21H31NO14S2 | 7.15–8.92 μmol/g dry weight | - | UPLC | [15] |
| Glucomoringin Isomer II | C21H31NO14S2 | 3.85–4.84 μmol/g dry weight | - | UPLC | [15] |
| Glucomoringin Isomer III | C21H31NO14S2 | 18.71–22.58 μmol/g dry weight | - | UPLC | [15] |
| Glucosoonjnain | - | Domesticated: 1.16 Wild: 33.79 | - | HPLC | [15] |
| β-carotene | C40H56 | 33.48 mg/100 g | - | HPLC | [16] |
| 27.22 mg/100 g | - | N/A | [16] | ||
| 36 mg/100 g | - | AOAC 2004 | [16] | ||
| 39.6 mg/100 g | - | N/A | [16] | ||
| 37.8 mg/100 g | - | N/A | [16] | ||
| 17.62 mg/100 g | - | N/A | [16] | ||
| 20.44 mg/100 g | - | HPLC | [16] | ||
| 66 mg/100 g | - | HPLC | [16] | ||
| Lutein | C40H56O2 | 35.05 mg/100 g | - | HPLC | [16] |
| 102 mg/100 g | - | HPLC | [16] | ||
| Caffeic acid | C9H8O4 | 0.409 mg/g | - | HPLC | [16] |
| 0.536 mg/g | - | HPLC | [16] | ||
| Chlorogenic acid | C16H18O9 | 0.489 mg/g | - | HPLC and MS/MS | [16] |
| o-Coumaric acid | C9H8O3 | 6.457 mg/g | - | HPLC | [16] |
| Ellagic acid | C14H6O8 | 0.018 mg/g | - | HPLC and MS/MS | [16] |
| 0.189 mg/g | - | HPLC and MS/MS | [16] | ||
| Ferulic acid | C10H10O4 | 0.078 mg/g | - | HPLC and MS/MS | [16] |
| 0.078 mg/g | - | HPLC | [16] | ||
| 0.128 mg/g | - | HPLC and MS/MS | [16] | ||
| Gallic acid | C7H6O5 | 1.034 mg/g | - | HPLC and MS/MS | [16] |
| 1.034 mg/g | - | HPLC and MS/MS | [16] | ||
| Epicatechin | C15H14O6 | 5.68 mg/g | - | HPLC | [16] |
| Isorhamnetin | C16H12O7 | 0.118 mg/g | - | HPLC | [16] |
| 0.52 mg/g e,f | - | HPLC-DAD-ESI-MS | [16] | ||
| 0.72 mg/g f,g | - | [16] | |||
| Kaempferol | C15H10O6 | 0.04 mg/g | - | HPLC | [16] |
| 2.360 mg/g | - | HPLC | [16] | ||
| 0.198 mg/g | - | HPLC and MS/MS | [16] | ||
| 1.412 mg/g | - | HPLC | [16] | ||
| 0.8 mg/g | - | LC/MS | [16] | ||
| 1.23 mg/g | - | [16] | |||
| 4.59 mg/g | - | [16] | |||
| 3.92 e,f | - | HPLC-DAD-ESI-MS | [16] | ||
| 2.16 mg/g f,g | - | [16] | |||
| 2.25 mg/g | - | HPLC-DAD | [16] | ||
| 1.75 mg/g | - | [16] | |||
| 1.05 mg/g | - | [16] | |||
| 2.9 mg/g d | - | LC/MS | [16] | ||
| 2.3 mg/g | - | [16] | |||
| 3.5 mg/g | - | [16] | |||
| 0.3 mg/g c | - | [16] | |||
| 0.16 mg/g d | - | [16] | |||
| Myricetin | C15H10O8 | 5.804 mg/g | - | HPLC | [16] |
| Quercetin | C15H10O7 | 0.281 mg/g | - | HPLC | [16] |
| 0.207 mg/g | - | HPLC and MS/MS | [16] | ||
| 0.207 mg/g | - | HPLC | [16] | ||
| 0.807 mg/g | - | HPLC and MS/MS | [16] | ||
| 3.529 mg/g | - | HPLC | [16] | ||
| 5.2 mg/g | - | LC/MS | [16] | ||
| 5.8 mg/g | - | [16] | |||
| 7.57 mg/g | - | [16] | |||
| 12.84 mg/g e,f | - | HPLC-DAD-ESI-MS | [16] | ||
| 16.64 mg/g f,g | - | [16] | |||
| 9.26 mg/g | - | HPLC-DAD | [16] | ||
| 6.34 mg/g | - | [16] | |||
| 7.70 mg/g | - | [16] | |||
| 5.47 mg/g b | - | LC-MS | [16] | ||
| 9.1 mg/g | - | [16] | |||
| 15.2 mg/g | - | [16] | |||
| 0.58 mg/g c | - | [16] | |||
| 0.46 mg/g d | - | [16] | |||
| Rutin | C27H30O16 | 1.674 mg/g | - | HPLC | [16] |
| 4-(α-L-rhamnopyranosyloxy)- benzyl-isothiocyanate (moringin) | C14H17NO5S | 22.56 mg/g e,f | - | HPLC-DAD-ESI-MS | [16] |
| 21.84 mg/g g,f | - | [16] | |||
| 33.9 mg/g c | - | LC-MS | [16] | ||
| 59.4 mg/g d | - | [16] | |||
| 4-O-(α-Lacetylrhamnopyranosyloxy)-benzyl-isothiocyanate isomer 1 | C16H19NO6S | 2.76 mg/g e,f | - | HPLC-DAD-ESI-MS | [16] |
| 2.16 mg/g g,f | - | [16] | |||
| 2.9 mg/g c | - | LC-MS | [16] | ||
| 5.0 mg/g d | - | [16] | |||
| 4-O-(α-Lacetylrhamnopyranosyloxy)-benzyl-isothiocyanate isomer 2 | C16H19NO6S | 1.80 mg/g e,f | - | HPLC-DAD-ESI-MS | [16] |
| 1.52 mg/g g,f | - | [16] | |||
| 1.2 mg/g c | - | LC-MS | [16] | ||
| 1.5 mg/g d | - | [16] | |||
| 4-O-(α-Lacetylrhamnopyranosyloxy)-benzyl isomer 3 | C16H19NO6S | 20.16 mg/g e,f | - | HPLC-DAD-ESI-MS | [16] |
| 12.76 mg/g g,f | - | [16] | |||
| 17.4 mg/g c | - | LC-MS | [16] | ||
| 50.2 mg/g d | - | [16] | |||
| Toluene | C7H8 | 1.27% | - | GC-MS | [12] |
| 0.03% a | - | GC-MS | [11] | ||
| 5-tert-Butyl-1,3-cyclopentadiene | C9H14 | 0.07% a | - | GC-MS | [11] |
| Benzaldehyde | C6H5CHO | 0.55% a | - | GC-MS | [11] |
| 5-Methyl-2-furaldehyde | C6H6O2 | 0.27% a | - | GC-MS | [11] |
| Benzeneacetaldehyde | C8H8O | 2.16% a | - | GC-MS | [11] |
| 2-Ethyl-3,6-dimethylpyrazine | C8H12N2 | 0.12% a | - | GC-MS | [11] |
| Undecane | C11H24 | 0.12% a | - | GC-MS | [11] |
| Isophoron | C9H14O | 0.10% a | - | GC-MS | [11] |
| Benzylnitrile | C8H7N | 1.10% a | - | GC-MS | [11] |
| 2,6,6-Trimethyl-2-cyclohexane-1,4-dione | C9H12O2 | 0.05% a | - | GC-MS | [11] |
| 2,2,4-Trimethyl-pentadiol | C12H24O3 | 0.09% a | - | GC-MS | [11] |
| 2,3-Epoxycarane | C10H16O | 0.16% a | - | GC-MS | [11] |
| p-Menth-1-en-8-ol | C10H18O | 0.08% a | - | GC-MS | [11] |
| 2,6,6-Trimethylcyclohexa-1,3-dienecarbaldehyde | C9H14 | 0.23% a | - | GC-MS | [11] |
| Indole | C8H7N | 1.20% a | - | GC-MS | [11] |
| Tridecane | C13H28 | 0.16% a | - | GC-MS | [11] |
| Ionone | C13H20O | 0.13% a | - | GC-MS | [11] |
| 0.03% a | - | GC-MS | [11] | ||
| 1,1,6-Trimethyl-1,2-dihydronaphthalene | C13H16 | 0.41% a | - | GC-MS | [11] |
| Ionene | C13H18 | 0.09% a | - | GC-MS | [11] |
| Damascenone | C13H18O | 0.28%a | - | GC-MS | [11] |
| Ledene oxide | C15H24O | 0.60% a | - | GC-MS | [11] |
| 2-tert-Butyl-1,4-dimethoxybenzene | C12H18O2 | 0.39% a | - | GC-MS | [11] |
| (E)-6,10-dimethylundeca-5,9-dien-2-one | C13H22O | 0.26% a | - | GC-MS | [11] |
| 4,6-Dimethyldodecane | C14H30 | 0.29% a | - | GC-MS | [11] |
| 3,3,5,6-Tetramethyl-1-indanone | C13H16O | 0.23% a | - | GC-MS | [11] |
| Dihydro-actiridioide | - | 1.21% a | - | GC-MS | [11] |
| 2,3,6-Trimethyl-naphthalene | C13H14 | 0.37% a | - | GC-MS | [11] |
| Megastigmatrienone | C13H18O | 0.57% a | - | GC-MS | [11] |
| 1-[2,3,6-Trimethyl-phenyl]-2-butanone | C13H18O | 3.44% a | - | GC-MS | [11] |
| 1-[2,3,6-Trimethyl-phenyl]-3-buten-2-one | C13H16O | 0.75% a | - | GC-MS | [11] |
| Isolongifolene | C15H24 | 0.56% a | - | GC-MS | [11] |
| Hexahydrofarnesylactone | C18H36O | 1.30% a | - | GC-MS | [11] |
| Farnesylacetone | C18H30O | 0.08% a | - | GC-MS | [11] |
| Methyl palmitate | C17H34O2 | 0.08% a | - | GC-MS | [11] |
| [6E,10E]-7,11,15-trimethyl-methylene-1,6,10,14-hexadeca-tetraene | C20H32 | 0.11% a | - | GC-MS | [11] |
| Phytol | C20H40O | 0.9664% a | - | GC-MS | [13] |
| 7.66% a | - | GC-MS | [11] | ||
| Docosane | C22H46 | 0.28% a | - | GC-MS | [11] |
| 1-Docosene | C22H44 | 0.41% a | - | GC-MS | [11] |
| Tetracosane | C24H50 | 1.45% a | - | GC-MS | [11] |
| Pentacosane | C25H52 | 17.41% a | - | GC-MS | [11] |
| 2.14% | - | GC-MS | [11] | ||
| Hexacosane | C26H54 | 11.20% a | - | GC-MS | [11] |
| Pyridine | C5H5N | 0.78% | - | GC-MS | [12] |
| Hepane,2-methyl- | C8H18 | 0.68% | - | GC-MS | [12] |
| Heptane,3-methyl- | C8H18 | 0.50% | - | GC-MS | [12] |
| Cyclohexane,1,3-dimethyl-,cis- | C8H16 | 0.78% | - | GC-MS | [12] |
| Octane | C8H18 | 1.75% | - | GC-MS | [12] |
| Cyclohexane,1,4-dimethyl-,cis- | C8H16 | 0.14% | - | GC-MS | [12] |
| Octane,2-methyl- | C9H20 | 0.17% | - | GC-MS | [12] |
| Cyclohexane,ethyl- | C8H16 | 0.43% | - | GC-MS | [12] |
| Ethylbenzene | C8H10 | 0.83% | - | GC-MS | [12] |
| Benzene,1,3-dimethyl- | C8H10 | 4.63% | - | GC-MS | [12] |
| o-Xylene | C8H10 | 2.82% | - | GC-MS | [12] |
| Nonane | C9H20 | 0.14% | - | GC-MS | [12] |
| Benzene,(1-methylethyl)- | C9H12 | 0.16% | - | GC-MS | [12] |
| Benzene,propyl- | C9H12 | 1.04% | - | GC-MS | [12] |
| Benzene,1-ethyl-3-methyl- | C9H12 | 5.95% | - | GC-MS | [12] |
| Benzene,1-ethyl-4-methyl- | C9H12 | 2.39% | - | GC-MS | [12] |
| Benzene,1,3,5-trimethyl- | C9H12 | 4.31% | - | GC-MS | [12] |
| Benzene,1-ethyl-2-methyl- | C9H12 | 2.55% | - | GC-MS | [12] |
| Benzene,1,2,4-trimethyl- | C9H12 | 16.96% | - | GC-MS | [12] |
| Benzene,1,2,3-trimethyl- | C9H12 | 4.14% | - | GC-MS | [12] |
| Indane | C9H10 | 0.39% | - | GC-MS | [12] |
| Benzene,1-ethyl-2,4- dimethyl- | C10H14 | 0.64% | - | GC-MS | [12] |
| Benzene,1-ethyl-2,3- dimethyl- | C10H14 | 0.99% | - | GC-MS | [12] |
| Benzene,1,2,3,5-tetramethyl- | C10H14 | 1.59% | - | GC-MS | [12] |
| Benzene,1,2,4,5-tetramethyl- | C10H14 | 2.12% | - | GC-MS | [12] |
| Benzene,1,2,3,4-tetramethyl- | C10H14 | 0.58% | - | GC-MS | [12] |
| Naphthalene | C10H8 | 1.21% | - | GC-MS | [12] |
| Dimethoate | C5H12NO3PS2 | 0.52% | - | GC-MS | [12] |
| Nonadecane | C19H40 | 0.22% | - | GC-MS | [12] |
| Eicosane | C20H42 | 0.31% | - | GC-MS | [12] |
| Heptacosane | C27H56 | 7.45% | - | GC-MS | [12] |
| γ-Tocopherol | C28H48O2 | 0.75% | - | GC-MS | [12] |
| Sulfurous acid, hexyl pentadecyl ester | C21H44O3S | 0.34% | - | GC-MS | [12] |
| dl-α-Tocopherol | C29H50O2 | 1.05% | - | GC-MS | [12] |
| β-Amyrin | C30H50O | 4.60% | - | GC-MS | [12] |
| 1,3-Dihydroxyacetone dimer | C6H12O6 | 3.8551% a | - | GC-MS | [13] |
| Acetic acid, [(amino carbonyl)amino]oxo- | C3H4N2O4 | 3.2396% a | - | GC-MS | [13] |
| 4(1H)-Pyrimidinone, 2,6-diamino- | C4H6N4O | 2.2433% a | - | GC-MS | [13] |
| 4H-Pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl- | C6H8O4 | 4.1801% | - | GC-MS | [13] |
| 8.9858% a | - | GC-MS | [13] | ||
| 2-Hexynoic acid | C6H8O2 | 1.1214% a | - | GC-MS | [13] |
| Butanedioic acid, 2-hydroxy-2-methyl-, (S)- | C7H12O5 | 3.1422% a | - | GC-MS | [13] |
| 3,3-Iminobispropylamine | C6H17N3 | 1.9275% a | - | GC-MS | [13] |
| 1-Hexanamine | C6H15N | 0.0774% a | - | GC-MS | [13] |
| 1,3-Dioxolan-2-one, 4,5-dimethyl- | C5H8O3 | 6.1627% a | - | GC-MS | [13] |
| 2-Butenethioic acid, 3-(ethylthio)-, S-(1-methylethyl) ester | C9H16OS2 | 1.3083% a | - | GC-MS | [13] |
| Propanamide, N,N-dimethyl- | C5H11NO | 3.0349% a | - | GC-MS | [13] |
| 2-Isopropoxyethyl propionate | C8H16O3 | 16.8738% a | - | GC-MS | [13] |
| D-Mannoheptulose | C7H14O7 | 2.5622% a | - | GC-MS | [13] |
| Azetidin-2-one 3,3-dimethyl-4-(1-aminoethyl)- | C7H14N2O | 4.6738% a | - | GC-MS | [13] |
| Carbonic acid, butyl 2-pentyl ester | C10H20O3 | 20.6431% a | - | GC-MS | [13] |
| Tetra acetyl-d-xylonic nitrile | C14H17NO9 | 5.0379% a | - | GC-MS | [13] |
| α-D-Glucose | C6H12O6 | 3.445% a | - | GC-MS | [13] |
| 1H-Cyclopenta[c]furan-3(3αH)-one, 6,6α-dihydro-1-(1,3-dioxolan-2-yl)-, (3αR,1-trans,6α-cis)- | C10H12O4 | 1.2587% a | - | GC-MS | [13] |
| 3-[1-(4-Cyano-1,2,3,4-tetrahydronaphthyl)]propanenitrile | C14H14N2 | 1.5253% a | - | GC-MS | [13] |
| Quinolinium, 1-ethyl-, iodide | C11H12IN | 1.4067% a | - | GC-MS | [13] |
| N-Isopropyl-3-phenylpropanamide | C12H17NO | 0.9462% a | - | GC-MS | [13] |
| 1,2-Ethanediamine, N-(2-aminoethyl)- | C4H13N3 | 0.8805% a | - | GC-MS | [13] |
| 1,4-Benzenediol, 2-methyl- | C7H8O2 | 4.3169% a | - | GC-MS | [13] |
| Ethene, ethoxy- | C4H8O | 0.5981% a | - | GC-MS | [13] |
| Dihydroxyacetone | C3H6O3 | 2.4651% a | - | GC-MS | [13] |
| Glycerine | C3H8O3 | 1.8656% a | - | GC-MS | [13] |
| Erythritol | C4H10O4 | 0.5327% a | - | GC-MS | [13] |
| Monomethyl malonate | C4H6O4 | 2.5684% a | - | GC-MS | [13] |
| 4,5-Diamino-6-hydroxypyrimidine | C8H12N8O2 | 0.6434% a | - | GC-MS | [13] |
| Furan, 2,3-dihydro-4-methyl- | C5H8O | 0.2105% a | - | GC-MS | [13] |
| Catecholborane | C6H5BO2 | 0.6806% a | - | GC-MS | [13] |
| 2-Fluoropyridine | C5H4FN | 0.8121% a | - | GC-MS | [13] |
| 1,2,3-Propanetriol, 1-acetate | C5H10O4 | 1.4375% a | - | GC-MS | [13] |
| 3,4-Furandiol, tetrahydro-, trans- | C4H8O | 0.1746% a | - | GC-MS | [13] |
| 1-Nitro-β-d-arabino furanose, tetraacetate | C13H17NO11 | 0.5114% a | - | GC-MS | [13] |
| 1,8-Diamino-3,6-dioxaoctane | C6H16N2O2 | 0.1172% a | - | GC-MS | [13] |
| 1,7-Diaminoheptane | C7H18N2 | 1.7997% a | - | GC-MS | [13] |
| N,N-Dimethylacetamide | C4H9ON | 0.454% a | - | GC-MS | [13] |
| 2-Oxoglutaric acid | C5H6O5 | 0.5466% a | - | GC-MS | [13] |
| Oxazolidine, 2-ethyl-2-methyl- | C6H13NO | 0.9008% a | - | GC-MS | [13] |
| Heptanal | C7H14O | 0.7112% a | - | GC-MS | [13] |
| 6-Methoxy-3-pyridazinethiol | C5H6N2OS | 0.4293% a | - | GC-MS | [13] |
| 3-Piperidinol | C5H11NO | 0.5971% a | - | GC-MS | [13] |
| 1,3-Propanediol, 2-ethyl-2-(hydroxymethyl)- | C6H14O3 | 21.1909% a | - | GC-MS | [13] |
| Benzeneacetonitrile, 4-hydroxy- | C8H7NO | 3.4763% a | - | GC-MS | [13] |
| Benzenebutanal, γ, 4-dimethyl- | C12H16O | 0.395% a | - | GC-MS | [13] |
| 2(4H)-Benzofuranone, 5,6,7,7α-tetrahydro-4,4,7α-trimethyl- | C11H16O2 | 0.4491% a | - | GC-MS | [13] |
| Ethanamine, N-ethyl-N-nitroso- | C4H10N2O | 5.2161% a | - | GC-MS | [13] |
| Propanoic acid, 2-methyl-, octyl ester | C12H24O | 15.0279% a | - | GC-MS | [13] |
| 3-Deoxy-d-mannoic lactone | C6H10O5 | 3.2947% a | - | GC-MS | [13] |
| d-Glycero-d-ido-heptose | C7H14O7 | 0.3814% a | - | GC-MS | [13] |
| D-erythro-Pentose, 2-deoxy- | C5H10O | 0.3314% a | - | GC-MS | [13] |
| N-Methoxy-1-ribofuranosyl-4-imidazolecarboxylic amide | C10H15N3O6 | 0.5345% a | - | GC-MS | [13] |
| Formamide, N,N-dimethyl- | C3H7NO | 0.5847% a | - | GC-MS | [13] |
| d-Talonic acid lactone | C6H10O6 | 0.5651% a | - | GC-MS | [13] |
| Sorbitol | C6H14O6 | 0.482% a | - | GC-MS | [13] |
| Allo-Inositol | C6H12O | 0.8749% a | - | GC-MS | [13] |
| 0.5189% a | - | GC-MS | [13] | ||
| 1.1254% a | - | GC-MS | [13] | ||
| D-chiro-Inositol, 3-O-(2-amino-4-((carboxyiminomethyl)amino)- 2,3,4,6-tetradeoxy-α-D-arabino-hexopyranosyl)- | C14H25N3O9 | 1.595% a | - | GC-MS | [13] |
| Muco-Inositol | C6H12O6 | 2.0264% a | - | GC-MS | [13] |
| Inositol | C6H12O6 | 2.0545% a | - | GC-MS | [13] |
| Cyclohexane, 1-methyl-4-(2-hydroxyethyl)- | C9H18O1 | 0.4822% a | - | GC-MS | [13] |
| Hexadecanoic acid, methyl ester | C17H34O | 0.8519% a | - | GC-MS | [13] |
| Phenol, 2-methyl- | C7H8O | 0.3737% a | - | GC-MS | [13] |
| (1S)-Propanol, (2S)-[(tert.butyloxycarbonyl)amino]-1-phenyl- | C14H21NO3 | 0.967% a | - | GC-MS | [13] |
| 9-Octadecenoic acid (Z)-, methyl ester | C19H36O2 | 1.0307% a | - | GC-MS | [13] |
| 9,12,15-Octadecatrienoic acid, (Z,Z,Z)- | C18H30O2 | 5.0063% a | - | GC-MS | [13] |
| 4-Allyl-3-(dimethylhydrazono)-2-methylhexane-2,5-diol | C12H24N2O2 | 0.4257% a | - | GC-MS | [13] |
| Benzyl- β -d-glucoside | C13H16O7 | 0.6056% a | - | GC-MS | [13] |
| 4,6-dimethyl-2-propyl-1,3,5-dithiazinane | C8H17NS2 | 0.1698% a | - | GC-MS | [13] |
| 1,3-Benzenediol, 2-methyl- | C7H8O2 | 0.545% a | - | GC-MS | [13] |
| 9-Octadecenamide, (Z)- | C18H35NO | 1.4694% a | - | GC-MS | [13] |
| Material Tested | Plant Organ | Disease | Model Used | Observed Effect | Active Principle | Quantity | References |
|---|---|---|---|---|---|---|---|
| Aqueous, ethanolic and methanolic extracts | Leaves | Anti-microbial | Enteropathogenic and orthopedics wounds bacteria and fungi | Inhibitory properties for wounds and certain fungal infections | n.i. | MIC 20 mg/mL enteropathogens and 3.75 to 30 µg/mL orthopedics wounds | [26] |
| Ethanol (80%), methanol (70%), hydroalcoholic extracts | Leaves | Metabolic disease and diabetes | Beta-carotene-linoleic acid system, Liposome peroxidation, and liver microsomes | Antioxidant | quercetin and kaempferol | 1 g 80% methanol, and 70% ethanol hydroalcoholic extracts have DPPH equal to 20, 37, 35 mg of pure quercetin | [27] |
| Ethanol solution (90%) | Leaves | Antioxidant | HPLC, MS-MS, chemometrics, DPPH, ABTS, FRAP | Correlation between phytochemical profile and antioxidant potential | kaempferol 3-O-rutinoside, quercetin 3-O-(6″-malonyl-glucoside), kaempferol 3-O-glucoside, and quercetin derivative | n.r. | [28] |
| Ethanolic and methanolic extract | Leaves | Antioxidant | DPPH | Higher antioxidant potential observed for ethanolic and methanolic extract | n.i. | DPPH from 46.32% ± 1.07% to 58.09% ± 0.92% for methanol extract and 56.76% ± 1.48% to 69.72% ± 1.15% for ethanol extract | [29] |
| Ethyl acetate extract | Leaves | Cardiovascular | RAW Macrophages | Decreased expression of inflammatory markers | phenolics | LC50, 212.73 μg/mL | [30] |
| LC10, 57.53 μg/mL | |||||||
| Hot water and ethanol extracts | Leaves | Cancer | Cancer breast cells | Inhibition of NF-kB signaling | n.i. | IC50 0.31 and 0.26 μg/mL, hot water and ethanolic extract, respectively | [31] |
| Hydroalcoholic extract(80% ethanol) | Leaves | NAFLD | Mice | nt Protected HFD-induced liver dam | n.i. | 150 mg/kg body weight, administered by gavage | [32] |
| Leaf powder | Leaves | Anti-inflammatory | Guinea pigs | Lower cholesterol and triglycerides and reduced inflammation in the liver | n.i. | 10% or 15% Moringa enriched diet, 6 weeks | [33] |
| Leaves | Cardiovascular | High-fat fed rats | Decrease cholesterol absorption | β-sitosterol | Bread fortified with extract at 10% & 15% for 45 days | [34] | |
| Leaves | NAFLD | Guinea pigs | Decrease expression of CD68, SERBP1c | n.i. | 10% or 15% Moringa diets with 0.25% dietary cholesterol to induce hepatic steatosis, 6 weeks | [35] | |
| Leaves | Protein deficiency | Fish | Protein supply | n.i. | 10%, 20%, and 30% of Moringa leaf meal of total dietary protein. The 10% gave the best response | [36] | |
| Methanolic extract | Leaves | Obesity | High-fat induced obesity rats | Anti-obesity properties | n.i. | 200 mg and 400 mg/kg reduced atherogenic index (1.7 ± 0.6 and 0.87 ± 0.76, respectively | [37] |
| N, α-L-Rhamnosyl vincosamide | Leaves | Cardiovascular | Isoproterenol (ISO)-induced cardiac toxicity in rats | Increasing the levels of troponin-T, creatine kinase-MB, lactate dehydrogenase and glutamate pyruvate transaminase | N, α-L-Rhamnosyl vincosamide | 40 mg/kg for 7 days, oral administration n | [38] |
| Tri-phenyl tetrazoliumchloride (TCC) | |||||||
| Total Alkaloids | Leaves | Antihypertensive | Frog hearth | Negative inotropic effect on the heart | Total Alkaloids | 3 to 48 ng | [39] |
| Guinea pigs | Calcium channel Blocking effect | ||||||
| Water extract (polyphenolics) Hydroalcoholic extract | Leaves | Antioxidant | DPPH | Gold metal ions reduction (Au+ to Au°) to form nanoparticles | n.i. | 5 mL extract + 100 mL 1 mM aqueous gold chloride soln | [40] |
| Cytotoxic and anticancer | Male Wistar rats | Decreasing tumor markers (α-fetoprotein and carcinoembryonic antigen) and increasing apoptosis | 150 mg/kg–250 mg/kg Male Wistar rats, 7–14 days | ||||
| Water extract | Leaves | Antioxidant | DPPH | Improvement extraction for antioxidant tests | tannins | T 80.54°C for 12.19 min. Protein 17.4%, anti-oxidant activity 10.3 μg/mL, tannin 7.8% | [41] |
| Leaves | Antioxidant | DPPH | Tannins were determined as the active principle for the activity | tannins | n.r. | [25] | |
| Leaves | Cardiovascular and Diabetes | Starch and phosphate buffer solution | Inhibition of α-glucosidase | quercetin | IC50 0.78 ± 0.21 mg/mL (inhibitor sucrose esterase) | [42] | |
| Pancreatic lipase solution | Inhibition of cholesterol esterase | At 10 mg/mL inhibited cholesterol micelle formation of 40.22 ± 2.64% | |||||
| Aqueous extracts (AE) | Seeds | Anti-allergic, and Immunosup-pressive prop-erties | Human peripheral blood mononuclear cells | Cytotoxic AE and cMOL, not cytotoxic wsMOL | Coagulant Moringa Lectins (cMOL) and Water-soluble Moringa Lectins (wsMOL) | 6.25 µg/mL | [43] |
| NCI-H292, HT-29 and HEp-2 cancer cell lines | Weekly/moderately cytotoxic AE, cMOL, wsMOL | ||||||
| Murine erythrocytes | Not hemolytic AE, cMOL, wsMOL | ||||||
| Lipopolyssaccharide-stimulated murine macrophages | AE, cMOL, and wsMOL regulated the production of nitric oxide, TNF-α and IL-1β | ||||||
| Mouse model of car-rageenan-induced pleurisy | AE reduced leukocyte migration | 2.000 mg/kg | |||||
| Ethanolic extracts | Seeds | Anti-inflammatory, and anti-diabetic | Rats |
Bio-accessibility of 1, using the TNO Intestinal Model (TIM-1), determined as 61% and 62% in fasted and fed states, respectively | Isothiocyanate 1 | 50 mg/kg intravenously with pure 1 or orally gavage with seed extract | [36] |
| Methanolic extract | Seeds | Antiviral | Influenza A virus (H1N1) | Inhibitory effects against the H1N1 virus | Moringa A, Glucomoringin, and Vitexin | IC50 0.26 ± 0.03, 0.98 ± 0.17, and 3.42 ± 0.37 μg/mL for Moringa A, Glucomoringin and Vi-texin, respectively | [44] |
| Anti-inflammatory | Raw264.7 cells | Decreasing the levels of TNF-α, IL-6, and IL-1β | |||||
| Phenolic extract | Seeds | Anti-microbial | Bacillus cereus, Staphylococcus aureus, Escherichia coli, Yersinia enterocolitica | The observed activity was due to the phenolic compounds obtained from defatted seed extract | n.i. | MIC 0.06–0.157% | [45] |
| Antioxidant | DPPH | IC50 0.9 ± 0.05 | |||||
| Stem powder | Stems | Anti-inflammatory | Keratinocytes and macrophages induced by PGN, TNF-α, or LPS | Suppression of inflammatory factors. Reduction of NLRP3 expression, and inhibition of caspase-1 activation | β-sitosterol | dose range: 7.5 to 30 μM | [46] |
| Ethanolic extracts | Leaves, seeds, pods | Anti-allergic | Rat basophilic leukemia (RBL- 2H3) cells | Extracts and compounds inhibited mast cell degranulation by inhibiting beta-hexosaminidase and histamine release from RBL-2H3 cells, and IL-4 and TNF-α release | β-Sitosterol-3-O-glucoside, Glucomoringin and Quercitin | Extracts of leaves, seeds, and pods: IC50 of 1.20 ± 0.29, 2.52 ± 0.33 and 2.52 ± 0.15 μg/mL, respectively | [47] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anzano, A.; Ammar, M.; Papaianni, M.; Grauso, L.; Sabbah, M.; Capparelli, R.; Lanzotti, V. Moringa oleifera Lam.: A Phytochemical and Pharmacological Overview. Horticulturae 2021, 7, 409. https://doi.org/10.3390/horticulturae7100409
Anzano A, Ammar M, Papaianni M, Grauso L, Sabbah M, Capparelli R, Lanzotti V. Moringa oleifera Lam.: A Phytochemical and Pharmacological Overview. Horticulturae. 2021; 7(10):409. https://doi.org/10.3390/horticulturae7100409
Chicago/Turabian StyleAnzano, Attilio, Mohammad Ammar, Marina Papaianni, Laura Grauso, Mohammed Sabbah, Rosanna Capparelli, and Virginia Lanzotti. 2021. "Moringa oleifera Lam.: A Phytochemical and Pharmacological Overview" Horticulturae 7, no. 10: 409. https://doi.org/10.3390/horticulturae7100409
APA StyleAnzano, A., Ammar, M., Papaianni, M., Grauso, L., Sabbah, M., Capparelli, R., & Lanzotti, V. (2021). Moringa oleifera Lam.: A Phytochemical and Pharmacological Overview. Horticulturae, 7(10), 409. https://doi.org/10.3390/horticulturae7100409







